Abstract
T cells must undergo two critical differentiation processes before they become competent effectors that can mediate actual immune responses. Progenitor T cells undergo defined stages of differentiation in the thymus, which include positive and negative selection, to generate a repertoire of T cells that will respond to foreign but not self antigens. When these immunocompetent T cells first migrate out of thymus into peripheral lymphoid tissues, they are naïve and are unable to mediate immune responses. However, upon antigen encounter, peripheral CD4+ naïve T cells undergo another differentiation process to become armed effector T cells including Th1, Th2, Th17 or regulatory T cells, all of which are capable of regulating immune responses. A canonical Wnt/β-catenin/T cell factor (TCF) pathway has been shown to regulate T cell differentiation in both the thymus and in peripheral lymphoid tissues. Dysfunction of this pathway at any stage of T cell differentiation could lead to severe autoimmunity including experimental autoimmune encephalomyelitis or immune deficiency. Understanding the role played by β-catenin/TCF-1 in T cell differentiation will facilitate our understanding of the mechanisms that regulate T cell function and assist in identifying novel therapy targets for treating both autoimmune and immune diseases. Therefore, in this review, we will focus on the function of β-catenin/TCF-1 pathway in the regulation of thymic and peripheral T cell differentiation processes.
1. Introduction
To become competent effector T cells that mediate actual immune responses, T cells must undergo two distinct differentiation processes that are carried out separately in thymus and the peripheral lymphoid tissues. T cell progenitors migrate from bone marrow into the thymus where they undergo defined stages of development that include positive and negative selection resulting in the elimination of more than 95% of immature T cells to avoid potential auto-immunity in the periphery. After selection, only T cells expressing T cell receptors (TCRs) that recognize foreign antigens presented by self-major histocompatibility complexes (MHCs) expressed on the surface of antigen presenting cells (APCs) are allowed to mature and migrate out of thymus into the periphery. This T cell development process in the thymus ensures that our immune system responds to foreign but not self antigens. T cells that migrate out of thymus into the peripheral lymphoid tissues are naïve; they are not competent to carry out actual immune responses unless they undergo an additional differentiation process. This process, which is initiated by the recognition and binding of antigens presented by APCs, arms naïve T cells to become competent in combating pathogens. Antigen engagement triggers TCR activation and the differentiation of CD4+ naïve T cells into effector T cells that include Th1, Th2, Th17 and regulatory T (Treg) effector T cells, all of which are capable of regulating actual immune responses. Both the thymic and peripheral differentiation processes are critical for the ability of T cells to mediate immune responses against foreign antigens. Dysfunction in either process will lead to immune deficiency or autoimmunity, including multiple sclerosis (MS) which is modeled by experimental autoimmune encephalomyelitis (EAE) in autoimmune disease studies. The canonical Wnt/β-catenin/TCF-1 signaling pathway in T cells is essential for regulation of both differentiation processes (1).
2. Wnt/β-catenin/TCF-1 pathway
Wnt is a family of secreted glycoproteins that control a variety of biological activities during development. Wnt signaling pathways include the canonical Wnt pathway, which is mediated by β-catenin/T cell factor (TCF), and non-canonical Wnt pathways, that involve intracellular calcium mobilization and the planar cell polarity pathway. Although non-canonical Wnt signaling has been implicated in some immunological processes such as regulation of thymocyte αβ-lineage survival (2), we will focus mainly on canonical Wnt signaling in T cell differentiation because it is the most significant and well studied pathway. Activation of the canonical Wnt signaling pathway is initiated by binding of one of the 19 different Wnt ligands, to the cell membrane-localized receptor complex composed of Frizzled and LDL receptor-related protein 5 (LRP5) or LRP6 (3). Ligand binding initiates a signaling cascade that dissociates the anchor protein, AXIN1, from a β-catenin destruction complex that contains glycogen synthase kinase 3β (GSK3β). In the absence of Wnt signals GSK3β phosphorylates β-catenin, which leads to its degradation via the ubiquitination. Wnt signaling and the dissociation of AXIN1 prevents β-catenin phosphorylation and degradation, allowing it to accumulate in the cytoplasm and then translocate into the nucleus. The ultimate effectors of the canonical Wnt pathway are the TCF/lymphocyte-enhancer-binding factor (LEF) transcription factors. In the absence of Wnt signals, TCF or LEF is bound by co-repressors including Groucho/Transducin-like enhancer (GRG/TLE), and target gene expression is suppressed. Following Wnt signaling, translocated β-catenin replaces the GRG/TLE repressor, and binds TCF/LEF as a co-activator, leading to transcriptional activation of the target genes. The β-catenin/TCF pathway regulates multiple basic developmental processes, including cell-fate specification, progenitor cell proliferation, establishment of the dorsal axis, and control of asymmetric cell division (4). In addition, this pathway has been linked to pathogenesis of diseases such as colon carcinoma (5) and leukemia (6). β-catenin/TCF signaling is also required for hematopoietic stem cell self-renewal and the proliferation of progenitor cells. In addition, it appears that β-catenin signals can confer a more undifferentiated phenotype on lineage-committed progenitors, inhibiting them from differentiating into various lineages (7). In T cells Wnt signals through β-catenin and TCF-1, which was isolated due to its affinity for a motif in CD3 and is expressed exclusively in T-cells in adult mice. In this review article, we discuss our research and how we believe it contributes to the big picture of the function of Wnt signaling through β-catenin and TCF-1 in the regulation of T cell development and differentiation.
3. Wnt/β-catenin/TCF-1 in T cell development
3.1 TCF-1 plays a critical role in T-lineage specification
TCF-1 is one of the first T cell-specific transcription factors induced during early T cell lineage commitment process (8–10). As for the roles of TCF-1 in T cell development, it was believed to be mainly involved in the regulation of cell proliferation and survival during T cell development, and not so much contributing to differentiation progress itself. However very recently, two different groups independently evaluated early thymocyte development when TCF-1 is deficient, and found that T cell lineage commitment process beyond early thymocyte progenitor (ETP) is completely blocked (11, 12). These results are suggestive of another critical role of TCF-1 in T cell lineage specification in addition to its most appreciated roles in regulating thymocyte proliferation and survival in later stages. In terms of the molecular mechanism of the “gate-keeping” of T lineage specification by TCF-1, it seems that TCF-1 can function as a multifaceted player by eliciting both global and T cell specific effects. While Germar et al. proposed that TCF-1 could control the survival of ETP by affecting DNA damage response (11), the work by Weber et al. indicate that TCF-1 might be able to regulate the expression of a few genes key to T lineage determination such as Gata-3 and Bcl11b, and also TCF-1 itself (12). Nonetheless, both groups identified the same CSL (CBF1, Suppressor of Hairless, and Lag-1) binding site at −31 kb upstream of TCF-1 transcription start site, indicating that Notch1 signal regulates TCF-1 expression. Therefore, TCF-1 is required throughout the entire thymocyte development process, starting from the earliest T cell lineage specification to the final positive/negative selection which will be covered in later sections of this review. It should be noted that TCF-1 might regulate T cell lineage commitment independent of β-catenin as ectopic expression of an inhibitor of β- and γ-catenin, ICAT, had no effect on the generation of Thy1+CD25+ cells in an in vitro thymocyte development system, indicating that TCF-1 is not regulated by canonical Wnt signals in this case, and other upstream signals such as Notch1, could be required for TCF-1 regulation, in line with the identification of CSL binding site at TCF-1 gene regulatory region (11, 12).
3.2 Wnt signaling is indispensable for thymocyte development
T cell development in thymus is usually divided into three stages (3): double negative (DN), double positive (DP), and single positive (SP). At the DN stage, thymocytes express neither CD4 nor CD8, but based on their expression of CD25 and CD44, they are divided into the DN1 (CD25−CD44+), DN2 (CD25+CD44+), DN3 (CD25+CD44−) and DN4 (CD25−CD44−) subsets. The heterogeneous DN1 population has 5 subsets (DN1a-DN1e) and the c-kit-expressing DN1a and DN1b cells are also called ETPs. DN1 and DN2 populations still have the potential to differentiate into other non-T cell lineages, such as natural killer (NK) cells, B cells, monocytes and dendritic cells until they pass the DN2b checkpoint regulated by Bcl11b and reach DN3 stage. (13–15). At the DN3 stage, thymocytes begin rearranging the TCR β chain locus and only those thymocytes that successfully rearrange and express a competent TCR β chain on the cell surface are allowed to survive and continue the process of T cell development. The cell surface TCR β chain combines with the invariant pre-Tα chain to form a pre-TCR, which is critical for proliferation of post-β thymocytes and the transition from the DN to DP stage. DN4 thymocytes proliferate extensively to prepare for the stringent DP selection process. Although more than 80% of total thymocytes are DP, only 5% are positively selected to mature into either CD4+ or CD8+ SP cells. Cells expressing αβTCRs that cannot recognize self-MHC peptides well enough to receive a signal die by neglect (16). If the TCR recognizes an MHC-self peptide with too high an affinity the cells are negatively selected and eliminated through apoptosis. However, DP thymocytes expressing pre-TCRs that interact with MHC-self peptides with a positive but sufficiently low affinity are positively selected to mature into SP cells. Mature SP cells migrate out of the thymus into the peripheral lymphoid organs, such as the lymph nodes and spleen, to participate in adaptive immunity. One of the earliest studies addressing the contribution of Wnt signaling pathway in the immune system added soluble Frizzled receptors as decoys for WNT proteins to disturb the pathway in Fetal Thymic Organ Cultures (FTOC) (17). The resulting inhibition of in vitro thymocyte differentiation, apparently through inhibition of proliferation, revealed an essential role for the Wnt signaling pathway in thymocyte development. Later studies using both transgenic gain-of-function and gene knockout loss-of-function animal models not only demonstrated that Wnt signaling was indispensible for thymocyte development, but also indicated that the Wnt signaling pathway is versatile. It regulates multiple molecular processes at multiple stages of T cell development. Here, we summarize an updated view of Wnt/β-catenin/TCF-1 function at different T cell development stages.
