Abstract
Iron homeostasis influences the development of pulmonary arterial hypertension (PAH) associated with hypoxia or hematologic disorders. To investigate whether severity of idiopathic PAH (IPAH) is impacted by alterations in iron metabolism, we assessed iron metabolic markers, including levels of zinc‐protoporphyrin (Zn‐pp), transferrin receptor, and red blood cell numbers and morphology in IPAH, associated PAH and sleep apnea‐induced pulmonary hypertension patients in comparison to healthy controls and asthmatics. Despite similarly normal measures of iron metabolism, Zn‐pp levels in IPAH and sleep apnea patients were elevated approximately twofold, indicating deficient iron incorporation to form heme and levels were closely related to measures of disease severity. Consistent with high Zn‐pp, PAH patients had increased red cell distribution width (RDW). In an expanded cohort including patients with IPAH and familial disease, the RDW was validated and related to clinical parameters of severity; including pulmonary artery pressures and 6‐minute walk distances. These results reveal an increased prevalence of subclinical functional iron deficiency in primary forms of PAH that is quantitatively related to disease severity. This suggests that altered iron homeostasis influences disease progression and demonstrates the importance of closely monitoring iron status in PAH patients. Clin Trans Sci 2011; Volume 4: 253–258
Keywords: pulmonary arterial hypertension, zinc‐protoporphyrin, iron metabolism
Introduction
Pulmonary arterial hypertension (PAH), a fatal disease characterized by an increase in pulmonary arterial pressure, may occur as a primary idiopathic (IPAH) or familial disease (FPAH), or as a secondary process associated with other diseases, for example, diseases of red blood cell production including anemias such as beta‐thalassemia and sickle cell anemia, mutations in iron regulatory proteins such as Chuvash polycythemia, and myeloproliferative disorders such as polycythemia vera. 1 , 2 , 3 Disorders of red blood cell production with less heme oxygen carrying capacity lead to tissue hypoxia, which is assumed to be the predisposing event for PAH associated with hematologic abnormalities. However, the attenuation of pulmonary hypertensive response to acute and sustained hypoxia by intravenous iron infusion also indicates that iron is a unique determinant in pulmonary vasomotor responses. 4 , 5 In contrast to secondary forms of PAH, idiopathic PAH (IPAH) occurs in the absence of ambient hypoxia or diseases known to lead to tissue hypoxia. 6 , 7 , 8 , 9 Yet, recent studies also point to a link between iron metabolism and IPAH. For example, low hemoglobin levels are related to decreased survival in all classes of pulmonary hypertension (PH), including IPAH, 10 and clinical assessments of iron stores suggest abnormalities in iron metabolism. 11
Iron is essential for productive heme synthesis, when iron is incorporated into the protoporphyrin ring by the mitochondrial enzyme ferrochelatase to produce heme and ultimately hemoproteins. In states of iron deficiency or block to iron incorporation, zinc is inserted in place of iron to form zinc‐protoporphyrin (Zn‐pp). 12 For example, lead blocks insertion of iron into protoporphyrin; hence, the Zn‐pp/heme ratio is commonly used to screen for lead poisoning. However, Zn‐pp is highly sensitive to any iron metabolic abnormalities as well as absolute iron deficiency. 12 , 13 , 14 Here, we hypothesized that abnormalities of iron metabolism are present and contribute to the pathophysiology of PAH. To test this, we first measured Zn‐pp in red blood cells of patients with IPAH in comparison to control healthy and asthmatic individuals as a nonvascular inflammatory lung disease control. Despite similarly normal red cell counts and hemoglobin levels, Zn‐pp was elevated in IPAH patients as compared to controls, and closely related to measures of disease severity. Levels of Zn‐pp were also elevated in sleep apnea‐related pulmonary hypertension (PH), but not in associated PAH (APAH) patients or individuals with asthma. Consistent with the finding of deficient iron incorporation to form heme, IPAH and sleep apnea PH patients also had greater variation in the size distribution of red blood cells, that is, an increase in the red cell distribution width (RDW), which was significantly related to Zn‐pp levels. In an expanded cohort that included PAH patients with the idiopathic and familial form of the disease, RDW was also elevated and tightly correlated with key clinical parameters of PAH severity, including pulmonary artery pressures and 6‐minute walk distances. These findings reveal that primary forms of PAH have a reduction in systemic iron availability that is related to parameters of disease severity, which suggests that a decrease in functional iron availability may contribute to PH.
