Abstract
Background
In adults it is well documented that there are substantial losses to the programme between HIV testing and start of antiretroviral therapy (ART). The magnitude and reasons for loss to follow-up and death between HIV diagnosis and start of ART in children are not well defined.
Methods
We searched the PubMed and EMBASE databases for studies on children followed between HIV diagnosis and start of ART in low-income settings. We examined the proportion of children with a CD4 cell count/percentage after after being diagnosed with HIV infection, the number of treatment-eligible children starting ART and predictors of loss to programme. Data were extracted in duplicate.
Results
Eight studies from sub-Saharan Africa and two studies from Asia with a total of 10,741 children were included. Median age ranged from 2.2 to 6.5 years. Between 78.0 and 97.0% of HIV-infected children subsequently had a CD4 cell count/percentage measured, 63.2 to 90.7% of children with an eligibility assessment met the eligibility criteria for the particular setting and time and 39.5 to 99.4% of the eligible children started ART. Three studies reported an association between low CD4 count/percentage and ART initiation while no association was reported for gender. Only two studies reported on pre-ART mortality and found rates of 13 and 6 per 100 person-years.
Conclusion
Most children who presented for HIV care met eligibility criteria for ART. There is an urgent need for strategies to improve the access to and retention to care of HIV-infected children in resource-limited settings.
Introduction
In 2010 worldwide about 3.4 million children younger than 15 years were HIV infected, of whom over 90% lived in sub-Saharan Africa [1]. In low-income countries, more than half of vertically-infected children die before the age of two years if they remain untreated [2]. Although early antiretroviral therapy (ART) has been shown to dramatically reduce early mortality and progression of HIV [3], the estimated coverage of ART in low and middle income countries is still much lower in children than in adults: in 2010 23% of the children in need of therapy received ART compared to 51% of adults [1]. However, the reasons for poor uptake of HIV testing and low therapy coverage in children are poorly understood.
A major challenge of health care programmes in the context of the rapid scale-up of ART is to retain patients in care after they tested positive for HIV. Retention is particularly poor in patients not yet eligible for ART. A recent systematic review in adults showed that only 59% of HIV positive patients had a CD4 count to determine treatment eligibility and that only 68% of ART eligible patients started ART [4]. Higher pre-ART retention in adults was associated with the availability of a point-of-care CD4 test [5], [6] and a better health status of the patient [7]. There are few data on loss to programme (mortality, loss to follow-up and transfer out) between HIV testing and start of ART in children. Good retention in care in children may be associated with similar factors as in adults but will in addition depend on the caregiver.
We performed a systematic review to estimate the magnitude and reasons for loss to programme between HIV testing and start of ART in HIV infected children in low-income settings.
Materials and Methods
Data sources
We searched the PubMed and EMBASE bibliographic databases on August 9, 2011. We limited the search to English-language publications which reported on patients in low-income settings. We further limited the search to studies published from 2002 onwards because the scale-up of ART in resource-limited settings (as defined by the World Bank classification) happened after 2002 [8], [9]. We used both free text and Medical Subject Headings (MeSH) and used a combination of the following words and their variations: ‘antiretroviral agents’, ‘therapeutic use’, ‘pre treatment’, ‘pre-ART’, ‘prior to treatment’, ‘eligibility’, ‘loss to care’ and ‘loss to follow-up’. We examined the references of all included studies. Further details of the search strategy are shown in the Appendix S1.
Study selection
We included all studies that reported on numbers of children followed between HIV diagnosis and start of ART, including studies that did not cover the entire time period. We excluded studies on adults and on the prevention of mother-to-child transmission (PMTCT). We also excluded qualitative studies, data from clinical trials and reports from national programmes (as there was a risk of duplicated data and lack of detailed information). Articles were excluded if they reported on the same study population and time period as another article that was more complete. Two reviewers (C.M., O.K.) assessed the eligibility of articles and abstracts. Discrepancies were resolved by consensus between the two reviewers.
Data extraction and analysis
We extracted the data of each publication in duplicate using a standardised data extraction sheet. The following information was extracted for each study: inclusion criteria, characteristics of the programme (setting, location, country), characteristics of the children (age, gender, CD4 cell counts or percentages at different time points), eligibility criteria for ART initiation and methods for tracing children lost to follow-up. In addition we extracted the number of children alive or lost to programme (i.e. lost to follow-up, transferred-out or died) during different time periods. The following time points were of interest: HIV testing, CD4 testing or clinical staging with eligibility assessment for ART, becoming eligible for ART and start of ART. We also assessed the number of children staying in pre-ART care and overall losses to programme and mortality before ART initiation or during pre-ART care. Finally we extracted predictors for loss to programme, mortality and loss to follow-up between HIV testing and CD4 testing and between meeting eligibility criteria for ART and ART initiation. We recorded if there was a positive or negative statistically significant association (p<0.05) or if there was no statistically significant association (p≥0.05). Discrepancies were resolved by consensus. Data were entered into an EpiData database (version 3.1). We calculated the percentage of children completing each time step and displayed the results as forest plots. Data were analysed using STATA version 11.2 (StataCorp, Texas, USA).
