Abstract
Objective
To determine whether starting antidepressant medication at the start of ECT reduces postECT relapse and to determine whether continuation pharmacotherapy with nortriptyline and lithium (NT-Li) differs in efficacy or side effects from continuation pharmacotherapy with venlafaxine and lithium (VEN-Li).
Method
During an acute ECT phase, 319 patients were randomized to treatment with moderate dosage bilateral ECT or high dosage right unilateral ECT. They were also randomized to concurrent treatment with placebo, NT, or VEN. Of 181 patients to meet postECT remission criteria, 122 (67.4%) participated in a second continuation pharmacotherapy phase. Patients earlier randomized to NT or VEN continued on the antidepressant, while patients earlier randomized to placebo were now randomized to NT or VEN. Li was added for all patients who were followed until relapse or 6 months.
Results
Starting an antidepressant medication at the beginning of the ECT course did not impact on the rate or timing of relapse relative to starting pharmacotherapy after ECT completion. The combination of NT-Li did not differ from VEN-Li in any relapse or side effect measure. Older age was strongly associated with lower relapse risk, whereas the type of ECT administered in the acute phase and medication resistance were not predictive. Across sites, 50% of patients relapsed, 33.6% continued in remission 6 months postECT, and 16.4% dropped out.
Conclusions
Starting an antidepressant medication during ECT does not impact relapse and there are concerns about administering Li during an acute ECT course. Nortriptyline and venlafaxine were equally effective in prolonging remission, although relapse rates following ECT are substantial despite intensive pharmacology. As opposed to the usual abrupt cessation of ECT, the impact of an ECT taper should be evaluated.
Relapse is common following successful acute phase treatment with ECT. Naturalistic studies1-4 and randomized controlled trials of alternative continuation therapies5-7 have documented relapse rates of 40% or greater in the first six months following termination of acute phase ECT. Virtually all patients will relapse if no continuation therapy is used, while monotherapy with a tricyclic antidepressant (TCA), nortriptyline (NT), has a modest beneficial effect.5 The combination of NT and lithium (Li) and continuation ECT appear to exert equivalent and pronounced benefit, each reducing the relapse rate to approximately 40-50%.5,6
ECT is the only biological treatment in psychiatry that is abruptly discontinued once found to be effective.8 In the treatment of major depression, the most common strategy has been to use pharmacological interventions as continuation therapy following ECT.9 Relapse following ECT is heavily skewed toward the period immediately following ECT termination. For example, in an earlier masked, randomized trial we conducted, of those who relapsed within 6 months of ECT termination, 67%, 62%, and 89% did so within 8 weeks while receiving placebo, nortriptyline alone, or combined nortriptyline and lithium, respectively.5 This pattern of early relapse may reflect the fact that antidepressant medications usually show a delay in onset of acute therapeutic action, and this delay may also apply to protection from relapse. Thus, in the period immediately following ECT, patients who are just starting pharmacotherapy may be especially vulnerable to relapse. They are exposed to both the abrupt discontinuation of an effective treatment and the introduction of a new form of treatment with a delay in onset of therapeutic action.
This study tested the hypothesis that starting antidepressant pharmacotherapy from the onset of ECT, as opposed to following ECT completion, results in a substantial improvement in relapse rate.8 During the acute ECT phase, patients were randomized to pharmacological treatment with placebo (PL), NT, or venlafaxine (VEN). They were also randomized to receive either high dosage RUL or moderate dosage BL ECT. In a second 6-month, triple-masked, continuation therapy trial, patients treated with placebo during the ECT course were randomized to continuation therapy with NT or VEN, those who received active medication during ECT continued on that medication, and lithium was added in all cases.
The findings regarding acute phase efficacy and side effects have been reported elsewhere.10 This report focuses on the randomized controlled trial of continuation pharmacotherapy following ECT, and addressed two primary questions: (1) Does starting an antidepressant medication prior to ECT reduce the postECT relapse rate relative to starting the antidepressant medication (and lithium) after ECT? (2) How does the efficacy of VEN-Li compare to NT-Li in relapse prevention following ECT? An alternative to NT could be of special utility to patients with a contraindication to treatment with a TCA.
Methods
Study Site and Study Participation
The study was conducted at the Wake Forest University (WF), Western Psychiatric Institute and Clinic (WPIC), and Washington University in St. Louis, MO (WU). The New York State Psychiatric Institute (NYSPI) was the coordinating and monitoring center. Using the Structured Clinical Interview for Axis I DSM-IV Disorders — Patient Edition (with Psychotic Screen),11 patients who entered Phase 1 of the study met the Diagnostic and Statistical Manual IV12 criteria for major depressive episode (unipolar or bipolar). They also had a pretreatment score of 21 or greater on the Hamilton Rating Scale for Depression (HRSD, 24-item),13 and treatment with ECT was indicated. Patients were excluded with a history of schizophrenia, schizoaffective disorder, non-mood disorder psychosis, neurological illness or insult, alcohol or drug abuse within the past year, ECT within the past 6 months, or severe medical illness that markedly increased the risks of ECT. Patients were also excluded with known allergy or medical contraindication to treatment with NT or VEN.
Participants were recruited from the approximately 750 consecutive patients who were clinically referred for ECT at the 3 sites. Over a 4-year period, 340 patients consented to study participation. Of the 21 patients who did not contribute acute Phase 1 outcome data, 17 left the study prior to the start of ECT (diagnostic exclusion identified: n=7; patient withdrew consent: n=7; family opposed to participation: n=3). After starting ECT, 4 additional patients were dropped from the intent-to-treat (ITT) sample due to identification of an exclusion criterion. The ITT sample for Phase 1 outcomes comprised 319 patients.
Patients were classified as Phase 1 remitters if they had at least a 60% reduction in HRSD scores relative to preECT baseline, with a maximum score of 10 both at an assessment within 2 days of ECT discontinuation and reassessment 4-8 days following ECT termination. Patients provided separate informed consent for participation in the acute ECT and continuation pharmacotherapy phases, and capacity to consent was assessed at each time point. The Institutional Review Boards at each enrollment site and NYSPI approved the study.
To enter the randomized continuation trial (Phase 2) patients had to be classified as remitters following ECT, and have no contraindication to treatment with Li. Of the 155 patients who were remitters, 122 (79%) participated in the continuation trial. Ås noted, the criteria for remission required a score of 10 or below on the HRSD as well as a minimum 60% reduction in score relative to preECT baseline. The threshold of 10 may appear lenient relative to some pharmacological studies, but the criteria are standard in ECT research and are based on use of the 24-item HRSD, whereas a threshold of 7 is commonly used when the 17-item HRSD is administered.
Study Design
In Phase 1 of the study,10 patients were randomized to receive either right unilateral (RUL) administered at 6 times the seizure threshold (6×ST) or bilateral (BL) ECT at 1.5×ST. ECT was given three times per week with a standard MECTA Spectrum 5000Q device (MECTA Corp, Tualatin, OR). Patients (N=62) in either ECT group who did not show substantial improvement after 8 or more treatments were crossed over to high dosage (2.5×ST) BL ECT. ECT was continued as long as clinical progress was observed and terminated after no further improvement was observed over at least two treatments.
Patients were also randomized to receive nortriptyline (NT), venlafaxine (VEN), or placebo (PL) starting the afternoon following the first ECT treatment using a “double-dummy” technique to maintain the mask. A standard dose escalation schedule was used, and the goal was to achieve therapeutic blood levels (100-120 ng/ml) of NT or a minimum daily dose of 225 mg of VEN in all patients by the end of the ECT course.