Several studies using different approaches to block Wnt signaling have clearly demonstrated that Wnt signaling is indispensable for thymocyte development. In addition to the studies observing inhibition of thymocyte differentiation when soluble Frizzled is used as a decoy for Wnt proteins in the FTOC system (17), mice deficient in either TCF-1 or β-catenin exhibit defective thymocyte development (18, 19). Evidence indicates Wnt is involved during the consecutive molecular selection events needed to generate mature, naive T cells. Furthermore, inhibition of Wnt signaling in postnatal mice by Dickkopf-related protein 1 led to a loss of progenitor thymic epithelial cells (TECs) and resulted in thymic degeneration (20), suggesting that Wnt signaling is also required for the maintenance of properly organized thymic epithelial microenvironment. A controversy arose when the thymic development of T cells was found to be normal in the combined absence of β- and γ-catenin (21, 22). Compared to the β-catenin-deficient mice used by Xu et al. (2008), where the deletion was limited to the T cells, β-catenin was specifically deleted in the cells of hematopoietic compartments in the mouse model used by Jeannet et al. (2008). It is possible that the differences in gene targeting used in the two animal models could have caused the discrepancy. Interestingly, in spite of the deletion of both β- and γ-catenin, Wnt signaling remained active in the thymocytes according to reporter gene activity, strongly suggesting that active Wnt signaling is a requirement for successful T cell development, and that alternative co-activators of TCF-1 may take over under the condition of β- and γ-catenin double deficiency. As yet however, few studies have looked for these alternative Wnt-mediated activators of TCF-1.
3.3 Wnt signaling supports proliferation of DN thymocytes
At the DN stage, the major role of Wnt signaling is the regulation of thymocyte proliferation. There are two critical checkpoints during the DN stage; the final T lineage determination step and β-selection. After gradually losing the potential to develop into other lineages, some DN thymocytes (DN2b) pass the transcription factor Bcl11b-dependent checkpoint, and their development fate is eventually determined as T cells (13, 14). DN2 thymocytes that successfully pass this first checkpoint proliferate extensively to expand cell numbers and this proliferation step is reported as the earliest time point at which Wnt signaling begins to regulate the T cell development process. The cells then proceed to the second checkpoint where DN2 thymocytes that fail to rearrange and express a competent TCR β chain are purged from the immature thymocyte pool. Another surge of proliferation takes place post-β-chain selection when DN3/4 thymocytes undergo extensive expansion to enlarge the thymocyte pool in preparation for the even more restrictive positive/negative selection that occurs at the DP stage. If Wnt signaling is blocked by various approaches including the addition of soluble Frizzled receptors to FTOC (17), or the genetic knock out of TCF-1 (18), β-catenin (19) or Wnt1 and Wnt4 (23) in mice, the proliferation of both the DN2 and post-β-selection DN3/4 thymocytes is severely impaired, leading to strikingly low thymic cellularity. In line with these results, a microarray analysis study using immature human CD34+ thymocytes also identified several proliferation-inducing transcription factors such as c-fos and c-jun as Wnt target genes (24). Together these experiments strongly indicate that Wnt signaling through β-catenin/TCF-1 is required for proliferation of the thymocytes during both expansion periods. The mechanisms by which the Wnt signaling specifically regulates thymocyte proliferation remains largely unknown, due both to the complexity of the signaling pathway itself and the even less well understood thymic microenvironment. More than 100 genes have been reported to be Wnt signaling targets, among which only FoxN1 is known to be expressed in the thymus (25). However, it is unlikely that Wnt signaling-regulated thymocyte proliferation is mediated by FoxN1, because the primary function of FoxN1 is to support the epithelial morphogenesis of the thymus (25). Furthermore, it is unknown if FoxN1 is expressed in thymocytes. An evaluation of changes in the expression or activity level of genes that are specifically related to cell cycle in DN2 and post-selection DN3/4 thymocytes could help identify the potential target genes that act downstream of Wnt/β-catenin/TCF-1 to regulate thymocyte proliferation. However, not only are downstream molecular factors of Wnt signaling in thymocytes elusive, but how the signaling pathway is initiated and what the upstream events are, is still unknown. At the DN2 stage when the thymocytes do not yet express a pre-TCR, Wnt ligands and the Frizzled receptors might form the critical communication interface between the thymocyte and the thymic environment to initiate proliferation. In addition, direct regulation of intracellular β-catenin activity is also possible since there is cross-talk between Wnt signaling and other pathways such as nuclear factor of activated T cells (NFAT) (26, 27), which is known to be critical for the proliferation of armed effector T cells (28). Rapid proliferation of DN thymocytes post-β-selection requires pre-TCR signaling (29) which appears to upregulate early growth response gene 3 (Egr3) through activation of NFAT and/or the extracellular signal-regulated kinases (ERK) (30–34). Egr3 is only transiently upregulated; it is turned off when the thymocytes become DP and stop proliferating to rearrange the TCRα locus (30). Wnt signaling may be activated by pre-TCR signaling in post-β-selection DN thymocytes. Firstly, β-catenin expression was induced by anti-CD3 antibody treatment when it was used to mimic pre-TCR stimulation in Rag2-deficient mice (35). In addition, stimulation of isolated thymocytes with either phorbol 12-myristate 13-acetate (PMA) or CD3 antibody caused accumulation of increased levels of nuclear β-catenin compared to untreated cells, suggesting that pre-TCR signaling could turn on the Wnt signaling pathway by activating β-catenin (19). This theory was further corroborated by the recent observation that TCR signaling in primary human T cells leads to stabilization of β-catenin (36). However, in another study, non-phosphorylated, stabilized β-catenin was readily detected in Rag-1-deficient thymocytes which lack a functional pre-TCR due to impaired TCR β chain locus rearrangement, and when Rag-1-deficient mice were injected with anti-CD3 antibodies to induce pre-TCR signaling, neither total nor active β-catenin increased. However, the authors did find that TCF-1 levels were significantly increased after pre-TCR induction through anti-CD3 antibody injection, indicating that pre-TCR signals might induce TCF-1 independently of Wnt-induced β-catenin accumulation/translocation (37). However, because TCF-1 is usually repressed by Groucho/TLE which competes with activated β-catenin to bind to TCF-1, it is difficult to explain how an increase in TCF-1 expression alone would lead to the activation of target genes. One of the ultimate indicators of Wnt signaling activity is target gene transcription. Therefore, anti-CD3 antibody treatment of Rag2-deficient mice crossed with TCF-1 reporter mice (38) and expression analysis of the TCF-1 reporter gene would help demonstrate a direct relationship between pre-TCR and Wnt signaling. Nevertheless, the consensus is that pre-TCR signaling is able to induce Wnt signaling in post-β-selection DN thymocytes, although how this induction occurs requires further investigation. Because pre-TCR signaling is known to stimulate both the Egr3 and Wnt signaling pathways in post-β-selection DN thymocytes, the intriguing question is whether these two pathways function independently of each other or there is crosstalk between them. Xu et al. (2009b) has proposed that pre-TCR signaling induces β-catenin cooperation with TCF-1 to regulate Egr3 expression and facilitate β-selection (39). They demonstrated that β-catenin binds to the Egr3 proximal regulatory region and this finding, together with the presence of TCF-1 binding sites in Egr3 promoter region (39) and the observed stimulation of TCF-1 reporter activity in DN3 thymocytes (37), suggests that the upregulation of Egr3 by pre-TCR signaling is mediated, at least in part, by β-catenin/TCF-1.
3.4 β-catenin/TCF-1 is required for DN to DP transition
Post-β-selected DN thymocytes undergo several rounds of cell division to expand the immature thymocyte pool before they upregulate both CD4 and CD8 to become DP thymocytes. During the DN to DP transition, the failure to upregulate CD4 or CD8 expression leads to a block of thymocyte development at the DN4 or the CD8+ immature single positive stage. Wnt signaling has been proposed to play a role in the DN to DP transition because TCF-1-deficient mice exhibit the accumulation of CD8+ ISP thymocytes (40). Furthermore, overexpression of ICAT, which blocks the binding of β-catenin to TCF-1 and LEF-1, is known to result in a thymocyte development block at the DN to DP transition step in FTOC assays (41), suggesting an essential role for β-catenin/TCF-1 in the DN to DP transition. Gene expression profiling of thymocytes by microarray analysis revealed reduced CD4 mRNA levels in TCF-1−/− thymocytes but increased CD4 mRNA levels in β-catenin transgenic thymocytes (42), indicating Wnt signaling stimulates CD4 expression. We have also shown that TCF-1 can directly target the CD4 regulatory region and stimulate its transcription (43), suggesting that one of the potential mechanisms regulated by Wnt signaling during the DN to DP transition is the induction of CD4 expression. However, it appears that there are also other transcription factors that can stimulate CD4 expression, because TCF-1 deficiency leads to a reduction but not the abolishment of CD4 expression.
