Abstract
Objective
We aimed to determine if the severity of inhalation injury evokes an immune response measurable at the systemic level and to further characterize the balance of systemic pro- and anti-inflammation early after burn and inhalation injury.
Summary Background Data
Previously we reported that the pulmonary inflammatory response is enhanced with worse grades of inhalation injury, and that those who die from their injuries have a blunted pulmonary immune profile compared to survivors.
Methods
From August 2007 to June 2011, bronchoscopy was performed on 80 patients admitted to the burn intensive care unit when smoke inhalation was suspected. Of these, inhalation injury was graded into one of five categories (0, 1, 2, 3, and 4), with Grade 0 being the absence of visible injury and Grade 4 corresponding to massive injury. Plasma was collected at the time of bronchoscopy and analyzed for 28 immunomodulating proteins via multiplex bead array or ELISA.
Results
The concentrations of several plasma immune mediators were increased with worse inhalation injury severity, even after adjusting for age and % TBSA. These included interleukin (IL)-1RA (p=0.002), IL-6 (p=0.002), IL-8 (p=0.026), granulocyte colony stimulating factor (p=0.002), and monocyte chemotactic protein (MCP)-1 (p=0.007). Differences in plasma immune mediator concentrations in surviving and deceased patients were also identified. Briefly, plasma concentrations of IL-1RA, IL-6, IL-8, IL-15, Eotaxin, and MCP-1 were higher in deceased patients compared to survivors (p<0.05 for all), while IL-4 and IL-7 were lower (p<0.05). After adjusting for the effects of age, % TBSA, and inhalation injury grade, plasma IL-1RA remained significantly associated with mortality (OR 3.12, 95% CI 1.03–9.44). Plasma IL-1RA also correlated with % TBSA, inhalation injury grade, fluid resuscitation, Baux score, revised Baux score, Denver score, and the Sequential Organ Failure Assessment score.
Conclusion
The severity of smoke inhalation injury has systemically reaching effects, which argues in favor of treating inhalation injury in a graded manner. Additionally, several plasma immune mediators measured early after injury were associated with mortality. Of these, IL-1RA appeared to have the strongest correlation with injury severity and outcomes measures, which may explain the blunted pulmonary immune response we previously found in non-survivors.
Keywords: Burn, Inhalation injury, Cytokines, Chemokines, Growth Factors, Interleukin-1 receptor antagonist, Mortality
Introduction
Early excision of the burn wound, improved understanding of the hypermetabolic response to injury, and modern advancements in critical care with appreciation for acute lung injury have all dramatically improved our ability to support the severely burn-injured patient.1 Despite these advancements, however, inhalation injury remains a significant contributor to mortality, increasing its likelihood by as much as 25%.2 Inhalation injury is also relatively common among patients with burns, yet there remains no standard for its diagnosis, scoring, and subsequent treatment. Further, as surgeons and critical care specialists, our understanding of the local and systemic inflammatory response to injury and sepsis continues to evolve, particularly in light of failed clinical trials designed to modulate the immune response to these conditions. Recent studies are also challenging the paradigm of a systemic inflammatory response syndrome that shifts over time to a compensatory anti-inflammatory response syndrome, in that the pro- and anti-inflammatory responses to injury and sepsis appear as processes with considerable overlap.3 Understanding the interplay between these mediators in the face of critical illness remains at the forefront of clinical and laboratory research efforts.
Concurrent to the evolving understanding of local and systemic inflammatory responses to injury and sepsis, we have found in burn patients with smoke inhalation that the pulmonary inflammatory response is enhanced with worse grades of inhalation injury, and that those who die from their injuries have a blunted pulmonary immune responsiveness compared to survivors.4,5 Based on the conclusions of these prior studies, we sought to answer two important questions: 1) Does the severity of inhalation injury evoke an immune response measurable at the systemic level, and 2) Does the systemic immune response to burn and smoke inhalation explain the pulmonary immune hyporesponsiveness that we have seen in non-survivors? Therefore, the present report investigated the plasma immune response early after burn and smoke inhalation, aiming to describe the balance of circulating pro- and anti-inflammatory mediators, and to correlate the systemic immune profile with both the severity of inhalation injury and outcomes.
Methods
Patients and Parameters
From August 2007 to June 2011, bronchoscopy was performed on 80 consecutive patients admitted to the burn intensive care unit (ICU) when smoke inhalation was suspected by history, physical, and/or laboratory findings. Specifically, the criteria for bronchoscopy included evidence of carbonaceous sputum, soot at the nares, burn of the head and neck, injury within a confined space, prolonged extrication, and/or and elevated % plasma carboxyhemoglobin (% COHb). Participants were excluded for the following: under age 18, use of immunosuppressive medications, the presence of malignancy, autoimmune condition, chronic inflammatory disease, or declining study participation. Plasma was collected at the time of bronchoscopy, which was within 15 hours of injury in all cases. The plasma of 17 non-smoking healthy adult volunteers, free of known pulmonary, cardiac, infectious, allergic, autoimmune, or cancerous disease, was also assessed for comparison.
Clinical variables and outcomes of interest were recorded, including: age, gender, race/ethnicity, % total body surface area burn (% TBSA), Baux score (Age + % TBSA), revised Baux score [Age + % TBSA + 17*Inhalation injury (0=No, 1=Yes)],6 Denver score, Sequential Organ Failure Assessment (SOFA) score, 24- and 72-hour fluid resuscitation (cc/kg), grade of inhalation injury, % plasma COHb, initial 48-hour and full hospitalization nadir of the ratio of plasma oxygen content to fraction of inspired oxygen (P:F ratio), initial 48-hour and full hospitalization peak of plasma creatinine and bilirubin, frequency of pneumonia, sepsis, and tracheostomy, ventilator days, ventilator free days, ICU length of stay (LOS), hospital LOS, and mortality. Pneumonia and sepsis were defined according to American Burn Association Consensus Conference criteria.7 Mortality was defined as any death from the time of injury to discharge from our tertiary burn center. This study was conducted under institutional review board approval.
Grading of Inhalation Injury and Sample Processing
During bronchoscopy, inhalation injury was graded into one of five categories (0, 1, 2, 3, and 4) based on Abbreviated Injury Score criteria, with Grade 0 being the absence of visible injury and Grade 4 corresponding to massive injury.8 Plasma was analyzed in duplicate for 28 immunomodulating proteins via multiplex immunoassay (Bio-Rad Laboratories, Hercules, CA) or ELISA (R&D Systems, Minneapolis, MN) as previously described, and according to manufacturer instructions.9 These results were compared between those with different grades of inhalation injury and outcomes.
Statistical analysis
All data were assessed for normality and parametric or non-parametric tests applied where appropriate. Dichotomous variables are reported as a percent and number, non-parametric variables are reported as median with the interquartile range, and continuous variables of parametric tests are reported as mean with standard deviation. Multivariable regression was performed based on log-transformed data in order to adjust for relevant confounders. Statistical analyses were calculated with SAS Version 9.1 (SAS Institute Inc., Cary, NC).
Results
Demographics, Clinical Characteristics, and Outcomes
Bronchoscopy was performed on 80 patients suspected of inhalation injury; of these, 72 had no exclusion criteria and were enrolled in these investigations. The median (interquartile range) time from injury to BALF and plasma collection was 6 (5–10) hours. Patients had a median age of 49 years, were 40% female, and had the following distribution of inhalation injury grades: Grade 0 (n=13); Grade 1, (n=16); Grade 2, (n=18); Grade 3, (n=20); Grade 4, (n=5). The mean (± SD) % TBSA burn of the entire cohort was 20.2 (± 23.2) %, and the median (interquartile range) % TBSA burn was 12.5 (1.0 to 31.3) %. No patient died during the initial 72 hours of resuscitation, and the subsequent primary causes of mortality were the following: multiple organ failure (n=8), cardiogenic shock (n=4), septic shock (n=1), acute respiratory distress syndrome (n=1), CO poisoning (n=1), and massive cerebrovascular accident (n=1). Of the 16 mortalities, 11 involved withdrawal of care when further heroic efforts became futile.
