Abstract
Efflux pump mechanisms perform important physiological functions such as prevention of toxin absorption from the gastrointestinal tract, elimination of bile from the hepatocytes, effective functioning of the blood–brain barrier and placental barrier, and renal excretion of drugs. They exist in all living cells, but those in the bacterial and mammalian cells are more important to the clinician and pharmacologist, as they constitute an important cause of antimicrobial drug resistance, which contributes to treatment failure, high medical bills, and increased mortality / morbidity. This review was aimed at highlighting the role of efflux pump mechanisms in microbial resistance to chemotherapeutic agents. It was also aimed to elucidate their structure and mechanisms of action so as to integrate the efflux pump mechanisms in the design and development of novel antimicrobial agents. Findings from previous studies and research on this subject assessed through Google search, Pubmed, Hinari websites, as well as standard textbooks on chemotherapy, provided the needed information in the process of this review. Efflux pump inhibitors are promising strategies for preventing and reverting efflux-mediated resistance to chemotherapeutic agents. They are usually employed as adjuncts in antimicrobial and cancer chemotherapy. Toxicity, more common with the older-generation inhibitors such as verapamil and reserpine, constitutes the greatest impediment to their clinical applications. No efflux pump inhibitor has been approved for routine clinical use, as a result of doubtful clinical efficacy and unacceptably high incidence of adverse effects, particularly inhibition of the P-450 drug metabolizing enzyme. At present, their applications are mainly restricted to epidemiological studies. Nonetheless, the search for efficacious and tolerable efflux pump inhibitors continues because of the potential benefits. There is a need to consider efflux pump substrate selectivity in the design and development of novel chemotherapeutic agents.
Keywords: Efflux pump, Chemotherapy, Drug resistance
Introduction
The problem of resistance to chemotherapeutic agents is perhaps one of the greatest challenges in clinical medicine. Drug resistance in malaria, Human Immunodeficiency Virus (HIV)/Acquired Immune Deficiency Syndrome (AIDS), bacterial infections, and cancer has continued to challenge the ingenuity of pharmacologists and physicians alike. Causes of drug resistance include irrational drug prescription and usage, abuse of antimicrobials, and substandard pharmaceuticals.[1–3] Implications of drug resistance include increased mortality and morbidity, increased cost of medical treatment, diagnostic uncertainties, and loss of faith in orthodox medicines. There is a need, therefore, for a continued search for more efficacious and more tolerable antimicrobial agents. This review helps in elucidating the mechanisms of action and chemical structures of efflux pumps, and thus helps in the design of novel antimicrobial agents.
The Methods of Literature Search
The information needed in this review was sourced from Google search, Pubmed, and Hinari websites, as well as from standard textbooks on chemotherapy. Key words used for the search include, efflux pump, resistance, chemotherapy and antimicrobials.
Mechanisms of Resistance to Chemotherapy
Antimicrobial challenge to microorganisms is a stress to which the organisms respond by developing resistance. Resistance in an organism can be acquired vertically from an organism to its offspring or horizontally between organisms by conjugation, transduction, and transformation. Genes for resistance are usually carried on plasmids. There are four main mechanisms by which an organism or a cell exhibits resistance to chemotherapeutic agents:
Drug inactivation
Inactivation of penicillin G by β-lactamases in penicillin-resistant bacteria.
Inactivation of chloramphenicol by the production of chloramphenicol acetyltransferase in resistant organisms.
Inactivation of aminoglycosides through phosphorylation, adenylation, or acetylation via aminoglycoside modifying enzymes.
Modification of drug binding sites
Alteration of the 30s subunits of the ribosome in aminoglycoside resistance.
Alteration of the DNA gyrase protein in fluoroquinolone resistance.
Alteration of the penicillin binding protein (PBP) in penicillin-resistant bacteria.
Alteration of the metabolic pathway
Alteration of dihydropteroate synthase in sulfonamide resistance.
Reduced intracellular drug accumulation
By decreasing drug influx as a result of mutations involving polysaccharide outer membrane (porins) in Gram-negative organisms.[4]
By increasing efflux of drugs from the intracellular compartment via energy-dependent efflux pumps. This mechanism is very common in resistance to tetracyclines, erythromycin, and fluoroquinolones. It is also the main mechanism of resistance in cancer chemotherapy.[5] This efflux pump-mediated resistance to chemotherapy is the focus of this review.
Efflux Pump Mechanisms
Efflux pumps, expressed in all living cells, protect the cells from the toxic effects of organic chemicals. An individual pump recognizes a large number of compounds as substrates because recognition is based on physical properties rather than on defined chemical structures, as in enzyme–substrate recognition. The efflux pump systems may be broadly divided into two:
Prokaryotic efflux pumps that mediate resistance in bacteria and viruses.
Eukaryotic efflux pumps that mediate drug resistance in fungi, protozoa, and cancer cells. The division is incomplete as some pumps mediate resistance in both prokaryotic and eukaryotic cells.
