Abstract
Background
The cornerstone of the adult hematopoietic system and clinical treatments for blood-related disease is the cohort of hematopoietic stem cells (HSC) that is harbored in the adult bone marrow microenvironment. Interestingly, this cohort of HSCs is generated only during a short window of developmental time. In mammalian embryos, hematopoietic progenitor and HSC generation occurs within several extra- and intra-embryonic microenvironments, most notably from ‘hemogenic’ endothelial cells lining the major vasculature. HSCs are made through a remarkable transdifferentiation of endothelial cells to a hematopoietic fate that is long-lived and self-renewable. Recent studies are beginning to provide an understanding of the biochemical signaling pathways and transcription factors/complexes that promote their generation.
Scope of the review
The focus of this review is on the biochemistry behind the generation of these potent long-lived self-renewing stem cells of the blood system. Both the intrinsic (master transcription factors) and extrinsic regulators (morphogens and growth factors) that affect the generation, maintenance and expansion of HSCs in the embryo will be discussed.
Major conclusions
The generation of HSCs is a stepwise process involving many developmental signaling pathways, morphogens and cytokines. Pivotal hematopoietic transcription factors are required for their generation. Interestingly, whereas these factors are necessary for HSC generation, their expression in adult bone marrow HSCs is oftentimes not required. Thus, the biochemistry and molecular regulation of HSC development in the embryo is overlapping, but differs significantly from the regulation of HSCs in the adult.
General significance
HSC numbers for clinical use are limiting, and despite much research into the molecular basis of HSC regulation in the adult bone marrow, no panel of growth factors, interleukins and/or morphogens has been found to sufficiently increase the number of these important stem cells. An understanding of the biochemistry of HSC generation in the developing embryo provides important new knowledge on how these complex stem cells are made, sustained and expanded in the embryo to give rise to the complete adult hematopoietic system, thus stimulating novel strategies for producing increased numbers of clinically useful HSCs.
Keywords: hematopoietic stem cells, AGM, development, transcription factors, signaling pathways
1. Ontogeny of hematopoietic stem cells
1.1. Multiple waves of de novo hematopoietic generation in the embryo
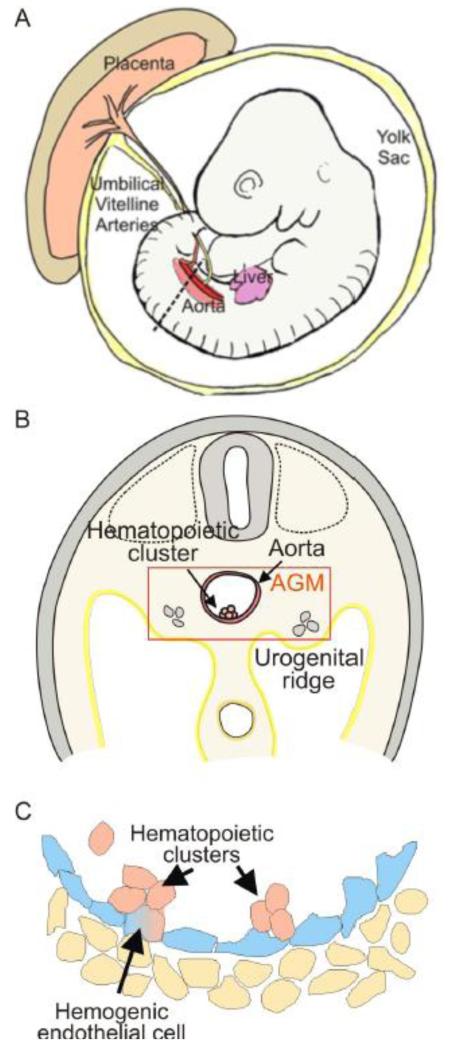
To understand the biochemistry behind HSC development, the cells that make up the vertebrate embryo blood system need some introduction. Blood cell specification occurs at least three separate times in the mammalian embryo – resulting in three de novo waves of hematopoietic cell production (reviewed in [1]). While it seems strange for embryos to establish the hematopoietic system multiple times, this in fact is a recurrent theme during ontogeny. For example, the mouse excretory system is generated first as the transient pronephric kidney, a secondary transient mesonephric kidney and finally as a third long-lived metanephric kidney that functions throughout adult life. The three distinct wave-like generations of the hematopoietic system provide a means by which the embryo can be temporarily supplied with rapidly produced hematopoietic cells, while generating a highly complex adult hematopoietic system with long-lived self-renewing hematopoietic stem cells (HSC) at its foundation. Hematopoiesis in the embryo occurs in several tissues that include the yolk sac, aorta-gonad-mesonephros (AGM) region, placenta and liver (Figure 1A).
Figure 1.
Hematopoietic stem cell development in the mouse embryo. A) Depiction of a mouse embryo at day 10.5 at the time when the first hematopoietic stem cells are generated in the aorta. Sites harboring (and/or generating) hematopoietic cells are shown: the extraembryonic yolk sac and placenta, the intraembryonic aorta and liver, and the umbilical and vitelline vessels that respectively connect the placenta and yolk sac to the aorta. The dotted line through the trunk of the embryo indicates the transverse section shown in panel B. B) Depiction of a transverse section through an E10.5 mouse embryo with the AGM (aorta-gonad-mesonephros/ aorta and urogenital ridges) region in the red rectangle. The AGM is flanked on the dorsal side by the neural tube and the somites, and on the ventral side by the gut and peritoneum. A hematopoietic cluster is indicated on the ventral wall of the dorsal aorta. Hematopoietic stem cells are localized in the clusters. C) A close up of the ventral wall of the aorta showing cluster formation. A hemogenic endothelial cell is undergoing the transition from endothelial cell to a hematopoietic cell.
The first wave of blood generation produces short-lived primitive erythrocytes that are necessary to carry oxygen through the rapidly growing conceptus and also primitive macrophages and megakaryocytes. Primitive erythrocytes are generated from aggregates of mesodermal precursors or ‘hemangioblasts’, in the yolk sac blood islands. Described over 100 years ago, the overlapping ontogenic appearance of both erythroid and endothelial cells indicates a common mesodermal precursor with at least bi-lineage potential [2-3]. This is further supported by the overlap in genetic programs for the two lineages (i.e. expression of Flk-1 (KDR), Scl (Tal1) and CD34) and the lack of both lineages in embryos deficient for some of these genes [4-6]. Surprisingly, hemangioblasts in vivo are localized not in the yolk sac but in the posterior primitive streak [7]. As they migrate to the yolk sac they begin their commitment to endothelial and hematopoietic progenitors, with several of these cells contributing to the formation of each blood island [8]. The first wave of primitive hematopoietic cell generation begins at embryonic day (E)7.5 in the mouse conceptus and is highly conserved across vertebrate species, including man (at 16-20 days of gestation [9].
In the mouse embryo the second wave of hematopoietic cell generation begins at E8/8.5, and overlaps with the first wave [10]. Definitive hematopoietic progenitors are de novo generated and some clusters of hematopoietic cells begin to appear in the major vasculature at E9.5. These hematopoietic progenitors are functionally more complex than primitive progenitors – they have multilineage potential (producing erythroid, myeloid and/or lymphoid cells), but they are not long-lived or self-renewing HSCs. De novo definitive progenitor generation occurs in the yolk sac, chorio-allantoic/placenta and the intraembryonic region around the aorta, as revealed by mouse embryo explant cultures and the Ncx1−/− mouse model (embryos lack circulation due to no heartbeat [11-12] (reviewed in [13]). Thus, ‘definitive hematopoietic progenitors’ constitute the second wave of hematopoietic specification.
The third wave of hematopoietic cell specification provides for the generation of adult type HSCs. Grafting studies in avian embryos provided unequivocal proof that the adult blood system is not derived from the yolk sac, but instead from an intraembryonic source of cells localizing to the dorsal aorta (reviewed in [1, 13]). Clusters of hematopoietic cells consistently found on the ventral wall of the dorsal aorta and major arteries of the chick embryo, led to the proposition that HSCs for the adult hematopoietic system arise from vascular endothelial cells (Figure 1B). Work in the mouse embryo showed that the first adult-type HSCs are generated in the intrabody AGM region (Figure 1A and B). These transplantable HSCs (as potent as adult bone marrow HSCs) are generated beginning at E10.5 and are thought to be contained within the vascular clusters within the aorta and vitelline and umbilical arteries [14-16]. The real-time generation of hematopoietic cells from ‘hemogenic endothelial cells’ lining the aorta (Figure 1C) has been demonstrated by vital confocal imaging in the mouse and zebrafish embryo [17-19] . The third wave of hematopoietic cell (HSC) generation is what generates the long-lived self-renewing HSCs that migrate, colonize and reside in the bone marrow throughout adult life.
1.2. Hemogenic endothelium as a source of definitive hematopoietic progenitors and HSCs
The generation of definitive hematopoietic progenitors (wave 2) and HSCs (wave 3) parallels the appearance of vascular hematopoietic clusters in the aorta, vitelline and umbilical arteries (Figure 1C). Histologic/immunostained sections through the midgestation embryo AGM region show that ‘hemogenic’ endothelial cells express some hematopoietic markers and some hematopoietic cluster cells express endothelial markers [20]. Cluster numbers peak at E10.5, when HSCs first appear. However, not all hematopoietic cells in the clusters are HSCs and not all clusters contain HSCs. There are many more cluster cells in the aorta than there are HSCs at this time point. Genetic studies using Cre-Lox recombination methods for deletion of pivotal intrinsic regulatory molecules show that HSC generation occurs only during a short window of developmental time [21-22]. It is unclear as yet whether all HSCs/cluster cells emerge from hemogenic endothelium, whether larger clusters form by proliferation of the emerging cell or through the recruitment of circulating cells. Recently, it was suggested that already hematopoietic committed cells (perhaps coming from the yolk sac, circulation [23] or other areas [24]) could mature to HSCs as they integrate into the appropriate microenvironment [24].