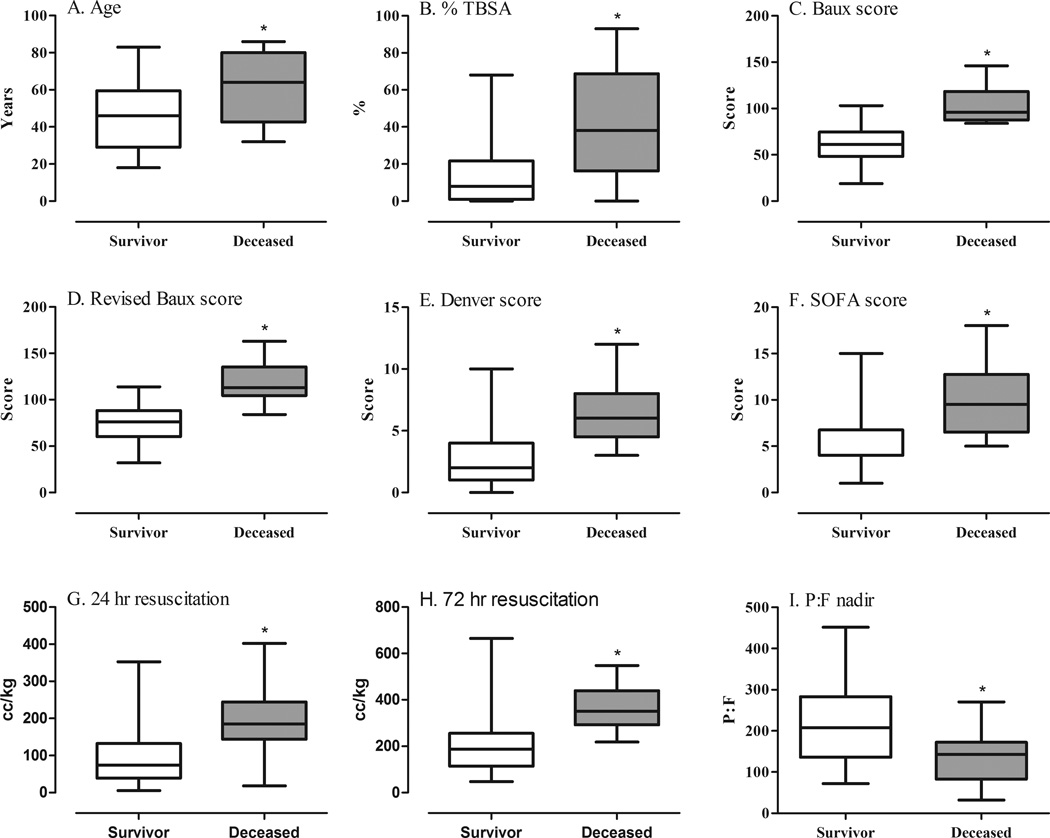
Table 1 demonstrates data corresponding to survival status, with select comparisons represented in Figure 1. Compared to survivors (n=56), those who succumbed to their injuries (n=16) were older (median 64 vs 46 years; p=0.005), had fewer ventilator-free days (median 0 vs 6; p<0.001), and had a greater % TBSA burn (median 38 vs 8; p<0.001), Baux score (median 96.0 vs 61.0; p<0.0001), Revised Baux score (median 113.0 vs 76.0; p<0.0001), Denver score (median 6 vs 2, p<0.0001), SOFA score (median 10 vs 4; p<0.0001), 24-hour fluid resuscitation (median 185 cc/kg vs 74 cc/kg; p<0.001), and 72-hour fluid resuscitation (median 351 cc/kg vs 187 cc/kg; p<0.001). Indeed, 25% of deceased patients compared to only 5% of survivors exceeded a 24-hour fluid resuscitation of 250 cc/kg (p=0.039), re-affirming this potentially important cutoff.10 Likewise, deceased patients also had a lower initial 48-hour P:F ratio nadir (median 142.5 vs 207.5; p=0.003), and had a greater peak of plasma creatinine and bilirubin levels during their hospitalization [median 2.50 vs 1.03 (p<0.001) and median 1.3 vs 0.8 (p<0.001), respectively]. Finally, deceased patients had a greater frequency of sepsis (38% vs 8%) and pneumonia (69% vs 45%), though these clinically relevant comparisons fell short of statistical significance (p<0.10 for both).
Table 1.
Demographics, clinical characteristics, outcomes, and mortality
| Survivors n=56 |
Deceased n=16 |
p Value | |
|---|---|---|---|
| Age (years) | 46.0 (29.0–59.5) | 64.0 (42.5–80.0) | 0.005 |
| Gender | 0.897 | ||
| Male | 34 (61) | 10 (63) | |
| Female | 22 (39) | 6 (38) | |
| Race/Ethnicity | 0.096 | ||
| Caucasian | 34 (61) | 9 (56) | |
| African American | 17 (30) | 2 (13) | |
| Hispanic | 3 (5) | 3 (19) | |
| Asian | 2 (4) | 2 (13) | |
| TBSA (%) | 8.0 (1.0–21.8) | 38.0 (16.3–68.8) | <0.001 |
| Baux score | 61.0 (48.3–74.5) | 96.0 (87.5–118.5) | <0.0001 |
| Revised Baux score | 76.0 (60.3–88.5) | 113.0 (104.5–135.5) | <0.0001 |
| Denver Score | 2 (1–4) | 6 (5–8) | <0.0001 |
| SOFA Score | 4 (4–8) | 10 (7–13) | <0.0001 |
| 24 hr fluid resuscitation (cc/kg) | 73.9 (39.0–132.1) | 184.8 (143.9–244.1) | <0.001 |
| 72 hr fluid resuscitation (cc/kg) | 186.9 (113.6–256.2) | 351.3 (292.0–439.3) | <0.001 |
| Inhalation injury | 44 (79) | 15 (94) | 0.272 |
| Inhalation injury grade | 2 (1–3) | 2 (1–3) | 0.436 |
| COHb (%) | 7.5 (3.7–12.2) | 4.1 (1.1–18.1) | 0.162 |
| Lowest P:F | |||
| (Initial 48 hours) | 207.5 (136.0–282.5) | 142.5 (82.5–172.5) | 0.003 |
| (Full hospitalization) | 143.0 (97.1–227.5) | 89.4 (65.3–135.2) | 0.001 |
| Peak Creatinine | |||
| (Initial 48 hours) | 1.01 (0.80–1.20) | 1.17 (0.98–1.53) | 0.092 |
| (Full hospitalization) | 1.03 (0.82–1.63) | 2.50 (1.33–3.37) | <0.001 |
| Peak Bilirubin | |||
| (Full hospitalization) | 0.8 (0.6–1.0) | 1.3 (0.9–1.9) | <0.001 |
| Pneumonia | 25 (45) | 11 (69) | 0.089 |
| Sepsis | 10 (18) | 6 (38) | 0.096 |
| Tracheostomy | 19 (34) | 7 (44) | 0.471 |
| Ventilator days | 9.5 (3.0–25.0) | 25.0 (6.3–37.5) | 0.061 |
| Ventilator free days | 6.0 (1.5–16.0) | 0.0 (0.0–0.0) | <0.001 |
| ICU Days | 17.5 (9.0–33.8) | 24.0 (7.0–43.3) | 0.750 |
| Hospital LOS | 23.0 (9.3–36.3) | 25.5 (6.3–44.0) | 0.973 |
Data represented as n (%) or median (interquartile range)
Baux score = Age + % TBSA
Revised Baux score = Age + % TBSA + 17*Inhalation injury (0=No, 1=Yes)
COHb, carboxyhemoglobin; ICU, intensive care unit; LOS, length of stay; SOFA, sequential organ failure assessment; TBSA, total body surface area burn
Figure 1.
Comparison of clinical parameters and outcomes measures between surviving and deceased patients of combined burn and smoke inhalation injury. Age (A), % TBSA (B), Baux score (C), Revised Baux score (D), Denver score (E), SOFA Score (F), Initial 24 hour resuscitation (G), Initial 72 hour resuscitation (H), P:F nadir within the first 48 hours of injury. *p<0.05 vs Survivor, Kruskal-Wallis test.
Plasma immune mediator concentrations according to smoke inhalation severity and burn size
The primary focus of this study was to determine whether or not the severity of smoke inhalation induced systemic immune mediator changes. Of the 72 patients enrolled in the study, 64 had plasma collected at the time of bronchoscopic diagnosis of their inhalation injury. We found that the concentrations of several plasma immune mediators were increased with worse inhalation injury severity, even after adjusting for age and % TBSA (Table 2). These included interleukin (IL)-1RA (p=0.002), IL-6 (p=0.002), IL-8 (p=0.026), granulocyte colony-stimulating factor (G-CSF) (p=0.002), and monocyte chemotactic protein (MCP)-1 (p=0.007). Likewise, the following plasma mediators were also increased with worse inhalation injury, though failed to reach statistical significance: IL-1β (p=0.078), IL-10 (p=0.075), IL-15 (p=0.060), and tumor necrosis factor (TNF)-α (p=0.099).
Table 2.
