Abstract
Purpose
To investigate the curative efficacy of osteonecrosis of the femoral head (ONFH) in a hip-preserving operative approach, by grafting a vascularized greater trochanter flap combined with a free iliac flap, in an attempt to seek an innovative approach for patients who suffered middle to late stage ONFH without total hip arthroplasty (THA) surgery.
Method
Our research included a total of 60 patients (66 hips) who accepted hip-preserving surgery by grafting a vascularized greater trochanter flap combined with a free iliac flap which was tightly filled by hammering because of ONFH (most were Association Research Circulation Osseous (ARCO) stage III patients) from January, 2006 to December, 2010. A Harris Hip Score was obtained during follow-ups, evaluating the clinical efficacy, X-rays were taken regularly for image assessing, and the SF-36 scale was used for estimating quality of life. Terminal observation time was considered when patients had symptom-dependant indications for performing another hip-preserving surgery or THA surgery.
Results
Fifty-eight patients (64 hips) were eventually contacted by telephone for an out-patient clinic return visit, with a mean follow-up time of 35.8 months (varied from 12 months to 60 months), but two patients lost contact for various reasons. The demographic data were as follows: there were 16 ARCO IIIA cases, 22 ARCO IIIB cases, and 26 ARCO IIIC cases, respectively. Postoperative X-rays revealed a well-repaired necrotic area of the femoral head and improvement of femoral-acetabulum coverage. The last follow-up mean Harris Hip Score was 86.56 ± 7.38 (excellent results reached 87.50 %), which were greatly improved compared to 50.95 ± 6.86 pre-operatively. Also the postoperative mean scores of all dimensions of the SF-36 scale were improved to some extent. Additionally the physical component summary (PSC) scores were enhanced from 42 ± 13 pre-operatively to 78 ± 11, while the postoperative mental component summary (MCS) scores (76 ± 11) largely increased in contrast to pre-operative scores (51 ± 10), with both target indices having statistical significance (p = 0.005, p = 0.01), signifying hugely improvement of the quality of life of the patients. A correlation was found between Harris Hip Score and all dimensions of SF-36 scale (r = 0.32–0.72), especially closely correlated with physical functioning (PF), role-physical (RP) and bodily pain (BP) in PCS aspect (r = 0.72, p < 0.01; r = 0.58, p < 0.01; r = 0.65, p < 0.01, respectively).
Conclusion
There is definite curative efficacy for the treatment of ONFH with an hip-preserving operative approach by grafting a vascularized greater trochanter flap combined with a free iliac flap which was tightly filled by hammering. This kind of operative approach reconstructs the biological stability of femoral head, which promotes repair of necrotic areas and indirectly preserves the femoral head of patients and a majority of hip function. It possesses vast clinical as well as practical significance, because the long-term efficacy can satisfy fundamental life requirements, especially for those young and middle-aged patients who suffer ONFH to avoid or put off the time of total hip arthroplasty(THA) surgery.
Introduction
Osteonecrosis of the femoral head (ONFH) has posed a great challenge for orthopaedic surgeons. With the change of diet (especially alcohol addiction), increase in traffic accidents (most result in femoral-neck fracture), and abuse of pain-killer drugs or immunosuppressive drugs (commonly glucocorticoids), and so on, the morbidity of ONFH presents an apparently rising tendency, and most are young and middle-aged patients [1–3]. If not treated in time, it will greatly affect the patient's life. [4, 5]. The technical improvement of total hip arthroplasty (THA) seems to give hope of complete recovery from the degenerate diseases [6, 7]. But for those young and middle-aged patients, it seems too early and patients may face the risk of more than one revision surgery and other complications [8, 9]. Thus, the theory of joint preserving management has been widely accepted by orthopaedic clinicians. The purpose of hip-preserving surgery is intended to maximally preserve and improve hip function by mending and reconstructing the blood supply, strengthening braced force of the subchondral bone of femoral head, and promoting repair and reconstruction of the necrotic bone. There exist multiple hip-preserving operative approaches, such as osteotomy [10], medullary core decompression surgery [11–13], fibular support surgery [14–16], and vascularized or non-vascularized bone flap grafting surgery [17, 18], etc. However, the definite clinical efficacy of all the above approaches is full of controversy.
Yasunaga et al. [19] conducted research regarding a histologic analysis of the femoral head that had underwent rotary osteotomy for the treatment of ONFH. They showed there was no osteogenesis repairment in the necrotic area, while various degrees of collapse were revealed in the remaining weight-bearing area. A number of articles reported definite positive efficacy of the treatment for early-stage (mainly (Association Research Circulation Osseous [ARCO] I and II) ONFH with medullary core decompression surgery [21, 22]. However, a finite-elements analysis [20] of the femoral head showed that it had a harmful effect on the structural integrity of the femoral head when that surgery was used alone, and suggested it could achieve better outcomes when combined with fibular graft support [22]. A meta-analysis [23] manifested that further surgery was indicated for those who suffered Steinberg stages I, II, and III of ONFH when performing medullary core decompression (16 %, 37 %, and 71 %, respectively). Patients in Ficat II and III stage of ONFH frequently acquired satisfactory outcomes by grafting vascularized bone flaps [17, 18].
