Abstract
Aluminium (Al) is highly abundant in the environment and can elicit a variety of toxic responses in biological systems. Here we characterize the effects of Al on Caenorhabditis elegans by identifying phenotypic abnormalities and disruption in whole-body metal homeostasis (metallostasis) following Al exposure in food. Widespread changes to the elemental content of adult nematodes were observed when chronically exposed to Al from the first larval stage (L1). Specifically, we saw increased barium, chromium, copper and iron content, and a reduction in calcium levels. Lifespan was decreased in worms exposed to low levels of Al, but unexpectedly increased when the Al concentration reached higher levels (4.8 mM). This bi-phasic phenotype was only observed when Al exposure occurred during development, as lifespan was unaffected by Al exposure during adulthood. Lower levels of Al slowed C. elegans developmental progression, and reduced hermaphrodite self-fertility and adult body size. Significant developmental delay was observed even when Al exposure was restricted to embryogenesis. Similar changes in Al have been noted in association with Al toxicity in humans and other mammals, suggesting that C. elegans may be of use as a model for understanding the mechanisms of Al toxicity in mammalian systems.
Introduction
Aluminium (Al) is the most abundant crustal metal and third most abundant crustal element. Although Al is largely insoluble at neutral pH, its bioavailability increases sharply with decreasing pH.1 The potential for exposure to bioavailable Al is significant owing to its ubiquitous presence in cookware, deodorants, culinary anti-caking agents, etc. It is therefore important to identify the biological effects of Al in order to further understand the risks arising from exposure.1,2 Studies of Al toxicity in humans have suggested a link between Al exposure and some forms of dementia, osteomalacia and microcytic anaemia.3 While the molecular mechanisms underlying these disorders are largely unknown, it has been suggested that Al exposure disrupts the physiological balance of some metals, including changes in iron (Fe) and calcium (Ca) levels.3 This may result from the fact that Al has higher binding affinities compared to the physiological binding partners. For example, the higher binding affinity of Al over magnesium (Mg) for ATP and free phosphate confers the potential for Al to interfere with Mg-catalyzed enzyme activities, resulting in altered metabolism.1 The homeostasis of copper (Cu) is also altered by Al in some biological systems.1 While studying the role of changes in individual metals and linking them to toxic pathology is important, we suggest there is great value in considering the effects of a toxic challenge on the metallome. Previous reports have correlated high levels of Al with altered elemental homeostasis, from animal models through to humans.4-7 It has been hypothesized that elevated Al may be the cause of the disruption of metallostasis, and that this is achieved through changes in the levels of several key elements, namely Cu, sodium (Na), phosphorus (P), Fe and sulphur (S).6,7 Since multiple pathologies arise following Al exposure and it is already known to affect the levels of some metals in a range of species, we hypothesized that Al alters global metal homeostasis.1,3,4,6
C. elegans is widely utilized for the study of toxicity.8-10 In particular, this animal is a powerful model for studies probing the molecular mechanisms of metal toxicology.11-14 Recent publications have shown Al, in the form of AlCl3, Al2O3 nanoparticles and Al2(SO4)3, exert toxic effects on C. elegans lifespan, body size, development, reproduction, locomotion, behavioral plasticity and memory.15-18 It has also been shown that these Al toxicity phenotypes can be transferred to the unexposed next generation.16 These studies were conducted with acute Al toxicity in liquid media. Since C. elegans is a terrestrial nematode and commonly grown on agar in the laboratory, we aimed to establish a toxicity model in standard laboratory growth conditions. Chronic Al exposure is more relevant to the constant exposure of humans to Al in everyday life, and therefore is likely to provide a better model for Al toxicity. Here we show that chronic Al exposure on agar plates produces comparable results to those previously described following acute exposure. We then undertook whole body elemental profile analysis to explore the potential link between these C. elegans Al toxicity phenotypes and metal homeostasis (metallostasis). We found marked alterations in metallostasis, with trends similar to those observed in mammalian systems, suggesting that C. elegans is a relevant model for future mechanistic studies of Al toxicity.
Materials & methods
C. elegans strain maintenance
The C. elegans strains used in this study were wild type Bristol N2, and TJ1060[spe-9(hc88)I; fer-15(b26)II]. They were obtained from the Caenorhabditis Genetics Center (http://www.cbs.umn.edu/CGC/). Unless otherwise stated, worms were grown at 20 °C on nematode growth agar (NGA) plates; 17 g bacto agar, 3 g NaCl, 2.5 g bacto peptone, 25 ml KH2PO4, 1 ml MgSO4, 1 ml CaCl, 1 ml cholesterol, and 925 ml H2O for 1 L NGA.19 C. elegans was fed the common laboratory attenuated E. coli strain OP50.
Al exposure of C. elegans cohorts
The Al exposure plates were set up using 3 ml NGA plates spotted with 100 μl OP50 solution. Aluminium was delivered to the worms by mixing Al(NO3)3·9H2O (unless otherwise stated) with the food source (concentrated E. coli OP50 strain). The latter was made from 1 L of OP50 overnight culture, concentrated by spinning at 11 000 × g for 15 min. This pellet was then re-suspended in 25 ml of S. Basal (20 ml 5 M NaCl, 50 ml 1 M KPO4, 930 ml H2O). The bacteria in the Al food solution (50% concentrated OP50 and 50% Al(NO3)3·9H2O dissolved in H2O) did not appear to be affected by the exposure to Al since they showed continued growth when plated out onto fresh growth plates, although it appears some Al was taken up by the bacteria due to the presence of Al in repeatedly washed bacteria samples (Al exposed); Table S4, ESI. Once the food solution was spotted onto the plates they were left overnight and used the next day, if possible; however, the number of days before use did not affect the results. Aluminium added into the agar rather than the E. coli solution produced similar phenotypes (Table S1, ESI†). It is known that the bioavailability of Al increases with increased acidity.20 Therefore, a pH3 Al solution was used for all Al preparations in order to maximize the bioavailability of Al. Unless otherwise stated H2O (low milli-ohm) at pH3 was used for all controls. The low pH did not result in significant difference from the control for the phenotypes analyzed (developmental progression in the P0 and Fl and fertility) when compared to neutral pH (Fig. SI, ESI†).
The Al solutions (to be added to the food) were all made from a stock solution of 300 mM Al dissolved in sterile H2O (pH3), which was pre-made, and stored at 4 °C until required; all other solutions were made as required. All solutions were inspected for precipitation before use and re-made if any precipitation was found. All Al solutions used were tested with ICP to confirm the concentration of Al. The concentration of Al that the worms were exposed to was calculated as: (Al food concentration × amount of food added to plate)/final plate volume. This method assumes an even distribution of Al throughout the agar. These values were used for comparison purposes. Local concentrations of Al are likely to be higher if Al is taken up by the bacterial food at the surface of the agar. However, we observed that a similar magnitude of effects on the worm were present whether the Al is provided in the bacterial food or the agar (Table S1, ESI†) suggesting that both methods result in similar Al bioavailability. The final plate concentrations of Al used varied; the commonly used concentrations were 0.3 mM Al (lowest concentration to cause significant difference to developmental delay phenotype), 1.9 mM Al (consistently produced approximately 50% adults in the developmental delay assay), and 4.8 mM (consistently produced close to 0% adults in the developmental delay assay).
Four replicates were prepared for each condition. A mass culture of worms was treated with hypochlorite solution (1 ml 5 M KOH, 0.8 ml hypochlorite, 8.2 ml H2O), washed three times with M9 buffer (3 g KH2PO4, 6 g Na2HPO4, 5 g NaCl, 1 ml 1 M MgSO4 in 1 L H2O),19 then transferred to a glass petri-dish containing M9 overnight at room temperature to arrest at the L1 larval stage. Approximately 30 larvae were then added to each control and Al-containing plates, and the plates incubated at 20 °C until moved to experimental temperature or scored as indicated. Where calcium or sodium NO3 salts were used to determine the effect of the NO3 anion, an equimolar concentration of the anion was used for each salt when compared to Al(NO3)3·9H2O.
The M9 solution and control NGA were tested for Al content but the levels were below the detection limit for the volume analyzed (1 ml).
Lifespan analysis
Al exposure plates were set up at 0.03 mM, 1.9 mM and 4.8 mM Al. TJ1060 hermaphrodites were cultured at 25 °C from L1 arrest resulting in sterility. After reaching adulthood all worms were scored for live vs. dead (no movement when touched with platinum wire pick) every two to three days until all worms were dead. The lifespan data were plotted as a survival curve using Graphpad Prism™, and compared using a log rank test implemented in Graphpad Prism™. Replicates were carried out blind, by three independent observers.
Developmental delay experiment
For the P0 experiments (first generation exposed to Al), the exposure plates were kept at 20 °C until the controls were mostly adult (~ 3 days). The number of adults and total number of worms were scored for each plate, and the percentage adult was calculated from these values (adults/total). Replicates were carried out blind, by three independent observers.