Results
Study characteristics
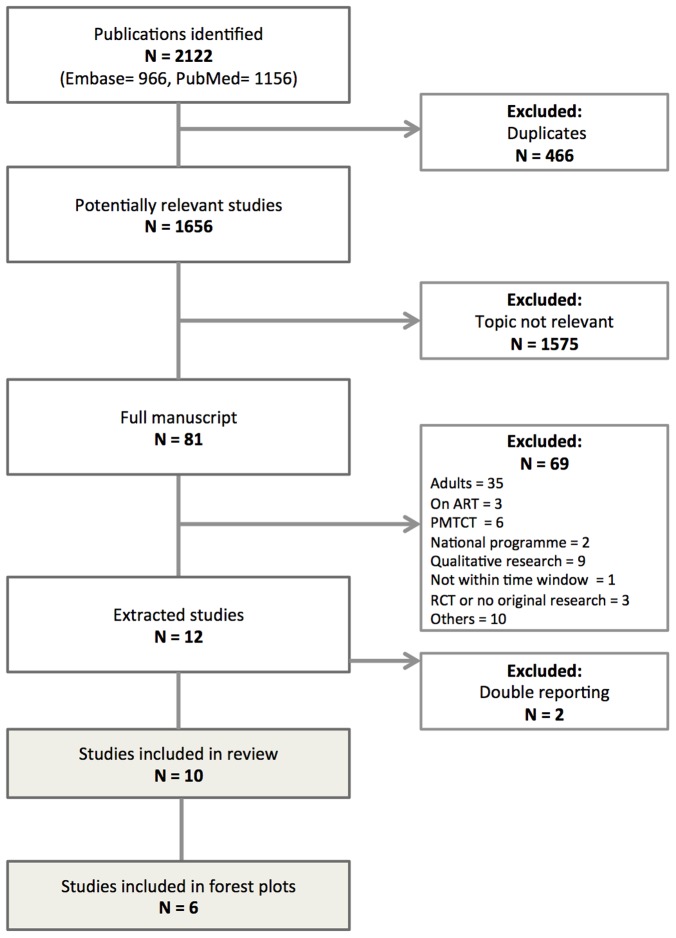
We identified 1,656 potentially relevant articles ( Figure 1 ). Ten studies were included in the systematic review ( Table 1 ): eight studies from sub-Saharan Africa (one study each from Côte d'Ivoire, the Democratic Republic of Congo, Ethiopia, Lesotho, Malawi, South Africa, Zambia and Kenya); one study from India; one study from Cambodia. The studies contributed data on 10,741 children and included all children who attended routine clinics during a specified time period, except for one study [10], in which a random sample of children was analyzed. Most of the studies were multi-site (n = 8); two studies were conducted at one site. The majority of studies reported on programmes in urban clinics where patients were seen by medical doctors. Table 1 shows a detailed description of the programmes studied, including the level of care delivery and the funding sources for the study. The median age of the children ranged from 2.2 to 6.5 years. Median or mean CD4 cell count at presentation ranged from 385 to 622 cells/µl, and median CD4 percentage from 14.5% to 16%. A detailed listing of CD4 cell measurement at presentation and corresponding age group in the study can be found in Table 1 .
Figure 1. Identification and selection of studies.
ART: Antiretroviral therapy; RCT: Randomized controlled trial; PMTCT: Prevention of mother to child transmission.
Table 1. Characteristics of studies included in review, first CD4 cell count measurement after HIV diagnosis and mortality rates prior to initiation of antiretroviral therapy (ART).
