Abstract
Purpose.
To test the feasibility of covalent loading of daunorubicin into oxidized porous silicon (OPS) and to evaluate the ocular properties of sustained delivery of daunorubicin in this system.
Methods.
Porous silicon was heat oxidized and chemically functionalized so that the functional linker on the surface was covalently bonded with daunorubicin. The drug loading rate was determined by thermogravimetric analysis. Release of daunorubicin was confirmed in PBS and excised rabbit vitreous by mass spectrometry. Daunorubicin-loaded OPS particles (3 mg) were intravitreally injected into six rabbits, and ocular properties were evaluated through ophthalmic examinations and histology during a 3-month study. The same OPS was loaded with daunorubicin using physical adsorption and was evaluated similarly as a control for the covalent loading.
Results.
In the case of covalent loading, 67 ± 10 μg daunorubicin was loaded into each milligram of the particles while 27 ± 10 μg/mg particles were loaded by physical adsorption. Rapid release of daunorubicin was observed in both PBS and excised vitreous (∼75% and ∼18%) from the physical adsorption loading, while less than 1% was released from the covalently loaded particles. Following intravitreal injection, the covalently loaded particles demonstrated a sustained degradation of OPS with drug release for 3 months without evidence of toxicity; physical adsorption loading revealed a complete release within 2 weeks and localized retinal toxicity due to high daunorubicin concentration.
Conclusions.
OPS with covalently loaded daunorubicin demonstrated sustained intravitreal drug release without ocular toxicity, which may be useful to inhibit unwanted intraocular proliferation.
Oxidized porous silicon can function as an efficient sustained intravitreal delivery system for daunorubicin; this system is novel and may be very useful for treating unwanted vitreoretinal proliferation.
Introduction
Proliferative vitreoretinopathy (PVR) is the most frequent cause of failure of retinal detachment surgery.1 PVR could be described as the uncontrolled growth of physiologically altered cells, including transformed retinal pigment epithelium (RPE) cells and glial cells at the vitreoretinal interface, leading to the formation of contractile membranes causing tractional retinal detachment. Inhibition of RPE proliferation by chemotherapeutic agents remains a primary target of PVR prevention.2–4 Daunorubicin is reported to be a very potent cell growth inhibitory agent in in vitro studies,4 although it has a short intravitreal half-life (131 minutes) and narrow therapeutic range.5 Daunorubicin has also been shown to be effective for treatment of experimental PVR.6,7 However, clinical studies have shown only a moderate effect following an intraoperative infusion or intravitreal injection.8,9 A large multicenter randomized study showed that the intraoperative application of daunorubicin (7.5 μg/mL infusion for 10 minutes) is safe, helps reduce the number of reoperations, and increases the time until first vitreoretinal reoperation.8 A single intravitreal injection of daunorubicin (5 μg) before the conclusion of vitrectomy has also been shown to yield higher attachment.9 These studies support the use of daunorubicin and illustrate the need for a sustainable drug delivery system to overcome the short therapeutic window that results from intraoperative infusion or intravitreal injection.
We previously reported the possibility of using porous silicon photonic crystals as an intraocular drug delivery system.10 Subsequently we demonstrated a chemical reaction to load daunorubicin covalently via a linker grafted to the inner pore walls of porous silicon particles by a hydrosilylation reaction. Both in vitro and in vivo studies showed successful drug loading11,12 and no ocular toxicity in vivo after a 6-month safety study. However, the particle formulation was found to induce chemical degradation of the daunorubicin payload, which was attributed to the presence of residual Si-H species and elemental silicon in the particles.11 We hypothesized that elemental Si in the skeleton of the porous nanostructure and Si-H species on the pore wall surfaces acted as reducing agents for daunorubicin, and this redox reaction led to degradation of the drug either before or after its release from the nanostructure. In a subsequent study,13 we demonstrated that partial or complete oxidation of the porous silicon nanostructure prior to drug attachment mitigated this deleterious reaction, with no drug degradation observed on material that had been completely oxidized. The high-temperature (800°C) conditions used for complete oxidation of porous silicon (yielding porous silica, SiO2) are not compatible with the linker chemistry. However, a silanol-based linker (3-aminopropyltrimethoxysilane) can be attached postoxidation, which can then be converted to a carboxylic acid functionality (via treatment with succinic anhydride) that allows covalent attachment of daunorubicin via N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC) coupling chemistry.13
In the present study we characterized the ocular properties and safety of the oxidized and silanized porous silicon particles covalently loaded with daunorubicin, as well as those of the oxidized porous silicon microparticles loaded with daunorubicin by physical adsorption following intravitreal injection, in order to identify an optimal formulation for safe and effective long-lasting ocular delivery of this potent antiproliferation agent.
Materials and Methods
Synthesis of Porous Silicon Microparticles
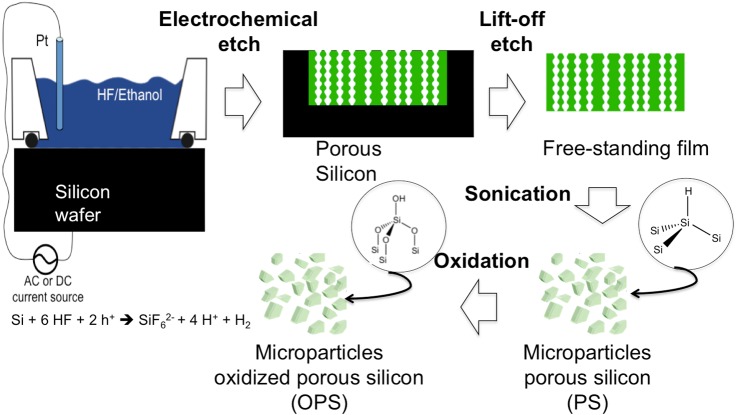
Porous silicon microparticles were prepared by electrochemical etch of highly doped, (100)-oriented p-type silicon wafers (boron-doped, 0.99 mΩ·cm resistivity; Siltronix Inc., Archamps, France) in a 3:1 solution of 48% aqueous hydrofluoric acid to ethanol (Thermo Fisher Scientific, Pittsburg, PA). A silicon wafer with an exposed area of 8.04 cm2 was contacted on the backside with a strip of aluminum foil and mounted in a Teflon etching cell with a platinum counter-electrode. The wafer was etched at a constant current density of 90.2 mA/cm2 for 300 seconds. The resulting porous layer was then lifted off by electropolishing in a 1:29 solution of 48% aqueous hydrofluoric acid to ethanol (Thermo Fisher Scientific) for 120 seconds at a current density of 6.2 mA/cm2. The etching and electropolishing procedure was repeated 20 times per wafer. The particles were harvested every four etches, and the resulting porous layers were ultrasonicated (model FS5 dual-action ultrasonic cleaner; Thermo Fisher Scientific) in ethanol for 30 minutes to form the microparticles. The porous silicon microparticles obtained showed an average particle size of 30 × 46 × 15 μm and pore size of 15 nm as measured from the scanning electron microscopic (SEM) images using Adobe Photoshop software (Adobe Photoshop CS6 for Mac; Adobe Systems Inc., San Jose, CA).14 A schematic diagram of the setup used for synthesis of the porous silicon microparticles is shown in Figure 1.
