Abstract
Animals use both pendular and elastic mechanisms to minimize energy expenditure during terrestrial locomotion. Elastic gaits can be either bilaterally symmetric (e.g. run and trot) or asymmetric (e.g. skip, canter and gallop), yet only symmetric pendular gaits (e.g. walk) are observed in nature. Does minimizing metabolic and mechanical power constrain pendular gaits to temporal symmetry? We measured rates of metabolic energy expenditure and calculated mechanical power production while healthy humans walked symmetrically and asymmetrically at a range of step and stride times. We found that walking with a 42 per cent step time asymmetry required 80 per cent (2.5 W kg−1) more metabolic power than preferred symmetric gait. Positive mechanical power production increased by 64 per cent (approx. 0.24 W kg−1), paralleling the increases we observed in metabolic power. We found that when walking asymmetrically, subjects absorbed more power during double support than during symmetric walking and compensated by increasing power production during single support. Overall, we identify inherent metabolic and mechanical costs to gait asymmetry and find that symmetry is optimal in healthy human walking.
Keywords: symmetry, biomechanics, energetics, locomotion, limb work
1. Introduction
Terrestrial animals minimize energy expenditure during locomotion using two different energy conserving mechanisms [1]. Elastic gaits (e.g. run, trot and gallop) rely on elastic energy storage and release through tendons and ligaments [2]. In contrast, pendular gaits (e.g. walk and pace) use the exchange of kinetic and gravitational potential energy to minimize the muscular work required for locomotion [3,4]. Gaits can also be classified as either bilaterally symmetric (walk, trot and pace) or asymmetric (canter and gallop), a classification made on the phase difference between the left and right limbs of a pair [5–7]. Elastic gaits can be either symmetric (e.g. run and trot) or asymmetric (e.g. skip, canter and gallop), yet only symmetric pendular gaits are observed in nature. Does minimizing metabolic and mechanical power constrain pendular gaits to temporal symmetry?
Bilaterally symmetric walking is modelled as an inverted pendulum; during single support, kinetic and gravitational potential energy are exchanged as the centre of mass (CoM) arcs over a rigid leg [1,8]. A coordination of simultaneous push-off and collision work is then necessary to transition to the next leg, much of it during double support [9–12]. It has also been shown that preferred symmetric walking is optimal; individuals choose step and stride lengths, widths and durations that minimize both metabolic and mechanical power [13–19].
Here we asked, is symmetry optimal during healthy human walking? Our approach involved comparing asymmetric strides to symmetric strides by varying right and left step time. As an asymmetric stride is composed of two unequal steps, at least one step is not preferred. To test asymmetry independent of non-preferred steps, asymmetric walking was compared with symmetric conditions with both comparable step and stride times.
We hypothesized that: (i) The metabolic cost of walking with asymmetric steps will be greater than for walking with symmetric steps. (ii) Greater mechanical power production will explain the greater metabolic cost. (iii) The majority of the additional positive and negative mechanical power required will be performed during double support.
2. Material and methods
(a). Overview
Ten healthy individuals volunteered for this study (five males and five females, height 1.74 ± 0.20 m, mass 68 ± 10 kg, age: 26 ± 6 year, mean ± 1 s.d.). All gave written consent prior to participation as per the University of Colorado Boulder IRB.
During a single session, subjects walked both symmetrically and asymmetrically at different step and stride times at one speed (1.25 m s−1) on a motorized dual-belt force treadmill while matching an auditory metronome and receiving visual symmetry feedback. We measured each subject's rates of metabolic energy expenditure via expired gas analysis and used the individual limbs method [20] to calculate the external mechanical power production during each condition.
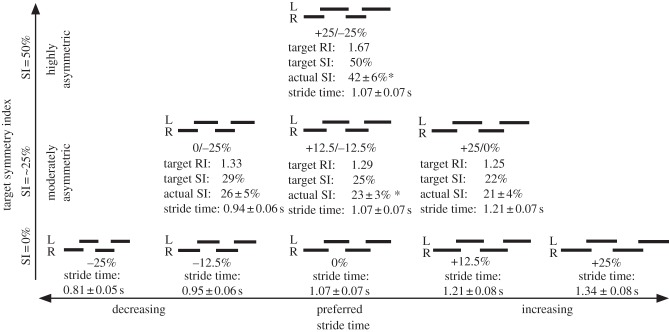
Subjects performed five symmetric trials (target step times −25, −12.5, 0, +12.5 and +25% different from preferred), three moderately asymmetric trials (right/left step times 0/−25, +12.5/−12.5 and +25/0% different from preferred) and one highly asymmetric trial (R/L step times +25/−25% of preferred; figure 1). Here, 0 refers to a preferred step-time step (i.e. 0% different from preferred). Each asymmetric trial therefore had a stride time and both right and left step times comparable to one of the symmetric trials. For example, during the +25/−25% condition, the right leg had a step time comparable with the +25 per cent trial, a left step time comparable with the −25 per cent trial, and thus an overall stride time comparable with the 0 per cent trial.
Figure 1.