| First author, year | Location | Setting | Facilities | Funding | Care delivery by | Study period | Nr children | Age (yrs) | HIV diagnosis | Eligibility for ART* | Median baseline CD4** |
| Anaky, 2011 [11] | Cote d'Ivoire | Semi-urban, urban | 17 urban and 2 semi-urban clinics in Abidjan | PEPFAR | trained health care workers | 2004 - 2007 | 1766 | median (IQR):4.5 (1.8 - 8.2) | >18 mths rapid assay,<18 mths RNA PCR | CD4% <25%: age <12mths,CD4% <20%: 12-35mths,CD4% <15%: ≥36mths | A: 487P: 15.6 |
| Berhan, 2011 [10] | Ethiopia | n.r. | 7 referral public hospitals, 2 are located in the capital | n.r. | general practitioner physicians, pediatricians | 2008 - 2009 | 1163 | mean (SD):4.9 (3.2)range: 1 mo – 14 yrs | n.r. | n.r. | n.r. n.r. |
| Edmonds, 2011 [12] | DRC | urban | 2 hospitals in Kinshasa | n.r. | comprehensive HIV care and treatment programme | 2007 - 2010 | 790 | median (IQR):5.9 (2.7 - 9.8) | <18 mths DNA PCR,>18 mths serological testing or HIV viral load | WHO guidelines 2006/2010, national guidelines | P: 15 |
| Feucht, 2007 [19] | South Africa | urban | Regional state hospital, pediatric ART clinic | n.r. | n.r. | 2004 | 276 | mean (range):4.3 (0.1 - 13) | n.r. | South African guidelines 2003 | A: mean: 622P: mean 15.3 |
| Leyenaar, 2010 [13] | Lesotho | urban | Pediatric HIV/AIDS care and treatment facility | Bristol-Myers Squibb, Baylor college | Nurses, social workers, physicians | 2005 - 2007 | 567 | median (range):2.2 (0 - 15.5) | n.r. | National guidelines (based on WHO 2006) | P: mean 15 |
| McGuire, 2010 [14] | Malawi | rural | 1 district hospital, 10 health centers | Médecins sans Frontières | n.r. | 2001 - 2007 | 107 | n.r. | n.r. | n.r. | n.r. |
| Nyandiko, 2009 [17] | Kenya | rural, urban | 1 urban referral clinic, 17 outpatient services | USAID-AMPATH | Paediatricians, medical and clinical officers | 2002 - 2008 | 4017 | median (range):4.5 (0 - 14.2) | <18 mths DNA PCR,>18 mths 2 parallel ELISAs | CD4% <15%: <6 yrs,CD4 <200 cells/µl: >6 yrs | A: 484P: 16.0 |
| Raguenaud, 2009 [16] | Cambodia | rural | 2 hospitals, 1 pediatric clinic, 1 referral hospital | n.r. | Doctor based clinical care, follow up by multidisciplinary team | 2002 - 2008 | 1168 | n.r. | <18 months RT PCR since 2006 | CD4% <15%: 36-59mths,CD4% <20%: 12-35mths,CD4 <200 cells/µl: ≥5 yrs WHO stage 3/ 4 | A: 410P: 14.5 |
| Seth, 2011 [18] *** | India | urban | 1 tertiary teaching hospital. New Dehli | n.r. | n.r. | 2006 - 2010 | 24 | n.r. | <18 months DNA PCR,>18 months reactive HIV serology | n.r. | n.r. |
| Sutcliffe, 2010 [15] | Zambia | rural, urban | 1 urban clinic: in a low income community in Lusaka, 2 rural clinics | Rural hospitals (churches, urban facility (Ministry of Health) | n.r. | 2004 - 2008 | 863 | median (IQR):urban: 6.5 (3.2 - 9.9),rural: 3.4 (1.8 - 7.4) | n.r. | WHO 2003/2006, national guidelines | A: urban: 385, rural: 572;P: n.r. |
n.r. not reported.
-
WHO 2003 guidelines [33]: all children if WHO paediatric stage III.WHO paediatric stage I (only when CD4 count available) or paediatric stage II:<18 months: CD4 percentage <20%.≥18 months: CD4 percentage <15%.
-
WHO 2006 guidelines [20]: all children if WHO stage 3 or 4 (there are specific rules for WHO stage 3 in case of co-infections).WHO stage 1 or 2 (total lymphocyte counts are used in sites where CD4 values cannot be determined):<1 year: CD4 percentage <25% or absolute CD4 cell count <1500 cells/µl.1 to <3 years: CD4 percentage <20% or absolute CD4 cell count <750 cells/µl.3 to <5 years: CD4 percentage <15% or absolute CD4 cell count <350 cells/µl.≥5 years: CD4 percentage <15% or absolute CD4 cell count <200 cells/µl.
-
• WHO 2010 guidelines [21]: all children if <2 years or in WHO stage 3 or 4.WHO stage 1 or 2:2 to <5 years: CD4 percentage ≤25% or absolute CD4 cell count ≤750 cells/µl.≥5 years: ≤350 cells/µl.
Absolute CD4 count A, percentage CD4 P.
Of 162 HIV exposed children, all 24 children who were diagnosed HIV positive were included.