Patients and Methods
Study population
Patients with IPAH, APAH, and PH related to sleep apnea were enrolled in the study. Controls included healthy individuals and asthmatics, who were recruited by advertisement. Controls were chosen to match IPAH subjects based on age, gender, and race. The initial IPAH cohort was later expanded by additional IPAH and FPAH patients for evaluation of red cell parameters that reflect iron metabolism. PAH was defined by the Third World Symposium on Pulmonary Arterial Hypertension 15 and IPAH patients were classified based on the National Institutes of Health (NIH) registry diagnostic criteria for PH subclass 1.1 (according to the World Health Organization criteria). 16 Sleep apnea was defined based on a >90% reduction in tidal volume lasting >10 seconds accompanied by >3% decrease in oxyhemoglobin saturation or terminated by arousal from sleep. 17 Asthma was verified based on positive methacholine challenge test and/or reversible airway obstruction by documentation of change in forced expiratory volume in one second or forced vital capacity by 12% and greater than 200 mL after two puffs of inhaled bronchodilator. 18 Exclusion criteria for all groups included a hemoglobin level below the reference range for the Cleveland Clinic laboratories, <11.5 g/dL for females and <13.0 g/dL for males. The study was approved by the Institutional Review Boards of the Cleveland Clinic (06–493) and written informed consent was obtained from all individuals.
Iron metabolism measures
Zn‐pp was measured in red blood cells that were oxygenated using a hematofluorometer (Hematoflourometer Model Zpp 206D, AVIV Biomedical Incorporated, Lakewood, NJ, USA). The fluorescence of Zn‐pp at 596 nm relative to heme absorption at 420 nm was determined; the ratio of μmol Zn‐pp/mol heme is reported. Transferrin receptor concentrations were measured in patient plasma by enzyme‐linked immunosorbant assay (ELISA R&D systems, Minneapolis, MN, USA). Complete blood counts were obtained under standard laboratory conditions.
Clinical assessment
Measure of pulmonary artery hypertension in all PAH subjects was estimated by right ventricular systolic pressure from echocardiography and/or by right heart catheterization and values reported for those measures in close proximity to the date of blood draw for analyses. Pulmonary arterial systolic pressure was estimated from the systolic pressure gradient between the right ventricle and the right atrium by the peak continuous wave Doppler velocity of the tricuspid regurgitant jet using the modified Bernoulli equation plus right atrial pressure estimated from the inferior vena caval size and collapsibility with respiration, as previously validated and described by others. 19 Patients’ functional status was determined through 6‐minute walk distances.
Statistical analysis
Continuous variables are presented as mean ± standard error of the mean or as median (25–75 percentile). Noncontinuous variables are presented as percentages. Comparisons of continuous variables between groups were performed using one‐way analysis of variance. The log transformation was employed when necessary to achieve normality. Associations between variables were assessed using Pearson correlation coefficients. A multivariable linear regression model was employed when comparing Zn‐pp levels among the groups to include covariate adjustment for the suspected confounding factors gender, age, and race. Statistical significance was assumed with p < 0.05. All analyses were performed using JMP version 8.0 (SAS Institute, Cary, NC, USA).
Results
Study population characteristics
Participants were similar in age and gender ( Table 1 ). PAH patients’ medications were similar and included prostacyclin (62%), warfarin (45%), diuretics (55%), endothelin receptor antagonists (45%), sildenafil (31%), and digoxin (24%). APAH patients’ primary diseases were rheumatoid arthritis, monoclonal gammopathy, Sweet’s syndrome, polycythemia vera, autoimmune hemolytic anemia, hypogammaglobulinemia, portopulmonary hypertension, appetite suppressant use, and congenital heart disease. Four IPAH patients (21%) and one sleep apnea patient (25%) were taking iron supplements, although they had hemoglobin levels above the set reference ranges for the study. Asthmatic volunteers were taking β‐agonists and inhaled corticosteroids as needed to control disease symptoms. IPAH patients had a range of mild to severe disease as determined by pulmonary artery systolic pressures (74 mmHg [interquartile range (IQR) 50–92]), cardiac index (2.5 L/min/m2[IQR 1.8–3.5]), and 6‐minute walk distances (445 m [IQR 363–542]). APAH and sleep apnea‐related PH had similar severity of disease as assessed by pulmonary artery systolic pressure (APAH 72.5 mmHg [IQR 61–99 mmHg]; sleep apnea 58.5 mmHg [IQR 51–75 mmHg]), cardiac index (APAH 2.7 L/min/m2[IQR 1.7–3.4 L/min/m2]; sleep apnea 1.8 L/min/m2[IQR 1.5–2.1 L/min/m2]), and 6‐minute walk distance (APAH 389 m [IQR 302–521 m]; sleep apnea 439 m [IQR 187–549 m]).
Table 1.
Characteristics of participants.
| Controls | Asthma | IPAH | FPAH | APAH | Sleep apnea PH | p value | |
|---|---|---|---|---|---|---|---|
| Number of patients | 12 | 14 | 19 | 11 | 6 | 4 | |
| Age (years) | 36 ± 3 | 40 ± 3 | 48 ± 3 | 41 ± 4 | 53 ± 5 | 57 ± 6 | 0.008 |
| Female/male | 9/3 | 11/3 | 15/4 | 9/2 | 5/1 | 3/1 | 0.99 |
| Race (C/AA/other) | 6/3/3 | 5/7/2 | 14/2/3 | 11/0/0 | 5/1/0 | 4/0/0 | 0.02 |
Data are expressed as mean ± SEM.