1.3 Molecular programming of HSC generation is distinct from programming of transient waves of hematopoietic cell generation
What makes the generation of HSCs so special as compared to earlier waves of blood generation? Each wave produces differentiated hematopoietic cells that circulate and move through the embryo to perform specific functions that are not so different from the differentiated hematopoietic cells arising from HSCs. Is it the microenvironment that directs the differences in cell potency, or is it the intrinsic program of the cells as they take on hematopoietic fate? Or is it both? These questions are presently driving investigations on the field of developmental hematopoiesis. The description of the complete genetic program of the nascent HSC and its direct precursor cell, the hemogenic endothelium, would open new possibilities and provide clues as to how to induce existing adult endothelial cells to become ‘hemogenic’ and thus, direct the programming of HSCs.
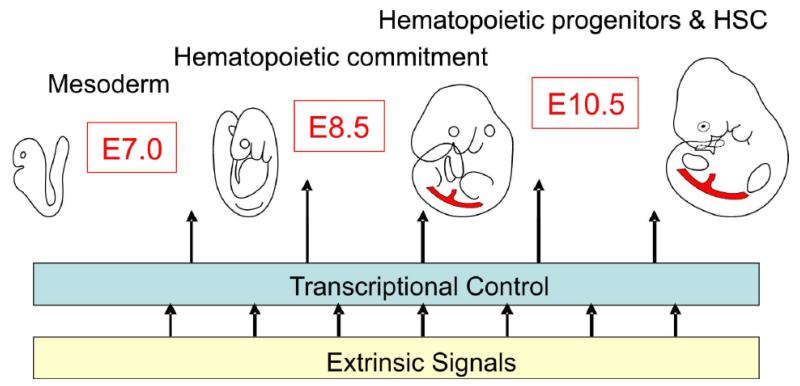
Indeed, the notion of different genetic requirements for the emergence of hematopoietic activities in specific embryonic sites and times has been revealed through targeted mutagenesis in mice. Differential requirements for the Runx1, Gata2, Gata3 transcription factors in fetal liver and AGM as compared to primitive yolk sac hematopoiesis has been shown (reviewed in [1, 13, 25]). Growth factor signaling, for example the receptor tyrosine kinase c-kit with its ligand SCF, is known to play roles during AGM and fetal liver stages, in activation of pathways that control hematopoietic cell survival, proliferation and/or differentiation (see review by Sharma and colleagues [26]). In contrast, the mutation of growth factor receptor Flk-1/VEGF ligand and the transcription factor Tal-l/SCL, results in the much earlier impairment at the embryonic yolk sac hematopoietic stage. Thus, the genetic programs of embryonic and adult hematopoietic cells appear to be somewhat overlapping during stages determining hematopoietic fate, but become unique as more complex properties are required in cells destined to become part of the adult hematopoietic system and may later become redundant. The molecular program guiding hematpoietic fate is initiated by extrinsic signals (growth factors and morphogens) coming from the surrounding microenvironment in the distinct stages of development and these signaling pathways activate the expression of the pivotal transcription factors (Figure 2).
Figure 2.
De novo generation of hematopoietic cells begins shortly after mesoderm formation and continues through mouse midgestation. Extrinsic factors that include morphogens such as FGF, Hh and BMPs produced in surrounding microenvironment affect mesodermal cells in hematopoietic fate choice and differentiation. These signaling pathways, as well as the Notch and VEGF pathways, impact directly or indirectly on the expression of several hematopoietic transcription factors in the presumptive hematopoietic cells in different hematopoietic sites and stages of development. The transcription factors directing hematopoietic fate and blood cell production include SCL, Gata2 and Runx1, amongst others. The specific temporal and spatial sequence of extrinsic signals, the combination and/or the levels of extrinsic signals play a role in the differential transcription factor expression and production of the distinct waves of hematopoietic cells in the developing embryo.
2. Intrinsic regulators of hematopoietic stem cell generation
Discoveries of three intrinsic regulators of HSC specification came from the genetic deletion experiments in the mouse. Pivotal hematopoietic transcription factors Scl, Gata2 and Runx1 were initially identified through chromosomal translocations and dysregulated expression in leukemic cells. Germ line deletion of each gene leads to midgestation embryonic lethality and profound anemia. Interestingly, these factors appear to work in complexes that bind to regulatory regions in a large overlapping set of hematopoietic specific genes.
2.1 SCL
The Scl (Scl/TAL-1/TCL-5) transcription factor is characterized by a basic helix-loop-helix domain (bHLH). As a member of the bHLH protein family, it acts through the HLH domain in a heterodimerization complex with E-proteins that recognize specific DNA sequences called Ebox motifs (G/ACANNTGG/A) [27-28]. Whereas Scl is most commonly known for its activity in acute T-cell leukemias (reviewed by [29]), the importance of Scl in normal hematopoiesis is still under research, using approaches that include germline and conditional deletion of the transcription factor in embryonic stem (ES) cells and mice.
Scl germline deficient mice show early embryonic lethality at E9.5, with an absence of hematopoiesis. This extensive anemic phenotype is also thought to be the cause of death of these embryos. No primitive or definitive hematopoietic cells are produced, yolk sac vasculature is abnormal and no vitelline vessels are formed [30-32] . Scl is required for hematopoietic specification in ventral mesoderm or ‘hemangioblast’ generation (wave 1). Its requirement for the subsequent 2nd and 3rd waves of mouse hematopoietic development is as yet unknown. The contribution of Scl−/− ES cells to the formation of mouse chimeras has also been tested. Scl−/− ES cells do not contribute to the formation of hematopoietic tissues, whereas they contribute to other various tissues. Further studies in this chimeric context proved Scl to be essential for lymphoid development [33]. An in vitro approach has been followed to test the hematopoietic potential of Scl−/− ES cells. These cells show a complete lack of myeloid and erythroid hematopoiesis, an effect that can be rescued by retroviral introduction of Scl cDNA in these cells or by tamoxifen inducible Scl expression [31, 33-34].
Scl is normally expressed in the developing embryo in the hematopoietic, endothelial and neural tissues. Expression in endothelial cells and hematopoietic tissues is of particular interest because of the developmental connection of these two cell lineages (hemangioblast, hemogenic endothelium) [35-36]. Several studies demonstrate the specific role of Scl in the proper development of both endothelial and hematopoietic cell lineages (reviewed by Lecuyer and Hoang 2004) [27] and suggest that it functions at the interface between vascular endothelial and hematopoietic lineages. Lancrin et al. found that Scl−/− Flk1+ ES-derived cells were unable to generate blast colonies and also that there was a complete lack of cells expressing hematopoietic markers c-kit and CD41, indicating that Scl is critical for the generation of the hemogenic endothelial population. Further studies by this group places its role in hematopoietic specification before the Runx1 requirement [37] (see below). Differentiation studies in Zebrafish embryos and in ES cells show that Scl is a regulator of mesodermal patterning [38-39]. Scl serves as an indirect negative regulator of cardiogenesis, and this function is independent of its later role in hematopoietic differentiation [40-41]. Thus, Scl plays an important role in the presumptive hematopoietic progenitor/stem cell mesoderm during embryonic development [41-43].
To define the effects of Scl deletion in adult mice, conditional knock out approaches have been taken, circumventing the embryonic lethality resulting from its germline deletion. When deleted in the adult, lineage specific effects of Scl deficiency are found in erythroid cells and megakaryocytes, but not other myeloid lineage cells. Interestingly, Scl participates in a “core transcriptional network” (together with Gata1 and Klf1) to regulate erythroid lineage differentiation[44]. It has been shown to be a direct regulator of a large group (>300) of erythroid specific genes, such as Eklf1, Gata1, glycophorin A, c-kit and globin [45]. Another study has revealed that Scl, through the transcriptional regulation of the cell cycle controller p21, regulates megakaryopoiesis. Importantly, although HSCs (and multipotent progenitors) express Scl, they do not rely on it for their ability to self-renew, differentiate and long-term reconstitute the blood system of irradiated adult recipients [43]. The apparent lack of effect of Scl deletion on adult HSCs is due to its redundancy with Lyl1[46]. Recent findings indicate that the levels of Scl expression control adult HSC quiescence by restricting the G1 entry of dormant HSCs [47].
2.2 Gata2
The Gata2 transcription factor which plays a role during the development of hematopoietic progenitor and stem cells is a member of the Gata family of factors. The 6 evolutionarily conserved proteins - Gata1 to Gata6 - recognize and bind the A/TGATAA/G DNA sequence from which they take their name, and have two highly conserved zinc finger (ZnF) domains [48]. Gata1, 2 and 3 are relevant to the hematopoietic system – Gata1 for the erythroid lineage, Gata2 for hematopoietic stem and progenitor cells, and Gata3 for T lymphocytes.
The expression pattern of Gata2 in the mouse embryo provides some information on its importance in the hematopoietic development. It is highly expressed hematopoietic cells of the fetal liver, placenta and in endothelial cells including the endothelium of the dorsal aorta from which HSCs emerge and intra-aortic hematopoietic cluster cells[49-52]. Moreover, high levels of Gata2 mRNA are detected in cell populations enriched for HSCs [53]. It is also expressed in the CNS and fetal heart,
The importance of Gata2 in the process of HSC generation was first highlighted by the creation of Gatat2 deficient mice. Gata2-/- embryos exhibit embryonic lethality between E10-E10.5 characterized by severe anemia [54]. Primitive hematopoietic progenitor numbers are decreased. Definitive hematopoietic progenitors are most profoundly affected and no HSCs are produced[55]. The rescue of this lethal Gata2−/− phenotype by functional complementation was performed using a YAC clone bearing the murine Gata2 genomic locus [56]. Further experiments performed with Gata2−/− ES cells differentiated into EBs under specific hematopoietic conditions, examined the hematopoietic progenitor compartment. The greatly reduced number of definitive hematopoietic progenitors and their poor expansion capacity lead to the conclusion that Gata2 is required for proliferation and survival of early hematopoietic cells [57]. Analysis of ES cells chimeric mice reveal a lack of contribution of Gata2−/− cells to any hematopoietic tissue [54]. Interestingly, Gata2+/- embryos are greatly reduced in AGM HSC numbers as shown by transplantation experiments [55]. In these studies the number of Ly6A-GFP cells, in which the HSC population can be found, is decreased 10 fold. However, by the time such animals reach adulthood the number of HSCs is normal but they are qualitatively impaired, as shown in competitive transplantation assays. Thus, Gata2 plays several distinct roles during development - HSC production, expansion and potency.