Plasma immune mediator concentrations according to smoke inhalation severity and burn size
| Mediator (pg/ml) |
Control | Burn Size (% TBSA) |
None (Grade 0) |
Low (Grade 1/2) |
High (Grade 3/4) |
p-value | Adjusted p-value* |
|---|---|---|---|---|---|---|---|
| C5a | 1494 | ≤10 % >10 % |
2795 516.9 |
1640 648.8 |
1738 527.6 |
0.529 0.829 |
0.921 |
| IL-1β | 1.3 | ≤10 % >10 % |
1.8 4.0 |
3.2 3.9 |
3.3 4.7 |
0.312 0.196 |
0.078 |
| IL-1RA | 123.2 | ≤10 % >10 % |
167.8 484.4 |
218.2 781.9 |
409.4 1656 |
0.191 0.045 |
0.002 |
| IL-2 | 0.0 | ≤10 % >10 % |
3.5 8.5 |
7.6 5.5 |
8.0 9.3 |
0.926 0.618 |
0.940 |
| IL-4 | 9.0 | ≤10 % >10 % |
1.3 7.7 |
4.0 2.1 |
3.9 3.5 |
0.350 0.116 |
0.650 |
| IL-5 | 3.7 | ≤10 % >10 % |
2.6 3.0 |
2.2 1.6 |
2.9 1.7 |
0.937 0.229 |
0.319 |
| IL-6 | 11.5 | ≤10 % >10 % |
33.8 116.5 |
54.2 140.7 |
234.6 274.4 |
0.010 0.211 |
0.002 |
| IL-7 | 19.8 | ≤10 % >10 % |
20.0 31.3 |
22.3 6.2 |
9.3 7.1 |
0.717 0.157 |
0.198 |
| IL-8 | 18.9 | ≤10 % >10 % |
16.5 26.8 |
11.4 28.5 |
59.3 44.1 |
0.014 0.344 |
0.026 |
| IL-9 | 0.0 | ≤10 % >10 % |
2.6 56.7 |
45.1 44.0 |
36.5 29.8 |
0.404 0.894 |
0.259 |
| IL-10 | 0.0 | ≤10 % >10 % |
4.1 17.6 |
5.5 15.0 |
12.1 19.2 |
0.085 0.354 |
0.075 |
| IL-12 | 4.8 | ≤10 % >10 % |
4.5 25.3 |
12.7 12.3 |
10.4 13.2 |
0.313 0.229 |
0.714 |
| IL-13 | 3.9 | ≤10 % >10 % |
1.2 6.1 |
5.7 5.0 |
5.4 4.9 |
0.123 0.938 |
0.313 |
| IL-15 | 0.0 | ≤10 % >10 % |
10.4 7.4 |
7.7 10.8 |
10.7 14.5 |
0.401 0.103 |
0.060 |
| IL-17 | 0.0 | ≤10 % >10 % |
0.3 31.6 |
18.3 11.5 |
25.0 31.5 |
0.178 0.138 |
0.291 |
| Eotaxin | 0.0 | ≤10 % >10 % |
154.5 247.8 |
166.4 182.1 |
147.3 185.9 |
0.945 0.209 |
0.470 |
| FGF-basic | 0.0 | ≤10 % >10 % |
0.0 20.1 |
10.0 30.4 |
26.1 20.7 |
0.087 0.890 |
0.392 |
| G-CSF | 24.3 | ≤10 % >10 % |
27.5 58.9 |
37.9 56.8 |
145.3 93.9 |
0.003 0.191 |
0.002 |
| GM-CSF | 0.0 | ≤10 % >10 % |
30.1 52.3 |
28.3 18.6 |
35.4 35.0 |
0.658 0.110 |
0.604 |
| IFN-γ | 118.4 | ≤10 % >10 % |
106.1 259.9 |
212.3 163.6 |
219.6 230.3 |
0.436 0.601 |
0.993 |
| IP-10 | 1152 | ≤10 % >10 % |
1914 656.4 |
883.7 810.9 |
680.6 1283 |
0.587 0.164 |
0.736 |
| MCP-1 | 13.6 | ≤10 % >10 % |
33.4 94.5 |
98.2 372.6 |
174.3 234.9 |
0.018 0.117 |
0.007 |
| MIP-1α | 0.0 | ≤10 % >10 % |
0.0 3.7 |
1.8 0.1 |
1.8 8.3 |
0.439 0.230 |
0.281 |
| MIP-1β | 37.4 | ≤10 % >10 % |
60.8 29.2 |
46.0 46.2 |
23.6 78.6 |
0.532 0.637 |
0.612 |
| PDGF | 6575 | ≤10 % >10 % |
446.0 2316 |
792.6 1484 |
1301 3679 |
0.633 0.334 |
0.902 |
| TNF-α | 0.0 | ≤10 % >10 % |
32.9 55.5 |
71.0 69.5 |
105.1 133.2 |
0.061 0.324 |
0.099 |
| VEGF | 1.2 | ≤10 % >10 % |
8.3 80.7 |
26.8 41.4 |
47.5 33.4 |
0.221 0.358 |
0.988 |
Adjusted for Age and % TBSA; all values in pg/ml plasma
Plasma cytokine, chemokine, and growth factors as related to mortality
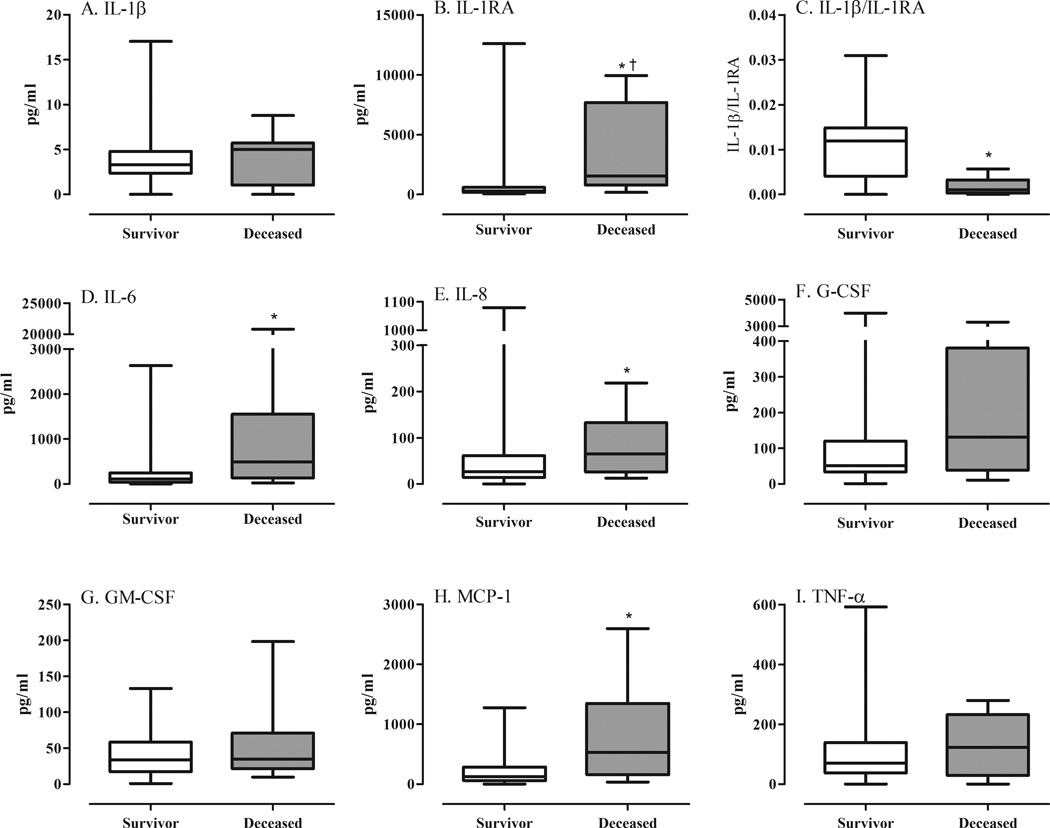
Differences in plasma immune mediator concentrations in surviving and deceased patients were identified (Table 3). Those most researched in burns, trauma, and sepsis, and/or which we found at physiologically relevant levels, are demonstrated in Figure 2. Plasma concentrations of IL-1RA, IL-6, IL-8, IL-15, Eotaxin, and MCP-1 were higher in deceased patients compared with survivors (p<0.05 for all), while IL-4 and IL-7 were lower (p<0.05). After adjusting for the effects of age, % TBSA, and inhalation injury grade, plasma IL-1RA remained significantly associated with mortality (OR 3.12, 95% CI 1.03–9.44). Likewise, deceased patients had a much lower IL-1β to IL-1RA ratio than did survivors (p=0.0001) (Figure 2). Of note is that the timing of plasma sample collection did not differ between survivors and non-survivors (p=0.479).
Table 3.