Mortality, loss to follow-up and transfer out
Deaths among patients lost to follow-up were ascertained in 5 studies by phone calls or home visits. Two studies reported on pre-ART mortality and found rates of 13.0 [11] and 6.0 [12] per 100 person-years (Table S1). A loss to follow-up rate and transfer-out rate before ART initiation were reported by one study [11] and were 50.3 and 2.9 per 100 person-years respectively. Four studies reported on percentages of children who died, were lost to follow-up/defaulted or transferred out before starting ART [13]–[16], and one study reported the proportion of patients lost to follow-up [17]. A small study of 24 children reported on the proportion of children who died, with no children lost to follow-up [18]. The estimates ranged between 3.2% and 45.8% for mortality, 0.0% and 37% for loss to follow-up and 3.4% and 4.6% for transfer out (Table S1). Since the person-time at risk was not reported these percentages are difficult to interpret.
Pre ART cascade
Studies reporting at least one of the three steps (HIV diagnosis to CD4 cell determination, ART eligibility assessment to meeting eligibility criteria, meeting ART eligibility criteria to ART start) were included in this analysis.
From HIV diagnosis to CD4 cell determination
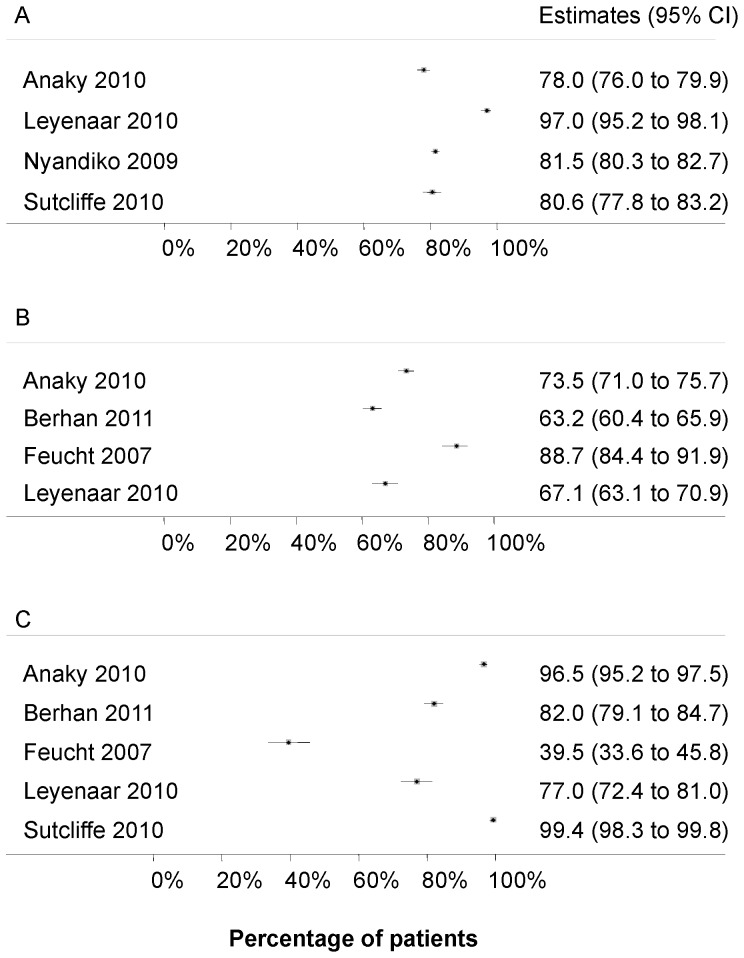
Four studies [11], [13], [15], [17] reported on the step from HIV diagnosis to CD4 cell measurement. The percentage of children with a CD4 cell count ranged from 78.0% to 97.0% ( Figure 2 , panel A).
Figure 2. Forest plots – time from HIV diagnosis to start of antiretroviral therapy (ART).
A): Percentage of HIV positive children with a CD4 cell count/percentage. B): Percentage of children with an eligibility assessement who meet eligibility criteria for ART. C): Percentage of ART eligible children starting ART.
From assessment of eligibility to start of ART
Four studies [10], [11], [13], [19] reported on the period from assessment of eligibility to start of ART. The percentage of children diagnosed with HIV who had a CD4 cell count/percentage measured and were eligible for ART ranged from 63.2% to 88.7% ( Figure 2 , panel B). Five studies [10], [11], [13], [15], [19] reported on the number of children starting ART after meeting eligibility criteria. Estimates ranged from 39.5% to 99.4% ( Figure 2 , panel C).