Comparisons were performed using one‐way analysis of variance for continuous variables and Fischer’s exact test for categorical data.
IPAH = idiopathic pulmonary arterial hypertension; APAH = associated pulmonary arterial hypertension; FPAH = familial pulmonary arterial hypertension; PH = pulmonary hypertension.
Peripheral blood cells and iron studies
Cell counts varied among the groups ( Table 2 ). APAH patients had lowest platelet counts, but PAH patients generally had low platelet counts. PH sleep apnea and FPAH patients had the highest red blood cell counts. The Zn‐pp/heme ratio in red blood cells was nearly twofold greater in IPAH and sleep apnea patients as compared to asthma and healthy controls, but was similar among APAH and controls (Zn‐pp/heme μmol/mol: IPAH 102 ± 14.4, sleep apnea PH 89.7 ± 17.7, APAH 66.3 ± 15.4, asthma 59.6 ± 6.4, healthy controls 65 ± 7.8, p= 0.02) ( Figure 1 ). All PAH and sleep apnea samples were characterized by higher RDW ( Table 2 ).Despite higher Zn‐pp and RDW, plasma transferrin receptor evaluated in the subgroup of IPAH patients (2.4 ± 0.2 mg/L) was not different than control (1.9 ± 0.2 mg/L) or asthmatic (2.0 ± 0.1 mg/L) samples, p= 0.15.
Table 2.
Blood counts.
| Controls | Asthma | IPAH | FPAH | APAH | Sleep apnea PH | p value | |
|---|---|---|---|---|---|---|---|
| Red blood cell count (M/μL) | 4.5 ± 0.1 | 4.6 ± 0.2 | 4.9 ± 0.1 | 5.2 ± 0.1 | 4.4 ± 0.2 | 5.0 ± 0.2 | 0.009* |
| Hemoglobin (g/dL) | 13.3 ± 0.3 | 13.5 ± 0.4 | 13.6 ± 0.3 | 14.5 ± 0.4 | 12.7 ± 0.6 | 14.1 ± 0.7 | 0.17 |
| Hematocrit (%) | 40.4 ± 0.9 | 40.9 ± 1.1 | 41.9 ± 0.9 | 43.5 ± 1.1 | 39.6 ± 1.7 | 42.8 ± 2.2 | 0.28 |
| Mean corpuscular volume (fL) | 89.1 ± 1.6 | 87.2 ± 1.4 | 86.6 ± 1.0 | 84.3 ± 1.5 | 89.8 ± 1.4 | 86.1 ± 2.8 | 0.15 |
| Mean corpuscular hemoglobin (pG) | 29.4 ± 0.6 | 29.0 ± 0.6 | 28.1 ± 0.4 | 28.0 ± 0.8 | 29.1 ± 0.8 | 28.2 ± 1.1 | 0.52 |
| Mean corpuscular hemoglobin concentration (g/dL) | 33.0 ± 0.3 | 33.2 ± 0.3 | 32.4 ± 0.3 | 33.2 ± 0.4 | 32.3 ± 0.5 | 33.0 ± 0.6 | 0.31 |
| Red blood cell distribution width (%) | 13.2 ± 0.3 | 13.7 ± 0.3 | 14.7 ± 0.4 | 15.1 ± 0.8 | 14.6 ± 0.4 | 15.7 ± 1.7 | 0.05* |
| White blood cell count (k/μL) | 6.0 ± 0.4 | 6.7 ± 0.5 | 6.0 ± 0.4 | 8.6 ± 0.6 | 5.4 ± 0.5 | 6.5 ± 1.1 | 0.004* |
| Platelets (k/μL) | 285.4 ± 20.1 | 315.4 ± 26.3 | 202.8 ± 20.9 | 213 ± 24.6 | 166.5 ± 25.1 | 300.5 ± 101.0 | 0.004* |
Data are expressed as mean ± SEM.
Comparisons were performed using one‐way analysis of variance.
*log transformation and ANOVA was used for unequal variances.
Figure 1.
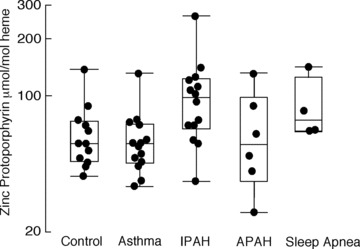
Zinc‐protoporphyrin/heme ratio (μmol/mol heme) is increased in IPAH and sleep apnea PH patients. Zn‐pp is increased in IPAH and sleep apnea PH patients compared to controls, asthmatics, and APAH patients (p= 0.02).