Plasma cytokine, chemokine, and growth factors as related to mortality
| Immune Mediator |
Survivors n=51 |
Deceased n=13 |
p Value |
|---|---|---|---|
| C5a | 516.9 (303.5–1996) | 1453 (628.2–2295) | 0.154 |
| IL-1β | 3.3 (2.4–4.8) | 5.0 (1.0–5.7) | 0.634 |
| IL-1RA | 280.1 (191.6–584.0) | 1562 (781.9–7670) | <0.001* |
| IL-2 | 7.4 (3.1–13.1) | 9.8 (3.2–17.0) | 0.531 |
| IL-4 | 4.6 (2.0–10.2) | 1.9 (1.2–4.5) | 0.045 |
| IL-5 | 2.3 (1.2–4.0) | 1.4 (0.9–4.3) | 0.381 |
| IL-6 | 114.2 (44.0–250.2) | 491.0 (136.8–1552) | 0.005 |
| IL-7 | 11.4 (5.5–35.9) | 5.2 (3.4–11.5) | 0.044 |
| IL-8 | 26.8 (14.2–61.2) | 65.1 (26.2–132.6) | 0.028 |
| IL-9 | 43.3 (20.1–61.8) | 34.2 (22.9–69.0) | 0.778 |
| IL-10 | 11.5 (6.0–19.2) | 16.0 (11.2–42.1) | 0.142 |
| IL-12 | 14.6 (7.9–25.3) | 10.8 (6.4–16.8) | 0.390 |
| IL-13 | 5.0 (3.8–7.3) | 5.8 (4.0–8.6) | 0.515 |
| IL-15 | 10.4 (5.3–14.8) | 13.6 (10.5–32.1) | 0.038 |
| IL-17 | 18.0 (0–76.5) | 16.2 (12.2–43.7) | 0.724 |
| Eotaxin | 173.0 (111.4–238.1) | 233.4 (181.3–342.8) | 0.024 |
| FGF-basic | 15.7 (0–36.7) | 18.9 (0–41.8) | 0.679 |
| G-CSF | 50.9 (33.6–119.9) | 131.3 (38.3–380.5) | 0.072 |
| GM-CSF | 34.0 (17.5–58.5) | 34.8 (21.7–71.0) | 0.433 |
| IFN-γ | 218.2 (118.0–319.4) | 246.8 (99.3–376.0) | 1.000 |
| IP-10 | 818.2 (534.3–1341) | 1221 (479.0–2429) | 0.489 |
| MCP-1 | 129.3 (61.1–285.8) | 531.5 (158.8–1348) | 0.006 |
| MIP-1α | 2.3 (0–10.9) | 1.0 (0–10.6) | 0.663 |
| MIP-1β | 43.8 (21.0–72.4) | 70.5 (10.7–123.7) | 0.548 |
| PDGF | 1641 (493.5–5842) | 869.4 (514.5–5773) | 0.606 |
| TNF-α | 71.0 (37.9–138.9) | 123.3 (29.7–232.3) | 0.537 |
| VEGF | 41.3 (21.0–83.4) | 41.4 (15.1–75.8) | 0.701 |
Data represented as median (interquartile range); all values are pg/ml;
Statistically significant after adjusting for Age, % TBSA, and Inhalation injury grade (OR 3.12; 95% CI 1.03–9.44)
Figure 2.
Select immune mediators in the plasma of patients within 15 hours of burn and smoke inhalation injury, comparing survivors and deceased. IL-1RA (B), IL-6 (D), IL-8 (E), and MCP-1 (H) were increased in deceased patients while the ratio of IL-1β to IL-1RA was markedly decreased (*p<0.05). The concentrations of IL-1β (A), G-CSF (F), GM-CSF (G), and TNF-α (I) were statistically no different between groups. After adjusting for the effects of Age, % TBSA, and inhalation injury grade, the concentration of IL-1RA remained significantly associated with mortality (†OR 3.12; 95% CI 1.03–9.44).
Correlations of plasma immune mediators with injury severity and outcomes
Of all the immune mediators measured in the BALF and the plasma, only the plasma concentration of macrophage inhibitory protein (MIP)-1β correlated with the time of sample collection (r=−0.35; p=0.005). In addition to being associated with burn patient mortality, numerous plasma immune mediator concentrations correlated significantly with injury severity and outcomes. Those mediators most studied in burn care and with the strongest correlations to % TBSA, inhalation injury grade, 24-hour fluid resuscitation, revised Baux score, Denver score, SOFA score, initial 48-hour P:F nadir, hospital LOS, and mortality are demonstrated in Table 4. Again, high levels of plasma IL-1RA were the most associated with injury severity and numerous outcomes measures, though plasma IL-6, IL-8, and MCP-1 had similarly significant correlations. Finally, the ratio of plasma IL-1β to IL-1RA was negatively correlated with Baux score (r=−0.380, p=0.002), revised Baux score (r=−0.372, p=0.003), Denver score (r=−0.250, p=0.046), 72h fluid resuscitation (r=−0.278, p=0.030), and mortality (r=−0.487, p<0.0001).
Table 4.
Spearman correlation of select plasma immune mediators with injury severity and outcomes
| % TBSA | Inhalation Injury Grade |
Initial 24 hr Fluid |
Revised Baux |
Denver Score |
SOFA Score |
Initial 48hr P:F Nadir |
Hospital LOS |
Mortality | ||
|---|---|---|---|---|---|---|---|---|---|---|
| IL-1β | p | 0.274 0.028 |
0.146 0.249 |
0.398 0.001 |
0.197 0.119 |
0.185 0.143 |
0.232 0.065 |
−0.037 0.774 |
0.128 0.312 |
0.061 0.632 |
| IL-1RA | p | 0.411 <0.001 |
0.282 0.024 |
0.512 <0.0001 |
0.495 <0.0001 |
0.424 <0.001 |
0.449 <0.001 |
−0.171 0.182 |
0.130 0.307 |
0.453 <0.001 |
| IL-6 | p | 0.233 0.068 |
0.438 <0.001 |
0.444 <0.001 |
0.429 <0.001 |
0.463 <0.001 |
0.438 <0.001 |
−0.267 0.038 |
0.269 0.035 |
0.364 0.004 |
| IL-8 | p | 0.225 0.074 |
0.365 0.003 |
0.358 0.004 |
0.340 0.006 |
0.354 0.004 |
0.344 0.005 |
−0.183 0.150 |
0.282 0.024 |
0.279 0.026 |
| IL-10 | p | 0.330 0.008 |
0.200 0.113 |
0.389 0.002 |
0.233 0.064 |
0.320 0.010 |
0.305 0.014 |
−0.172 0.177 |
0.217 0.085 |
0.186 0.141 |
| IL-15 | p | 0.193 0.126 |
0.195 0.123 |
0.280 0.025 |
0.309 0.013 |
0.238 0.058 |
0.195 0.123 |
−0.093 0.466 |
0.110 0.389 |
0.263 0.036 |
| G-CSF | p | 0.195 0.123 |
0.418 <0.001 |
0.302 0.015 |
0.310 0.013 |
0.376 0.002 |
0.370 0.003 |
−0.243 0.055 |
0.307 0.014 |
0.228 0.070 |
| GM-CSF | p | 0.189 0.189 |
0.056 0.697 |
0.082 0.571 |
0.123 0.394 |
0.069 0.633 |
0.125 0.386 |
−0.083 0.568 |
0.211 0.141 |
0.114 0.432 |
| IFN-γ | p | 0.123 0.334 |
0.010 0.936 |
0.008 0.949 |
0.029 0.818 |
0.163 0.200 |
0.180 0.154 |
−0.141 0.269 |
0.209 0.098 |
0.001 0.993 |
| MCP-1 | p | 0.302 0.015 |
0.368 0.003 |
0.499 <0.0001 |
0.444 <0.001 |
0.420 <0.001 |
0.417 <0.001 |
−0.274 0.030 |
0.219 0.082 |
0.348 0.005 |
| MIP-1β | p | 0.267 0.033 |
0.044 0.731 |
0.264 0.035 |
0.175 0.168 |
0.097 0.447 |
0.118 0.353 |
−0.082 0.521 |
0.026 0.840 |
0.077 0.547 |
| PDGF | p | 0.205 0.145 |
−0.058 0.683 |
0.377 0.006 |
−0.002 0.989 |
−0.137 0.333 |
−0.005 0.973 |
0.276 0.050 |
0.186 0.188 |
−0.074 0.603 |
| TNF-α | p | 0.145 0.252 |
0.239 0.057 |
0.283 0.024 |
0.266 0.034 |
0.234 0.063 |
0.167 0.187 |
−0.135 0.292 |
0.121 0.340 |
0.079 0.536 |
| VEGF | p | 0.169 0.181 |
−0.020 0.876 |
0.097 0.447 |
0.013 0.917 |
0.050 0.697 |
0.135 0.288 |
−0.104 0.416 |
0.111 0.383 |
−0.049 0.698 |
Each cell represents Spearman’s rank correlation coefficient (top) and p-value (bottom)
Revised Baux score = Age + % TBSA + 17*Inhalation injury (0=No, 1=Yes)
LOS, length of stay; SOFA, sequential organ failure assessment; TBSA, total body surface area burn.
