Abstract
Cells release into the extracellular environment diverse types of membrane vesicles of endosomal and plasma membrane origin called exosomes and microvesicles, respectively. These extracellular vesicles (EVs) represent an important mode of intercellular communication by serving as vehicles for transfer between cells of membrane and cytosolic proteins, lipids, and RNA. Deficiencies in our knowledge of the molecular mechanisms for EV formation and lack of methods to interfere with the packaging of cargo or with vesicle release, however, still hamper identification of their physiological relevance in vivo. In this review, we focus on the characterization of EVs and on currently proposed mechanisms for their formation, targeting, and function.
Introduction
Intercellular communication is an essential hallmark of multicellular organisms and can be mediated through direct cell–cell contact or transfer of secreted molecules. In the last two decades, a third mechanism for intercellular communication has emerged that involves intercellular transfer of extracellular vesicles (EVs). Although the release of apoptotic bodies during apoptosis has been long known (Hristov et al., 2004), the fact that also perfectly healthy cells shed vesicles from their plasma membrane has only recently become appreciated. These vesicles are generally referred to as microvesicles, ectosomes, shedding vesicles, or microparticles among others (Holme et al., 1994; Hess et al., 1999; Cocucci et al., 2009; György et al., 2011). The term exosome was initially used for vesicles ranging from 40 to 1,000 nm that are released by a variety of cultured cells (Trams et al., 1981), but the subcellular origin of these vesicles remained unclear. Later, this nomenclature was adopted for 40–100-nm vesicles released during reticulocyte differentiation as a consequence of multivesicular endosome (MVE) fusion with the plasma membrane (Harding et al., 1984; Pan et al., 1985). One decade later, exosomes were found to be released by B lymphocytes and dendritic cells through a similar route (Raposo et al., 1996; Zitvogel et al., 1998). The involvement of MVEs was demonstrated by the observation that fusion with the plasma membrane released exosomes together with previously endocytosed colloidal gold (Fig. 1; see Harding et al. in this issue). Several additional cell types of both hematopoietic and nonhematopoietic origin, such as cytotoxic T cells, platelets, mast cells, neurons, oligodendrocytes, Schwann cells, and intestinal epithelial cells, were also shown to release exosomes through MVE fusion with the cell surface (Simons and Raposo, 2009; Théry et al., 2009). That exosomes can also be secreted in vivo had already been proposed by observations that vesicles from prostate epithelial cells (prostasomes) correspond in size to the intraluminal vesicles (ILVs) of storage vacuoles (the equivalent of MVEs) in these cells (Ronquist and Brody, 1985). Vesicles with hallmarks of exosomes have been isolated from diverse body fluids, including semen (Ronquist and Brody, 1985; Park et al., 2011; Aalberts et al., 2012), blood (Caby et al., 2005), urine (Pisitkun et al., 2004), saliva (Ogawa et al., 2011), breast milk (Admyre et al., 2007), amniotic fluid (Asea et al., 2008), ascites fluid (Andre et al., 2002), cerebrospinal fluid (Vella et al., 2007), and bile (Masyuk et al., 2010). Most of these studies attributed the isolated vesicles to exosomes because of their exosome-like protein contents. However, circulating vesicles are likely composed of both exosomes and microvesicles (MVs), and currently available purification methods, as discussed later, do not allow one to fully discriminate between exosomes and MVs. That a single cell type releases both exosomes and MVs has, for example, either been demonstrated or suggested for platelets (Heijnen et al., 1999), endothelial cells (Deregibus et al., 2007), and breast cancer cells (Muralidharan-Chari et al., 2009). Confusion on the origin and nomenclature of EVs has spread through the literature as well because vesicles with the size of exosomes that bud at the plasma membrane have also been called exosomes (Booth et al., 2006). For clarity, in this review, we will exclusively refer to exosomes as EVs originating from MVEs and to MVs for those EVs that are shed from the plasma membrane (Fig. 2). It should be noted that most studies have not clearly defined the origin of EVs under study; therefore, we will mostly refer to EVs rather than MVs or exosomes. A major ongoing challenge is to establish methods that will allow one to discriminate between exosomes and MVs. Differences in properties such as size, morphology, buoyant density, and protein composition seem insufficient for a clear distinction (Bobrie et al., 2011). Only when we are able to interfere with the molecular machineries required for EV formation and cargo sequestration will their origins be optimally determined. Such knowledge will also open new avenues to resolve their respective functions. In this review, we will highlight and discuss current experimental limitations that need to be resolved and the state of the art on the cell biology of EVs and their possible functions.
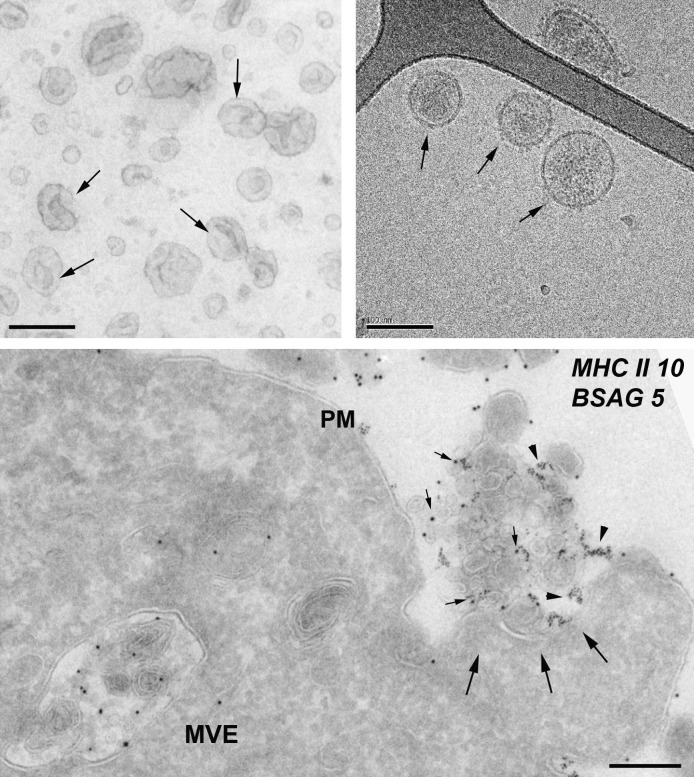
Figure 1.
Ultrastructure of exosomes. (top left) Exosomes isolated from melanoma cells were contrasted with uranyl-acetate and embedded as whole mount preparations in methylcellulose. Note their artificial cup shape appearance (examples are indicated with arrows) and heterogeneous size ranging from 30 to 100 nm. (top right) Exosomes from prostate epithelial cells (prostasomes) were directly frozen and observed by cryo–electron microscopy without chemical fixation or contrasting. Exosomes appear round and are visualized with improved resolution (arrows). The elongated structure (top right of the micrograph) is the Formvar film on the EM grid. (bottom) EBV-transformed B lymphocytes were allowed to endocytose BSA coupled to 5-nm gold particles (BSAG 5) for 10 min and then chased for 20 min in the absence of BSAG 5. Ultrathin cryosections were immunolabeled for MHC class II with 10-nm protein A gold. An MVE fusion profile (arrows) is defined by regurgitated 5-nm BSAG 5 that had previously been endocytosed. In addition to BSAG 5 (arrowheads), the exocytic profile contains exosomes labeled for MHC class II with 10-nm gold (MHC II 10; small arrows). PM, plasma membrane. Bars, 100 nm.
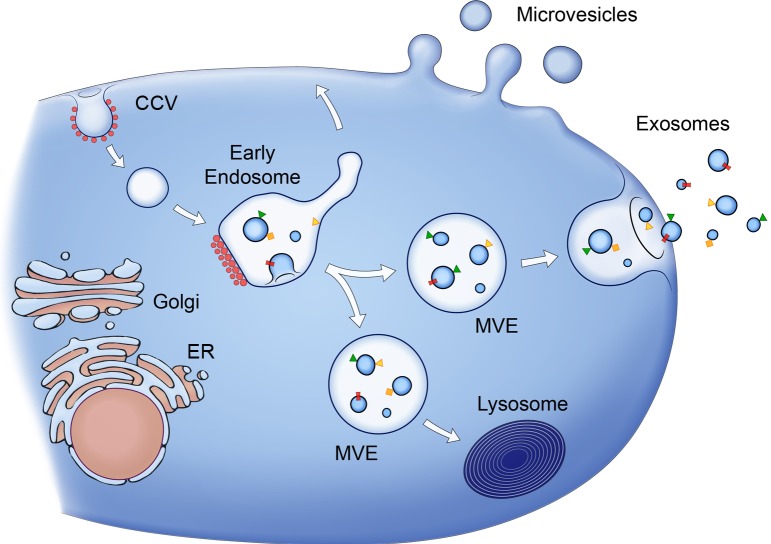
Figure 2.
Release of MVs and exosomes. MVs bud directly from the plasma membrane, whereas exosomes are represented by small vesicles of different sizes that are formed as the ILV by budding into early endosomes and MVEs and are released by fusion of MVEs with the plasma membrane. Other MVEs fuse with lysosomes. The point of divergence between these types of MVEs is drawn at early endosomes, but the existence of distinct early endosomes feeding into these two pathways cannot be excluded. Red spots symbolize clathrin associated with vesicles at the plasma membrane (clathrin-coated vesicles [CCV]) or bilayered clathrin coats at endosomes. Membrane-associated and transmembrane proteins on vesicles are represented as triangles and rectangles, respectively. Arrows represent proposed directions of protein and lipid transport between organelles and between MVEs and the plasma membrane for exosome secretion.
