Abstract
Caspase-1-mediated detection of pathogens is a potent arm of the innate immune system. La Rock and Cookson (2012) show that the Yersinia Type III secretion effector, YopM, directly inhibits Caspase-1.
The innate immune system detects pathogens through cytosolic inflammasomes, including the Nod-like receptors NLRP3, NLRC4, and NLRP1b. These signaling platforms recruit the adaptor protein ASC into a single focus. The ASC focus subsequently recruits the enzyme Caspase-1, which auto-processes and promotes the secretion of the pro-inflammatory cytokines IL-1β and IL-18 that initiate immune responses. Underscoring the importance of Caspase-1 in the innate immune response, mice deficient in Caspase-1 fail to control many pathogenic infections. Many viruses have developed direct mechanisms to suppress either Caspase-1 or IL-1β, delaying the onset of the innate immune response. For example, poxviruses encode direct inhibitors of Caspase-1, such as CrmA, as wells as analogs of the mammalian pyrin-only proteins that likely prevent ASC oligomerization (Taxman et al., 2010). Further, Kaposi’s sarcoma-associated herpesvirus (KSHV) encodes a Nod-like receptor (NLR) analog that inhibits inflammasome oligomerization (Gregory et al., 2011). Macrophages detect Salmonella typhimurium though the NLRC4 inflammasome when the S. typhimurium T3SS inadvertently translocates flagellin or T3SS rod proteins into the host cell cytosol. S. typhimurium indirectly evade this detection by repressing flagellin expression and using an alternate NLRC4-evading T3SS rod proteins (Miao and Rajan, 2011). However, bacterial virulence proteins that directly inhibit inflammasome pathways have not been identified. Yersinia drastically modulates the host environment and suppresses immune signaling by delivering various effectors (Yops) into the host cytosol via its type III secretion system (T3SS). In this issue of Cell Host and Microbe, La Rock and Cookson elegantly demonstrate that the Yersinia T3SS effector YopM directly inhibits Caspase-1, and that this inhibition is essential for Yersinia pseudotuberculosis virulence in vivo (La Rock, 2012).
In their report, wild type Y. pseudotuberculosis failed to activate Caspase-1, whereas Caspase-1 was robustly activated in response to a strain deficient in all known T3SS effector Yop genes (YersiniaΔ lacking YopJOEHKM). Remarkably, La Rock and Cookson found that evasion of Caspase-1 was entirely attributed to yopM. This was proven using a strain only deficient in yopM, complementing ΔyopJOEHKM with plasmid borne yopM, and retrovirally transducing macrophages with yopM in the absence of bacterial infection. It has long been known that the ΔyopM mutant is highly attenuated, although its function in vivo has remained elusive. La Rock and Cookson nail down the importance of YopM-mediated Caspase-1 inhibition by demonstrating that ΔyopM becomes fully virulent in Caspase-1 deficient mice (La Rock, 2012).
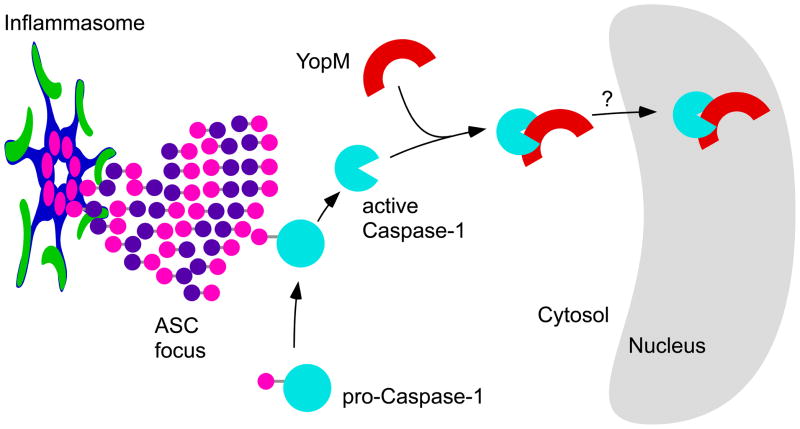
Significantly, La Rock and Cookson provide a mechanism by which a bacterial virulence factor inhibits Caspase-1 activity (Figure 1). Several viruses inhibit Caspase-1 by expressing proteins that mimic the Caspase-1 binding and cleavage motif and thus act as pseudo-substrates that block the Caspase-1 catalytic site. By aligning YopM to known Caspase-1 substrates and inhibitors, the authors identified a Caspase-1-binding motif. The critical aspartic acid residue within this motif was required for direct YopM binding to activated Caspase-1. Interestingly, the YopM C-terminus directs nuclear localization, and this domain is distinct from the Caspase-1-binding site (Benabdillah et al., 2004). La Rock and Cookson’s data suggest an intriguing model in which YopM carts Caspase-1 into the nucleus, making it unavailable for recruitment to the cytosolic ASC focus. Indeed, the NLRP3-ASC focus still forms, yet YopM prevents Caspase-1 accumulation in this focus.