Discussion
The results of our study are threefold. First, numerous routine clinical parameters and indicators of outcome were associated with mortality, which substantiates the generalizability of our data.1,2,8,11– 16 Second, the severity of smoke inhalation injury appeared to induce systemically measureable effects, in that plasma concentrations of IL-1RA, IL-6, IL-8, G-CSF, and MCP-1 were increased with worse grades of inhalation injury. And third, several plasma immune mediators measured early after injury were associated with mortality, including IL-1RA, IL-4, IL-6, IL-7, IL-8, IL-15, Eotaxin, and MCP-1. Of these, increased plasma concentrations of anti-inflammatory IL-1RA (and therefore a reduced ratio of IL-1β to IL-1RA) had the strongest correlations with injury severity, outcomes measures, and mortality, which may have contributed to the blunted pulmonary immune response we previously found in non-survivors.5
Abundant animal and human studies have investigated the immune response to burns, trauma, and sepsis, implicating both the pro- and anti-inflammatory response as potential determinants of outcome and survival. To date, however, no single mediator, be it pro- or anti-inflammatory, has been identified as a successful candidate for immunomodulation, despite attempts in phase I, phase II, and even phase III clinical trials.17–39 These findings suggest either that our understanding of the immune response to burns, trauma, and sepsis is incomplete, or that our approach to immunomodulation of inherently heterogenous patients with a single mediator is inappropriate. Indeed, our study affirms the recent one of Xiao et al, which indicated a simultaneous induction of pro- and anti-inflammatory processes after injury.3 These data suggests that an appreciation for the balance of inflammatory signaling may be most crucial to efforts at modulating the immune profile after burn injury.
The current study is important as it is only the second of which we are aware as having identified inhalation injury as inducing systemic immune mediator changes. Previously, Rodriguez et al reported that the addition of inhalation injury magnified the amount of circulating IL-8 detected.40 In our study, the systemic effects of inhalation injury are evidenced by numerous plasma immune mediator concentrations being elevated in response to more severe grades of inhalation injury, even after adjusting for relevant variables such as age and % TBSA. These findings are in contrast to those of Finnerty et al, who also tested the hypothesis that inhalation injury augments the systemic inflammatory response.41 In a study of 72 children, they failed to demonstrate that the presence of inhalation injury altered the systemic immune profile as determined by changes in plasma immune mediator concentrations. However, as compared to our study of adults with a mean % TBSA burn of 20%, the sample population in the study of Finnerty et al consisted of children with and without inhalation injury, who had a mean % TBSA burn of approximately 60% or greater. Not only then was the size of burn injury considerably different in our study, so was the population age, which is known to influence the immune response to injury and is a factor actively investigated at our own institution. Therefore, comparisons between our study and theirs may be difficult, especially as the study of Finnerty et al did not take into account the severity of inhalation injury, which, as we have observed, is an important determinant of the systemic immune response to burns combined with smoke inhalation. Further yet, plasma and BALF samples were collected in our study in a well-defined post-injury window of approximately 5–10 hours, thus minimizing the effect of time since injury on immune mediator concentrations.
Having previously found that the pulmonary immune response is blunted in non-survivors of burn and smoke inhalation injury,5 we also sought to identify a circulating mediator of anti-inflammation potentially responsible for this phenomenon. Of all 28 plasma immunomodulating proteins that we examined within 15 hours of injury, an elevated plasma concentration of IL-1RA was most predictive of mortality, independent of age, % TBSA, and inhalation injury grade. This finding provides a plausible explanation for the pulmonary immune anergy that we previously identified in non-survivors of burn and inhalation injury.5 Moreover, IL-1RA, and the imbalance of IL-1α/β to IL-1RA signaling, could serve as a legitimate target for future research.42 For instance, in a study of 31 burn-injured patients, Cannon et al found that low circulating IL-1β was associated with mortality.43 As other studies had noted a similar role of IL-1α/β in human and animal survival from sepsis,44–49 Cannon et al concluded that IL-1β is an essential component of the host defense to injury. Indeed, Kupper et al found that defective antigen presentation after burn injury could be restored with IL-1β,50 and Mancilla et al found that high doses of IL-1RA decreased survival in an animal model of Klebsiella infection.47 Finally, more recent studies in burns, trauma, and sepsis validate the hypothesis that insufficient IL-1α/β or excessive IL-1RA results in systemic immune dysfunction that may be detrimental to survival.51–53 As phase III trials investigating IL-1RA therapy in severe sepsis have been disappointing,19 one may surmise then that some degree of IL-1 signaling could be important for various facets of the immune response to injury and sepsis, such as B- and T-cell proliferation, activation of neutrophils and macrophages, and release of hematopoietic factors.54–66 Yet, data are conflicting regarding the role of IL-1 after burn injury, particularly as mice deficient for IL-1 receptor associated kinase-1 have been shown to have less cardiac contractile dysfunction after burns than their wild-type counterparts.67 Additionally, IL-1RA synthesis may be differentially regulated amongst individuals, which could explain why some have an enhanced production of this anti-inflammatory cytokine, independent from even age, % TBSA, and inhalation injury grade.68 Future studies are warranted to examine the role of IL-1RA as a mediator of subsequent burn patient outcome by understanding its mechanistic role in immune signaling, which could very well involve IL-6 and IL-8 through mitogen-activated protein kinase and NF-κB pathways, in addition to steric inhibition of IL-1α/β binding to their membrane-bound receptors.69 Alternatively, early measurement of IL-1RA may serve as an ideal candidate for predicting burn patient outcomes.
Several other cytokines and chemokines which our study identified as correlating with morbidity and mortality have been frequently implicated as important mediators of injury response and outcome, most notably IL-6 and IL-8. For instance, we found that plasma concentrations of IL-6 were elevated early after injury in non-survivors, and that higher levels of IL-6 correlated with inhalation injury grade, fluid resuscitation, pulmonary dysfunction, and subsequent multiple organ failure and death. Similar findings to ours have been routinely published in other studies of burns, trauma, and sepsis, whereby IL-6 is elevated in those at greatest risk for poor outcomes.70–78 As with many mediators of the immune response to injury, the precise role of IL-6 (which has demonstrated both pro- and anti-inflammatory effects) other than as a marker of disease severity remains unclear.79–84 Along these lines, knock-out (KO) studies in mice have demonstrated IL-6 to be important for the stimulation of hepatic glutamine transport after burn injury, and in mediating myocardial dysfunction after burn injury in the setting of sepsis.67,85,86 Moreover, animal models of burn injury using IL-6 deficient mice and wild-type mice given neutralizing concentrations of anti-IL-6 antibody have suggested beneficial effects to cell-mediated immunity, cytokine production, and organ function with reduced or absent IL-6.87–89
Our study also appears to confirm IL-8 as an important determinant and/or marker of burn patient outcome. As a chemokine, IL-8 primarily serves to recruit neutrophils to the site of injury and to activate their expression of surface adhesion molecules, degranulation, and oxidative-burst capacity. Likewise, IL-8 is frequently identified as a correlate to morbidity and mortality in animals as well as patients with burns, trauma, and sepsis.34,35,40,76,90–92 As with IL-1RA, IL-6, and the balance of pro- and anti-inflammation, further studies are required to better elucidate the role for locally produced and circulating IL-8 as an effecter of burn patient outcomes. Such investigations are ongoing at our burn center and research laboratories, with the aim through translational studies to further refine our understanding of the immune response to injury, especially among specific groups who vary in terms of gender, age, and alcohol intoxication. These investigations will continue to involve injury-response modification through antibody suppression, transgenic animal models, and elucidation of intracellular signaling pathways.
We acknowledge important limitations to our study. First, though a sample size of 72 patients is relatively robust, the specificity of our results is hindered by the heterogeneity of the study population, particularly along the lines of age and % TBSA. However, our regression approach accounted for the most important effectors of outcome. Second, our study has an inherent selection bias, in that only those with a suspected inhalation injury were candidates for enrollment, thereby excluding many with skin burns alone. Nonetheless, many widely accepted correlates with outcomes were applicable to our study population, such as age, % TBSA, and measures of organ dysfunction, rendering our results interpretable and generalizable to the majority of burn-injured patients. Third, the data are limited to the acute phase of injury response, in that the concentrations of plasma immune mediators were assessed within 15 hours of injury. Therefore, our study neither examines the immune response to injury during the full resuscitation phase nor over the course of hospitalization. Lastly, our study does not provide mechanistic data that may hold additional clues as to how seemingly important mediators of injury, such as IL-1β, IL-1RA, IL-6, and IL-8, may induce their effects. We hope that continued patient enrollment, collaboration with other centers, and additional bench-side studies will advance our understanding of the immune response to burn injury, trauma, and sepsis.
We conclude that the early anti-inflammatory response to burn and smoke inhalation injury may play a significant role in subsequent outcome, and that it could still serve as a potential target for therapeutic intervention. We also conclude that the severity of smoke inhalation injury evokes systemically-reaching effects that alter the immune response to injury, arguing in favor of treating inhalation injury in a graded manner.
Acknowledgements
Without the dedicated assistance of research nurses, clinical nurses, respiratory therapists, and staff in the burn ICU, this prospectively designed study would not be possible. We especially thank Ellen L. Burnham, MD, MS, from the University of Colorado School of Medicine, for her assistance with this study and in the manuscript drafting process.