Putative genome wide target genes of Gata2 have been described recently in the hematopoietic progenitor cell line HPC-7. In genes important for hematopoiesis, Gata2 binding sites overlap with those of other transcriptional factors (such as Scl,,Runx1, Lmo2, Lyl1, Fli-1 and Erg), implying the collaboration of these factors in a “heptameric” transcription factor complex [58]. Another study in bone marrow progenitor cell populations suggests that Gata2 and Scl function in combination with Ldb1, another transcriptional factor essential for hematopoiesis [59].
Although, the Gata2−/− mouse studies provide unequivocal proof that Gata2 is one of the major key players in hematopoiesis, the precise function of this protein is still uncertain. Retroviral driven Gata2 overexpression in adult bone marrow hematopoietic progenitor cells blocks their ability to contribute to hematopoiesis as well as their ability to expand [60]. Gata2/ER enforced expression in FDCP cells (hematopoietic progenitor cell model) and in BA/F3 cells (IL-3 dependent hematopoietic progenitor cell line) triggers differentiation to monocyte and granulocyte cell fate, while it blocks self-renewal potential [61].These results reveal varying roles for Gata2 in multipotent hematopoietic progenitors and suggest that Gata2 levels make the difference. Particularly in embryonic development, the diploid dose of Gata2 is required to make the normal number of HSCs. The ability to reach the threshold level of Gata2 expression is probabilistic in the haploid context, thus many fewer HSCs are generated. Recently, GATA2 haploinsufficiency has been found to underly some human immunodeficiency syndromes [62]. Additionally, the effect of Gata2 in the growth suppression of hematopoietic progenitor cells may be explained by the formation of different complexes with other transcription factors that are expressed during later developmental stages [63]. Despite progress in understanding the function of Gata2, its exact role in HSC generation still needs further investigation.
2.3 Runx1
Runx1 (AML1, Cbfα) belongs to the family of the DNA binding proteins that include Runx2 and Runx3 that have high homology with the Drosophila gene runt (reviewed in [64]. Runx1 binds directly to DNA sequence TGT/CGGT through its Runt homology domain (RHD), which is a 118 amino acids domain centrally located in the protein. Runx1 heterodimerizes with Cbfβ to form the core binding factor (CBF). This interaction increases its affinity for the DNA consensus sequence [65-66].
The high frequency of chromosomal rearrangements in the Runx1 (AML) locus in acute myeloid leukemia (AML) cells was one of the reasons that made Runx1 an interesting subject for intense study in the blood system (reviewed by [67]). Runx1 binds to the regulatory elements (enhancer core motifs) of several genes and provides for tissue specific gene expression of molecules known to be important for hematopoiesis. Some of these genes encode GM-CSF, IL-3 and CSF1 receptor [68].
The importance of Runx1 in the development of the hematopoietic system was first revealed in Runx1 deficient mice. Deletion of this transcription factor results in severe hematopoietic defects that lead to embryonic lethality at E12.5. In contrast the early normal production of the primitive hematopoietic system, the most obvious effect of Runx1 deficiency is the complete absence of definitive hematopoiesis in the yolk sac (YS) and the fetal liver (FL) at midgestation. The AGM region was found to contain no HSCs. Lack of contribution to the hematopoietic tissues by Runx1−/− cells was also observed in chimeric mice made with Runx1−/− ES cells. EB differentiation of Runx1−/− ES cells and analysis for the presence of hematopoietic progenitors confirmed the lack of definitive hematopoiesis [65, 68-69]. Interestingly, deficiency of core binding factor β (Cbfβ), the partner of Runx1 in the core binding factor complex (CBF), results in the same phenotype as Runx1 deficiency [65, 70-71].
The temporal and spatial expression pattern of Runx1 argues for its importance in the generation of the HSCs and progenitor cells. Runx1 expression localizes to the endothelial cells of the yolk sac, the vitelline and umbilical arteries, the placenta and most importantly the ventral wall of the dorsal aorta [12, 16, 72]. It is expressed in the aorta just before the emergence of the HSCs and the progenitor cells, implicating Runx1 in the hematopoietic fate process in hemogenic endothelial cells. Evidence to support this theory showed that Runx1-/- embryos lack hematopoietic clusters and ex vivo cultures of Runx1-/- endothelial cells possess no hematopoietic potential [16, 73]. A more recent study [21] confirmed the specific role of Runx1 in the generation of hematopoietic cells from the hemogenic endothelial cells. Specific deletion of Runx1 in VE-cadherin (VEC) expressing endothelial cells (VEC-Cre mediated Runx1 excision) resulted in lack of HSC and progenitor generation, phenocopying the hematopoietic defects found in the germline deleted embryos. Interestingly, the specific deletion of the gene in a hematopoietic cell context (Vav-cre mediated Runx1 excision) showed that Runx1 is no longer required after HSCs are generated. Thus, Runx1 plays an important role in the endothelial to hematopoietic transition process by which definitive progenitors and HSCs are generated. However, although it is expressed in HSCs after they are generated, Runx1 function is no longer required.
Similarly to Gata2, haploinsufficiency of Runx1 has notable effects on HSC generation. A haploid dose of the transcriptional factor leads to only half the number of HSCs in the AGM region [74]. It also results in the slightly earlier appearance of HSCs in the normal sites of hematopoiesis in the embryo [75]. Haploinsufficiency and trisomy of Runx1 (on chromosome 21) in man predisposes the individual and/or results in hematopoietic dysfunction (leukemia and Downs Syndrome). Thus, proper regulated control of transcription factor levels in HSCs is an important but often less studied aspect. An interesting experiment to test whether Gata2 and Runx1 function in the same cells (precursors, hemogenic endothelial, hematopoietic cells) was performed. Since adult Runx1+/− and Gata2+/− mice are viable and present with a relatively normal hematopoietic blood profile, matings were established to generate double heterozygous mice. Interestingly, no Runx1+/−:Gata2+/− mice were born. However, Runx1+/−:Gata2+/− embryos could be harvested and fetal livers were found to contain fewer hematopoietic progenitors than single heterozygous mutants [58]. These findings, together with chromatin immunoprecipitation studies in hematopoietic cell lines, strongly suggest that Runx1 and Gata2 act in concert to control expression of hematopoietic genes involved in HSC and progenitor cell generation.
3. Extrinsic regulators of hematopoietic stem cell generation
The embryonic microenvironment surrounding the major vasculature – the dorsal aorta, vitelline and umbilical arteries – has been a major focus of study to identify and characterize the extrinsic regulators involved in the generation of HSCs. Signaling pathways such as TGFβ, BMP, Hedgehog, Wnt and Notch control hematopoiesis in the embryo. Since HSCs emerge from the ventral endothelium of the aorta, it is thought that the underlying mesenchymal cells produce signaling molecules that impact directly or indirectly on hemogenic endothelial and emerging hematopoietic cells. Indeed, the midline expression of Hh in the gut and the ventral expression of BMP4 in the mouse embryo suggest that these factors play an in vivo physiologic role in hematopoietic specification and generation in the aorta. In Zebrafish, BMP4 affects Runx1 and Gata2 expression [76] and in the adult mouse, the BMP pathway controls the number of HSCs by regulating the size of the niche[77]. Moreover, the Hedgehog (Hh) signaling pathway has also been implicated in Zebrafish hematopoietic development through its induction of VEGF production, subsequent Notch expression and thereafter, Notch mediated expression of Gata2 in the aorta[78]. In the adult mouse, several independent groups have demonstrated that Hh does not regulate HSCs (reviewed by [79]). In contrast, HSCs have been found to be controlled in a dose dependent manner by the Wnt signaling pathway[80]. At early developmental times, the Wnt/β-catenin signaling pathway is required for the transition between hemogenic endothelium to HSC in the AGM region of the mouse embryo, but it is not required for their maintenance[81]. This pathway also plays a role in the specification of HSC in the Zebrafish AGM[82]. We describe here some of the known factors and signaling pathways, including FGF, VEGF, Notch, TGFβ, Hh and BMP4, that although they have been studied individually, they presumably converge in the same cohort of cells to affect hematopoietic ontogeny in the mammalian embryo.
3.1 Hedgehog
The Hedgehog signaling pathway is critical during development of the major organ systems of vertebrates and acts by controlling proliferation, cell survival, migration and differentiation in a time and dose dependent manner [83]. Endoderm-mesoderm interactions are essential for hematopoietic induction in the developing mouse conceptus, with Hh being produced by endodermal cells. Visceral endoderm and Hh signals have been shown to divert the fate of prospective neuroectoderm to hematopoietic fate [84]. In zebrafish, Hh induces VEGF expression in the somites that leads to the migration of lateral mesodermal cells to the midline [85]. Gene targeting studies show that Hh is required for murine YS hematopoiesis/angiogenesis. Ihh expression is found in the endoderm layer of the YS, and Gli1 (intracellular transducer of Hh pathway) expressing cells are found in the mesoderm beginning at E6.5 [86]. In parallel, VEGF, a downstream target of Hh, is expressed in YS endoderm. The Flk-1 gene, encoding the receptor of VEGF, is expressed in the mesoderm and in the embryo body and placenta in the vasculature [5, 87]. Despite its important role in vasculogenesis, only 50% of Ihh−/− embryos do not have any hematological defects at E11. However, they become anemic and have a much smaller fetal liver (FL) at E13.5. It is during this developmental stage when definitive hematopoietic cells are established and maintained in the fetal liver microenvironment [88]. This study, as well as studies in zebrafish, indicate that the Hh signaling pathway controls definitive but not primitive hematopoiesis [78, 88]. Moreover, during Drosophila development, Hh signaling molecules instruct cells within the hematopoietic niche to maintain hematopoietic progenitors in a precursor state [89]. We examined the effects of the ventrally located gut tissue, which is Hh producing, on the generation of the definitive HSC in the AGM region of mouse embryo. Gut tissue specifically induced HSC activity in early E10 AGM, as did exogenously added Hh [90]. Thus, Hh signaling is early player in AGM HSC generation although other pathways may compensate in its absence.