Figure 1. .
Schematic diagram of the synthesis of porous silicon microparticles. The electrochemical etch cell and apparatus used, as well as the Si electro corrosion half-reaction responsible for porous Si formation, is shown. The synthesis of porous Si involves the electrochemical corrosion of a single crystal wafer. In the presence of an electrolyte containing hydrofluoric acid, electrochemical corrosion removes Si in the form of SiF62−, generating myriads of nanometer-scale pores. Pores nucleate uniformly across the surface of the Si wafer, and they propagate preferentially in the <100> crystallographic direction.15
Surface Modification of Porous Silicon Microparticles
For complete oxidation of porous silicon (Si, PS) to porous silica (SiO2, OPS) particles were placed in a ceramic boat and heated from room temperature to 800°C in a furnace chamber for 1 hour (Thermo Fisher Scientific). The furnace was allowed to cool to room temperature for an additional 3 hours prior to removal of the samples. The resulting porous silica particles (OPS) were then reacted with 2% concentrated hydrochloric acid (HCl; Sigma-Aldrich, St. Louis, MO) in water for 1 hour, rinsed three times with water, and dried. The particles were then vortexed in a 1% 3-aminopropyltrimethoxysilane (APS; Sigma-Aldrich) in ethanol solution for 1 hour for amine functionalization, rinsed with ethanol, and dried. Finally, amine-functionalized OPS were reacted with 0.1 M succinic anhydride (99%; Sigma-Aldrich) in N,N-dimethylformamide (DMF; Sigma-Aldrich) for 16 hours and rinsed with water to obtain a carboxylic acid functional surface (OPS-CO2H). The carboxylic acid group resulted from the ring opening of succinic anhydride through reaction with the amine group on the surface of the particles, as previously described.13
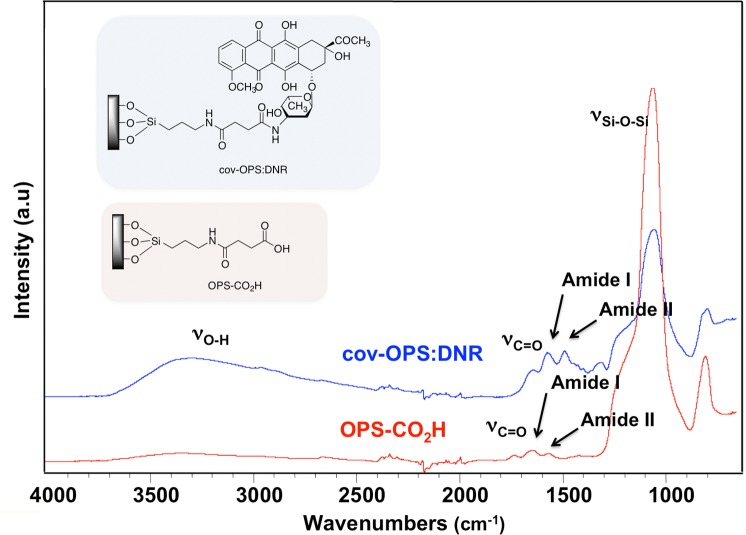
The presence of the functional linker on the OPS-CO2H surface was confirmed by attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy using a Nicolet 6700 FT-IR spectrometer with Smart-ATR attachment (Thermo Fisher Scientific). Characteristic vibrational bands were observed at 1719 cm−1 (v C=O), 1640 cm−1 (amide I), and 1556 cm−1 (amide II) (Fig. 2). Additionally, the shift in the characteristic vibrational bands associated with amide bonds before and after daunorubicin grafting provided evidence of successful drug loading by covalent attachment.
Figure 2. .
Fourier transform infrared (FTIR) spectra of oxidized porous silicon microparticles (OPS-CO2H) before daunorubicin loading and after daunorubicin loading by covalent attachment (cov-OPS:DNR).
Loading of Daunorubicin
Two drug loading strategies were compared in this work: (1) covalent attachment (cov-OPS:DNR), in which the drug was loaded into the linker-modified porous silicon oxide microparticles (OPS-CO2H) by an EDC-mediated coupling reaction, as shown in Figure 3A; and (2) physical adsorption (ads-OPS:DNR), in which the drug was loaded by simple solution infiltration, resulting in physisorption of the drug to the inner pore walls of the oxidized porous silicon microparticles.
Figure 3. .
(A) Schematic illustration of loading of daunorubicin into oxidized porous silicon microparticles by covalent attachment. (B) Oxidized porous silicon microparticles before (B1) and after (B2) daunorubicin loading.
Drug loading by covalent attachment (cov-OPS:DNR). For loading of daunorubicin by covalent attachment, aqueous solutions containing 68 mM EDC (8.45 mg; Sigma-Aldrich) and 6.5 mM N-hydroxysulfosuccinimide (Sulfo-NHS, 0.92 mg; Pierce Biotechnology Inc., Rockford, IL) were added to a dispersion of OPS-CO2H (7 mg) particles in aqueous phosphate-buffered saline at pH 7.4 (PBS, 650 mL; Thermo Fisher Scientific) containing 10% by volume dimethyl sulfoxide (DMSO; Alfa Aesar, Ward Hill, MA). An aqueous solution (200 μL) containing 1 mg/mL daunorubicin hydrochloride (DNR; Tocris Biosciences, Minneapolis, MN) was added to the particle mixture, and the sample was vortexed for 2 hours at room temperature. Particles were pelletized by centrifugation and then carefully rinsed with ethanol five times, until the washing solution was close to transparent, in order to remove unloaded drug and any excess cross linkers. At this point, the particles were observed to have changed from an initial white translucent appearance (Fig. 3B1) to the deep orange color of daunorubicin (Fig. 3B2). A wash with ethanol after synthesis, functionalization, and drug loading was enough to ensure that the particles were sterile prior to in vivo injection.