Middle to late stage ONFH usually cannot acquire satisfactory results for both surgeons and patients, resulting in the view that most researchers [24–26] consider THA surgery as the last treatment. However, as ONFH patients become increasingly younger, a proper and improved hip-preserving approach with clinical efficacy seems extremely important and urgently needed. We are eagerly expecting to improve the clinical efficacy of hip-preservation through a new approach to treat those middle to late stage ONFH patients.
Methods and materials
Included in our research were a total 60 patients (66 hips) who underwent hip-preserving surgery over one year, by grafting a vascularized greater trochanter flap combined with a free iliac flap tightly filled by hammering because of ONFH (mostly are ARCO III stage patients), in the department of orthopaedics of the first affiliated hospital of Guangzhou University of Traditional Chinese Medicine, from January, 2006 to December, 2010. All the patients were divided into three groups according to the ARCO classification system: ARCO IIIA group, ARCO IIIB group, and ARCO IIIC group, respectively. Pre-operative data included anterior-posterior and frog X-rays of pelvic area, CT scan and MRI of both hips, along with the Harris Hip Score and score of the SF-36 scale. 58 patients (64 hips) were finally contacted by telephone- for an out-patient clinic return visit (Table 1).
Table 1.
The demographic data of included patients
| Stages | Gender | Total | Mean age | |
|---|---|---|---|---|
| M | F | |||
| ARCO III A | 11 hips | 5 hips | 16 hips | 31.25 (18 ∼ 48) Y |
| ARCO III B | 19 hips | 3 hips | 22 hips | 30.95 (18 ∼ 46) Y |
| ARCO III C | 18 hips | 8 hips | 26 hips | 30.31 (18 ∼ 46) Y |
| Total | 48 hips | 16 hips | 64 hips | 30.76 (18 ∼ 48) Y |
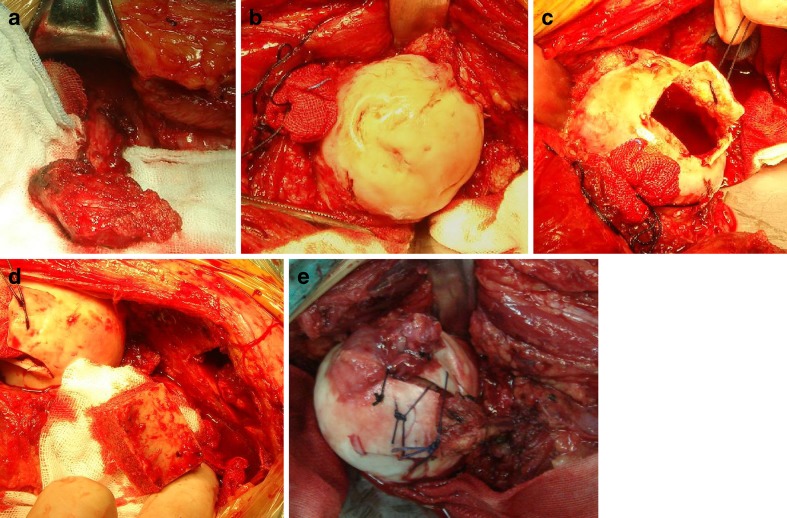
All the followed up patients had the hip-preserving surgery performed by professor Yirong Zeng et al., the primary inventor of the operation, in the department of orthopaedics of the first affiliated hospital of Guangzhou University of Traditional Chinese Medicine. The detailed procedures were as follows: Firstly, exposing the transverse branch of the lateral femoral circumflex artery, laterally separating it toward to the greater trochanter. This was followed by chiseling out the vascularized greater trochanter flap, and carefully observing beating of the exposed artery, as well as the vascularity of the out-taken flap. Secondly, the femoral head was revealed by dissecting the joint capsule covering the conjunction of head-neck, clearly opening a window-like incision in the necrotic area of femoral head with a scalpel, without taking off the whole cut bone, but linked to the remaining cortical bone. Then necrotic bone was completely cleared out until fresh blood was oozing close to the boundary of the hardening zone. After the above was completed, an ipsilateral autologous free iliac flap was taken out and shaped in the necessary way. Finally, came the grafting of allograft bone combined with a free iliac flap tightly filled by hammering in the opened surgical site, together with the already exposed vascularized greater trochanter. The joint capsule was washed with normal saline and the whole femoral head covered with antiblocking membrane. Reducing the hip joint and closing the incision was done gradually, step by step (Fig. 1).