3.2 BMP
Hh and BMP signaling act in a polarizing manner on adult blood formation in the Zebrafish aorta, with Hh expression in the notochord (dorsal) and BMP expressed ventrally [91]. Deficiencies in the intracellular transducers of the BMP signaling pathway, Smad1 or Smad5, results in the failure of the embryo to generate definitive hematopoietic progenitors [92]. In vitro, BMP4 plays a major role in regulating hematopoietic differentiation in murine and human embryonic stem cells [93-94]. At high concentration, BMP4 mediated signaling acts to maintain the proliferation of HSCs derived from human umbilical cord blood rather than promoting their differentiation [95]. In mouse, we found BMP4 mRNA and protein expressed in the ventral mesenchyme underlying the E10/11 mouse aorta. Ly6A-GFP+ hemogenic endothelial cells, HSCs and hematopoietic clusters are in close association with these mesenchymal cells and express BMP receptors, suggesting the direct action of BMP4 on these cells [96]. Addition of BMP4 to E11 AGM explants enhances HSC numbers, while Gremlin, a BMP antagonist suppresses HSCs. Our recent data show that BMP4 is indispensable for HSC growth in AGM explants cultured under serum-free conditions (Crisan, M, unpublished observations). This is consistent with the data showing that Smad5 deficient mice die between E9.5 and E11.5 with variable phenotypes affecting amnion, gut, heart, neural tube and the YS where the absence of a well organised vasculature is observed [97] Indeed, immunostainings with an anti-phospho (P)-Smad1/5/8 antibody (BMP4/2 specific Smads) show P-Smad expression n ventral aortic endothelium and hematopoietic cells (unpublished results, A. Zwijsen, Leuven, BE; P ten Dijke, Leiden, NL), suggesting that the pathway is highly active in the E11 AGM. Our most recent preliminary data in BRE-GFP (BMP Response Element) transgenic mice show GFP+ cells in the vascular hematopoietic clusters and aortic endothelium at E11, and that transplantable AGM HSCs are BMP activated. However, the canonical BMP signaling pathway is not required for hematopoietic stem and progenitor cells in either the adult bone marrow or fetal liver suggesting that other pathways may regulate HSC development [98]. In the same time, Gli1−/− mice show increased long-term HSC engraftment [99]. This is consistent with the effect of cyclopamine, a Hh antagonist, on hES cells where blood development was observed to be BMP-dependent [100]. Thus, BMP and Hh pathways may converge to control definitive hematopoiesis during mouse ontogeny.
3.3 FGF
FGFs regulate cellular proliferation, survival, migration, and differentiation. It is important for mesoderm induction during gastrulation followed by the dorso-ventral patterning of the mesoderm [101-104] FGF is also acting as a inducer of neovascularization and neoangiogenesis [105-106] and is involved in promoting proliferation and maintenance of hematopoietic progenitors and stem cells [107]. It is unclear whether FGF is required during hematopoietic ontogeny. Studies show that FGF positively regulates hematopoietic stem and progenitor cells [108-110] although recent data demonstrate that FGF-1 alone was unable to support hematopoietic progenitor proliferation in vitro [111]. However, in combination with SCF, TPO and IGF-2, a cytokine cocktail so-called STIF medium defined by Zhang, FGF-1 efficiently supported the in vitro amplification of murine and human HSCs [112]. Hematopoietic stem and progenitor expansion was further increased in the presence of mesenchymal stromal cells, most likely due to the synergic effects of the growth factor cocktail containing FGF-1 and cell-cell contacts between the cells [111].
Using mice overexpressing constitutively active FGFR2 (under control of the Tie-2 promoter), Shigematsu et al. demonstrate that FGFR2 signaling facilitates donor hematopoietic engraftment in a transplantation scenario and that this is due to its anti-apoptotic effects [113]. Finally, high FGF activity negatively regulates the formation of primitive blood in chick embryo and promotes endothelial cell differentiation. Opposing results are obtained when FGFR2 is blocked [114]. The negative influence of FGF signaling was also reported in Xenopus embryos through downregulation of Gata2 transcription factor, a key hematopoietic target gene of BMP4 [115]. Cooperation between FGF and other signaling pathways also occurs in other cells such as osteoblasts, which serve as the hematopoietic niche for adult HSPC (reviewed by [116]). Altogether, these studies demonstrate that FGF acts not only directly through hematopoietic stem and progenitor cells, but also acts indirectly through their niche.
3.4 VEGF
VEGFA is well known angiogenic factor and acts mainly through VEGFR2, also named Flk-1 or KDR. Complete deficiency and haploinsufficiency of VEGF during murine development causes embryonic lethality between E9.5 and E10 due to an absence of angiogenesis and hematopoiesis [87, 117]. Mice lacking Flk-1 fail to develop definitive HSCs and die between E8.5 and E9.5 [5]. Consistent with this study, Flk-1-/- ES cells do not contribute to primitive or definitive hematopoiesis in chimeric embryos [118].
VEGF is also essential for definitive hematopoiesis in the zebrafish embryo. This is supported by the expression of Flk-1 in Runx 1 expressing blood cells [78]. In zebrafish, VEGF is downstream to Hh signaling pathway, but can also be regulated by canonical BMP pathway as its promoter contains BMP activated Smad-binding elements [78, 119].
During mouse development, VEGF is produced by the visceral endoderm (as is Ihh). Gata4-/- ES cells differentiated to embryoid bodies demonstrate a disruption in the formation of the visceral endoderm, VEGF and Ihh expression and definitive hematopoietic progenitor growth. However, exogenously added VEGF and Ihh can rescue definitive hematopoietic progenitor production in Gata 4-/- embryoid bodies [120]. Also, VEGF can act in synergy with bFGF in human ES cell cultures to specify hemogenic endothelial and hematopoietic cells. In serum-free conditions a combination of BMP4 and VEGF induce hematopoiesis from murine ES cells and induced pluripotent stem cells [121-123].
VEGFA is also required to maintain adult hematopoietic stem cells. Although VEGF is present in the host environment, VEGF deficient HSCs do not engraft adult irradiated recipient mice suggesting that HSC are regulated through an autocrine mechanism [124]. However, adult HSCs express the VEGFR and other experiments suggest that HSC engraftment can also be dependent on the VEGF signaling pathway [125-126].
3.5 Notch
Notch signaling is required for the specification and generation of definitive HSCs from hemogenic endothelium during development [127-129]. Inhibition of different components of Notch signaling by mutation or targeted deletion results in severe vascular defects. Arterial vessels, the location of the first generated definitive HSCs, are lacking and there is a total absence of HSCs [52, 128, 130]. In contrast to the severe defects in definitive hematopoiesis in Notch1-/- embryos, YS hematopoietic development is not affected. Thus, Notch1 is required only for definitive hematopoietic progenitor and HSC generation [127, 129]. In mouse embryo, Notch1 activates Gata2 expression (a direct Notch target) in the aorta. Notch1 is expressed in some endothelial cells lining the ventral wall of the aorta and transcriptional transactivation studies suggest regulation of AGM HSCs through activation of Gata2 expression [129].
In Zebrafish Notch mutants, Runx1 (used as a hematopoietic progenitor/stem cells marker) is not expressed. However, when heat shock inducible Notch1a intracellular domain is transiently activated during embryogenesis, the number of Runx1 expressing cells as compared to control is expanded, suggesting that Runx1 is a Notch target gene in hematopoietic progenitor/stem cells [131]. Downregulation of Notch 1 by the morpholino approach results in highly altered definitive myelopoiesis from the early stages of development [132]. Whereas some Notch ligands may be expressed in the aorta, the somitic expression of the Notch ligands is required for the establishment of definitive haematopoiesis, as recently shown in the zebrafish embryo [133].
The Notch signaling pathway cooperates with other pathways to control hematopoiesis. Studies in zebrafish and mice have shown that Notch specification of HSCs is regulated by VEGFA, downstream of Hh signaling pathway (reviewed by [128]. Moreover in Drosophila, Notch gain and loss of function phenotypes are similar to that of hypoxic genes controlling hematopoietic cells. Data recently showed that interactions between Notch and Hif-signaling pathways control hematopoietic development [134].
4. Future Perspectives
The challenges currently at the leading edge of research into HSC development, are to define in vivo the biochemical and cellular processes that affect the onset and implementation of the hematopoietic gene expression program in each of the de novo waves of hematogenesis, and to identify the specific multistep program and network of interactions that lead to the genesis of adult HSCs in the 3rd wave. Increasingly complex molecular programs directed by pivotal transcription factors Scl, Gata2 and Runx1 culminate in the generation of HSCs and the adult hematopoietic system and are only the starting point in our understanding of how HSCs are made. Taking a multidisciplinary approach and using state-of-the-art molecular, cellular and microscopic (3-D whole embryo confocal and real-time vital confocal imaging) techniques, a wide variety of reagents and mouse models, it should be possible to track, isolate and characterize presumptive HSCs during all ontogenic stages as these cells metamorphose through the mesoderm, endothelium, hemogenic endothelium and emerge as HSCs. Important issues include whether the presumptive cells simultaneously express the pivotal transcription factors Scl, Gata2 and Runx1 or whether there is a sequential/orderly factor requirement; how levels of transcription factors affect the expression of downstream targets, how many signaling pathways impact on HSC development and what are the specific effects of each pathway and how the pathways syngergise. The results of such studies will lead to insights allowing the development of novel strategies for the generation of HSCs from human vascular tissue or human iPS cells.
Highlights.