3.5 β-catenin/TCF-1 enhances DP thymocyte survival
Although it appears that Wnt signaling activity decreases once thymocytes become DP (35, 38), another critical aspect of thymocyte development at the DP stage that is dependent on Wnt signaling appears to be their survival. DP thymocytes rearrange their TCRα loci and the newly formed TCRs are tested by interaction with MHC-self peptide complexes. Only thymocytes expressing TCRs that interact with the complexes positively, but with a sufficiently low affinity, are able to survive and eventually develop into T cells. However, DP thymocytes are able to initiate multiple rounds of TCRα locus rearrangement during the window of positive and negative selection. Therefore, the lifespan of a thymocyte is critical for shaping the T cell repertoire because the longer the thymocyte can survive, the greater the chances are it will generate a TCR that will be positively selected.
Ioannidis et al. (2001) clearly demonstrated that TCF-1-deficient thymocytes undergo accelerated apoptosis which can be prevented by ectopically introducing the anti-apoptotic factor Bcl-2. We (44), and also another group (45), have shown that β-catenin enhances DP thymocyte survival. We found that thymocytes from β-catenin transgenic mice which ectopically express a stabilized β-catenin (β-catTg) have enhanced survival compared to WT thymocytes (44). More importantly, the enhanced survival exhibited by the β-catTg thymocytes is mediated by Bcl-xL upregulation (44). This result is consistent with the low Bcl-xL levels we and others reported in TCF-1−/− thymocytes (46, 47), and indicates that Wnt signaling protects DP thymocytes from premature apoptosis by upregulating Bcl-xL. In spite of the absolute requirement for β-catenin/TCF-1 in the regulation of DP thymocyte survival, the activity of Wnt signaling is under tight control. Guo et al. reported that β-catenin stabilization led to a block of DP to SP transitions, and the accumulated DP thymocytes are prone to malignant transformation (54), indicating that Wnt signaling might be involved in a fail-safe mechanism responsive for eliminating unwanted thymocytes at DP stage.
In addition to β-catenin/TCF-1, there are other factors implicated in the regulation of DP thymocyte survival, including Retinoid-related orphan nuclear receptor γt (RORγt) (48, 49), NFAT4 (26), Egr3 (50), Hela E-box binding protein (HEB) (51) and c-myb. A more challenging task than the identification of individual factors involved in DP thymocyte survival will be to investigate how each of the factors is integrated into the molecular network regulating DP thymocyte survival. RORγt is the thymus-specific isoform of RORγ, and a member of the steroid receptor family. The phenotype of massive thymocyte apoptosis exhibited by RORγt-deficient mice indicates an essential role for RORγt in supporting DP thymocyte survival (48). Similar to that observed in TCF-1−/− thymocytes, RORγt-deficient thymocytes also have reduced Bcl-xLand the ectopic expression of Bcl-xL successfully rescues RORγt−/− thymocyte survival. Given the resemblance of DP thymocyte apoptosis kinetics, and the similarly reduced Bcl-xL levels observed in TCF-1−/− and RORγt−/− thymocytes, we suspected that TCF-1 and RORγt could be involved in the same DP thymocyte survival regulation pathway mediated by Bcl-xL. We found that TCF-1-deficient thymocytes express strikingly lower levels of RORγt, whereas β-catenin transgenic thymocytes have higher expression of RORγt, compared to WT counterparts (53), indicating that the expression of RORγt in the thymocytes may be under control of β-catenin/TCF-1. Using an in vitro thymocyte differentiation system, we showed that forced expression of RORγt rescued the survival of TCF-1-deficient DP thymocytes, but ectopically expressed β-catenin or TCF-1 failed to rescue survival of RORγt-deficient DP thymocytes (53). Both results indicate that RORγt expression occurs downstream of β-catenin/TCF-1. To confirm this molecular mechanism, we tested the ability of β-catenin/TCF-1 to stimulate a reporter construct containing the RORγt promoter region, and found that the β-catenin/TCF-1 complex activated the RORγt promoter. A fragment of the RORγt promoter region contained in our construct was specifically enriched in thymocyte chromatin precipitated by anti-β-catenin antibodies in Chromatin immuno-precipitation assays (53), and we identified a TCF-1 binding motif (CTTTG) in the enriched RORγt regulatory fragment. The mutation of this element to disrupt TCF-1 binding led to a ~50% reduction of RORγt promoter activity (53). Taken together, our data clearly demonstrates that β-catenin/TCF-1 activates RORγt transcription by directly binding its promoter region, and RORγt subsequently upregulates Bcl-xL to enhance DP thymocyte survival.
3.6 Wnt signaling is involved in positive/negative selection
Based on reporter studies, the Wnt signaling activity is reduced in DP and SP thymocytes compared to DN cells (38). However, the importance of Wnt signaling at these stages is clearly indicated by its involvement not only in DP thymocyte survival but also in the regulation of both positive and negative selection (55–57). Yu et al. used a transgenic mouse strain that expressed a stabilized β-catenin via a Lck promoter, to show that expression of the transgene accelerated the generation of CD8 SP thymocytes. This step usually occurs later than the generation of CD4 SP thymocytes but in these mice the generation of both types of thymocytes took place with the same kinetics (56). It appears that the accelerated generation of CD8+ SP thymocytes is dependent on enhanced IL-7 signals that are caused by the β-catenin overexpression (55). Interestingly, in another study using mouse models that carried an exon 3 deletion of β-catenin, generated by crossing conditional β-catenin stabilization Ctnnbex3 mice (57–59) with CD4 or Lck-cre mice, the high level of β-catenin was found to enhance negative selection. This result is consistent with the impaired negative selection observed in TCF-1−/− mice (57). One possible explanation for these seemingly contradictory observations is the strategy used to stabilize β-catenin expression in the different experimental systems. The mouse model used by Yu et al. expresses both a “modest” level of transgenic stabilized β-catenin and a lower but detectable level of endogenous β-catenin, and possibility that there is an influence on thymic selection from the endogenous β-catenin cannot be excluded (56). In contrast, the mice used in the negative selection study expressed undetectable levels of endogenous β-catenin (57, 59). In addition, the actual amount of stabilized or active β-catenin might also differ in these systems. It is possible that a subtle change of the level of active β-catenin in DP thymocytes could lead to an opposite thymic selection outcome.
4. Wnt/β-catenin/TCF-1 in peripheral T cell differentiation
4.1 Introduction
Upon antigen engagement together with specific cytokines, naïve CD4+ T cells up-regulate master transcription factors that determine their differentiation into Th1, Th2, Th17 or Treg effector cells (60). When stimulated with IL-12 or INF-γ, naïve T cells express the T-box transcription factor T-bet as their lineage specific transcriptional factor and differentiate into Th1 cells, which secrete interferon-γ (IFN-γ) as their signature cytokine. In the presence of IL-4, naïve T cells express GATA3 as their lineage specific transcriptional factor and differentiate into Th2 cells, which secrete the signature cytokines IL-4, IL-5 and IL-13 (61). TCR stimulation in the presence of IL-6 and TGF-β leads to up-regulation of RORγt and differentiation of naïve T cells into Th17 cells that produce IL-17, IL-21, IL-22 and GM-CSF. Stimulation of naïve T cells in the presence of TGF-β without IL-6 induces Foxp3, which directs their differentiation into Tregs, and they secrete IL-10 and TGF-β. Th1, Th2 and Th17 cells are considered inflammatory effector T cells, because they generally stimulate immune responses, but Tregs suppress inflammatory T cell function partially via IL-10 and TGF-β. Therefore, the same naïve T cells can differentiate into inflammatory and regulatory T cells, two subsets of cells with opposite function. It is critical therefore, to study the mechanisms that regulate the differentiation of naïve T cells into different types of effector T cells in order to facilitate the development of novel treatments for persons with dysregulated immune systems. Great efforts have been made to understand β-catenin/TCF-1 pathway because it is known to regulate many important cellular functions in peripheral T cell differentiation.
4.2 TCF-1 promotes Th2 but represses Th1 differentiation
TCF-1 has been shown to be critical for initiation of Th2 differentiation, a GATA-3-dependent process (62). Early expression of GATA-3 seems to require TCR stimulation but is independent of IL-4, the cytokine essential for Th2 differentiation. TCF-1 and β-catenin stimulate early GATA-3-1b expression via direct binding of TCF-1 to GATA-3-1b transcriptional regulatory element. Furthermore, TCF-1-induced GATA-3 expression stimulates IL-4 production, and the deletion of TCF-1 impairs Th2 responses in vivo (62), suggesting that TCF-1 promotes Th2 differentiation via stimulation of GATA-31b expression and IL-4 production. Because the differentiation of Th1 and Th2 helper T cells is reciprocal (63, 64), it is not surprising that TCF-1, which promotes Th2 differentiation, inhibits Th1 formation. This is most likely by TCF-1 negatively regulates the production of IFN-γ, which promotes Th1 differentiation. In contrast to the requirement for both β-catenin and TCF-1 for Th2 differentiation, the TCF-1 suppressive effect on Th1 differentiation is independent of β-catenin (62). T-bet, a master transcription factor for Th1 differentiation, functionally represses Th2 differentiation (65) and T-bet has been shown to negatively regulate TCF-1 through a direct interaction with the transcriptional repressor Bcl-6. As a result of TCF-1 suppression by T-bet, Th2 differentiation is inhibited and Th1 differentiation is reciprocally enhanced (65).