To maintain the mask, the treating psychiatrist prescribed both NT and VEN for each patient. The site pharmacist had access to the randomization code and substituted PL for NT and/or VEN, as needed. The randomization to ECT and pharmacological conditions (6 combinations) was based on permutted blocks, with equal representation within each block of BL and RUL ECT, and a 1.5:1 ratio of PL relative to either NT or VEN. At each site, other than the individuals involved in the administration of ECT (none of whom provided clinical ratings), patients, treatment teams, and outcome assessors were masked to ECT treatment assignment, and, other than the pharmacist, patients and all personnel were masked to pharmacotherapy assignment.
Eligibility for Phase 2 required only that patients be classified as remitters following ECT and all eligible patients were approached for participation. Excessive distance, lack of transportation, and preference to be treated openly by the referring physician were the leading reasons for non-participation. Patients who received NT or VEN in Phase 1 continued on these medications, while patients who had received PL were randomized to NT or VEN. The mask was maintained for whether the patient was treated with NT or VEN and whether patients received active medication or PL in Phase 1. The double-dummy technique was followed for at least 4 weeks, or until patients had been maintained at a steady regimen for at least two weeks. At this point, the medication was converted to one set of masked pills (either NT or VEN). All patients also received open continuation treatment with lithium (Li). Blood samples were obtained at every visit during Phase 2 and dosing of NT was targeted to achieve a steady state blood level of 100-120 ng/ml and dosing of Li was adjusted to achieve a steady state level of 0.5-0.7 mEq/l. The dosing of VEN was targeted for titrating up to 300 mg/d.
Assessments
Information from interviews with patients, family, and health care providers and from medical records was obtained to complete the Antidepressant Treatment History Form (ATHF) to quantify the extent of medication resistance (i.e., number of failed adequate antidepressant trials in the current episode and total number of trials of antidepressant trials).14 Prior to ECT, and twice weekly, prior to crossover ECT, and at ECT termination, a clinical rater (CR) and a study psychiatrist (SP) not involved in ECT administration completed the Hamilton Rating Scale for Depression.13 The CR also completed the Clinical Global Impression (CGI) scales (severity and improvement: CGI-S and CGI-I)15 and the Global Assessment of Function scale (GAF)12 at the same intervals. Patients completed the Beck Depression Inventory-II (BDI)16 at before and following the ECT course.
During the continuation phase patients were followed until relapse or for six months. Patients were evaluated at weekly intervals for the first four weeks, and at two-week intervals for the remaining 20 weeks. They were contacted by phone at weekly intervals between visits. Clinical ratings during the continuation phase were obtained by the same blinded CR and blinded SP that evaluated patients throughout the ECT course. During the continuation trial, a separate blinded SP assessed side effects, vital signs, adjusted medication or PL dosage based on plasma levels reported by NYSPI and side effects. This individual did not complete clinical ratings of symptomatic status.
Time to relapse was the main outcome measure. The criteria for relapse were a mean HRSD score (CR and blinded SP) of at least 16 that was maintained for at least one week (over two consecutive visits) and a mean absolute increase of at least 10 points at two consecutive visits relative to continuation trial baseline. These criteria reflected a clinical worsening such that most clinicians would abandon the current treatment in favor of an alternative. Patients could also meet criteria for relapse if the patient was rated as considerably worse on the CGI by both raters at each of two consecutive visits over at least one week, and the SP documented that was in the patient's clinical interest to exit the protocol based on the emergence of suicidal ideation or intent, psychotic symptoms, hypomania or mania, or significant functional impairment (GAF score < 50).
Side effects were assessed in terms of the frequency of adverse and serious adverse events (AEs and SAEs), and scores on the Udvalg for Kliniske Undersogelser (UKU) Side Effect Rating Scale.17 AEs and SAEs were defined following standard conventions. The UKU scale was completed by a treating SP at the same intervals as HRSD interviews. The dependent measures were average and maximal total scores over the continuation pharmacotherapy trial.
Statistical Methods
The sites were compared in remitter rate and Phase 2 study participation using chi-square analyses. Patients who met remitter criteria following ECT and who did or did not participate in the continuation trial were compared in demographic, clinical, and previous treatment features with t-tests for continuous measures and chi-square analyses for dichotomous variables. Similarly, the four treatment groups in the continuation trial were contrasted in these features using analyses of variance (ANOVAs), with each treatment condition representing a different level of the single main effect. Unless otherwise specified, significant main effects in ANOVAs and analyses of covariance (ANCOVAs) were followed by Tukey's Honest Significant Difference (HSD) post hoc test.
The primary analysis testing the key hypotheses of the continuation trial used survival analysis for right-censored failure-time data. A simultaneous regression model was fit to the relapse-time data using the Weibull distribution.18 Covariates in the regression model were the pharmacological condition in Phase 1 (Drug vs. PL), pharmacological status in Phase 2 (NT-Li vs. VEN-Li), the interaction of these two terms, site (3 levels), number of adequate antidepressant trials in the current episode, HRSD score at the start of the continuation trial, and patient age. To confirm the findings from the parametric survival analysis regarding treatment group differences, nonparametric estimates of the survival distribution function for each group were computed using the Kaplan-Meier method and contrasted with the logrank test (Mantel-Cox).19
A second parametric survival analysis was conducted to explore clinical correlates of relapse. In this analysis, the model included effects of site, HRSD score at continuation trial baseline, age, treatment resistance (total number of antidepressant trials in the current episode), psychosis (psychotic vs. nonpsychotic depression), polarity (unipolar vs. bipolar depression), depression severity (HRSD score at preECT baseline), randomized Phase 1 ECT assignment (6×ST RUL ECT vs. 1.5×ST BL ECT), and total number of ECT administered.
The Phase 2 treatment groups were contrasted in side effects, examining average and maximum UKU scores during the continuation trial. ANCOVAs were conducted on these measures, modeling Phase 1 medication condition (Drug vs. PL) and Phase 2 medication condition (NT-Li vs. VEN-Li) as main effects, the interaction of these two terms, and the main effect of site, and using age as a covariate. Additional ANOVAs examined the effects of Phase 2 clinical outcome on UKU scores, with site and Phase 2 outcome (relapse, dropout, complete without relapse) as main effects.
ANCOVAs were conducted on the average and maximum oral dosages of NT, VEN, and Li and the average and maximum blood levels of NT and Li. For patients not receiving a medication, the oral dosage that was believed to be administered was substituted, as was the dummy blood level (for NT) that was reported. The between-subject factors included Phase 1 and Phase 2 medication conditions, their interaction, and site, with age serving as a covariate. Parametric survival analyses were conducted separately for patients treated with NT-Li and VEN-Li to examine relations between oral dosage, blood levels, and relapse.
Results
Sample Characteristics
Of 319 patients that received at least one treatment with ECT, as described elsewhere,10 181 patients (56.7%) were remitters (Table 1). The remission rate was higher in patients randomized to right unilateral relative to bilateral ECT and in patients randomized to receive NT during the ECT course, and to a lesser extent, VEN, in comparison to placebo. The sites differed in remitter rate, X22 = 12.36, P=0.002 (Table 1). The remission rate was highest at WPIC, lowest at WU, and intermediate at WF.
Table 1. Number of Patients at Each Site who Started ECT, Remitted with ECT, and Entered and Completed the Continuation Trial.
| Site | Started ECT | ECT Remitter | Entered Continuation Trial | Continuation Trial | |
|---|---|---|---|---|---|
|
| |||||
| Dropout | Relapse | ||||
| Wake Forest University | 106 | 61 | 36 | 12 | 12 |
| Washington University | 86 | 36 | 21 | 3 | 14 |
| Western Psychiatric Clinic and Institute | 127 | 84 | 65 | 5 | 35 |
| Total | 319 | 181 | 122 | 20 | 61 |
Of 181 remitters, 122 patients (67.4%) entered the randomized, double-masked, continuation pharmacotherapy trial. The principal reasons for non-participation in the continuation trial were travel limitations and preference to be treated openly by their referring physician. The rate of participation in the continuation trial among remitters differed among the sites, X22 = 7.11, P=0.03 (Table 1), with the participation rate at WPIC (77.4%) higher than at WF (59.0%) and WU (58.3%).