Bacterial efflux pumps
The prokaryotic (bacterial) efflux pumps are divided into six classes:[6]
Major facilitator superfamily (MFS)
ATP-binding cassette (ABC) superfamily
Small multidrug resistance (SMR) family
Resistance-nodulation cell division (RND) superfamily
Multi-antimicrobial extension (MATE)
Drug metabolite transporter (DMT) superfamily
Antibiotics can act as inducers and regulators of expression of efflux pumps.[7] Several efflux pumps can be expressed on a given bacterial species, thus conferring on it resistance to many antimicrobials. The ABC efflux pumps are adenosine-5′-triphosphate (ATP)-dependent (primary transporters) and others are drug-proton antiporters and are the major efflux pumps involved in multidrug resistance.[8]
Efflux pumps in eukaryotes
These are divided into five groups:
Monocarboxylate transporter (MCT)
Multidrug resistance protein (MDR, P-glycoprotein)
Multidrug resistance-associated proteins (MRPs)
Peptide transporters (PEPTs)
Na+ phosphate transporters (NPTs)
Structure of Efflux Pumps
In Gram-negative bacteria, characterized by a protective double membrane system, a typical efflux pump consists of the following four components:
Outer membrane proteins
Middle periplasmic protein
Inner membrane protein
Transmembrane duct
The periplasmic membrane protein interacts with the outer and inner membranes to stabilize the duct (channel) in a closed state. Opening of the duct is triggered by binding of the drug to the inner membrane protein and the energy-dependent, protein–protein interaction between the outer membrane protein and periplasmic membrane protein. The inner membrane transporter provides energy by exchanging the substrate (drug) with H+. Figure 1 shows schematic drawing of the structure of an efflux pump.[9]
Figure 1.
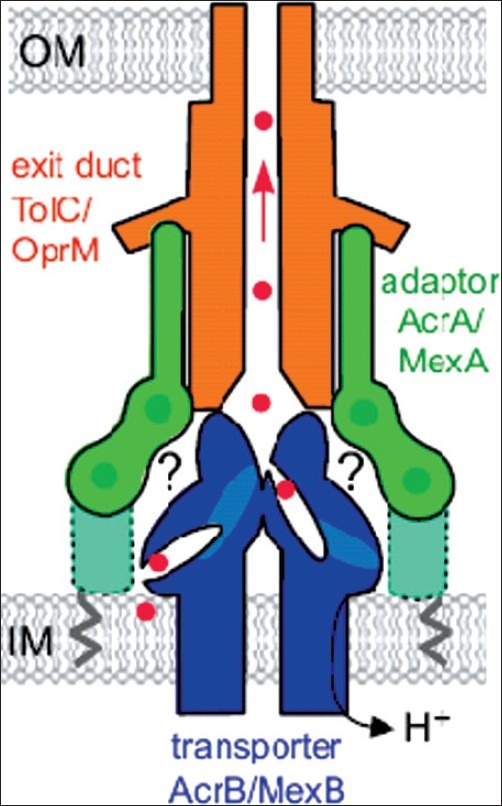
Schematic of the tripartite multidrug efflux pump. The pumps comprise an exit duct (shown in orange; TolC in E. coli, OprM in P. aeruginosa) anchored in the outer membrane (OM), an integral inner membrane (IM) transporter (in blue; AcrB and MexB) and a periplasmic adaptor protein (in green; AcrA and MexA) linked to the inner membrane by a fatty acid (zigzagline). The adaptor binds the exit duct α-helical domain via it's α-hairpin (23) and thre transporter via it's α-hairpin (23) and the transporter via unknown interactions (indicated by ?). The adaptor linear multidomain structure is characterized by interdomain flexibility, but it is incompletye, missing the MP domain indicated by the dotted outline. Red dots indicate antibacterial drugs bound to putative pockets in the transporter, passing through TolC (arrowed), and out of the cell[9]
Physiological Functions of Efflux Pumps
Under normal physiological conditions, efflux pumps are expressed in organs involved in elimination of endogenous waste and xenobiotics, such as the kidney, liver, and epithelial tissues that protect important organs like the small intestine, placenta, blood–brain barrier, and the testes.[10] In the small intestine, the efflux pump-mediated mechanism limits the absorption of several drugs.[11]
The brain is well protected against toxins and drugs. In addition to the anatomical modification of cerebral endothelial cells in the blood–brain barrier, the efflux pumps ensure that harmful substances that enter the brain cells are pumped out.[10] Wang et al. demonstrated that knock-out mice lacking P-glycoprotein exhibited higher brain concentrations of peripherally administered vincristine compared to normal mice.[12]
The active efflux of drugs by placental efflux pumps help to maintain their barrier function. These pumps, mainly the ABC type, are located on the maternal surface of the syncytial membrane of placental microvilli.[13]
The blood–testis barrier (BTB), formed by the adjacent membranes of Sertoli cells, protects the spermatozoa from the effect of harmful substances and drugs as well as forms immunological sanctuaries so that antibodies are not formed against sperm cells during spermatogenesis. Efflux pumps located in the Sertoli cell membranes help in this protection by pumping out drugs and other harmful substances.[14]
P-glycoprotein and MRP2 secrete metabolites such as conjugated bilirubin out of the hepatocytes into the bile canaliculi;[15,16] genetic absence of MRP2 causes decreased excretion of bilirubin in the Dubin–Johnson syndrome.[17] P-glycoprotein mediates the tubular secretion of cholesterol and uric acid, thereby protecting the proximal tubular epithelial cells from cellular injury.[18,19] P-glycoprotein located on the luminal membrane of renal epithelial cells actively secretes digoxin, ceimetidine, and many other drugs, and the efflux pump inhibitors such as verapamil, reserpine, vinblastin, and daunorubicin inhibit the tubular secretion of digoxin.[20]
Overcoming Efflux-mediated Resistance
The strategies to overcome efflux-mediated drug resistance include the following:
Bypassing the efflux pump
Structural analogs of an antimicrobial agent show differences as substrates for efflux pumps. The newer generation agents are less susceptible to efflux pumps than the older generation agents of the same class. For example, the glycyclines are less susceptible than tetracyclines, and ketolides are less susceptible than macrolides.[21,22]
Biological inhibition of the efflux pump
Being proteins, the efflux pumps could be neutralized by antibodies. Alternatively, translation of the genes coding for these efflux pumps could be prevented by using antisense oligonucleotides. Oethinger et al. demonstrated that deletion of the AcrB gene in E. coli restored its sensitivity to fluoroquinolones.[23]
Pharmacological inhibition of efflux pumps
Drugs that are competitive and non-competitive inhibitors of efflux pumps are used to reverse or prevent the development of efflux-mediated drug resistance. These efflux pump inhibitors are usually used as adjuncts in therapy.