Fig. 1.
a chiseling out the vascularized greater trochanter flap; b exposing the femoral head; c opening femoral head with a window-like incision and clearing out necrotic bone; d taking out free iliac flap, shaping the iliac flap in a proper style and grafting it into the femoral head; e grafting the vascularized greater trochanter flap, suturing the opened articular cartilage and reshaping the femoral head
There are several intra-operative matters to draw your attention to: First of all, the length, quantity, and quality of isolated vessels must be well guaranteed. Secondly, the bone tunnel under the incision must be totally excavated without breaking through cartilage surface, as well as having enough space for grafting the vascularized greater trochanter flap without compression, to facilitate the creeping substitution of neovascularization. Thirdly, the free iliac flap must be embedded lateral to the external wall of the femoral head to provide mechanical support.
Postoperative treatments are mainly the prevention of infection and deep venous thrombosis of the lower limbs (DVT). Recovery exercise concludes the six-week traction of tibial tubercle, and hip function training, especially flexion, occurs during the entire recovery stage of lying in bed. Upon removing the bone traction, patients are allowed to walk with the assistance of crutches. In this situation, the unaffected hip sustains almost the total weight of the body. Besides, holster traction for five to six hours per day for half a year postoperatively is strongly suggested to achieve good to excellent outcomes. Wheelchair use is allowed for the entire postoperative recovery, especially if both affected hips have undergone hip-preserving surgery. For those patients who underwent the operations, all were asked to come back to the out-patient orthopedic clinic for a return visit through telephone contact. X-rays of anteroposterior and frog position of the pelvis are appropriate for efficacy evaluation. The follow-up times are as follows: three month intervals within one year postoperatively, six month intervals between one year to two years postoperatively, and one year intervals thereafter. In patients' dietary suggestions, wine is banned and oral glucocorticoid should decrease or even be discontinued.
Results
Fifty-eight cases (64 hips) have been contacted by telephone for out-patient clinic return visits postoperatively. There were 16 ARCO IIIA cases (11 males and five females, mean age 31.25 years, range 18–48 years), 22 ARCO IIIB cases (19 males and three females, mean age 30.95 years, range 18–46 years), and 26 ARCO IIIC cases (18 males and eight females, mean age 30.31 years,range 18–48 years) (Tables 1, 2 and 3). Follow-up time ranged from 12 months to 60 months (mean 35.8 months). Postoperative X-rays revealed a well-repaired necrotic zone as well as improved coverage of femoral head to acetabulum (Fig. 2). Some patients suffered secondary femoral acetabulum impingement (FAI), which may lead to early osteoarthritis. In the follow-up process, we discovered osteophyte formation around the head-neck junction in six patients on postoperative X-rays. Some other patients complained of long-term mild hip pain, regardless of a good femoral head shape, while other patients acquired good function and no pain, even with poor or even bad femoral head shape on their postoperative X-rays. Among them, the Harris Hip Score of five patients was fair (range 70–79 points), while only three patients got a poor Harris Hip Score (below 70 points). We then analysed these eight patients for results. From the aspect of pain score of the Harris Hip Score, six patients had relatively low points (less than or equal to 30 points), and may have needed nonsteroidal anti-inflammatory drugs (NSAIDs), such as Meloxicam, for pain relief. However, among those six patients, three acquired good to excellent function points (greater than or equal to 45 points). Taking their latest X-rays of anteroposterior and frog position of pelvis into consideration, we assumed that obtaining good to excellent hip function may account for the good repairment of femoral head, which to some extent matches with the acetabulum. On the other hand, the reason for hip mild pain may due to relatively deficiency repairment of the subchondral bone because of blood supply obstacles. Also, one patient complained of poor function (function score was 35 points) without any hip pain (pain score was 45 points); this may be accounted for by lack of early functional training of the operated hip without following our guidance, which may lead to poor function score because of the formation of scar tissues (Table 4).
Table 2.
Harris Hip Score preoperatively and postoperatively
| Stages | Preoperative | Postoperative | P value |
|---|---|---|---|
| ARCO III A | 54.00 ± 6.57 | 87.00 ± 6.93 | P < 0.01 |
| ARCO III B | 51.00 ± 6.89 | 85.00 ± 7.16 | P < 0.01 |
| ARCO III C | 48.00 ± 7.32 | 83.00 ± 7.65 | P < 0.05 |
| Total | 50.95 ± 6.86 | 86.56 ± 7.38 | P < 0.01 |
Table 3.