Current knowledge about hematopoietic stem cell ontogeny in vertebrate embryos
Intrinsic/transcription factors – SCL, Runx, Gata2 – that direct HSC generation
Developmental morphogens/factors that are extrinsic cues for HSC generation
Acknowledgements
We thank all lab members for helpful discussions and the NIH (R37 DK51077 to ED), ZonMW (911.09.036 to ED; ZonMW VENI (016.126.088 to MC), EMBO LTF (ALTF 206-2009 to MC), FES NIRM (ED) for support.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Dzierzak E, Speck NA. Of lineage and legacy: the development of mammalian hematopoietic stem cells. Nat Immunol. 2008;9:129–136. doi: 10.1038/ni1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Murray P. The development in vitro of the blood of the early chick embryo. Proc Roy Soc London. 1932;11:497–521. [Google Scholar]
- [3].Sabin F. Studies on the origin of blood vessels and of red blood corpuscles as seen in the living blastoderm of chicks during the second day of incubation, Carnegie Inst. Wash. Pub. # 272. Contrib. Embryol. 1920;9:214. [Google Scholar]
- [4].Park C, Ma YD, Choi K. Evidence for the hemangioblast. Exp Hematol. 2005;33:965–970. doi: 10.1016/j.exphem.2005.06.003. [DOI] [PubMed] [Google Scholar]
- [5].Shalaby F, Rossant J, Yamaguchi TP, Gertsenstein M, Wu XF, Breitman ML, Schuh AC. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature. 1995;376:62–66. doi: 10.1038/376062a0. [DOI] [PubMed] [Google Scholar]
- [6].Tavian M, Hallais MF, Peault B. Emergence of intraembryonic hematopoietic precursors in the pre-liver human embryo. Development. 1999;126:793–803. doi: 10.1242/dev.126.4.793. [DOI] [PubMed] [Google Scholar]
- [7].Huber TL, Kouskoff V, Fehling HJ, Palis J, Keller G. Haemangioblast commitment is initiated in the primitive streak of the mouse embryo. Nature. 2004;432:625–630. doi: 10.1038/nature03122. [DOI] [PubMed] [Google Scholar]
- [8].Ueno H, Weissman IL. Clonal analysis of mouse development reveals a polyclonal origin for yolk sac blood islands. Dev Cell. 2006;11:519–533. doi: 10.1016/j.devcel.2006.08.001. [DOI] [PubMed] [Google Scholar]
- [9].Tavian M, Peault B. Embryonic development of the human hematopoietic system. Int J Dev Biol. 2005;49:243–250. doi: 10.1387/ijdb.041957mt. [DOI] [PubMed] [Google Scholar]
- [10].Palis J, Robertson S, Kennedy M, Wall C, Keller G. Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development. 1999;126:5073–5084. doi: 10.1242/dev.126.22.5073. [DOI] [PubMed] [Google Scholar]
- [11].Lux CT, Yoshimoto M, McGrath K, Conway SJ, Palis J, Yoder MC. All primitive and definitive hematopoietic progenitor cells emerging before E10 in the mouse embryo are products of the yolk sac. Blood. 2008;111:3435–3438. doi: 10.1182/blood-2007-08-107086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Rhodes KE, Gekas C, Wang Y, Lux CT, Francis CS, Chan DN, Conway S, Orkin SH, Yoder MC, Mikkola HK. The emergence of hematopoietic stem cells is initiated in the placental vasculature in the absence of circulation. Cell Stem Cell. 2008;2:252–263. doi: 10.1016/j.stem.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Medvinsky A, Rybtsov S, Taoudi S. Embryonic origin of the adult hematopoietic system: advances and questions. Development. 2011;138:1017–1031. doi: 10.1242/dev.040998. [DOI] [PubMed] [Google Scholar]
- [14].de Bruijn MF, Ma X, Robin C, Ottersbach K, Sanchez MJ, Dzierzak E. Hematopoietic stem cells localize to the endothelial cell layer in the midgestation mouse aorta. Immunity. 2002;16:673–683. doi: 10.1016/s1074-7613(02)00313-8. [DOI] [PubMed] [Google Scholar]
- [15].de Bruijn MF, Speck NA, Peeters MC, Dzierzak E. Definitive hematopoietic stem cells first develop within the major arterial regions of the mouse embryo. Embo J. 2000;19:2465–2474. doi: 10.1093/emboj/19.11.2465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].North T, Gu TL, Stacy T, Wang Q, Howard L, Binder M, Marin-Padilla M, Speck NA. Cbfa2 is required for the formation of intra-aortic hematopoietic clusters. Development. 1999;126:2563–2575. doi: 10.1242/dev.126.11.2563. [DOI] [PubMed] [Google Scholar]
- [17].Boisset JC, van Cappellen W, Andrieu-Soler C, Galjart N, Dzierzak E, Robin C. In vivo imaging of haematopoietic cells emerging from the mouse aortic endothelium. Nature. 2010;464:116–120. doi: 10.1038/nature08764. [DOI] [PubMed] [Google Scholar]
- [18].Kissa K, Herbomel P. Blood stem cells emerge from aortic endothelium by a novel type of cell transition. Nature. 2010;464:112–115. doi: 10.1038/nature08761. [DOI] [PubMed] [Google Scholar]
- [19].Bertrand JY, Chi NC, Santoso B, Teng S, Stainier DY, Traver D. Haematopoietic stem cells derive directly from aortic endothelium during development. Nature. 2010;464:108–111. doi: 10.1038/nature08738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Yokomizo T, Yamada-Inagawa T, Yzaguirre AD, Chen MJ, Speck NA, Dzierzak E. Whole-mount three-dimensional imaging of internally localized immunostained cells within mouse embryos. Nat Protoc. 2012;7:421–431. doi: 10.1038/nprot.2011.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Chen MJ, Yokomizo T, Zeigler BM, Dzierzak E, Speck NA. Runx1 is required for the endothelial to haematopoietic cell transition but not thereafter. Nature. 2009;457:887–891. doi: 10.1038/nature07619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Zovein AC, Hofmann JJ, Lynch M, French WJ, Turlo KA, Yang Y, Becker MS, Zanetta L, Dejana E, Gasson JC, Tallquist MD, Iruela-Arispe ML. Fate tracing reveals the endothelial origin of hematopoietic stem cells. Cell Stem Cell. 2008;3:625–636. doi: 10.1016/j.stem.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Yoder MC, Hiatt K, Dutt P, Mukherjee P, Bodine DM, Orlic D. Characterization of definitive lymphohematopoietic stem cells in the day 9 murine yolk sac. Immunity. 1997;7:335–344. doi: 10.1016/s1074-7613(00)80355-6. [DOI] [PubMed] [Google Scholar]
- [24].Rybtsov S, Sobiesiak M, Taoudi S, Souilhol C, Senserrich J, Liakhovitskaia A, Ivanovs A, Frampton J, Zhao S, Medvinsky A. Hierarchical organization and early hematopoietic specification of the developing HSC lineage in the AGM region. J Exp Med. 2011;208:1305–1315. doi: 10.1084/jem.20102419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Orkin SH, Zon LI. Hematopoiesis: an evolving paradigm for stem cell biology. Cell. 2008;132:631–644. doi: 10.1016/j.cell.2008.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Sharma S, Gurudutta GU, Satija NK, Pati S, Afrin F, Gupta P, Verma YK, Singh VK, Tripathi RP. Stem cell c-KIT and HOXB4 genes: critical roles and mechanisms in self-renewal, proliferation, and differentiation. Stem Cells Dev. 2006;15:755–778. doi: 10.1089/scd.2006.15.755. [DOI] [PubMed] [Google Scholar]
- [27].Lecuyer E, Hoang T. SCL: from the origin of hematopoiesis to stem cells and leukemia. Exp Hematol. 2004;32:11–24. doi: 10.1016/j.exphem.2003.10.010. [DOI] [PubMed] [Google Scholar]
- [28].Murre C. Helix-loop-helix proteins and lymphocyte development. Nat Immunol. 2005;6:1079–1086. doi: 10.1038/ni1260. [DOI] [PubMed] [Google Scholar]
- [29].Begley CG, Green AR. The SCL gene: from case report to critical hematopoietic regulator. Blood. 1999;93:2760–2770. [PubMed] [Google Scholar]
- [30].Shivdasani RA, Mayer EL, Orkin SH. Absence of blood formation in mice lacking the T-cell leukaemia oncoprotein tal-1/SCL. Nature. 1995;373:432–434. doi: 10.1038/373432a0. [DOI] [PubMed] [Google Scholar]
- [31].Robb L, Elwood NJ, Elefanty AG, Kontgen F, Li R, Barnett LD, Begley CG. The scl gene product is required for the generation of all hematopoietic lineages in the adult mouse. EMBO J. 1996;15:4123–4129. [PMC free article] [PubMed] [Google Scholar]
- [32].Robb L, Lyons I, Li R, Hartley L, Kontgen F, Harvey RP, Metcalf D, Begley CG. Absence of yolk sac hematopoiesis from mice with a targeted disruption of the scl gene. Proc Natl Acad Sci U S A. 1995;92:7075–7079. doi: 10.1073/pnas.92.15.7075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Porcher C, Swat W, Rockwell K, Fujiwara Y, Alt FW, Orkin SH. The T cell leukemia oncoprotein SCL/tal-1 is essential for development of all hematopoietic lineages. Cell. 1996;86:47–57. doi: 10.1016/s0092-8674(00)80076-8. [DOI] [PubMed] [Google Scholar]
- [34].Endoh M, Ogawa M, Orkin S, Nishikawa S. SCL/tal-1-dependent process determines a competence to select the definitive hematopoietic lineage prior to endothelial differentiation. EMBO J. 2002;21:6700–6708. doi: 10.1093/emboj/cdf674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Elefanty AG, Begley CG, Hartley L, Papaevangeliou B, Robb L. SCL expression in the mouse embryo detected with a targeted lacZ reporter gene demonstrates its localization to hematopoietic, vascular, and neural tissues. Blood. 1999;94:3754–3763. [PubMed] [Google Scholar]
- [36].Kallianpur AR, Jordan JE, Brandt SJ. The SCL/TAL-1 gene is expressed in progenitors of both the hematopoietic and vascular systems during embryogenesis. Blood. 1994;83:1200–1208. [PubMed] [Google Scholar]