4.3 TCF-1 promotes Th17 differentiation
Identification of the Th17 lineage has shifted the traditional Th1/Th2 paradigm by adding a new lineage of T helpers (66). In 2003, Cua et al. (2003) made the seminal observation that IL-23, but not the Th1 promoting cytokine IL-12, was critical for the development of EAE, which had long been believed to be a Th1-dependent autoimmune disease (67). Th17 cells produce IL-17, IL-21 and IL-22 cytokines, and IL-17 induces massive tissue inflammation due to the broad distribution of its receptors on both immune and nonimmune cells (68). In addition to EAE, circumstantial evidence has also accumulated suggesting a pathogenic role for Th17 cells in multiple human autoimmune disorders, including multiple sclerosis (69), rheumatoid arthritis (70), asthma (71), IBD (72) and psoriasis (73). Stimulation of naïve T cells in the presence of TGF-β and IL-6 is sufficient to form IL-17-producing T cells (74). IL-6 activates Stat3, which is essential for formation of Th17 cells (75) and IL-6 and TGF-β together are required to induce RORγt, a lineage-specific transcriptional factor of Th17 cells. RORγt is the master transcription factor Th17 differentiation, since T cells deficient in RORγt fail to become IL-17 producing cells, even in the presence of TGF-β and IL-6 (76).
Data from both Yu et al. (2001) and our lab (77) have shown that TCF-1 is critical for the control Th17 differentiation. TCF-1−/− T cells show enhanced differentiation into Th17 cells, with the increased production of Th17 cytokines, including IL-17A and IL-17F, and the up-regulation of IL-23 receptor (IL-23R). TCF-1−/− T cells also induce more severe EAE disease in an adoptive transferred model (77). In contrast to most known Th17 regulating factors, including Stat3 (78), Runx1(79), IRF4 (80), Ahr (81), Batf (82), Foxp3 (83) and Ets-1 (84, 85), all of which either positively or negatively regulate Th17 differentiation via RORγt expression, TCF-1 regulates Th17 differentiation without changing the expression levels of RORγt (77, 86). Yu et al. (2001) has suggested that TCF-1 regulates IL-17 at the mature T cell stage. But TCF-1 expression in T cells decreases rapidly after T cell activation in vivo in both humans and mice (87, 88), suggesting that this may not be the case. Indeed, we have shown that forced expression of WT or dominant negative TCF-1 does not affect the Th17 differentiation potential of either WT or TCF-1−/− T cells (77). Instead we propose there is a TCF-1 regulated, epigenetic modification of the IL-17 locus during T cell development in the thymus, which leads to a more open state of the IL-17 gene, and these epigenetic modifications are imprinted in the T cells even after they mature and migrate out of thymus (77). We found that 1) TCF-1−/− mice had a larger natural Th17 cell population in the thymus and periphery, that produces Th17 cytokines without further differentiation (89); 2) Expression of a full length, dominant negative TCF-1 or β-catenin in mature TCF-1−/− T cells did not interfere with Th17 differentiation; 3) Deletion of TCF-1 does not alter the expression of factors known to regulate Th17 differentiation (77). Our results therefore, do not favor a model in which TCF-1 regulates Th17 differentiation at mature T cell stage, and suggests the possibility that TCF-1 may regulate Th17 differentiation potential during developmental T cell stages. Previous studies have demonstrated that the IFN-γ and IL-4 gene loci undergo dramatic epigenetic changes in chromatin modifications, resulting in the activation of their expression upon differentiation to Th1 and Th2 cells, respectively (90). Similarly, during Th17 differentiation, the IL-17 locus also undergoes extensive epigenetic modification, leading to a significant increase in DNA accessibility for transcription activation (84). Acetylation and methylation of histone proteins are two of the critical epigenetic modifications for regulating gene expression because they alter DNA-protein binding and the accessibility of the DNA to transcription and other regulatory factors (91). During Th17 differentiation, the IL-17 locus is slowly transformed into an open, accessible state as indicated by the hyper-acetylation and hyper-trimethylation of histone H3. Our data shows that the H3 protein at the IL-17 locus in TCF-1−/− thymocytes is more acetylated and trimethylated than in WT cells, indicating a more open state of the IL-17 gene in developing T cells. This result suggests that there is a TCF-1-mediated process that represses the IL-17 locus via epigenetic modifications during T cell development in thymus (77). Furthermore, the presence of increased IL-17 mRNA levels and more natural Th17 in peripheral T cells of TCF-1−/− mice further suggests that TCF-1-mediated epigenetic modifications of IL-17 locus are preserved after T cells migrate out of the thymus (77). Altogether, we propose that TCF-1 regulates a program including epigenetic modifications to repress IL-17 locus during T cell development. This TCF-1-mediated repression is critical for prevention of autoimmunity, as suggested by susceptibility of TCF-1−/− mice to EAE. Although β-catenin activates target gene expression together with TCF-1 in the canonical Wnt pathway, (92, 93) it may not be involved in Th17 differentiation. We expressed a stabilized β-catenin transgene in the thymus and Th17 differentiation was not affected (77). TCF-1 target gene expression is inhibited when the protein is associated with its co-repressor GRG in mouse (Groucho in Drosophila) (94), but it remains to be determined if GRG is required for TCF-1-mediated epigenetic modification of the IL-17 locus. It is therefore important to emphasize that TCF-1 can regulate T cell function via both β-catenin dependent and independent pathways.
4.4 Function of β-catenin in Tregs
Although little work has been done to investigate the role of Wnt/β-catenin/TCF-1 signaling in Treg development, differentiation and function, one study by Ding et al (2008) showed that β-catenin promoted survival of CD4+CD25+ Treg cells. The expression of a stabilized β-catenin in Tregs via retroviral transduction increased Treg survival through enhanced expression of Bcl-XL. More importantly, forced expression of β-catenin enhanced Treg function to protect against inflammatory bowel disease (95). The induction of an anergic phenotype in CD4+ CD25− effector T cells after transduction of a stabilized β-catenin was also reported (95), but this result is not supported by another study (87).
4.5 TCF-1 is required for establishment of CD8 memory T cells
Wnt signaling plays a prominent role in hematopoietic stem cells (HSCs) maintenance and cell lineage specification in a variety of tissues (96, 97). Because long-life memory T cells have the capacity to self-renew by slow homeostatic turnover (98), and they share certain transcriptional patterns with HSCs (99), the role of the Wnt/β-catenin/TCF-1 pathway in T cell memory formation has been intensively investigated. In a gain-of-function study, Zhao et al. (2010) showed that constitutive activation of the canonical Wnt pathway favors memory CD8 T cell formation during the initial immune challenge, resulting in an enhanced immunity upon the second encounter with the same pathogen (87). TCF-1 is highly expressed in naïve T cells, then down-regulated in effector T cells, and up-regulated again in memory T cells, indicating it may play an important role for T cell memory formation. Forced expression of TCF-1 alone limited the expansion of antigen-specific CD8 T cells in response to Listeria monocytogenes infection, whereas forced expression of the TCF-1 transgene coupled with that of stabilized β-catenin, generated more antigen-specific memory CD8 T cells. These cells expanded into a larger number of secondary effectors that cleared pathogens faster when the immunized mice were rechallenged with virulent L. monocytogenes, vaccinia virus or lymphocytic choriomeningitis virus (87). These results support that the requirement for β-catenin, acting as a TCF-1 coactivator, in the establishment of memory T cells. Consistent with this hypothesis, two loss-of-function studies (100, 101) have shown TCF-1 is essential for the establishment of functional CD8 T cell memory. Although it is not essential for mounting a primary protective CD8 T cell response to viral infection, for the maintenance of a pool of virus-specific effector T cells, or for effector memory cell formation, TCF-1 is critical for the secondary expansion of virus-specific CD8 T cells upon re-infection, for the generation of CD8 central memory precursor cells and for long-lived CD8 T cell memory (101). Mice deficient in β-catenin and γ-catenin also show impaired replication of CD8 T cell memory (101).