Funded by the National Institutes of Health [T32 GM008750 (RLG), T32 AA013527 (EJK), R01 AA012034 (EJK), P30 AA019373 (EJK)], the Department of Defense (W81XWH-09-1-0619), and the Dr. Ralph and Marian C. Falk Medical Research Trust (EJK)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Pruitt BA, Wolf SE, Mason AD. Total Burn Care. 3rd Edition. Philadelphia: Elsevier; 2007. Epidemiological, demographic, and outcome characteristics of burn injury; pp. 14–32. [Google Scholar]
- 2.Latenser BA, Miller SF, Bessey PQ, et al. National Burn Repository 2006: a ten-year review. J Burn Care Res. 2007;28(5):635–658. doi: 10.1097/BCR.0B013E31814B25B1. [DOI] [PubMed] [Google Scholar]
- 3.Xiao W, Mindrinos MN, Seok J, et al. A genomic storm in critically injured humans. J Exp Med. 2011;208(13):2581–2590. doi: 10.1084/jem.20111354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Albright JM, Davis CS, Bird MD, et al. The acute pulmonary inflammatory response to the graded severity of smoke inhalation injury. Crit Care Med. 2012;40(4):1113–1121. doi: 10.1097/CCM.0b013e3182374a67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Davis CS, Albright JM, Carter SR, et al. Early pulmonary immune hyporesponsiveness is associated with mortality after burn and smoke inhalation injury. J Burn Care Res. 2012;33(1):26–35. doi: 10.1097/BCR.0b013e318234d903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Osler T, Glance LG, Hosmer DW. Simplified estimates of the probability of death after burn injuries: extending and updating the baux score. J Trauma. 2010;68(3):690–697. doi: 10.1097/TA.0b013e3181c453b3. [DOI] [PubMed] [Google Scholar]
- 7.Greenhalgh DG, Saffle JR, Holmes JH, et al. American Burn Association consensus conference to define sepsis and infection in burns. J Burn Care Res. 2007;28(6):776–790. doi: 10.1097/BCR.0b013e3181599bc9. [DOI] [PubMed] [Google Scholar]
- 8.Endorf FW, Gamelli RL. Inhalation injury, pulmonary perturbations, and fluid resuscitation. J Burn Care Res. 2007;28(1):80–83. doi: 10.1097/BCR.0B013E31802C889F. [DOI] [PubMed] [Google Scholar]
- 9.Bird MD, Zahs A, Deburghgraeve C, et al. Decreased pulmonary inflammation following ethanol and burn injury in mice deficient in TLR4 but not TLR2 signaling. Alcohol Clin Exp Res. 2010;34(10):1733–1741. doi: 10.1111/j.1530-0277.2010.01260.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ivy ME, Atweh NA, Palmer J, et al. Intra-abdominal hypertension and abdominal compartment syndrome in burn patients. J Trauma. 2000;49(3):387–391. doi: 10.1097/00005373-200009000-00001. [DOI] [PubMed] [Google Scholar]
- 11.Miller SF, Bessey P, Lentz CW, et al. ABA NBR Committee. National burn repository 2007 report: a synopsis of the 2007 call for data. J Burn Care Res. 2008;29(6):862–870. doi: 10.1097/BCR.0b013e31818cb046. discussion 871. [DOI] [PubMed] [Google Scholar]
- 12.Hassan Z, Wong JK, Bush J, et al. Assessing the severity of inhalation injuries in adults. Burns. 2010;36(2):212–216. doi: 10.1016/j.burns.2009.06.205. [DOI] [PubMed] [Google Scholar]
- 13.Klein MB, Hayden D, Elson C, et al. The association between fluid administration and outcome following major burn: a multicenter study. Ann Surg. 2007;245(4):622–628. doi: 10.1097/01.sla.0000252572.50684.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mosier MJ, Pham TN, Klein MB, et al. Early acute kidney injury predicts progressive renal dysfunction and higher mortality in severely burned adults. J Burn Care Res. 2010;31(1):83–92. doi: 10.1097/BCR.0b013e3181cb8c87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Coca SG, Bauling P, Schifftner T, et al. Contribution of acute kidney injury toward morbidity and mortality in burns: a contemporary analysis. Am J Kidney Dis. 2007;49(4):517–523. doi: 10.1053/j.ajkd.2006.12.018. [DOI] [PubMed] [Google Scholar]
- 16.Mosier MJ, Pham TN, Park DR, et al. Predictive value of bronchoscopy in assessing the severity of inhalation injury. J Burn Care Res. 2012;33(1):65–73. doi: 10.1097/BCR.0b013e318234d92f. [DOI] [PubMed] [Google Scholar]
- 17.Fisher CJ, Jr, Dhainaut JF, Opal SM, et al. Recombinant human interleukin 1 receptor antagonist in the treatment of patients with sepsis syndrome. Results from a randomized, double-blind, placebo-controlled trial. Phase III rhIL-1ra Sepsis Syndrome Study Group. JAMA. 1994;271(23):1836–1843. [PubMed] [Google Scholar]
- 18.Fisher CJ, Jr, Agosti JM, Opal SM, et al. Treatment of septic shock with the tumor necrosis factor receptor:Fc fusion protein. The Soluble TNF Receptor Sepsis Study Group. N Engl J Med. 1996;334(26):1697–1702. doi: 10.1056/NEJM199606273342603. [DOI] [PubMed] [Google Scholar]
- 19.Opal SM, Fisher CJ, Jr, Dhainaut JF, et al. Confirmatory interleukin-1 receptor antagonist trial in severe sepsis: a phase III, randomized, double-blind, placebo-controlled, multicenter trial. The Interleukin-1 Receptor Antagonist Sepsis Investigator Group. Crit Care Med. 1997;25(7):1115–1124. doi: 10.1097/00003246-199707000-00010. [DOI] [PubMed] [Google Scholar]
- 20.Reinhart K, Karzai W. Anti-tumor necrosis factor therapy in sepsis: update on clinical trials and lessons learned. Crit Care Med. 2001;29(7 Suppl):S121–S125. doi: 10.1097/00003246-200107001-00037. [DOI] [PubMed] [Google Scholar]
- 21.Deans KJ, Haley M, Natanson C, et al. Novel therapies for sepsis: a review. J Trauma. 2005;58(4):867–874. doi: 10.1097/01.ta.0000158244.69179.94. [DOI] [PubMed] [Google Scholar]
- 22.Schröder O, Laun RA, Held B, et al. Association of interleukin-10 promoter polymorphism with the incidence of multiple organ dysfunction following major trauma: results of a prospective pilot study. Shock. 2004;21(4):306–310. doi: 10.1097/00024382-200404000-00003. [DOI] [PubMed] [Google Scholar]
- 23.Schroeder O, Schulte KM, Schroeder J, et al. The-1082 interleukin-10 polymorphism is associated with acute respiratory failure after major trauma: a prospective cohort study. Surgery. 2008;143(2):233–242. doi: 10.1016/j.surg.2007.07.040. [DOI] [PubMed] [Google Scholar]
- 24.Huebinger RM, Rivera-Chavez F, Chang LY, et al. IL-10 polymorphism associated with decreased risk for mortality after burn injury. J Surg Res. 2010;164(1):e141–e145. doi: 10.1016/j.jss.2010.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gu W, Zeng L, Zhou J, et al. Clinical relevance of 13 cytokine gene polymorphisms in Chinese major trauma patients. Intensive Care Med. 2010;36(7):1261–1265. doi: 10.1007/s00134-010-1797-5. [DOI] [PubMed] [Google Scholar]
- 26.McDaniel DO, Hamilton J, Brock M, et al. Molecular analysis of inflammatory markers in trauma patients at risk of postinjury complications. J Trauma. 2007;63(1):147–157. doi: 10.1097/TA.0b013e31806bf0ab. discussion 157–8. [DOI] [PubMed] [Google Scholar]
- 27.Ami K, Kinoshita M, Yamauchi A, et al. IFN-gamma production from liver mononuclear cells of mice in burn injury as well as in postburn bacterial infection models and the therapeutic effect of IL-18. J Immunol. 2002;169(8):4437–4442. doi: 10.4049/jimmunol.169.8.4437. [DOI] [PubMed] [Google Scholar]
- 28.Göebel A, Kavanagh E, Lyons A, et al. Injury induces deficient interleukin-12 production, but interleukin-12 therapy after injury restores resistance to infection. Ann Surg. 2000;231(2):253–261. doi: 10.1097/00000658-200002000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O'Suilleabhain C, O'Sullivan ST, Kelly JL, et al. Interleukin-12 treatment restores normal resistance to bacterial challenge after burn injury. Surgery. 1996;120(2):290–296. doi: 10.1016/s0039-6060(96)80300-x. [DOI] [PubMed] [Google Scholar]
- 30.Lyons A, Goebel A, Mannick JA, et al. Protective effects of early interleukin 10 antagonism on injury-induced immune dysfunction. Arch Surg. 1999;134(12):1317–1323. doi: 10.1001/archsurg.134.12.1317. discussion 1324. [DOI] [PubMed] [Google Scholar]