-
Drug loading by physical adsorption (ads-OPS:DNR). For loading by physical adsorption or infiltration, an aqueous solution (200 μL) containing 1 mg/mL daunorubicin hydrochloride (DNR; Tocris Biosciences) was added to 7 mg porous silicon oxide microparticles (OPS-CO2H), which were suspended in 650 μL 10% DMSO (Alfa Aesar) in PBS. The particles were then vortexed for 2 hours at room temperature and rinsed briefly with water five times until the washing solution was free of orange color, followed by one final ethanol rinse, and then isolated for in vivo use. As with the particles loaded by covalent attachment, the drug-loaded particles were observed to change from an initial white translucent appearance to the deep orange color of daunorubicin after loading of the drug.
After drug loading and ethanol rinse, the particles were dried and stored in a capped vial in the dark, at 4°C in a refrigerator, under an argon protective atmosphere. Daunorubicin-loaded particles were resuspended in balanced salt solution (BSS) immediately before injection.
Thermogravimetric determination of drug loading. The daunorubicin-loaded samples (∼1 mg) were placed in a 90 μL alumina sample cup. Samples were heated at a constant rate of 10 °C/min up to 800°C in nitrogen atmosphere with a purge rate of 10 mL/min using a Q600 simultaneous TGA/DSC apparatus (TA Instruments, Newcastle, DL). As determined by thermogravimetric analysis (TGA), the mass loading of daunorubicin by covalent attachment was 67 ± 10 μg/mg particles and was 27 ± 10 μg/mg particles for physical adsorption loading (Fig. 4).16,17
Figure 4. .
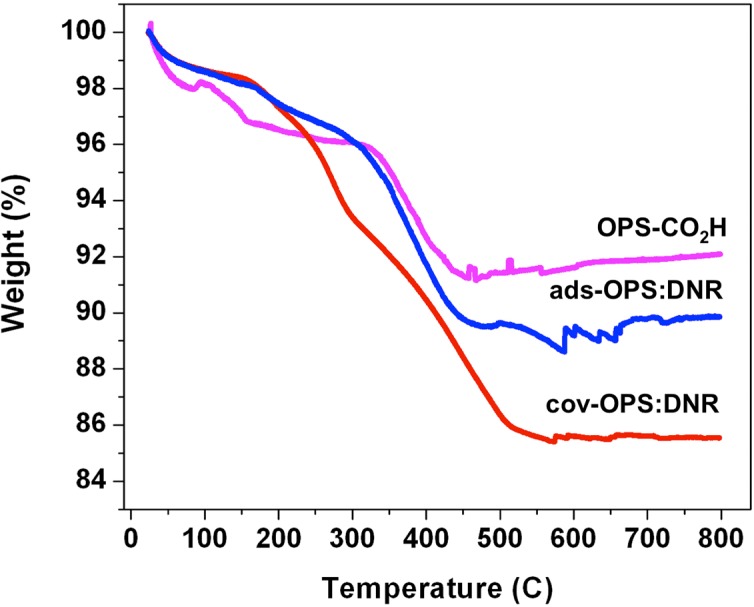
Thermogravimetric analysis (TGA) curves of OPS-CO2H (oxidized porous silicon with carboxylic acid functional surface), ads-OPS:DNR (oxidized porous silicon particles containing physically adsorbed daunorubicin), and cov-OPS:DNR (oxidized porous silicon particles containing daunorubicin covalently attached to the pore walls as described in the text). Weight percent is reported relative to initial weight of sample, prior to heating. The initial decrease in weight at 120°C is attributed to the loss of water from the sample. The decrease in weight observed from 120°C to 600°C is attributed to the organic functionalization and the drug loaded into the particles. The weight loss difference obtained by subtraction between particles before (OPS-CO2H) and after drug loading (ads-OPS:DNR or cov-OPS:DNR) accounts exclusively for the organic matter corresponding to the drug loaded into the particles.
Ex Vivo Drug Release Study in Rabbit Vitreous
It has been reported that daunorubicin degrades in biological fluids.18 In order to test the release profile of daunorubicin from the oxidized porous silicon formulations under conditions mimicking the volume and temperature of rabbit vitreous, 3 mg drug-infiltrated or covalently loaded microparticles were weighted into a 250 μL conical vial, and 100 μL BSS was added. The 100 μL particle suspension was injected into 1.5 mL excised rabbit vitreous in a vial using a 25-gauge needle and then incubated at 37°C. No stirring was applied because stirring can disturb and liquefy the vitreous and cause the drug release profile to deviate from that in physiological condition. At time points 1, 3, 5, 24, 120, and 192 hours, the vial was centrifuged at 5000 rpm for 20 minutes, and 300 μL of the upper portion of the vitreous fluid was extracted and stored at −20°C, protected from light, until analysis. Fresh BSS (300 μL) was added to the original vial in order to maintain a constant volume.
The drug released into fluids from particles containing daunorubicin loaded by covalent attachment (cov-OPS:DNR) was analyzed using positive ion mode electrospray ionization (ESI) liquid chromatography–mass spectrometry (LC-MS/MS) in a 1260 HPLC chromatographic system (Agilent, Palo Alto, CA) coupled with a Thermo LCQdeca-MS spectrometer (Thermo Fisher Scientific). The chromatographic separation was performed on a Shiseido Capcell Pak MG III C18 column (2.0 mm inner diameter × 50 mm length, 3 μm) with a guard column (Phenomenex, Torrance, CA). Mobile phase A was 2.5% by volume methanol (CH3OH, HPLC grade; Sigma-Aldrich) in water with 0.1% formic acid (HCOOH; Sigma-Aldrich). Pure methanol with 0.1% by volume formic acid was used as mobile phase B. The mobile phase was delivered at a rate of 200 μL/min under gradient conditions as follows: 30% phase B to 95% phase B in 10 minutes, followed by 6 minutes with 95% phase B and then back to 30% phase B in 1 minute. An additional time of 8 minutes at 30% phase B was found to be sufficient to equilibrate the column and return the system to the initial conditions for subsequent analysis. The LC-ESI-MS/MS was operated under selected reaction monitoring (SRM) scan mode to detect daunorubicin and the spiked internal standard doxorubicin (DOX; Thermo Fisher Scientific). A LC-ESI-MS/MS run of pure vitreous was used as a blank control. Under these conditions, daunorubicin revealed a single peak at a retention time of 10.9 minutes. The internal standard doxorubicin showed a retention time of 9.9 minutes. Quantitative detection of drug released from particles containing physisorbed daunorubicin (ads-OPS:DNR) was performed by LC with UV detection at 254 nm.