Demographic and clinical characteristics of the continuation trial participants are presented in Table 2. Remitters who did or did not enter the continuation trial were contrasted in the features listed in Table 2. There were no significant differences. The four treatment groups in the continuation trial, defined by whether they received an antidepressant or placebo during ECT and NT-Li or VEN-Li as postECT continuation therapy, were contrasted in these clinical features. For the continuous measures, the four continuation therapy groups differed in years of education, F(3, 118) = 2.94, P=0.04, and preECT BDI score, F(3, 118) = 3.02, P=0.03. Post hoc comparisons (Tukey's HSD) indicated that the group that received placebo during ECT followed by NT-Li continuation therapy had more years of education (15.30 ± 3.31) compared to patients treated with VEN during ECT and followed by VEN-Li (13.38 ± 2.24). Post hoc comparisons revealed no significant pair-wise differences among the groups in preECT BDI scores. In the discrete variables, the four continuation therapy groups differed only in the representation of bipolar disorder, X23 = 8.39, P=0.04. The rates were highest for those who received placebo during ECT followed by NT-Li (30.00%) compared to patients who received VEN during ECT followed by VEN-Li (5.13%). Overall, it appeared that the randomizations were successful in creating groups with comparable demographic and clinical features.
Table 2.
Demographic and Clinical Features of the Total Continuation Trial Sample and for Each of the Four Randomized Treatment Groups.
| Variable | Total Continuation Sample | Phase 1: Placebo Phase 2: NT-Li |
Phase 1: Placebo Phase 2: VEN-Li |
Phase 1: NT Phase 2: NT-Li |
Phase 1: VEN Phase 2: VEN-Li |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||
| n=122 | n=20 | n=24 | n=39 | N=39 | ||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| Age (yr) | 48.90 | 15.01 | 48.30 | 16.76 | 45.50 | 9.86 | 47.08 | 16.79 | 53.13 | 14.38 |
| Gender (% female) | 64.75 | 65.00 | 58.33 | 58.97 | 74.36 | |||||
| Education, yr | 13.92 | 2.68 | 15.30 | 3.31 | 13.29 | 1.71 | 14.13 | 3.00 | 13.38 | 2.24 |
| PreECT HRSD | 29.80 | 5.80 | 30.30 | 5.71 | 28.17 | 5.25 | 31.36 | 6.49 | 28.97 | 5.19 |
| PreECT CGI [severity] | 5.30 | 0.71 | 5.30 | 0.47 | 5.33 | 0.70 | 5.49 | 0.60 | 5.10 | 0.88 |
| PreECT BDI | 37.24 | 10.60 | 39.75 | 8.80 | 34.92 | 10.02 | 40.33 | 10.68 | 34.28 | 10.88 |
| Psychotic (%) | 25.41 | 20.00 | 16.67 | 30.77 | 28.21 | |||||
| Polarity (% bipolar) | 19.67 | 30.00 | 20.83 | 28.21 | 5.13 | |||||
| Episode duration (wks.) § | 38.09 | 34.39 | 28.1 | 23.17 | 36.04 | 34.18 | 39.76 | 35.43 | 43.13 | 38.32 |
| Adequate antidepressant treatment trials, current episode (no.) | 1.21 | 1.29 | 1.45 | 1.00 | 1.54 | 1.69 | 0.95 | 1.05 | 1.15 | 1.33 |
| Total antidepressant treatment trials, current episode (no.) | 5.52 | 3.98 | 4.85 | 2.37 | 7.04 | 6.00 | 5.15 | 3.69 | 5.31 | 3.21 |
| Medication Resistant (%) | 75.41 | 85.00 | 75.00 | 71.79 | 74.36 | |||||
| Total ECT (no.) | 8.27 | 3.46 | 8.20 | 2.95 | 8.58 | 3.55 | 8.59 | 3.41 | 7.79 | 3.77 |
| PostECT HRSD | 5.43 | 2.78 | 6.50 | 2.86 | 5.25 | 2.27 | 5.18 | 2.99 | 5.23 | 2.79 |
| PostECT CGI [severity] | 1.82 | 0.83 | 1.90 | 1.02 | 1.97 | 0.81 | 1.58 | 0.74 | 1.94 | 0.80 |
| PostECT CGI [improvement] | 1.59 | 0.65 | 1.60 | 0.68 | 1.50 | 0.59 | 1.51 | 0.54 | 1.71 | 0.75 |
| PostECT BDI | 9.94 | 8.49 | 11.98 | 8.59 | 8.73 | 5.54 | 11.12 | 10.15 | 8.46 | 8.03 |
NT = Nortriptyline; VEN = Venlafaxine; Li = Lithium Carbonate; HRSD =Hamilton Rating Scale for Depression; CGI = Clinical Global Impression; BDI = Beck Depression Inventory-II.
Adequacy of each medication trial given during the index episode before ECT was evaluated with the Antidepressant Treatment History Form.14 Each trial was rated on a scale ranging from 0 to 5, with a score of 3 the threshold for classification as medication resistant. To be considered an adequate trial, the threshold for sufficient dosage corresponded, for example, to a minimum of 200 mg/d imipramine equivalents for tricyclic antidepressants and 20 mg/d for fluoxetine. The threshold for sufficient duration was a minimum of 4 weeks at or above the threshold for sufficient dosage. To be classified as resistant, patients with psychotic depression had to receive an adequate antidepressant trial and at least 3 weeks of concurrent treatment with an antipsychotic medication, with dosage at least 400 mg/d chlorpromazine equivalents.
An upper limit of 104 weeks was imposed.
Hypothesis Testing
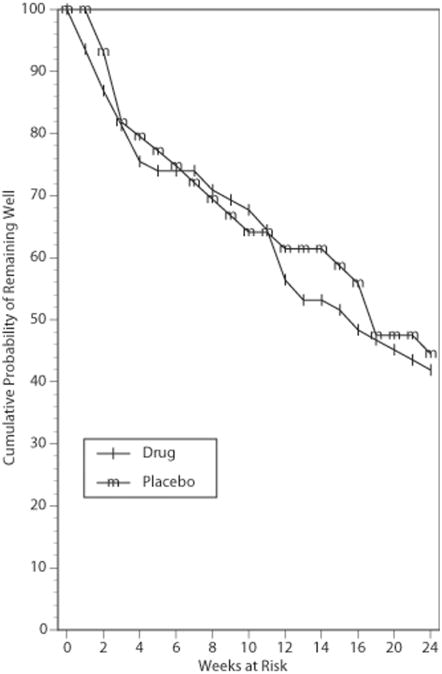
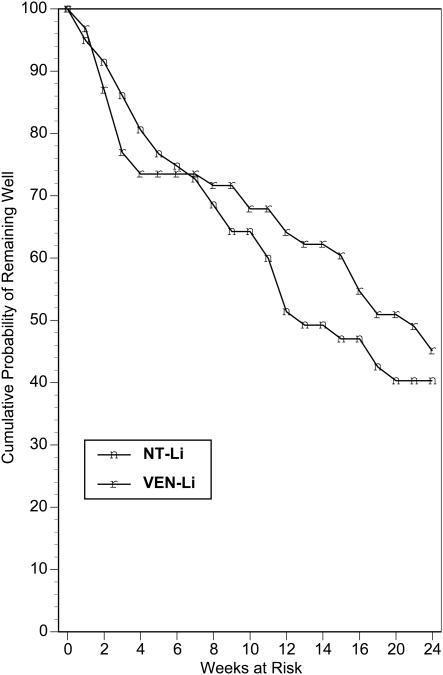
The findings from the parametric survival analysis of the relapse-time data are presented in Table 3. There was no indication that beginning an antidepressant agent at the start of ECT impacted on relapse relative to receiving placebo during the ECT course, X21 = 1.41, P=0.23. The Kaplan Meier survival plot for this comparison is presented in Figure 1. Likewise, in the parametric survival analysis there was no indication that treatment with NT-Li differed in relapse from treatment with VEN-Li, X21 = 0.36, P=0.55. The Kaplan Meier survival plot for this comparison is presented in Figure 2. In the parametric survival analysis there was also no indication of an interaction between the Phase 1 and Phase 2 medication conditions, X21 = 0.01, P=0.93. The Kaplan Meier survival plot representing the 4 groups individually is presented in Figure 3. Nonparametric tests of each these comparisons, as represented in Figures 1-3, all failed to approach significance (all P's > 0.61). Thus, there was no evidence that early start of an antidepressant or use of NT relative to VEN had any impact on relapse.