The function of β-catenin/TCF-1 in the establishment of memory T cells is partially via stimulation of eomesodermin expression. Zhou et al. (2010) showed that the Wnt-TCF-1 pathway was necessary and sufficient to induce eomesodermin expression. Eomesodermin, which is a paralogue of T-bet, is important for CD8+ T cell function, differentiation and memory formation (102–104). Using transcriptomic analysis on sorted WT and TCF-1−/− memory OT-I cells, their group revealed that in addition to CD8+ effector molecules, cytokines, chemokines and their receptors, TCF-1 controls a wide spectrum of other biological functions in memory T cells, including transcriptional regulation, cell cycle progression, and apoptosis. TCF-1−/− memory T cells had reduced levels of eomesodermin which is required to enhance IL-2Rβ expression, and subsequently confers IL-15 responsiveness to memory CD8+ T cells (103). TCF-1 is believed to bind to regulatory sequences on the eomesodermin promoter in vivo to stimulate eomesodermin expression, which in turn protects time-dependent loss of memory T cells. Support for the critical roles played by β-catenin/TCF-1 in memory T cell function is further confirmed by studies using a pharmacological inhibitor of GSK3β. In the absence of Wnt signaling, β-catenin is constantly degraded in cytoplasm due to phosphorylation by GSK3β (105). Forced activation of the Wnt signaling pathway in CD8 T cells using the GSK3β inhibitor promoted the differentiation of CD8 T cells into memory precursor cells and arrested effector T cell differentiation (106), suggesting a positive role for β-catenin in promoting memory T cell formation. This result however, has been challenged by other studies. Driessens et al. (2010) used genetic approaches to either express stabilized β-catenin or delete β-catenin in peripheral T cells. Their results indicated that expression of stabilized β-catenin or the deletion of β-catenin had no effect on memory phenotype compared to control cells. This work is further supported by a recent study using a conditional knockout mouse that specifically deleted β-catenin in mature T cells (107), and showed that β-catenin was dispensable for CD8+ memory development. Altogether, these results support a role for TCF-1 in memory T cell function, but the need for β-catenin, a coactivator of TCF-1, remains controversial.
5. Conclusions
The Wnt/β-catenin/TCF-1 pathway regulates T cell function in both the central and peripheral immune system. In the thymus, it is required at multiple stages of T cell development to regulate T cell proliferation, survival and apoptosis (Figure 1). These steps ensure the elimination of self-reactive T cells and the development of mature of T cells that only response to foreign antigens. In peripheral lymphoid tissues, TCF-1 regulates the differentiation of naïve CD4+ T cells into effector T cells that are competent to mediate immune responses (Figure 2). The function of this pathway in the thymus and periphery may not always be separate. For example, TCF-1 mediated-enhancement of Th17 differentiation in the periphery could be due to a role in epigenetic modification of the IL-17 gene locus during T cell development in the thymus. Despite the progress in our understanding, there is controversy about the role of β-catenin in T cell development and function, especially because a recent study demonstrated its function is dispensable in T cells (108), suggesting that much more research is needed. However, the controversy that exists may be due to the different experimental procedures, model systems and approaches that have been used. Additional studies on how different T cells integrate TCF-1/β-catenin signals to coordinate immune responses are essential, and with the availability of new technologies that allow analysis of global epigenetic modification, gene expression, and chromatin occupancy, there is the opportunity to identify the broad network of genes regulated by of the Wnt/β-catenin/TCF-1 pathway that determine individual T cell fate and effector function.
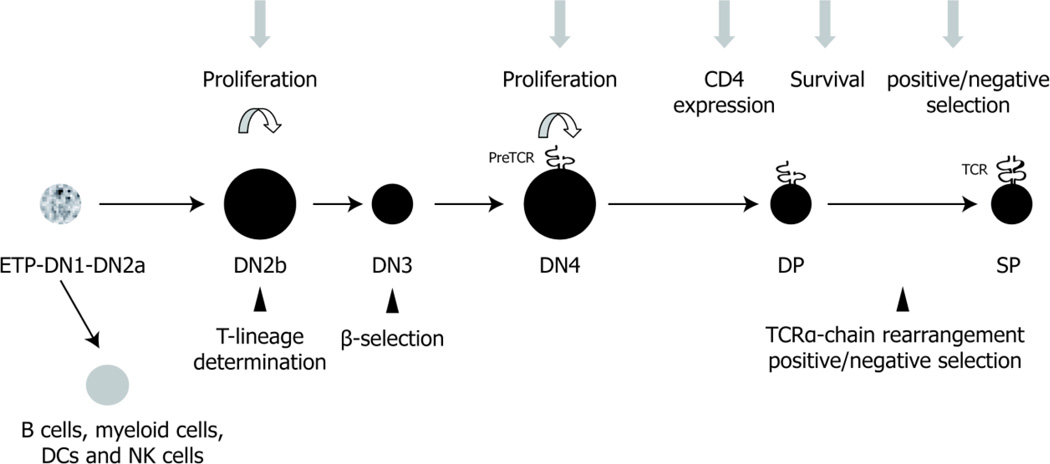
Figure 1.
Summary of function of Wnt pathway during thymic T cell development. Black triangles indicates the important events during T cell development. Arrows indicate the developmental stages that Wnt pathway potentially has important regulatory function. ETP, early T cell progenitors. Note that TCF-1 also plays a critical role in T cell lineage specification at “ETP-DN1-DN2a” stage, which is not shown in the figure.
Figure 2.
Summary of function of Wnt pathway during peripheral T cell differentiation. Potential positive or negative function of β-catenin/TCF-1 during naïve T cells differentiation into Th1, Th2, Th17, Treg and CD8 memory T cells are indicated. Cytokines produced by different types of T helper cells are also indicated.
Acknowledgements
We thank Dr. Margaret Morgan for expert editorial assistance.
Our work was supported by grants NIH R01-AI053147, NIH R56-AI072554, the Nesvig lymphoma Fellowship and Research Fund and City of Hope.
Footnotes
Authors of the paper have no financial conflict of interests in connection this manuscript.
References
- 1.Yu Q, Sharma A, Sen JM. TCF1 and beta-catenin regulate T cell development and function. Immunol Res. 2010;47:45–55. doi: 10.1007/s12026-009-8137-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liang H, Coles AH, Zhu Z, Zayas J, Jurecic R, Kang J, Jones SN. Noncanonical Wnt signaling promotes apoptosis in thymocyte development. The Journal of experimental medicine. 2007;204:3077–3084. doi: 10.1084/jem.20062692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xie H, Huang Z, Wang R, Sun Z. Regulation of thymocyte survival by transcriptional coactivators. Crit Rev Immunol. 2006;26:475–486. doi: 10.1615/critrevimmunol.v26.i6.10. [DOI] [PubMed] [Google Scholar]
- 4.Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–480. doi: 10.1016/j.cell.2006.10.018. [DOI] [PubMed] [Google Scholar]
- 5.Barker N, Clevers H. Mining the Wnt pathway for cancer therapeutics. Nat Rev Drug Discov. 2006;5:997–1014. doi: 10.1038/nrd2154. [DOI] [PubMed] [Google Scholar]
- 6.Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL, Gotlib J, Li K, Manz MG, Keating A, Sawyers CL, Weissman IL. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. The New England journal of medicine. 2004;351:657–667. doi: 10.1056/NEJMoa040258. [DOI] [PubMed] [Google Scholar]
- 7.Staal FJ, Luis TC, Tiemessen MM. WNT signalling in the immune system: WNT is spreading its wings. Nat Rev Immunol. 2008;8:581–593. doi: 10.1038/nri2360. [DOI] [PubMed] [Google Scholar]
- 8.Taghon TN, David ES, Zuniga-Pflucker JC, Rothenberg EV. Delayed, asynchronous, and reversible T-lineage specification induced by Notch/Delta signaling. Genes & development. 2005;19:965–978. doi: 10.1101/gad.1298305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rothenberg EV. T cell lineage commitment: identity and renunciation. J Immunol. 186:6649–6655. doi: 10.4049/jimmunol.1003703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Georgescu C, Longabaugh WJ, Scripture-Adams DD, David-Fung ES, Yui MA, Zarnegar MA, Bolouri H, Rothenberg EV. A gene regulatory network armature for T lymphocyte specification. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:20100–20105. doi: 10.1073/pnas.0806501105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Germar K, Dose M, Konstantinou T, Zhang J, Wang H, Lobry C, Arnett KL, Blacklow SC, Aifantis I, Aster JC, Gounari F. T-cell factor 1 is a gatekeeper for T-cell specification in response to Notch signaling. Proceedings of the National Academy of Sciences of the United States of America. 108:20060–20065. doi: 10.1073/pnas.1110230108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weber BN, Chi AW, Chavez A, Yashiro-Ohtani Y, Yang Q, Shestova O, Bhandoola A. A critical role for TCF-1 in T-lineage specification and differentiation. Nature. 476:63–68. doi: 10.1038/nature10279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ikawa T, Hirose S, Masuda K, Kakugawa K, Satoh R, Shibano-Satoh A, Kominami R, Katsura Y, Kawamoto H. An essential developmental checkpoint for production of the T cell lineage. Science (New York, N.Y. 329:93–96. doi: 10.1126/science.1188995. [DOI] [PubMed] [Google Scholar]