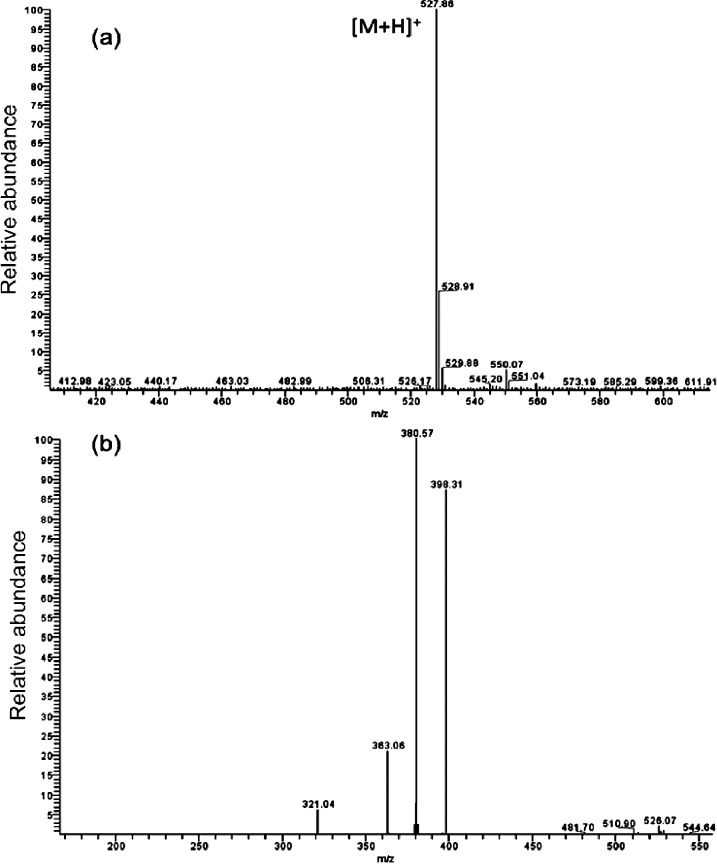
Figure 5a shows the protonated molecular ion peak of daunorubicin at m/z 528, and Figure 5b shows the MS/MS spectrum of this molecular ion peak. The most intense daughter ion peak at m/z 381 was selected for SRM in subsequent daunorubicin detection using LC-MS/MS analysis. A representative LC-ESI-MS/MS chromatogram of daunorubicin released from oxidized porous silicon microparticles loaded with daunorubicin by covalent grafting (cov-OPS:DNR) is displayed in Figure 6. The SRM plots of both DNR and DOX are presented.
Figure 5. .
Typical positive ion mode ESI-MS and ESI-MS/MS spectra of daunorubicin: (a) full scan mode; (b) MS/MS analysis on m/z 528 ([M+H]+, where M = the molecular mass of daunorubicin).
Figure 6. .
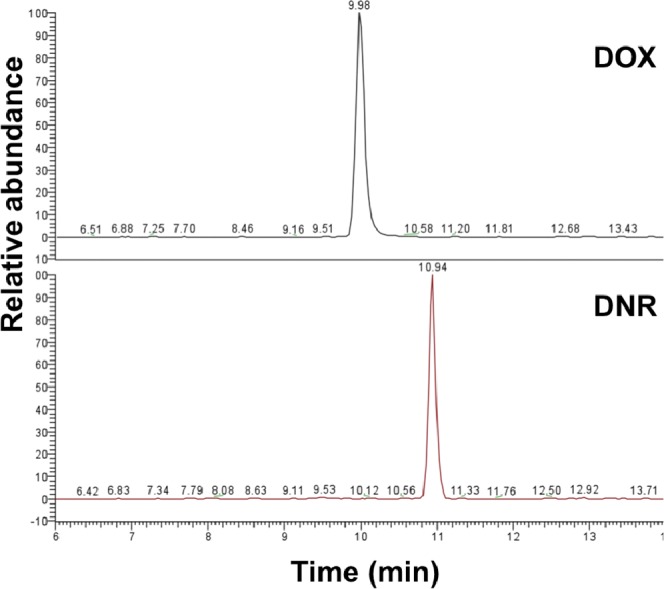
Selected reaction monitoring (SRM) LC-ESI-MS/MS chromatograms of daunorubicin released at 8 days from oxidized porous silicon microparticles loaded with daunorubicin by covalent attachment (cov-OPS:DNR). DOX was used as an internal standard.
Pretreatment of the vitreous fluid with an equal volume of 2-propanol (HPLC grade; Sigma-Aldrich) was used to extract daunorubicin, precipitate proteins, and decrease the viscosity of the system before LC analysis.19 Daunorubicin was quantitated in the vitreous by spiking the samples with an internal standard. Vitreous samples (100 μL) were spiked with doxorubicin hydrochloride (1 ng). The internal standard was calibrated by determining, after extraction of the sample, the ratio of the analyte peak height to the internal standard peak height. The extracts were dried down and resuspended in 100 μL mobile phase prior to injection (20 μL) into the chromatographic column.
In Vivo Ocular Safety and In Vitreous Degradation Profile
Nine New Zealand Red rabbits were used to study the intraocular safety of daunorubicin-loaded OPS particles. Three animals were injected with OPS particles containing physisorbed daunorubicin (ads-OPS:DNR), and the other six were injected with OPS particles containing daunorubicin loaded by cov-OPS:DNR. All animal handling was performed according to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research, and the studies were approved by institutional animal care and use committee of the University of California-San Diego. Baseline intraocular pressure (IOP) and anterior chamber and fundus examinations were documented. One eye of each rabbit was injected intravitreally with 3 mg OPS particles in 100 μL BSS. The fellow eye of each rabbit was injected with 100 μL BSS to serve as the control. The intravitreal injection was administered using the pars plana approach at 2 mm from the limbus following aseptic precautions. After intravitreal injection, the eyes were examined at day 3, at weeks 1, 2, 4, and 8, and at 3 months. Slit-lamp examination, IOP measurement with a handheld tonometer (Tonopen; Medtronic, Jacksonville, FL), and fundus examinations were performed at the indicated time points. Color fundus photographs were taken with a Canon film camera (Canon CF-60U; Canon Inc., Tokyo, Japan) at every exam. Electroretinograms (ERGs) were performed at weeks 2 and 12 as described previously.20 Three animals injected with ads-OPS:DNR particles were sacrificed at week 2 following intravitreal injection. For the group injected with the cov-OPS:DNR particle formulation, three animals were sacrificed at week 2 and three at week 12. After sacrifice, eye globes were enucleated for routine histopathological evaluation. In addition, the sections from cov-OPS:DNR–injected eyes at 2-week and 3-month time points were processed for immunohistochemistry to identify apoptotic cells in retina using the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining kit according to the manufacturer's protocol (TUNEL IHC, ApopTag Peroxidase In Situ; Millipore, Billeria, MA). The sections from the contralateral eye were used as the study controls. At the same time, negative and positive controls were also created and evaluated. For the negative control, sections were treated in the same way as the test sections except for omitting the step of incubation with terminal deoxynucleotidyl transferase (TdT); for the positive control, the sections were treated the same as the test sections except that before primary antibody incubation, the sections were incubated with DNase buffer 1X (Promega, Madison, WI) for 5 minutes at room temperature.