Table 3. Parametric Survival Analysis on Relapse-Time Data.
| Source | DF | Chi-Square | P |
|---|---|---|---|
| Site | 2 | 6.13 | 0.047 |
| Phase 1 Medication Condition (PL vs. Drug) | 1 | 1.41 | 0.23 |
| Phase 2 Medication Condition (NT-Li vs. VEN-Li) | 1 | 0.36 | 0.55 |
| Phase 1 × Phase 2 Medication Condition | 1 | 0.01 | 0.93 |
| No. Adequate Treatment Trials, Current Episode | 1 | 2.06 | 0.15 |
| HRSD at continuation trial baseline | 1 | 2.62 | 0.11 |
| Age | 1 | 14.90 | 0.0001 |
Figure 1.
Kaplan-Meier estimates of the proportion of patients who remained well during the continuation pharmacotherapy trial for patients randomized to treatment with placebo or antidepressant medication (NT or VEN) during the ECT course.
Figure 2.
Kaplan-Meier estimates of the proportion of patients who remained well during the continuation trial for patients randomized to treatment with nortriptyline and lithium (NT-Li) or venlafaxine and lithium (VEN-Li) as continuation pharmacotherapy.
Figure 3.
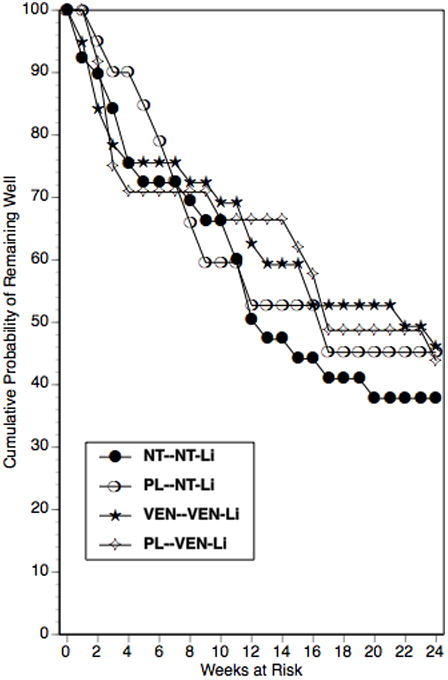
Kaplan-Meier estimates of the proportion of patients who remained well during the continuation trial for patients randomized to the four treatment conditions: placebo (PL) or drug (NT or VEN) during ECT and, during continuation pharmacotherapy, nortriptyline and lithium (NT-Li) or venlafaxine and lithium (VEN-Li) as continuation pharmacotherapy.
Overall the risk of relapse was substantial. Of the 122 patients to enter the trial, 61 (50.0%) relapsed during the six-month follow-up period, 41 (33.6%) completed without relapse, and 20 patients (16.4%) dropped out. There was a narrow range of relapse rates among the four treatment groups. As seen in Figure 3, the probability of remaining well at trial termination ranged from 37.8% (NT during ECT followed by NT-Li) to 46.1% (VEN during ECT followed by VEN-Li).
Correlates of Relapse
As seen in Table 3, the parametric analysis indicated that the sites differed in relapse, X21 = 6.13, P=0.047. The nonparametric test of this effect only yielded a trend, X21 = 5.10, P=0.08. Likelihood of survival was somewhat lower at WU than at the other two sites.
As also seen in Table 3, there was a strong association between patient age and relapse, X21 = 14.90, P<0.0001. Patients who completed without relapse (54.78±14.17) were on average nearly ten years older than patients who relapsed (44.89±14.05), with patients who dropped out intermediate (49.10±16.21). A one-way ANOVA indicated that the three groups differed in age, F(2, 119)=5.75, P=0.004, and Tukey's HSD indicated that patients who relapsed were significantly younger than those who completed.
In previous research, degree of treatment resistance, as quantified by the ATHF, has been a potent predictor of postECT relapse.1,3-5 However, in this study the term in the parametric model representing treatment resistance, the number of adequate failed treatment trials in the current episode, only had a weak, nonsignificant relationship to relapse, X21 = 2.06, P=0.15 (Table 3). It is widely known that adequate treatment trials constitute a relatively small proportion of the total attempts at antidepressant treatment. Indeed, in this study, on average, patients received more than 4.5 times as many attempts at antidepressant treatment (5.52±3.98) compared to the number of adequate trials they received (1.21±1.29) (Table 2). The parametric survival analysis summarized in Table 3 was repeated using the total number of treatment trials as the term representing treatment resistance. The previously observed effects of site and age were essentially unchanged. There was also a strong relationship between total number of antidepressant treatment trials and relapse, X21 = 6.81, P=0.009. No other effects emerged.
A one-way ANOVA conducted on the total number of antidepressant trials contrasting patients who relapsed (6.26±5.00), completed (4.59±2.51), or dropped out (5.20±2.19) yielded only an effect at a trend level, F(2, 119)=2.31, P=0.10. However, a t-test indicated that patients who relapsed had received more antidepressant trials than patients who completed without relapse, t(93.65)=2.33, P<0.03. In all subsequent analyses total number of antidepressant trials was retained in the parametric survival model as the term representing treatment resistance.
A parametric survival analysis was conducted to explore other factors that may be associated with relapse risk. As seen in Table 4, a new model was applied that included terms representing psychosis, unipolar vs. bipolar depression, severity of depression (HRSD score) at preECT baseline, duration of current episode, randomization to RUL vs. BL ECT, and total number of ECT. The effects of site, age, and total number of antidepressant trials remained significant, while the effect of HRSD score at continuation trial baseline approached significance, X21 = 2.88, P=0.09. There were no other effects. In particular, there was no evidence that the distinction between psychotic and nonpsychotic depression, depression severity at preECT baseline, receipt of BL vs. RUL ECT, or total number of ECT were related to risk of relapse.
Table 4. Parametric Survival Analysis of Clinical Predictors of Relapse-Time Data.
| Source | DF | Chi-Square | P |
|---|---|---|---|
| Site | 2 | 9.76 | 0.0078 |
| HRSD at continuation trial baseline | 1 | 3.38 | 0.07 |
| Age | 1 | 11.56 | 0.0007 |
| Total No. of Antidepressant Trials, Current Episode | 1 | 5.52 | 0.02 |
| Psychosis (Yes/No) | 1 | 0.11 | 0.74 |
| Unipolar vs. Bipolar | 1 | 2.47 | 0.12 |
| HRSD at preECT study entry | 1 | 0.02 | 0.88 |
| ECT assignment (BL VS RUL) | 1 | 0.01 | 0.91 |
| Total No. of ECT | 1 | 1.29 | 0.26 |
Side Effects and Adverse Events
ANCOVAs were conducted on the average UKU score during the continuation trial, as well as on the maximum score during the trial. In both cases, there was no main effect of Phase 1 or Phase 2 medication status, or their interaction. Thus, there was no impact on side effect burden whether patients received active medication or placebo in Phase 1 and whether patients received NT-Li or VEN-Li in Phase 2. There were no effects of age in either ANCOVA. However, there was a main effect of site in the ANCOVA on average UKU score, F(2, 107)=5.74, P=0.004, and maximum UKU score, F(2, 107)=3.81, P=0.025. In both cases, post hoc comparisons (Tukey's HSD) indicated that UKU scores were significantly higher at WU than at WPIC, with WF intermediate.