Wright stains of peripheral blood did not reveal evidence of basophilic stippling or Pappenheimer bodies, although increased rouleaux formation was often observed in IPAH samples as compared to controls ( Figure 2 ). Among the healthy or asthma control sample, Zn‐pp was not significantly related to any other hematologic measures. However, within IPAH samples, Zn‐pp was strongly related to RDW (R= 0.75, p= 0.002), mean corpuscular hemoglobin (MCH) (R=–0.87, p < 0.001), mean corpuscular hemoglobin concentration (MCHC) (R=–0.84, p < 0.001), and transferrin receptor (R= 0.70, p= 0.008). The same trends held true when considering all PH patients together: Zn‐pp was related to RDW (R= 0.63, p= 0.001), MCH (R=–0.82, p < 0.001), mean corpuscular volume (MCV) (R=–0.69, p < 0.001), and MCHC (R=–0.74, p < 0.001).
Figure 2.
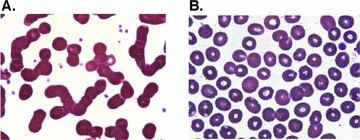
Rouleaux formation in IPAH blood smears. The peripheral blood smears of IPAH patients (A) shows increased rouleaux formation compared to control subjects (B).
Zn‐pp and clinical parameters of disease severity
Zn‐pp levels were associated with severity of PH in IPAH patients. Pulmonary artery systolic pressures measured by echocardiography and pulmonary artery pressures measured by right heart catheterization correlated well (R= 0.69, p= 0.0006). In IPAH patients, Zn‐pp levels paralleled pulmonary arterial pressures measured by echocardiography (R= 0.63, p= 0.02) and measured by right heart catherization (R= 0.57, p= 0.05] and were inversely related to distance walked in 6 minutes (R=–0.57, p= 0.04) ( Figure 3 ). The 6‐minute walk distance was similarly related to RDW (R=–0.65, p= 0.02). Further, Zn‐pp tended to correlate with other hemodynamic parameters such as right atrial pressure (R= 0.50, p= 0.1) and cardiac index (R=–0.55, p= 0.08). When patients receiving iron supplementation were excluded, there was a more robust correlation of Zn‐pp to pulmonary arterial systolic pressure (R= 0.75, p= 0.005) and 6‐minute walk distance (R=–0.67, p= 0.02). While the small numbers of sleep apnea PH patients precluded correlation analyses, Zn‐pp values of APAH patients were unrelated to hemodynamic measures or 6‐minute walk distance (all p > 0.1).
Figure 3.
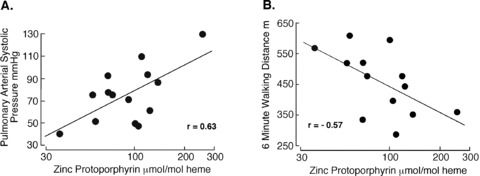
Zn‐pp is related to disease severity in IPAH. (A) Zn‐pp and pulmonary arterial systolic pressure in IPAH. In IPAH patients, the pulmonary artery systolic pressures increase with increasing Zn‐pp levels (R= 0.63, p= 0.02). (B) Zn‐pp and 6‐minute walk distance in IPAH. Functional status as assessed by distance able to walk within 6 minutes is inversely related to Zn‐pp levels (R=–0.57, p= 0.04).
There was no relationship between Zn‐pp and brain natriuretic factor (BNP), an established biomarker of cardiac failure in PAH, but we found the expected correlations between BNP and cardiopulmonary function including a direct relationship to pulmonary arterial systolic pressure (R= 0.77, p= 0.005), and inverse relation to 6‐minute walk distance (R=–0.62, p= 0.04).
Increased RDW in an expanded cohort of FPAH and IPAH is related to disease severity
Given the close association of Zn‐pp to RDW, and the relationship of both to clinical disease severity, we investigated RDW as a quantitative and readily available biomarker of clinical disease severity in an expanded cohort of PAH that included FPAH and IPAH individuals. FPAH patients generally had more severe disease than IPAH individuals (pulmonary artery systolic pressure 71.0 mmHg [IQR 40–90 mmHg]; cardiac index 1.6 L/min/m2[IQR 1.3–2.5 L/min/m2]; 6‐minute walk distance 389 m[IQR 373–530 m]). One FPAH patient was taking oral iron supplement but was not anemic ( Table 2 ). Among the IPAH and FPAH patients, RDW was related to pulmonary arterial systolic pressures measured by echocardiography (R= 0.47, p= 0.01) and pulmonary arterial pressure measured by right heart catherization (R= 0.51, p= 0.01) and inversely related to 6‐minute walk distance (R=−0.52, p= 0.01) ( Figure 4 ). In addition, RDW correlated with other hemodynamic measures such as right atrial pressure (R= 0.62, p= 0.002) and peripheral vascular resistance (R= 0.61, p= 0.02). Exclusion of patients receiving iron supplementation strengthened the relation of RDW to pulmonary artery systolic pressure (R= 0.52, p= 0.008) and 6‐minute walk distance (R= 0.61, p= 0.005).