Predictors of mortality, loss to follow-up and ART initiation
Three studies reported on predictors for starting ART while no study reported on predictors for determining a CD4 cell count/percentage or of mortality. A low CD4 cell count was a strong predictor for ART initiation in all three studies [10], [12], [15]. Two studies reported no association between gender and ART initiation. Sutcliffe et al [15] reported that a long distance to the clinic was associated with loss to follow-up (in particular in children living in rural areas) and Edmonds et al [12] found that advanced clinical stage was associated with ART initiation.
Discussion
Our systematic review included over 10,000 children from 10 low-income countries. In general, few data were available on the different outcomes and heterogeneity across studies was substantial. Our analysis showed that in low-income settings, 78% and 97% of HIV-infected children had a CD4 count/percentage measured and that the large majority of these patients met ART eligibility criteria at presentation. Among those who were assessed for ART eligibility, 63% to 91% of the children were already eligible for therapy, and of the eligible children 40% to 99% initiated ART. According to the WHO 2006 guidelines [20], most children in WHO stage 3 (with few exceptions) and all children in WHO stage 4 are eligible for ART irrespective of CD4 count or percentage, and children in WHO stage 1 and 2 are eligible if the CD4 percentage or absolute CD4 cell count is below an age-specific threshold. In the 2010 revision of the guidelines, WHO recommended that all children aged <2 years should start ART irrespective of the CD4 count, percentage or clinical stage [21]. Different eligibility criteria will influence the number of people deemed eligible for ART initiation while for children in advanced clinical stage it was not necessary to measure CD4 cells. Apart from two studies [12], [18] the follow-up ended before the new guidelines were published. Our study could thus not determine the impact of the change in guidelines on retention in care. Furthermore, operational and financial barriers were the source of differences between implementations of the new ART guidelines across countries. During the study period other major operational changes were made. This includes, for example scale-up of access to PCR HIV testing for children <18 months and use of paediatric syrups instead of soluble fixed dose combinations. This further limited comparison of treatment uptake overall, and for specific age groups and time periods in particular.
This systematic review has several other limitations: 1) The search strategy was limited to English-language publications from only two databases. 2) The generalizability of our findings was influenced by the geographic limitations of the search and the small number of studies. 3) Many studies did not explicitly report if the CD4 cell count used to determine treatment eligibility was the first measured CD4 cell count or if the measurement was taken during pre ART care. 4) The studies did not limit the analysis of predictors for ART initiation to ART eligible children only. 5) Only few studies reported on the outcomes of children not retained in care, as these were not traced systematically in most studies. 6) Finally, it was not always possible to distinguish between overall loss to programme and loss to follow-up if mortality and transfer out was not reported. No study reported on all outcomes and on predictors influencing these outcomes, which made it impossible to assess exactly what happened to these children and why they were lost from follow-up.
Presentation for HIV testing and treatment at a late stage of disease has been shown to increase mortality both before and after ART initiation [22]–[24]. It also increases the risk of developing infectious and non-infectious diseases in HIV-infected children [25]. In this study, we found that the large majority of children were eligible for ART at first presentation. This finding reflects the general failure of health systems to diagnose paediatric HIV-infections and enrol these patients into HIV care early in the course of the disease. Limited capacity to perform PCR testing in infants and to retrieve test results remain important barriers to the success of many ART programmes. Also the lack of integration of PMTCT with paediatric and maternal HIV care programmes [26], stigma, and other socio-cultural barriers are major problems. In a previous meta-analysis, we found that 64% of HIV-exposed infants received early infant diagnosis by PCR at around 6 weeks, and 55% were tested between 12–18 months [27].
Detailed reasons for poor uptake of HIV testing and treatment in low income settings remain poorly understood. No studies included in this review differentiated loss to follow-up in children tested by PCR versus those tested with rapid tests. The feasibility and effect of decentralization of paediatric HIV care could also not be assessed. Since studies evaluating PMTCT programme outcomes were excluded from the analysis, one of the reasons for the high proportion of children with advanced clinical disease at presentation could be that the majority of these children were not included in ART programmes after birth.
Several studies, including two systematic reviews [4], [28], have shown that about one third of adult patients who meet ART eligibility criteria never started ART. In the present study we found that the situation is more encouraging for children since 40% to 99% of treatment eligible children started ART. Table 2 compares the present review with the two previous systematic reviews in adults. Most importantly, more treatment-eligible children than adults start ART. In Rosen et al [4], fewer patients were assessed for eligibility but the definition of this second step differed slightly.