Previously we noted that UKU scores in the total Phase 1 sample decreased markedly from preECT baseline, an effect linked to extent of clinical improvement.10 This was also true in the subsample of 111 patients who had at least one UKU score at preECT baseline, during the ECT course, and during the continuation trial. Average UKU scores declined from 19.90±8.36 at preECT to 10.64±5.01 during ECT, t(110) = 15.40, P<0.0001. In contrast, there was a small but significant rise in average UKU scores during the continuation trial, 12.33±7.17, t(110) = 2.28, P=0.02. Thus, reports of side effects were somewhat lower during the ECT than the continuation pharmacotherapy phase of this study. However, the maximum UKU score during the ECT course, 17.30±7.12 did not differ from the maximum score during the continuation trial, 17.66±7.36, t(110) = 0.45, P=0.66.
ANOVAs were conducted on average and maximum UKU scores during the continuation trial with site and Phase 2 outcome (completed without relapse, relapse, dropout) as between-subject terms. In both analyses, the effect of site was significant, as described above. There was also a significant effect of Phase 2 outcome for both the average UKU score, F(2, 109)=18.26, P<0.0001, and the maximum UKU score, F(2, 109)=14.56, P<0.0001. Post hoc comparisons (Tukey's HSD) indicated that patients who relapsed had substantially higher average and maximum UKU scores than either patients who completed or dropped out (Table 5). Thus, clinical status appeared to exert a powerful effect on UKU scores in both Phase 1 and Phase 2.
Table 5. Average and Maximum UKU Score During the Continuation Trial as a Function of Clinical Outcome.
| Phase 2 Outcome | Average UKU Score | Maximum UKU Score | ||
|---|---|---|---|---|
| Mean | SD | Mean | SD | |
| Completed without relapse (N=40) | 7.73 | 3.83 | 13.53 | 5.35 |
| Relapse (N=59) | 15.69 | 7.28 | 20.93 | 7.04 |
| Dropout (N=15) | 11.76 | 6.82 | 15.80 | 7.40 |
Note. UKU data were not obtained in 8 patients.
Seven patients experienced single adverse events, one patient had two adverse events, one patient had two adverse events and one serious adverse event and one patient had a single serious adverse event. The number of events was too small to examine relationships with treatment conditions or outcome. The 11 adverse events included orthostasis (N=2), falls (N=2), emergence of mania, hypertension, rash, seizure-like attack, nausea, dizziness and slowed movement, and multiple symptoms related to high NT blood levels. The two serious adverse events included a patient with an acute subdural hematoma after a fall that was evacuated in neurosurgery and a suicide attempt by overdose.
Oral Dosage and Medication Levels
ANCOVAs were conducted on the average and maximum oral dosages of NT, VEN, and Li and the average and maximum blood levels of NT and Li. The between-subject terms were Phase 1 and Phase 2 medication conditions, their interaction, and site, and age was the covariate. Table 6 presents the descriptive statistics for patients randomized in Phase 2 to either NT-Li or VEN-Li. Values for patient groups not receiving a medication (e.g., NT values for patients treated with VEN-Li) reflect the oral dosage that was believed to be administered or the dummy blood level reported to the treating physician.
Table 6.
Oral Dosage and Blood Levels of Continuation Pharmacotherapy Medications by Treatment Condition.
| Continuation Pharmacotherapy Group | ||||
|---|---|---|---|---|
| NT-Li | VEN-Li | |||
| Mean | SD | Mean | SD | |
| Average Oral Dosage (mg) | ||||
| NT | 87.63 | 27.78 | 86.51* | 21.18 |
| VEN | 286.74* | 29.60 | 275.20 | 37.42 |
| Li | 686.91 | 219.19 | 664.08 | 242.10 |
| Maximum Oral Dosage (mg) | ||||
| NT | 99.11 | 30.88 | 93.85* | 23.57 |
| VEN | 294.64* | 24.17 | 291.39 | 30.92 |
| Li | 816.96 | 285.92 | 784.43 | 300.49 |
| Average Blood Level | ||||
| NT (ng/ml) | 107.99 | 45.68 | 104.80* | 30.06 |
| Li (mEq/L) | 0.51 | 0.16 | 0.48 | 0.17 |
| Maximum Blood Level | ||||
| NT (ng/ml) | 154.60 | 69.93 | 122.77* | 33.22 |
| Li (mEq/L) | 0.71 | 0.23 | 0.68 | 0.27 |
NT = nortriptyline; VEN = venlafaxine; Li = lithium carbonate.
For patient not receiving a medication (e.g., NT for patients treated with VEN-Li) oral dosage reflects the dosage believed to be administered and blood levels reflect dummy values reported to the treating physician.
In the ANCOVAs there was no effect involving Phase 1 medication condition. Thus, whether patients received a placebo in Phase1 and started an antidepressant in Phase 2 or continued on the same antidepressant in Phase 2 had no impact on the average or maximum oral dosage or blood levels of medication in Phase 2. Similarly, with but one exception, there was no effect involving Phase 2 medication condition. The exception was that patients treated with NT-Li had higher maximum blood levels of NT during the continuation trial than the maximum dummy levels reported for patients treated with VEN-Li, F(1, 109)=14.56, P=0.004 (see Table 6), despite the fact that the two groups were nearly equivalent in average NT levels. This effect was due to the fact that abnormally high NT blood levels (≥ 200 ng/ml) occurred exclusively among patients actually receiving NT (N=12), presumably reflecting slow metabolism of the medication. In hindsight, this threat to maintenance of the blind could have been eliminated by including a small number of very high dummy values in the reports given to the treating physicians. Other than this, there were no effects of the randomized Phase 1 or Phase 2 medication conditions on oral doses and blood levels, strongly supporting the integrity of the study.
There were effects of site in the ANCOVAs on the average, F(2, 107)=4.75, P=0.01, and maximum, F(2, 107)=4.38, P=0.01, Li blood level. Post hoc comparisons indicated that WPIC had higher average and maximum Li levels than either WF or WU. Indeed, these values on average were more than 25% higher at WPIC than either WF or WU. Despite the higher Li levels at WPIC, as noted earlier, the relapse rate was essentially equivalent at WF and WPIC. There were also effects of site on the average, F(2, 110)=7.02, P=0.001, and maximum, F(2, 110)=3.37, P=0.04, oral VEN dosage. Post hoc comparisons indicated that oral VEN dose (across the VEN-Li and NT-Li groups) was higher at WPIC than WF, with WU intermediate. The magnitudes of these site differences were small.
Age exerted a significant effect in the ANCOVAs on oral average and maximum dosages of NT, VEN, and Li (all P's ≤ 0.02). In each case, older patients received smaller oral dosages. However, there was no effect of age on blood levels of NT or Li (all P's > 0.21). Thus, it appeared that the sites successfully adjusted oral dosage of NT and Li to produce substantial equivalence in blood levels. Older patients had a substantial advantage in this trial in terms of relapse rate. This effect could not be attributed to differences in age groups in blood levels of the medications. Overall average oral dosage (VEN) and blood levels (NT and Li) were within the ranges targeted.