Figure 4.
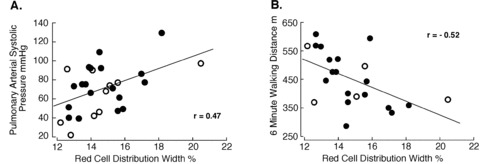
RDW and parameters of disease severity in IPAH and FPAH. (A) RDW parallels pulmonary arterial systolic pressures (R= 0.47, p= 0.01). (B) RDW is inversely associated to 6‐minute walking distance (R=–0.52, p= 0.01). Open circles, FPAH; closed circles, IPAH.
Discussion
The current study identifies low functional iron availability in IPAH patients. While other studies have shown that anemia, iron deficiency, as well as high RDW are prognosticators for poor outcomes in PAH patients, 10 , 11 , 20 to our knowledge, this is the first study that uncovers the direct relationship of iron utilization to severity of PAH. Iron‐containing proteins are essential to oxygen transfer and storage, such as hemoglobin in the red blood cell and myoglobin in cardiac muscle, but are also essential for numerous vital cellular metabolic functions, including the mitochondrial electron transport chain and catalysis reactions that form nitric oxide (NO). 21 Abnormalities of NO production by NO synthases, heme‐containing enzymes, and mitochondrial oxygen utilization and energy production by cytochrome oxidases have all been described as fundamental to PAH pathophysiology. 22 , 23 , 24 , 25 , 26 , 27 , 28
Greater levels of Zn‐pp and rouleaux formation may be found in patients with functional iron deficiency that develops from changes in iron homeostasis induced by inflammatory cytokines, that is, anemia of chronic disease. 29 Localized inflammation around pulmonary vessels has been described in IPAH 30 , 31 , 32 and might be sufficient to cause anemia of chronic disease. 14 Soon et al. showed increased inflammatory cytokines in the IPAH population and a possible relation of interleukin‐6 to iron deficiency. 33 , 34 In support of a functional iron deficiency, soluble transferrin receptor was similar among control groups and IPAH, suggesting that a block to iron use may be responsible for the greater levels of Zn‐pp. 29 , 35 Interestingly, four IPAH patients were receiving oral supplementation with iron. Excluding these patients from the analysis strengthened the association between hematologic parameters and disease severity suggesting that the negative impact of an uncorrected iron disturbance on disease might be alleviated by correcting the iron imbalance. On the other hand, iron supplementation should lower Zn‐pp, but two of the patients with iron supplementation had elevations of Zn‐pp, transferrin receptor, and RDW, which would support a block to either iron absorption and/or utilization at the cellular level. Despite the lack of basophilic stippling, future studies might consider testing blood levels of lead, high levels of which could contribute to block of iron incorporation to heme and subsequent high Zn‐pp levels. In this context, parenteral iron supplementation has been recently shown to be safe and effective in the treatment of left heart failure. 36 , 37 Intravenous iron infusions (but not oral ingestion of iron) and/or treatment with erythropoietin have been shown to increase hemoglobin and decrease the risk of hospitalizations due to cardiac events. 37 Although benefits have been assumed to be related to hemoglobin and oxygen‐carrying capacity, clinical symptoms, functional capacity, and quality of life improve in patients regardless of iron status prior to the study, which suggests that iron effects other than red blood cell hemoglobin content, for example, NO synthesis and/or mitochondria function may be involved. 36 In support of this concept, Smith et al. showed that decreasing serum iron acutely through chelation with desferoxamine amplified the pulmonary hypertensive responses of healthy subjects under hypoxia condition. 4 , 5 This suggests that even a slight decrease in functional iron availability to cells may significantly worsen disease in PAH patients.