Harris Hip Score scale
| Stages | Hips (cases) | Harris Hip Score scale | Good to excellent rates (%) | |||
|---|---|---|---|---|---|---|
| Excellent | Good | Fair | Poor | |||
| ARCO III A | 16 | 12 | 4 | 0 | 0 | 100 |
| ARCO III B | 22 | 16 | 4 | 2 | 0 | 90.90 |
| ARCO III C | 26 | 6 | 14 | 3 | 3 | 76.92 |
| Total | 64 | 34 | 22 | 5 | 3 | 87.50 |
Total 100 points. Excellent > 90 points, Good: 80–89 points, Fair: 70–79 points, Poor < 70 points
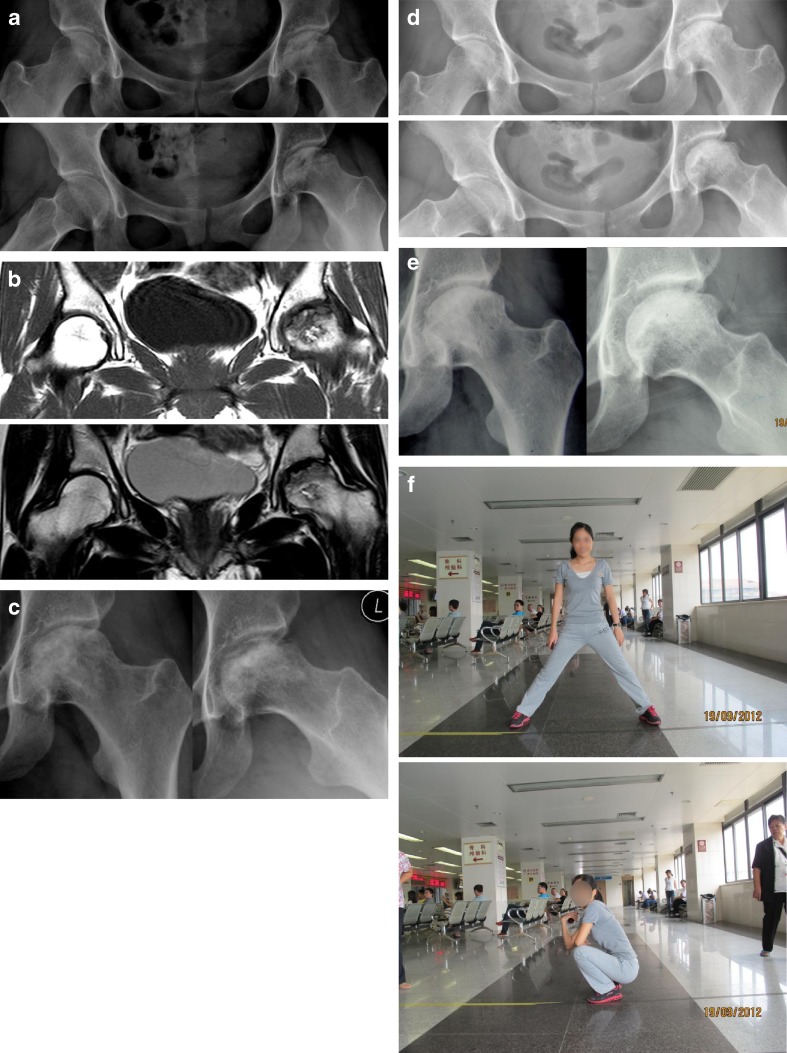
Fig. 2.
Case: female, 21 years, osteonecrosis of the femoral head (ONFH) of left hip, Y: a Pre-operative X-rays; b Pre-operative magnetic resonance imaging (MRI); c 3 months postoperatively; d 30 months postoperatively; e 48 months postoperatively; f Hip function of 48 months postoperatively
Table 4.
Details of eight patients
| Aspects of Harris Hip Score | Results Harris Hip score | |||||||
|---|---|---|---|---|---|---|---|---|
| Fair | Poor | |||||||
| ARCO III B | ARCO III C | ARCO III C | ||||||
| Pain score (total 44 points) | 44 | 30 | 30 | 40 | 30 | 20 | 30 | 20 |
| Function score (total 56 points) | 35 | 46 | 47 | 32 | 45 | 26 | 35 | 22 |
From the last return examination, we observed that Harris Hip Score reached to 86.56 ± 7.38 points postoperatively, from 50.95 ± 6.86 points pre-operatively (p < 0.01, which means an apparent statistical significance, and good-to-excellent rates were 87.50 %). With respect to the three groups, the ARCO IIIA group had 87.00 ± 6.93 points postoperatively compared to 54.00 ± 6.57(p < 0.01) points pre-operatively,100 % good to excellent rates, while Harris Hip Score in ARCO IIIB (good to excellent rates were 90.90 %) and ARCO IIIC (good to excellent rates were 76.92 %) groups improved from 51.00 ± 6.89 and 48.00 ± 7.32 points pre-operatively to 85.00 ± 7.16 (p < 0.01) and 83.00 ± 7.65 (p < 0.05) points postoperatively, respectively (Tables 2 and 3). The quality of postoperative life was strikingly enhanced indirectly, as revealed by the improved physical component summary (PSC) and mental component summary (MCS) points (Tables 5 and 6).
Table 5.
Score of each dimension of PCS of SF-36 scale
| Group | PF | RP | BP | GH | PCS |
|---|---|---|---|---|---|
| Preoperative | 36.00 ± 11.00 | 25.00 ± 14.00 | 43.00 ± 15.00 | 58.00 ± 12.00 | 42.00 ± 13.00 |
| Postoperatie | 76.00 ± 15.00 | 75.00 ± 13.00 | 82.00 ± 9.00 | 78.00 ± 10.00 | 78.00 ± 11.00 |
| Improvement (%) | 52.63 | 66.67 | 47.56 | 25.64 | 46.15 |
| P value | P = 0.005 | P = 0.0025 | P = 0.005 | P = 0.21 > 0.05 | P = 0.005 |
PCS physical component summary, PF physical functioning, RP role-physical, BP bodily pain, GH general health
Table 6.