Efflux Pump Inhibitors
Efflux pump inhibitors prevent the energy-dependent efflux of drugs and some endogenous metabolites from the cells. They are a promising strategy for restoring the activity of existing antimicrobial agents. A majority of the efflux pump inhibitors are not used as pump inhibitors in routine clinical practice because concentrations that achieve efflux inhibition in vitro are rarely achieved in vivo without serious toxicities.[24] At present, many are used for epidemiological surveys of drug-resistant organisms. In this direction, ethidium bromide is an efficient substrate for many MDR pumps and is used to assess the effect of inhibition on such pumps.
An ideal efflux pump inhibitor should:
restore the activity of an antimicrobial in both intrinsic and acquired resistance;
have a wide range of activity against Gram-positive and Gram-negative bacterial pumps; and
not affect the physiological efflux pumps so as to minimize adverse effects.
Mechanism of action of efflux pump inhibitors
Minimum inhibitory concentrations (MIC) are very important in assessing the inhibitory actions of efflux pumps. Couto et al. (2008) demonstrated that reduction of a drug's MIC by at least a quarter of its original value by addition of an efflux pump inhibitor was indicative of efflux activity.[25]
Mechanisms of action of efflux pump inhibitors
Non-competitive blocking of the drug-binding site on the efflux pump
Dissipating the energy source of the efflux pump[26]
Classification of efflux pump inhibitors
Some efflux pump inhibitors are naturally occurring lipophilic alkaloids, terpenoids, and flavonoids, while others are rationally designed by manipulation of molecular structures of pump substrates.[27] Classification of efflux pump inhibitors along the line of efflux pumps is difficult because just like the pumps, some inhibitors are pump specific, while others are not. The closest attempt at classification is based on the effect on bacterial and mammalian efflux pumps:
Microbial efflux pump inhibitors
Mammalian efflux pump inhibitors
Some efflux pump inhibitors such as verapamil and reserpine inhibit both microbial and mammalian efflux pumps.
Microbial efflux pump inhibitors
As Gram-negative bacteria usually have a thick outer wall protection, which does not allow easy access of substances, the effects of efflux pumps and consequently efflux pump inhibitors are more pronounced in them as compared to the Gram-positive organisms. Microbial efflux pump inhibitors include:
Analogs of antimicrobial agents such as tetracyclines, aminoglycosides, and fluoroquinolones. Modifications in these analogs enable them to escape efflux pumps.[22]
Peptidomimetics. The prototype here is phenylalanine arginyl-β-naphthylamide (PAβN).
Amide derivatives
Quinolone derivatives, for example, quinazolinones
Phenothiazines, for example, chlorpromazine
Selective serotonin reuptake inhibitors (SSRI), for example, paroxetine
Protein pump inhibitors, for example, omepraole, pantoprazole
Mammalian efflux pump inhibitors
A broad division of mammalian efflux pump inhibitors includes:
P-glycoprotein inhibitors
First-generation P-gp inhibitors, for example, verapamil, nifedipine, lovastatin, simvastatin, cyclosporine A, tamoxifen, ketoconazole, erythromycin, progesterone
Second-generation P-gp inhibitors, for example, valspodar, biricodar, timicodar
Third-generation P-gp inhibitors, for example, elacridar, zosupidar, tariquidar
MRP inhibitors, for example, argosterol A, ralozitene
Breast cancer receptor protein (BCRP) inhibitors, for example, elacridar, reserpine
Previous Works on Efflux Pump Inhibitors
Efflux pump inhibitors for microbial infections
Most reports on bacterial efflux pump inhibitors are based on in vitro laboratory experiments. One major impediment for the use of these inhibitors in treating clinical infections is the high plasma concentrations of these drugs needed to achieve efflux pump inhibition in vivo.
Zhang et al. demonstrated that reserpine reversed the resistance of Mycobacterium tuberculosis to isonicotinylhydrazine (INH) and pyrazinamide.[28] Also, Cui et al. observed decreased MIC of anti-TB agents to resistant M. tuberculosis strains in a liquid culture, after addition of verapamil and reserpine.