Step diagram of conditions. Subjects walked both symmetrically and asymmetrically at a range of target step and stride times. Subjects completed five symmetric trials (Target SI 0%), three moderately symmetric trials (Target SI approx. 25%), and one highly asymmetric trial (Target SI 50%). Here, 0% refers to a preferred step-time step. Subjects were able to match the target stride time for all conditions, but did not walk as asymmetrically as the target for the +12.5/−12.5% and +25/−25% conditions. Asterisks (*) actual SI significantly different from target. Significance at p < 0.05. Values are mean ± 1 s.d.
(b). Symmetry calculation
We define a step as being from heel strike of one foot to heel strike of the contralateral foot (i.e. a right step is from left heel strike to right heel strike). We calculated ratio index (RI) [21] as
| 2.1 |
We gave subjects RI feedback because it provided a simple, easily understood metric and implies a clear directionality. We chose target RI's greater than 1.0 for each asymmetric condition. Therefore, the right leg was always the ‘slow’ leg, with a step time greater than the left leg and slower than or equal to preferred. Conversely, the left leg was the ‘fast’ leg for all subjects with a step time less than the right leg and faster than or equal to preferred. We also calculated symmetry index (SI) [22–24] as
| 2.2 |
Note that SI = 0% for perfect symmetry and a larger SI indicates greater asymmetry. Although subjects received RI feedback, we present our results in terms of SI to more easily compare our results with the numerous previous studies that have used this metric.
(c). Experimental protocol
Subjects first stood quietly on the treadmill for 5 min while we measured their metabolic rate. Although all subjects had extensive experience with treadmill walking, we further familiarized all subjects to treadmill walking for 5 min at the experimental speed (1.25 m s−1). We measured their preferred stride frequency, which we used in all subsequent trials to determine target step and stride times.
We then introduced the subjects to the step time symmetry feedback. In real-time, a computer screen mounted at eye level graphically presented subjects with their actual step time RI as well as a target RI for that trial. Subjects walked for 5 min at 1.25 m s−1 with verbal instructions to explore how to increase and decrease their RI. Subjects had visual feedback of their RI for the remainder of the protocol, including during the symmetric trials. Additionally, all trials were accompanied by a metronome set to the appropriate step frequency and symmetry. The feedback and all instructions pertained only to step time. Throughout the experiment, subjects were free to choose their right and left step lengths within the constraint of matching the treadmill speed.
Subjects next completed the five symmetric trials (random order) for 5 min each. Subjects then performed the +12.5/−12.5% condition for 20 min and then completed the 0/−25, +25/0 and +25/−25% conditions in a random order for 7 min each. See the electronic supplementary materials for a demonstration video of the +12.5/−12.5% condition.
(d). Metabolic rate
We measured metabolic rate via expired gas analysis using a ParvoMedics TrueOne v. 2400 metabolic measurement system (Sandy, UT, USA). Subjects sat for 10 min prior to measurement of their standing metabolic rate and rested for 3 min between all trials. We calculated the average rates of O2 consumption and CO2 production over the last 2 min of each trial, calculated gross metabolic power [25] and subtracted standing power from gross power to find net metabolic power. These data are available in the electronic supplementary materials.
(e). Kinematics
Footswitches measured heel-strike and toe-off times throughout the experiment (B&L Engineering, Tustin, CA, USA). We used these data and equation (2.1) to provide subjects with real-time RI feedback. Following each asymmetric trial, we also used these data to calculate the percentage of the gait cycle taken up by the left step (39–45% depending on condition), which we used to synchronize the independently recorded ground reaction forces (GRFs).
(f). Mechanics
We measured the GRFs at 1000 Hz using a dual-belt treadmill with an AMTI force platform under one treadmill [26]. For all conditions, we recorded right foot GRF for 30 s during the last 1 min of each trial. For the asymmetric conditions, subjects then walked for 1 min in the other direction (i.e. with their left foot on the force treadmill). We measured left foot GRF for 30 s during this window.
For all conditions, we low-pass filtered the data at 20 Hz, used an 80 N force threshold to determine stance onset and offset times, and then constructed an average force profile for the first 15 complete strides. For the symmetric conditions, we duplicated and phase-shifted the right foot force profile by 50 per cent of the gait cycle to create a complete stride cycle from the right foot forces. For the asymmetric trials, we phase-shifted the left foot GRF by the percentage calculated using the footswitch data to create a right and left combined force profile. In software, we enforced zero net impulse across a stride such that average vertical force equalled the subject's weight and average horizontal force equalled zero. We then calculated individual limb power after Donelan et al. [20] (data available in the electronic supplementary materials).
(g). Statistics
Because steps both faster and slower than preferred are metabolically and mechanically different from preferred steps, we believe that the costs of asymmetry are best understood by comparing an asymmetric slow step/fast step stride to the corresponding symmetric slow–slow and fast–fast conditions. For example, we compared the +12.5/−12.5 per cent condition to the average of the same step time +12.5 per cent and −12.5 per cent conditions rather than to the same stride time 0 per cent condition. We therefore normalized the metabolic and mechanical power of each asymmetric trial to the average of these two relevant step time conditions. We then used a linear mixed effects regression to investigate how increasing SI affected metabolic and mechanical power.