Isolation and characterization of EVs
One major challenge in the field is to improve and standardize methods for EV isolation and analysis (Théry et al., 2006). Currently, EVs are mostly isolated from the supernatants of cultured cells grown in fetal calf serum depleted of EVs by performing differential ultracentrifugation. Next, EVs can be efficiently separated from nonmembranous particles, such as protein aggregates, by using their relatively low buoyant density (Raposo et al., 1996; Escola et al., 1998; van Niel et al., 2003; Wubbolts et al., 2003), and differences in floatation velocity can be used to separate differently sized classes of EVs (Aalberts et al., 2012). The size of exosomes is equivalent to that of the ILVs of the MVEs from which they originate (40–100-nm diameter). MVs are generally larger (up to ∼1,000 nm in diameter), but also small vesicles (100 nm) may bud from the cell surface (Booth et al., 2006). Additional purification can be achieved by immunoadsorption (Wubbolts et al., 2003) using a protein of interest, which also selects for vesicles with an exoplasmic or outward orientation. Because of the increasing interest in exosomes and other EVs and their potential use in therapeutics or as biomarkers for disease, commercially available kits that allow for “easy isolation procedures” are being developed and marketed. Such approaches should be taken cautiously because they often fail to distinguish between differently sized EVs and membrane-free macromolecular aggregates. Further characterization of isolated EVs requires complementary biochemical (immunoblotting), mass spectrometry, and imaging techniques. Whereas large EVs are most often analyzed by conventional electron microscopy, small EVs can also be observed as “whole mount” samples when deposited without sectioning on electron microscopy grids (Raposo et al., 1996). In the latter approach, EVs may collapse during drying, resulting in a cup-shaped morphology, which is often considered erroneously as a typical feature of exosomes (Raposo et al., 1996). Quickly frozen, vitrified vesicles analyzed by cryo–electron microscopy indeed show that exosomes and other EVs have a perfectly rounded shape (Fig. 1 and not depicted; Conde-Vancells et al., 2008). Complementary to electron microscopy, nanoparticle tracking analysis allows determination of the size distribution of isolated EVs based on the Brownian motion of vesicles in suspension (Soo et al., 2012). Because conventional flow cytometers cannot distinguish between vesicles that are <300 nm, a novel high resolution flow cytometry–based method has been recently developed for quantitative high throughput analysis of individual (immunolabeled) nanosized vesicles (Nolte-’t Hoen et al., 2012b; van der Vlist et al., 2012).
The molecular composition of EVs.
The protein content of EVs from different sources has been analyzed by SDS-PAGE followed by protein staining, immunoblotting, or proteomic analysis. Highly purified EVs should be devoid of pollutants, such as serum proteins and protein components of intracellular compartments (e.g., the endoplasmic reticulum or mitochondria), that are never in contact with EVs. As a consequence of their origin, exosomes from different cell types contain endosome-associated proteins (e.g., Rab GTPase, SNAREs, Annexins, and flotillin), some of which are involved in MVE biogenesis (e.g., Alix and Tsg101; van Niel et al., 2006). Membrane proteins that are known to cluster into microdomains at the plasma membrane or at endosomes often are also enriched on EVs. These include tetraspanins, a family of >30 proteins that are composed of four transmembrane domains (Hemler, 2003). Tetraspanins such as CD63, CD81, CD82, CD53, and CD37 were first identified in B cell exosomes in which they can be enriched >100-fold relative to the transferrin receptor, which in this cell type can be considered as a genuine marker for both the plasma membrane and early endosomes (Escola et al., 1998). Other studies confirmed that tetraspanins are abundant in EVs from other sources (Zöller, 2009). Although tetraspanin-enriched membrane domains are distinct from detergent-resistant lipid–protein rafts (Hemler, 2008), EVs are also enriched in proteins that associate with lipid rafts, including glycosylphosphatidylinositol-anchored proteins and flotillin (Théry et al., 1999; Wubbolts et al., 2003). In comparison to the plasma membrane, exosomes from a variety of cells (Wubbolts et al., 2003; Laulagnier et al., 2004; Subra et al., 2007; Brouwers et al., 2012) are highly enriched in cholesterol, sphingomyelin, and hexosylceramides at the expense of phosphatidylcholine and phosphatidylethanolamine. The fatty acids in exosomes are mostly saturated or monounsaturated. Together with the high concentration of cholesterol, this may account for lateral segregation of these lipids into ILVs/exosomes during their formation at MVEs. Less is known of the protein and lipid contents of MVs and whether particular components are enriched on MVs relative to their originating plasma membrane.
A major breakthrough was the demonstration that the cargo of EVs included both mRNA and miRNA and that EV-associated mRNAs could be translated into proteins by target cells (Ratajczak et al., 2006; Valadi et al., 2007). Later studies reported on the RNA contents of EV isolates from other cell cultures (Skog et al., 2008) and from body fluids (Hunter et al., 2008; Rabinowits et al., 2009; Michael et al., 2010). EVs with features of exosomes released by immune cells have been demonstrated to selectively incorporate miRNA that can be functionally transferred as a consequence of fusion with recipient cells (Mittelbrunn et al., 2011; Montecalvo et al., 2012). Recently, analysis of RNA from EVs by unbiased deep sequencing approaches demonstrated that, in addition to mRNA and miRNA, EVs also contain a large variety of other small noncoding RNA species, including RNA transcripts overlapping with protein coding regions, repeat sequences, structural RNAs, tRNA fragments, vault RNA, Y RNA, and small interfering RNAs (Bellingham et al., 2012; Nolte-’t Hoen et al., 2012a). Many RNAs that were isolated with EVs were found to be enriched relative to the RNA profiles of the originating cells (Ratajczak et al., 2006; Valadi et al., 2007; Skog et al., 2008; Nolte-’t Hoen et al., 2012a), indicating that RNA molecules are selectively incorporated into EVs. It is important to note that many studies failed to demonstrate whether identified extracellular RNAs were truly associated with EVs or rather with RNA–protein complexes that may have been co-isolated with EVs. Whether RNAs are within the cytosolic lumen or associated with the outer membrane of EVs can be achieved by measuring flotation into sucrose gradients and resistance to RNase digestion subsequent to protease treatment. Also, different RNA isolation methods give extensive variation in exosomal RNA yield and patterns (Eldh et al., 2012), and such experimental variations between studies, together with the lack of quantitative data, make it impossible to make a comparative inventory of the RNA species assigned to EVs so far.
The database ExoCarta (http://www.exocarta.org) catalogs proteins, lipids, and RNA that have been identified in EVs from different sources. As it is, the components listed may correspond to both MVs and exosomes. This catalog has recently been updated as the compendium Vesiclepedia that will continuously be supplemented by novel contributions from different groups working in the field, using as much as possible equivalent and standardized EV isolation protocols (Kalra et al., 2012).
Biogenesis of EVs and cargo selection
Because exosomes are formed in MVEs and MVs originate by direct budding from the plasma membrane (Fig. 1 and Fig. 2), the cellular machineries involved in their formation and release are likely to differ, although mechanistic elements may be shared. Within the lysosomal pathway, MVEs are prone to fuse with lysosomes for degradation of their contents, differently from the itinerary of secretory MVEs. We and others have provided biochemical and morphological evidence that these two distinct fates rely on distinct populations of MVEs that coexist within the same cell. Localization of cholesterol with the toxin perfringolysin indicated one cholesterol-rich MVE population for exosome secretion and another, morphologically identical, but cholesterol-poor population for lysosomal targeting (Möbius et al., 2002). Conversely, lysobisphosphatidic acid is absent in exosomes (Wubbolts et al., 2003) but clearly present in lysosomally destined epidermal growth factor–containing MVEs (White et al., 2006). In dendritic cells, sorting of MHC II into exosomes is, in contrast to lysosomal targeting, independent of MHC II ubiquitination and rather correlates with incorporation into the tetraspanin CD9-containing detergent-resistant membranes (Buschow et al., 2009). The generation of MVEs involves the lateral segregation of cargo at the delimiting membrane of an endosome and inward budding and pinching of vesicles into the endosomal lumen (Fig. 2). The molecular machineries involved in the biogenesis of MVEs en route for degradation have been resolved based on the initial discovery of yeast mutants that were defective in the transport to the vacuole, the yeast analogue of mammalian lysosomes. These evolutionarily conserved proteins assemble into four multiprotein complexes: endosomal sorting complex responsible for transport (ESCRT)-0, -I, -II, and -III, which associate with accessory proteins (e.g., Alix and VPS4). The ESCRT-0, -I, and -II complexes recognize and sequester ubiquitinated membrane proteins at the endosomal delimiting membrane, whereas the ESCRT-III complex is responsible for membrane budding and actual scission of ILVs (Raiborg and Stenmark, 2009; Hurley, 2010). The discovery of the machinery involved in MVE biogenesis gave rise to the speculation on its potential role in exosome formation. As we discuss next, however, the function of ESCRT components in the formation of secretory MVEs appears to be more complex than originally supposed.
ESCRT-independent and -dependent mechanisms.
In oligodendroglial cell lines, which secrete the proteolipid protein in association with exosomes, exosome biogenesis and secretion do not require ESCRT function but are dependent on sphingomyelinase, an enzyme that produces ceramide (Trajkovic et al., 2008). These observations are consistent with the presence of high concentrations of ceramide and derivatives thereof in exosomes (Wubbolts et al., 2003; Trajkovic et al., 2008; Brouwers et al., 2012). The existence of ESCRT-independent mechanisms for MVE formation is supported by the finding that cells concomitantly depleted of four subunits of the ESCRT complex are still able to generate CD63-positive MVEs (Stuffers et al., 2009). Recruitment of MHC II to exosomes from antigen-presenting cells occurs independently of MHC II ubiquitination, again consistent with sorting mechanisms that may operate independently of the ESCRT machinery (Buschow et al., 2009). Our own studies in pigment-producing melanocytes indicate that mammalian cells developed pathways for MVE formation independently of both ESCRTs (Theos et al., 2006) and ceramide (van Niel et al., 2011). Tetraspanins, which are highly enriched in MVEs, have often been proposed to play a role in the formation of ILVs and the exosome (Simons and Raposo, 2009). MHC class II molecules in exosomes are associated with large protein complexes also containing tetraspanins (Wubbolts et al., 2003; Buschow et al., 2009). In another cell system, we have shown that CD63 functions in ESCRT-independent sorting to ILVs of the melanosomal protein PMEL (van Niel et al., 2011), a protein that is targeted to exosomes in melanoma cells (Wolfers et al., 2001).