Score of each dimension of MCS of SF-36 scale
| Group | VT | SF | RE | MH | MCS |
|---|---|---|---|---|---|
| Preoperative | 55.00 ± 11.00 | 61.00 ± 9.00 | 34.00 ± 14.00 | 52.00 ± 11.00 | 51.00 ± 10.00 |
| Postoperatie | 74.00 ± 15.00 | 87.00 ± 8.00 | 68.00 ± 12.00 | 76.00 ± 12.00 | 76.00 ± 11.00 |
| Improvement (%) | 25.68 | 29.89 | 50.00 | 31.58 | 32.89 |
| P value | P = 0.16 > 0.05 | P = 0.05 | P = 0.01 | P = 0.025 | P = 0.01 |
MCS mental component summary, VT vitality, SF social functioning, RE role emotional, MH mental health
A linear correlation (r = 0.32 ∼ 0.72) between Harris Hip Score and SF-36 score was discovered, and an especially high correlation with RF,RP, and BP of SF-36 scale(r = 0.72, p < 0.01; r = 0.58, p < 0.01; r = 0.65, p < 0.01, respectively) (Table 7). Differentiating from SF-36 scale, the Harris Hip Score evaluation centered on pain, hip function, and daily life other than mental health evaluation.
Table 7.
Correlation between score of SF-36 and Harris Hip Score
| SF-36 | Harris | |
|---|---|---|
| PF | r = 0.72 | P < 0.01 |
| RP | r = 0.58 | P < 0.01 |
| BP | r = 0.65 | P < 0.01 |
| GH | r = 0.34 | P < 0.05 |
| VT | r = 0.38 | P < 0.05 |
| SF | r = 0.42 | P < 0.05 |
| RE | r = 0.43 | P < 0.05 |
| MH | r = 0.32 | P < 0.05 |
PF physical functioning, RP role-physical, BP bodily pain, GH general health, VT vitality, SF social functioning, RE role emotional, MH mental health
Discussion
It is a great pity to miss the best chance of hip-preserving surgery because of the insidious onset of ONFH, which usually develops quickly with various kinds of cartilage damage and following subchondral bone fracture of the femoral head. There are multiple hip-preserving operations available now, as well as high risks of failure [11, 27, 28]. It has been proved that most hip-preserving surgery can achieve good outcomes only when the necrotic areas are limited to small parts of the femoral head [29]. Current available treatments focusing on reconstruction of the blood supply of femoral head can frequently largely improve the curative effect [30–34]. So does vascularized greater trochanter flap grafting.
With the recently increasing research on the pathogenesis of ONFH, biomechanics factors are attracting attention more and more. Ueo et al. [35] discovered that necrotic change might even also appear in the area of good blood supply, which arouses the possible significant role of biomechanics that play in pathogenesis of ONFH. Mihara et al. [36] conducted animal research that forced standing rats to create strong stress to their spine cords; ONFH were found in those rats. The above studies revealed that only when the pathogenesis of ONFH is combined with dynamic function of the hip, namely biomechanics, can the onset of ONFH really be explained. Based on the above theories, we improved and designed a vascularized greater trochanter flap graft combined with a free iliac flap tightly filled by hammering hip-preserving surgery. For young and middle-aged patients who suffered middle to late stage of ONFH, good to excellent clinical efficacy has been achieved when performing this kind of surgery.
Once necrotic bone has formed, the repair reaction starts. The repair reaction is a process of revascularization, which extends from the normal blood supply around the necrotic area unidirectionally to the core of involved area. However, the repair capability is limited in most patients, which can only form granulation tissue and a hardening zone around the necrotic area. As a result, it may destroy the mechanical structure in the trabeculae of the femoral head and osteone of the subchondral bone, which leads to stress declination in the necrotic area and stress concentration around involved area. It can easily lead to fracture of the trabeculae and osteone of the femoral head when stress acts on pathological tissues. Consequently, fracture, even collapse of the femoral head, cannot finally be avoided.
Korompilias et al. [30] regarded that the affective factors of hip-preserving surgery were the pathogen of ONFH, and the classification as well as the range of necrosis area. According to research concerning the biomechanics of the hip [35, 36], the two key principles of hip-preserving surgery for the treatment of ONFH are promoting repair of the necrotic area and protecting the femoral head from collapsing. The main predictive risk factors of the incidence of collapse depend on the range and area of necrosis [4, 11, 36, 37]. Our research has shown that the more normal shape the femoral head retains, the more hip function is kept. However, this can only be realized on the basis of good repair-ability when the lateral walls are integral enough to prevent collapse, and a less involved area of the femoral head.