- 14.Li L, Leid M, Rothenberg EV. An early T cell lineage commitment checkpoint dependent on the transcription factor Bcl11b. Science (New York, N.Y. 329:89–93. doi: 10.1126/science.1188989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rothenberg EV, Moore JE, Yui MA. Launching the T-celllineage developmental programme. Nature reviews. 2008;8:9–21. doi: 10.1038/nri2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Goldrath AW, Bevan MJ. Selecting and maintaining a diverse T-cell repertoire. Nature. 1999;402:255–262. doi: 10.1038/46218. [DOI] [PubMed] [Google Scholar]
- 17.Staal FJ, Meeldijk J, Moerer P, Jay P, van de Weerdt BC, Vainio S, Nolan GP, Clevers H. Wnt signaling is required for thymocyte development and activates Tcf-1 mediated transcription. European journal of immunology. 2001;31:285–293. doi: 10.1002/1521-4141(200101)31:1<285::AID-IMMU285>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 18.Schilham MW, Wilson A, Moerer P, Benaissa-Trouw BJ, Cumano A, Clevers HC. Critical involvement of Tcf-1 in expansion of thymocytes. J Immunol. 1998;161:3984–3991. [PubMed] [Google Scholar]
- 19.Xu Y, Banerjee D, Huelsken J, Birchmeier W, Sen JM. Deletion of beta-catenin impairs T cell development. Nature immunology. 2003;4:1177–1182. doi: 10.1038/ni1008. [DOI] [PubMed] [Google Scholar]
- 20.Osada M, Jardine L, Misir R, Andl T, Millar SE, Pezzano M. DKK1 mediated inhibition of Wnt signaling in postnatal mice leads to loss of TEC progenitors and thymic degeneration. PLoS ONE. 5:e9062. doi: 10.1371/journal.pone.0009062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jeannet G, Scheller M, Scarpellino L, Duboux S, Gardiol N, Back J, Kuttler F, Malanchi I, Birchmeier W, Leutz A, Huelsken J, Held W. Long-term, multilineage hematopoiesis occurs in the combined absence of beta-catenin and gamma-catenin. Blood. 2008;111:142–149. doi: 10.1182/blood-2007-07-102558. [DOI] [PubMed] [Google Scholar]
- 22.Koch U, Wilson A, Cobas M, Kemler R, Macdonald HR, Radtke F. Simultaneous loss of beta- and gamma-catenin does not perturb hematopoiesis or lymphopoiesis. Blood. 2008;111:160–164. doi: 10.1182/blood-2007-07-099754. [DOI] [PubMed] [Google Scholar]
- 23.Mulroy T, McMahon JA, Burakoff SJ, McMahon AP, Sen J. Wnt-1 and Wnt-4 regulate thymic cellularity. European journal of immunology. 2002;32:967–971. doi: 10.1002/1521-4141(200204)32:4<967::AID-IMMU967>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 24.Staal FJ, Weerkamp F, Baert MR, van den Burg CM, van Noort M, de Haas EF, van Dongen JJ. Wnt target genes identified by DNA microarrays in immature CD34+ thymocytes regulate proliferation and cell adhesion. J Immunol. 2004;172:1099–1108. doi: 10.4049/jimmunol.172.2.1099. [DOI] [PubMed] [Google Scholar]
- 25.Balciunaite G, Keller MP, Balciunaite E, Piali L, Zuklys S, Mathieu YD, Gill J, Boyd R, Sussman DJ, Hollander GA. Wnt glycoproteins regulate the expression of FoxN1, the gene defective in nude mice. Nature immunology. 2002;3:1102–1108. doi: 10.1038/ni850. [DOI] [PubMed] [Google Scholar]
- 26.Oukka M, Ho IC, de la Brousse FC, Hoey T, Grusby MJ, Glimcher LH. The transcription factor NFAT4 is involved in the generation and survival of T cells. Immunity. 1998;9:295–304. doi: 10.1016/s1074-7613(00)80612-3. [DOI] [PubMed] [Google Scholar]
- 27.Beals CR, Sheridan CM, Turck CW, Gardner P, Crabtree GR. Nuclear export of NF-ATc enhanced by glycogen synthase kinase-3. Science. 1997;275:1930–1934. doi: 10.1126/science.275.5308.1930. [DOI] [PubMed] [Google Scholar]
- 28.Janeway CA, Walport TP, Shlomchik M. J. M. Immunobiology. New York, NY: Garland Publishing; 2001. [Google Scholar]
- 29.Godfrey DI, Zlotnik A. Control points in early T-cell development. Immunology today. 1993;14:547–553. doi: 10.1016/0167-5699(93)90186-O. [DOI] [PubMed] [Google Scholar]
- 30.Xi H, Schwartz R, Engel I, Murre C, Kersh GJ. Interplay between RORgammat, Egr3, and E proteins controls proliferation in response to pre- TCR signals. Immunity. 2006;24:813–826. doi: 10.1016/j.immuni.2006.03.023. [DOI] [PubMed] [Google Scholar]
- 31.Shao H, Kono DH, Chen LY, Rubin EM, Kaye J. Induction of the early growth response (Egr) family of transcription factors during thymic selection. The Journal of experimental medicine. 1997;185:731–744. doi: 10.1084/jem.185.4.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rengarajan J, Mittelstadt PR, Mages HW, Gerth AJ, Kroczek RA, Ashwell JD, Glimcher LH. Sequential involvement of NFAT and Egr transcription factors in FasL regulation. Immunity. 2000;12:293–300. doi: 10.1016/s1074-7613(00)80182-x. [DOI] [PubMed] [Google Scholar]
- 33.Tonks NK, Neel BG. Combinatorial control of the specificity of protein tyrosine phosphatases. Current opinion in cell biology. 2001;13:182–195. doi: 10.1016/s0955-0674(00)00196-4. [DOI] [PubMed] [Google Scholar]
- 34.Neel BG, Tonks NK. Protein tyrosine phosphatases in signal transduction. Current opinion in cell biology. 1997;9:193–204. doi: 10.1016/s0955-0674(97)80063-4. [DOI] [PubMed] [Google Scholar]
- 35.Xu M, Sharma A, Hossain MZ, Wiest DL, Sen JM. Sustained expression of pre-TCR induced beta-catenin in post-beta-selection thymocytes blocks T cell development. J Immunol. 2009;182:759–765. doi: 10.4049/jimmunol.182.2.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lovatt M, Bijlmakers MJ. Stabilisation of beta-catenin downstream of T cell receptor signalling. PLoS ONE. 5 doi: 10.1371/journal.pone.0012794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Goux D, Coudert JD, Maurice D, Scarpellino L, Jeannet G, Piccolo S, Weston K, Huelsken J, Held W. Cooperating pre-T-cell receptor and TCF-1-dependent signals ensure thymocyte survival. Blood. 2005;106:1726–1733. doi: 10.1182/blood-2005-01-0337. [DOI] [PubMed] [Google Scholar]
- 38.Weerkamp F, Baert MR, Naber BA, Koster EE, de Haas EF, Atkuri KR, van Dongen JJ, Herzenberg LA, Staal FJ. Wnt signaling in the thymus is regulated by differential expression of intracellular signaling molecules. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:3322–3326. doi: 10.1073/pnas.0511299103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu M, Sharma A, Wiest DL, Sen JM. Pre-TCR-induced betacatenin facilitates traversal through beta-selection. J Immunol. 2009;182:751–758. doi: 10.4049/jimmunol.182.2.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Verbeek S, Izon D, Hofhuis F, Robanus-Maandag E, te Riele H, van de Wetering M, Oosterwegel M, Wilson A, MacDonald HR, Clevers H. An HMG-box-containing T-cell factor required for thymocyte differentiation. Nature. 1995;374:70–74. doi: 10.1038/374070a0. [DOI] [PubMed] [Google Scholar]
- 41.Pongracz JE, Parnell SM, Jones T, Anderson G, Jenkinson EJ. Overexpression of ICAT highlights a role for catenin-mediated canonical Wnt signalling in early T cell development. European journal of immunology. 2006;36:2376–2383. doi: 10.1002/eji.200535721. [DOI] [PubMed] [Google Scholar]
- 42.Wang R, Xie H, Huang Z, Ma J, Fang X, Ding Y, Sun Z. TCF-1 regulates thymocyte survival via a RORγt-dependent pathway. J Immunol. 2011 doi: 10.4049/jimmunol.1101205. (paper in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huang Z, Xie H, Ioannidis V, Held W, Clevers H, Sadim MS, Sun Z. Transcriptional regulation of CD4 gene expression by T cell factor- 1/beta-catenin pathway. J Immunol. 2006;176:4880–4887. doi: 10.4049/jimmunol.176.8.4880. [DOI] [PubMed] [Google Scholar]
- 44.Xie H, Huang Z, Sadim MS, Sun Z. Stabilized beta-catenin extends thymocyte survival by up-regulating Bcl-xL. J Immunol. 2005;175:7981–7988. doi: 10.4049/jimmunol.175.12.7981. [DOI] [PubMed] [Google Scholar]
- 45.Hossain MZ, Yu Q, Xu M, Sen JM. ICAT expression disrupts beta-catenin-TCF interactions and impairs survival of thymocytes and activated mature T cells. International immunology. 2008;20:925–935. doi: 10.1093/intimm/dxn051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang R, Xie H, Huang Z, Ma J, Fang X, Ding Y, Sun Z. TCF-1 regulates thymocyte survival via a RORγ3t-dependent pathway. J Immunol. 2011 doi: 10.4049/jimmunol.1101205. (paper in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ioannidis V, Beermann F, Clevers H, Held W. The beta-catenin--TCF-1 pathway ensures CD4(+)CD8(+) thymocyte survival. Nature immunology. 2001;2:691–697. doi: 10.1038/90623. [DOI] [PubMed] [Google Scholar]