Overexpression of the efflux pump genes RV2459 and RV3728 was induced by administration of combined INH and ethambutol Gupta, and Ramon-Garcia et al. demonstrated that deletion of the RV1410C gene (which encodes the P55 efflux pump in the mycobacteria) made the organisms more susceptible to first- and second-line anti-TB drugs.[30,31] Farnesol significantly enhanced accumulation of ethidium bromide in Mycobacterium smegmatis and showed significant synergism when combined with rifampicin.[32]
Omeprazole inhibited the NorA pump of Gram-positive bacteria, while pantoprazole restored the antibiotic susceptibilities of the multidrug-resistant strains of Helicobacter pylori, as they significantly reduced the MIC of these antibiotics.[28,33] Reserpine enhanced the activity of fluoroquinolones on the multidrug-resistant, Gram-positive bacteria, decreased the emergence of resistant strains in Staphylococcus aureus and Streptococcus pneumoniae, and improved the susceptibility to tetracycline in methicillin-resistant Sta. aureus.[22,34]
Chlorpromazine enhanced the antimicrobial activity of aminoglycosides and macrolides, and also had a synergistic effect in combination with penicillin G against E. coli by inhibition of bacterial efflux pumps.[35,36]
Plasmodium falciparum (causative organism of malignant malaria) developed resistance to chloroquine, proguanil, and pyrimethamine, which was partly traced to a decreased accumulation of these drugs by the falciparum organism due to development of the P. falciparum multidrug-resistance gene.[37] In other in vitro studies, verapamil restored the chloroquine concentrating ability and sensitivity in P. falciparum. Bray et al. had earlier demonstrated that verapamil reversed chloroquine resistance in P. falciparum in a dose-dependent manner, suggesting competition between chloroquine and verapamil for the organism's MDR pump.[38]
Lee et al. observed that milbemycins were potent inhibitors of the CDR1 pump in Candida albicans and these drugs potentiated the antifungal activity of fluconazole against a wide variety of C. albicans clinical isolates.[39] Similarly, quinazolinones have been identified as inhibitors of fungal efflux pumps.[40]
Drug resistance is becoming a big challenge in the fight against HIV / AIDS. An efflux-mediated mechanism contributes to this resistance. It has been demonstrated that long-term administration of anti-retroviral drugs contribute to efflux-mediated resistance by inducing expression and function of P-glycoproteins.[41,42] Khaliq et al. demonstrated that ketoconazole (P-glycoprotein inhibitor) increased the cerebrospinal fluid (CSF) levels of ritonavir and saquinavir.[43]
Efflux pump inhibitors for cancers
P-glycoprotein is encoded by the MDR1 gene and its overexpression in cancer cells contributes to resistance in cancer chemotherapy. The P-glycoprotein inhibitors are competitive substrates for P-glycoproteins; therefore, they increase the intracellular concentration of co-administered anticancer drugs. Many of the agents entered clinical trials, but none was successful due to an unacceptably high incidence of adverse effects. The observed toxicities were mainly a result of the competitive inhibition of cytochrome P-450 enzymes, which increased the plasma concentration of the co-administered anticancer agents.[44] The physiological efflux pumps were also inhibited.
This high incidence of adverse effects was very common with the first- and second-generation P-glycoprotein inhibitors, such as verapamil, cyclosporine A, valspodar, and biricodar. Third-generation P-glycoprotein inhibitors were designed to specifically inhibit only P-glycoprotein, and thus reduce the adverse effects. However, clinical trials using the third-generation P-glycoprotein inhibitors produced conflicting results.[45] In a phase I clinical trial, van Zuylen et al. found that a combination of R101933 (laniquidar, a third-generation P-glycoprotein) with docetaxel, in the treatment of solid tumors, did not alter the plasma pharmacokinetics of the latter, suggesting that R101933 did not influence the disposition of docetaxel.[46] Some newer products, for example, R-verapamil had minimal side effects when combined with anticancer agents, but exhibited poor clinical response.[47,48] Toppmeyer et al. demonstrated the efficacy of biricodar (Incel) when combined with paclitaxel in the treatment of advanced breast cancer, as compared to paclitaxel alone.[49] However, in a phase II clinical trial in which biricodar was combined with doxorubicin and vincristine, in patients with recurrent small cell lung cancer, Gandhi and his colleagues did not observe enhanced antitumor activity.[50]
Flavonoids as efflux pump inhibitors
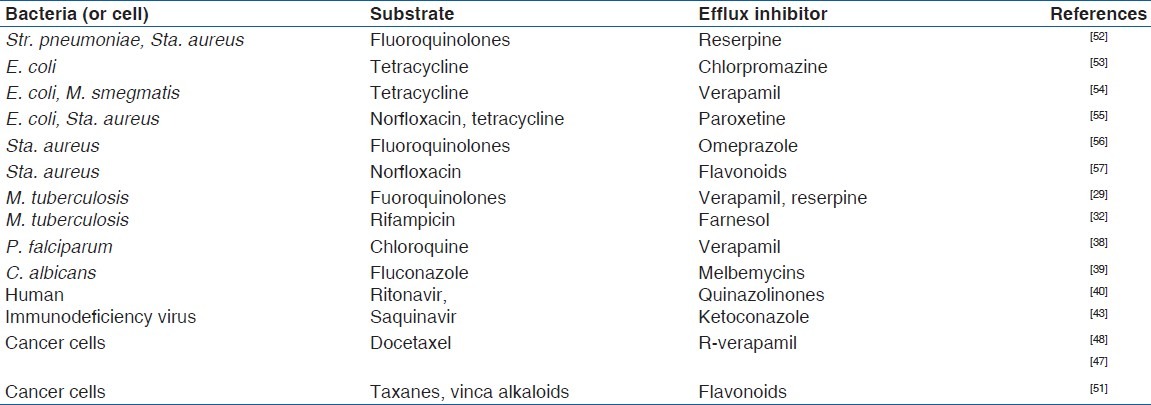
Flavonoids (herbal constituents) also exhibited P-gp inhibitory and direct antitumor activity, thereby acting synergistically with taxanes, vinca alkaloids, and campothecins, in cancer chemotherapy.[51] Table 1 shows some efflux pump substrates and their inhibitors.
Table 1.
Efflux pump substrates and inhibitors in some microorganisms and cancer cells
Adverse Effects of Efflux Pump Inhibitors
Available laboratory data indicate that many drugs are substrates for both efflux pumps and the cytochrome p-450 A34 metabolizing enzyme. P-glycoprotein inhibitors also inhibit cytochrome P-450 enzymes that metabolize anticancer agents, thus leading to increased toxicity when they are co-administered with these agents.