Statistical Analysis
In this study, IOP and ERG were recorded multiple times from the same eyes. In addition, each rabbit received daunorubicin-loaded OPS particles in the right eye, and the left eye received the equivalent volume of BSS as control. Therefore, to take the fellow eye association and the repeated measurements from the same animal into consideration, pooled IOP and ERG parameters were analyzed using a generalized estimating equation (GEE) to compare mean IOPs and ERG parameters between the right eyes and the left eyes.21 All analyses were performed using Statistical Analysis Software version 9.2 (SAS, Cary, NC), and P value smaller than 0.05 was considered significant.
Results
Ex Vivo Release of Daunorubicin
For the OPS particles loaded via simple infiltration (ads-OPS:DNR), very rapid release of daunorubicin was observed in both PBS and excised rabbit vitreous. By the end of the study (significant decomposition of the vitreous occurs at more than 8 days), ∼75% of loaded daunorubicin was released in PBS, and 18% was released in excised rabbit vitreous (Fig. 7, left panel; Figs. 7A, 7B, respectively). In contrast, covalently loaded OPS particles (cov-OPS:DNR) exhibited very slow release. By the end of the study (8 days), less than 1% of the loaded daunorubicin was released into PBS or excised rabbit vitreous (Fig. 7, right panel; Figs. 7C, 7D, respectively). For both drug loading strategies, daunorubicin was released into the excised rabbit vitreous more slowly than into PBS (Fig. 7).
Figure 7. .
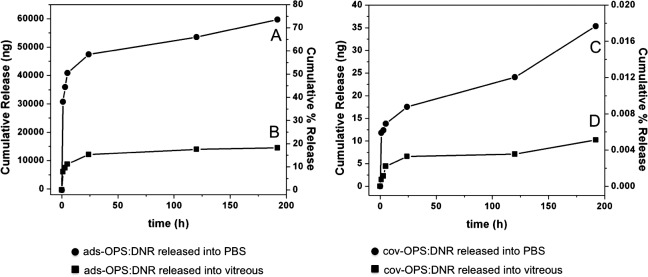
Left panel: Daunorubicin released ex vivo from ads-OPS:DNR (oxidized porous silicon particles containing physically adsorbed daunorubicin) into PBS (A) and into vitreous (B). Right panel: Daunorubicin released ex vivo from cov-OPS:DNR (oxidized porous silicon particles containing daunorubicin covalently attached to the pore walls) into PBS (C) and into vitreous (D). In each graph, the left y-axis represents cumulative amount of drug released over time, and the right y-axis represents cumulative percentage of drug released.
In Vivo Ocular Properties
Following the intravitreal injection, the ads-OPS:DNR particles were observed to aggregate in the vitreous and settle down into the inferior vitreous cavity. The vitreous appeared clear, and no anterior segment or vitreous inflammation was noted. However, the inferior retina showed localized whitening on the second day after injection, and the lesion became atrophic over time with retinal pigment disturbance (Fig. 8). These three rabbits had been sacrificed at the 2-week time point following intravitreal injection. At the time of sacrifice, the eyes had clear media, and the retina appeared normal elsewhere. The animals injected with cov-OPS:DNR particles were followed for a period of 3 months postinjection. Indirect ophthalmoscopy showed that the cov-OPS:DNR particles dispersed into the vitreous within a few days of injection and that the amount of the particles decreased over time (Fig. 9). For the cov-OPS:DNR particles, slit-lamp and fundus examination of all the rabbits showed no signs of toxicity. The average IOP of the study eyes was 18.36 ± 5 versus the control eye value of 18.12 ± 3.8 (P = 0.88, GEE) (Fig. 10).
Figure 8. .
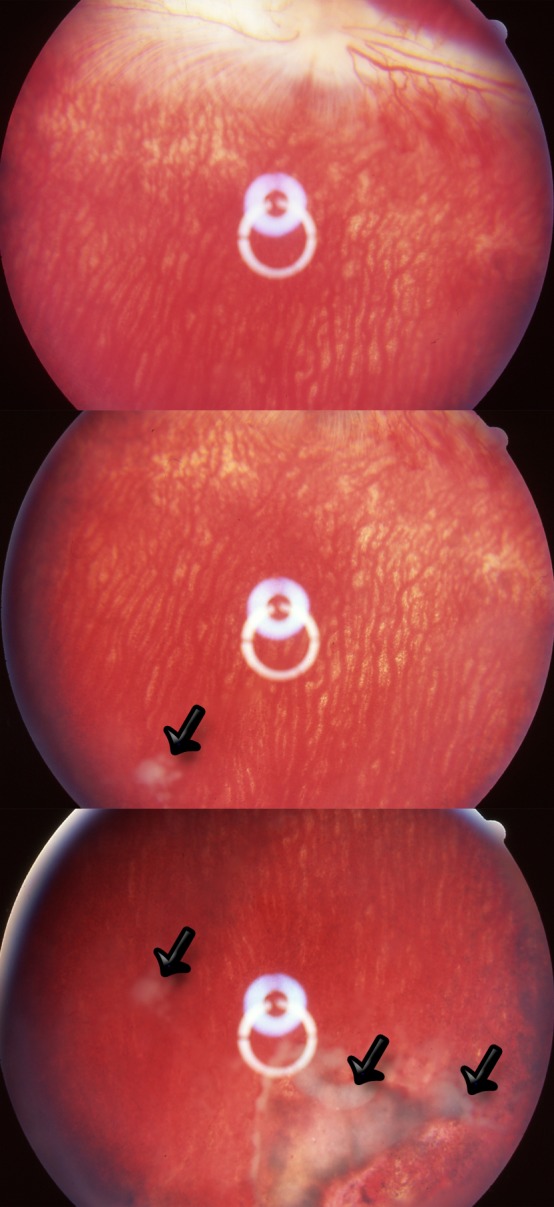
Fundus photographs obtained on day 14 of three rabbits injected with ads-OPS:DNR particles (oxidized porous silicon particles containing physically adsorbed daunorubicin). Localized pigmentation changes and atrophy are apparent in small regions of the inferior retina. The white material was the aggregates of empty porous silicon particles after daunorubicin leached out (arrows).