- [37].Lancrin C, Sroczynska P, Stephenson C, Allen T, Kouskoff V, Lacaud G. The haemangioblast generates haematopoietic cells through a haemogenic endothelium stage. Nature. 2009;457:892–895. doi: 10.1038/nature07679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Ismailoglu I, Yeamans G, Daley GQ, Perlingeiro RC, Kyba M. Mesodermal patterning activity of SCL. Exp Hematol. 2008;36:1593–1603. doi: 10.1016/j.exphem.2008.07.005. [DOI] [PubMed] [Google Scholar]
- [39].Peterkin T, Gibson A, Patient R. Common genetic control of haemangioblast and cardiac development in zebrafish. Development. 2009;136:1465–1474. doi: 10.1242/dev.032748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Schoenebeck JJ, Keegan BR, Yelon D. Vessel and blood specification override cardiac potential in anterior mesoderm. Dev Cell. 2007;13:254–267. doi: 10.1016/j.devcel.2007.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Van Handel B, Montel-Hagen A, Sasidharan R, Nakano H, Ferrari R, Boogerd CJ, Schredelseker J, Wang Y, Hunter S, Org T, Zhou J, Li X, Pellegrini M, Chen JN, Orkin SH, Kurdistani SK, Evans SM, Nakano A, Mikkola HK. Scl represses cardiomyogenesis in prospective hemogenic endothelium and endocardium. Cell. 2012;150:590–605. doi: 10.1016/j.cell.2012.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Hall MA, Curtis DJ, Metcalf D, Elefanty AG, Sourris K, Robb L, Gothert JR, Jane SM, Begley CG. The critical regulator of embryonic hematopoiesis, SCL, is vital in the adult for megakaryopoiesis, erythropoiesis, and lineage choice in CFU-S12. Proc Natl Acad Sci U S A. 2003;100:992–997. doi: 10.1073/pnas.0237324100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Mikkola HK, Klintman J, Yang H, Hock H, Schlaeger TM, Fujiwara Y, Orkin SH. Haematopoietic stem cells retain long-term repopulating activity and multipotency in the absence of stem-cell leukaemia SCL/tal-1 gene. Nature. 2003;421:547–551. doi: 10.1038/nature01345. [DOI] [PubMed] [Google Scholar]
- [44].Wontakal SN, Guo XY, Smith C, MacCarthy T, Bresnick EH, Bergman A, Snyder MP, Weissman SM, Zheng DY, Skoultchi AI. A core erythroid transcriptional network is repressed by a master regulator of myelo-lymphoid differentiation. Proc Natl Acad Sci U S A. 2012;109:3832–3837. doi: 10.1073/pnas.1121019109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Kassouf MT, Hughes JR, Taylor S, McGowan SJ, Soneji S, Green AL, Vyas P, Porcher C. Genome-wide identification of TAL1’s functional targets: insights into its mechanisms of action in primary erythroid cells. Genome Res. 2010;20:1064–1083. doi: 10.1101/gr.104935.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Souroullas GP, Salmon JM, Sablitzky F, Curtis DJ, Goodell MA. Adult hematopoietic stem and progenitor cells require either Lyl1 or Scl for survival. Cell Stem Cell. 2009;4:180–186. doi: 10.1016/j.stem.2009.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Lacombe J, Herblot S, Rojas-Sutterlin S, Haman A, Barakat S, Iscove NN, Sauvageau G, Hoang T. Scl regulates the quiescence and the long-term competence of hematopoietic stem cells. Blood. 2010;115:792–803. doi: 10.1182/blood-2009-01-201384. [DOI] [PubMed] [Google Scholar]
- [48].Ko LJ, Engel JD. DNA-binding specificities of the GATA transcription factor family. Mol Cell Biol. 1993;13:4011–4022. doi: 10.1128/mcb.13.7.4011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Minegishi N, Suzuki N, Yokomizo T, Pan X, Fujimoto T, Takahashi S, Hara T, Miyajima A, Nishikawa S, Yamamoto M. Expression and domain-specific function of GATA2 during differentiation of the hematopoietic precursor cells in midgestation mouse embryos. Blood. 2003;102:896–905. doi: 10.1182/blood-2002-12-3809. [DOI] [PubMed] [Google Scholar]
- [50].Nardelli J, Thiesson D, Fujiwara Y, Tsai FY, Orkin SH. Expression and genetic interaction of transcription factors GATA2 and GATA3 during development of the mouse central nervous system. Dev Biol. 1999;210:305–321. doi: 10.1006/dbio.1999.9278. [DOI] [PubMed] [Google Scholar]
- [51].Ng YK, George KM, Engel JD, Linzer DI. GATA factor activity is required for the trophoblast-specific transcriptional regulation of the mouse placental lactogen I gene. Development. 1994;120:3257–3266. doi: 10.1242/dev.120.11.3257. [DOI] [PubMed] [Google Scholar]
- [52].Robert-Moreno A, Espinosa L, de la Pompa JL, Bigas A. RBPjkappa-dependent Notch function regulates Gata2 and is essential for the formation of intra-embryonic hematopoietic cells. Development. 2005;132:1117–1126. doi: 10.1242/dev.01660. [DOI] [PubMed] [Google Scholar]
- [53].Orlic D, Anderson S, Biesecker LG, Sorrentino BP, Bodine DM. Pluripotent hematopoietic stem cells contain high levels of mRNA for c-kit, GATA2, p45 NF-E2, and c-myb and low levels or no mRNA for c-fms and the receptors for granulocyte colony-stimulating factor and interleukins 5 and 7. Proc Natl Acad Sci U S A. 1995;92:4601–4605. doi: 10.1073/pnas.92.10.4601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Tsai FY, Keller G, Kuo FC, Weiss M, Chen J, Rosenblatt M, Alt FW, Orkin SH. An early haematopoietic defect in mice lacking the transcription factor GATA2. Nature. 1994;371:221–226. doi: 10.1038/371221a0. [DOI] [PubMed] [Google Scholar]
- [55].Ling KW, Ottersbach K, van Hamburg JP, Oziemlak A, Tsai FY, Orkin SH, Ploemacher R, Hendriks RW, Dzierzak E. GATA2 plays two functionally distinct roles during the ontogeny of hematopoietic stem cells. J Exp Med. 2004;200:871–882. doi: 10.1084/jem.20031556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Zhou Y, Lim KC, Onodera K, Takahashi S, Ohta J, Minegishi N, Tsai FY, Orkin SH, Yamamoto M, Engel JD. Rescue of the embryonic lethal hematopoietic defect reveals a critical role for GATA2 in urogenital development. EMBO J. 1998;17:6689–6700. doi: 10.1093/emboj/17.22.6689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Tsai FY, Orkin SH. Transcription factor GATA2 is required for proliferation/survival of early hematopoietic cells and mast cell formation, but not for erythroid and myeloid terminal differentiation. Blood. 1997;89:3636–3643. [PubMed] [Google Scholar]
- [58].Wilson NK, Foster SD, Wang X, Knezevic K, Schutte J, Kaimakis P, Chilarska PM, Kinston S, Ouwehand WH, Dzierzak E, Pimanda JE, de Bruijn MF, Gottgens B. Combinatorial transcriptional control in blood stem/progenitor cells: genome-wide analysis of ten major transcriptional regulators. Cell Stem Cell. 2010;7:532–544. doi: 10.1016/j.stem.2010.07.016. [DOI] [PubMed] [Google Scholar]
- [59].Li L, Jothi R, Cui K, Lee JY, Cohen T, Gorivodsky M, Tzchori I, Zhao Y, Hayes SM, Bresnick EH, Zhao K, Westphal H, Love PE. Nuclear adaptor Ldb1 regulates a transcriptional program essential for the maintenance of hematopoietic stem cells. Nat Immunol. 2011;12:129–136. doi: 10.1038/ni.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Persons DA, Allay JA, Allay ER, Ashmun RA, Orlic D, Jane SM, Cunningham JM, Nienhuis AW. Enforced expression of the GATA2 transcription factor blocks normal hematopoiesis. Blood. 1999;93:488–499. [PubMed] [Google Scholar]
- [61].Heyworth C, Gale K, Dexter M, May G, Enver T. A GATA2/estrogen receptor chimera functions as a ligand-dependent negative regulator of self-renewal. Genes Dev. 1999;13:1847–1860. doi: 10.1101/gad.13.14.1847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Bresnick EH, Katsumura KR, Lee HY, Johnson KD, Perkins AS. Master regulatory GATA transcription factors: mechanistic principles and emerging links to hematologic malignancies. Nucleic Acids Res. 2012;40:5819–5831. doi: 10.1093/nar/gks281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Ezoe S, Matsumura I, Nakata S, Gale K, Ishihara K, Minegishi N, Machii T, Kitamura T, Yamamoto M, Enver T, Kanakura Y. GATA2/estrogen receptor chimera regulates cytokine-dependent growth of hematopoietic cells through accumulation of p21(WAF1) and p27(Kip1) proteins. Blood. 2002;100:3512–3520. doi: 10.1182/blood-2002-04-1177. [DOI] [PubMed] [Google Scholar]
- [64].Swiers G, de Bruijn M, Speck NA. Hematopoietic stem cell emergence in the conceptus and the role of Runx1. Int J Dev Biol. 2010;54:1151–1163. doi: 10.1387/ijdb.103106gs. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [65].Wang Q, Stacy T, Miller JD, Lewis AF, Gu TL, Huang X, Bushweller JH, Bories JC, Alt FW, Ryan G, Liu PP, Wynshaw-Boris A, Binder M, Marin-Padilla M, Sharpe AH, Speck NA. The CBFbeta subunit is essential for CBFalpha2 (AML1) function in vivo. Cell. 1996;87:697–708. doi: 10.1016/s0092-8674(00)81389-6. [DOI] [PubMed] [Google Scholar]
- [66].Wang S, Wang Q, Crute BE, Melnikova IN, Keller SR, Speck NA. Cloning and characterization of subunits of the T-cell receptor and murine leukemia virus enhancer core-binding factor. Mol Cell Biol. 1993;13:3324–3339. doi: 10.1128/mcb.13.6.3324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Rabbitts TH. Chromosomal translocations in human cancer. Nature. 1994;372:143–149. doi: 10.1038/372143a0. [DOI] [PubMed] [Google Scholar]