These aforementioned studies indicate that the ESCRT system may have distinct functions in EV production versus lysosomal protein sorting. Even though EV cargo proteins may not be selected through ubiquitination, some ESCRT components have been implicated in EV formation. For example, the transferrin receptor, which in reticulocytes is fated for exosome secretion, interacts with the ESCRT accessory protein Alix during its sorting at MVEs (Géminard et al., 2004). More recently, Alix was also shown to be involved in exosome biogenesis and exosomal sorting of syndecans through an interaction with syntenin (Baietti et al., 2012). Our unpublished data exploiting a medium throughput interference (RNAi) screen targeting 23 different components of ESCRT-0/I/II/III and associated proteins in HeLa CIITA cells expressing MHC class II indicate a role for only a few members of this family (STAM [signal-transducing adaptor molecule], Tsg101, Alix, Hrs, and VPS4; Colombo, M., and C. Théry, personal communication). The ESCRT-0 component Hrs (Hepatocyte growth factor–associated tyrosine kinase) has been reported to be involved in exosome formation/secretion in dendritic cells (Tamai et al., 2010). The tumor suppressor protein p53 and its transcriptional target TSAP6 have been implicated in the regulation of exosome secretion (Yu et al., 2006), illustrating potential couplings between signaling and exosome biogenesis (Hupalowska and Miaczynska, 2012). Moreover, p53 activity has been linked to the ESCRT-III component Chmp1A (Manohar et al., 2011), further explaining a role for p53 in MVE and maybe exosome biogenesis.
How cytosolic constituents are recruited into exosomes is unclear but may involve association of exosomal membrane proteins with chaperones such as Hsc70, that are found in exosomes from most cell types (Théry et al., 2001; Géminard et al., 2004). Using quantitative mass spectrometry, we identified a small subset of cytosolic proteins and proteins that, together with tetraspanins, coimmunoprecipitated with MHC II from lysed exosomes. These included Hsc70, Hsp90, 14–3-3 epsilon, and PKM2, all of which could potentially play a role in protein sorting to exosomes (Buschow et al., 2010). Hsc70 was also shown to interact with the transferrin receptor in maturing reticulocytes but not in other cell types (Géminard et al., 2004). Given the unfolding but still incomplete picture of both ESCRT-independent and -dependent aspects in the biogenesis of exosomes, the mechanism is likely to be complex. Similarly, the mechanism for generation of MVs from the plasma membrane is largely undefined. In principle, oligomerization of a cytoplasmic protein in addition to any plasma membrane anchor, including myristoylation and palmitoylation, appears sufficient to drive proteins into MVs (Shen et al., 2011). MV formation in breast cancer cells requires the actin–myosin machinery and the action of small GTPases, such as ARF6 (Muralidharan-Chari et al., 2009, 2010). Interestingly, a recent study provided evidence for the recruitment of the ESCRT-I subunit Tsg101 to the plasma membrane by means of a tetrapeptide PSAP motif that drives the ARRDC1 (Arrestin 1 domain–containing protein 1) into MVs (Nabhan et al., 2012). Thus, the molecular machineries for exosome and MV biogenesis may share mechanistic elements.
How RNA species are sorted into EVs is also far from being resolved. Recent observations suggest that RNAs in EVs share specific sequence motifs that may potentially function as cis-acting elements for targeting to EVs (Batagov et al., 2011). Although speculative, the finding that ESCRT-II is an RNA binding complex (Irion and St Johnston, 2007) opens the possibility that it may also function to select RNA for incorporation into EVs. Moreover, the observations that MVEs are sites of miRNA-loaded RISC (RNA-induced silencing complex) accumulation (Gibbings et al., 2009) and that exosome-like vesicles are considerably enriched in GW182 and AGO2 implicate functional roles of these proteins in RNA sorting to exosomes.
Mechanisms involved in the release of EVs
The machineries involved in scission/release of MVs from the plasma membrane and those implicated in the mobilization of secretory MVEs to the cell periphery, their docking, and fusion with the cell surface are still at an early stage of comprehension. These processes require the cytoskeleton (actin and microtubules), associated molecular motors (kinesins and myosins), molecular switches (small GTPases), and the fusion machinery (SNAREs and tethering factors; Cai et al., 2007). The first indications for the involvement of Rab GTPases in exosome secretion were from studies on reticulocyte cell lines, which required the function of Rab 11 for exosome secretion (Savina et al., 2002). More recently, in an RNAi screen in HeLa cells targeting 59 members of the Rab GTPase family, knockdown of Rab27a or Rab27b significantly reduced the amount of secreted exosomes (Ostrowski et al., 2010). Rab27 is associated with secretory lysosome–related organelles (Raposo et al., 2007), and these findings thus also directly strengthen a role for endocytic compartments in exosome secretion. By analogy with other cell systems hosting secretory endo/lysosomes, Rab27 could be involved directly or indirectly in the transport and tethering at the cell periphery of the secretory MVEs. Along this line, silencing of two known Rab27 effectors, Slp4 (also known as SYTL4 [synaptotagmin-like 4]) and Slac2b (also known as EXPH5 [exophilin 5]), inhibited exosome secretion and phenocopied silencing of Rab27a and Rab27b, respectively (Ostrowski et al., 2010). In a separate screen, targeting Rab GTPase-activating proteins, knockdown of the Rab GTPase-activating proteins TBC1D10A–C and interference with its effector, Rab35, reduced exosome secretion (Hsu et al., 2010). It should be noted that although Rab11, Rab27, and Rab35 all appear to be involved in exosome release, selective inactivation of each of these Rabs only partially impacted this pathway. The roles of these GTPases could be either complementary, cell type dependent, or only indirect by regulating pathways upstream of exosome secretion.
Release of EVs was found to be regulated in several cellular model systems. For example, MV release can be stimulated through activation of purinergic receptors with ATP (Wilson et al., 2004). Platelets are stimulated to shed vesicles from the plasma membrane and to release exosomes in response to thrombin receptor activation (Heijnen et al., 1998). Dendritic cells increase the release of MVs and change the protein composition thereof in response to activation by lipopolysaccharides (Obregon et al., 2006; Nolte-’t Hoen et al., 2012c), whereas peptide-loaded immature dendritic cells were stimulated to release exosomes in response to their interaction with T cells recognizing peptide-loaded MHC class II (Buschow et al., 2009). Similarly, plasma membrane depolarization increases the rapid secretion of exosomes by neuronal cells (Fauré et al., 2006; Lachenal et al., 2011), and cross-linking of CD3 in T cells stimulates exosome release by T cells (Blanchard et al., 2002). One central trigger for the release of EVs appears to involve increasing intracellular Ca2+ concentrations, as demonstrated, for example, for a human erythroleukemia cell line (Savina et al., 2005) and mast cells (Raposo et al., 1997).
Little is known about the machinery that drives MVE fusion with the plasma membrane. The SNARE complex involved in Ca2+-regulated exocytosis of conventional lysosomes includes VAMP7 and Ca2+ binding synaptotagmin VII (Rao et al., 2004). Whether the exocytic fusion of MVEs is similarly modulated and/or controlled by the same fusion machinery is debated: Exosome secretion by maturing reticulocytes appeared to rely on VAMP7 function (Fader et al., 2009), whereas in MDCK cells, expression of the Longin domain of VAMP7 selectively impaired lysosomal secretion but not the release of exosomes (Proux-Gillardeaux et al., 2007). In a recent study, it was demonstrated that secretion of exosomes carrying the morphogen Wnt is dependent on the R-SNARE Ykt6 (Gross et al., 2012).
The V0 subunit of the vacuolar V-ATPase, which is involved in fusion events independently of its proton pump activity, may, through its association with SNAREs, form fusion pores (Marshansky and Futai, 2008). The V0-ATPase has been proposed to regulate MVE secretion in Caenorhabditis elegans (Liégeois et al., 2006), but these findings await validation in mammalian cells.
Interactions of EVs with recipient cells
Functions of EVs in physiological and pathological processes depend on the ability of EVs to interact with recipient cells to deliver their contents of proteins, lipids, and RNAs (Fig. 3). Specificity of target cell binding is illustrated by the finding that isolated B cell exosomes selectively bind follicular dendritic cells in lymphoid follicles (Denzer et al., 2000). Similarly, EVs released by a human intestinal epithelial cell line interacted preferentially with dendritic cells rather than with B or T lymphocytes (Mallegol et al., 2007). The cellular and molecular basis for EV targeting is still undetermined, but several target cell–dependent and –conditional aspects are beginning to emerge. Target cell specificity for binding of exosomes (or other EVs) is likely to be determined by adhesion molecules, such as integrins, that are present in EVs. For example, T cells can recruit MHC class II–containing dendritic cell–derived exosomes that are secreted in response to cognate dendritic cell–T cell interactions (Buschow et al., 2009). Recruitment of these exosomes required T cell activation and was dependent on an induced high-affinity state of LFA-1 (leukocyte function-associated antigen-1) rather than on T cell receptor specificity (Nolte-’t Hoen et al., 2009). Exosomes carrying MHC class II and ICAM-1 from mature dendritic cells can also be recruited by bystander dendritic cells with help of LFA-1 (Segura et al., 2007). Differences in exosomal tetraspanin complexes also appear to influence target cell selection in vitro and in vivo (Rana et al., 2012), possibly by modulating the functions of associated proteins, including adhesion molecules such as integrins (Hemler, 2003). Yet other molecules, such as galactin-5 and galectin-9, are involved in the clearance of reticulocyte exosomes by macrophages (Barrès et al., 2010) and in the targeting of nasopharyngeal carcinoma–derived EVs to CD4+ T cells (Klibi et al., 2009), respectively.