Finally, parametric survival analyses on survival time data were conducted separately for patients treated with NT-Li or VEN-Li in Phase 2, with Phase 1 medication condition, site, and age as predictors. In separate analyses, average blood level of NT, average blood level of Li, and average oral dosage of VEN were added as predictors. In none of the six analyses was there an effect of Phase 1 medication condition (medication vs. placebo). In all six analyses there was a significant effect of age (all P's ≤ 0.02), indicating that the protective effect of age on relapse was significantly manifested in both Phase 2 treatment conditions. In no case was the oral dosage (VEN) or blood level (NT or Li) related to relapse.
Discussion
In this study, despite aggressive continuation pharmacotherapy, the overall relapse rate in the six months after remission of a depressive episode with ECT was 50%. The size of the patient sample makes it one of the larger randomized ECT data sets in the modern literature. This geographically diverse sample included unipolar and bipolar depressed patients and was representative of the severely depressed, often treatment-resistant, patients who are common in modern ECT practice. One site reported significantly higher relapse rates than the others, and, of course, these data contributed to raising the overall relapse rate observed in the study as a whole. Other than the overall relapse rate, though, this difference did not change the major findings of the study, which were the same whether the site was included in the analyses or not – mirroring a similar set of findings in the report of the outcomes in the acute phase of ECT treatment. 10.
The first aim of this study was to determine whether starting an antidepressant medication at the start of ECT resulted in a reduction in relapse when compared to the standard practice of starting continuation pharmacotherapy only once ECT is completed. There was no evidence in this trial that the early start of an antidepressant medication had any effect on postECT relapse (Figure 1). The second aim of this study was to determine whether there was a difference in the efficacy or safety of postECT continuation therapy with NT-Li compared to VEN-Li. There was no evidence in this trial for a difference between the postECT continuation therapy regimens in relapse or side effects. These findings were consistent across the sites.
It was surprising that we failed to observe any benefit of the earlier start of antidepressant medication on relapse rate. As seen in Figures 1-3, relapse in this trial and in previous prospective studies1-6 was most common in the period immediately following completion of the ECT course. When designing this trial, it seemed commonsensical that an earlier start of antidepressant treatment would prove beneficial, especially given the view that antidepressant treatment is associated with a substantial delay in onset of therapeutic benefit. However, the findings of this trial were clearly not supportive of this hypothesis. It should be recognized, of course, that this trial only tested the potential benefit of starting early the antidepressant component of the combined treatment with Li. Due to the potential of a negative interaction between Li and ECT, Li treatment is typically discontinued during the acute ECT course.20-22 In our previous multicenter, prospective trial, monotherapy with NT was distinctly inferior in relapse prevention compared to combination NT-Li. Thus, it appears that Li contributes important protection from relapse, and that starting an antidepressant alone before the completion of ECT does not contribute to relapse prevention.
Practically speaking, the implications of this negative finding regarding the timing of antidepressant administration should be considered in tandem with the earlier findings of this study.10. The short-term efficacy of ECT was improved by the concomitant administration of NT or VEN compared to PL. This effect was substantial, corresponding to an approximately 15% improvement in remission rate. There was also suggestive evidence that NT may have a protective effect on aspects of neuropsychological function. Thus, the overall results of this trial provide evidence of benefit for the co-administration of ECT and antidepressant medications. The benefit, perhaps surprisingly, is reflected in short-term ECT outcome and not in the prevention of relapse.
This study had careful standardization of medication management procedures. As a result, NT levels averaged close to the target of 100 ng/ml, and average VEN oral dosage was near the target of 300 mg/d. Li levels were typically around 0.5 mEq/L. Perhaps, due to the adequacy of dosing and medication management, no relationship was seen between oral dose or blood level of medications and relapse. Of note, older patients were able to maintain similar blood levels compared to younger patients, once again providing assurance as to the feasibility of adequate administration of these medication regimens in the elderly.
The second aim of this study was to contrast NT-Li and VEN-Li as continuation therapies. Similar maintenance of remission was obtained whether NT or VEN was used as the antidepressant medication in the combination treatment. This finding confirms that a second-generation antidepressant with both serotonergic and noradrenergic effects can be used as the antidepressant component of a pharmacologic relapse prevention strategy. This may be of special import for patients who have a contraindication to the use of NT. Both treatments were well tolerated, apparently equally, and there was no difference in side effect burden whether NT-Li or VEN-Li was used. The presumed better tolerability of a second generation agent did not materialize in the quantitative analyses of side effects.
A variety of demographic and clinical variables were examined as potential predictors of relapse. Across the analyses, and confirmed within each of the two pharmacological continuation treatments, patient age was a robust and consistent predictor of relapse. Older patients were substantially less likely to relapse than younger patients, replicating an effect we first reported in our earlier multisite, postECT randomized, continuation pharmacological trial.5 This effect was not statistically significant (P=0.13) in the recent multisite comparison of continuation ECT and NT-Li.6 Of note, approximately half the acute ECT trials that examined an association between patient age and short-term efficacy reported a positive relationship,23 including a large recent multisite study.24 Thus, it would appear that older patients have an advantage in both short-term and longer-term clinical outcome following ECT. It is very unusual in medicine for therapeutic benefit for a treatment to increase with aging. The fact that the benefit pertains to both acute efficacy and freedom from relapse provides justification for the preferential use of this intervention in late-life depression. At a mechanistic level, the aspect of aging responsible for these associations is unknown.
Treatment resistance has been a predictor of ECT short-term outcome in many,2-4,25,26 but not all,27 studies. Indeed, treatment resistance, as indexed by the number of failed adequate antidepressant treatment trials in the current episode, was a consistent predictor of short-term ECT clinical outcome in this prospective, multisite study.10 Similarly, in a number of studies treatment-resistance has been a potent predictor of postECT relapse.1,3-5,28 However, in this study the traditional measure of treatment resistance, the number of failed adequate trials, was a significant predictor of acute ECT outcome but had only a weak, nonsignificant relationship with relapse. Secondary analyses demonstrated that another measure of treatment resistance, the total number of antidepressant trials in the current episode (regardless of adequacy), had a robust relationship with relapse. Inconsistency among studies in the relations of ATHF measures of treatment resistance to ECT outcomes may be related to the effort required to query sources about past treatment trials and the limited reliability in determining the adequacy of treatments given incomplete information regarding dose, duration, and compliance. In this study, the measure that showed robust relations with relapse, total number of trials, has significant practical advantages. Its determination only requires knowledge of what trials were attempted in the current episode, without requiring knowledge of the determinants of adequacy. Determining which measures of treatment resistance are most predictive of ECT outcomes may have broad ramifications. Assessment of treatment resistance has become common in defining samples in antidepressant trials and treatment resistance has shown strong predictive value of antidepressant outcomes for other brain stimulation interventions29,30 and for pharmacological treatment of major depression.31,32
Also of consequence were the clinical features and treatment parameters that were unrelated to relapse. Polarity of depression, the presence or absence of psychosis, the severity of depressive symptoms at preECT baseline, and current episode duration (data not shown) had no relations with relapse. These negative findings may help rule out some alternative explanations for the link of age or treatment resistance with relapse. Of note, however, Axis II co-morbidity was not assessed, and there is substantial evidence that the short- and long-term efficacy of antidepressant treatment may be influenced by this dimension.33-35 It is also noteworthy that the number of ECT received in the acute treatment phase and randomization to high dosage RUL ECT or low dosage BL ECT had no impact on relapse. Indeed, to our knowledge no study has ever found vulnerability to relapse following ECT to be related to ECT technique. Indeed, in several prospective trials in which patients were randomized to ECT modalities that often differed markedly in efficacy, there was no indication that forms of ECT differed in relapse potential.2-4,10 In other words, it may be safely concluded that how one achieves remission with ECT (number of treatments, form of ECT administered) is independent of the likelihood of relapse.
