Abstract
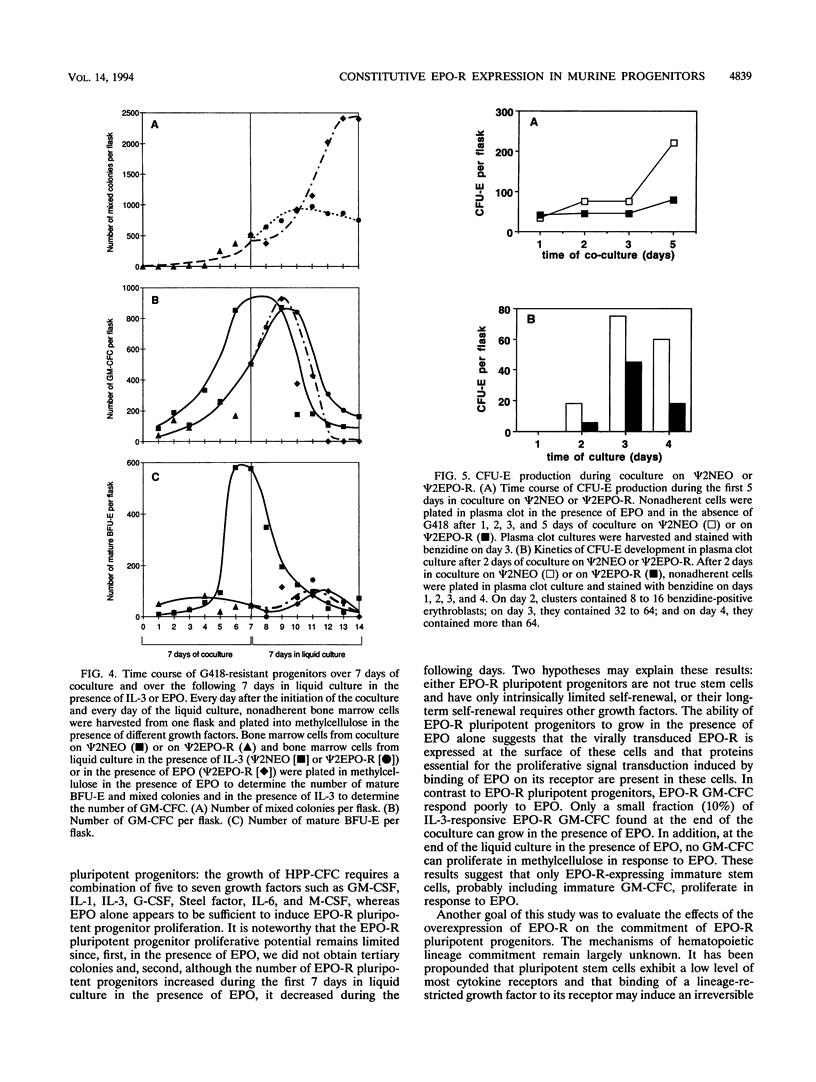
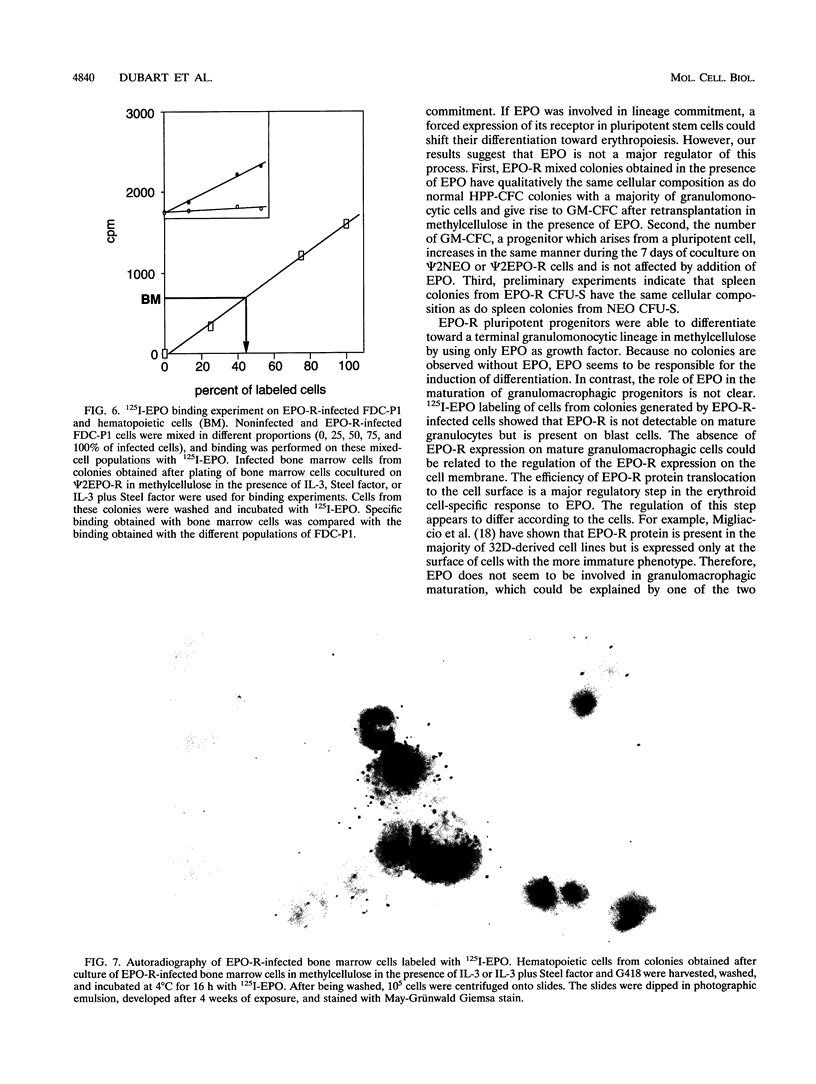
Erythropoietin (EPO) is a prime regulator of the growth and differentiation of erythroid blood cells. The EPO receptor (EPO-R) is expressed in late erythroid progenitors (mature BFU-E and CFU-E), and EPO induces proliferation and differentiation of these cells. By introducing, with a retroviral vector, a normal EPO-R cDNA into murine adult bone marrow cells, we showed that EPO is also able to induce proliferation in pluripotent progenitor cells. After 7 days of coculture with virus-producing cells, bone marrow cells were plated in methylcellulose culture in the presence of EPO, interleukin-3, or Steel factor alone or in combination. In the presence of EPO alone, EPO-R virus-infected bone marrow cells gave rise to mixed colonies comprising erythrocytes, granulocytes, macrophages and megakaryocytes. The addition of interleukin-3 or Steel factor to methylcellulose cultures containing EPO did not significantly modify the number of mixed colonies. The cells which generate these mixed colonies have a high proliferative potential as shown by the size and the ability of the mixed colonies to give rise to secondary colonies. Thus, it appears that EPO has the same effect on EPO-R-expressing multipotent cell proliferation as would a combination of several growth factors. Finally, our results demonstrate that inducing pluripotent progenitor cells to proliferate via the EPO signaling pathway has no major influence on their commitment.
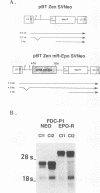
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barber D. L., D'Andrea A. D. The erythropoietin receptor and the molecular basis of signal transduction. Semin Hematol. 1992 Oct;29(4):293–304. [PubMed] [Google Scholar]
- Broudy V. C., Lin N., Brice M., Nakamoto B., Papayannopoulou T. Erythropoietin receptor characteristics on primary human erythroid cells. Blood. 1991 Jun 15;77(12):2583–2590. [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Damen J. E., Liu L., Cutler R. L., Krystal G. Erythropoietin stimulates the tyrosine phosphorylation of Shc and its association with Grb2 and a 145-Kd tyrosine phosphorylated protein. Blood. 1993 Oct 15;82(8):2296–2303. [PubMed] [Google Scholar]
- Dexter T. M., Garland J., Scott D., Scolnick E., Metcalf D. Growth of factor-dependent hemopoietic precursor cell lines. J Exp Med. 1980 Oct 1;152(4):1036–1047. doi: 10.1084/jem.152.4.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson G. R., Gonda T. J., Metcalf D., Hariharan I. K., Cory S. A lethal myeloproliferative syndrome in mice transplanted with bone marrow cells infected with a retrovirus expressing granulocyte-macrophage colony stimulating factor. EMBO J. 1989 Feb;8(2):441–448. doi: 10.1002/j.1460-2075.1989.tb03396.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones S. S., D'Andrea A. D., Haines L. L., Wong G. G. Human erythropoietin receptor: cloning, expression, and biologic characterization. Blood. 1990 Jul 1;76(1):31–35. [PubMed] [Google Scholar]