This is particularly so with the first- and second-generation P-glycoprotein inhibitors. For example, Ebert et al. demonstrated that the co-administration of the P-glycoprotein inhibitor erythromycin and cardiac glycosides (digoxin) in hospitalized patients was associated with increased serum concentration of the latter.[58] Wakasugi et al. had earlier shown that clarithromycin increased the plasma concentration of co-administered digoxin by inhibition of P-glycoprotein–mediated renal excretion.[59] Verapamil and reserpine increased the cytotoxicity of taxols, anthracyclines, and vinca alkaloids by inhibition of P-glycoprotein–mediated efflux.[60]
The second-generation P-glycoprotein inhibitor valspodar inhibited the P-450 3A4-mediated metabolism of paclitaxel and vinblastine, resulting in an increased serum concentration of these agents.[61] This often necessitated a reduction in doses of anticancer agents with attendant reduction in clinical response.
These older-generation P-glycoprotein inhibitors also inhibit physiological efflux pumps such as those involved in blood–brain barrier, BTB, and placental functions.[62]
Generally, third-generation P-glycoprotein inhibitors exhibit a decreased incidence of toxicity when co-administered with other drugs.[49,63] Thus, tariquidar, laniquidar, and zosuquidar do not affect cytochrome P-450 3A4 at relevant concentrations; also, they do not affect physiological efflux pumps.[64]
Efflux pump inhibitors also exhibit adverse effects not related to efflux pump or the cytochrome P-450 enzyme function. Such effects include arrythmias (verapamil), immunosuppression (cyclosporin A), vaginal bleeding (tamoxifen), allergic hepatitis (ketoconazole), and cholestatic hepatitis (erythromycin).
Conclusion
Efflux pump-mediated mechanisms contribute to resistance in chemotherapy. As promising as efflux pump inhibitors appear to be, none has been approved for routine clinical use as a result of doubtful clinical efficacy and unacceptably high incidence of adverse effects. At present, their applications are mainly restricted to epidemiological studies. These drawbacks, notwithstanding, the search for efficacious and tolerable pump inhibitors continues because of the potential benefits. With such an agent, most chemotherapeutic agents rendered useless by efflux-mediated resistance will become useful again.
Way forward
Consider efflux pump substrate selectivity in the design and development of novel chemotherapeutic agents.
Structural elucidation of efflux pumps will help to develop more effective / specific inhibitors.
There is a need to screen natural herbs for efflux pump inhibitory activity.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
References
- 1.McNutty CA, Boyle P, Nichols T, Clappison P, Davey P. The public's attitudes to and compliance with antibiotics. J AntimicrobChemother. 2007;60(Suppl 1):i63–8. doi: 10.1093/jac/dkm161. [DOI] [PubMed] [Google Scholar]
- 2.Castanon JI. History of the use of antibiotic as growth promoters in European poultry fees. PoultSci. 2007;86:2466–71. doi: 10.3382/ps.2007-00249. [DOI] [PubMed] [Google Scholar]
- 3.Larsson DG, Fick J. Transparency throughout the production chain-a way to reduce pollution from the manufacturing of pharmaceuticals? RegulToxicolPharmacol. 2009;53:161–3. doi: 10.1016/j.yrtph.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 4.Vila J, Marti S, Sanchez-Cespede J. Porins, efflux pumps and multidrug resistance in Acinetobacterbaumannii. J AntimicrobChemother. 2007;59:1210–5. doi: 10.1093/jac/dkl509. [DOI] [PubMed] [Google Scholar]
- 5.Rang HP, Dale MM, Ritter JM, More PK. Pharmacology. 5th ed. London: Churchill Livingstone; 2004. p. 631. [Google Scholar]
- 6.Poole K. Outer membranes and efflux: the path to multidrug resistance in Gram-negative bacteria. Curr Pharm Biotechnol. 2002;3:77–98. doi: 10.2174/1389201023378454. [DOI] [PubMed] [Google Scholar]
- 7.Morita Y, Sobel ML, Poole K. Antibiotic inducibility of the MexXY multidrug efflux system of Pseudomonas aeruginosa: involvement of the antibiotic-inducible PA5471 gene product. J Bacteriol. 2006;188:1847–55. doi: 10.1128/JB.188.5.1847-1855.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Magnet S, Courvalin P, Lambert T. Resistance-nodulation-cell division-type efflux pump involved in aminoglycoside resistance in Acinetobacterbaumannii strain BM4454. Antimicrob Agents Chemother. 2001;45:3375–80. doi: 10.1128/AAC.45.12.3375-3380.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Symmons MF, Bokma E, Koronakis E, Hughes G, Koronakis V. The assembled structure of a complete tripartite bacterial efflux pump. [Last accessed on 2012 Feb 24];ProcNatlAcadSci USA. 2009 106:7173–8. doi: 10.1073/pnas.0900693106. Available from: http://www.pnas.org/cgi/doi/10.1073/ pnas.0900693106 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hagenbuch B, Gao B, Meier PJ. Transport of zenobiotics across the blood-brain barrier. News PhysiolSci. 2002;17:231–4. doi: 10.1152/nips.01402.2002. [DOI] [PubMed] [Google Scholar]