When performing this hip-preserving surgery, totally clear out the necrosis tissues until the hardening zone and fresh bleeding can be watched. This kind of management decreases not only the compression of femoral head, but also stress concentration around necrotic zone. Following this, we need to graft bone and vessels into the necrotic zone to maintain the balance and blood supply of the femoral head. It promotes an extremely strong induction of osteogenesis with grafting allogeneic cancellous bone in the initiating stage [38]. In order to prevent collapse of the femoral head, the free iliac graft needs to be close to the lateral wall to provide powerful mechanical support in the repair stage. Vascularized greater trochanter flap grafts are placed under the surgical site near the head-neck conjunction to provide a blood supply for the femoral head. When closing the window-like surgical site, we need to pay attention to articular cartilage trimming, as well as femoral head reshaping, which can provide good conditions for the repairment of femoral head.
It can effectively avoid the incidence of complications that are caused by donor site and grafts. There has been no complication in our study. Gaskill et al. [39] found that complications occurred in 215 patients from 1,270 cases who accepted autologous bone graft surgery. Among them, there were146 cases that appeared to be donor site complications, while 29 cases appeared to be graft complications. However, they both had been well managed. They considered that the above-mentioned incidence of complications were not out of our acceptance. Besides, serious complications were not commonly seen, while mild complications temporarily occurred and recovery was easily completed.
We discovered that some patients revealed early-stage hip osteoarthritis (OA) lesions and mild heterotopic ossification (HO) in the follow-up process. There were four patients that revealed early-stage hip OA without any complaint of symptoms, and two patients were found to have mild HO (one was Brooker stage I and the other was Brooker stage II) from postoperative X-rays. Hip OA may account for articular cartilage impairment. Intra-operatively, we found that the majority of cartilages were out-of-flatness, rugae-like, and even stripped from the articular cartilage surface. In order to minimize the articular cartilage damage, we chose to use the rugae-like zone as the window-like incision. Hip OA may appear if the articular cartilage surface is not well repaired. The occurrence of HO may be relevant to multiple factors, so we douched the hip joint thoroughly with normal saline intra-operatively and prescribed oral NSAIDs (mainly Meloxicam) to patients postoperatively.
Our study aimed to evaluate the efficacy of that hip-preserving surgery. In fact, patients who underwent that kind of surgery mostly obtained satisfactory hip function, and no patient from the last return visit needed a total hip replacement. Maybe, total hip replacement for those patients cannot be avoided in the end. However, when that kind of surgery is performed, patients will receive less revision surgery or even avoid them for their entire life. Besides, there existed several limitations in our study. Firstly, the included samples are relatively small, and the follow-up time was relatively short, which may lead to suspicion of its availability and limit its wide application. Secondly, after performing this kind of surgery, complete weight bearing is forbidden for one year. It is hard to make sure that all operated patients follow our guidance, which is shown to be extremely important for the final outcomes. Furthermore, when they heard of the one year rest from work, some patients were hesitant to accept the surgery. Thirdly, in our research, we evaluated the clinical efficacy mainly depending on patient’s pain relief, hip function improvement, and radiog raphic changes other than estimation of overall quality of life improvement. More clinical practice and longer follow-up time are needed for better evaluation of this kind of hip-preserving surgery. All in all, the authors show great interest in its study and are eager for it to become a widely available application to do more benefits to patients who suffer from ONFH.
References