A prior study by Hampole et al. showed that RDW is a sensitive prognostic marker of mortality in PAH patients that is superior to BNP, a well‐validated biomarker of heart failure. 20 Similarly, studies of Patel et al. and Perlstein et al. found RDW to be a strong predictor of mortality in the general population that was associated with increased risk of death due to cardiovascular disease, cancer, and chronic lower respiratory tract disease. 38 , 39 In this study, RDW was directly related to pulmonary artery pressures and functional status of patients. The elevation of Zn‐pp may occur due to transient, intermittent episodes of decreased systemic iron availability or block to utilization, and produce the changes in mean corpuscular volume without the development of overt anemia in PAH. In addition to the alterations in iron homeostasis, platelet counts were generally decreased in PAH groups, which confirms previous reports of platelet dysfunction and hypercoagulation in this patient population. 40 , 41 However, another commonly reported reason for the low observed platelet count may be prostacyclins used in the majority of these patients. A pattern of hypercoagulability and increased thrombosis has been suggested to account for the PH seen in splenectomized patients with beta‐thalassemia major and intermedia, unstable hemoglobin mutants, and certain inherited and acquired red cell membrane disorders. 42 , 43 Here, our APAH sample had blood disorders, including polycythemia vera and monoclonal gammopathy, but did not have elevation of Zn‐pp or any relationship of Zn‐pp to blood counts or hemodynamic parameters, which is in contrast to the findings in IPAH, FPAH, and sleep apnea patients. It is possible that the variety of primary diseases in the APAH group confound relationships. A larger sample size accounting for differences within the group would be helpful to address this possibility in the future. On the other hand, sleep apnea PH patients had a profile very similar to the IPAH patients, which supports the idea that PH, such as induced by transient hypoxia, may be exacerbated by concurrent functional iron deficiency. Overall, this study together with prior reports provide evidence that altered iron homeostasis may contribute to the development and/or severity of PH. The finding that RDW is related to Zn‐pp in this study also suggests that RDW may serve as a surrogate metric of abnormalities of iron handling in the cell and PAH severity.
Conclusion
This study reveals that Zn‐pp, which is a quantitative and sensitive measure of functional deficiency of iron utilization, is elevated in IPAH and sleep apnea‐related PH and is strongly related to pulmonary artery pressure. The results provide evidence in favor of altered iron homeostasis in PH pathophysiology. The findings suggest that iron status should be monitored and iron supplementation considered in the care of patients. In this context, our study demonstrates a close relationship of Zn‐pp and disease severity to RDW, a readily available clinical measure. Further research to identify the causes for, and many potential downstream consequences of iron metabolic abnormalities that mechanistically contribute to PH is needed in order to plan optimal therapies in the care of patients.
Conflict of Interest
None.
Acknowledgments
This work was supported by the National Institute of Health [HL60917]. Ilka Decker was a Howard Hughes Medical Institute Medical Research Training Fellow. The authors thank G. Brittenham for helpful discussions and J. Hanson for technical support of experiments.
References
- 1. Adir Y, Humbert M. Pulmonary hypertension in patients with chronic myeloproliferative disorders. Eur Respir J. 2010; 35: 1396–1406. [DOI] [PubMed] [Google Scholar]
- 2. Hickey MM, Richardson T, Wang T, Mosqueira M, Arguiri E, Yu H, Yu QC, Solomides CC, Morrisey EE, Khurana TS, Christofidou‐Solomidou M., and Simon M.C. The von Hippel‐Lindau Chuvash mutation promotes pulmonary hypertension and fibrosis in mice. J Clin Invest. 2010; 120: 827–839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Rich S, Hart K. Familial pulmonary hypertension in association with an abnormal hemoglobin. Insights into the pathogenesis of primary pulmonary hypertension. Chest. 1991; 99: 1208–1210. [DOI] [PubMed] [Google Scholar]