In the F1 experiments (generation after Al exposure; P0), both the P0, and F1 worms were exposed to a final Al plate concentration of 1.9 mM; this Al concentration gave a consistent score of ~50% P0 adults. Eggs were picked from P0 worms and transferred to the F1 exposure plates. The F1 exposure plates were incubated at 20 °C for three days (or until controls were adult). The number of adults and the total number of worms of all stages were scored for each plate (percentage adults calculated as above).
For the F2 experiments (generation after F1), the P0 worms were exposed to Al at a final plate concentration of 1.9 mM. F1 eggs were collected after the P0 worms were exposed to Al for three days (from L1) and transferred to non-Al plates. F2 eggs were collected after a further three days and also transferred to non-Al plates. After three days the total number of worms and the number of adults were determined (percentage adults calculated as above).
Fig. S2 (ESI†) presents a diagram of the exposure protocols for all three generational experiments (P0, F1 and F2).
The mean values for all developmental delay experiments, ±standard error of the mean (SEM), were calculated in Microsoft Excel™, and graphed in GraphPad Prism™. Conditions were compared by a two-tailed, one-way ANOVA (two-tailed, two-way for F1) with Tukey’s post-hoc test.
Vulval phenotypic analysis
Worms exposed to Al from 0.03 mM to 4.8 mM were examined for altered vulval morphology. The worms were grown on Al or control plates for 3 days, and then transferred to 50 mg ml−1 sodium azide solution (to induce paralysis to aid photomicroscopy) on a thin 2% agarose pad mounted on a glass microscope slide. Photomicrographs were taken of the 4.8 mM exposed cohort using an Olympus BX51 microscope and a Hamatsu Ocra-ER camera at 4x (Fig. 4A and C and 60x (Fig. 4B and D).
Fig. 4. Al exposure causes protruding vulva (Pvl) and egg-laying defective (Egl) phenotypes.
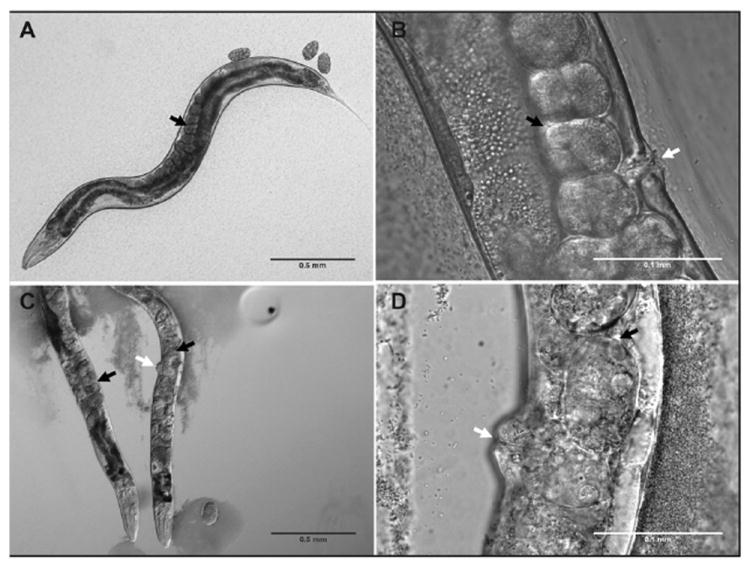
A & B show control worms with normal egg stacking (black arrows) and vulval formation (white arrow). C & D show worms exposed to 4.8 mM Al with egg-laying defective (Egl) & protruding vulva (Pvl) phenotypes; the black arrows indicate locations of eggs showing abnormal stacking and unfertilized oocytes, and the white arrows indicate protruding vulvas. Error bars represent 0.5 mm (grey) and 0.1 mm (white).
Hermaphrodite self-fertility
Exposure plates were set up using 0.03 mM and 0.003 mM Al. Additional control plates of NaNO3 (0.1 mM and 0.01 mM) or Ca(NO3)2 (0.05 mM and 0.005 mM) were also prepared to control for osmotic potential. The worms were then transferred to fresh exposure plates (Al or control) after two days at 20 °C (one worm per plate; ten plates per condition) and then transferred again to new plates every 24 h. The number of eggs laid in each 24 h was scored, and the percentage reaching adulthood along with the length of time taken was recorded. The data were plotted in GraphPad Prism™ as an X & Y graph (± SEM) and Boltzmann sigmoidal curves of non-linear fit. A two-way ANOVA and Tukey’s post-hoc test was used to compare data sets; implemented in GraphPad Prism™.
Pharyngeal pumping assay
Exposure plates were set up using 0.03 mM, 1.9 mM and 4.8 mM Al from L1. Worms were kept at 20 °C for approximately 3.5 days, or until adult, and then scored for the number of pharyngeal pumps per minute, by visual observation using a dissection microscope. A pharyngeal pump is a contraction and near-simultaneous relaxation of the pharyngeal muscles required to suck bacteria into the pharynx. This was repeated on 20 individual worms per condition, n = 20. The averages for each condition (± SEM) were calculated in GraphPad Prism™, and Student’s T-tests were used to determine significant differences between the data sets.
Body length measurement
Aluminium exposure plates were set up using 0.03 mM, 1.9 mM and 4.8 mM Al. Worms were scored for growth stage and length every 6 h by mounting onto slides and capturing a photomicrograph of the entire worm (including the thin tail) using an Olympus BX51 microscope and a Hamatsu Ocra-ER camera at a range of magnifications (40× to 4× where appropriate). The photomicrographs were analyzed at each time-point using a micro-measure to determine the scale for each picture set, and Image J software (NIH; http://rsbweb.nih.gov/ij/docs/faqs.html) was used to quantify length. The results were calculated from measured lengths of individual worms in each of 10 photomicrographs taken from each time point. Image J was used to determine the mid-line length of all worms (Image J score), and the length in mm was calculated in Microsoft Excel™ as follows:
where l; actual worm length (mm), Jl; the length of the worm measured in image J (pixels), Jm; the length of the micro-measure in Image J (pixels), and m; actual length of the micro-measure (mm). The data were plotted in GraphPad Prism™ as a two-category X & Y graph (± SEM) and Boltzmann sigmoidal curves of non-linear fit. A Type 1 one-way ANOVA and Tukey’s post-hoc test were used to analyze data sets and implemented in GraphPad Prism™.
Elemental analysis
Worms used for elemental analysis were washed off plates with M9 buffer without disturbing the bacterial lawn. The worms were pelleted by centrifugation (420 × g for 30 s) and washed three times with M9 buffer over a total of 20 min to promote clearance of gut content. The worm samples were then transferred to pre-weighed, polypropylene tubes and dried overnight in a 60 °C oven. The desiccated pellets were weighed and digested in OmniTrace 70% HNO3 (EMD Chemicals) overnight at 60 °C with 150–200 rpm orbital shaking; 250 μl of acid was used for all samples. Worm acid lysates were diluted to 5% HNO3 with OmniTrace water (EMD Chemicals); the samples were diluted to a final volume of 3.125 ml for analysis. The samples were clarified by centrifugation (3000 × g for 10 min), and introduced using a pneumatic concentric nebulizer with argon as the carrier gas into a Vista Pro inductively coupled plasma—atomic emissions spectrophotometer (ICP-AES; Varian Inc) within 1–2 h of sample preparation as previously described.21 Further dilutions (10× and 100×) of the samples were used, where necessary in order to generate accurate readings of all elements in the sample; final volume for samples tested was kept at 3.125 ml in 5% acid.
The ICP-AES was calibrated using National Institute of Standards and Technology (NIST)-traceable elemental standards and validated using NIST-traceable 1577b bovine liver reference material (BLRM). Elements analyzed were Al, B, Ba, Ca, Cd, Cr, Cu, Fe, K, Mg, Mn, Na, Ni, P, S, Si, Sn, Sr, Ti, Zn, and Zr; wavelengths and detection limits for each element are listed in Supplemental Table 3†. The coefficient of variations (CV) for intra-assay and inter-assay precision for all elements measured with the NIST reference material were typically <10%. Specifically for Al, the detection range was calibrated for 0.05-5 ppm using the 237.312 nm Al emission line; mean intra-assay CV ranged from 1.1% (1 ppm in BLRM, n = 10 in 1 run) to 4.4% (0.1 ppm in BLRM, n = 10 in 1 run); and mean inter-assay percent error of measurement ranged from 0.1 (5 ppm in 10 independent runs) to 9.1% (0.05 ppm in 10 independent runs). Cs (50 ppm) was used for ionization suppression and Y (5 ppm) was used as an internal standard for all samples. All reagents and plastic ware were certified or routinely tested for trace metal work. Elemental content data was summarized using native software (ICP Expert; Varian Inc) and normalized to dry weight of worm sample.