Table 2. Comparison of different systematic reviews about linkage to care in adults and children.
| Present study | Rosen et al [4] | Mugglin et al [28] | |
| Population | Children | Adults | Adults |
| Study location | Sub-Saharan Africa, India and Cambodia | Sub-Saharan Africa | Sub-Saharan Africa |
| Separation into mortality, loss to follow-up and transfer out | Yes | No | Yes |
| Assessed predictors for loss to follow-up | Yes | No | Yes |
| Assessed first CD4 cell count/percentage | Yes | No | Yes(only absolute CD4 cell count) |
| Number of studies | 10 | 28 | 29 |
| Total number of patients analyzed | 10,741 | 66,926 | 148,912 |
| Period of review | January 2002 to August 2011 | All up to April 2011 | January 2002 to August 2011 |
| Percent assessed for eligibility * # | range: 78.0 - 97.0% | 59% (35 - 88%) | 78% (71 - 84%) |
| Percent eligible starting ART * | range: 39.5 - 99.4% | 68% (14 - 84%) | 63% (55.71%) |
Percentages with 95% confidence intervals are shown if not stated otherwise
# Rosen: staged and referred for ART or pre-ART care
Mugglin: Provided CD4 sample irrespective or referral to ART or pre-ART care
Not surprisingly low CD4 cell counts and advanced clinical stage of disease were important predictors for starting ART. However, the high proportion of children starting ART in advanced clinical stage could also mean that these children are more likely to access and remain in HIV care whereas healthier children might not be linked to ART programmes or may drop out during follow-up. In rural areas time from diagnosis to ART initiation was longer if the distance to the clinic increased [15]. Structural barriers for retention in HIV care have been described in adults [4] and the same factors also influence clinical outcomes in children.
Our findings underline the need to improve linkage to care and access to ART for HIV-infected children in low-income countries. A wide range of measures and interventions have been proposed to improve clinical outcomes of these patients. These include more widely available point-of-care CD4 cell count testing [5], [6] and removal of barriers to ART in specific populations. The WHO 2010 guidelines that recommend ART for all children <2 years old, regardless of CD4 cell count/percentage [21] are one example. Other approaches to improve retention in PMTCT programmes (e.g. staff training and active defaulter tracing systems [29], [30]) and access to general health care programmes, such as family-centred models [31] have also been proposed. In order to increase the coverage of ART, especially in remote rural settings, HIV testing and care have to be decentralized and brought to the communities [32]. This is particularly important for PCR diagnosis in infants <18 months old.
In conclusion, this systematic review shows that the large majority of children accessing HIV care meet ART eligibility criteria, suggesting that efforts should be made to link children to HIV testing and ART programmes at earlier stages. Pre-ART mortality and loss to follow-up remain important barriers to the improvement of ART coverage in HIV infected children in resource-limited settings. Importantly, data on clinical outcomes and predictors of retention in care during the pre-ART time period are scarce. Future studies should document mortality, loss to follow-up and transfer-out for all pre-ART time periods. HIV testing by PCR in children less than 18 months should be distinguished from other HIV tests. Assessment of eligibility criteria for starting ART should be separated into immunological (absolute and percentage CD4) and clinical criteria and stratified by age. Children lost to programme should be traced and reasons for attrition recorded. Finally, future studies should specifically examine whether universal ART for all children less than two years (irrespective of CD4 cell determination) decreases pre-ART loss to programme in these children.
Supporting Information
Search Terms of Electronic Databases.
(DOCX)
Mortality, loss to follow-up (LTFU) and transfer out before start of antiretroviral therapy in studies included in the systematic review. Rates per 100 person-years (pyrs) are given if reported in the study. Percentages refer to the proportion of patients enrolled in care, irrespective of the follow-up time.
(DOCX)
PRISMA Flow Diagram.
(DOC)
PRISMA Checklist.
(DOC)
Funding Statement
The study was supported by the National Institute of Allergy and Infectious Diseases (NIAID), Grant 5U01-AI069924-05, a PROSPER fellowship grant to O.K. funded by the Swiss National Science Foundation (Grant 32333B_131629) and a PhD student fellowship to J.E. from the Swiss School of Public Health. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.World Health Organization, UNAIDS, UNICEF (2011) Global HIV/AIDS Response. Epidemic update and health sector progress towards Universal Access. Progress Report 2011. Available: http://whqlibdoc.who.int/publications/2011/9789241502986_eng.pdf. Accessed 4 Jul 2012.