We also tested whether subjects met the target stride time and SI for each condition using a one-sample t-test. Finally, we tested for a change in positive and negative mechanical work across three symmetric or asymmetric conditions using a one-factor repeated measure ANOVA. We performed this test for each foot when that foot was the leading leg, during single support and as the trailing leg. If we found a significant difference, we then followed up this comparison with three paired t-tests to identify specific within-comparison differences. We used R v. 2.13.1 (2011) for the regression analysis and Matlab v. 7.11.0 for all t-tests and ANOVA's. Our significance criterion was α < 0.05.
3. Results
Before explaining our novel findings for asymmetric gait, it is important to validate our approach with the symmetric results. We confirm previous findings on the metabolic cost of varying step time; symmetric steps slower and faster than preferred increased metabolic power [13–19]. Symmetric steps 25 per cent slower than preferred increased metabolic power by 30 per cent (approx. 0.97 W kg−1), while steps 25 per cent faster than preferred increased metabolic power by 42 per cent (1.35 W kg−1; figure 2a). Similarly, external mechanical power increased with increasing symmetric step time (i.e. longer steps; figure 2b). Each 12.5 per cent increase in symmetric step time resulted in a 6 per cent (0.024 W kg−1) increase in positive mechanical power production (figure 2b), consistent with previous findings [11,15].
Figure 2.
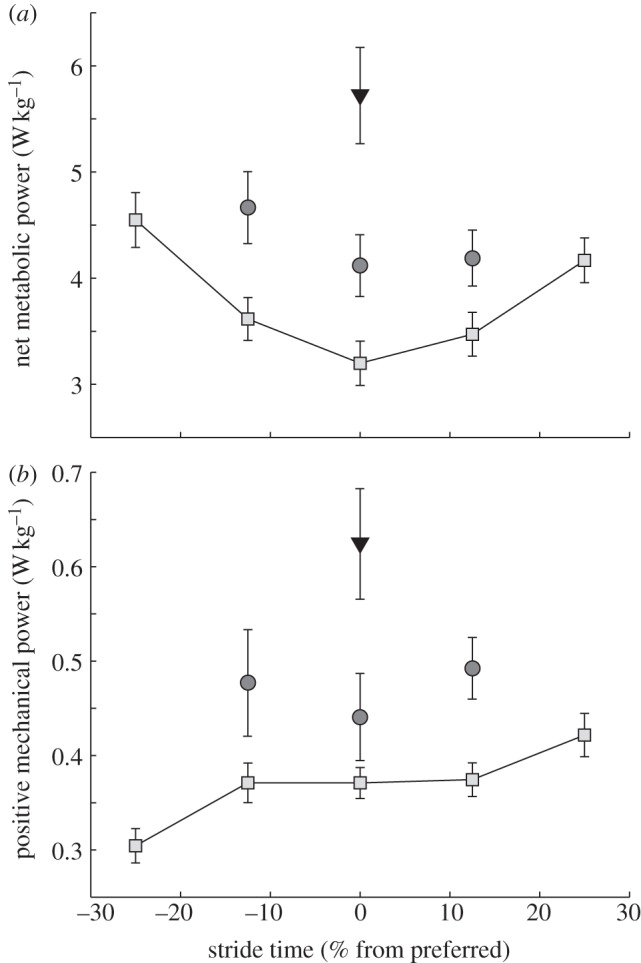
(a) Net metabolic power and (b) external mechanical power plotted versus stride time. Deviation from preferred in either dimension (symmetry or stride time) resulted in increasing metabolic and mechanical demands. Error bars are ± 1 s.e. Inverted triangles, highly asymmetric; circles, moderately asymmetric; squares, symmetric.
The moderately asymmetric conditions required 21–29 per cent (0.7–1.0 W kg−1) more metabolic power than symmetric steps at the same stride time (figure 2a). Moreover, the highly asymmetric, +25/−25% condition required 80 per cent (2.5 W kg−1) more metabolic power. Regression analysis revealed that asymmetric walking was also more expensive than the average cost of symmetric walking with corresponding fast and slow steps. We found that a 23 per cent asymmetry required 17 per cent (approx. 0.55 W kg−1) more metabolic power while a 42 per cent asymmetry required 31 per cent (approx. 1.0 W kg−1) more metabolic power than corresponding symmetric walking (p < 0.0001) (figure 2b). We also observed increases in external mechanical power production. Subjects produced 35 per cent more positive power for the moderately asymmetric conditions and 64 per cent more positive power for the highly asymmetric conditions (approx. 0.13 and 0.24 W kg−1, respectively, p < 0.0001; figure 2b).
We next examined subject's gait timings and mechanics more closely to better understand specifically how subjects responded to the perturbations. For both the symmetric and asymmetric conditions, subjects were able to successfully walk at the target stride time but did not walk as asymmetrically as the target for the +12.5/−12.5% and +25/−25% conditions (figure 1). We do not believe that this affected our conclusions because our regression analysis used subjects' actual rather than target asymmetry.
Subjects adjusted both their stance and swing times to reach the target asymmetry. In order to increase step time with their right leg, subjects chose correspondingly longer stance durations with their left leg. Increasing asymmetry resulted in a longer left stance time, a shorter right stance time and a faster left-to-right transition relative to symmetric walking at the same stride time. We also observed changes in the vertical and horizontal GRF produced by each leg and in CoM velocity (figure 3). Consistent with symmetric walking, during asymmetric trials, subjects generally moved forward more quickly during double support and slower at mid-stance. Unlike symmetric walking, however, horizontal CoM velocity was slower at left mid-stance than at right mid-stance, well beyond the small CoM trajectory asymmetries observed in healthy human gait [27].