- [68].Okuda T, vanDeursen J, Hiebert SW, Grosveld G, Downing JR. AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell. 1996;84:321–330. doi: 10.1016/s0092-8674(00)80986-1. [DOI] [PubMed] [Google Scholar]
- [69].Mukouyama Y, Chiba N, Hara T, Okada H, Ito Y, Kanamaru R, Miyajima A, Satake M, Watanabe T. The AML1 transcription factor functions to develop and maintain hematogenic precursor cells in the embryonic aorta-gonad-mesonephros region. Dev Biol. 2000;220:27–36. doi: 10.1006/dbio.2000.9617. [DOI] [PubMed] [Google Scholar]
- [70].Sasaki K, Yagi H, Bronson RT, Tominaga K, Matsunashi T, Deguchi K, Tani Y, Kishimoto T, Komori T. Absence of fetal liver hematopoiesis in mice deficient in transcriptional coactivator core binding factor beta. Proc Natl Acad Sci U S A. 1996;93:12359–12363. doi: 10.1073/pnas.93.22.12359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [71].Wang Q, Stacy T, Binder M, Marin-Padilla M, Sharpe AH, Speck NA. Disruption of the Cbfa2 gene causes necrosis and hemorrhaging in the central nervous system and blocks definitive hematopoiesis. Proc Natl Acad Sci U S A. 1996;93:3444–3449. doi: 10.1073/pnas.93.8.3444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [72].Ottersbach K, Dzierzak E. The murine placenta contains hematopoietic stem cells within the vascular labyrinth region. Dev Cell. 2005;8:377–387. doi: 10.1016/j.devcel.2005.02.001. [DOI] [PubMed] [Google Scholar]
- [73].Yokomizo T, Ogawa M, Osato M, Kanno T, Yoshida H, Fujimoto T, Fraser S, Nishikawa S, Okada H, Satake M, Noda T, Ito Y. Requirement of Runx1/AML1/PEBP2alphaB for the generation of haematopoietic cells from endothelial cells. Genes Cells. 2001;6:13–23. doi: 10.1046/j.1365-2443.2001.00393.x. [DOI] [PubMed] [Google Scholar]
- [74].Robin C, Ottersbach K, Durand C, Peeters M, Vanes L, Tybulewicz V, Dzierzak E. An unexpected role for IL-3 in the embryonic development of hematopoietic stem cells. Dev Cell. 2006;11:171–180. doi: 10.1016/j.devcel.2006.07.002. [DOI] [PubMed] [Google Scholar]
- [75].Cai Z, de Bruijn M, Ma X, Dortland B, Luteijn T, Downing RJ, Dzierzak E. Haploinsufficiency of AML1 affects the temporal and spatial generation of hematopoietic stem cells in the mouse embryo. Immunity. 2000;13:423–431. doi: 10.1016/s1074-7613(00)00042-x. [DOI] [PubMed] [Google Scholar]
- [76].Liu F, Walmsley M, Rodaway A, Patient R. Fli1 acts at the top of the transcriptional network driving blood and endothelial development. Curr Biol. 2008;18:1234–1240. doi: 10.1016/j.cub.2008.07.048. [DOI] [PubMed] [Google Scholar]
- [77].Zhang J, Niu C, Ye L, Huang H, He X, Tong WG, Ross J, Haug J, Johnson T, Feng JQ, Harris S, Wiedemann LM, Mishina Y, Li L. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003;425:836–841. doi: 10.1038/nature02041. [DOI] [PubMed] [Google Scholar]
- [78].Gering M, Patient R. Hedgehog signaling is required for adult blood stem cell formation in zebrafish embryos. Dev Cell. 2005;8:389–400. doi: 10.1016/j.devcel.2005.01.010. [DOI] [PubMed] [Google Scholar]
- [79].Mar BG, Amakye D, Aifantis I, Buonamici S. The controversial role of the Hedgehog pathway in normal and malignant hematopoiesis. Leukemia. 2011;25:1665–1673. doi: 10.1038/leu.2011.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [80].Luis TC, Naber BA, Roozen PP, Brugman MH, de Haas EF, Ghazvini M, Fibbe WE, van Dongen JJ, Fodde R, Staal FJ. Canonical wnt signaling regulates hematopoiesis in a dosage-dependent fashion. Cell Stem Cell. 2011;9:345–356. doi: 10.1016/j.stem.2011.07.017. [DOI] [PubMed] [Google Scholar]
- [81].Ruiz-Herguido C, Guiu J, D’Altri T, Ingles-Esteve J, Dzierzak E, Espinosa L, Bigas A. Hematopoietic stem cell development requires transient Wnt/beta-catenin activity. J Exp Med. 2012;209:1457–1468. doi: 10.1084/jem.20120225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [82].Goessling W, North TE, Loewer S, Lord AM, Lee S, Stoick-Cooper CL, Weidinger G, Puder M, Daley GQ, Moon RT, Zon LI. Genetic interaction of PGE2 and Wnt signaling regulates developmental specification of stem cells and regeneration. Cell. 2009;136:1136–1147. doi: 10.1016/j.cell.2009.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Ingham PW, McMahon AP. Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 2001;15:3059–3087. doi: 10.1101/gad.938601. [DOI] [PubMed] [Google Scholar]
- [84].Dyer MA, Farrington SM, Mohn D, Munday JR, Baron MH. Indian hedgehog activates hematopoiesis and vasculogenesis and can respecify prospective neurectodermal cell fate in the mouse embryo. Development. 2001;128:1717–1730. doi: 10.1242/dev.128.10.1717. [DOI] [PubMed] [Google Scholar]
- [85].Ciau-Uitz A, Walmsley M, Patient R. Distinct origins of adult and embryonic blood in Xenopus. Cell. 2000;102:787–796. doi: 10.1016/s0092-8674(00)00067-2. [DOI] [PubMed] [Google Scholar]
- [86].Farrington SM, Belaoussoff M, Baron MH. Winged-helix, Hedgehog and Bmp genes are differentially expressed in distinct cell layers of the murine yolk sac. Mech Dev. 1997;62:197–211. doi: 10.1016/s0925-4773(97)00664-3. [DOI] [PubMed] [Google Scholar]
- [87].Carmeliet P, Ferreira V, Breier G, Pollefeyt S, Kieckens L, Gertsenstein M, Fahrig M, Vandenhoeck A, Harpal K, Eberhardt C, Declercq C, Pawling J, Moons L, Collen D, Risau W, Nagy A. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature. 1996;380:435–439. doi: 10.1038/380435a0. [DOI] [PubMed] [Google Scholar]
- [88].Cridland SO, Keys JR, Papathanasiou P, Perkins AC. Indian hedgehog supports definitive erythropoiesis. Blood Cells Mol Dis. 2009;43:149–155. doi: 10.1016/j.bcmd.2009.04.004. [DOI] [PubMed] [Google Scholar]
- [89].Tokusumi Y, Tokusumi T, Stoller-Conrad J, Schulz RA. Serpent, suppressor of hairless and U-shaped are crucial regulators of hedgehog niche expression and prohemocyte maintenance during Drosophila larval hematopoiesis. Development. 2010;137:3561–3568. doi: 10.1242/dev.053728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [90].Peeters M, Ottersbach K, Bollerot K, Orelio C, de Bruijn M, Wijgerde M, Dzierzak E. Ventral embryonic tissues and Hedgehog proteins induce early AGM hematopoietic stem cell development. Development. 2009;136:2613–2621. doi: 10.1242/dev.034728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Wilkinson RN, Pouget C, Gering M, Russell AJ, Davies SG, Kimelman D, Patient R. Hedgehog and Bmp polarize hematopoietic stem cell emergence in the zebrafish dorsal aorta. Dev Cell. 2009;16:909–916. doi: 10.1016/j.devcel.2009.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].McReynolds LJ, Gupta S, Figueroa ME, Mullins MC, Evans T. Smad1 and Smad5 differentially regulate embryonic hematopoiesis. Blood. 2007;110:3881–3890. doi: 10.1182/blood-2007-04-085753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Chadwick K, Wang L, Li L, Menendez P, Murdoch B, Rouleau A, Bhatia M. Cytokines and BMP4 promote hematopoietic differentiation of human embryonic stem cells. Blood. 2003;102:906–915. doi: 10.1182/blood-2003-03-0832. [DOI] [PubMed] [Google Scholar]
- [94].Johansson BM, Wiles MV. Evidence for involvement of activin A and bone morphogenetic protein 4 in mammalian mesoderm and hematopoietic development. Mol Cell Biol. 1995;15:141–151. doi: 10.1128/mcb.15.1.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Bhatia M, Bonnet D, Wu D, Murdoch B, Wrana J, Gallacher L, Dick JE. Bone morphogenetic proteins regulate the developmental program of human hematopoietic stem cells. J Exp Med. 1999;189:1139–1148. doi: 10.1084/jem.189.7.1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [96].Durand C, Robin C, Bollerot K, Baron MH, Ottersbach K, Dzierzak E. Embryonic stromal clones reveal developmental regulators of definitive hematopoietic stem cells. Proc Natl Acad Sci U S A. 2007;104:20838–20843. doi: 10.1073/pnas.0706923105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [97].Chang H, Huylebroeck D, Verschueren K, Guo Q, Matzuk MM, Zwijsen A. Smad5 knockout mice die at mid-gestation due to multiple embryonic and extraembryonic defects. Development. 1999;126:1631–1642. doi: 10.1242/dev.126.8.1631. [DOI] [PubMed] [Google Scholar]
- [98].Singbrant S, Karlsson G, Ehinger M, Olsson K, Jaako P, Miharada K, Stadtfeld M, Graf T, Karlsson S. Canonical BMP signaling is dispensable for hematopoietic stem cell function in both adult and fetal liver hematopoiesis, but essential to preserve colon architecture. Blood. 2010;115:4689–4698. doi: 10.1182/blood-2009-05-220988. [DOI] [PubMed] [Google Scholar]
- [99].Merchant A, Joseph G, Wang Q, Brennan S, Matsui W. Gli1 regulates the proliferation and differentiation of HSCs and myeloid progenitors. Blood. 2010;115:2391–2396. doi: 10.1182/blood-2009-09-241703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [100].Cerdan C, Bhatia M. Novel roles for Notch, Wnt and Hedgehog in hematopoesis derived from human pluripotent stem cells. Int J Dev Biol. 2010;54:955–963. doi: 10.1387/ijdb.103067cc. [DOI] [PubMed] [Google Scholar]