- 31.O'Riordain MG, O'Riordain DS, Molloy RG, et al. Dosage and timing of anti-TNF-alpha antibody treatment determine its effect of resistance to sepsis after injury. J Surg Res. 1996;64(1):95–101. doi: 10.1006/jsre.1996.0312. [DOI] [PubMed] [Google Scholar]
- 32.Gamelli RL, He LK, Liu H. Recombinant human granulocyte colony-stimulating factor treatment improves macrophage suppression of granulocyte and macrophage growth after burn and burn wound infection. J Trauma. 1995;39(6):1141–1146. doi: 10.1097/00005373-199512000-00023. discussion 1146-7. [DOI] [PubMed] [Google Scholar]
- 33.Molloy RG, Holzheimer R, Nestor M, et al. Granulocyte-macrophage colony-stimulating factor modulates immune function and improves survival after experimental thermal injury. Br J Surg. 1995;82(6):770–776. doi: 10.1002/bjs.1800820618. [DOI] [PubMed] [Google Scholar]
- 34.Laffon M, Pittet JF, Modelska K, et al. Interleukin-8 mediates injury from smoke inhalation to both the lung endothelial and the alveolar epithelial barriers in rabbits. Am J Respir Crit Care Med. 1999;160(5 Pt 1):1443–1449. doi: 10.1164/ajrccm.160.5.9901097. [DOI] [PubMed] [Google Scholar]
- 35.Sakurai H, Soejima K, Schmalstieg FC, et al. Inhibition of lung permeability changes after burn and smoke inhalation by an anti-interleukin-8 antibody in sheep. Surg Today. 2009;39(5):399–406. doi: 10.1007/s00595-008-3879-3. [DOI] [PubMed] [Google Scholar]
- 36.Polk HC, Jr, Cheadle WG, Livingston DH, et al. A randomized prospective clinical trial to determine the efficacy of interferon-gamma in severely injured patients. Am J Surg. 1992;163(2):191–196. doi: 10.1016/0002-9610(92)90099-d. [DOI] [PubMed] [Google Scholar]
- 37.Dries DJ. Interferon gamma in trauma-related infections. Intensive Care Med. 1996;22(Suppl 4):S462–S467. doi: 10.1007/BF01743725. [DOI] [PubMed] [Google Scholar]
- 38.Wasserman D, Ioannovich JD, Hinzmann RD, et al. Interferon-gamma in the prevention of severe burn-related infections: a European phase III multicenter trial. The Severe Burns Study Group. Crit Care Med. 1998;26(3):434–439. doi: 10.1097/00003246-199803000-00010. [DOI] [PubMed] [Google Scholar]
- 39.Park KH, Lee KH, Kim H, et al. The anti-inflammatory effects of ulinastatin in trauma patients with hemorrhagic shock. J Korean Med Sci. 2010;25(1):128–134. doi: 10.3346/jkms.2010.25.1.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rodriguez JL, Miller CG, Garner WL, et al. Correlation of the local and systemic cytokine response with clinical outcome following thermal injury. J Trauma. 1993;34(5):684–694. doi: 10.1097/00005373-199305000-00011. discussion 694-5. [DOI] [PubMed] [Google Scholar]
- 41.Finnerty CC, Herndon DN, Jeschke MG. Inhalation injury in severely burned children does not augment the systemic inflammatory response. Crit Care. 2007;11(1):R22. doi: 10.1186/cc5698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Drost AC, Larsen B, Aulick LH. The effects of thermal injury on serum interleukin 1 activity in rats. Lymphokine Cytokine Res. 1993;12(3):181–185. [PubMed] [Google Scholar]
- 43.Cannon JG, Friedberg JS, Gelfand JA, et al. Circulating interleukin-1 beta and tumor necrosis factor-alpha concentrations after burn injury in humans. Crit Care Med. 1992;20(10):1414–1419. doi: 10.1097/00003246-199210000-00009. [DOI] [PubMed] [Google Scholar]
- 44.Cannon JG, Tompkins RG, Gelfand JA, et al. Circulating interleukin-1 and tumor necrosis factor in septic shock and experimental endotoxin fever. J Infect Dis. 1990;161(1):79–84. doi: 10.1093/infdis/161.1.79. [DOI] [PubMed] [Google Scholar]
- 45.Luger A, Graf H, Schwarz HP, et al. Decreased serum interleukin 1 activity and monocyte interleukin 1 production in patients with fatal sepsis. Crit Care Med. 1986;14(5):458–461. doi: 10.1097/00003246-198605000-00004. [DOI] [PubMed] [Google Scholar]
- 46.Munoz C, Carlet J, Fitting C, et al. Dysregulation of in vitro cytokine production by monocytes during sepsis. J Clin Invest. 1991;88(5):1747–1754. doi: 10.1172/JCI115493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mancilla J, García P, Dinarello CA. The interleukin-1 receptor antagonist can either reduce or enhance the lethality of Klebsiella pneumoniae sepsis in newborn rats. Infect Immun. 1993;61(3):926–932. doi: 10.1128/iai.61.3.926-932.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Silver GM, Gamelli RL, O'Reilly M, Hebert JC. The effect of interleukin 1 alpha on survival in a murine model of burn wound sepsis. Arch Surg. 1990;125(7):922–925. doi: 10.1001/archsurg.1990.01410190120020. [DOI] [PubMed] [Google Scholar]
- 49.O'Reilly M, Silver GM, Davis JH, et al. Interleukin 1 beta improves survival following cecal ligation and puncture. J Surg Res. 1992;52(5):518–522. doi: 10.1016/0022-4804(92)90321-p. [DOI] [PubMed] [Google Scholar]
- 50.Kupper TS, Green DR, Durum SK, Baker CC. Defective antigen presentation to a cloned T helper cell by macrophages from burned mice can be restored with interleukin-1. Surgery. 1985;98(2):199–206. [PubMed] [Google Scholar]
- 51.de Pablo R, Monserrat J, Reyes E, et al. Mortality in patients with septic shock correlates with anti-inflammatory but not proinflammatory immunomodulatory molecules. J Intensive Care Med. 2011;26(2):125–132. doi: 10.1177/0885066610384465. [DOI] [PubMed] [Google Scholar]
- 52.Tadros T, Traber DL, Heggers JP, Herndon DN. Effects of interleukin-1alpha administration on intestinal ischemia and reperfusion injury, mucosal permeability, and bacterial translocation in burn and sepsis. Ann Surg. 2003;237(1):101–109. doi: 10.1097/00000658-200301000-00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Erdag G, Morgan JR. Interleukin-1alpha and interleukin-6 enhance the antibacterial properties of cultured composite keratinocyte grafts. Ann Surg. 2002;235(1):113–124. doi: 10.1097/00000658-200201000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kampschmidt RF. Infection, inflammation, and interleukin 1 (IL-1) Lymphokine Res. 1983;2(3):97–102. [PubMed] [Google Scholar]
- 55.Oppenheim JJ, Charon JA, Luger TA. Evidence for an in vivo inflammatory role of interleukin 1 (IL 1) Transplant Proc. 1982;14(3):553–555. [PubMed] [Google Scholar]
- 56.Lipsky PE. Role of interleukin-1 in human B-cell activation. Contemp Top Mol Immunol. 1985;10:195–217. doi: 10.1007/978-1-4684-4838-2_8. [DOI] [PubMed] [Google Scholar]
- 57.Fibbe WE, van Damme J, Billiau A, et al. Interleukin 1 induces human marrow stromal cells in long-term culture to produce granulocyte colony-stimulating factor and macrophage colony-stimulating factor. Blood. 1988;71(2):430–435. [PubMed] [Google Scholar]
- 58.Czuprynski CJ, Brown JF. Recombinant murine interleukin-1 alpha enhancement of nonspecific antibacterial resistance. Infect Immun. 1987;55(9):2061–2065. doi: 10.1128/iai.55.9.2061-2065.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Czuprynski CJ, Brown JF, Young KM, et al. Effects of murine recombinant interleukin 1 alpha on the host response to bacterial infection. J Immunol. 1988;140(3):962–968. [PubMed] [Google Scholar]
- 60.Ozaki Y, Ohashi T, Minami A, Nakamura S. Enhanced resistance of mice to bacterial infection induced by recombinant human interleukin-1a. Infect Immun. 1987;55(6):1436–1440. doi: 10.1128/iai.55.6.1436-1440.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gladue R, Girard A, Newborg M. Enhanced antibacterial resistance in neutropenic mice treated with human recombinant interleukin-1 beta. Agents Actions. 1988;24(1–2):130–136. doi: 10.1007/BF01968091. [DOI] [PubMed] [Google Scholar]