Figure 3.
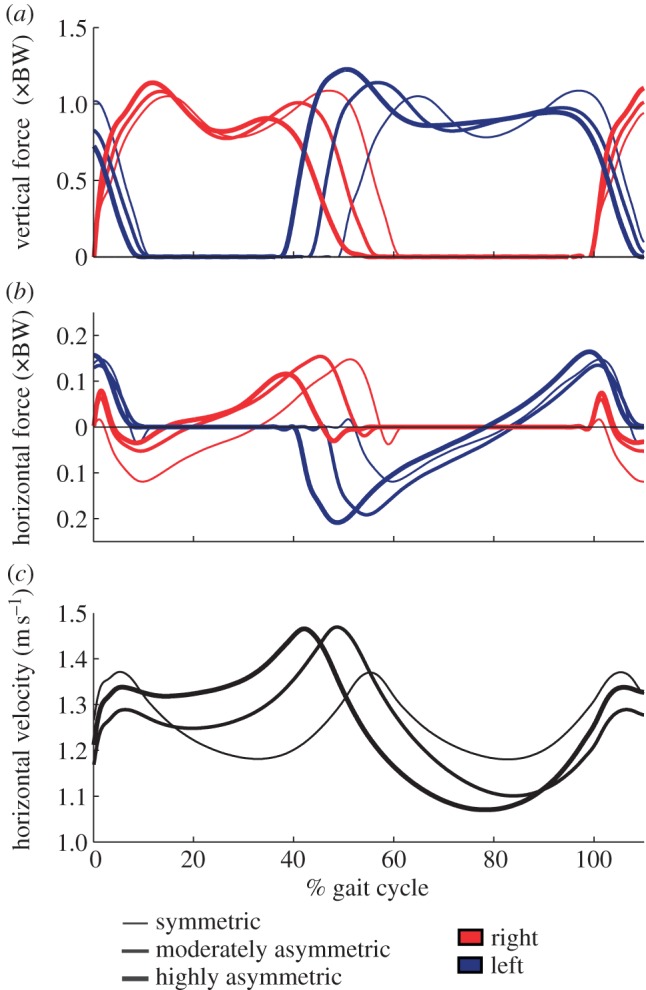
(a) Ensemble average vertical and (b) horizontal GRF and horizontal CoM velocity (c) profiles for the 0% (symmetric), +12.5/−12.5% (moderately asymmetric) and +25/−25% (highly asymmetric) conditions. The right, ‘slow’ leg demonstrated reduced vertical force during late stance and reduced braking force during early stance. The left, ‘fast’ leg produced more vertical and braking force during early stance. Consistent with symmetric walking, horizontal CoM velocity was faster during double support and slower at mid-stance. Unlike symmetric walking, however, horizontal CoM velocity was slower at left mid-stance than during right mid-stance. (Online version in colour.)
Subjects also shifted when during a stride they were absorbing and producing power with each leg (figure 4). We broke down stance into three phases (double support as the leading leg, single support and double support as the trailing leg) and calculated the work performed by each leg during each phase (figure 5). To control for the effects of stance and step duration on mechanics, we compared the work performed across the −25, 0/−25 and +25/−25% conditions for the right leg, because stance duration was nearly constant across these conditions (contact times of 0.49, 0.51 and 0.52 s, respectively). Similarly, we made this comparison across the +25, +25/0 and +25/−25% conditions for the left leg (contact times of 0.79, 0.77 and 0.72 s).
Figure 4.
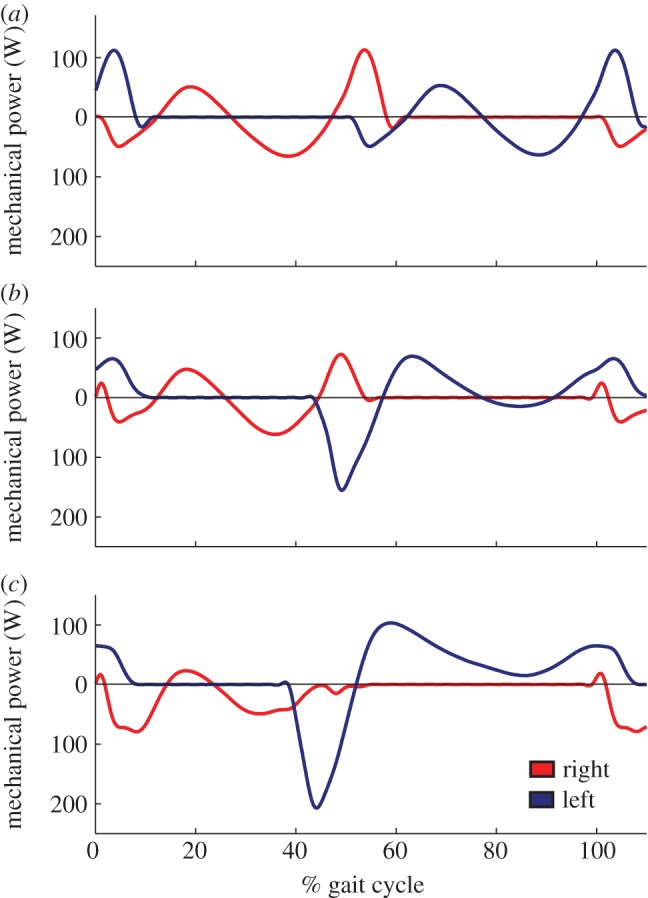
Ensemble average mechanical power across a stride for (a) the 0% (symmetric), (b) +12.5/−12.5% (moderately asymmetric) and (c) +25/−25% (highly asymmetric) conditions. (Online version in colour.)