- 4. Smith TG, Balanos GM, Croft QP, Talbot NP, Dorrington KL, Ratcliffe PJ, Robbins PA. The increase in pulmonary arterial pressure caused by hypoxia depends on iron status. J Physiol. 2008; 586: 5999–6005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Smith TG, Talbot NP, Privat C, Rivera‐Ch M, Nickol AH, Ratcliffe PJ, Dorrington KL, Leon‐Velarde F, Robbins, PA Effects of iron supplementation and depletion on hypoxic pulmonary hypertension: two randomized controlled trials. JAMA. 2009; 302: 1444–1450. [DOI] [PubMed] [Google Scholar]
- 6. Fagan KA. Selected Contribution: pulmonary hypertension in mice following intermittent hypoxia. J Appl Physiol. 2001; 90: 2502–2507. [DOI] [PubMed] [Google Scholar]
- 7. Farber HW, Loscalzo J. Pulmonary arterial hypertension. N Engl J Med. 2004; 351: 1655–1665. [DOI] [PubMed] [Google Scholar]
- 8. McLaughlin VV, McGoon MD. Pulmonary arterial hypertension. Circulation. 2006; 114: 1417–1431. [DOI] [PubMed] [Google Scholar]
- 9. Weitzenblum E, Chaouat A, Canuet M, Kessler R. Pulmonary hypertension in chronic obstructive pulmonary disease and interstitial lung diseases. Semin Respir Crit Care Med. 2009; 30: 458–470. [DOI] [PubMed] [Google Scholar]
- 10. Krasuski RA, Hart SA, Smith B, Wang A, Harrison JK, Bashore TM. Association of anemia and long‐term survival in patients with pulmonary hypertension. 2010 May 14 [Epub ahead of print]. [DOI] [PubMed]
- 11. Ruiter G, Lankhorst S, Boonstra A, Postmus PE, Zweegman S, Westerhof N, van der Laarse WJ, Vonk‐Noordegraaf A. Iron deficiency is common in idiopathic pulmonary arterial hypertension. Eur Respir J. 2011; 37:1386–1391. [DOI] [PubMed] [Google Scholar]
- 12. Labbe RF, Vreman HJ, Stevenson DK. Zinc protoporphyrin: a metabolite with a mission. Clin Chem. 1999; 45: 2060–2072. [PubMed] [Google Scholar]
- 13. Hastka J, Lasserre JJ, Schwarzbeck A, Hehlmann R. Central role of zinc protoporphyrin in staging iron deficiency. Clin Chem. 1994; 40: 768–773. [PubMed] [Google Scholar]
- 14. Hastka J, Lasserre JJ, Schwarzbeck A, Strauch M, Hehlmann R. Zinc protoporphyrin in anemia of chronic disorders. Blood. 1993; 81: 1200–1204. [PubMed] [Google Scholar]
- 15. Simonneau G, Galie N, Rubin LJ, Langleben D, Seeger W, Domenighetti G, Gibbs S, Lebrec D, Speich R, Beghetti, M. , et al Clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2004; 43: 5S–12S. [DOI] [PubMed] [Google Scholar]
- 16. Ghamra ZW, Dweik RA. Primary pulmonary hypertension: an overview of epidemiology and pathogenesis. Cleve Clin J Med. 2003; 70( Suppl 1): S2–S8. [DOI] [PubMed] [Google Scholar]
- 17. Sleep‐related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999; 22: 667–689. [PubMed] [Google Scholar]
- 18. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide. Am J Respir Crit Care Med 2005; 171: 912–930. [DOI] [PubMed] [Google Scholar]
- 19. Kircher BJ, Himelman RB, Schiller NB. Noninvasive estimation of right atrial pressure from the inspiratory collapse of the inferior vena cava. Am J Cardiol. 1990; 66: 493–496. [DOI] [PubMed] [Google Scholar]
- 20. Hampole CV, Mehrotra AK, Thenappan T, Gomberg‐Maitland M, Shah SJ. Usefulness of red cell distribution width as a prognostic marker in pulmonary hypertension. Am J Cardiol. 2009; 104: 868–872. [DOI] [PubMed] [Google Scholar]
- 21. Stuehr DJ, Tejero J, Haque MM Structural and mechanistic aspects of flavoproteins: electron transfer through the nitric oxide synthase flavoprotein domain. Febs J. 2009; 276: 3959–3974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Archer SL, Marsboom G, Kim GH, Zhang HJ, Toth PT, Svensson EC, Dyck JR, Gomberg‐Maitland M, Thebaud B, Husain AN, et al Epigenetic attenuation of mitochondrial superoxide dismutase 2 in pulmonary arterial hypertension: a basis for excessive cell proliferation and a new therapeutic target. Circulation. 2010; 121: 2661–2671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fijalkowska I, Xu W, Comhair SA, Janocha AJ, Mavrakis LA, Krishnamachary B, Zhen L, Mao T, Richter A, Erzurum SC, et al Hypoxia inducible‐factor1alpha regulates the metabolic shift of pulmonary hypertensive endothelial cells. Am J Pathol. 2010; 176: 1130–1138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kaneko FT, Arroliga AC, Dweik RA, Comhair SA, Laskowski D, Oppedisano R, Thomassen MJ Erzurum SC. Biochemical reaction products of nitric oxide as quantitative markers of primary pulmonary hypertension. Am J Respir Crit Care Med. 1998; 158: 917–923. [DOI] [PubMed] [Google Scholar]
- 25. Machado RF, Londhe Nerkar MV, Dweik RA, Hammel J, Janocha A, Pyle J, Laskowski D, Jennings C, Arroliga AC, Erzurum SC. Nitric oxide and pulmonary arterial pressures in pulmonary hypertension. Free Radic Biol Med. 2004; 37: 1010–1017. [DOI] [PubMed] [Google Scholar]