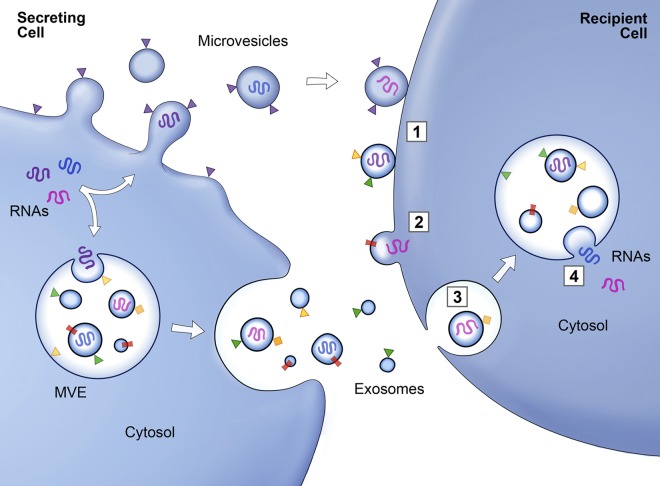
Figure 3.
Schematic of protein and RNA transfer by EVs. Membrane-associated (triangles) and transmembrane proteins (rectangles) and RNAs (curved symbols) are selectively incorporated into the ILV of MVEs or into MVs budding from the plasma membrane. MVEs fuse with the plasma membrane to release exosomes into the extracellular milieu. MVs and exosomes may dock at the plasma membrane of a target cell (1). Bound vesicles may either fuse directly with the plasma membrane (2) or be endocytosed (3). Endocytosed vesicles may then fuse with the delimiting membrane of an endocytic compartment (4). Both pathways result in the delivery of proteins and RNA into the membrane or cytosol of the target cell. Fusion and endocytosis are only represented for exosomal vesicles, but plasma membrane–derived MVs may have similar fates.
After binding to recipient cells, EVs may remain stably associated with the plasma membrane or dissociate, directly fuse with the plasma membrane, or be internalized through distinct endocytic pathways (Fig. 3). When endocytosed, EVs may subsequently fuse with the endosomal delimiting membrane or be targeted to lysosomes for degradation. Stable and persistent cell surface exposure can be expected, particularly on cells that display little if any endocytic activity, as was proposed for MHC class II–carrying exosomes associated with follicular dendritic cells that do not synthesize MHC class II themselves but function in the maintenance of T cell memory (Denzer et al., 2000). Detection of fusion of small EVs with the plasma membrane by fluorescence microscopy in live cells is limited by resolution and the fast dynamics of fusion events. Nevertheless, direct evidence for fusion of exosomes with target cell membranes has been obtained by labeling exosomes with the lipophilic dye R18, in which self-quenching is relieved upon dilution as a consequence of fusion (Montecalvo et al., 2012), resulting in flashing and an increase in the fluorescence of target cells. Several other studies provided evidence for the accumulation of captured EVs in endocytic or phagocytic compartments, with uptake depending on the actin cytoskeleton, phosphatidylinositol 3-kinase activity, and dynamin-2 function (Morelli, 2006; Barrès et al., 2010; Tian et al., 2010).
Functions of EVs
To our knowledge, one the first studies reporting the functional interaction of EVs with cells is the promotion of sperm cell motility by prostasomes (Stegmayr and Ronquist, 1982). Over the past years, very diverse biological functions have been attributed to EVs (also summarized by Harding et al. in this issue), and it is now commonly accepted that exosomes and MVs represent important vehicles of intercellular communication in between cells locally or at a distance.
In the early 80’s, exosome secretion by reticulocytes was reported as a mechanism to eradicate obsolete molecules (Harding et al., 2013). Later, the capacity of exosomes to act as antigen-presenting vesicles, to stimulate antitumoral immune responses, or rather to induce tolerogenic effects has stimulated the interest of immunologists to investigate their potential use in clinics (Bobrie et al., 2011; Chaput and Théry, 2011). Tumor cells as well as other cells in tumor microenvironments also secrete EVs (exosomes and microvesicles), and there is evidence that these contribute to tumor progression by promoting angiogenesis and tumor cell migration in metastases (Rak, 2010; Hood et al., 2011). Tumor-derived vesicles also bear immunosuppressive molecules, which can inactivate T lymphocytes or natural killer cells, or promote the differentiation of regulatory T lymphocytes or myeloid cells to suppress immune responses (Zhang and Grizzle, 2011).
Functions of EVs have also been reported in epithelia and in the nervous system. EVs released apically or basolaterally by intestinal epithelial cells appear to be involved in antigen presentation at inflammatory conditions, and these EVs may confer the ability of static epithelial cells to act at a distance (van Niel et al., 2001). In the airways, EVs present in the bronchoalveolar fluid bear tolerizing molecules (e.g., in allergen-tolerized mice) or, conversely, may increase proinflammatory cytokine secretion by airway epithelial cells in asthmatic human patients (Prado et al., 2008; Qazi et al., 2010).
Recent studies reported the association of membrane-bound morphogens to EVs, including Wnt (Gross et al., 2012; Luga et al., 2012; Beckett et al., 2013), and the Notch ligand DII4 (Sheldon et al., 2010). Through Wnt signaling, fibroblast exosomes have recently been demonstrated to promote breast cancer cell dynamics (Luga et al., 2012). Also, in Drosophila melanogaster, Wnt-associated EVs have been implicated in signal transduction, although here, exosomes do not appear to be essential for Wnt gradient formation in tissues (Beckett et al., 2013). In addition to exosomes or MVs, small lipoprotein particles may also contribute to the secretion and be responsible for tissue gradient formation of Wnt and Hedgehog (Panáková et al., 2005; Eaton, 2006; Neumann et al., 2009).
Within the nervous system, neurons, oligodendroglial cells, and microglia secrete EVs that could be targeted from one cell type to the other (Fauré et al., 2006; Krämer-Albers et al., 2007; Lachenal et al., 2011). EVs have recently been proposed to participate in myelin formation (Bakhti et al., 2011) as well as in neurite outgrowth and neuronal survival (Wang et al., 2011). Within the central nervous system, several pathogenic proteins that are involved in central nervous system diseases, such as prions (Fevrier et al., 2004), β-amyloid peptide (Rajendran et al., 2006), superoxide dismutase (Gomes et al., 2007), and α-synuclein (Emmanouilidou et al., 2010), are released from cells in association with EVs. These secreted vesicles are thought to participate in disseminating pathogenesis through interaction with recipient cells. Ultrastructural observations in situ in the gut of prion-infected mice showed the presence of A33 antigen-positive EVs (Kujala et al., 2011), supporting their existence in vivo. Interestingly, α-synuclein can be detected in the plasma and cerebrospinal fluid of humans, extending the interest for EVs as biomarkers in disease (Alvarez-Llamas et al., 2008; Al-Nedawi et al., 2009; Simpson et al., 2009).
The interest of scientists and physicians in EVs has expanded logarithmically over the past decade in response to the discoveries that EVs are not only generated in cell culture but are also abundantly present in body fluids, carry RNA, and show a wide range of regulatory functions. As discussed, we are still at an early stage of deciphering the molecular mechanisms involved in EV biogenesis and recruitment of cargo therein. Specific knowledge of these mechanisms will help us to intervene with EV function in vivo, an absolute requirement to decipher their precise role in physiological processes. Also, more accurate and standardized purification methods are required for the implementation in a clinical setting of EVs as biomarkers, vaccines, or drug delivery devices. To help coordinate these enormous challenges, the International Society for Extracellular Vesicles was launched in 2011.
Acknowledgments
We thank Guillaume van Niel, Alessandra Lo Cicero, Clotilde Théry, Richard Wubbolts, Marca Wauben, and Esther Nolte-’t Hoen for many stimulating discussions. We are grateful to our laboratory members for their continuous support. We are grateful to Phil Stahl for reading the manuscript. We will always keep in memory Rose Johnstone, her contributions, and enthusiasm, which have been stimulating for all of us. We apologize to all colleagues whose work could not be cited as a result of space limitations.
We thank Institut Curie, Centre National de la Recherche Scientifique, Agence Nationale pour la Recherche (Programme Blanc and Maladies infectieuses, immunité et environnement), Association pour la Recherche Contre le Cancer, Clarins, and Utrecht University for support and funding.