Figure 5.
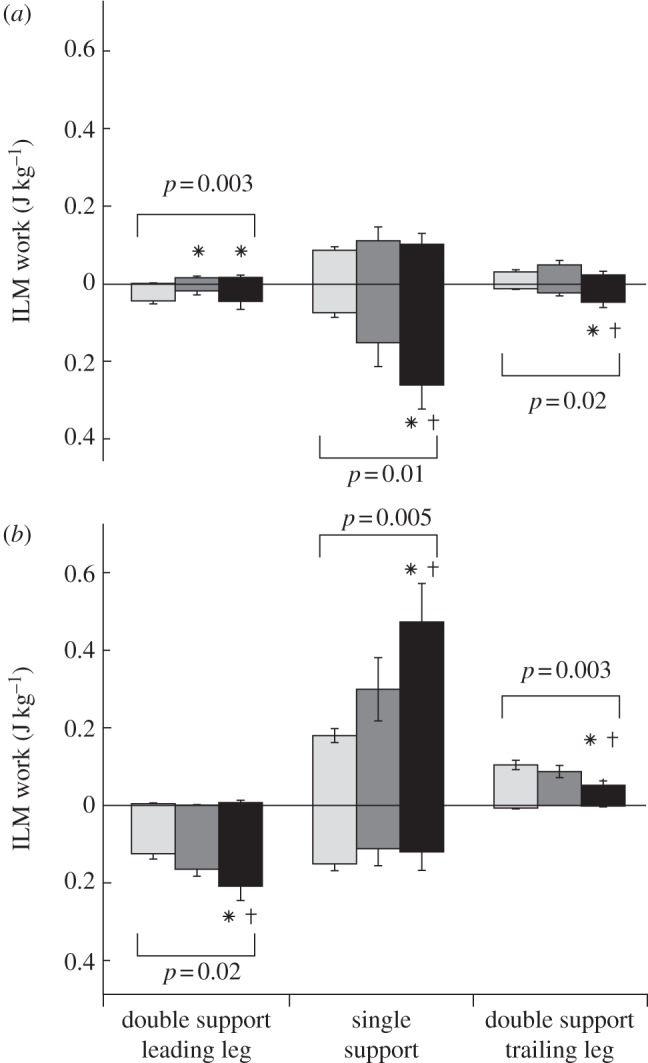
Individual limb work for the (a) right and (b) left leg as the leading leg, during single support and as the trailing leg. For the right leg, we compared the −25, 0/−25 and +25/−25% conditions, whereas for the left leg, we compared the +25%, +25/0% and +25%/−25% conditions because stance duration was nearly constant across these conditions. With increasing asymmetry, subjects performed more negative work with their right leg during single support, and with their left leg as the leading leg. Subjects performed more positive work during single support on the left leg. Asterisks (*) significantly different from symmetric and dagger symbols (†) significantly different from moderately asymmetric. Significance at p < 0.05. Error bars are ± 1 s.e. black bars, highly asymmetric; dark grey bars, moderately asymmetric; light grey bars, symmetric.
Increasing asymmetry resulted in a redistribution of when during the gait cycle subjects performed positive and negative work (figure 5). For the right, ‘slow’ leg, increasing asymmetry resulted in an increase in negative work performed during single support. The left, ‘fast’ leg performed substantially more negative work as the lead leg during the left-to-right transition and less positive work as the trailing leg during the right-to-left transition. Subjects then performed dramatically more positive work during left leg single support.
4. Discussion
We accept our first hypothesis; walking with asymmetric step times required 21–80% (0.7–2.5 W kg−1) more metabolic power than symmetric walking at comparable stride times. Asymmetric walking was also 17–31% (approx. 0.55–1.0 W kg−1) more expensive than symmetric walking at comparable, non-preferred step times (figure 2a). We find that gait asymmetry itself is intrinsically metabolically expensive beyond the costs imposed by non-preferred step times.
Many previous studies have shown that preferred human gait involves combinations of step width, length and duration that minimize energy expenditure [14,15]. Here, we have identified symmetry as another energy-minimizing objective in healthy human gait. Interestingly, walking asymmetrically at non-preferred stride times required increased metabolic power in parallel with the increases observed during symmetric walking (figure 2a, moderately symmetric points). We therefore clarify two discrete metabolic costs in human walking; both step symmetry and stride time are independently optimized during preferred human walking.
Mechanical power production during the asymmetric conditions paralleled the observed increases in metabolic power (figure 2b). For example, a 23 per cent asymmetry required 35 per cent more positive mechanical power production than symmetric walking at comparable step times. We therefore accept our second hypothesis and suggest that increases in external mechanical power production largely explain the observed increases in metabolic power.