- Kitamura T., Tange T., Terasawa T., Chiba S., Kuwaki T., Miyagawa K., Piao Y. F., Miyazono K., Urabe A., Takaku F. Establishment and characterization of a unique human cell line that proliferates dependently on GM-CSF, IL-3, or erythropoietin. J Cell Physiol. 1989 Aug;140(2):323–334. doi: 10.1002/jcp.1041400219. [DOI] [PubMed] [Google Scholar]
- Komatsu N., Nakauchi H., Miwa A., Ishihara T., Eguchi M., Moroi M., Okada M., Sato Y., Wada H., Yawata Y. Establishment and characterization of a human leukemic cell line with megakaryocytic features: dependency on granulocyte-macrophage colony-stimulating factor, interleukin 3, or erythropoietin for growth and survival. Cancer Res. 1991 Jan 1;51(1):341–348. [PubMed] [Google Scholar]
- Kuramochi S., Chiba T., Amanuma H., Tojo A., Todokoro K. Growth signal erythropoietin activates the same tyrosine kinases as interleukin 3, but activates only one tyrosine kinase as differentiation signal. Biochem Biophys Res Commun. 1991 Dec 31;181(3):1103–1109. doi: 10.1016/0006-291x(91)92052-l. [DOI] [PubMed] [Google Scholar]
- Kuramochi S., Ikawa Y., Todokoro K. Characterization of murine erythropoietin receptor genes. J Mol Biol. 1990 Dec 5;216(3):567–575. doi: 10.1016/0022-2836(90)90384-x. [DOI] [PubMed] [Google Scholar]
- Lansdorp P. M., Dragowska W., Mayani H. Ontogeny-related changes in proliferative potential of human hematopoietic cells. J Exp Med. 1993 Sep 1;178(3):787–791. doi: 10.1084/jem.178.3.787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longmore G. D., Pharr P., Neumann D., Lodish H. F. Both megakaryocytopoiesis and erythropoiesis are induced in mice infected with a retrovirus expressing an oncogenic erythropoietin receptor. Blood. 1993 Oct 15;82(8):2386–2395. [PubMed] [Google Scholar]
- Mann R., Mulligan R. C., Baltimore D. Construction of a retrovirus packaging mutant and its use to produce helper-free defective retrovirus. Cell. 1983 May;33(1):153–159. doi: 10.1016/0092-8674(83)90344-6. [DOI] [PubMed] [Google Scholar]
- Maouche L., Tournamille C., Hattab C., Boffa G., Cartron J. P., Chrétien S. Cloning of the gene encoding the human erythropoietin receptor. Blood. 1991 Nov 15;78(10):2557–2563. [PubMed] [Google Scholar]
- McNiece I. K., Bradley T. R., Kriegler A. B., Hodgson G. S. Subpopulations of mouse bone marrow high-proliferative-potential colony-forming cells. Exp Hematol. 1986 Oct;14(9):856–860. [PubMed] [Google Scholar]