- 11.Dahan A, Sabit H, Amidon GL. Multiple efflux pumps are involved in the transepithelial transport of colchicine: combined effect of p-glycoprotein and multidrug resistance-associated protein 2 leads to decreased intestinal absorption throughout the entire small intestine. Drug MetabDispos. 2009;37:2028–36. doi: 10.1124/dmd.109.028282. [DOI] [PubMed] [Google Scholar]
- 12.Wang F, Zhou F, Krah GD, Gallo JM. Influence of blood-brain barrier efflux pumps on the distribution of vincristine in brain and brain tumors. Neuro Oncol. 2010;12:1043–9. doi: 10.1093/neuonc/noq056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Evsenko D, Paxton JW, Keelan JA. Active transport across the human placenta. The impact on drug efficacy and toxicity. Expert Opin Drug Metab Toxicol. 2006;2:51–69. doi: 10.1517/17425255.2.1.51. [DOI] [PubMed] [Google Scholar]
- 14.Su L, Mruk DD, Cheug CY. Drugs transporters, the blood-testis barrier and spermatogenesis. [Last accessed on 2012 Feb 24];J Endocrinol. 2011 208:207–23. doi: 10.1677/JOE-10-0363. Available from: http://www.ncbi.nlm.nih.gob/pubmed/21134990? dopt . Abstract . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mottino AD, Hottman T, Jennes L, Vore M. Expression and localization of multidrug resistance protein mrp2 in rat small intestine. J PharmacolExpTher. 2000;293:717–23. [PubMed] [Google Scholar]
- 16.Kruh GD, Belinsky M. The MRP family of drug efflux pumps. Oncogene. 2003;22:7537–52. doi: 10.1038/sj.onc.1206953. [DOI] [PubMed] [Google Scholar]
- 17.Wilkinson GR. The dynamics of drug absorption, distribution, and elimination. In: Hardman JG, Limbird E, editors. The pharmacological basis of therapeutics. 10th ed. New York: McGraw-Hill; 2000. p. 12. [Google Scholar]
- 18.Zager RA. P glycoprotein-mediated cholesterol cycling determines proximal tubular cell viability. Kidney Int. 2001;60:944–56. doi: 10.1046/j.1523-1755.2001.060003944.x. [DOI] [PubMed] [Google Scholar]
- 19.Karyekar C, Eddington N, Briglia A, Gubbins PO, Thomas C. Renal interaction between itraconazole and cimetidine. J ClinPharmacol. 2004;44:919–27. doi: 10.1177/0091270004266783. [DOI] [PubMed] [Google Scholar]
- 20.Hori R, Okamura N, Aiba T, Tanigawara Y. Role of P-glycoprotein in renal tubular secretion of digoxin in the isolated perfused rat kidney. J Pharmacol Exp Ther. 1993;266:1620–5. [PubMed] [Google Scholar]
- 21.Hooper DC. Mechanisms of action and resistance of older and newer fluoroquinolones. Clin Infect Dis. 2000;31(Suppl 2):S24–8. doi: 10.1086/314056. [DOI] [PubMed] [Google Scholar]
- 22.Li HZ, Nikaido H. Efflux-mediated drug resistance in bacteria. Drugs. 2004;64:159–204. doi: 10.2165/00003495-200464020-00004. [DOI] [PubMed] [Google Scholar]
- 23.Oethinger M, Kern WV, Jellen-Ritter AS, McMurry LM, Levy SB. Ineffectiveness of topoisomerase mutations in mediating clinically significant fluoroquinolone resistance in Escherichia coli in the absence of the AcrAB efflux pump. Antimicrob Agents Chemother. 2000;44:10–3. doi: 10.1128/aac.44.1.10-13.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Van Bambeke F, Pages JM, Lee VJ. Inhibitors of bacterial efflux pumps as adjuvants in antibiotic treatments and diagnostic tools for detections of resistance by efflux. Recent Pat Antiinfect Drug Discov. 2006;1:157–75. doi: 10.2174/157489106777452692. [DOI] [PubMed] [Google Scholar]
- 25.Couto I, Costa SS, Viveiros M, Martins M, Amaral L. Efflux-mediated response of Staphylococcus aureus exposed to ethidium bromide. J Antimicrob Ther. 2008;62:504–13. doi: 10.1093/jac/dkn217. [DOI] [PubMed] [Google Scholar]
- 26.Zechini B, Versace I. Inhibitors of multidrug resistant efflux systems in bacteria. Recent Pat Antiinfect Drug Discov. 2009;4:37–50. doi: 10.2174/157489109787236256. [DOI] [PubMed] [Google Scholar]
- 27.Nelson ML, Levy SB. Reversal of tetracycline resistance mediated by different bacterial tetracycline resistance determinants by an inhibitor of the Tet(B) antiport protein. Antimicrob Agents Chemother. 1999;43:1719–24. doi: 10.1128/aac.43.7.1719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang Y, Permar S, Sun Z. Conditions that may affect the results of susceptibility testing of Mycobacterium tuberculosis to pyrazinamide. J Med Microbiol. 2002;51:42–9. doi: 10.1099/0022-1317-51-1-42. [DOI] [PubMed] [Google Scholar]
- 29.Cui ZL, Wang XL, Wang J, Lu JM, Hu ZY. The effects of gene mutation related to drug resistance and drug efflux pump in extensively drug resistant tuberculosis clinical isolates. ZhonghuaJie He He Hu Xi ZaZhi. 2010;33:505–9. [PubMed] [Google Scholar]