- 2. Newell ML, Coovadia H, Cortina-Borja M, Rollins N, Gaillard P, et al. (2004) Mortality of infected and uninfected infants born to HIV-infected mothers in Africa: a pooled analysis. Lancet 364: 1236–1243. [DOI] [PubMed] [Google Scholar]
- 3. Violari A, Cotton MF, Gibb DM, Babiker AG, Steyn J, et al. (2008) Early antiretroviral therapy and mortality among HIV-infected infants. N Engl J Med 359: 2233–2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Rosen S, Fox MP (2011) Retention in HIV care between testing and treatment in sub-Saharan Africa: a systematic review. PLoS Med 8: e1001056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Jani IV, Sitoe NE, Alfai ER, Chongo PL, Quevedo JI, et al. (2011) Effect of point-of-care CD4 cell count tests on retention of patients and rates of antiretroviral therapy initiation in primary health clinics: an observational cohort study. Lancet 378: 1572–1579. [DOI] [PubMed] [Google Scholar]
- 6. Faal M, Naidoo N, Glencross DK, Venter WD, Osih R (2011) Providing immediate CD4 count results at HIV testing improves ART initiation. J Acquir Immune Defic Syndr 58: e54–59. [DOI] [PubMed] [Google Scholar]
- 7. Zachariah R, Tayler-Smith K, Manzi M, Massaguoi M, ven Griensven J, et al. (2011) Retention and attrition during the preparation phase and after start of antiretroviral treatment in Thyolo, Malawi, and Kibera, Kenya: implications for programmes? Trans R Soc Trop Med Hyg 105: 421–430. [DOI] [PubMed] [Google Scholar]
- 8. Gilks CF, Crowley S, Ekpini R, Gove S, Perriens J, et al. (2006) The WHO public-health approach to antiretroviral treatment against HIV in resource-limited settings. Lancet 368: 505–510. [DOI] [PubMed] [Google Scholar]
- 9. Keiser O, Anastos K, Schechter M, Balestre E, Myer L, et al. (2008) Antiretroviral therapy in resource-limited settings 1996 to 2006: patient characteristics, treatment regimens and monitoring in sub-Saharan Africa, Asia and Latin America. Trop Med Int Health 13: 870–879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Berhan Y (2011) Age and CD4 count of vertically HIV-infected children at the time of diagnosis: what are independent predictors for being symptomatic and CD4 counts drop? J Trop Pediatr 57: 14–23. [DOI] [PubMed] [Google Scholar]
- 11. Anaky MF, Duvignac J, Wemin L, Kouakoussui A, Karcher S, et al. (2010) Scaling up antiretroviral therapy for HIV-infected children in Côte d'Ivoire: determinants of survival and loss to programme. Bull World Health Organ 88: 490–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Edmonds A, Yotebieng M, Lusiama J, Matumona Y, Kitetele F, et al. (2011) The effect of highly active antiretroviral therapy on the survival of HIV-infected children in a resource-deprived setting: a cohort study. PLoS Med 8: e1001044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Leyenaar JK, Novosad PM, Ferrer KT, Thahane LK, Mohapi EQ, et al. (2010) Early clinical outcomes in children enrolled in human immunodeficiency virus infection care and treatment in lesotho. Pediatr Infect Dis J 29: 340–345. [DOI] [PubMed] [Google Scholar]
- 14. McGuire M, Munyenyembe T, Szumilin E, Heinzelmann A, Le Paih M, et al. (2010) Vital status of pre-ART and ART patients defaulting from care in rural Malawi. Trop Med Int Health 15: 55–62. [DOI] [PubMed] [Google Scholar]
- 15. Sutcliffe CG, van Dijk JH, Bolton-Moore C, Cotham M, Tambatamba B, et al. (2010) Differences in presentation, treatment initiation, and response among children infected with human immunodeficiency virus in urban and rural Zambia. Pediatr Infect Dis J 29: 849–854. [DOI] [PubMed] [Google Scholar]
- 16. Raguenaud ME, Isaakidis P, Zachariah R, Te V, Soeung S, et al. (2009) Excellent outcomes among HIV+ children on ART, but unacceptably high pre-ART mortality and losses to follow-up: a cohort study from Cambodia. BMC Pediatr 9: 54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Nyandiko WM, Mwangi A, Ayaya SO, Nabakwe EC, Tenge CN, et al. (2009) Characteristics of HIV-infected children seen in western Kenya. East Afr Med J 86: 364–373. [DOI] [PubMed] [Google Scholar]
- 18. Seth A, Chandra J, Gupta R, Kumar P, Aggarwal V, et al. (2012) Outcome of HIV Exposed Infants: Experience of a Regional Pediatric Center for HIV in North India. Indian J Pediatr 79: 188–193. [DOI] [PubMed] [Google Scholar]