- Migliaccio A. R., Migliaccio G., D'Andrea A., Baiocchi M., Crotta S., Nicolis S., Ottolenghi S., Adamson J. W. Response to erythropoietin in erythroid subclones of the factor-dependent cell line 32D is determined by translocation of the erythropoietin receptor to the cell surface. Proc Natl Acad Sci U S A. 1991 Dec 15;88(24):11086–11090. doi: 10.1073/pnas.88.24.11086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miura O., D'Andrea A., Kabat D., Ihle J. N. Induction of tyrosine phosphorylation by the erythropoietin receptor correlates with mitogenesis. Mol Cell Biol. 1991 Oct;11(10):4895–4902. doi: 10.1128/mcb.11.10.4895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pharr P. N., Hankins D., Hofbauer A., Lodish H. F., Longmore G. D. Expression of a constitutively active erythropoietin receptor in primary hematopoietic progenitors abrogates erythropoietin dependence and enhances erythroid colony-forming unit, erythroid burst-forming unit, and granulocyte/macrophage progenitor growth. Proc Natl Acad Sci U S A. 1993 Feb 1;90(3):938–942. doi: 10.1073/pnas.90.3.938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quelle D. E., Wojchowski D. M. Localized cytosolic domains of the erythropoietin receptor regulate growth signaling and down-modulate responsiveness to granulocyte-macrophage colony-stimulating factor. Proc Natl Acad Sci U S A. 1991 Jun 1;88(11):4801–4805. doi: 10.1073/pnas.88.11.4801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoemaker C. B., Mitsock L. D. Murine erythropoietin gene: cloning, expression, and human gene homology. Mol Cell Biol. 1986 Mar;6(3):849–858. doi: 10.1128/mcb.6.3.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watowich S. S., Yoshimura A., Longmore G. D., Hilton D. J., Yoshimura Y., Lodish H. F. Homodimerization and constitutive activation of the erythropoietin receptor. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2140–2144. doi: 10.1073/pnas.89.6.2140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witthuhn B. A., Quelle F. W., Silvennoinen O., Yi T., Tang B., Miura O., Ihle J. N. JAK2 associates with the erythropoietin receptor and is tyrosine phosphorylated and activated following stimulation with erythropoietin. Cell. 1993 Jul 30;74(2):227–236. doi: 10.1016/0092-8674(93)90414-l. [DOI] [PubMed] [Google Scholar]
- Worton R. G., McCulloch E. A., Till J. E. Physical separation of hemopoietic stem cells from cells forming colonies in culture. J Cell Physiol. 1969 Oct;74(2):171–182. doi: 10.1002/jcp.1040740209. [DOI] [PubMed] [Google Scholar]
- Ymer S., Tucker W. Q., Sanderson C. J., Hapel A. J., Campbell H. D., Young I. G. Constitutive synthesis of interleukin-3 by leukaemia cell line WEHI-3B is due to retroviral insertion near the gene. Nature. 1985 Sep 19;317(6034):255–258. doi: 10.1038/317255a0. [DOI] [PubMed] [Google Scholar]
- Yoshimura A., D'Andrea A. D., Lodish H. F. Friend spleen focus-forming virus glycoprotein gp55 interacts with the erythropoietin receptor in the endoplasmic reticulum and affects receptor metabolism. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4139–4143. doi: 10.1073/pnas.87.11.4139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssoufian H., Longmore G., Neumann D., Yoshimura A., Lodish H. F. Structure, function, and activation of the erythropoietin receptor. Blood. 1993 May 1;81(9):2223–2236. [PubMed] [Google Scholar]