We reject our third hypothesis. With increasing asymmetry, subjects performed less positive push-off work and more negative collision work during double support (figure 5) but compensated with dramatically more positive work during left leg single support. These findings are generally consistent with those of Soo & Donelan [10], who suggest that an appropriate coordination of push-off and collision minimizes the step-to-step transition work required for human walking. That subjects compensate during single support also emphasizes the importance of single support work during walking [28].
Our results also have important clinical applications. Substantial gait asymmetries are common in people recovering from stroke, injury, joint replacement and amputation [29–32]. A substantial portion of the higher metabolic cost of walking observed in these populations [33] may be due to the asymmetry itself. We also find that the costs of slight asymmetry are probably relatively small. Our regression results suggest that an asymmetry of 1 per cent would require only a 0.74 per cent increase in net metabolic power, consistent with those of Srinivasan [34], who predicts only minimal costs to small amounts of gait asymmetry.
One limitation is that we examined only external mechanical power. To complete the asymmetric trials, subjects tended to delay their right leg swing to achieve a slower step time while swinging their left leg more quickly, altering the demand for internal mechanical power. Part of the increase in metabolic cost may therefore be attributed to changing leg-swing dynamics, which have been shown to exact a metabolic cost during human walking [35]. We estimated internal power (the rate at which work is performed to swing the legs relative to the CoM) during each of our conditions using previously published equations [36,37]. We found that the duty factor and stride frequency observed in the asymmetric conditions do not predict any change in internal work relative to symmetric walking. We believe that although swinging the fast leg required additional internal power, this was offset by the reduced internal power demand for swinging the other leg.
Although subjects received identical forms of feedback during both the symmetric and asymmetric trials, part of the increased metabolic cost may be due to the novel nature of the task. Asymmetric gait required dramatically more mental effort than walking symmetrically, even at non-preferred stride times. We did not observe any significant changes in metabolic power during the initial 20 min trial, but it remains possible that additional practice would reduce metabolic power. Additionally, subjects may have used different control strategies to complete the asymmetric trials, for example, walking with increased muscle co-activation, which would probably impose a metabolic penalty.
We did not constrain spatial kinematic variables beyond indirectly constraining stride length; subjects were free to choose both step lengths. Most subjects had a preferred step length asymmetry direction; their ‘fast’ leg always took a step either longer or shorter than their ‘slow’ leg, which was consistent for a given subject but varied between subjects.
We found severe metabolic costs associated with temporal asymmetry. We can only speculate that spatial asymmetry would impose similar metabolic and mechanical costs. Previously, Kim & Eng [38] showed that temporal but not spatial gait symmetry was correlated with GRF symmetry in stroke patients, suggesting that imposed spatial asymmetry may be less expensive than observed for temporal asymmetry in the present study. However, both spatial and temporal asymmetry would probably require asymmetric CoM displacements and velocities (figure 3), which might require additional mechanical work. Measurement of the spatial features of imposed asymmetry and quantification of the results might be a fruitful area for future investigation.
Although we find that symmetry is broadly optimal in healthy adults, these findings do not necessarily support symmetry as an end goal in rehabilitation for individuals with pathological gait asymmetry [38]. Asymmetric gait may be a less metabolically costly strategy for controlling a highly asymmetric system, such as in an individual with the large, physical asymmetries associated with many gait pathologies. Additionally, minimizing metabolic cost is only one possible optimality criteria in human gait; gait asymmetry may be adaptive, for example, by improving impaired balance [39], minimizing joint pain [40] or simplifying control [41]. Indeed, we suspect that asymmetry may be overall optimal in individuals with large physical asymmetries.
In summary, walking with asymmetric step times required substantial metabolic costs above those imposed by non-preferred step times. Increased positive, external mechanical power production paralleled the observed increase in metabolic cost. Subjects walked by performing less positive push-off work and more negative collision work, while compensating with additional positive work during single support. We identify inherent metabolic and mechanical costs to gait asymmetry and find that symmetry is optimal in healthy adults.