Footnotes
Abbreviations used in this paper:
- ESCRT
- endosomal sorting complex responsible for transport
- EV
- extracellular vesicle
- ILV
- intraluminal vesicle
- MV
- microvesicle
- MVE
- multivesicular endosome
References
- Aalberts M., van Dissel-Emiliani F.M., van Adrichem N.P., van Wijnen M., Wauben M.H., Stout T.A., Stoorvogel W. 2012. Identification of distinct populations of prostasomes that differentially express prostate stem cell antigen, annexin A1, and GLIPR2 in humans. Biol. Reprod. 86:82 10.1095/biolreprod.111.095760 [DOI] [PubMed] [Google Scholar]
- Admyre C., Johansson S.M., Qazi K.R., Filén J.J., Lahesmaa R., Norman M., Neve E.P., Scheynius A., Gabrielsson S. 2007. Exosomes with immune modulatory features are present in human breast milk. J. Immunol. 179:1969–1978 [DOI] [PubMed] [Google Scholar]
- Al-Nedawi K., Meehan B., Rak J. 2009. Microvesicles: messengers and mediators of tumor progression. Cell Cycle. 8:2014–2018 10.4161/cc.8.13.8988 [DOI] [PubMed] [Google Scholar]
- Alvarez-Llamas G., de la Cuesta F., Barderas M.E., Darde V., Padial L.R., Vivanco F. 2008. Recent advances in atherosclerosis-based proteomics: new biomarkers and a future perspective. Expert Rev. Proteomics. 5:679–691 10.1586/14789450.5.5.679 [DOI] [PubMed] [Google Scholar]
- Andre F., Schartz N.E., Movassagh M., Flament C., Pautier P., Morice P., Pomel C., Lhomme C., Escudier B., Le Chevalier T., et al. 2002. Malignant effusions and immunogenic tumour-derived exosomes. Lancet. 360:295–305 10.1016/S0140-6736(02)09552-1 [DOI] [PubMed] [Google Scholar]
- Asea A., Jean-Pierre C., Kaur P., Rao P., Linhares I.M., Skupski D., Witkin S.S. 2008. Heat shock protein-containing exosomes in mid-trimester amniotic fluids. J. Reprod. Immunol. 79:12–17 10.1016/j.jri.2008.06.001 [DOI] [PubMed] [Google Scholar]
- Baietti M.F., Zhang Z., Mortier E., Melchior A., Degeest G., Geeraerts A., Ivarsson Y., Depoortere F., Coomans C., Vermeiren E., et al. 2012. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat. Cell Biol. 14:677–685 10.1038/ncb2502 [DOI] [PubMed] [Google Scholar]
- Bakhti M., Winter C., Simons M. 2011. Inhibition of myelin membrane sheath formation by oligodendrocyte-derived exosome-like vesicles. J. Biol. Chem. 286:787–796 10.1074/jbc.M110.190009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrès C., Blanc L., Bette-Bobillo P., André S., Mamoun R., Gabius H.J., Vidal M. 2010. Galectin-5 is bound onto the surface of rat reticulocyte exosomes and modulates vesicle uptake by macrophages. Blood. 115:696–705 10.1182/blood-2009-07-231449 [DOI] [PubMed] [Google Scholar]
- Batagov A.O., Kuznetsov V.A., Kurochkin I.V. 2011. Identification of nucleotide patterns enriched in secreted RNAs as putative cis-acting elements targeting them to exosome nano-vesicles. BMC Genomics. 12(Suppl. 3):S18 10.1186/1471-2164-12-S3-S18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beckett K., Monier S., Palmer L., Alexandre C., Green H., Bonneil E., Raposo G., Thibault P., Borgne R.L., Vincent J.P. 2013. Drosophila s2 cells secrete wingless on exosome-like vesicles but the wingless gradient forms independently of exosomes. Traffic. 14:82–96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellingham S.A., Coleman B.M., Hill A.F. 2012. Small RNA deep sequencing reveals a distinct miRNA signature released in exosomes from prion-infected neuronal cells. Nucleic Acids Res. 40:10937–10949 10.1093/nar/gks832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanchard N., Lankar D., Faure F., Regnault A., Dumont C., Raposo G., Hivroz C. 2002. TCR activation of human T cells induces the production of exosomes bearing the TCR/CD3/zeta complex. J. Immunol. 168:3235–3241 [DOI] [PubMed] [Google Scholar]
- Bobrie A., Colombo M., Raposo G., Théry C. 2011. Exosome secretion: molecular mechanisms and roles in immune responses. Traffic. 12:1659–1668 10.1111/j.1600-0854.2011.01225.x [DOI] [PubMed] [Google Scholar]
- Booth A.M., Fang Y., Fallon J.K., Yang J.M., Hildreth J.E., Gould S.J. 2006. Exosomes and HIV Gag bud from endosome-like domains of the T cell plasma membrane. J. Cell Biol. 172:923–935 10.1083/jcb.200508014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brouwers J.F., Aalberts M., Jansen J.W.A., van Niel G., Stout T.A.E., Helms J.B., Stoorvogel W. 2012. Distinct lipid compositions of two types of human prostasomes. Proteomics. In press [DOI] [PubMed] [Google Scholar]
- Buschow S.I., Nolte-’t Hoen E.N., van Niel G., Pols M.S., ten Broeke T., Lauwen M., Ossendorp F., Melief C.J., Raposo G., Wubbolts R., et al. 2009. MHC II in dendritic cells is targeted to lysosomes or T cell-induced exosomes via distinct multivesicular body pathways. Traffic. 10:1528–1542 10.1111/j.1600-0854.2009.00963.x [DOI] [PubMed] [Google Scholar]
- Buschow S.I., van Balkom B.W., Aalberts M., Heck A.J., Wauben M., Stoorvogel W. 2010. MHC class II-associated proteins in B-cell exosomes and potential functional implications for exosome biogenesis. Immunol. Cell Biol. 88:851–856 10.1038/icb.2010.64 [DOI] [PubMed] [Google Scholar]
- Caby M.P., Lankar D., Vincendeau-Scherrer C., Raposo G., Bonnerot C. 2005. Exosomal-like vesicles are present in human blood plasma. Int. Immunol. 17:879–887 10.1093/intimm/dxh267 [DOI] [PubMed] [Google Scholar]
- Cai H., Reinisch K., Ferro-Novick S. 2007. Coats, tethers, Rabs, and SNAREs work together to mediate the intracellular destination of a transport vesicle. Dev. Cell. 12:671–682 10.1016/j.devcel.2007.04.005 [DOI] [PubMed] [Google Scholar]
- Chaput N., Théry C. 2011. Exosomes: immune properties and potential clinical implementations. Semin. Immunopathol. 33:419–440 10.1007/s00281-010-0233-9 [DOI] [PubMed] [Google Scholar]
- Cocucci E., Racchetti G., Meldolesi J. 2009. Shedding microvesicles: artefacts no more. Trends Cell Biol. 19:43–51 10.1016/j.tcb.2008.11.003 [DOI] [PubMed] [Google Scholar]
- Conde-Vancells J., Rodriguez-Suarez E., Embade N., Gil D., Matthiesen R., Valle M., Elortza F., Lu S.C., Mato J.M., Falcon-Perez J.M. 2008. Characterization and comprehensive proteome profiling of exosomes secreted by hepatocytes. J. Proteome Res. 7:5157–5166 10.1021/pr8004887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denzer K., van Eijk M., Kleijmeer M.J., Jakobson E., de Groot C., Geuze H.J. 2000. Follicular dendritic cells carry MHC class II-expressing microvesicles at their surface. J. Immunol. 165:1259–1265 [DOI] [PubMed] [Google Scholar]
- Deregibus M.C., Cantaluppi V., Calogero R., Lo Iacono M., Tetta C., Biancone L., Bruno S., Bussolati B., Camussi G. 2007. Endothelial progenitor cell derived microvesicles activate an angiogenic program in endothelial cells by a horizontal transfer of mRNA. Blood. 110:2440–2448 10.1182/blood-2007-03-078709 [DOI] [PubMed] [Google Scholar]
- Eaton S. 2006. Release and trafficking of lipid-linked morphogens. Curr. Opin. Genet. Dev. 16:17–22 10.1016/j.gde.2005.12.006 [DOI] [PubMed] [Google Scholar]
- Eldh M., Lötvall J., Malmhäll C., Ekström K. 2012. Importance of RNA isolation methods for analysis of exosomal RNA: evaluation of different methods. Mol. Immunol. 50:278–286 10.1016/j.molimm.2012.02.001 [DOI] [PubMed] [Google Scholar]
- Emmanouilidou E., Melachroinou K., Roumeliotis T., Garbis S.D., Ntzouni M., Margaritis L.H., Stefanis L., Vekrellis K. 2010. Cell-produced alpha-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J. Neurosci. 30:6838–6851 10.1523/JNEUROSCI.5699-09.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Escola J.M., Kleijmeer M.J., Stoorvogel W., Griffith J.M., Yoshie O., Geuze H.J. 1998. Selective enrichment of tetraspan proteins on the internal vesicles of multivesicular endosomes and on exosomes secreted by human B-lymphocytes. J. Biol. Chem. 273:20121–20127 10.1074/jbc.273.32.20121 [DOI] [PubMed] [Google Scholar]
- Fader C.M., Sánchez D.G., Mestre M.B., Colombo M.I. 2009. TI-VAMP/VAMP7 and VAMP3/cellubrevin: two v-SNARE proteins involved in specific steps of the autophagy/multivesicular body pathways. Biochim. Biophys. Acta. 1793:1901–1916 10.1016/j.bbamcr.2009.09.011 [DOI] [PubMed] [Google Scholar]
- Fauré J., Lachenal G., Court M., Hirrlinger J., Chatellard-Causse C., Blot B., Grange J., Schoehn G., Goldberg Y., Boyer V., et al. 2006. Exosomes are released by cultured cortical neurones. Mol. Cell. Neurosci. 31:642–648 10.1016/j.mcn.2005.12.003 [DOI] [PubMed] [Google Scholar]
- Fevrier B., Vilette D., Archer F., Loew D., Faigle W., Vidal M., Laude H., Raposo G. 2004. Cells release prions in association with exosomes. Proc. Natl. Acad. Sci. USA. 101:9683–9688 10.1073/pnas.0308413101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Géminard C., De Gassart A., Blanc L., Vidal M. 2004. Degradation of AP2 during reticulocyte maturation enhances binding of hsc70 and Alix to a common site on TFR for sorting into exosomes. Traffic. 5:181–193 10.1111/j.1600-0854.2004.0167.x [DOI] [PubMed] [Google Scholar]