- 30.Gupta AK, Katoch VM, Chauhan DS, Sharma R, Singh M, Venkatesan K, et al. Microarray analysis of efflux pump genes in multidrug-resistant mycobacterium tuberculosis during stress induced by common anti-tuberculosis drugs. Microb Drug Resist. 2010;16:21–8. doi: 10.1089/mdr.2009.0054. [DOI] [PubMed] [Google Scholar]
- 31.Ramon-Garcia S, Martin C, Thompson CJ, Ainsa JA. Role of the mycobacternium tuberculosis p55 efflux pump in intrinsic drug resistance, oxidative stress responses and growth. Antimicrob Agents Chemother. 2009;53:3675–82. doi: 10.1128/AAC.00550-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jin J, Zhang JY, Guo N, Shang H, Li L, Liang JC, et al. Farnesol, a potential efflux pump inhibitor in Mycobacterium smegmatis. Molecules. 2010;15:7750–62. doi: 10.3390/molecules15117750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Vidaillac C, Guillon J, Arpin C, Forfar-Bares I, Ba BB, Grellet J, et al. Synthesis of omeprazole analogues and evaluation of these as potential inhibitors of multidrug efflux pump NorA of Staphylococcus aureus. Antimicrob Agents Chemother. 2007;51:831–8. doi: 10.1128/AAC.01306-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Poelands GJ, Mazurkiewicz P, Konings WN. Multidrug transporters and antibiotic resistance in Lactococcuslactis. BiochimBiophysActa. 2002;1555:1–7. doi: 10.1016/s0005-2728(02)00246-3. [DOI] [PubMed] [Google Scholar]
- 35.Chan YY, Ong YM, Chua KL. Synergistic interaction between phenothiazines and antimicrobial agents against Burkholderiapseudomallei. Antimicrob Agents Chemother. 2007;51:623–30. doi: 10.1128/AAC.01033-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lehtinen J, Lilius EM. Promethazine renders Escherichia coli susceptible to penicillin G: real-time measurement of bacterial susceptibility by fluoro-luminotry. Int J Antimicrob Agents. 2007;30:44–51. doi: 10.1016/j.ijantimicag.2007.02.019. [DOI] [PubMed] [Google Scholar]
- 37.Martin RE, Marchetti RV, Cowan AL, Howitt SM, Bröer S, Kirk K. Chloroquine transport via the malaria parasite's chloroquine resistance transporter. Science. 2009;325(94):1680–2. doi: 10.1126/science.1175667. [DOI] [PubMed] [Google Scholar]
- 38.Bray PG, Howells RE, Ritchie GY, Ward SA. Rapid chloroquine efflux phenotype in both chloroquine-sensitive and chroloquine-resistant Plasmodium falciparum: A correlation of chloroquine sensitivity with energy - dependent drug accumulation. BiochemPharmacol. 1992;44:1317–24. doi: 10.1016/0006-2952(92)90532-n. [DOI] [PubMed] [Google Scholar]
- 39.Lee MD, Galazzo GL, Stanley AH, Lee JC, Warren MS, FuernKranz H, et al. Microbial fermentation-derived inhibitors of efflux-pump-mediated drug resistance. Farmaco. 2001;56:81–5. doi: 10.1016/s0014-827x(01)01002-3. [DOI] [PubMed] [Google Scholar]
- 40.Watkins WJ, Lemoine RC, Chong L, Cho A, Renau TE, Kuo B, et al. Quinazolinone fungal efflux pump inhibitors part 2: In vitro structure activity relationships of (N - methyl/ - piperazynil) - containing derivatives. Bioorg Med ChemLett. 2004;14:5133–7. doi: 10.1016/j.bmcl.2004.07.071. [DOI] [PubMed] [Google Scholar]
- 41.Perloff MD, von Moltke LL, Fahey JM, Daily JP, Greenblat DJ. Induction of p-glycoprotein expression by HIV protease inhibitors in cell culture. AIDS. 2000;14:1287–9. doi: 10.1097/00002030-200006160-00034. [DOI] [PubMed] [Google Scholar]
- 42.Zastre JA, Chan GN, Ronaldson PT, Ramaswamy M, Courand PO, Romero IA, et al. Up-regulation of p-glycoprotein by HIV protease inhibitors in a human brain microvessel endothelial cell line. J Neurosci Res. 2009;87:1023–36. doi: 10.1002/jnr.21898. [DOI] [PubMed] [Google Scholar]
- 43.Khaliq Y, Gallicano K, Venance S, Seguin I, Fake K, Kravcik S, et al. Effect of ketoconazole, the p-glycoprotein inhibitor on ritonavir and saquinavir plasma and cerebrospinal fluid concentrations. IntersciConfAntimicrob Agents Chemother. 1999;39:24–30. [Google Scholar]
- 44.Relling MV. Are the major effects of p-glycoprotein modulators due to altered pharmacokinetics of anticancer drugs? Ther Drug Monit. 1996;18:350–6. doi: 10.1097/00007691-199608000-00006. [DOI] [PubMed] [Google Scholar]
- 45.Kurnik D, Sofowora GG, Donahue JP, Nair UB, Wilkinson GR, Wood AJ, et al. Tariquidar, a selective P-glycoprotein inhibitor, does not potentiate loperamide's opioid brain effects in humans despite full inhibition of hymphocyte P-glycoprotein. Anaesthesiology. 2008;109:1092–9. doi: 10.1097/ALN.0b013e31818d8f28. [DOI] [PubMed] [Google Scholar]
- 46.van Zuylen L, Sparreboom A, van der Gaast A, van der Burg ME, van Beurden V, Bol CJ, et al. The orally administered P-glycoprotein inhibitor R101933 does not alter the plasma pharmacokinetics of docetaxel. Clin Cancer Res. 2000;6:1365. [PubMed] [Google Scholar]