While we did not find a difference between the two pharmacological continuation therapies in side effect burden, these scores were strongly related to clinical outcome during the follow-up period. Patients who relapsed reported more severe side effects during the trial than those who completed without relapse. In the earlier phase of the study, we found that side effect (UKU) scores dropped markedly during ECT compared to preECT baseline and that the magnitude of this change was associated with the change in HRSD scores.10 Thus, in both phases of this study clinical outcome was strongly related to side effect scores. This pattern may not be surprising given the overlap between depressive symptomatology and some systemic side effects. Regardless, our findings indicate that the reports of side effects were more influenced by clinical state than the forms of treatment received (ECT or pharmacology).
Each the three recent, large, multisite, prospective, randomized trials of postECT continuation therapy (including this study) found relapse rates on the order of 40-50% for optimal forms of continuation pharmacotherapy or continuation therapy with BL ECT.5,6 These are underestimates since some patients dropout before relapse can be established. Thus, even with aggressive continuation ECT or continuation pharmacotherapy, it appears that 50% or more of remitted patients will relapse within 6 months of ECT completion, with the bulk of relapse skewed to the early weeks following ECT. Two factors might be weighed when considering this conclusion. First, it is possible that the methods used in these trials to declare “relapse” are too sensitive and that a number of patients may experience worsening a few weeks after ECT that is transient. This account is speculative. In this study we required a substantial increase in symptomatology that was maintained for at least a week to designate relapse. We had no information on the duration of symptomatic worsening in patients who were declared relapsed and this might be a subject of future investigation. Second, while a 50% relapse rate following ECT is high and worrisome, it may not compare unfavorably with the relapse rate reported in treatment-resistant patients who achieve remission with pharmacological agents. It has long been thought that continuing the pharmacological strategy that achieved remission was key to relapse prevention following acute response to antidepressant medication.36 The findings of the STAR*D trial indicate that relapse following remission increases at greater levels of treatment resistance and are comparable to the rates found here.31,32
Due to unexpected low collection of cognitive data, analyses of cognitive outcomes were not possible, and represent a limitation of the study.
In summary, this study confirmed that use of lithium in combination with two different antidepressants provides moderate protection against relapse, and broadened the choice of antidepressant to include a second generation compound in common use, VEN. A recent study also found similar outcomes using a fixed schedule of continuation ECT without use of concurrent psychotropics.6 Along with the skewing of relapse to the first several weeks postECT, these observations suggest exploration of other strategies to maintain remission. One strategy is to taper ECT over a few weeks, thereby providing coverage while medication regimens are being put in place. A second approach common in community practice is to augment pharmacological continuation therapy with ECT scheduled according to symptomatic exacerbations. Alternatively, the first phase of this study demonstrated augmentation of acute response to ECT by concomitant antidepressant pharmacotherapy.10 This would suggest that the combination of complete courses of continuation pharmacotherapy with continuation ECT should be more effective than either alone. Using other forms of brain stimulation to maintain remission is also worthy of exploration, although rTMS seems most useful for less treatment resistant depression29,30 and VNS, the only other brain stimulation treatment labeled for use in depression and with evidence of long-term benefit,37 is largely unreimbursed. Study of other antidepressants and mood stabilizers may also be warranted, and there is justification to test agents with novel mechanisms. There remains an urgent need for treatments that will improve on current practice options to maintain the still superior recovery from depression that is achieved with ECT.
Acknowledgments
This study was supported in part by grants RO1 MH35636 (Dr Sackeim), RO1 MH61609 (Dr Sackeim), RO1 MH61594 (Dr McCall), RO1 MH61621 (Dr Isenberg), and RO1 MH61591 (Dr Haskett), from the US Public Health Service, Rockville, MD. A grant was obtained from Wyeth Pharmaceuticals for the purchase of the medications used in this study. The ECT devices used were loaned by the MECTA Corporation.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Sackeim HA, Prudic J, Devanand DP, Decina P, Kerr B, Malitz S. The impact of medication resistance and continuation pharmacotherapy on relapse following response to electroconvulsive therapy in major depression. J Clin Psychopharmacol. 1990;10:96–104. doi: 10.1097/00004714-199004000-00004. [DOI] [PubMed] [Google Scholar]
- 2.Sackeim HA, Prudic J, Devanand DP, Kiersky JE, Fitzsimons L, Moody BJ, McElhiney MC, Coleman EA, Settembrino JM. Effects of stimulus intensity and electrode placement on the efficacy and cognitive effects of electroconvulsive therapy. N Engl J Med. 1993;328:839–846. doi: 10.1056/NEJM199303253281204. [DOI] [PubMed] [Google Scholar]
- 3.Sackeim HA, Prudic J, Devanand DP, Nobler MS, Lisanby SH, Peyser S, Fitzsimons L, Moody BJ, Clark J. A prospective, randomized, double-blind comparison of bilateral and right unilateral electroconvulsive therapy at different stimulus intensities. Arch Gen Psychiatry. 2000;57:425–434. doi: 10.1001/archpsyc.57.5.425. [DOI] [PubMed] [Google Scholar]
- 4.Sackeim HA, Prudic J, Nobler MS, Fitzsimons L, Lisanby SH, Payne N, Berman RM, Brakemeier EL, Perera T, Devanand DP. Effects of pulse width and electrode placement on the efficacy and cognitive effects of electroconvulsive therapy. Brain Stimulation. 2008;1:71–83. doi: 10.1016/j.brs.2008.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sackeim HA, Haskett RF, Mulsant BH, Thase ME, Mann JJ, Pettinati HM, Greenberg RM, Crowe RR, Cooper TB, Prudic J. Continuation pharmacotherapy in the prevention of relapse following electroconvulsive therapy: a randomized controlled trial. JAMA. 2001;285:1299–1307. doi: 10.1001/jama.285.10.1299. [DOI] [PubMed] [Google Scholar]
- 6.Kellner CH, Knapp RG, Petrides G, Rummans TA, Husain MM, Rasmussen K, Mueller M, Bernstein HJ, O'Connor K, Smith G, Biggs M, Bailine SH, Malur C, Yim E, McClintock S, Sampson S, Fink M. Continuation electroconvulsive therapy vs pharmacotherapy for relapse prevention in major depression: a multisite study from the Consortium for Research in Electroconvulsive Therapy (CORE) Arch Gen Psychiatry. 2006;63:1337–1344. doi: 10.1001/archpsyc.63.12.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lauritzen L, Odgaard K, Clemmesen L, Lunde M, Ohrstrom J, Black C, Bech P. Relapse prevention by means of paroxetine in ECT-treated patients with major depression: a comparison with imipramine and placebo in medium-term continuation therapy. Acta Psychiatr Scand. 1996;94:241–251. doi: 10.1111/j.1600-0447.1996.tb09856.x. [DOI] [PubMed] [Google Scholar]