Figure 9. .
Fundus photographs of rabbit eye at 3 days (3d), 2 weeks (2wk), 5 weeks (5wk), and 9 weeks (9wk) postinjection of cov-OPS:DNR particles. The oxidized porous silicon particles contained daunorubicin covalently attached to the inner pore walls as described in the text. Initially, the OPS particles appear red due to the presence of daunorubicin. The image obtained at 9 weeks shows significantly fewer particles with a less intense red color, indicative of particle degradation and drug release.
Figure 10. .
Mean (n = 3) IOP observed in the cov-OPS:DNR group (pSi_Ox_D) and control group (BSS) as a function of day postinjection. pSi_Ox_D: Oxidized porous silicon particles containing daunorubicin covalently attached to the pore walls as described in the text. No significant difference in IOP between study and control eyes was observed at any of the measured time points.
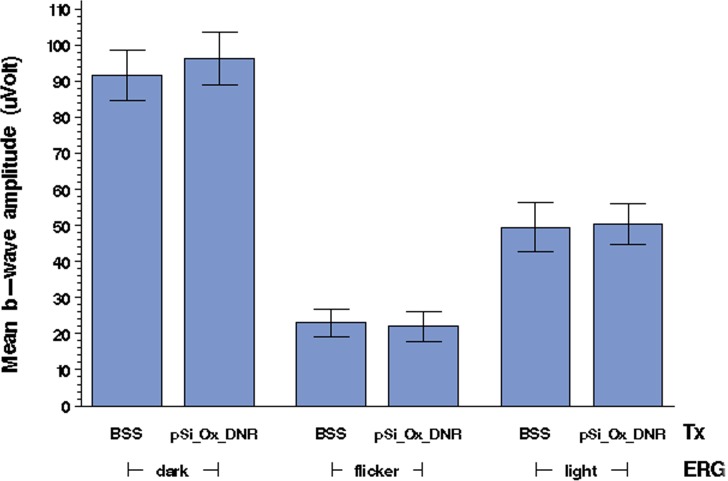
ERG examination revealed no difference in either amplitudes or implicit times for the particle-injected eyes as compared to their control eyes. The b-wave amplitudes obtained from ERG measurements (Fig. 11) were similar between study eyes and their fellow eyes under different types of ERG examination (P = 0.9 for light-adapted ERG, P = 0.86 for flicker ERG, P = 0.64 for dark-adapted ERG).
Figure 11. .
Mean (n = 3) b-wave amplitude values, in microvolts, obtained by ERG measurements on study (pSi_Ox_D, cov-OPS:DNR) and control (BSS) rabbit eyes. Dark-adapted, flicker, and light-adapted ERG measurements, as indicated, were performed on days 14 and 90 postinjection. BSS, control eyes injected with balanced salt solution; cov-OPS:DNR, eyes injected with oxidized porous silicon particles containing daunorubicin, covalently attached to the pore walls as described in the text. No significant difference in b-wave amplitude was observed between the treatment group and the control group.
The half-life of the cov-OPS:DNR particles in vivo, estimated from the fundus photographs and indirect ophthalmoscopy, was 2 weeks, and the retina/vitreous appeared completely normal through the observation course of 3 months (Fig. 9). The cov-OPS:DNR particles in the vitreous differed from the ads-OPS:DNR particles in that the former showed decreasing quantity over time but without much color change, while the latter changed back to white or clear at the 2-week time point following intravitreal injection (Fig. 8), indicating complete leaching out of daunorubicin.
The histopathological examination of the eyes injected with cov-OPS:DNR revealed normal retinal structures as compared to the control eyes. TUNEL staining revealed no discernible apoptosis in any of retinal cell layers compared with the contralateral control eyes (Fig. 12).
Figure 12. .
Detection of apoptotic cells by TUNEL staining from the treated and control eyes harvested 2 weeks following intravitreal injection. (A) A section from a study control eye showing normal retinal structures and no apoptotic cells. (B) A section from a cov-OPS:DNR-injected eye, showing normal retinal structures similar to those in the section in (A) from its contralateral control eye. No apoptotic activity was detected. (C) Negative control section. (D) Positive control section, showing brownish positive staining of cells in all three layers of the retina. The sections were counterstained with hematoxylin. All four images are at ×62.5 magnification.
Discussion
To improve biocompatibility, porous silicon is usually modified by various surface chemistries.17 In addition to surface modifications, the skeleton of the material can be converted from silicon to silicon dioxide or silicon carbide. These skeletal chemistries dramatically change the dissolution and drug loading properties of the nanoporous material.22 The drug payload often imposes its own specific surface chemistry requirements. Daunorubicin is a potent antiproliferation compound4 that has shown to be promising for prevention of PVR.6–9 However, the presence of a quinone-like structure in daunorubicin makes it a redox-active drug, and it can be reduced by surface hydrides (Si-H species) or elemental silicon present in porous silicon. One of the more common methods to functionalize porous silicon, the hydrosilylation reaction, leaves these reactive species in the porous silicon matrix; and hydrosilylated porous silicon has been found to react with and degrade daunorubicin, both in vitro and ex vivo.12,13 In the current study, we oxidized the skeleton of porous silicon to completely remove the deleterious Si-H and elemental silicon species. The thermal oxidation reaction employed has previously been shown to be compatible with daunorubicin, and it does not destroy the porous nanostructure of the material.13
Daunorubicin has a very short vitreous half-life (131 minutes)5 and narrow therapeutic window (1.88 μg/mL to 5.64 μg/mL) following a single intravitreal injection.23,24 Therefore, the therapeutic effect of daunorubicin against PVR is limited without a sustained drug delivery system. The intravitreal bolus injection follows first-order kinetics, and drug level quickly drops below the therapeutic level. We hypothesized that with a sustainable delivery system, daunorubicin might provide a significantly better antiproliferation effect with a broadened therapeutic window by lowering the minimum concentration needed for efficacy. Indeed, even a subdivided administration of a given dose of daunorubicin has been shown to be more effective in preventing experimental PVR than a single dose of the same quantity of drug.7 This improvement in efficacy was likely due to the extended therapeutic duration afforded by the subsequent injection.