- 47.Nobili S, Landini I, Gighoni B, Mini E. Pharmacological strategies to overcoming multidrug resistance. Curr Drug Targets. 2006;7:861–79. doi: 10.2174/138945006777709593. [DOI] [PubMed] [Google Scholar]
- 48.Perez Tomas R. Multidrug resistance: Retrospect and prospects in anti-cancer drug treatment. Curr Med Chem. 2006;13:1859–76. doi: 10.2174/092986706777585077. [DOI] [PubMed] [Google Scholar]
- 49.Toppmeyer D, Seidman AD, Pollak M, Russel C, Tkaczick K, Verma S, et al. Safety and efficacy of the multidrug resistance inhibitor lncel (biricodar, VX-710) in combination with paclitaxel for advanced breast cancer refractory to paclitaxel. Clin Cancer Res. 2002;8:670–8. [PubMed] [Google Scholar]
- 50.Gandhi L, Harding MW, Neubauer M, Langer CJ, Moore M, Ross HJ, et al. A phase II study of the safety and efficacy of the multidrug resistance inhibitor VX-710 combined with doxorubicin and vincristine in patients with recurrent small cell lung cancer. Cancer. 2007;109:924–32. doi: 10.1002/cncr.22492. [DOI] [PubMed] [Google Scholar]
- 51.Bansal T, Jaggi M, Khar R, Talegaonkar S. Emerging significance of flavonoids as p-glycoprotein inhibitors in cancer chemotherapy. J Pharm Pharm Sci. 2009;12:46–78. doi: 10.18433/j3rc77. [DOI] [PubMed] [Google Scholar]
- 52.Baucheron S, Imberechts H, Chaslus-Darcla E, Cloeckaert A. The AcrB multidrug transporter play a major role in high-level fluoroquinolone resistance in Salmonella entericaserovartyphimurium phage type DT204. Microb Drug Resist. 2002;8:281–9. doi: 10.1089/10766290260469543. [DOI] [PubMed] [Google Scholar]
- 53.Molnar J, Hever A, Fakla I, Fischer J, Ocsovski I, Aszalós A. Inhibition of the transport function of membrane proteins by some substituted phenothiazines in E. coli and multidrug resistant tumor cells. Anticancer Res. 1997;17:481–6. [PubMed] [Google Scholar]
- 54.Gibbons S, Udo EE. The effect of reserpine, a modulator of multidrug efflux pumps, on the in vitro activity of tetracycline against clinical isolates of methicillin resistant Staphylococcus aureus (MRSA) possessing the tet(K) determinant. Phytother Res. 2000;14:139–40. doi: 10.1002/(sici)1099-1573(200003)14:2<139::aid-ptr608>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 55.Kaatz GW, Moudgal VV, Seo SM, Hansen JB, Kristiansen JE. Phenylpiperidine selective serotonin reuptake inhibitors interfere with multidrug efflux pump activity in Staphylococcus aureus. Int J Antimicrob Agents. 2003;22:254–61. doi: 10.1016/s0924-8579(03)00220-6. [DOI] [PubMed] [Google Scholar]
- 56.Coban AY, Ekinci AB, Durupinar B. A mutidrug efflux inhibitor reduces fluoroquinoline resistance in Pseudomonas aeruginosa isolates. Chemotherapy. 2004;50:22–6. doi: 10.1159/000077280. [DOI] [PubMed] [Google Scholar]
- 57.Musumeci R, Speciale A, Costanzo R, Berberisaetnensis C. Presl. extracts: antimicrobial properties and interaction with ciprofloxacin. Int J Antimicrob Agents. 2003;22:48–53. doi: 10.1016/s0924-8579(03)00085-2. [DOI] [PubMed] [Google Scholar]
- 58.Ebert S, Renner B, Neubert A, Reisig M, Bachmakov I, Konig J, et al. Role of p-glycoprotein inhibitor for drug interactions; Evidence from in vitro and pharmacological studies. Clin Pharmacokinet. 2007;46:1039–49. doi: 10.2165/00003088-200746120-00004. [DOI] [PubMed] [Google Scholar]
- 59.Wakasugi H, Yano I, Ito T, Hashida T, Futami T, Nohara R, et al. Effect of clarithromycin on renal excretion of digoxin: interaction with P. glycoprotein. ClinPharmacolTher. 1998;64:123–8. doi: 10.1016/S0009-9236(98)90030-3. [DOI] [PubMed] [Google Scholar]
- 60.Drori S, Eytan GD, Assaraf YG. Potentiation of anticancer-drug cytotoxicity by multidrug-resistance chemosensitizers involves alterations in membrane fluidity leading to increased membrane permeability. Eur J Biochem. 1995;228:1020–9. doi: 10.1111/j.1432-1033.1995.tb20352.x. [DOI] [PubMed] [Google Scholar]
- 61.Wandel C, Kim RB, Kajiji S. P-glycoprotein and cytochrome p-450 3A inhibition: dissociation of inhibitory potencies. Cancer Res. 1999;59:3944–8. [PubMed] [Google Scholar]
- 62.Lum BL, Gosland MP. MDR expression in normal tissues. Pharmacologic implications for the clinical use of P-glycoprotein inhibitors. Haematol Oncol Clin North Am. 1995;9:319–36. [PubMed] [Google Scholar]
- 63.Bramwell VH, Morris D, Ernst DS, Hings I, Blackstein M, Venner PM, et al. Safety and efficacy of the multidrug- resistance inhibitor biricodar (VX-710) with concurrent doxorubicin in patients with anthracycline-resistant advanced soft tissue sarcoma. Clin Cancer Res. 2002;8:383–93. [PubMed] [Google Scholar]
- 64.Mistry P, Stewart AJ, Dangerfield W, Okiji S, Liddle C, Bootle D, et al. In vitro and in vivo reversal of P-glycoprotein-mediated multidrug resistance by a novel potent modulator XR9576. Cancer Res. 2001;61:749–58. [PubMed] [Google Scholar]