References
- 1.Cavagna GA, Heglund NC, Taylor CR. 1977. Mechanical work in terrestrial locomotion: two basic mechanisms for minimizing energy expenditure. Am. J. Physiol. 233, 243–261 [DOI] [PubMed] [Google Scholar]
- 2.Alexander RMcN, Bennet-Clark HC. 1977. Storage of elastic strain energy in muscle and other tissues. Nature 265, 114–117 10.1038/265114a0 (doi:10.1038/265114a0) [DOI] [PubMed] [Google Scholar]
- 3.Cavagna GA, Saibene FP, Margaria R. 1964. External work in walking. J. Appl. Physiol. 18, 1–9 [DOI] [PubMed] [Google Scholar]
- 4.Cavagna GA, Thys H, Zamboni A. 1976. The sources of external work in level walking and running. J. Physiol. 262, 639–657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hildebrand M. 1977. Analysis of asymmetrical gaits. J. Mammal. 58, 131–56 10.2307/1379571 (doi:10.2307/1379571) [DOI] [Google Scholar]
- 6.Hildebrand M. 1968. Symmetrical gaits of dogs in relation to body build. J. Morph. 124, 353–359 10.1002/jmor.1051240308 (doi:10.1002/jmor.1051240308) [DOI] [PubMed] [Google Scholar]
- 7.Hildebrand M. 1976. Analysis of tetrapod gaits: general considerations and symmetrical gaits. In Neural control of locomotion (eds Herman RM, Grillner S, Stein P, Stuart DG.), pp. 203–236 New York, NY: Plenum Press [Google Scholar]
- 8.Farley CT, Ferris DP. 1998. Biomechanics of walking and running: center of mass movements to muscle action. Exerc. Sport Sci. Rev. 26, 253–286 [PubMed] [Google Scholar]
- 9.Soo CH, Donelan JM. 2010. Mechanics and energetics of step-to-step transitions isolated from human walking. J. Exp. Biol. 213, 4265–4271 10.1242/jeb.044214 (doi:10.1242/jeb.044214) [DOI] [PubMed] [Google Scholar]
- 10.Soo CH, Donelan JM. 2012. Coordination of push-off and collision determine the mechanical work of step-to-step transitions when isolated from human walking. Gait Posture 35, 292–297 10.1016/j.gaitpost.2011.09.102 (doi:10.1016/j.gaitpost.2011.09.102) [DOI] [PubMed] [Google Scholar]
- 11.Donelan JM, Kram R, Kuo AD. 2002. Mechanical work for step-to-step transitions is a major determinant of the metabolic cost of human walking. J. Exp. Biol. 205, 3717–3727 [DOI] [PubMed] [Google Scholar]
- 12.Kuo AD, Donelan JM, Ruina A. 2005. Energetic consequences of walking like an inverted pendulum. Exerc. Sport Sci. Rev. 33, 88–97 10.1097/00003677-200504000-00006 (doi:10.1097/00003677-200504000-00006) [DOI] [PubMed] [Google Scholar]
- 13.Umberger BR, Martin PE. 2007. Mechanical power and efficiency of level walking with different stride rates. J. Exp. Biol. 210, 3255–3265 10.1242/jeb.000950 (doi:10.1242/jeb.000950) [DOI] [PubMed] [Google Scholar]
- 14.Donelan JM, Kram R, Kuo AD. 2001. Mechanical and metabolic determinants of the preferred step width in human walking. Proc. R. Soc. Lond. B 268, 1985–1992 10.1098/rspb.2001.1761 (doi:10.1098/rspb.2001.1761) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Minetti AE, Capelli C, Zamparo P, Di Prampero PE, Saibene FP. 1995. Effects of stride frequency on mechanical power and energy expenditure of walking. Med. Sci. Sports Exerc. 27, 1194–1202 10.1249/00005768-199508000-00014 (doi:10.1249/00005768-199508000-00014) [DOI] [PubMed] [Google Scholar]
- 16.Molen NH, Rozendal RH, Boon W. 1972. Graphic representation of the relationship between oxygen-consumption and characteristics of normal gait of the human male. Proc. K. Ned. Akad. Wet. C 75, 305–314 [PubMed] [Google Scholar]
- 17.Zarrugh MY, Radcliffe CW. 1978. Predicting metabolic cost of level walking. Eur. J. Appl. Physiol. 38, 215–223 10.1007/BF00430080 (doi:10.1007/BF00430080) [DOI] [PubMed] [Google Scholar]
- 18.Minetti AE, Alexander RMcN. 1997. Theory of metabolic costs for bipedal gaits. J. Theor. Biol. 186, 467–476 10.1006/jtbi.1997.0407 (doi:10.1006/jtbi.1997.0407) [DOI] [PubMed] [Google Scholar]
- 19.Cotes JE, Meade F. 1960. The energy expenditure and mechanical energy demand in walking. Ergonomics 3, 97–119 10.1080/00140136008930473 (doi:10.1080/00140136008930473) [DOI] [Google Scholar]
- 20.Donelan JM, Kram R, Kuo AD. 2002. Simultaneous positive and negative external mechanical work in human walking. J. Biomech. 35, 117–124 10.1016/S0021-9290(01)00169-5 (doi:10.1016/S0021-9290(01)00169-5) [DOI] [PubMed] [Google Scholar]
- 21.Sadeghi H, Allard P, Prince F, Labelle H. 2000. Symmetry and limb dominance in able-bodied gait: a review. Gait Posture 12, 34–45 10.1016/S0966-6362(00)00070-9 (doi:10.1016/S0966-6362(00)00070-9) [DOI] [PubMed] [Google Scholar]
- 22.Bosch K, Rosenbaum D. 2010. Gait symmetry improves in childhood: a 4-year follow-up of foot loading data. Gait Posture 32, 464–468 10.1016/j.gaitpost.2010.07.002 (doi:10.1016/j.gaitpost.2010.07.002) [DOI] [PubMed] [Google Scholar]