Statistical analysis was conducted using Prism (GraphPad Software, Inc). A two-tailed T-test for two samples with unequal variance was used to analyze the control and treated populations for each element; P values of less than 0.05 were used to determine significance. Elemental values for treated populations were compared to the mean value for the control population to produce the percentage change from control values
used to calculate the mean percentage change from control for the graphical representation of the data (Fig. 6B); absolute values are shown in Table 3. Correlation significance was determined using one- or two-way ANOVA as appropriate, with Tukey’s post-hoc test.
Fig. 6. Al exposure alters whole-worm elemental homeostasis.

(A) Elemental profile of unexposed control worms. Error bars show SEM, and n for each element and condition is shown in Table 3. (B) Changes in elemental composition measured by ICP-AES in Al exposed compared to control worms. * Represents significant difference when compared to control levels (P < 0.05). Error bars show SEM. A list of all elements tested, compared to control, and n for each element is listed in Table 3.
Table 3.
Al exposure alters elemental homeostasisa
| Element | Control
|
0.03 mM
|
1.9 mM
|
4.8 mM
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ave (μg mg-1) | SEM | n | Ave (μg mg-1) | SEM | n | P value/Significance (from control) | Ave (μg mg-1) | SEM | n | P value/Significance (from control) | Ave (μg mg-1) | SEM | n | P value/Significance (from control) | ||||
| Al | 0.0306 | 0.0088 | 6 | 0.1542 | 0.0333 | 4 | 0.0299 | * | 0.6821 | 0.1584 | 4 | 0.0258 | * | 5.1356 | 0.2939 | 4 | 0.0000 | * |
| B | 0.0156 | 0.0021 | 8 | 0.0154 | 0.0022 | 4 | 0.9587 | ns | 0.0145 | 0.0019 | 4 | 0.7074 | ns | 0.0228 | 0.0019 | 4 | 0.0167 | * |
| Ba | 0.0059 | 0.0019 | 6 | 0.0067 | 0.0010 | 4 | 0.7493 | ns | 0.0069 | 0.0016 | 4 | 0.7242 | ns | 0.0471 | 0.0113 | 4 | 0.0060 | * |
| Ca | 6.7276 | 0.4305 | 8 | 6.3070 | 0.3941 | 4 | 0.4895 | ns | 8.3817 | 0.8770 | 4 | 0.1576 | ns | 4.2597 | 0.3107 | 4 | 0.0005 | * |
| Cd | 0.0062 | 0.0008 | 8 | 0.0044 | 0.0013 | 4 | 0.2774 | ns | 0.0042 | 0.0015 | 4 | 0.3093 | ns | 0.0034 | 0.0010 | 2 | 0.0242 | * |
| Cr | 0.0053 | 0.0030 | 8 | 0.0031 | 0.0005 | 4 | 0.5064 | ns | 0.0034 | 0.0007 | 4 | 0.5675 | ns | 0.0233 | 0.0050 | 4 | 0.0055 | * |
| Cu | 0.0135 | 0.0032 | 8 | 0.0076 | 0.0016 | 4 | 0.1274 | ns | 0.0131 | 0.0014 | 4 | 0.9107 | ns | 0.0602 | 0.0158 | 4 | 0.0141 | * |
| Fe | 0.2186 | 0.0653 | 8 | 0.1642 | 0.0123 | 4 | 0.4388 | ns | 0.1560 | 0.0190 | 4 | 0.3840 | ns | 0.4241 | 0.0712 | 4 | 0.0368 | * |
| K | 26.9525 | 2.4043 | 8 | 27.6455 | 2.0446 | 4 | 0.8309 | ns | 27.0088 | 1.8890 | 4 | 0.9857 | ns | 31.5816 | 1.4034 | 4 | 0.1134 | ns |
| Mg | 2.6072 | 0.1926 | 8 | 2.5340 | 0.1606 | 4 | 0.7768 | ns | 2.6094 | 0.2458 | 4 | 0.9946 | ns | 3.3969 | 0.3952 | 4 | 0.0671 | ns |
| Mn | 0.0177 | 0.0024 | 8 | 0.0203 | 0.0036 | 4 | 0.5701 | ns | 0.0224 | 0.0048 | 4 | 0.4178 | ns | 0.0465 | 0.0153 | 4 | 0.0694 | ns |
| Na | 69.5007 | 11.8925 | 8 | 76.8124 | 16.0885 | 4 | 0.7266 | ns | 68.6054 | 13.5515 | 4 | 0.9617 | ns | 100.8975 | 8.4030 | 4 | 0.0435 | * |
| Ni | 0.0291 | 0.0018 | 3 | 0.0222 | 0.0019 | 3 | 0.0564 | ns | 0.0209 | 0.0049 | 4 | 0.1958 | ns | 0.0244 | 0.0047 | 4 | 0.3025 | ns |
| P | 55.0049 | 5.3131 | 8 | 58.8806 | 4.7429 | 4 | 0.5994 | ns | 58.5377 | 4.4789 | 4 | 0.6229 | ns | 74.5452 | 3.9621 | 4 | 0.0094 | * |
| S | 5.2047 | 0.2261 | 8 | 5.0121 | 0.2519 | 4 | 0.5858 | ns | 5.5281 | 0.0948 | 4 | 0.2197 | ns | 6.0105 | 0.2741 | 4 | 0.0267 | * |
| Si | 7.1296 | 1.9178 | 8 | 8.4316 | 2.1393 | 4 | 0.6630 | ns | 10.0223 | 2.3777 | 4 | 0.3756 | ns | 12.6482 | 2.6877 | 4 | 0.0849 | ns |
| Sr | 0.0114 | 0.0020 | 8 | 0.0222 | 0.0015 | 4 | 0.0018 | * | 0.0126 | 0.0015 | 4 | 0.6371 | ns | 0.0160 | 0.0022 | 4 | 0.1178 | ns |
| Zn | 0.0575 | 0.0029 | 8 | 0.0608 | 0.0032 | 4 | 0.4658 | ns | 0.0740 | 0.0204 | 4 | 0.4791 | ns | 0.0483 | 0.0051 | 4 | 0.1033 | ns |
Changes in composition of elements present in control, and Al exposed (0.03 mM, 1.9 mM and 4.8 mM Al) worms as measured by ICP-AES. The average μg of element/mg of dry worm sample mass (Ave), and SEM is shown for each concentration. P Values are shown for each value compared to the control via Student’s T-test, ns represents no significance (P > 0.05), and
represents significant difference (P < 0.05).
Results
Developmental Al exposure alters lifespan
To begin to understand the effect of chronic Al exposure (from L1 until death) on C. elegans populations, we first tested whether Al influenced normal life span. In survival analysis experiments, 0.03 mM Al caused a significant reduction in median lifespan (P < 0.05) when the worms were exposed to Al from the first larval stage (L1) (Fig. 1A & Table 1). However, increasing the Al to 1.9 mM resulted in no significant effect on lifespan (P > 0.05), and the higher Al concentration of 4.8 mM significantly increased the median lifespan (P < 0.0001) compared to untreated controls (Fig. 1A & Table 1). Since worms were exposed at the L1 stage, we considered whether exposure during development was critical for the Al lifespan phenotypes. To test this, we exposed 1 day-old adults to Al and failed to observe alterations in lifespan (Fig. 1B), indicating that Al exposure during development was an important factor in determining the response to Al.
Fig. 1. Developmental Al exposure influences lifespan.
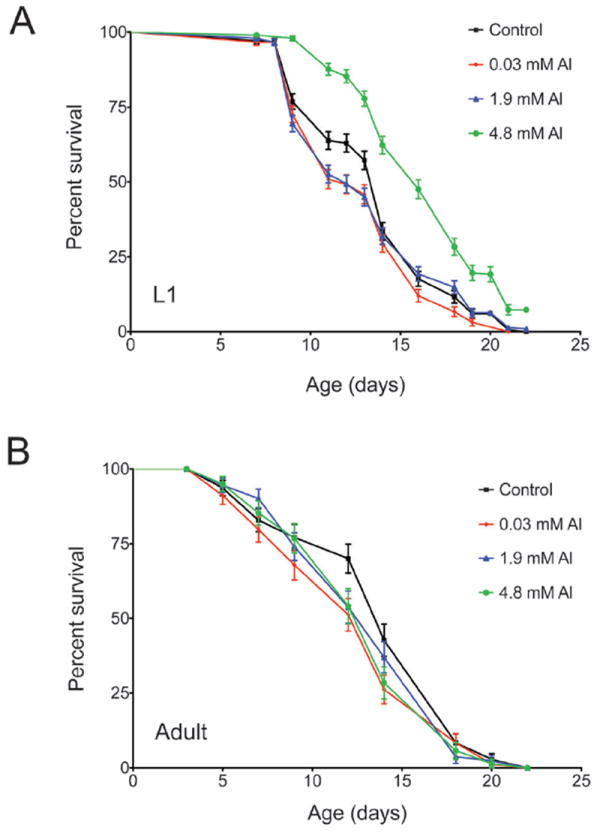
Survival curves of strain TJ1060 at 25 °C during continuous Al exposure. Error bars show SEM; n for each condition is shown in Table 1. (A) Worms were exposed from L1. The survival curves for 0.03 mM and 4.8 mM Al-exposed worms are significantly different from the control, P < 0.05. (B) Worms were exposed from day 1 of adulthood (day 3 on graph). No Al-exposed cohort is significantly different from the control (0 mM Al), P > 0.05.