- 19. Feucht UD, Kinzer M, Kruger M (2007) Reasons for delay in initiation of antiretroviral therapy in a population of HIV-infected South African children. J Trop Pediatr 53: 398–402. [DOI] [PubMed] [Google Scholar]
- 20.World Health Organization (2006) Antiretroviral therapy of HIV infection in infants and children: Towards universal access. Recommendations for a public health approach. Available: http://www.who.int/hiv/pub/guidelines/paediatric020907.pdf. Accessed 4 July 2012. [PubMed]
- 21.World Health Organization (2010) Antiretroviral therapy for HIV infection in infants and children: Towards universal access. Recommendations for a public health approach: 2010 revision. Available: http://whqlibdoc.who.int/publications/2010/9789241599801_eng.pdf. Accessed 4 July 2012. [PubMed]
- 22. Davies MA, Keiser O, Technau K, Eley B, Rabie H, et al. (2009) Outcomes of the South African national antiretroviral treatment programme for children: The IeDEA southern Africa collaboration. S Afr Med J 99: 730–737. [PMC free article] [PubMed] [Google Scholar]
- 23. KIDS-ART-LINC Collaboration (2008) Low Risk of Death, but Substantial Program Attrition, in Pediatric HIV Treatment Cohorts in Sub-Saharan Africa. J Acquir Immune Defic Syndr 49: 523–531. [DOI] [PubMed] [Google Scholar]
- 24. Togun T, Peterson I, Jaffar S, Oko F, Okomo U, et al. (2011) Pre-treatment mortality and loss-to-follow-up in HIV-1, HIV-2 and HIV-1/HIV-2 dually infected patients eligible for antiretroviral therapy in The Gambia, West Africa. AIDS Res Ther 8: 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Sutcliffe CG, van Dijk JH, Bolton C, Persaud D, Moss WJ (2008) Effectiveness of antiretroviral therapy among HIV-infected children in sub-Saharan Africa. Lancet Infect Dis 8: 477–489. [DOI] [PubMed] [Google Scholar]
- 26. Meyers T, Moultrie H, Naidoo K, Cotton M, Eley B, et al. (2007) Challenges to pediatric HIV care and treatment in South Africa. J Infect Dis 196 Suppl 3: S474–481. [DOI] [PubMed] [Google Scholar]
- 27. Wettstein C, Mugglin C, Egger M, Blaser N, Salazar L, et al. (2012) Missed opportunities to prevent mother-to-child transmission in sub-Saharan Africa. AIDS 26: 2361–2373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Mugglin C, Estill J, Wandeler G, Bender N, Egger M, et al. (2012) Loss to programme between HIV diagnosis and initiation of antiretroviral therapy in sub-Saharan Africa: systematic review and meta-analysis. Trop Med Int Health 2012 epub ahead of print [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Labhardt ND, Manga E, Ndam M, Balo JR, Bischoff A, et al. (2009) Early assessment of the implementation of a national programme for the prevention of mother-to-child transmission of HIV in Cameroon and the effects of staff training: a survey in 70 rural health care facilities. Trop Med Int Health 14: 288–293. [DOI] [PubMed] [Google Scholar]
- 30. Thomson KA, Cheti EO, Reid T (2011) Implementation and outcomes of an active defaulter tracing system for HIV, prevention of mother to child transmission of HIV (PMTCT), and TB patients in Kibera, Nairobi, Kenya. Trans R Soc Trop Med Hyg 105: 320–326. [DOI] [PubMed] [Google Scholar]
- 31. Leeper SC, Montague BT, Friedman JF, Flanigan TP (2010) Lessons learned from family-centred models of treatment for children living with HIV: current approaches and future directions. J Int AIDS Soc 13 Suppl 2: S3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Sweat M, Morin S, Celentano D, Mulawa M, Singh B, et al. (2011) Community-based intervention to increase HIV testing and case detection in people aged 16–32 years in Tanzania, Zimbabwe, and Thailand (NIMH Project Accept, HPTN 043): a randomised study. Lancet Infect Dis 11: 525–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.World Health Organization (2004) Scaling up antiretroviral therapy in resource-limited settings: treatment guidelines for a public health approach. Available: http://www.who.int/hiv/pub/prev_care/en/arvrevision2003en.pdf. Accessed 23 January 2013.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Search Terms of Electronic Databases.
(DOCX)
Mortality, loss to follow-up (LTFU) and transfer out before start of antiretroviral therapy in studies included in the systematic review. Rates per 100 person-years (pyrs) are given if reported in the study. Percentages refer to the proportion of patients enrolled in care, irrespective of the follow-up time.
(DOCX)
PRISMA Flow Diagram.
(DOC)
PRISMA Checklist.
(DOC)