- 8.Sackeim HA. Continuation therapy following ECT: directions for future research. Psychopharmacol Bull. 1994;30:501–521. [PubMed] [Google Scholar]
- 9.Prudic J, Olfson M, Sackeim HA. Electro-convulsive therapy practices in the community. Psychol Med. 2001;31:929–934. doi: 10.1017/s0033291701003750. [DOI] [PubMed] [Google Scholar]
- 10.Sackeim HA, Dillingham EM, Prudic J, Cooper T, McCall WV, Rosenquist P, Isenberg K, Garcia K, Mulsant BH, Haskett RF. Effect of concomitant pharmacotherapy on electroconvulsive therapy outcomes: short-term efficacy and adverse effects. Archives of General Psychiatry. 2009;66:729–737. doi: 10.1001/archgenpsychiatry.2009.75. [DOI] [PubMed] [Google Scholar]
- 11.First MB, Spitzer RL, Gibbon M, Williams JBW. Structured Clinical Interview for Axis I DSM-IV Disorders — Patient Edition (with Psychotic Screen) (SCID-I/P) New York: Biometrics Research Department, New York State Psychiatric Institute; 1996. [Google Scholar]
- 12.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. Fourth. Washington, D. C.: American Psychiatric Association; 1994. [Google Scholar]
- 13.Hamilton M. Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol. 1967;6:278–296. doi: 10.1111/j.2044-8260.1967.tb00530.x. [DOI] [PubMed] [Google Scholar]
- 14.Sackeim HA. The definition and meaning of treatment-resistant depression. J Clin Psychiatry. 2001;62(16):10–17. [PubMed] [Google Scholar]
- 15.Guy W. ECDEU Assessment Manual for Psychopharmacology. Washington, D.C.: Superintendent of Documents, U.S Government Printing Office, U.S. Department of Health, Education, and Welfare Publication, No. 76-338; 1976. [Google Scholar]
- 16.Beck AT, Steer RA, Brown GK. Beck Depression Inventory Manual. 2nd. San Antonio, TX: Psychological Corporation; 1996. [Google Scholar]
- 17.Lingjaerde O, Ahlfors UG, Bech P, Dencker SJ, Elgen K. The UKU side effect rating scale. A new comprehensive rating scale for psychotropic drugs and a cross-sectional study of side effects in neuroleptic-treated patients. Acta Psychiatr Scand Suppl. 1987;334:1–100. doi: 10.1111/j.1600-0447.1987.tb10566.x. [DOI] [PubMed] [Google Scholar]
- 18.Kalbfleisch JD, Prentice RL. Survival Models and Data Analysis. New York: John Wiley; 1980. [Google Scholar]
- 19.Peto R, Peto J. Asymptomatically efficient rank invariant procedure. J R Statist Soc Series A. 1972;135:185–207. [Google Scholar]
- 20.Weiner RD, Whanger AD, Erwin CW, Wilson WP. Prolonged confusional state and EEG seizure activity following concurrent ECT and lithium use. Am J Psychiatry. 1980;137:1452–1453. doi: 10.1176/ajp.137.11.1452. [DOI] [PubMed] [Google Scholar]
- 21.American Psychiatric Association. The Practice of ECT: Recommendations for Treatment, Training and Privileging. Second. Washington, D.C.: American Psychiatric Press; 2001. [Google Scholar]
- 22.Small JG, Milstein V. Lithium interactions: lithium and electroconvulsive therapy. J Clin Psychopharmacol. 1990;10:346–350. [PubMed] [Google Scholar]
- 23.Sackeim HA. Electroconvulsive therapy in late-life depression. In: Roose SP, Sackeim HA, editors. Late-Life Depression. New York: Oxford University Press; 2004. pp. 241–278. [Google Scholar]
- 24.O'Connor MK, Knapp R, Husain M, Rummans TA, Petrides G, Smith G, Mueller M, Snyder K, Bernstein H, Rush AJ, Fink M, Kellner C. The influence of age on the response of major depression to electroconvulsive therapy: a C.O.R.E. Report. Am J Geriatr Psychiatry. 2001;9:382–390. [PubMed] [Google Scholar]
- 25.Prudic J, Sackeim HA, Devanand DP. Medication resistance and clinical response to electroconvulsive therapy. Psychiatry Res. 1990;31:287–296. doi: 10.1016/0165-1781(90)90098-p. [DOI] [PubMed] [Google Scholar]
- 26.Prudic J, Haskett RF, Mulsant B, Malone KM, Pettinati HM, Stephens S, Greenberg R, Rifas SL, Sackeim HA. Resistance to antidepressant medications and short-term clinical response to ECT. Am J Psychiatry. 1996;153:985–992. doi: 10.1176/ajp.153.8.985. [DOI] [PubMed] [Google Scholar]
- 27.Kellner CH, Fink M. The efficacy of ECT and “treatment resistance”. J ECT. 2002;18:1–2. doi: 10.1097/00124509-200203000-00001. [DOI] [PubMed] [Google Scholar]
- 28.Rasmussen KG, Mueller M, Rummans TA, Husain MM, Petrides G, Knapp RG, Fink M, Sampson SM, Bailine SH, Kellner CH. Is baseline medication resistance associated with potential for relapse after successful remission of a depressive episode with ECT? Data from the Consortium for Research on Electroconvulsive Therapy (CORE) Journal of Clinical Psychiatry. 2009;70:232–237. doi: 10.4088/jcp.08m04092. [DOI] [PubMed] [Google Scholar]
- 29.Lisanby SH, Husain MM, Rosenquist PB, Maixner D, Gutierrez R, Krystal A, Gilmer W, Marangell LB, Aaronson S, Daskalakis ZJ, Canterbury R, Richelson E, Sackeim HA, George MS. Daily left prefrontal repetitive transcranial magnetic stimulation in the acute treatment of major depression: clinical predictors of outcome in a multisite, randomized controlled clinical trial. Neuropsychopharmacology. 2009;34:522–534. doi: 10.1038/npp.2008.118. [DOI] [PubMed] [Google Scholar]
- 30.George MS, Lisanby SH, Avery D, McDonald WM, Durkalski V, Pavlicova M, Anderson B, Nahas Z, Bulow P, Zarkowski P, Holtzheimer PE, 3rd, Schwartz T, Sackeim HA. Daily left prefrontal transcranial magnetic stimulation therapy for major depressive disorder: a sham-controlled randomized trial. Archives of General Psychiatry. 2010;67:507–516. doi: 10.1001/archgenpsychiatry.2010.46. [DOI] [PubMed] [Google Scholar]
- 31.Rush AJ. STAR*D: what have we learned? Am J Psychiatry. 2007;164:201–204. doi: 10.1176/ajp.2007.164.2.201. [DOI] [PubMed] [Google Scholar]
- 32.Rush AJ, Trivedi MH, Wisniewski SR, Nierenberg AA, Stewart JW, Warden D, Niederehe G, Thase ME, Lavori PW, Lebowitz BD, McGrath PJ, Rosenbaum JF, Sackeim HA, Kupfer DJ, Luther J, Fava M. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163:1905–1917. doi: 10.1176/ajp.2006.163.11.1905. [DOI] [PubMed] [Google Scholar]
- 33.DeBattista C, Mueller K. Is electroconvulsive therapy effective for the depressed patient with comorbid borderline personality disorder? J ECT. 2001;17:91–98. doi: 10.1097/00124509-200106000-00002. [DOI] [PubMed] [Google Scholar]
- 34.Feske U, Mulsant BH, Pilkonis PA, Soloff P, Dolata D, Sackeim HA, Haskett RF. Clinical outcome of ECT in patients with major depression and comorbid borderline personality disorder. Am J Psychiatry. 2004;161:2073–2080. doi: 10.1176/appi.ajp.161.11.2073. [DOI] [PubMed] [Google Scholar]
- 35.Thase ME. The role of Axis II comorbidity in the management of patients with treatment-resistant depression. Psychiatr Clin North Am. 1996;19:287–309. doi: 10.1016/s0193-953x(05)70289-6. [DOI] [PubMed] [Google Scholar]
- 36.Klein D, Gittelman R, Quitkin F, Rifkin A. Diagnosis and Drug Treatment of Psychiatric Disorders: Adults and Children. Baltimore: Williams and Wilkins; 1980. [Google Scholar]
- 37.Sackeim HA, Brannan SK, John Rush A, George MS, Marangell LB, Allen J. Durability of antidepressant response to vagus nerve stimulation (VNSTM) Int J Neuropsychopharmacol. 2007:1–10. doi: 10.1017/S1461145706007425. [DOI] [PubMed] [Google Scholar]