Table 1.
Effect of Al exposure on wild type C. elegans lifespana
| Mean | Median | n/replicates | P values | ||
|---|---|---|---|---|---|
| L1 | Control | 13 | 14 | 277/10 | — |
| 0.03 mM Al | 12 | 12 | 270/9 | 0.0249 | |
| 1.9 mM Al | 13 | 12 | 291/10 | ns | |
| 4.8 mM Al | 15 | 16 | 293/10 | < 0.0001 | |
| Adult | Control | 13 | 14 | 85/3 | — |
| 0.03 mM Al | 12 | 14 | 85/3 | ns | |
| 1.9 mM Al | 13 | 14 | 85/3 | ns | |
| 4.8 mM Al | 12 | 14 | 71/3 | ns |
Average lifespans (mean and median) for L1 or adult exposed animals; Al exposed and control. The n signifies the total number of worms used, and replicates are shown for the number of times the lifespan was repeated. Comparative analysis was made by the log rank test, and P values are shown in the table, ns = not significant (P < 0.05).
Exposure to Al causes slowed development
Since exposure to Al during development was important for effects in adults, we further characterized the effects of Al in early life. C. elegans were exposed to a range of Al concentrations on NGA plates throughout development from the first larval (L1) stage. An age-synchronous population was generated by starvation arrest of L1 worms. Following Al exposure for 1–2 generations, we monitored the populations over three generations: P0, Fl, and F2 where the P0 is the parental and F1 and F2 are the subsequent generations (Fig. S2, ESI†). Exposure of the P0 generation to Al(NO3)3·9H2O ranging between 0.03 mM and 4.8 mM from L1 caused a significant, dose dependent developmental delay compared to non-Al-exposed control worms (Fig. 2A). Higher concentrations arrested development completely. The in vivo whole body Al concentration as measured by ICP-AES increased significantly (P < 0.05) in response to increased Al concentration in the food (Fig. 2B and Table 3). Control Al levels were 0.03 (±0.008) μg Al/mg dry weight (Table 3). For 0.03 mM Al, the whole body Al value was 0.15 (±0.03) μg Al/mg dry weight (403% higher than control; Fig. 2B), P < 0.03 (Table 3). For 1.9 mM Al, the Al value was 0.68 (±0.16) μg Al/mg dry weight (2126% higher than control; Fig. 2B), P < 0.03 (Table 3). For 4.8 mM Al, the Al value was 5.14 (±0.20) μg Al/mg dry weight (16 663% higher than control; Fig. 2B), P < 0.001 (Table 3). To confirm that the developmental delay was due to the Al cation rather than the nitrate salt (NO3−), we substituted the nitrate salt with chloride as the counterion. There was no difference in the developmentally delayed phenotype when comparing equimolar Al(NO3)3·9H2O to AlCl3, suggesting that the delayed phenotype resulted from exposure to the Al cation (Fig. 2C). Furthermore, developmental delay was not due to non-specific osmotic stress since worms exposed to similar concentrations of NaNO3 did not exhibit developmental delay (Fig. 3A). We also investigated whether the developmental delay could be due to suppression of feeding. However, there was no significant change in feeding behaviour as detected by the pharyngeal pumping rate assay, which is a measure of the rate of feeding (Fig. 3, ESI†).
Fig. 2. Al exposure causes developmental delay.
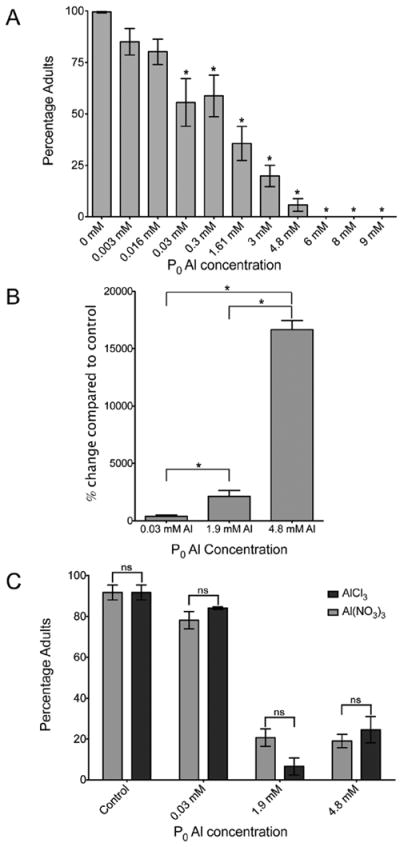
(A) Developmental delay in the P0 (Al-exposed) generation. Bars represent percentage adults after 3 days exposure, the X-axis shows Al(NO3)3·9H2O plate concentrations for the exposures. Asterisk represents significant difference from the control (P < 0.05), and error bars are ±SEM, n = 16 biological repeats for each condition. (B) Changes in Al content of worms: a comparison of the levels of Al by ICP-AES present in Al exposed compared to control worms. The Y-axis represents the percentage change of whole worm Al levels for Al exposed worms at 0.3 mM, 1.9 mM and 4.8 mM Al to control worms. * represents significant difference from the conditions indicated (P < 0.05), and error bars are ±SEM; n = 4, 4 & 6 biological repeats respectively. (C) A comparison of developmental delay caused by Al(NO3)3·9H2O vs. AlCl3—exposures were carried out on the P0 generation and percentage adults was recorded after 3 days. The exposure condition is shown on the X-axis and the Al salt is shown in the graph key, dark grey = AlCl3, light grey = Al(NO3)3·9H2O. There is no significant difference between conditions (ns), and error bars are ±SEM, n = 4 biological repeats.
Fig. 3. Al-induced developmental delay is transferred to F1, but not F2.

(A) Developmental delay in the F1. Bars represent F1 percentage adults (3 days from egg). The P0 and F1 exposure conditions are shown on the X-axis (P0: F1); Al=Al(NO3)3·9H2O and Na=NaNO3. * Represents significant difference (P < 0.05) from the control F1 condition, ** and *** represent significant difference (P < 0.05) from both F1 controls (C and Na). Error bars are ±SEM, n = 20. (B) Developmental delay in the F2—bars represent F2 percentage adults (3 days from egg), the P0 exposure condition is shown on the X-axis (± SEM). The Al exposure is 1.9 mM Al; the F1 and F2 generations were both grown on control plates. There is no significant difference between conditions, n = 4 biological repeats. (C) Multi-generational changes in Al content of worms by ICP-AES: a comparison of the levels of Al present in each generation when only P0 is exposed to Al, at 1.9 mM Al. The Y-axis represents the fold change of Al levels when comparing Al exposed (P0) to control for each generation. * Represents significant difference (P < 0.05). Error bars show ±SEM, n = 4.
Al exposure causes developmental delay in the progeny of exposed animals
We examined the progeny of Al-exposed P0 worms (F1) on control plates lacking Al. The developmental rate of these F1 worms mimicked that of the Al-exposed parent population. The percentage of adults that reached adulthood after 3 days in the P0 Al–F1 control (C) animals was not significantly different from P0 C–F1 Al-treated animals (Fig. 3A). When the progeny of Al-exposed P0 worms (F1) were themselves exposed to Al during development (P0 Al–F1 Al), the developmental delay was significantly enhanced, P < 0.05 (Fig. 3A). This demonstrates that the apparent transfer of toxicity was additive with additional toxic effects observed from repeated (multi-generational) larval exposure. The developmentally delayed phenotype was not transferred to the F2 when, after the P0, subsequent generations were grown on control plates; P0 Al–F1 C–F2 C did not show developmental delay (Fig. 3B). No Al accumulation was seen in either the F1 or the F2 non-exposed generations (Fig. 3C) indicating that the transfer of toxicity from P0 to F1 is either caused by direct embryonic exposure to Al, or indirectly through, for example, alteration of other elements.
Developmental Al exposure causes vulval defects
Aluminium-exposed worms showed significant developmental abnormalities, particularly with respect to specific cell lineage defects. Protruding vulva (Pvl) and egg-laying defective (Egl) phenotypes were observed in approximately ten percent of the population at 0.03 mM Al (data not shown), and in approximately ninety percent of the population at 4.8 mM Al exposure (Fig. 4C and D), when compared to control worms (Fig. 4A and B).