- 23.Robinson RO, Herzog W, Nigg BM. 1987. Use of force platform variables to quantify the effects of chiropractic manipulation on gait symmetry. J. Manip. Physiol. Ther. 10, 172–176 [PubMed] [Google Scholar]
- 24.Herzog W, Nigg BM, Read L, Olsson E. 1989. Asymmetries in ground reaction force patterns in normal human gait. Med. Sci. Sports Exerc. 21, 110–114 10.1249/00005768-198902000-00020 (doi:10.1249/00005768-198902000-00020) [DOI] [PubMed] [Google Scholar]
- 25.Brockway JM. 1987. Derivation of formulae used to calculate energy-expenditure in man. Hum. Nutr. Clin. Nutr. 41C, 463–471 [PubMed] [Google Scholar]
- 26.Browning RC, Kram R. 2007. Effects of obesity on the biomechanics of walking at different speeds. Med. Sci. Sports Exerc. 39, 1632–1641 10.1249/mss.0b013e318076b54b (doi:10.1249/mss.0b013e318076b54b) [DOI] [PubMed] [Google Scholar]
- 27.Minetti AE, Cisotti C, Mian OS. 2011. The mathematical description of the body centre of mass 3D path in human and animal locomotion. J. Biomech. 44, 1471–1477 10.1016/j.jbiomech.2011.03.014 (doi:10.1016/j.jbiomech.2011.03.014) [DOI] [PubMed] [Google Scholar]
- 28.Neptune RR, Zajac FE, Kautz SA. 2004. Muscle mechanical work requirements during normal walking: the energetic cost of raising the body's center-of-mass is significant. J. Biomech. 37, 817–825 10.1016/j.jbiomech.2003.11.001 (doi:10.1016/j.jbiomech.2003.11.001) [DOI] [PubMed] [Google Scholar]
- 29.Hesse S, Reiter F, Jahnke M, Dawson M, Sarkodie-Gyan T, Mauritz KH. 1997. Asymmetry of gait initiation in hemiparetic stroke subjects. Arch. Phys. Med. Rehab. 78, 719–724 10.1016/S0003-9993(97)90079-4 (doi:10.1016/S0003-9993(97)90079-4) [DOI] [PubMed] [Google Scholar]
- 30.Isakov E, Keren O, Benjuya N. 2000. Transtibial amputee gait: time-distance parameters and EMG activity. Prosth. Orthot. Int. 24, 216–220 10.1080/03093640008726550 (doi:10.1080/03093640008726550) [DOI] [PubMed] [Google Scholar]
- 31.Noyes FR, Barber SD, Mangine RE. 1991. Abnormal lower limb symmetry determined by function hop tests after anterior cruciate ligament rupture. Am. J. Sports Med. 19, 513–518 10.1177/036354659101900518 (doi:10.1177/036354659101900518) [DOI] [PubMed] [Google Scholar]
- 32.McCrory JL, White SC, Lifeso RM. 2001. Vertical ground reaction forces: objective measures of gait following hip arthroplasty. Gait Posture 14, 104–109 10.1016/S0966-6362(01)00140-0 (doi:10.1016/S0966-6362(01)00140-0) [DOI] [PubMed] [Google Scholar]
- 33.Waters RL, Mulroy S. 1999. The energy expenditure of normal and pathologic gait. Gait Posture 9, 207–231 10.1016/S0966-6362(99)00009-0 (doi:10.1016/S0966-6362(99)00009-0) [DOI] [PubMed] [Google Scholar]
- 34.Srinivasan M. 2011. Fifteen observations on the structure of energy-minimizing gaits in many simple biped models. J. R. Soc Interface 8, 74–98 10.1098/rsif.2009.0544 (doi:10.1098/rsif.2009.0544) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Doke J, Donelan JM, Kuo AD. 2005. Mechanics and energetics of swinging the human leg. J. Exp. Biol. 208, 439–445 10.1242/jeb.01408 (doi:10.1242/jeb.01408) [DOI] [PubMed] [Google Scholar]
- 36.Minetti AE. 1998. A model equation for the prediction of mechanical internal work of terrestrial locomotion. J. Biomech. 31, 463–468 10.1016/S0021-9290(98)00038-4 (doi:10.1016/S0021-9290(98)00038-4) [DOI] [PubMed] [Google Scholar]
- 37.Nardello F, Ardigò LP, Minetti AE. 2011. Measured and predicted mechanical internal work in human locomotion. Hum. Mov. Sci. 30, 90–104 10.1016/j.humov.2010.05.012 (doi:10.1016/j.humov.2010.05.012) [DOI] [PubMed] [Google Scholar]
- 38.Kim CM, Eng JJ. 2003. Symmetry in vertical ground reaction force is accompanied by symmetry in temporal but not distance variables of gait in persons with stroke. Gait Posture 18, 23–28 10.1016/S0966-6362(02)00122-4 (doi:10.1016/S0966-6362(02)00122-4) [DOI] [PubMed] [Google Scholar]
- 39.Hof AL, van Bockel RM, Schoppen T, Postema K. 2007. Control of lateral balance in walking: experimental findings in normal subjects and above-knee amputees. Gait Posture 25, 250–258 10.1016/j.gaitpost.2006.04.013 (doi:10.1016/j.gaitpost.2006.04.013) [DOI] [PubMed] [Google Scholar]
- 40.Powers CM, Heino JG, Rao S, Perry J. 1999. The influence of patellofemoral pain on lower limb loading during gait. Clin. Biomech. 14, 722–728 10.1016/s0268-0033(99)00019-4 (doi:10.1016/s0268-0033(99)00019-4) [DOI] [PubMed] [Google Scholar]
- 41.Collins JJ, Stewart IN. 1993. Coupled nonlinear oscillators and the symmetries of animal gaits. J. Nonlinear Sci. 3, 349–392 10.1007/BF02429870 (doi:10.1007/BF02429870) [DOI] [Google Scholar]