- 1.Razik F, Alexopoulos AS, El-Osta B, Connolly MJ, Brown A, Hassan S, Ravikumar K. Time to internal fixation of femoral neck fractures in patients under sixty years--does this matter in the development of osteonecrosis of femoral head? Int Orthop. 2012;36(10):2127–2132. doi: 10.1007/s00264-012-1619-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Li Y, Han R, Geng C, Wang Y, Wei L. A new osteonecrosis animal model of the femoral head induced by microwave heating and repaired with tissue engineered bone. Int Orthop. 2009;33(2):573–580. doi: 10.1007/s00264-008-0672-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Akinyoola AL, Adediran IA, Asaleye CM, Bolarinwa AR. Risk factors for osteonecrosis of the femoral head in patients with sickle cell disease. Int Orthop. 2009;33(4):923–926. doi: 10.1007/s00264-008-0584-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Merle D’Aubigné R, Postel M, Mazabraud A, Massias P, Gueguen J. Idiopathic necrosis of the femoral head in adults. Bone Joint Surg Br. 1965;47:612–633. [PubMed] [Google Scholar]
- 5.Ohzono K, Saito M, Takaoka K, Ono K, Saito S, Nishina T, Kadowaki T. Natural history of nontraumatic avascular necrosis of the femoral head. Bone Joint Surg Br. 1991;73:68–72. doi: 10.1302/0301-620X.73B1.1991778. [DOI] [PubMed] [Google Scholar]
- 6.Moriya M, Uchiyama K, Takahira N, Fukushima K, Yamamoto T, Hoshi K, Itoman M, Takaso M. Evaluation of bipolar hemiarthroplasty for the treatment of steroid-induced osteonecrosis of the femoral head. Int Orthop. 2012;36(10):2041–2047. doi: 10.1007/s00264-012-1612-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim YH, Choi Y, Kim JS. Cementless total hip arthroplasty with ceramic-on-ceramic bearing in patients younger than 45 years with femoral-head osteonecrosis. Int Orthop. 2010;34(8):1123–1127. doi: 10.1007/s00264-009-0878-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Katz RL, Bourne RB, Rorabeck CH, McGee H. Total hip arthroplasty in patients with avascular necrosis of the hip: follow-up observations on cementless and cemented operations. Clin Orthop Relat Res. 1992;281:145–151. [PubMed] [Google Scholar]
- 9.Kim YH, Oh JH, Oh SH. Cementless total hip arthroplasty in patients with osteonecrosis of the femoral head. Clin Orthop Relat Res. 1995;320:73–84. [PubMed] [Google Scholar]
- 10.Sugioka Y, Hotokebuchi T, Tsutsui H. Transtrochanteric anterior rotational osteotomy for idiopathic and streoid-induced necrosis of the femoral head. Indications and long-term results. [J] Clin Orthop Relat Res. 1992;277:111–120. [PubMed] [Google Scholar]
- 11.Fairbank AC, Bhatia D, Jinnah RH, Hungerford DS. Long-term results of core decompression for ischaemic necrosis of the femoral head. Bone Joint Surg Br. 1995;77:42–49. [PubMed] [Google Scholar]
- 12.Koo KHI, Kim R, Ko GH, Song HR, Jeong ST, Cho SH. Preventing collapse in early osteonecrosis of the femoral head: A randomised clinical trial of core decompression. Bone Joint Surg Br. 1995;77:870–874. [PubMed] [Google Scholar]
- 13.Mont MA, Carbone JJ, Firbank AC. Core decompression versus nonoperative management for osteonecrosis of the hip. Clin Orthop Relat Res. 1996;324:169–178. doi: 10.1097/00003086-199603000-00020. [DOI] [PubMed] [Google Scholar]
- 14.Buckley PD, Gearen PF, Petty RW. Structural bone-graft-ing for early atraumatic avascular necrosis of the femoral head. Bone Joint Surg Am. 1991;73:1357–1364. [PubMed] [Google Scholar]
- 15.Scully SP, Aaron RK, Urbaniak JR. Survival analysis of hips treated with core decompression or vascularized fibular grafting because of avascular necrosis. Bone Joint Surg Am. 1998;80:1270–1275. doi: 10.2106/00004623-199809000-00004. [DOI] [PubMed] [Google Scholar]
- 16.Smith KR, Bonfiglio M, Montgomery WJ. Non-traumatic necrosis of the femoral head treated with tibial bone-grafting: A follow-up note. Bone Joint Surg Am. 1980;62:845–847. [PubMed] [Google Scholar]
- 17.Meyers MH. The treatment of osteonecrosis of the hip with fresh osteochondral allografts and with the muscle pedicle graft technique. Clin Orthop. 1978;130:202–209. [PubMed] [Google Scholar]
- 18.Matsusaki H, Noguchi M, Kawakami T, et al. Use of vascularized pedicle iliac bone graft combined with transtrochanteric rotational osteotomy in the treatment of avascular necrosis of the femoral head. Arch Orthop Trauma Surg. 2005;125(2):95–101. doi: 10.1007/s00402-004-0777-z. [DOI] [PubMed] [Google Scholar]
- 19.Yasunaga Y, Hisatome T, Ikuta Y, et al. A histological study of the necrotic area after transtrochanteric anterior rotational osteotomy for osteonecrosis of the femoral head. J Bone Joint Surg Br. 2001;83(2):167–170. doi: 10.1302/0301-620X.83B2.11503. [DOI] [PubMed] [Google Scholar]