In this investigation, we studied a particle formulation in which daunorubicin is loaded by covalent attachment to an oxidized porous silicon skeleton (cov-OPS:DNR). As a comparison, we also studied a formulation in which the drug is simply adsorbed to the inner pore walls of an oxidized porous silicon surface (ads-OPS:DNR). This infiltration-loaded formulation displayed a much larger rate of release than the covalent formulation, with 75% of infiltrated daunorubicin released within 8 days. By contrast, less than 1% of the covalently loaded daunorubicin was released during the same period. This difference was also clearly reflected in the in vivo study; an intravitreal injection of 3 mg infiltration-loaded particles caused obvious localized retinal toxicity, while an equivalent intravitreal injection of covalently loaded particles demonstrated a completely safe profile according to IOP, ERG, and histology measurements. It should be noted that both particle types are expected to reduce the retinal exposure to daunorubicin; the quantity of daunorubicin injected in either formulation was sufficiently large that an equivalent intravitreal injection of the free drug would have resulted in extensive toxicity in the rabbit.23,24 In addition, daunorubicin displays strong cardiac toxicity in systemic circulation. Many intravitreally administered drugs enter systemic circulation following a bolus intravitreal injection.25–28 The high aqueous solubility of daunorubicin (39.2 μg/mL) (provided in the public domain by DrugBank, http://www.drugbank.ca/drugs/DB00694) allows it to enter systemic circulation quite readily, where it could lead to cardiac toxicity. The advantage of a slow-releasing covalent delivery vehicle such as the porous silicon formulation studied here is that it provides a means of introducing a large quantity of drug in a single injection while limiting the steady-state drug concentration below the local and systemic toxic limits.
We, along with others, have shown that daunorubicin is a much more potent antiproliferation agent than the benchmark 5-Fluororacil (5-FU).4 Endothelial cells are extremely sensitive to daunorubicin, and the minimum effective concentration can be as low as 0.001 μM, which is 104 more potent than 5-FU.4 Therefore daunorubicin may have a strong ability to inhibit angiogenesis, which is a key factor in massive proliferation.
The current in vivo study of the cov-OPS:DNR formulation showed clear vitreous and normal retina through 3 months. The cov-OPS:DNR particles were gradually degraded and disappeared from the vitreous. From the photographic recordings and visual observations, the elimination half-life of the cov-OPS:DNR particles from the vitreous is ∼2 weeks. Due to the covalent means of loading the drug, cov-OPS:DNR particles degrade simultaneously with daunorubicin release, which was substantiated by observing the color change of the cov-OPS:DNR formulation during the course of the in vivo study. The red to red-orange color of the particles is due to the spectral absorbance of daunorubicin, as the OPS particles themselves are colorless. The cov-OPS:DNR particles gradually disappeared over time without much loss in color. These data are consistent with the method used to load the drug; the covalent linker attaching the drug to the porous skeleton in the cov-OPS:DNR formulation is stable, and the drug is expected to be released only as the particle degrades and dissolves. By contrast, the ads-OPS:DNR formulation showed a similar red color immediately after injection, but the color faded rapidly as the loaded drug diffused out. Two weeks following the intravitreal injection, the ads-OPS:DNR formulation became colorless as it had been before the drug loading. In the in vitro drug release study, both formulations, ads-OPS:DNR and cov-OPS:DNR, displayed larger release rates in PBS than in excised rabbit vitreous under static conditions. This may be due to the higher viscosity of vitreous, which impedes the diffusion of daunorubicin from porous silicon to the free fluid phase of the vitreous. In a dynamic living eye, porous silicon degradation and the drug release could be different. Indeed, 3 mg cov-OPS:DNR was completely cleared from the vitreous within a time window of 3 months. Because the risk of PVR development has a time window, after injury or surgery, of 1 to 4 months,29 the covalent loading method appears to provide a vitreous residence time and a safety profile for daunorubicin that is appropriate for this indication.
To take the advantage of the high potency of daunorubicin and to extend the therapeutic duration, daunorubicin has been formulated into liposomes,6 microspheres,30 and even implants.31 Compared to the porous silicon delivery system reported here, liposomes and microspheres have short shelf lives, and prolonged controlled release of the drugs for weeks or months is difficult. For implants, infection and other complications associated with invasive surgeries to place and remove them are of concern.
In summary, the current study demonstrated a prototype of a safe intravitreal daunorubicin delivery system using oxidized porous silicon particles and a covalent drug loading strategy. Although this daunorubicin delivery system needs to be further evaluated, including its pharmacokinetics and efficacy, the present study demonstrates feasibility of the system for extended drug release (3 months) with no ocular toxicity. It is most likely that the duration of drug release and vitreous drug concentration will vary according to porosity, surface chemistry, pore wall thickness, and size of the porous Si-derived particles. The electrochemical synthesis of the nanomaterial allows these parameters to be easily adjusted for optimization of vitreous drug release and pharmacokinetic profiles.
Footnotes
Supported by NIH EY020617, the National Science Foundation under Grant DMR-1210417, and a postdoctoral grant from Fundacion Alfonso Martin Escudero (AN).