- 26. Ozkan M, Dweik RA, Laskowski D, Arroliga AC, Erzurum SC. High levels of nitric oxide in individuals with pulmonary hypertension receiving epoprostenol therapy. Lung. 2001; 179: 233–243. [DOI] [PubMed] [Google Scholar]
- 27. Xu W, Kaneko FT, Zheng S, Comhair SA, Janocha AJ, Goggans T, Thunnissen FB, Farver C, Hazen SL, Jennings C C., et al Increased arginase II and decreased NO synthesis in endothelial cells of patients with pulmonary arterial hypertension. Faseb J. 2004; 18: 1746–1748. [DOI] [PubMed] [Google Scholar]
- 28. Xu W, Koeck T, Lara AR, Neumann D, DiFilippo FP, Koo M, Janocha AJ, Masri FA, Arroliga AC, Jennings C. et al. Alterations of cellular bioenergetics in pulmonary artery endothelial cells. Proc Natl Acad Sci U S A. 2007; 104: 1342–1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med. 2005; 352: 1011–1023. [DOI] [PubMed] [Google Scholar]
- 30. Asosingh K, Aldred MA, Vasanji A, Drazba J, Sharp J, Farver C, Comhair SA, Xu W, Licina L, Huang L, et al Circulating angiogenic precursors in idiopathic pulmonary arterial hypertension. Am J Pathol. 2008; 172: 615–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Aytekin M, Comhair SA, de la Motte C, Bandyopadhyay SK, Farver CF, Hascall VC, Erzurum SC, Dweik RA. High levels of hyaluronan in idiopathic pulmonary arterial hypertension. Am J Physiol Lung Cell Mol Physiol. 2008; 295: L789–L799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Dorfmuller P, Perros F, Balabanian K, Humbert M. Inflammation in pulmonary arterial hypertension. Eur Respir J. 2003; 22: 358–363. [DOI] [PubMed] [Google Scholar]
- 33. Soon E, Holmes AM, Treacy CM, Doughty NJ, Southgate L, Machado RD, Trembath RC, Jennings S, Barker L, Nicklin P, et al Elevated levels of inflammatory cytokines predict survival in idiopathic and familial pulmonary arterial hypertension. Circulation. 2010; 122: 920–927. [DOI] [PubMed] [Google Scholar]
- 34. Soon E, Treacy CM, Toshner MR, Mackenzie‐Ross R, Manglam V, Busbridge M, Sinclair‐McGarvie M, Arnold J, Sheares KK, Morrell NW, Pepke‐Zaba J. Unexplained iron deficiency in idiopathic and heritable pulmonary arterial hypertension. Thorax. 2011; 66: 326–332. [DOI] [PubMed] [Google Scholar]
- 35. Punnonen K, Irjala K, Rajamaki A Serum transferrin receptor and its ratio to serum ferritin in the diagnosis of iron deficiency. Blood. 1997; 89: 1052–1057. [PubMed] [Google Scholar]
- 36. Anker SD, Comin Colet J, Filippatos G, Willenheimer R, Dickstein K, Drexler H, Luscher TF, Bart B, Banasiak W, Niegowska J, et al Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med. 2009; 361: 2436–2448. [DOI] [PubMed] [Google Scholar]
- 37. Comin‐Colet J, Ruiz S, Cladellas M, Rizzo M, Torres A, Bruguera J. A pilot evaluation of the long‐term effect of combined therapy with intravenous iron sucrose and erythropoietin in elderly patients with advanced chronic heart failure and cardio‐renal anemia syndrome: influence on neurohormonal activation and clinical outcomes. J Card Fail. 2009; 15: 727–735. [DOI] [PubMed] [Google Scholar]
- 38. Patel KV, Ferrucci L, Ershler WB, Longo DL, Guralnik JM. Red blood cell distribution width and the risk of death in middle‐aged and older adults. Arch Intern Med. 2009; 169: 515–523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Perlstein TS, Weuve J, Pfeffer MA, Beckman JA. Red blood cell distribution width and mortality risk in a community‐based prospective cohort. Arch Intern Med. 2009; 169: 588–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Bakouboula, B. , Morel, O. , Faure, A. , Zobairi, F. , Jesel, L. , Trinh, A. , Zupan, M. , Canuet, M. , Grunebaum, L. , Brunette, A. , et al Procoagulant membrane microparticles correlate with the severity of pulmonary arterial hypertension. Am J Respir Crit Care Med. 2008; 177: 536–543. [DOI] [PubMed] [Google Scholar]
- 41. Can MM, Tanboga IH, Demircan HC, Ozkan A, Koca F, Keles N, Sonmez K, Kaymaz C, Serebruany V. Enhanced hemostatic indices in patients with pulmonary arterial hypertension: an observational study. Thromb Res. 2010; 126: 280–282. [DOI] [PubMed] [Google Scholar]
- 42. Singer ST, Kuypers FA, Styles L, Vichinsky EP, Foote D, Rosenfeld H. Pulmonary hypertension in thalassemia: association with platelet activation and hypercoagulable state. Am J Hematol. 2006; 81: 670–675. [DOI] [PubMed] [Google Scholar]
- 43. Villagra J, Shiva S, Hunter LA, Machado RF, Gladwin MT, Kato GJ Platelet activation in patients with sickle disease, hemolysis‐associated pulmonary hypertension, and nitric oxide scavenging by cell‐free hemoglobin. Blood 2007; 110: 2166–2172. [DOI] [PMC free article] [PubMed] [Google Scholar]