Figure 1. YopM inhibits Caspase-1.
Yersinia T3SS activity is detected by cytosolic inflammasomes by unknown mechanisms. Oligomerized NLR inflammasomes recruit the adaptor protein ASC through homotypic interactions between CARD (pink) or pyrin (purple) domains. ASC is composed of a CARD and a pyrin domain, and subsequently collects the entire complement of cellular ASC into a single focus. The ASC-CARD domains then recruit pro-Caspase-1 via it’s CARD domain, resulting in its auto-proteolytic processing and activation. YopM contains a pseudo-substrate binding site that binds and retains activated Caspase-1. Because YopM contains a nuclear localization signal, sequestration of activated Caspase-1 in the nucleus may be a key mechanism by which YopM separates Caspase-1 from it’s intended targets.
Several groups previously investigated inflammasome detection of Y. enterocolitica, Y. pseudotuberculosis, and Y. pestis, focusing on YopE, YopJ, and YopK. These reports must now be re-interpreted in light of YopM suppression. It is worth noting that the use of different cell lines (primary versus immortalized), macrophage priming conditions, and Yersinia strains may explain the differences in the relative contribution of different Yops in suppressing Caspase-1 activation. For example, Schotte et al. reported that Y. enterocolitica ΔyopHOPEMT triggered IL-1β secretion, and suggested that YopE, but not YopM, was the critical effector blocking this response (Schotte et al., 2004). However, they only examined a single macrophage cell line in the absence of priming, with complementary studies in Caspase-1 over-expressing cell lines. Does Caspase-1 activation in these cell lines accurately reflect detection pathways occurring in primary macrophages and in vivo?
Later, using a plasmid-cured Y. pseudotuberculosis strain containing the Y. pestis virulence plasmid deleted for all known Yop effectors (yopM, yopT, yopK, ylpA, yopE, yadA, ypkA, yopJ, yopH) and several other open reading frames, YopK was implicated in decreasing Caspase-1 activation and IL-1β secretion (Brodsky et al., 2010). YopK interacts with the YopBD translocon, and in the absence of all known Yops, adding back YopK results in decreased Caspase-1 activation. Pore forming toxins are detected through NLRP3. Thus in the absence of YopK, does the YopBD translocon pore become dysregulated, permitting potassium efflux from the cell and subsequent NLRP3 activation? Interestingly, yopK mutants have an increased rate of translocation of other effectors, including YopM (Dewoody et al., 2011). Are increased amounts of T3SS rod protein and flagellin aberrantly injected into the cell to be detected though NLRC4? Perhaps in the absence of YopK, rapid detection kinetics though multiple pathways overcome the ability of YopM to block Caspase-1.
In stark contrast to YopM/YopK suppressing inflammasome activation, YopJ (a MAPK and IKKβ inhibitor also known as YopP in Y. enterocolitica) induces NLRP3-dependent Caspase-1 activation and IL-1β secretion in the absence of priming (Viboud and Bliska, 2005). Interestingly, YopJ/P isoforms that are more catalytically active are more readily detected, with IL-1β secretion at 8–24 hours post infection (Zheng et al., 2012). This is quite delayed compared to typical inflammasome agonists such as S. typhimurium, ATP, or Alum. Are these delayed kinetics due in part to YopM? To fully understand the ability of inflammasomes to detect the catalytic activity of YopJ, experiments comparing wild type, ΔyopM, ΔyopJ, and ΔyopMJ in primed as well as unprimed macrophages would be informative.