- [101].Furthauer M, Van Celst J, Thisse C, Thisse B. Fgf signalling controls the dorsoventral patterning of the zebrafish embryo. Development. 2004;131:2853–2864. doi: 10.1242/dev.01156. [DOI] [PubMed] [Google Scholar]
- [102].Griffin K, Patient R, Holder N. Analysis of FGF function in normal and no tail zebrafish embryos reveals separate mechanisms for formation of the trunk and the tail. Development. 1995;121:2983–2994. doi: 10.1242/dev.121.9.2983. [DOI] [PubMed] [Google Scholar]
- [103].Slack JM, Darlington BG, Heath JK, Godsave SF. Mesoderm induction in earlyXenopus embryos by heparin-binding growth factors. Nature. 1987;326:197–200. doi: 10.1038/326197a0. [DOI] [PubMed] [Google Scholar]
- [104].Yamaguchi TP, Harpal K, Henkemeyer M, Rossant J. fgfr-1 is required for embryonic growth and mesodermal patterning during mouse gastrulation. Genes Dev. 1994;8:3032–3044. doi: 10.1101/gad.8.24.3032. [DOI] [PubMed] [Google Scholar]
- [105].Auguste P, Javerzat S, Bikfalvi A. Regulation of vascular development by fibroblast growth factors. Cell Tissue Res. 2003;314:157–166. doi: 10.1007/s00441-003-0750-0. [DOI] [PubMed] [Google Scholar]
- [106].Presta M, Dell’Era P, Mitola S, Moroni E, Ronca R, Rusnati M. Fibroblast growth factor/fibroblast growth factor receptor system in angiogenesis. Cytokine Growth Factor Rev. 2005;16:159–178. doi: 10.1016/j.cytogfr.2005.01.004. [DOI] [PubMed] [Google Scholar]
- [107].Moroni E, Dell’Era P, Rusnati M, Presta M. Fibroblast growth factors and their receptors in hematopoiesis and hematological tumors. J Hematother Stem Cell Res. 2002;11:19–32. doi: 10.1089/152581602753448513. [DOI] [PubMed] [Google Scholar]
- [108].Allouche M, Bikfalvi A. The role of fibroblast growth factor-2 (FGF-2) in hematopoiesis. Prog Growth Factor Res. 1995;6:35–48. doi: 10.1016/0955-2235(95)00041-0. [DOI] [PubMed] [Google Scholar]
- [109].Gordon-Thomson C, Fabian BC. Hypoblastic tissue and fibroblast growth factor induce blood tissue (haemoglobin) in the early chick embryo. Development. 1994;120:3571–3579. doi: 10.1242/dev.120.12.3571. [DOI] [PubMed] [Google Scholar]
- [110].Huber TL, Zhou Y, Mead PE, Zon LI. Cooperative effects of growth factors involved in the induction of hematopoietic mesoderm. Blood. 1998;92:4128–4137. [PubMed] [Google Scholar]
- [111].Walenda T, Bokermann G, Ventura Ferreira MS, Piroth DM, Hieronymus T, Neuss S, Zenke M, Ho AD, Muller AM, Wagner W. Synergistic effects of growth factors and mesenchymal stromal cells for expansion of hematopoietic stem and progenitor cells. Exp Hematol. 2011;39:617–628. doi: 10.1016/j.exphem.2011.02.011. [DOI] [PubMed] [Google Scholar]
- [112].Zhang CC, Lodish HF. Murine hematopoietic stem cells change their surface phenotype during ex vivo expansion. Blood. 2005;105:4314–4320. doi: 10.1182/blood-2004-11-4418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [113].Shigematsu A, Shi M, Okigaki M, Adachi Y, Koike N, Che J, Iwasaki M, Matsubara H, Imamura M, Ikehara S. Signaling from fibroblast growth factor receptor 2 in immature hematopoietic cells facilitates donor hematopoiesis after intra-bone marrow-bone marrow transplantation. Stem Cells Dev. 2010;19:1679–1686. doi: 10.1089/scd.2009.0370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [114].Nakazawa F, Nagai H, Shin M, Sheng G. Negative regulation of primitive hematopoiesis by the FGF signaling pathway. Blood. 2006;108:3335–3343. doi: 10.1182/blood-2006-05-021386. [DOI] [PubMed] [Google Scholar]
- [115].Xu RH, Ault KT, Kim J, Park MJ, Hwang YS, Peng Y, Sredni D, Kung H. Opposite effects of FGF and BMP4 on embryonic blood formation: roles of PV.1 and GATA2. Dev Biol. 1999;208:352–361. doi: 10.1006/dbio.1999.9205. [DOI] [PubMed] [Google Scholar]
- [116].Marie PJ, Miraoui H, Severe N. FGF/FGFR signaling in bone formation: progress and perspectives. Growth Factors. 2012;30:117–123. doi: 10.3109/08977194.2012.656761. [DOI] [PubMed] [Google Scholar]
- [117].Ferrara N, Carver-Moore K, Chen H, Dowd M, Lu L, O’Shea KS, Powell-Braxton L, Hillan KJ, Moore MW. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature. 1996;380:439–442. doi: 10.1038/380439a0. [DOI] [PubMed] [Google Scholar]
- [118].Shalaby F, Ho J, Stanford WL, Fischer KD, Schuh AC, Schwartz L, Bernstein A, Rossant J. A requirement for Flk1 in primitive and definitive hematopoiesis and vasculogenesis. Cell. 1997;89:981–990. doi: 10.1016/s0092-8674(00)80283-4. [DOI] [PubMed] [Google Scholar]
- [119].He C, Chen X. Transcription regulation of the vegf gene by the BMP/Smad pathway in the angioblast of zebrafish embryos. Biochem Biophys Res Commun. 2005;329:324–330. doi: 10.1016/j.bbrc.2005.01.133. [DOI] [PubMed] [Google Scholar]
- [120].Pierre M, Yoshimoto M, Huang L, Richardson M, Yoder MC. VEGF and IHH rescue definitive hematopoiesis in Gata-4 and Gata-6-deficient murine embryoid bodies. Exp Hematol. 2009;37:1038–1053. doi: 10.1016/j.exphem.2009.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [121].Grigoriadis AE, Kennedy M, Bozec A, Brunton F, Stenbeck G, Park IH, Wagner EF, Keller GM. Directed differentiation of hematopoietic precursors and functional osteoclasts from human ES and iPS cells. Blood. 2010;115:2769–2776. doi: 10.1182/blood-2009-07-234690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [122].Irion S, Clarke RL, Luche H, Kim I, Morrison SJ, Fehling HJ, Keller GM. Temporal specification of blood progenitors from mouse embryonic stem cells and induced pluripotent stem cells. Development. 2010;137:2829–2839. doi: 10.1242/dev.042119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [123].Wang C, Tang X, Sun X, Miao Z, Lv Y, Yang Y, Zhang H, Zhang P, Liu Y, Du L, Gao Y, Yin M, Ding M, Deng H. TGFbeta inhibition enhances the generation of hematopoietic progenitors from human ES cell-derived hemogenic endothelial cells using a stepwise strategy. Cell Res. 2012;22:194–207. doi: 10.1038/cr.2011.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [124].Gerber HP, Malik AK, Solar GP, Sherman D, Liang XH, Meng G, Hong K, Marsters JC, Ferrara N. VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism. Nature. 2002;417:954–958. doi: 10.1038/nature00821. [DOI] [PubMed] [Google Scholar]
- [125].Chan CK, Chen CC, Luppen CA, Kim JB, DeBoer AT, Wei K, Helms JA, Kuo CJ, Kraft DL, Weissman IL. Endochondral ossification is required for haematopoietic stem-cell niche formation. Nature. 2009;457:490–494. doi: 10.1038/nature07547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [126].Hooper AT, Butler JM, Nolan DJ, Kranz A, Iida K, Kobayashi M, Kopp HG, Shido K, Petit I, Yanger K, James D, Witte L, Zhu Z, Wu Y, Pytowski B, Rosenwaks Z, Mittal V, Sato TN, Rafii S. Engraftment and reconstitution of hematopoiesis is dependent on VEGFR2-mediated regeneration of sinusoidal endothelial cells. Cell Stem Cell. 2009;4:263–274. doi: 10.1016/j.stem.2009.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [127].Bertrand JY, Cisson JL, Stachura DL, Traver D. Notch signaling distinguishes 2 waves of definitive hematopoiesis in the zebrafish embryo. Blood. 2010;115:2777–2783. doi: 10.1182/blood-2009-09-244590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [128].Gering M, Patient R. Notch signalling and haematopoietic stem cell formation during embryogenesis. J Cell Physiol. 2010;222:11–16. doi: 10.1002/jcp.21905. [DOI] [PubMed] [Google Scholar]
- [129].Kumano K, Chiba S, Kunisato A, Sata M, Saito T, Nakagami-Yamaguchi E, Yamaguchi T, Masuda S, Shimizu K, Takahashi T, Ogawa S, Hamada Y, Hirai H. Notch1 but not Notch2 is essential for generating hematopoietic stem cells from endothelial cells. Immunity. 2003;18:699–711. doi: 10.1016/s1074-7613(03)00117-1. [DOI] [PubMed] [Google Scholar]
- [130].Bigas A, Espinosa L. Hematopoietic stem cells: to be or Notch to be. Blood. 2012;119:3226–3235. doi: 10.1182/blood-2011-10-355826. [DOI] [PubMed] [Google Scholar]
- [131].Burns CE, Traver D, Mayhall E, Shepard JL, Zon LI. Hematopoietic stem cell fate is established by the Notch-Runx pathway. Genes Dev. 2005;19:2331–2342. doi: 10.1101/gad.1337005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [132].Bugeon L, Taylor HB, Progatzky F, Lin MI, Ellis CD, Welsh N, Smith E, Vargesson N, Gray C, Renshaw SA, Chico TJ, Zon LI, Lamb J, Dallman MJ. The NOTCH pathway contributes to cell fate decision in myelopoiesis. Haematologica. 2011;96:1753–1760. doi: 10.3324/haematol.2011.044115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [133].Clements WK, Kim AD, Ong KG, Moore JC, Lawson ND, Traver D. A somiticWnt16/Notch pathway specifies haematopoietic stem cells. Nature. 2011;474:220–224. doi: 10.1038/nature10107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [134].Mukherjee T, Kim WS, Mandal L, Banerjee U. Interaction between Notch and Hif-alpha in development and survival of Drosophila blood cells. Science. 2011;332:1210–1213. doi: 10.1126/science.1199643. [DOI] [PMC free article] [PubMed] [Google Scholar]