Al exposure reduces fertility
We analyzed the egg-laying ability of worms exposed to Al(NO3)3·9H2O compared to the same concentrations of the control salts NaNO3 and Ca(NO3)2. At 0.03 mM, Al significantly reduced both the total number of eggs, and the age-specific number of eggs laid over the fertile period (Fig. 5A & Table 2). This phenotype was not observed for Na and Ca exposures, or with the unexposed controls.
Fig. 5. Al exposure affects fertility and body size.

(A) Number of eggs laid each 24 h after exposure to given plate concentrations. The curve for 0.03 mM Al exposed worms is significantly different from the control worms (P < 0.05). Error bars represent ±SEM, n = 10 biological repeats. (B) Growth curve: scatter plot with non-linear Boltzmann sigmoidal curves, showing differences in growth following Al exposure at given plate concentrations. The curves for 1.9 mM and 4.8 mM Al are significantly different from the control (P < 0.05). Error bars represent ±SEM, n = 10 worms per condition per time-point. (C) Unexposed adult worm cultured on control plate for 3 days (72 h), (D) Adult worm exposed to 0.03 mM Al from L1 for 72 h, (E) adult worm exposed to 1.9 mM Al from L1 for 72 h, (F) adult worm exposed to 4.8 mM Al from L1 for 72 h. Scale bars represent 0.2 mm.
Table 2.
Effect of Al on total reproductive outputa
| Total Eggs Laid
|
n | P Value | ||
|---|---|---|---|---|
| Ave | SEM | |||
| Control | 262 | 26 | 10 | — |
| Ca 0.15 mM | 269 | 32 | 10 | ns |
| Ca 1.5 mM | 269 | 28 | 10 | ns |
| Na 0.3 mM | 304 | 12 | 10 | ns |
| Na 3 mM | 308 | 14 | 10 | ns |
| Al 0.1 mM | 291 | 7 | 10 | ns |
| Al 1 mM | 150 | 42 | 10 | P < 0.05 |
Comparison of average total number of eggs laid for all conditions tested, ±SEM. P values are shown in the table, ns = not significant (P < 0.05).
Al exposure suppresses growth
The lengths of worms exposed to Al and control worms were measured at intervals from 6 h after reintroduction of food from L1 arrest. Both developmental progression and final body length were inversely related with Al exposure, being significantly reduced at 1.9 mM and 4.8 mM Al (Fig. 5B). Fig. 5C–F shows representative examples of the difference in body size at 72 h. The control (Fig. 5C) and 0.03 mM Al-exposed worms (Fig. 5D) were similar in size, whereas the 1.9 mM Al exposed worms (Fig. 5E) were slightly smaller, and the 4.8 mM Al-exposed worms (Fig. 5F) were very small in comparison.
Al exposure alters systemic elemental composition
Previous toxicology studies in mammalian systems suggest that altered metal homeostasis (metallostasis) is key to producing Al toxic phenotypes.1,3,6 We therefore determined the extent of disruption to in vivo metallostasis following Al exposure in C. elegans. The elemental profile of unexposed adult worms is very tightly regulated, evidenced by the small variability between samples and experiments (Fig. 6A). The elemental profiles of the worms changed significantly (P < 0.05) in response to Al exposure at concentrations of 0.03 mM, 1.9 mM and 4.8 mM (Table 3). The concentrations of most elements analyzed exhibited a statistically significant, dose-dependent change (Fig. 6B); the levels of Al along with other elements (B, Ba, Ca, Cd, Cr, Cu, Fe, Na, P, S, and Sr) altered during Al exposure were significantly changed (P < 0.01) with increasing Al concentration. As expected, the percentage change in Al content when compared to control animals, was significantly higher (P < 0.001) when exposed to greater Al concentrations, resulting in approximately 5-fold increase over control at 0.03 mM Al, 23-fold increase at 1.9 mM Al, and 171-fold increase at 4.8 mM Al; 0.03 μg of element/mg of dry sample (μg mg−1) in control to 0.15 μg mg−1, 0.68 μg mg−1, and 5.14 μg mg−1 respectively in the Al-exposed animals.
Aluminium exposure resulted in dose-dependent alterations in the levels of the essential elements Ca, Cu, Fe, Cr, Na, K, P, and S (Fig. 6B). The worm Ca content for 4.8 mM Al exposure was significantly lower (P < 0.001) than control levels (Table 3) indicating a decrease in Ca levels with increased Al concentration (P < 0.0001) (Fig. 6B). The worm contents for Cu, Fe, P, Cr, Na, K, and S at 4.8 mM Al exposure were all significantly higher (P < 0.05) than the controls (Fig. 6B, Table 3).
We found significantly higher levels (P < 0.05) of B, and Sr, in the 4.8 mM Al exposed samples when compared to the controls (Fig. 6B, Table 3). The levels of Sr followed a bi-phasic pattern, where 1.9 mM Al caused a drop in Sr levels (P < 0.05), but no significant change was seen for 0.03 mM or 4.8 mM Al. This bi-phasic pattern was significant (P < 0.0001). Silicon has been reported to alleviate Al toxicity via increased Si uptake in the freshwater snail L. stagnalis.22 Although there was an apparent trend for increased Si with Al levels (Table 3 and Fig. 6), this was not significant (P = 0.08).
We also found Al-induced changes in the levels of metals which are known to be toxic at low concentrations in biological systems, including Cd, and Ba14,23 (Table 3). Amounts of Cd were significantly lower (P < 0.01) in 4.8 mM Al-exposed worms (0.003 μg mg−1) compared to controls (0.006 μg mg−1; Table 3). The worm content for Ba was significantly higher (P < 0.05) in 4.8 mM Al exposed worms (0.047 μg mg−1) when compared to controls (0.006 μg mg−1); Table 3. No differences were observed (P > 0.05) in levels of Mn, Ni, or Zn (Table 3). The values for other metals, including Sn, Ti, and Zr were below the threshold for detection for at least one independent experimental set, and were therefore excluded from the analysis (data not shown).
The change in elemental profile for worms was not due to the change in bacterial elemental profiles; elemental profiles (percentage change Al exposed compared to controls) for C. elegans were significantly different (P < 0.05) from those of E. coli (Table 4, ESI†).
Discussion
Phenotypes of Al toxicity
Here we have described the phenotypes that result from chronic Al exposure in the nematode C. elegans: growth retardation (also observed in the progeny of exposed worms), developmental delay, reduced fertility, and decreased lifespan. We used standard terrestrial-like solid agar (NGA) media; similar phenotypes have previously been reported following acute Al exposure in liquid media where exposure of L1 C. elegans to AlCl3 and nanoparticulate Al2O3 reduced body length, number of eggs present inside adult hermaphrodites and overall fecundity.17 The preferred method of cultivation for laboratory strains of C. elegans is NGA plates because C. elegans is a terrestrial organism with an optimal brood size observed when the worms are cultured on agar. The liquid culture method is a markedly different environment, but has been previously used for toxicological studies.14,24,25 Solid agar medium, NGA, was used throughout this study so that we could compare the results to the wider C. elegans knowledge base where agar culturing is the norm. The elemental analysis indicated that Al was readily taken up by C. elegans, probably via ingestion since Al was added into the food source. However, some studies suggest that small molecules can be absorbed through the cuticle, and this could also be the case in our experiments, owing to the solubility of Al at pH3.20,26
Wang et al. (2009) suggest that the mechanisms of toxicity associated with Al-induced growth and fecundity phenotypes are similar to those previously reported for Cd toxicity where abnormalities in the pharynx and intestine are observed, resulting in decreased food intake and absorption, and leading to starvation.17,27 However, in the present study, a pharyngeal pumping assay of the Al-exposed nematodes showed no effect of Al on feeding behaviour, and analysis of the levels of internalised Al by ICP-AES showed that considerable Al uptake occurred. Therefore it is unlikely that Al limits food intake of C. elegans. However, the speciation of Al has been shown to profoundly influence toxicity, therefore Al accumulation alone is not an indicator of toxicity.28 Although, due to the combination of Al accumulation and toxicity phenotypes seen we can hypothesise that the phenotypes and elemental changes reported here are due to intracellular Al toxicity.