Although La Rock and Cookson show that Caspase-1 inhibition is the primary function of YopM, it may have other functions. YopM is polymorphic, and the number of leucine-rich repeats, which mediate protein-protein interactions, varies between strains. One could imagine that by adding more leucine-rich repeats, a greater surface area for binding host proteins could be created. YopM binds two host serine/threonine kinases (RSK1 and PKN2) {Viboud, 2005 #33}. Does YopM bind Caspase-1 and RSK1/PKN2 at the same time and sequester all three in the nucleus? Thus, it is possible that YopM plays different functions in different Yersinia species and/or targeted cell types.
In summary, La Rock and Cookson provide convincing data that the T3SS effector YopM promotes Yersinia virulence by inhibiting Caspase-1 activation, presumably permitting early replication in the absence of an inflammatory response. This story exemplifies the complex interplay between innate immune responses and bacterial evasion strategies. Whereas delivery of effectors by the T3SS into the host cytosol is a highly efficient mechanism by which pathogens modify the host environment to its advantage, it also provides a smorgasbord of bacterial ligands and cellular perturbations that can be detected by cytosolic sensors. In Yersinia, YopM counters this inherent weakness in the system.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Benabdillah R, Mota LJ, Lutzelschwab S, Demoinet E, Cornelis GR. Identification of a nuclear targeting signal in YopM from Yersinia spp. Microb Pathog. 2004;36:247–261. doi: 10.1016/j.micpath.2003.12.006. [DOI] [PubMed] [Google Scholar]
- Brodsky IE, Palm NW, Sadanand S, Ryndak MB, Sutterwala FS, Flavell RA, Bliska JB, Medzhitov R. A Yersinia effector protein promotes virulence by preventing inflammasome recognition of the type III secretion system. Cell Host Microbe. 2010;7:376–387. doi: 10.1016/j.chom.2010.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewoody R, Merritt PM, Houppert AS, Marketon MM. YopK regulates the Yersinia pestis type III secretion system from within host cells. Mol Microbiol. 2011;79:1445–1461. doi: 10.1111/j.1365-2958.2011.07534.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gregory SM, Davis BK, West JA, Taxman DJ, Matsuzawa S, Reed JC, Ting JP, Damania B. Discovery of a viral NLR homolog that inhibits the inflammasome. Science. 2011;331:330–334. doi: 10.1126/science.1199478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Rock CN, Cookson BT. A bacterial virulence protein arrests inflammasome assembly and processing. Cell Host Microbe. 2012 doi: 10.1016/j.chom.2012.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao EA, Rajan JV. Salmonella and Caspase-1: A complex Interplay of Detection and Evasion. Front Microbiol. 2011;2:85. doi: 10.3389/fmicb.2011.00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schotte P, Denecker G, Van Den Broeke A, Vandenabeele P, Cornelis GR, Beyaert R. Targeting Rac1 by the Yersinia effector protein YopE inhibits caspase-1-mediated maturation and release of interleukin-1beta. J Biol Chem. 2004;279:25134–25142. doi: 10.1074/jbc.M401245200. [DOI] [PubMed] [Google Scholar]
- Taxman DJ, Huang MT, Ting JP. Inflammasome inhibition as a pathogenic stealth mechanism. Cell Host Microbe. 2010;8:7–11. doi: 10.1016/j.chom.2010.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viboud GI, Bliska JB. Yersinia outer proteins: role in modulation of host cell signaling responses and pathogenesis. Annu Rev Microbiol. 2005;59:69–89. doi: 10.1146/annurev.micro.59.030804.121320. [DOI] [PubMed] [Google Scholar]
- Zheng Y, Lilo S, Mena P, Bliska JB. YopJ-induced caspase-1 activation in Yersinia-infected macrophages: independent of apoptosis, linked to necrosis, dispensable for innate host defense. PLoS One. 2012;7:e36019. doi: 10.1371/journal.pone.0036019. [DOI] [PMC free article] [PubMed] [Google Scholar]