- 20.Clark JC, Dass CR, Choong PF. A review of clinical and molecular prognostic factors in osteosarcoma. Cancer Res Clin Oncol. 2008;134(3):281–297. doi: 10.1007/s00432-007-0330-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Floerkemeier T, Lutz A, Nackenhorst U, Thorey F, Waizy H, Windhagen H, von Lewinski G. Core decompression and osteonecrosis intervention rod in osteonecrosis of the femoral head: clinical outcome and finite element analysis. Int Orthop. 2011;35(10):1461–1466. doi: 10.1007/s00264-010-1138-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baksi DP, Pal AK, Baksi DD. Long-term results of decompression and muscle-pedicle bone grafting for osteonecrosis of the femoral head. Int Orthop. 2009;33(1):41–47. doi: 10.1007/s00264-007-0455-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dass CR, Nadesapillai AP, Robin D, et al. Downregulation of uPAR confirms link in growth and metastasis of osteosarcoma. Clin Exp Metastasis. 2005;22(8):643–652. doi: 10.1007/s10585-006-9004-3. [DOI] [PubMed] [Google Scholar]
- 24.Keizer SB, Kock NB, Dijkstra PD, Taminiau AH, Nelissen RG. Treatment of avascular necrosis of the hip by a non-vascularised cortical graft. Bone Joint Surg Br. 2006;88:460–466. doi: 10.1302/0301-620X.88B4.16950. [DOI] [PubMed] [Google Scholar]
- 25.Dailiana ZH, Toth AP, Gunneson E, Berend KR, Urbaniak JR. Free vascularized fibular grafting following failed core decompression for femoral head osteonecrosis. Arthroplasty. 2007;22:679–688. doi: 10.1016/j.arth.2006.12.042. [DOI] [PubMed] [Google Scholar]
- 26.Israelite C, Nelson CL, Ziarani CF, Landa J, Steinberg ME. Bilateral core decompression for osteonecrosis of the femoral head. Clin Orthop Relat Res. 2005;441:285–290. doi: 10.1097/01.blo.0000192365.58958.84. [DOI] [PubMed] [Google Scholar]
- 27.Athanasios E, Bassounas A, Apostolos H, Karantanas B, Dimit-rios I, Fotiadis C, Konstantinos N, Malizos D. Femoral head osteonecrosis: volumetric MRI assessment and outcome. Eur J Radiol. 2007;63:10–15. doi: 10.1016/j.ejrad.2007.03.028. [DOI] [PubMed] [Google Scholar]
- 28.Mont MA, Hungerford DS. Non-traumatic avascular necrosis of the femoral head. Bone Joint Surg Am. 1995;77:459–474. doi: 10.2106/00004623-199503000-00018. [DOI] [PubMed] [Google Scholar]
- 29.Zhao FC, Li ZR, Zhang NF, Wang BL, Sun W, Cheng LM, Liu ZH. Lesion size changes in osteonecrosis of the femoral head: a long-term prospective study using MRI. Int Orthop. 2010;34(6):799–804. doi: 10.1007/s00264-009-0829-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Korompilias AV, Lykissas MG, Beris AE, et al. Vascularised fibular graft in the management of femoral head osteonecrosis: twenty years later. J Bone Joint Surg Br. 2009;91(3):287–293. doi: 10.1302/0301-620X.91B3.21846. [DOI] [PubMed] [Google Scholar]
- 31.Mont MA, Jones LG, Seyler TM, et al. New treatment approaches for osteonecrosis of the femoral head. Instr Course Lect. 2007;56(2):197–212. [PubMed] [Google Scholar]
- 32.Kawate K, Yajima H, Sugimoto K, et al. Indications for free vascularized fibular grafting for the treatment of osteonecrosis of the femoral hend. BMC Musculoskelet Disord. 2007;8(1):78. doi: 10.1186/1471-2474-8-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jin H, Xia B, Yu N, He B, Shen Y, Xiao L, Tong P. The effects of autologous bone marrow mesenchymal stem cell arterial perfusion on vascular repair and angiogenesis in osteonecrosis of the femoral head in dogs. Int Orthop. 2012;36(12):2589–2596. doi: 10.1007/s00264-012-1674-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yin S, Zhang C, Jin D, Chen S, Sun Y, Sheng J. Treatment of osteonecrosis of the femoral head in lymphoma patients by free vascularised fibular grafting. Int Orthop. 2011;35(8):1125–1130. doi: 10.1007/s00264-010-1031-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ueo T, Tsutsumi S, Yamamuro T, et al. Biomechanical aspect of the development of aseptic necrosis of the femoral head. Arch Orthop Trauma Surg. 1985;04(3):145–149. doi: 10.1007/BF00454690. [DOI] [PubMed] [Google Scholar]
- 36.Mihara K, Hirano T. Standing Is a Causative Fact or in Osteonecr osis of the Femoral Head in Gr owing Rats. Pediatr Orthop. 1998;18:665–669. doi: 10.1097/00004694-199809000-00022. [DOI] [PubMed] [Google Scholar]
- 37.Shimizu K, Moriya H, Akita T, Sakamoto M, Suguro T. Pre-diction of collapse with magnetic resonance imaging of avascular necrosis of the femoral head. Bone Joint Surg Am. 1994;76:215–223. doi: 10.2106/00004623-199402000-00007. [DOI] [PubMed] [Google Scholar]
- 38.Gamradt SC, Lieberman JR. Genetic modification of stem cells to en2 hance bone rapair. Ann Biomed Eng. 2004;1:136–147. doi: 10.1023/B:ABME.0000007798.78548.b8. [DOI] [PubMed] [Google Scholar]
- 39.Gaskill TR, Urbaniak JR, Aldridge JM, et al. Vascularized fibular transfer for femoral head osteonecrosis: donor and graft site morbidity. Bone Joint Surg Am. 2009;91(8):1861–1867. doi: 10.2106/JBJS.H.01105. [DOI] [PubMed] [Google Scholar]