Early exposure to Al causes problems in development and later life
Aluminium not only causes developmental delay in the exposed generation (P0), but also in their progeny (F1); in those eggs that develop into worms a maternal transferable toxicity phenotype is seen. During exposure of the P0 generation the gonadal tissues and/or oocytes and embryos are exposed to Al; that is, the F1 eggs are exposed to Al whilst developing in utero. This early exposure to Al appears to be enough to cause phenotypic abnormalities in later life since we found no evidence for elevated whole-body Al levels in the F1 progeny when only the P0 was exposed to Al. Similar transferable toxicity phenotypes have been reported for worms exposed to Zn, Ni, lead (Pb) and cobalt (Co)29-32 and it has been suggested that this phenomenon could be due to the build-up of toxic metals inside the developing egg leading to Al toxicity during embryogenesis29,32—a process termed ’chemical legacy’. Maternal transfer of silver (Ag), selenium (Se) and Zn to developing embryos has been reported for the cuttlefish Sepia officinalis, indicating this as a possible mechanism of action.33 In utero Al exposure causes toxic effects in mammals such as rats and mice, suggesting that early-life exposure could also present a risk to humans.34 Our finding that the Al content in the F1 animals is not significantly different from control levels, also adds credence to this theory that early-life exposure to Al results in delayed development. When we compared LI-exposed to adult-exposed worms, we observed a significantly reduced lifespan for the former but not the latter, also suggesting that early-life exposure to Al is more detrimental than adult exposure. There are many reported genetic mutations where lifespan is altered (both increased and decreased) coincidentally with disruption to normal developmental progression.35-39 This is an important observation since all the cell divisions occur in C. elegans during development; none occur during adulthood outside of the germ-line.40 Since the longevity phenotype seen following high Al exposure during L1 was not present when worms were exposed from 1 day-old adult, it is probable that Al triggers, or prevents, an event from occurring during development, producing more stress resistant worms that are longer-lived.
Hormesis in aluminium toxicity
Perhaps surprisingly, we found that Al increased or decreased lifespan depending on the exposure concentration. An extension of lifespan at the higher Al concentration (4.8 mM) could be due to a hormetic effect, where previous or early exposure to a toxicant or stressor results in later-life tolerance.41-45 A similar hormetic effect has been shown for Al toxicity in wheat seedlings, where pre-treatment with H2O2 before Al exposure alleviates Al-induced oxidative stress.46 Survival of early high Al exposure may trigger a hormetic effect, where worms are more resistant to later life Al toxicities. Mitochondrially generated reactive oxygen species (ROS) have been shown to increase survival by triggering the induction of the stress response system, promoting longevity and increased stress resistance.47 If Al induces oxidative stress,48-53 a hormetic mechanism could contribute to survival.
Proposed mechanism(s) of aluminium toxicity
Although the precise mechanism(s) by which Al enters and accumulates within the nematode tissue is still unclear, there are similarities when comparing the phenotypes in this model with those from other organisms reported in the literature. In humans, Al interferes with the binding of Ca to extracellular ligands and extracellular biological surfaces (e.g. the outside of cell membranes), and alters the homeostasis of Cu and Fe.1,3-7 We observed a reduction in Ca levels in worms exposed to Al, which is consistent with the theory derived from humans which proposes that Al displaces Ca due to a higher binding affinity, causing osteomalacia.3 A similar mechanism could contribute to the reduction in body size of C. elegans, via disruption of Ca’s role in cell structure and adhesion, for example, Ca’s role in the structure of the cuticle. We also found an increase in levels of the essential metals Cu and Fe in Al-exposed worms. This is consistent with the altered metallostasis seen in humans and other mammals after Al exposure.1,3-7,54 Cadmium has been shown to decrease in association with elevated Al levels in humans.6 Similarly, we found that Al-exposed worms had lower Cd levels than controls. These findings suggest similar mechanism(s) of Al toxicity to those in mammalian systems.
Aluminium toxicity disrupts metallostasis
We have shown here that Al causes changes to elemental profiles of whole worms. For the higher Al concentration, we found large, significant increases in multiple metals known to cause oxidative stress, e.g. Ba, Fe, Cr, and Cu.23,55,56 In addition, some of the phenotypes we observed following Al exposure are known to also occur as a result of oxidative stress, e.g. accelerated aging,57 and infertility.58 We hypothesize that the accumulation of Al inside the worms causes oxidative stress, thereby inducing the Al-exposure phenotypes.
Yang et al. (1998) reported that chronic exposure to AlCl3 increased Al levels in the mouse brain, together with decreased Cu content and increased Zn levels.4 These authors suggested that the increased Zn levels could be the cause of Al-induced dementia. Yang et al.’s model also suggests that decreased levels of Cu could lead to decreased Cu/Zn superoxide dismutase (SOD), thereby reducing the defense against this oxidative stress. In our model however, although there was an apparent slight reduction in Cu concentrations (at lower Al concentrations of 0.03 mM and 1.9 mM Al) compared to control, this change was not significant. Mustak et al. (2008) analyzed elemental levels in the serum of bipolar mood disorder patients, and found Al and Cu levels to be higher, and Fe and Zn to be lower than in control subjects.7 The authors proposed that elevated Al levels affect metallostasis by causing alterations in the trace element pool, and that this disruption causes an imbalance in elemental concentrations and promotes the production of oxidative stress. Ahmed et al. (2010) analyzed elemental content of serum in Parkinson’s disease patients, and found that elevated levels of Al were correlated with increased levels of the redox active metals, Cu and Mn. They hypothesized that Al alters P homeostasis, causing widespread disruption in metallostasis, leading to the oxidative stress, apoptosis, and neuronal dysfunction associated with the disease. We propose a similar mechanism for chronic Al exposure in worms as we also found an increase in P levels associated with high Al levels. Since Al binds P,1 we propose a mechanism in which increased Al causes more P internalization as a result of Al–P binding, leading to widespread disruption of P homeostasis.
Copper, Fe and Al all increase in mouse brain during the natural course of aging, and may precipitate oxidative stress.4,59-61 This is consistent with the oxidative theory of aging, where increased oxidative damage causes a reduction in lifespan62 and could account for the reduced lifespan seen here at the lower Al concentrations. At high concentrations, B has been shown to cause detrimental effects on fertility in mice, rats and humans.63-67 Since B increased in worms exposed to Al it is possible that the fertility phenotypes seen during Al exposure are due to increased B levels. The increased oxidative stress associated with increased B concentrations could be the cause of this decreased fertility.
Summary
Chronic exposure to aluminium is toxic to C. elegans, inducing changes in growth, development, lifespan and fertility. Aluminium also induces changes in elemental composition of whole worms. It is possible that altered levels of specific elements, both essential and non-essential, may contribute to the phenotypes seen during chronic Al toxicity. We hypothesize that Al causes changes in the levels of key elements in the worm, which in turn leads to oxidative stress and toxic responses. This is relevant to human studies showing similar elemental changes in response to Al. Since the proposed toxicity models have been suggested for other animals, we propose common mechanisms of toxicity from C. elegans to mammalian systems.
Supplementary Material
Acknowledgments
We thank the members of the Lithgow and Gill (Scripps Research Institute, Florida) labs for their intellectual input, Ida Klang, Mark Lucanic, Dipa Bhaumik, David Zucker, and Tai C. Holland for technical support, and the Caenorhabditis Genetics Center for C. elegans strains.
Funding
This work was supported by grants from the Larry L. Hillblom Foundation, and the National Institutes of Health [UL1 DE019608 and R01 AG029631-01A1].
Footnotes
Electronic supplementary information (ESI) available. See DOI: 10.1039/c2mt00146b
Contributor Information
David W. Killilea, Email: dkillilea@chori.org.
Gordon J. Lithgow, Email: glithgow@buckinstitute.org.