- Gibbings D.J., Ciaudo C., Erhardt M., Voinnet O. 2009. Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity. Nat. Cell Biol. 11:1143–1149 10.1038/ncb1929 [DOI] [PubMed] [Google Scholar]
- Gomes C., Keller S., Altevogt P., Costa J. 2007. Evidence for secretion of Cu,Zn superoxide dismutase via exosomes from a cell model of amyotrophic lateral sclerosis. Neurosci. Lett. 428:43–46 10.1016/j.neulet.2007.09.024 [DOI] [PubMed] [Google Scholar]
- Gross J.C., Chaudhary V., Bartscherer K., Boutros M. 2012. Active Wnt proteins are secreted on exosomes. Nat. Cell Biol. 14:1036–1045 10.1038/ncb2574 [DOI] [PubMed] [Google Scholar]
- György B., Szabó T.G., Pásztói M., Pál Z., Misják P., Aradi B., László V., Pállinger E., Pap E., Kittel A., et al. 2011. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell. Mol. Life Sci. 68:2667–2688 10.1007/s00018-011-0689-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harding C., Heuser J., Stahl P. 1984. Endocytosis and intracellular processing of transferrin and colloidal gold-transferrin in rat reticulocytes: demonstration of a pathway for receptor shedding. Eur. J. Cell Biol. 35:256–263 [PubMed] [Google Scholar]
- Harding C.V., Heuser J.E., Stahl P.D. 2013. Exosomes: Looking back three decades and into the future. J. Cell Biol. 200:367–371 10.1083/jcb.201212113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heijnen H.F., Debili N., Vainchencker W., Breton-Gorius J., Geuze H.J., Sixma J.J. 1998. Multivesicular bodies are an intermediate stage in the formation of platelet alpha-granules. Blood. 91:2313–2325 [PubMed] [Google Scholar]
- Heijnen H.F., Schiel A.E., Fijnheer R., Geuze H.J., Sixma J.J. 1999. Activated platelets release two types of membrane vesicles: microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha-granules. Blood. 94:3791–3799 [PubMed] [Google Scholar]
- Hemler M.E. 2003. Tetraspanin proteins mediate cellular penetration, invasion, and fusion events and define a novel type of membrane microdomain. Annu. Rev. Cell Dev. Biol. 19:397–422 10.1146/annurev.cellbio.19.111301.153609 [DOI] [PubMed] [Google Scholar]
- Hemler M.E. 2008. Targeting of tetraspanin proteins—potential benefits and strategies. Nat. Rev. Drug Discov. 7:747–758 10.1038/nrd2659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hess C., Sadallah S., Hefti A., Landmann R., Schifferli J.A. 1999. Ectosomes released by human neutrophils are specialized functional units. J. Immunol. 163:4564–4573 [PubMed] [Google Scholar]
- Holme P.A., Solum N.O., Brosstad F., Røger M., Abdelnoor M. 1994. Demonstration of platelet-derived microvesicles in blood from patients with activated coagulation and fibrinolysis using a filtration technique and western blotting. Thromb. Haemost. 72:666–671 [PubMed] [Google Scholar]
- Hood J.L., San R.S., Wickline S.A. 2011. Exosomes released by melanoma cells prepare sentinel lymph nodes for tumor metastasis. Cancer Res. 71:3792–3801 10.1158/0008-5472.CAN-10-4455 [DOI] [PubMed] [Google Scholar]
- Hristov M., Erl W., Linder S., Weber P.C. 2004. Apoptotic bodies from endothelial cells enhance the number and initiate the differentiation of human endothelial progenitor cells in vitro. Blood. 104:2761–2766 10.1182/blood-2003-10-3614 [DOI] [PubMed] [Google Scholar]
- Hsu C., Morohashi Y., Yoshimura S., Manrique-Hoyos N., Jung S., Lauterbach M.A., Bakhti M., Grønborg M., Möbius W., Rhee J., et al. 2010. Regulation of exosome secretion by Rab35 and its GTPase-activating proteins TBC1D10A–C. J. Cell Biol. 189:223–232 10.1083/jcb.200911018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter M.P., Ismail N., Zhang X., Aguda B.D., Lee E.J., Yu L., Xiao T., Schafer J., Lee M.L., Schmittgen T.D., et al. 2008. Detection of microRNA expression in human peripheral blood microvesicles. PLoS ONE. 3:e3694 10.1371/journal.pone.0003694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hupalowska A., Miaczynska M. 2012. The new faces of endocytosis in signaling. Traffic. 13:9–18 10.1111/j.1600-0854.2011.01249.x [DOI] [PubMed] [Google Scholar]
- Hurley J.H. 2010. The ESCRT complexes. Crit. Rev. Biochem. Mol. Biol. 45:463–487 10.3109/10409238.2010.502516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irion U., St Johnston D. 2007. bicoid RNA localization requires specific binding of an endosomal sorting complex. Nature. 445:554–558 10.1038/nature05503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalra H., Simpson R.J., Ji H., Aikawa E., Altevogt P., Askenase P., Bond V.C., Borràs F.E., Breakefield X., Budnik V., et al. 2012. Vesiclepedia: a compendium for extracellular vesicles with continuous community annotation. PLoS Biol. 10:e1001450 10.1371/journal.pbio.1001450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klibi J., Niki T., Riedel A., Pioche-Durieu C., Souquere S., Rubinstein E., Le Moulec S., Guigay J., Hirashima M., Guemira F., et al. 2009. Blood diffusion and Th1-suppressive effects of galectin-9-containing exosomes released by Epstein-Barr virus-infected nasopharyngeal carcinoma cells. Blood. 113:1957–1966 10.1182/blood-2008-02-142596 [DOI] [PubMed] [Google Scholar]
- Krämer-Albers E.M., Bretz N., Tenzer S., Winterstein C., Möbius W., Berger H., Nave K.A., Schild H., Trotter J. 2007. Oligodendrocytes secrete exosomes containing major myelin and stress-protective proteins: Trophic support for axons? Proteomics Clin. Appl. 1:1446–1461 10.1002/prca.200700522 [DOI] [PubMed] [Google Scholar]
- Kujala P., Raymond C.R., Romeijn M., Godsave S.F., van Kasteren S.I., Wille H., Prusiner S.B., Mabbott N.A., Peters P.J. 2011. Prion uptake in the gut: identification of the first uptake and replication sites. PLoS Pathog. 7:e1002449 10.1371/journal.ppat.1002449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lachenal G., Pernet-Gallay K., Chivet M., Hemming F.J., Belly A., Bodon G., Blot B., Haase G., Goldberg Y., Sadoul R. 2011. Release of exosomes from differentiated neurons and its regulation by synaptic glutamatergic activity. Mol. Cell. Neurosci. 46:409–418 10.1016/j.mcn.2010.11.004 [DOI] [PubMed] [Google Scholar]
- Laulagnier K., Motta C., Hamdi S., Roy S., Fauvelle F., Pageaux J.F., Kobayashi T., Salles J.P., Perret B., Bonnerot C., Record M. 2004. Mast cell- and dendritic cell-derived exosomes display a specific lipid composition and an unusual membrane organization. Biochem. J. 380:161–171 10.1042/BJ20031594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liégeois S., Benedetto A., Garnier J.M., Schwab Y., Labouesse M. 2006. The V0-ATPase mediates apical secretion of exosomes containing Hedgehog-related proteins in Caenorhabditis elegans. J. Cell Biol. 173:949–961 10.1083/jcb.200511072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luga V., Zhang L., Viloria-Petit A.M., Ogunjimi A.A., Inanlou M.R., Chiu E., Buchanan M., Hosein A.N., Basik M., Wrana J.L. 2012. Exosomes mediate stromal mobilization of autocrine Wnt-PCP signaling in breast cancer cell migration. Cell. 151:1542–1556 10.1016/j.cell.2012.11.024 [DOI] [PubMed] [Google Scholar]
- Mallegol J., Van Niel G., Lebreton C., Lepelletier Y., Candalh C., Dugave C., Heath J.K., Raposo G., Cerf-Bensussan N., Heyman M. 2007. T84-intestinal epithelial exosomes bear MHC class II/peptide complexes potentiating antigen presentation by dendritic cells. Gastroenterology. 132:1866–1876 10.1053/j.gastro.2007.02.043 [DOI] [PubMed] [Google Scholar]
- Manohar S., Harlow M., Nguyen H., Li J., Hankins G.R., Park M. 2011. Chromatin modifying protein 1A (Chmp1A) of the endosomal sorting complex required for transport (ESCRT)-III family activates ataxia telangiectasia mutated (ATM) for PanC-1 cell growth inhibition. Cell Cycle. 10:2529–2539 10.4161/cc.10.15.15926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marshansky V., Futai M. 2008. The V-type H+-ATPase in vesicular trafficking: targeting, regulation and function. Curr. Opin. Cell Biol. 20:415–426 10.1016/j.ceb.2008.03.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masyuk A.I., Huang B.Q., Ward C.J., Gradilone S.A., Banales J.M., Masyuk T.V., Radtke B., Splinter P.L., LaRusso N.F. 2010. Biliary exosomes influence cholangiocyte regulatory mechanisms and proliferation through interaction with primary cilia. Am. J. Physiol. Gastrointest. Liver Physiol. 299:G990–G999 10.1152/ajpgi.00093.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michael A., Bajracharya S.D., Yuen P.S., Zhou H., Star R.A., Illei G.G., Alevizos I. 2010. Exosomes from human saliva as a source of microRNA biomarkers. Oral Dis. 16:34–38 10.1111/j.1601-0825.2009.01604.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittelbrunn M., Gutiérrez-Vázquez C., Villarroya-Beltri C., González S., Sánchez-Cabo F., González M.A., Bernad A., Sánchez-Madrid F. 2011. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2:282 10.1038/ncomms1285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Möbius W., Ohno-Iwashita Y., van Donselaar E.G., Oorschot V.M., Shimada Y., Fujimoto T., Heijnen H.F., Geuze H.J., Slot J.W. 2002. Immunoelectron microscopic localization of cholesterol using biotinylated and non-cytolytic perfringolysin O. J. Histochem. Cytochem. 50:43–55 10.1177/002215540205000105 [DOI] [PubMed] [Google Scholar]