- 62.Van der Meer JW, Helle M, Aarden L. Comparison of the effects of recombinant interleukin 6 and recombinant interleukin 1 on nonspecific resistance to infection. Eur J Immunol. 1989;19(2):413–416. doi: 10.1002/eji.1830190229. [DOI] [PubMed] [Google Scholar]
- 63.Van der Meer JW, Barza M, Wolff SM, Dinarello CA. A low dose of recombinant interleukin 1 protects granulocytopenic mice from lethal gram-negative infection. Proc Natl Acad Sci U S A. 1988;85(5):1620–1623. doi: 10.1073/pnas.85.5.1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Alexander HR, Doherty GM, Fraker DL, et al. Human recombinant interleukin-1 alpha protection against the lethality of endotoxin and experimental sepsis in mice. J Surg Res. 1991;50(5):421–424. doi: 10.1016/0022-4804(91)90018-h. [DOI] [PubMed] [Google Scholar]
- 65.Parker KP, Benjamin WR, Kaffka KL, Kilian PL. Presence of IL-1 receptors on human and murine neutrophils. Relevance to IL-1-mediated effects in inflammation. J Immunol. 1989;142(2):537–542. [PubMed] [Google Scholar]
- 66.Sullivan GW, Carper HT, Sullivan JA, et al. Both recombinant interleukin-1 (beta) and purified human monocyte interleukin-1 prime human neutrophils for increased oxidative activity and promote neutrophil spreading. J Leukoc Biol. 1989;45(5):389–395. doi: 10.1002/jlb.45.5.389. [DOI] [PubMed] [Google Scholar]
- 67.Thomas JA, Tsen MF, White DJ, Horton JW. IRAK contributes to burn-triggered myocardial contractile dysfunction. Am J Physiol Heart Circ Physiol. 2002;283(2):H829–H836. doi: 10.1152/ajpheart.00416.2001. [DOI] [PubMed] [Google Scholar]
- 68.Danis VA, Millington M, Hyland VJ, Grennan D. Cytokine production by normal human monocytes: inter-subject variation and relationship to an IL-1 receptor antagonist (IL-1Ra) gene polymorphism. Clin Exp Immunol. 1995;99(2):303–310. doi: 10.1111/j.1365-2249.1995.tb05549.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Garat C, Arend WP. Intracellular IL-1Ra type 1 inhibits IL-1-induced IL-6 and IL-8 production in Caco-2 intestinal epithelial cells through inhibition of p38 mitogen-activated protein kinase and NF-kappaB pathways. Cytokine. 2003;23(1–2):31–40. doi: 10.1016/s1043-4666(03)00182-0. [DOI] [PubMed] [Google Scholar]
- 70.Drost AC, Burleson DG, Cioffi WG, Jr, et al. Plasma cytokines following thermal injury and their relationship with patient mortality, burn size, and time postburn. J Trauma. 1993;35(3):335–339. doi: 10.1097/00005373-199309000-00001. [DOI] [PubMed] [Google Scholar]
- 71.Hack CE, De Groot ER, Felt-Bersma RJ, et al. Increased plasma levels of interleukin-6 in sepsis. Blood. 1989;74(5):1704–1710. [PubMed] [Google Scholar]
- 72.Gebhard F, Pfetsch H, Steinbach G, et al. Is interleukin 6 an early marker of injury severity following major trauma in humans? Arch Surg. 2000;135(3):291–295. doi: 10.1001/archsurg.135.3.291. [DOI] [PubMed] [Google Scholar]
- 73.S C, Mahadevan C, Thomas N, et al. Do baseline IL-6 levels predict post-operative morbidity after surgery (CABG) in cardiac patients? J Card Surg. 2006;21(5):521–522. doi: 10.1111/j.1540-8191.2006.00289.x. [DOI] [PubMed] [Google Scholar]
- 74.Tateishi Y, Oda S, Nakamura M, et al. Depressed heart rate variability is associated with high IL-6 blood level and decline in the blood pressure in septic patients. Shock. 2007;28(5):549–553. doi: 10.1097/shk.0b013e3180638d1. [DOI] [PubMed] [Google Scholar]
- 75.Drost AC, Burleson DG, Cioffi WG, Jr, et al. Plasma cytokines after thermal injury and their relationship to infection. Ann Surg. 1993;218(1):74–78. doi: 10.1097/00000658-199307000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gauglitz GG, Finnerty CC, Herndon DN, et al. Are serum cytokines early predictors for the outcome of burn patients with inhalation injuries who do not survive? Crit Care. 2008;12(3):R81. doi: 10.1186/cc6932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Schlüter B, König B, Bergmann U, et al. Interleukin 6--a potential mediator of lethal sepsis after major thermal trauma: evidence for increased IL-6 production by peripheral blood mononuclear cells. J Trauma. 1991;31(12):1663–1670. doi: 10.1097/00005373-199112000-00017. [DOI] [PubMed] [Google Scholar]
- 78.Ueyama M, Maruyama I, Osame M, et al. Marked increase in plasma interleukin-6 in burn patients. J Lab Clin Med. 1992;120(5):693–698. [PubMed] [Google Scholar]
- 79.Steensberg A, Fischer CP, Keller C, et al. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am J Physiol Endocrinol Metab. 2003;285(2):E433–E437. doi: 10.1152/ajpendo.00074.2003. [DOI] [PubMed] [Google Scholar]
- 80.Cicco NA, Lindemann A, Content J, et al. Inducible production of interleukin-6 by human polymorphonuclear neutrophils: role of granulocyte-macrophage colony-stimulating factor and tumor necrosis factor-alpha. Blood. 1990;75(10):2049–2052. [PubMed] [Google Scholar]
- 81.Xing Z, Gauldie J, Cox G, et al. IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest. 1998;101(2):311–320. doi: 10.1172/JCI1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Guo Y, Dickerson C, Chrest FJ, et al. Increased levels of circulating interleukin 6 in burn patients. Clin Immunol Immunopathol. 1990;54(3):361–371. doi: 10.1016/0090-1229(90)90050-z. [DOI] [PubMed] [Google Scholar]
- 83.Calandra T, Gerain J, Heumann D, et al. High circulating levels of interleukin-6 in patients with septic shock: evolution during sepsis, prognostic value, and interplay with other cytokines. The Swiss-Dutch J5 Immunoglobulin Study Group. Am J Med. 1991;91(1):23–29. doi: 10.1016/0002-9343(91)90069-a. [DOI] [PubMed] [Google Scholar]
- 84.Chishti AD, Shenton BK, Kirby JA, Baudouin SV. Neutrophil chemotaxis and receptor expression in clinical septic shock. Intensive Care Med. 2004;30(4):605–611. doi: 10.1007/s00134-004-2175-y. [DOI] [PubMed] [Google Scholar]
- 85.Pawlik TM, Carter EA, Bode BP, et al. Central role of interleukin-6 in burn induced stimulation of hepatic amino acid transport. Int J Mol Med. 2003;12(4):541–548. [PubMed] [Google Scholar]
- 86.Zhang H, Wang HY, Bassel-Duby R, et al. Role of interleukin-6 in cardiac inflammation and dysfunction after burn complicated by sepsis. Am J Physiol Heart Circ Physiol. 2007;292(5):H2408–H2416. doi: 10.1152/ajpheart.01150.2006. [DOI] [PubMed] [Google Scholar]
- 87.Plackett TP, Gamelli RL, Kovacs EJ. Gender-based differences in cytokine production after burn injury: a role of interleukin-6. J Am Coll Surg. 2010;210(1):73–78. doi: 10.1016/j.jamcollsurg.2009.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Fontanilla CV, Faunce DE, Gregory MS, et al. Anti-interleukin-6 antibody treatment restores cell-mediated immune function in mice with acute ethanol exposure before burn trauma. Alcohol Clin Exp Res. 2000;24(9):1392–1399. [PubMed] [Google Scholar]
- 89.Gomez CR, Nomellini V, Baila H, et al. Comparison of the effects of aging and IL-6 on the hepatic inflammatory response in two models of systemic injury: scald injury versus i.p. LPS administration. Shock. 2009;31(2):178–184. doi: 10.1097/SHK.0b013e318180feb8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Carvalho GL, Wakabayashi G, Shimazu M, et al. Anti-interleukin-8 monoclonal antibody reduces free radical production and improves hemodynamics and survival rate in endotoxic shock in rabbits. Surgery. 1997;122(1):60–68. doi: 10.1016/s0039-6060(97)90265-8. [DOI] [PubMed] [Google Scholar]
- 91.Hack CE, Hart M, van Schijndel RJ, et al. Interleukin-8 in sepsis: relation to shock and inflammatory mediators. Infect Immun. 1992;60(7):2835–2842. doi: 10.1128/iai.60.7.2835-2842.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Endo S, Inada K, Ceska M, et al. Plasma interleukin 8 and polymorphonuclear leukocyte elastase concentrations in patients with septic shock. J Inflamm. 1995;45(2):136–142. [PubMed] [Google Scholar]