References
- 1.Williams RJP. What is wrong with aluminium?: The J.D. Birchall memorial lecture. J Inorg Biochem. 1999;76:81–88. doi: 10.1016/s0162-0134(99)00118-x. [DOI] [PubMed] [Google Scholar]
- 2.Yokel RA, McNamara PJ. Aluminium toxicokinetics: an updated minireview. Pharmacol Toxicol. 2001;88:159–167. doi: 10.1034/j.1600-0773.2001.d01-98.x. [DOI] [PubMed] [Google Scholar]
- 3.Becaria A, Campbell A, Bondy SC. Aluminum as a toxicant. Toxicol Ind Health. 2002;18:309–320. doi: 10.1191/0748233702th157oa. [DOI] [PubMed] [Google Scholar]
- 4.Yang MS. Determination of endogenous trace metal contents in various mouse brain regions after prolonged oral administration of aluminum chloride. J Toxicol Environ Health Part A. 1998;55:445–453. doi: 10.1080/009841098158359. [DOI] [PubMed] [Google Scholar]
- 5.Zhang L, Li X, Gu Q, Zhu Y, Zhao H, Li Y, Zhang Z. Effects of subchronic aluminum exposure on serum concentrations of iron and iron-associated proteins in rats. Biol Trace Elem Res. 2011;141:246–253. doi: 10.1007/s12011-010-8725-y. [DOI] [PubMed] [Google Scholar]
- 6.Ahmed SSSJ, Santosh W. Metallomic profiling and linkage map analysis of early parkinson’s disease: A new insight to aluminum marker for the possible diagnosis. PLoS One. 2010;5:e11252. doi: 10.1371/journal.pone.0011252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mustak MS, Rao TSS, Shanmugavelu P, Sundar NMS, Menon RB, Rao RV, Rao KSJ. Assessment of serummacro and trace element homeostasis and the complexity of inter-element relations in bipolar mood disorders. Clin Chim Acta. 2008;394:47–53. doi: 10.1016/j.cca.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 8.Avila D, Helmcke K, Aschner M. The Caenorhabiditis elegans model as a reliable tool in neurotoxicology. Hum Exp Toxicol. 2010 doi: 10.1177/0960327110392084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Leung MCK, Williams PL, Benedetto A, Au C, Helmcke KJ, Aschner M, Meyer JN. Caenorhabditis elegans: An emerging model in biomedical and environmental toxicology. Toxicol Sci. 2008;106:5–28. doi: 10.1093/toxsci/kfn121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Graves AL, Boyd WA, Williams PL. Using transgenic Caenorhabditis elegans in soil toxicity testing. Arch Environ Contam Toxicol. 2005;48:490–494. doi: 10.1007/s00244-004-0031-2. [DOI] [PubMed] [Google Scholar]
- 11.Au C, Benedetto A, Anderson J, Labrousse A, Erikson K, Ewbank JJ, Aschner M. SMF-1, SMF-2 and SMF-3 DMT1 orthologues regulate and are regulated differentially by manganese level in C. elegans. PLoS One. 2009;4:e7792. doi: 10.1371/journal.pone.0007792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Helmcke KJ, Aschner M. Hormetic effect of methylmercury on Caenorhabditis elegans. Toxicol Appl Pharmacol. 2010;248:156–164. doi: 10.1016/j.taap.2010.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ye B, Rui Q, Wu Q, Wang D. Metallothioneins are required for formation of cross-adaptation response to neurobehavioral toxicity from lead and mercury exposure in nematodes. PLoS One. 2010;5:e14052. doi: 10.1371/journal.pone.0014052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barsyte D, Lovejoy DA, Lithgow GJ. Longevity and heavy metal resistance in daf-2 and age-1 long-lived mutants of Caenorhabditis elegans. FASEB J. 2001;15:627–634. doi: 10.1096/fj.99-0966com. [DOI] [PubMed] [Google Scholar]
- 15.Wang D, Wang Y, Shen L. Confirmation of combinational effects of calcium with other metals in a paper recycling mill effluent on nematode lifespan with toxicity identification evaluation method. J Environ Sci. 2010;22:731–737. doi: 10.1016/s1001-0742(09)60170-4. [DOI] [PubMed] [Google Scholar]
- 16.Wang DY, Yang YC, Wang Y. Aluminium toxicosis causing transferable defects from exposed animals to their progeny in Caenorhabditis elegans. Zhonghua Yu Fang Yi Xue Za Zhi. 2009;43:45–51. [PubMed] [Google Scholar]
- 17.Wang H, Wick RL, Xing B. Toxicity of nanoparticulate and bulk ZnO, Al2O3 and TiO2 to the nematode Caenorhabditis elegans. Environ Pollut. 2009;157:1171–1177. doi: 10.1016/j.envpol.2008.11.004.S0269-7491(08)00590-3 [DOI] [PubMed] [Google Scholar]
- 18.Ye H, Ye B, Wang D. Trace administration of vitamin E can retrieve and prevent UV-irradiation- and metal exposure-induced memory deficits in nematode Caenorhabditis elegans. Neurobiol Learn Mem. 2008;90:10–18. doi: 10.1016/j.nlm.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 19.Hope IA. C elegans A Practical Approach. Oxford University Press; 1999. [Google Scholar]
- 20.Alexopoulos E, McCrohan CR, Powell JJ, Jugdaohsingh R, White KN. Bioavailability and toxicity of freshly neutralised aluminium to the freshwater crayfish Pacifasticus leniusculus. Arch Environ Contam Toxicol. 2003;45:509–514. doi: 10.1007/s00244-003-0228-9. [DOI] [PubMed] [Google Scholar]
- 21.McColl G, Killilea DW, Hubbard AE, Vantipalli MC, Melov S, Lithgow GJ. Pharmacogenetic analysis of lithiuminduced delayed aging in Caenorhabditis elegans. J Biol Chem. 2008;283:350–357. doi: 10.1074/jbc.M705028200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dobranskyte A, Jugdaohsingh R, Stuchlik E, Powell JJ, White KN, McCrohan CR. Role of exogenous and endogenous silicon in ameliorating behavioural responses to aluminium in a freshwater snail. Environ Pollut. 2004;132:427–433. doi: 10.1016/j.envpol.2004.05.023. [DOI] [PubMed] [Google Scholar]
- 23.Wang DY, Wang Y. Phenotypic and behavioral defects caused by barium exposure in nematode Caenorhabditis elegans. Arch Environ Contam Toxicol. 2008;54:447–453. doi: 10.1007/s00244-007-9050-0. [DOI] [PubMed] [Google Scholar]
- 24.Williams PL, Dusenbery DB. Aquatic toxicity testing using the nematode, Caenorhabditis elegans. Environ Toxicol Chem. 1990;9:1285–1290. [Google Scholar]
- 25.Chu KW, Chow KL. Synergistic toxicity of multiple heavy metals is revealed by a biological assay using a nematode and its transgenic derivative. Aquat Toxicol. 2002;61:53–64. doi: 10.1016/s0166-445x(02)00017-6. [DOI] [PubMed] [Google Scholar]
- 26.Page AP, Johnstone IL. In: WormBook. T. C. e. R. Community, WormBook, editor. 2007. [Google Scholar]
- 27.Popham JD, Webster JM. Cadmium toxicity in the free-living nematode, Caenorhabditis elegans. Environ Res. 1979;20:183–191. doi: 10.1016/0013-9351(79)90096-3. [DOI] [PubMed] [Google Scholar]
- 28.Walton RC, McCrohan CR, Livens FR, White KN. Tissue accumulation of aluminium is not a predictor of toxicity in the freshwater snail, Lymnaea stagnalis. Environ Pollut. 2009;157:2142–2146. doi: 10.1016/j.envpol.2009.02.009. [DOI] [PubMed] [Google Scholar]
- 29.Wang D, Shen L, Wang Y. The phenotypic and behavioral defects can be transferred from zinc-exposed nematodes to their progeny. Environ Toxicol Pharmacol. 2007;24:223–230. doi: 10.1016/j.etap.2007.05.009. [DOI] [PubMed] [Google Scholar]
- 30.Wang D, Wang Y. Nickel sulfate induces numerous defects in Caenorhabditis elegans that can also be transferred to progeny. Environ Pollut. 2008;151:585–592. doi: 10.1016/j.envpol.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 31.Wang D-y, Yang P. Multi-biological defects caused by lead exposure exhibit transferable properties from exposed parents to their progeny in Caenorhabditis elegans. J Environ Sci. 2007;19:1367–1372. doi: 10.1016/s1001-0742(07)60223-x. [DOI] [PubMed] [Google Scholar]
- 32.Wang Y, Xie W, Wang D. Transferable properties of multi-biological toxicity caused by cobalt exposure in Caenorhabditis elegans. Environ Toxicol Chem. 2007;26:2405–2412. doi: 10.1897/06-646R1.1. [DOI] [PubMed] [Google Scholar]
- 33.Lacoue-Labarthe T, Warnau M, Oberhänsli F, Teyssié J-L, Jeffree R, Bustamante P. First experiments on the maternal transfer of metals in the cuttlefish Sepia officinalis. Mar Pollut Bull. 2008;57:826–831. doi: 10.1016/j.marpolbul.2008.01.042. [DOI] [PubMed] [Google Scholar]
- 34.Domingo JL. Reproductive and developmental toxicity of aluminum: A review. Neurotoxicol Teratol. 1995;17:515–521. doi: 10.1016/0892-0362(95)00002-9. [DOI] [PubMed] [Google Scholar]