- 48.Sun Z, Unutmaz D, Zou YR, Sunshine MJ, Pierani A, Brenner-Morton S, Mebius RE, Littman DR. Requirement for RORgamma in thymocyte survival and lymphoid organ development. Science (New York, N.Y. 2000;288:2369–2373. doi: 10.1126/science.288.5475.2369. [DOI] [PubMed] [Google Scholar]
- 49.Xie H, Sadim MS, Sun Z. RORgammat recruits steroid receptor coactivators to ensure thymocyte survival. J Immunol. 2005;175:3800–3809. doi: 10.4049/jimmunol.175.6.3800. [DOI] [PubMed] [Google Scholar]
- 50.Xi H, Kersh GJ. Sustained early growth response gene 3 expression inhibits the survival of CD4/CD8 double-positive thymocytes. J Immunol. 2004;173:340–348. doi: 10.4049/jimmunol.173.1.340. [DOI] [PubMed] [Google Scholar]
- 51.D'Cruz LM, Knell J, Fujimoto JK, Goldrath AW. An essential role for the transcription factor HEB in thymocyte survival, Tcra rearrangement and the development of natural killer T cells. Nature immunology. 11:240–249. doi: 10.1038/ni.1845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yuan J, Crittenden RB, Bender TP. c-Myb promotes the survival of CD4+CD8+ double-positive thymocytes through upregulation of Bcl-xL. J Immunol. 184:2793–2804. doi: 10.4049/jimmunol.0902846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang R, Xie H, Huang Z, Ma J, Fang X, Ding Y, Sun Z. TCF-1 regulates thymocyte survival via a RORγt-dependent pathway. J Immunol. doi: 10.4049/jimmunol.1101205. (paper in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Guo Z, Dose M, Kovalovsky D, Chang R, O'Neil J, Look AT, von Boehmer H, Khazaie K, Gounari F. Beta-catenin stabilization stalls the transition from double-positive to single-positive stage and predisposes thymocytes to malignant transformation. Blood. 2007;109:5463–5472. doi: 10.1182/blood-2006-11-059071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yu Q, Xu M, Sen JM. Beta-catenin expression enhances IL-7 receptor signaling in thymocytes during positive selection. J Immunol. 2007;179:126–131. doi: 10.4049/jimmunol.179.1.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yu Q, Sen JM. Beta-catenin regulates positive selection of thymocytes but not lineage commitment. J Immunol. 2007;178:5028–5034. doi: 10.4049/jimmunol.178.8.5028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kovalovsky D, Yu Y, Dose M, Emmanouilidou A, Konstantinou T, Germar K, Aghajani K, Guo Z, Mandal M, Gounari F. Beta-catenin/Tcf determines the outcome of thymic selection in response to alphabetaTCR signaling. J Immunol. 2009;183:3873–3884. doi: 10.4049/jimmunol.0901369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Harada N, Tamai Y, Ishikawa T, Sauer B, Takaku K, Oshima M, Taketo MM. Intestinal polyposis in mice with a dominant stable mutation of the beta-catenin gene. The EMBO journal. 1999;18:5931–5942. doi: 10.1093/emboj/18.21.5931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gounari F, Aifantis I, Khazaie K, Hoeflinger S, Harada N, Taketo MM, von Boehmer H. Somatic activation of beta-catenin bypasses pre-TCR signaling and TCR selection in thymocyte development. Nature immunology. 2001;2:863–869. doi: 10.1038/ni0901-863. [DOI] [PubMed] [Google Scholar]
- 60.Dong C. Helper T-cell heterogeneity: a complex developmental issue in the immune system. Cell Mol Immunol. 2010;7:163. doi: 10.1038/cmi.2010.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Murphy KM, Reiner SL. The lineage decisions of helper T cells. Nature reviews. Immunology. 2002;2:933–944. doi: 10.1038/nri954. [DOI] [PubMed] [Google Scholar]
- 62.Yu Q, Sharma A, Oh SY, Moon HG, Hossain MZ, Salay TM, Leeds KE, Du H, Wu B, Waterman ML, Zhu Z, Sen JM. T cell factor 1 initiates the T helper type 2 fate by inducing the transcription factor GATA-3 and repressing interferon-gamma. Nature immunology. 2009;10:992–999. doi: 10.1038/ni.1762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Mosmann TR, Coffman RL. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annual review of immunology. 1989;7:145–173. doi: 10.1146/annurev.iy.07.040189.001045. [DOI] [PubMed] [Google Scholar]
- 64.Liew FY. T(H)1 and T(H)2 cells: a historical perspective. Nature reviews. Immunology. 2002;2:55–60. doi: 10.1038/nri705. [DOI] [PubMed] [Google Scholar]
- 65.Oestreich KJ, Huang AC, Weinmann AS. The lineage-defining factors T-bet and Bcl-6 collaborate to regulate Th1 gene expression patterns. The Journal of experimental medicine. 2011;208:1001–1013. doi: 10.1084/jem.20102144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dong C. TH17 cells in development: an updated view of their molecular identity and genetic programming. Nature reviews. Immunology. 2008;8:337–348. doi: 10.1038/nri2295. [DOI] [PubMed] [Google Scholar]
- 67.Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, Lucian L, To W, Kwan S, Churakova T, Zurawski S, Wiekowski M, Lira SA, Gorman D, Kastelein RA, Sedgwick JD. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature. 2003;421:744–748. doi: 10.1038/nature01355. [DOI] [PubMed] [Google Scholar]
- 68.Ouyang W, Kolls JK, Zheng Y. The biological functions of T helper 17 cell effector cytokines in inflammation. Immunity. 2008;28:454–467. doi: 10.1016/j.immuni.2008.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Matusevicius D, Kivisakk P, He B, Kostulas N, Ozenci V, Fredrikson S, Link H. Interleukin-17 mRNA expression in blood and CSF mononuclear cells is augmented in multiple sclerosis. Multiple sclerosis. 1999;5:101–104. doi: 10.1177/135245859900500206. [DOI] [PubMed] [Google Scholar]
- 70.Kirkham BW, Lassere MN, Edmonds JP, Juhasz KM, Bird PA, Lee CS, Shnier R, Portek IJ. Synovial membrane cytokine expression is predictive of joint damage progression in rheumatoid arthritis: a two-year prospective study (the DAMAGE study cohort) Arthritis and rheumatism. 2006;54:1122–1131. doi: 10.1002/art.21749. [DOI] [PubMed] [Google Scholar]
- 71.Molet S, Hamid Q, Davoine F, Nutku E, Taha R, Page N, Olivenstein R, Elias J, Chakir J. IL-17 is increased in asthmatic airways and induces human bronchial fibroblasts to produce cytokines. The Journal of allergy and clinical immunology. 2001;108:430–438. doi: 10.1067/mai.2001.117929. [DOI] [PubMed] [Google Scholar]
- 72.Duerr RH, Taylor KD, Brant SR, Rioux JD, Silverberg MS, Daly MJ, Steinhart AH, Abraham C, Regueiro M, Griffiths A, Dassopoulos T, Bitton A, Yang H, Targan S, Datta LW, Kistner EO, Schumm LP, Lee AT, Gregersen PK, Barmada MM, Rotter JI, Nicolae DL, Cho JH. A genome-wide association study identifies IL23R as an inflammatory bowel disease gene. Science. 2006;314:1461–1463. doi: 10.1126/science.1135245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Krueger GG, Langley RG, Leonardi C, Yeilding N, Guzzo C, Wang Y, Dooley LT, Lebwohl M. A human interleukin-12/23 monoclonal antibody for the treatment of psoriasis. The New England journal of medicine. 2007;356:580–592. doi: 10.1056/NEJMoa062382. [DOI] [PubMed] [Google Scholar]
- 74.Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, Weaver CT. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nature immunology. 2005;6:1123–1132. doi: 10.1038/ni1254. [DOI] [PubMed] [Google Scholar]
- 75.Harris TJ, Grosso JF, Yen HR, Xin H, Kortylewski M, Albesiano E, Hipkiss EL, Getnet D, Goldberg MV, Maris CH, Housseau F, Yu H, Pardoll DM, Drake CG. Cutting edge: An in vivo requirement for STAT3 signaling in TH17 development and TH17-dependent autoimmunity. Journal of immunology. 2007;179:4313–4317. doi: 10.4049/jimmunol.179.7.4313. [DOI] [PubMed] [Google Scholar]
- 76.Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A, Lafaille JJ, Cua DJ, Littman DR. The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126:1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 77.Ma J, Wang R, Fang X, Ding Y, Sun Z. Critical Role of TCF-1 in Repression of the IL-17 Gene. PloS one. 2011;6:e24768. doi: 10.1371/journal.pone.0024768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yang XO, Panopoulos AD, Nurieva R, Chang SH, Wang D, Watowich SS, Dong C. STAT3 regulates cytokine-mediated generation of inflammatory helper T cells. The Journal of biological chemistry. 2007;282:9358–9363. doi: 10.1074/jbc.C600321200. [DOI] [PubMed] [Google Scholar]
- 79.Li LN, Zhang HD, Yuan SJ, Yang DX, Wang L, Sun ZX. Differential sensitivity of colorectal cancer cell lines to artesunate is associated with expression of beta-catenin and E-cadherin. European journal of pharmacology. 2008;588:1–8. doi: 10.1016/j.ejphar.2008.03.041. [DOI] [PubMed] [Google Scholar]