- Montecalvo A., Larregina A.T., Shufesky W.J., Stolz D.B., Sullivan M.L., Karlsson J.M., Baty C.J., Gibson G.A., Erdos G., Wang Z., et al. 2012. Mechanism of transfer of functional microRNAs between mouse dendritic cells via exosomes. Blood. 119:756–766 10.1182/blood-2011-02-338004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morelli A.E. 2006. The immune regulatory effect of apoptotic cells and exosomes on dendritic cells: its impact on transplantation. Am. J. Transplant. 6:254–261 10.1111/j.1600-6143.2005.01197.x [DOI] [PubMed] [Google Scholar]
- Muralidharan-Chari V., Clancy J., Plou C., Romao M., Chavrier P., Raposo G., D’Souza-Schorey C. 2009. ARF6-regulated shedding of tumor cell-derived plasma membrane microvesicles. Curr. Biol. 19:1875–1885 10.1016/j.cub.2009.09.059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muralidharan-Chari V., Clancy J.W., Sedgwick A., D’Souza-Schorey C. 2010. Microvesicles: mediators of extracellular communication during cancer progression. J. Cell Sci. 123:1603–1611 10.1242/jcs.064386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nabhan J.F., Hu R., Oh R.S., Cohen S.N., Lu Q. 2012. Formation and release of arrestin domain-containing protein 1-mediated microvesicles (ARMMs) at plasma membrane by recruitment of TSG101 protein. Proc. Natl. Acad. Sci. USA. 109:4146–4151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neumann S., Coudreuse D.Y., van der Westhuyzen D.R., Eckhardt E.R., Korswagen H.C., Schmitz G., Sprong H. 2009. Mammalian Wnt3a is released on lipoprotein particles. Traffic. 10:334–343 10.1111/j.1600-0854.2008.00872.x [DOI] [PubMed] [Google Scholar]
- Nolte-’t Hoen E.N., Buschow S.I., Anderton S.M., Stoorvogel W., Wauben M.H. 2009. Activated T cells recruit exosomes secreted by dendritic cells via LFA-1. Blood. 113:1977–1981 10.1182/blood-2008-08-174094 [DOI] [PubMed] [Google Scholar]
- Nolte-’t Hoen E.N., Buermans H.P., Waasdorp M., Stoorvogel W., Wauben M.H., ’t Hoen P.A. 2012a. Deep sequencing of RNA from immune cell-derived vesicles uncovers the selective incorporation of small non-coding RNA biotypes with potential regulatory functions. Nucleic Acids Res. 40:9272–9285 10.1093/nar/gks658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolte-’t Hoen E.N., van der Vlist E.J., Aalberts M., Mertens H.C., Bosch B.J., Bartelink W., Mastrobattista E., van Gaal E.V., Stoorvogel W., Arkesteijn G.J., Wauben M.H. 2012b. Quantitative and qualitative flow cytometric analysis of nanosized cell-derived membrane vesicles. Nanomedicine. 8:712–720 10.1016/j.nano.2011.09.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolte-’t Hoen E.N., van der Vlist E.J., de Boer-Brouwer M., Arkesteijn G.J., Stoorvogel W., Wauben M.H. 2012c. Dynamics of dendritic cell-derived vesicles: high-resolution flow cytometric analysis of extracellular vesicle quantity and quality. J. Leukoc. Biol. 10.1189/jlb.0911480 [DOI] [PubMed] [Google Scholar]
- Obregon C., Rothen-Rutishauser B., Gitahi S.K., Gehr P., Nicod L.P. 2006. Exovesicles from human activated dendritic cells fuse with resting dendritic cells, allowing them to present alloantigens. Am. J. Pathol. 169:2127–2136 10.2353/ajpath.2006.060453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa Y., Miura Y., Harazono A., Kanai-Azuma M., Akimoto Y., Kawakami H., Yamaguchi T., Toda T., Endo T., Tsubuki M., Yanoshita R. 2011. Proteomic analysis of two types of exosomes in human whole saliva. Biol. Pharm. Bull. 34:13–23 10.1248/bpb.34.13 [DOI] [PubMed] [Google Scholar]
- Ostrowski M., Carmo N.B., Krumeich S., Fanget I., Raposo G., Savina A., Moita C.F., Schauer K., Hume A.N., Freitas R.P., et al. 2010. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat. Cell Biol. 12:19–30 10.1038/ncb2000 [DOI] [PubMed] [Google Scholar]
- Pan B.T., Teng K., Wu C., Adam M., Johnstone R.M. 1985. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J. Cell Biol. 101:942–948 10.1083/jcb.101.3.942 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panáková D., Sprong H., Marois E., Thiele C., Eaton S. 2005. Lipoprotein particles are required for Hedgehog and Wingless signalling. Nature. 435:58–65 10.1038/nature03504 [DOI] [PubMed] [Google Scholar]
- Park K.H., Kim B.J., Kang J., Nam T.S., Lim J.M., Kim H.T., Park J.K., Kim Y.G., Chae S.W., Kim U.H. 2011. Ca2+ signaling tools acquired from prostasomes are required for progesterone-induced sperm motility. Sci. Signal. 4:ra31 10.1126/scisignal.2001595 [DOI] [PubMed] [Google Scholar]
- Pisitkun T., Shen R.F., Knepper M.A. 2004. Identification and proteomic profiling of exosomes in human urine. Proc. Natl. Acad. Sci. USA. 101:13368–13373 10.1073/pnas.0403453101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prado N., Marazuela E.G., Segura E., Fernández-García H., Villalba M., Théry C., Rodríguez R., Batanero E. 2008. Exosomes from bronchoalveolar fluid of tolerized mice prevent allergic reaction. J. Immunol. 181:1519–1525 [DOI] [PubMed] [Google Scholar]
- Proux-Gillardeaux V., Raposo G., Irinopoulou T., Galli T. 2007. Expression of the Longin domain of TI-VAMP impairs lysosomal secretion and epithelial cell migration. Biol. Cell. 99:261–271 10.1042/BC20060097 [DOI] [PubMed] [Google Scholar]
- Qazi K.R., Torregrosa Paredes P., Dahlberg B., Grunewald J., Eklund A., Gabrielsson S. 2010. Proinflammatory exosomes in bronchoalveolar lavage fluid of patients with sarcoidosis. Thorax. 65:1016–1024 10.1136/thx.2009.132027 [DOI] [PubMed] [Google Scholar]
- Rabinowits G., Gerçel-Taylor C., Day J.M., Taylor D.D., Kloecker G.H. 2009. Exosomal microRNA: a diagnostic marker for lung cancer. Clin. Lung Cancer. 10:42–46 10.3816/CLC.2009.n.006 [DOI] [PubMed] [Google Scholar]
- Raiborg C., Stenmark H. 2009. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature. 458:445–452 10.1038/nature07961 [DOI] [PubMed] [Google Scholar]
- Rajendran L., Honsho M., Zahn T.R., Keller P., Geiger K.D., Verkade P., Simons K. 2006. Alzheimer’s disease beta-amyloid peptides are released in association with exosomes. Proc. Natl. Acad. Sci. USA. 103:11172–11177 10.1073/pnas.0603838103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rak J. 2010. Microparticles in cancer. Semin. Thromb. Hemost. 36:888–906 10.1055/s-0030-1267043 [DOI] [PubMed] [Google Scholar]
- Rana S., Yue S., Stadel D., Zöller M. 2012. Toward tailored exosomes: the exosomal tetraspanin web contributes to target cell selection. Int. J. Biochem. Cell Biol. 44:1574–1584 10.1016/j.biocel.2012.06.018 [DOI] [PubMed] [Google Scholar]
- Rao S.K., Huynh C., Proux-Gillardeaux V., Galli T., Andrews N.W. 2004. Identification of SNAREs involved in synaptotagmin VII-regulated lysosomal exocytosis. J. Biol. Chem. 279:20471–20479 10.1074/jbc.M400798200 [DOI] [PubMed] [Google Scholar]
- Raposo G., Nijman H.W., Stoorvogel W., Liejendekker R., Harding C.V., Melief C.J., Geuze H.J. 1996. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 183:1161–1172 10.1084/jem.183.3.1161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raposo G., Tenza D., Mecheri S., Peronet R., Bonnerot C., Desaymard C. 1997. Accumulation of major histocompatibility complex class II molecules in mast cell secretory granules and their release upon degranulation. Mol. Biol. Cell. 8:2631–2645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raposo G., Marks M.S., Cutler D.F. 2007. Lysosome-related organelles: driving post-Golgi compartments into specialisation. Curr. Opin. Cell Biol. 19:394–401 10.1016/j.ceb.2007.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ratajczak J., Wysoczynski M., Hayek F., Janowska-Wieczorek A., Ratajczak M.Z. 2006. Membrane-derived microvesicles: important and underappreciated mediators of cell-to-cell communication. Leukemia. 20:1487–1495 10.1038/sj.leu.2404296 [DOI] [PubMed] [Google Scholar]
- Ronquist G., Brody I. 1985. The prostasome: its secretion and function in man. Biochim. Biophys. Acta. 822:203–218 10.1016/0304-4157(85)90008-5 [DOI] [PubMed] [Google Scholar]
- Savina A., Vidal M., Colombo M.I. 2002. The exosome pathway in K562 cells is regulated by Rab11. J. Cell Sci. 115:2505–2515 [DOI] [PubMed] [Google Scholar]