Disclosure: J. Chhablani, None; A. Nieto, None; H. Hou, None; E.C. Wu, None; W.R. Freeman, Spinnaker Biosciences (C); M.J. Sailor, Spinnaker Biosciences (I); L. Cheng, None
References
- 1. Pastor JC. Proliferative vitreoretinopathy: an overview. Surv Ophthalmol. 1998; 43: 3–18 [DOI] [PubMed] [Google Scholar]
- 2. Cheng L, Hostetler K, Valiaeva N, et al. Intravitreal crystalline drug delivery for intraocular proliferation diseases. Invest Ophthalmol Vis Sci. 2010; 51: 474–481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Turgut B, Uyar F, Ustundag B, Celiker U, Akpolat N, Demir T. The impact of tacrolimus on growth factors in experimental proliferative vitreoretinopathy. Retina. 2012; 32: 232–241 [DOI] [PubMed] [Google Scholar]
- 4. Hou J, Li Y, Zhou Z, et al. Antiproliferative property of hexadecyloxypropyl 9-[2-(phosphonomethoxy) ethyl] guanine (HDP-PMEG) for unwanted ocular proliferation. Mol Vis. 2011; 17: 627–637 [PMC free article] [PubMed] [Google Scholar]
- 5. Wiedemann P, Sorgente N, Bekhor C, Patterson R, Tran T, Ryan SJ. Daunomycin in the treatment of experimental proliferative vitreoretinopathy. Effective doses in vitro and in vivo. Invest Ophthalmol Vis Sci. 1985; 26: 719–725 [PubMed] [Google Scholar]
- 6. Shinohara K, Tanaka M, Sakuma T, Kobayashi Y. Efficacy of daunorubicin encapsulated in liposome for the treatment of proliferative vitreoretinopathy. Ophthalmic Surg Lasers Imaging. 2003; 34: 299–305 [PubMed] [Google Scholar]
- 7. Khawly JA, Saloupis P, Hatchell DL, Machemer R. Daunorubicin treatment in a refined experimental model of proliferative vitreoretinopathy. Graefes Arch Clin Exp Ophthalmol. 1991; 229: 464–467 [DOI] [PubMed] [Google Scholar]
- 8. Wiedemann P, Hilgers RD, Bauer P, Heimann K. Porous silicon particles function as an intravitreal delivery system for daunorubicin. Paper presented at: 8th International Conference Porous Silicon Semiconductors Science and Technology; March 2012; Malaga, Spain [Google Scholar]
- 9. Kumar A, Nainiwal S, Choudhary I, Tewari HK, Verma LK. Role of daunorubicin in inhibiting proliferative vitreoretinopathy after retinal detachment surgery. Clin Experiment Ophthalmol. 2002; 30: 348–351 [DOI] [PubMed] [Google Scholar]
- 10. Cheng L, Anglin E, Cunin F, et al. Intravitreal properties of porous silicon photonic crystals: a potential self-reporting intraocular drug-delivery vehicle. Br J Ophthalmol. 2008; 92: 705–711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Wu EC, Andrew JS, Cheng L, Freeman WR, Pearson L, Sailor MJ. Real-time monitoring of sustained drug release using the optical properties of porous silicon photonic crystal particles. Biomaterials. 2011; 32: 1957–1966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Nieto A, Hartmann KI, Wu EC. et al. Porous silicon particles function as an intravitreal delivery system for daunorubicin. Paper presented at: 8th International Conference Porous Silicon Semiconductors Science and Technology; March 2012; Malaga, Spain [Google Scholar]
- 13. Wu EC, Andrew JS, Buyanin A, Kinsella JM, Sailor MJ. Suitability of porous silicon microparticles for the long-term delivery of redox-active therapeutics. Chem Commun. 2011; 47: 5699–5701 [DOI] [PubMed] [Google Scholar]
- 14. Wu EC, Park J-H, Park J, Segal E, Cunin F, Sailor MJ. Oxidation-triggered release of fluorescent molecules or drugs from mesoporous Si microparticles. ACS Nano. 2008; 2: 2401–2409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Park JH, Gu L, von Maltzahn G, Ruoslahti E, Bhatia SN, Sailor MJ. Biodegradable luminescent porous silicon nanoparticles for in vivo applications. Nat Mat. 2009; 8: 331–336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Lehto VP, Vähä-Heikkilä K, Paski J, Salonen J. Use of thermoanalytical methods in quantification of drug load in mesoporous silicon microparticles. J Therm Anal Calorim. 2005; 80: 393–397 [Google Scholar]
- 17. Salonen J, Kaukonen AM, Hirvonen J, Lehto VP. Mesoporous silicon in drug delivery applications. J Pharm Sci. 2008; 97: 632–653 [DOI] [PubMed] [Google Scholar]
- 18. Maniez-Devos DM, Baurain R, Lesne M, Trouet A. Degradation of doxorubicin and daunorubicin in human and rabbit biological fluids. J Pharm Biomed Anal. 1986; 4: 353–365 [DOI] [PubMed] [Google Scholar]
- 19. Marney LC, Laha TJ, Baird GS, Rainey PM, Hoofnagle AN. Isopropanol protein precipitation for the analysis of plasma free metanephrines by liquid chromatography-tandem mass spectrometry. Clin Chem. 2008; 54: 1729–1732 [DOI] [PubMed] [Google Scholar]
- 20. Cheng L, Hostetler KY, Chaidhawangul S, et al. Treatment or prevention of herpes simplex virus retinitis with intravitreally injectable crystalline 1-O-hexadecylpropanediol-3-phospho-ganciclovir. Invest Ophthalmol Vis Sci. 2002; 43: 515–521 [PubMed] [Google Scholar]
- 21. Liang KY, Zeger SL. Longitudinal data-analysis using generalized linear-models. Biometrika. 1986; 73: 13–22 [Google Scholar]
- 22. Anglin EJ, Cheng L, Freeman WR, Sailor MJ. Porous silicon in drug delivery devices and materials. Adv Drug Del Rev. 2008; 60: 1266–1277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wiedemann P, Kirmani M, Santana M, Sorgente N, Ryan SJ. Control of experimental massive periretinal proliferation by daunomycin: dose-response relation. Graefes Arch Clin Exp Ophthalmol. 1983; 220: 233–235 [DOI] [PubMed] [Google Scholar]
- 24. Santana M, Wiedemann P, Kirmani M, et al. Daunomycin in the treatment of experimental proliferative vitreoretinopathy: retinal toxicity of intravitreal daunomycin in the rabbit. Graefes Arch Clin Exp Ophthalmol. 1984; 221: 210–213 [DOI] [PubMed] [Google Scholar]
- 25. Bakri SJ, Snyder MR, Reid JM, Pulido JS, Singh RJ. Pharmacokinetics of intravitreal bevacizumab (Avastin). Ophthalmology. 2007; 114: 855–859 [DOI] [PubMed] [Google Scholar]
- 26. Bakri SJ, Snyder MR, Reid JM, Pulido JS, Ezzat MK, Singh RJ. Pharmacokinetics of intravitreal ranibizumab (Lucentis). Ophthalmology. 2007; 114: 2179–2182 [DOI] [PubMed] [Google Scholar]
- 27. Nan K, Sun S, Li Y, et al. Characterisation of systemic and ocular drug level of triamcinolone acetonide following a single sub-Tenon injection. Br J Ophthalmol. 2010; 94: 654–658 [DOI] [PubMed] [Google Scholar]
- 28. Kovacs K, Wagley S, Quirk MT, et al. Pharmacokinetic study of vitreous and serum concentrations of triamcinolone acetonide after posterior sub-tenon's injection. Am J Ophthalmol. 2012; 153: 939–948 [DOI] [PubMed] [Google Scholar]
- 29. Mietz H, Heimann K. Onset and recurrence of proliferative vitreoretinopathy in various vitreoretinal disease. Br J Ophthalmol. 1995; 79: 874–877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Metaxa AF, Efthimiadou EK, Boukos N, Kordas G. Polysaccharides as a source of advanced materials: Cellulose hollow microspheres for drug delivery in cancer therapy. J Colloid Interface Sci. 2012; 384: 198–206 [DOI] [PubMed] [Google Scholar]
- 31. Rabowsky JH, Dukes AJ, Lee DA, Leong KW. The use of bioerodible polymers and daunorubicin in glaucoma filtration surgery. Ophthalmology. 1996; 103: 800–807 [DOI] [PubMed] [Google Scholar]