- 80.Huber M, Brustle A, Reinhard K, Guralnik A, Walter G, Mahiny A, von Low E, Lohoff M. IRF4 is essential for IL-21-mediated induction, amplification, and stabilization of the Th17 phenotype. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:20846–20851. doi: 10.1073/pnas.0809077106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Veldhoen M, Hirota K, Westendorf AM, Buer J, Dumoutier L, Renauld JC, Stockinger B. The aryl hydrocarbon receptor links TH17-cellmediated autoimmunity to environmental toxins. Nature. 2008;453:106–109. doi: 10.1038/nature06881. [DOI] [PubMed] [Google Scholar]
- 82.Schraml BU, Hildner K, Ise W, Lee WL, Smith WA, Solomon B, Sahota G, Sim J, Mukasa R, Cemerski S, Hatton RD, Stormo GD, Weaver CT, Russell JH, Murphy TL, Murphy KM. The AP-1 transcription factor Batf controls T(H)17 differentiation. Nature. 2009;460:405–409. doi: 10.1038/nature08114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhou L, Lopes JE, Chong MM, Ivanov II, Min R, Victora GD, Shen Y, Du J, Rubtsov YP, Rudensky AY, Ziegler SF, Littman DR. TGF-beta-induced Foxp3 inhibits T(H)17 cell differentiation by antagonizing RORgammat function. Nature. 2008;453:236–240. doi: 10.1038/nature06878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Akimzhanov AM, Yang XO, Dong C. Chromatin remodeling of interleukin-17 (IL-17)-IL-17F cytokine gene locus during inflammatory helper T cell differentiation. The Journal of biological chemistry. 2007;282:5969–5972. doi: 10.1074/jbc.C600322200. [DOI] [PubMed] [Google Scholar]
- 85.Moisan J, Grenningloh R, Bettelli E, Oukka M, Ho IC. Ets-1 is a negative regulator of Th17 differentiation. The Journal of experimental medicine. 2007;204:2825–2835. doi: 10.1084/jem.20070994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Yu Q, Sharma A, Ghosh A, Sen JM. T cell factor-1 negatively regulates expression of IL-17 family of cytokines and protects mice from experimental autoimmune encephalomyelitis. Journal of immunology. 2011;186:3946–3952. doi: 10.4049/jimmunol.1003497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zhao DM, Yu S, Zhou X, Haring JS, Held W, Badovinac VP, Harty JT, Xue HH. Constitutive activation of Wnt signaling favors generation of memory CD8 T cells. Journal of immunology. 2010;184:1191–1199. doi: 10.4049/jimmunol.0901199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Willinger T, Freeman T, Herbert M, Hasegawa H, McMichael AJ, Callan MF. Human naive CD8 T cells down-regulate expression of the WNT pathway transcription factors lymphoid enhancer binding factor 1 and transcription factor 7 (T cell factor-1) following antigen encounter in vitro and in vivo. Journal of immunology. 2006;176:1439–1446. doi: 10.4049/jimmunol.176.3.1439. [DOI] [PubMed] [Google Scholar]
- 89.Marks BR, Nowyhed HN, Choi JY, Poholek AC, Odegard JM, Flavell RA, Craft J. Thymic self-reactivity selects natural interleukin 17-producing T cells that can regulate peripheral inflammation. Nature immunology. 2009;10:1125–1132. doi: 10.1038/ni.1783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lee CG, Sahoo A, Im SH. Epigenetic regulation of cytokine gene expression in T lymphocytes. Yonsei medical journal. 2009;50:322–330. doi: 10.3349/ymj.2009.50.3.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pokholok DK, Harbison CT, Levine S, Cole M, Hannett NM, Lee TI, Bell GW, Walker K, Rolfe PA, Herbolsheimer E, Zeitlinger J, Lewitter F, Gifford DK, Young RA. Genome-wide map of nucleosome acetylation and methylation in yeast. Cell. 2005;122:517–527. doi: 10.1016/j.cell.2005.06.026. [DOI] [PubMed] [Google Scholar]
- 92.Huang Z, Xie H, Ioannidis V, Held W, Clevers H, Sadim MS, Sun Z. Transcriptional regulation of CD4 gene expression by T cell factor- 1/beta-catenin pathway. Journal of immunology. 2006;176:4880–4887. doi: 10.4049/jimmunol.176.8.4880. [DOI] [PubMed] [Google Scholar]
- 93.Huang Z, Wang R, Xie H, Shang W, Manicassamy S, Sun Z. Stabilized beta-catenin potentiates Fas-mediated T cell apoptosis. Journal of immunology. 2008;180:6586–6592. doi: 10.4049/jimmunol.180.10.6586. [DOI] [PubMed] [Google Scholar]
- 94.Roose J, Molenaar M, Peterson J, Hurenkamp J, Brantjes H, Moerer P, van de Wetering M, Destree O, Clevers H. The Xenopus Wnt effector XTcf-3 interacts with Groucho-related transcriptional repressors. Nature. 1998;395:608–612. doi: 10.1038/26989. [DOI] [PubMed] [Google Scholar]
- 95.Ding Y, Shen S, Lino AC, Curotto de Lafaille MA, Lafaille JJ. Beta-catenin stabilization extends regulatory T cell survival and induces anergy in nonregulatory T cells. Nature medicine. 2008;14:162–169. doi: 10.1038/nm1707. [DOI] [PubMed] [Google Scholar]
- 96.Reya T, Clevers H. Wnt signalling in stem cells and cancer. Nature. 2005;434:843–850. doi: 10.1038/nature03319. [DOI] [PubMed] [Google Scholar]
- 97.Reya T, Duncan AW, Ailles L, Domen J, Scherer DC, Willert K, Hintz L, Nusse R, Weissman IL. A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature. 2003;423:409–414. doi: 10.1038/nature01593. [DOI] [PubMed] [Google Scholar]
- 98.Tough DF, Sprent J. Turnover of naive- and memory-phenotype T cells. The Journal of experimental medicine. 1994;179:1127–1135. doi: 10.1084/jem.179.4.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Luckey CJ, Bhattacharya D, Goldrath AW, Weissman IL, Benoist C, Mathis D. Memory T and memory B cells share a transcriptional program of self-renewal with long-term hematopoietic stem cells. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:3304–3309. doi: 10.1073/pnas.0511137103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhou X, Yu S, Zhao DM, Harty JT, Badovinac VP, Xue HH. Differentiation and persistence of memory CD8(+) T cells depend on T cell factor 1. Immunity. 2010;33:229–240. doi: 10.1016/j.immuni.2010.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Jeannet G, Boudousquie C, Gardiol N, Kang J, Huelsken J, Held W. Essential role of the Wnt pathway effector Tcf-1 for the establishment of functional CD8 T cell memory. Proceedings of the National Academy of Sciences of the United States of America. 2010;107:9777–9782. doi: 10.1073/pnas.0914127107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Intlekofer AM, Takemoto N, Wherry EJ, Longworth SA, Northrup JT, Palanivel VR, Mullen AC, Gasink CR, Kaech SM, Miller JD, Gapin L, Ryan K, Russ AP, Lindsten T, Orange JS, Goldrath AW, Ahmed R, Reiner SL. Effector and memory CD8+ T cell fate coupled by T-bet and eomesodermin. Nature immunology. 2005;6:1236–1244. doi: 10.1038/ni1268. [DOI] [PubMed] [Google Scholar]
- 103.Takemoto N, Intlekofer AM, Northrup JT, Wherry EJ, Reiner SL. Cutting Edge: IL-12 inversely regulates T-bet and eomesodermin expression during pathogen-induced CD8+ T cell differentiation. Journal of immunology. 2006;177:7515–7519. doi: 10.4049/jimmunol.177.11.7515. [DOI] [PubMed] [Google Scholar]
- 104.Pearce EL, Mullen AC, Martins GA, Krawczyk CM, Hutchins AS, Zediak VP, Banica M, DiCioccio CB, Gross DA, Mao CA, Shen H, Cereb N, Yang SY, Lindsten T, Rossant J, Hunter CA, Reiner SL. Control of effector CD8+ T cell function by the transcription factor Eomesodermin. Science. 2003;302:1041–1043. doi: 10.1126/science.1090148. [DOI] [PubMed] [Google Scholar]
- 105.Staal FJ, Luis TC, Tiemessen MM. WNT signalling in the immune system: WNT is spreading its wings. Nature reviews. Immunology. 2008;8:581–593. doi: 10.1038/nri2360. [DOI] [PubMed] [Google Scholar]
- 106.Gattinoni L, Zhong XS, Palmer DC, Ji Y, Hinrichs CS, Yu Z, Wrzesinski C, Boni A, Cassard L, Garvin LM, Paulos CM, Muranski P, Restifo NP. Wnt signaling arrests effector T cell differentiation and generates CD8+ memory stem cells. Nature medicine. 2009;15:808–813. doi: 10.1038/nm.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Prlic M, Bevan MJ. Cutting edge: beta-catenin is dispensable for T cell effector differentiation, memory formation, and recall responses. Journal of immunology. 2011;187:1542–1546. doi: 10.4049/jimmunol.1100907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Prlic M, Bevan MJ. Cutting edge: beta-catenin is dispensable for T cell effector differentiation, memory formation, and recall responses. J Immunol. 2011;187:1542–1546. doi: 10.4049/jimmunol.1100907. [DOI] [PMC free article] [PubMed] [Google Scholar]