- Savina A., Fader C.M., Damiani M.T., Colombo M.I. 2005. Rab11 promotes docking and fusion of multivesicular bodies in a calcium-dependent manner. Traffic. 6:131–143 10.1111/j.1600-0854.2004.00257.x [DOI] [PubMed] [Google Scholar]
- Segura E., Guérin C., Hogg N., Amigorena S., Théry C. 2007. CD8+ dendritic cells use LFA-1 to capture MHC-peptide complexes from exosomes in vivo. J. Immunol. 179:1489–1496 [DOI] [PubMed] [Google Scholar]
- Sheldon H., Heikamp E., Turley H., Dragovic R., Thomas P., Oon C.E., Leek R., Edelmann M., Kessler B., Sainson R.C., et al. 2010. New mechanism for Notch signaling to endothelium at a distance by Delta-like 4 incorporation into exosomes. Blood. 116:2385–2394 10.1182/blood-2009-08-239228 [DOI] [PubMed] [Google Scholar]
- Shen B., Fang Y., Wu N., Gould S.J. 2011. Biogenesis of the posterior pole is mediated by the exosome/microvesicle protein-sorting pathway. J. Biol. Chem. 286:44162–44176 10.1074/jbc.M111.274803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons M., Raposo G. 2009. Exosomes—vesicular carriers for intercellular communication. Curr. Opin. Cell Biol. 21:575–581 10.1016/j.ceb.2009.03.007 [DOI] [PubMed] [Google Scholar]
- Simpson R.J., Lim J.W., Moritz R.L., Mathivanan S. 2009. Exosomes: proteomic insights and diagnostic potential. Expert Rev. Proteomics. 6:267–283 10.1586/epr.09.17 [DOI] [PubMed] [Google Scholar]
- Skog J., Würdinger T., van Rijn S., Meijer D.H., Gainche L., Sena-Esteves M., Curry W.T., Jr, Carter B.S., Krichevsky A.M., Breakefield X.O. 2008. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 10:1470–1476 10.1038/ncb1800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soo C.Y., Song Y., Zheng Y., Campbell E.C., Riches A.C., Gunn-Moore F., Powis S.J. 2012. Nanoparticle tracking analysis monitors microvesicle and exosome secretion from immune cells. Immunology. 136:192–197 10.1111/j.1365-2567.2012.03569.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stegmayr B., Ronquist G. 1982. Promotive effect on human sperm progressive motility by prostasomes. Urol. Res. 10:253–257 10.1007/BF00255932 [DOI] [PubMed] [Google Scholar]
- Stuffers S., Sem Wegner C., Stenmark H., Brech A. 2009. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic. 10:925–937 10.1111/j.1600-0854.2009.00920.x [DOI] [PubMed] [Google Scholar]
- Subra C., Laulagnier K., Perret B., Record M. 2007. Exosome lipidomics unravels lipid sorting at the level of multivesicular bodies. Biochimie. 89:205–212 10.1016/j.biochi.2006.10.014 [DOI] [PubMed] [Google Scholar]
- Tamai K., Tanaka N., Nakano T., Kakazu E., Kondo Y., Inoue J., Shiina M., Fukushima K., Hoshino T., Sano K., et al. 2010. Exosome secretion of dendritic cells is regulated by Hrs, an ESCRT-0 protein. Biochem. Biophys. Res. Commun. 399:384–390 10.1016/j.bbrc.2010.07.083 [DOI] [PubMed] [Google Scholar]
- Theos A.C., Truschel S.T., Tenza D., Hurbain I., Harper D.C., Berson J.F., Thomas P.C., Raposo G., Marks M.S. 2006. A lumenal domain-dependent pathway for sorting to intralumenal vesicles of multivesicular endosomes involved in organelle morphogenesis. Dev. Cell. 10:343–354 10.1016/j.devcel.2006.01.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Théry C., Regnault A., Garin J., Wolfers J., Zitvogel L., Ricciardi-Castagnoli P., Raposo G., Amigorena S. 1999. Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein hsc73. J. Cell Biol. 147:599–610 10.1083/jcb.147.3.599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Théry C., Boussac M., Véron P., Ricciardi-Castagnoli P., Raposo G., Garin J., Amigorena S. 2001. Proteomic analysis of dendritic cell-derived exosomes: a secreted subcellular compartment distinct from apoptotic vesicles. J. Immunol. 166:7309–7318 [DOI] [PubMed] [Google Scholar]
- Théry C., Amigorena S., Raposo G., Clayton A. 2006. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr. Protoc. Cell Biol. Chapter 3:Unit 3.22. [DOI] [PubMed] [Google Scholar]
- Théry C., Ostrowski M., Segura E. 2009. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 9:581–593 10.1038/nri2567 [DOI] [PubMed] [Google Scholar]
- Tian T., Wang Y., Wang H., Zhu Z., Xiao Z. 2010. Visualizing of the cellular uptake and intracellular trafficking of exosomes by live-cell microscopy. J. Cell. Biochem. 111:488–496 10.1002/jcb.22733 [DOI] [PubMed] [Google Scholar]
- Trajkovic K., Hsu C., Chiantia S., Rajendran L., Wenzel D., Wieland F., Schwille P., Brügger B., Simons M. 2008. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science. 319:1244–1247 10.1126/science.1153124 [DOI] [PubMed] [Google Scholar]
- Trams E.G., Lauter C.J., Salem N., Jr, Heine U. 1981. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim. Biophys. Acta. 645:63–70 10.1016/0005-2736(81)90512-5 [DOI] [PubMed] [Google Scholar]
- Valadi H., Ekström K., Bossios A., Sjöstrand M., Lee J.J., Lötvall J.O. 2007. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9:654–659 10.1038/ncb1596 [DOI] [PubMed] [Google Scholar]
- van der Vlist E.J., Nolte-’t Hoen E.N., Stoorvogel W., Arkesteijn G.J., Wauben M.H. 2012. Fluorescent labeling of nano-sized vesicles released by cells and subsequent quantitative and qualitative analysis by high-resolution flow cytometry. Nat. Protoc. 7:1311–1326 10.1038/nprot.2012.065 [DOI] [PubMed] [Google Scholar]
- van Niel G., Raposo G., Candalh C., Boussac M., Hershberg R., Cerf-Bensussan N., Heyman M. 2001. Intestinal epithelial cells secrete exosome-like vesicles. Gastroenterology. 121:337–349 10.1053/gast.2001.26263 [DOI] [PubMed] [Google Scholar]
- van Niel G., Mallegol J., Bevilacqua C., Candalh C., Brugière S., Tomaskovic-Crook E., Heath J.K., Cerf-Bensussan N., Heyman M. 2003. Intestinal epithelial exosomes carry MHC class II/peptides able to inform the immune system in mice. Gut. 52:1690–1697 10.1136/gut.52.12.1690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Niel G., Porto-Carreiro I., Simoes S., Raposo G. 2006. Exosomes: a common pathway for a specialized function. J. Biochem. 140:13–21 10.1093/jb/mvj128 [DOI] [PubMed] [Google Scholar]
- van Niel G., Charrin S., Simoes S., Romao M., Rochin L., Saftig P., Marks M.S., Rubinstein E., Raposo G. 2011. The tetraspanin CD63 regulates ESCRT-independent and -dependent endosomal sorting during melanogenesis. Dev. Cell. 21:708–721 10.1016/j.devcel.2011.08.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vella L.J., Sharples R.A., Lawson V.A., Masters C.L., Cappai R., Hill A.F. 2007. Packaging of prions into exosomes is associated with a novel pathway of PrP processing. J. Pathol. 211:582–590 10.1002/path.2145 [DOI] [PubMed] [Google Scholar]
- Wang S., Cesca F., Loers G., Schweizer M., Buck F., Benfenati F., Schachner M., Kleene R. 2011. Synapsin I is an oligomannose-carrying glycoprotein, acts as an oligomannose-binding lectin, and promotes neurite outgrowth and neuronal survival when released via glia-derived exosomes. J. Neurosci. 31:7275–7290 10.1523/JNEUROSCI.6476-10.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White I.J., Bailey L.M., Aghakhani M.R., Moss S.E., Futter C.E. 2006. EGF stimulates annexin 1-dependent inward vesiculation in a multivesicular endosome subpopulation. EMBO J. 25:1–12 10.1038/sj.emboj.7600759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson H.L., Francis S.E., Dower S.K., Crossman D.C. 2004. Secretion of intracellular IL-1 receptor antagonist (type 1) is dependent on P2X7 receptor activation. J. Immunol. 173:1202–1208 [DOI] [PubMed] [Google Scholar]
- Wolfers J., Lozier A., Raposo G., Regnault A., Théry C., Masurier C., Flament C., Pouzieux S., Faure F., Tursz T., et al. 2001. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat. Med. 7:297–303 10.1038/85438 [DOI] [PubMed] [Google Scholar]
- Wubbolts R., Leckie R.S., Veenhuizen P.T., Schwarzmann G., Möbius W., Hoernschemeyer J., Slot J.W., Geuze H.J., Stoorvogel W. 2003. Proteomic and biochemical analyses of human B cell-derived exosomes. Potential implications for their function and multivesicular body formation. J. Biol. Chem. 278:10963–10972 10.1074/jbc.M207550200 [DOI] [PubMed] [Google Scholar]
- Yu X., Harris S.L., Levine A.J. 2006. The regulation of exosome secretion: a novel function of the p53 protein. Cancer Res. 66:4795–4801 10.1158/0008-5472.CAN-05-4579 [DOI] [PubMed] [Google Scholar]
- Zhang H.G., Grizzle W.E. 2011. Exosomes and cancer: a newly described pathway of immune suppression. Clin. Cancer Res. 17:959–964 10.1158/1078-0432.CCR-10-1489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zitvogel L., Regnault A., Lozier A., Wolfers J., Flament C., Tenza D., Ricciardi-Castagnoli P., Raposo G., Amigorena S. 1998. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4:594–600 10.1038/nm0598-594 [DOI] [PubMed] [Google Scholar]
- Zöller M. 2009. Tetraspanins: push and pull in suppressing and promoting metastasis. Nat. Rev. Cancer. 9:40–55 10.1038/nrc2543 [DOI] [PubMed] [Google Scholar]