- 35.Dillin A, Hsu A-L, Arantes-Oliveira N, Lehrer-Graiwer J, Hsin H, Fraser AG, Kamath RS, Ahringer J, Kenyon C. Rates of behavior and aging specified by mitochondrial function during development. Science. 2002;298:2398–2401. doi: 10.1126/science.1077780. [DOI] [PubMed] [Google Scholar]
- 36.Fisher AL, Lithgow GJ. The nuclear hormone receptor DAF-12 has opposing effects on Caenorhabditis elegans lifespan and regulates genes repressed in multiple long-lived worms. Aging Cell. 2006;5:127–138. doi: 10.1111/j.1474-9726.2006.00203.x. [DOI] [PubMed] [Google Scholar]
- 37.Harrington LA, Harley CB. Effect of vitamin E on lifespan and reproduction in Caenorhabditis elegans. Mech Ageing Dev. 1988;43:71–78. doi: 10.1016/0047-6374(88)90098-x. [DOI] [PubMed] [Google Scholar]
- 38.Henderson ST, Johnson TE. daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr Biol. 2001;11:1975–1980. doi: 10.1016/s0960-9822(01)00594-2. [DOI] [PubMed] [Google Scholar]
- 39.Johnson TE. Aging can be genetically dissected into component processes using long-lived lines of Caenorhabditis elegans. Proc Natl Acad Sci U S A. 1987;84:3777–3781. doi: 10.1073/pnas.84.11.3777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sulston JE, Horvitz HR. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev Biol. 1977;56:110–156. doi: 10.1016/0012-1606(77)90158-0. [DOI] [PubMed] [Google Scholar]
- 41.Lithgow GJ, White TM, Melov S, Johnson TE. Thermotolerance and extended life-span conferred by single-gene mutations and induced by thermal stress. Proc Natl Acad Sci U S A. 1995;92:7540–7544. doi: 10.1073/pnas.92.16.7540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cypser JR, Johnson TE. Multiple stressors in Caenorhabditis elegans induce stress hormesis and extended longevity. J Gerontol Ser A. 2002;57:B109–B114. doi: 10.1093/gerona/57.3.b109. [DOI] [PubMed] [Google Scholar]
- 43.Cypser JR, Tedesco P, Johnson TE. Hormesis and aging in Caenorhabditis elegans. Exp Gerontol. 2006;41:935–939. doi: 10.1016/j.exger.2006.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Damelin LH, Alexander JJ. Metal-induced hormesis requires cPKC dependent glucose transport and lowered respiration. Hum Exp Toxicol. 2001;20:347–358. doi: 10.1191/096032701680350596. [DOI] [PubMed] [Google Scholar]
- 45.Nascarella MA, Stoffolano JG, Stanek EJ, Kostecki PT, Calabrese EJ. Hormesis and stage specific toxicity induced by cadmium in an insect model, the queen blowfly. Environ Pollut. 2003;124:257–262. doi: 10.1016/s0269-7491(02)00479-7. [DOI] [PubMed] [Google Scholar]
- 46.Xu FJ, Jin CW, Liu WJ, Zhang YS, Lin XY. Pretreatment with H2O2 alleviates aluminum-induced oxidative stress in wheat seedlings. Journal of Integrative Plant Biology. 2011;53:44–53. doi: 10.1111/j.1744-7909.2010.01008.x. [DOI] [PubMed] [Google Scholar]
- 47.Ristow M, Zarse K. How increased oxidative stress promotes longevity and metabolic health: The concept of mitochondrial hormesis (mitohormesis) Exp Gerontol. 2010;45:410–418. doi: 10.1016/j.exger.2010.03.014. [DOI] [PubMed] [Google Scholar]
- 48.Becaria A, Lahiri DK, Bondy SC, Chen D, Hamadeh A, Li H, Taylor R, Campbell A. Aluminum and copper in drinking water enhance inflammatory or oxidative events specifically in the brain. J Neuroimmunol. 2006;176:16–23. doi: 10.1016/j.jneuroim.2006.03.025. [DOI] [PubMed] [Google Scholar]
- 49.Bhalla P, Dhawan D. Protective role of lithium in ameliorating the aluminium-induced oxidative stress and histological changes in rat brain. Cellular and Molecular Neurobiology. 2009 doi: 10.1007/s10571-008-9343-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ding R, Yang Y. Aluminum chloride induced oxidative damage on cells derived from hippocampus and cortex of ICR mice. Brain Res. 2010;1324:96–102. doi: 10.1016/j.brainres.2010.02.024. [DOI] [PubMed] [Google Scholar]
- 51.García T, Esparza JL, Nogués MR, Romeu M, Domingo JL, Gómez M. Oxidative stress status and RNA expression in hippocampus of an animal model of Alzheimer’s disease after chronic exposure to aluminum. Hippocampus. 2010;20:218–225. doi: 10.1002/hipo.20612. [DOI] [PubMed] [Google Scholar]
- 52.Sánchez-Iglesias S, Méndez-Alvarez E, Iglesias-González J, Muñoz-Patiño A, Sánchez-Sellero I, Labandeira-García JL, Soto-Otero R. Brain oxidative stress and selective behaviour of aluminium in specific areas of rat brain: potential effects in a 6-OHDA-induced model of Parkinson’s disease. J Neurochem. 2009;109:879–888. doi: 10.1111/j.1471-4159.2009.06019.x. [DOI] [PubMed] [Google Scholar]
- 53.Sharma D, Sethi P, Hussain E, Singh R. Curcumin counteracts the aluminium-induced ageing-related alterations in oxidative stress, Na+, K+ ATPase and protein kinase C in adult and old rat brain regions. Biogerontology. 2009;10:489–502. doi: 10.1007/s10522-008-9195-x. [DOI] [PubMed] [Google Scholar]
- 54.Li X, Hu C, Zhu Y, Sun H, Li Y, Zhang Z. Effects of aluminum exposure on bone mineral density, mineral, and trace elements in rats. Biol Trace Elem Res. 2011;143:378–385. doi: 10.1007/s12011-010-8861-4. [DOI] [PubMed] [Google Scholar]
- 55.Kasprzak KS. Oxidative DNA and protein damage in metal-induced toxicity and carcinogenesis. Free Radical Biol Med. 2002;32:958–967. doi: 10.1016/s0891-5849(02)00809-2. [DOI] [PubMed] [Google Scholar]
- 56.Xu J, Knutson MD, Carter CS, Leeuwenburgh C. Iron accumulation with age, oxidative stress and functional decline. PLoS One. 2008;3:e2865. doi: 10.1371/journal.pone.0002865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vanfleteren JR. Oxidative stress and ageing in Caenorhabditis elegans. Biochem J. 1993;292:605–608. doi: 10.1042/bj2920605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Doreswamy K, Muralidhara Genotoxic consequences associated with oxidative damage in testis of mice subjected to iron intoxication. Toxicology. 2005;206:169–178. doi: 10.1016/j.tox.2004.07.010. [DOI] [PubMed] [Google Scholar]
- 59.Stohs SJ, Bagchi D. Oxidative mechanisms in the toxicity of metal ions. Free Radical Biol Med. 1995;18:321–336. doi: 10.1016/0891-5849(94)00159-h. [DOI] [PubMed] [Google Scholar]
- 60.Tripathi S, Mahdi AA, Nawab A, Chander R, Hasan M, Siddiqui MS, Mahdi F, Mitra K, Bajpai VK. Influence of age on aluminum induced lipid peroxidation and neurolipofuscin in frontal cortex of rat brain: A behavioral, biochemical and ultrastructural study. Brain Res. 2009;1253:107–116. doi: 10.1016/j.brainres.2008.11.060. [DOI] [PubMed] [Google Scholar]
- 61.Donahue AN, Aschner M, Lash LH, Syversen T, Sonntag WE. Growth hormone administration to aged animals reduces disulfide glutathione levels in hippocampus. Mech Ageing Dev. 2006;127:57–63. doi: 10.1016/j.mad.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 62.Lithgow GJ, Walker GA. Stress resistance as a determinate of C. elegans lifespan. Mech Ageing Dev. 2002;123:765–771. doi: 10.1016/s0047-6374(01)00422-5. [DOI] [PubMed] [Google Scholar]
- 63.Trumbo P, Yates AA, Schlicker S, Poos M. Dietary reference intakes: Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. J Am Diet Assoc. 2001;101:294–301. doi: 10.1016/S0002-8223(01)00078-5. [DOI] [PubMed] [Google Scholar]
- 64.Turkez H, Geyikoglu F, Tatar A, Keles MS, Kaplan I. The effects of some boron compounds against heavy metal toxicity in human blood. Exp Toxicol Pathol. 2012;64(1-2):93–101. doi: 10.1016/j.etp.2010.06.011. [DOI] [PubMed] [Google Scholar]
- 65.Treinen KA, Chapin RE. Development of testicular lesions in F344 rats after treatment with boric acid. Toxicol Appl Pharmacol. 1991;107:325–335. doi: 10.1016/0041-008x(91)90212-w. [DOI] [PubMed] [Google Scholar]
- 66.Ku WW, Chapin RE, Wine RN, Gladen BC. Testicular toxicity of boric acid (BA): Relationship of dose to lesion development and recovery in the F344 rat. Reprod Toxicol. 1993;7:305–319. doi: 10.1016/0890-6238(93)90020-8. [DOI] [PubMed] [Google Scholar]
- 67.Fail PA, George JD, Seely JC, Grizzle TB, Heindel JJ. Reproductive toxicity of boric acid in Swiss (CD-1) mice: Assessment using the continuous breeding protocol. Fundam Appl Toxicol. 1991;17:225–239. doi: 10.1016/0272-0590(91)90215-p. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


