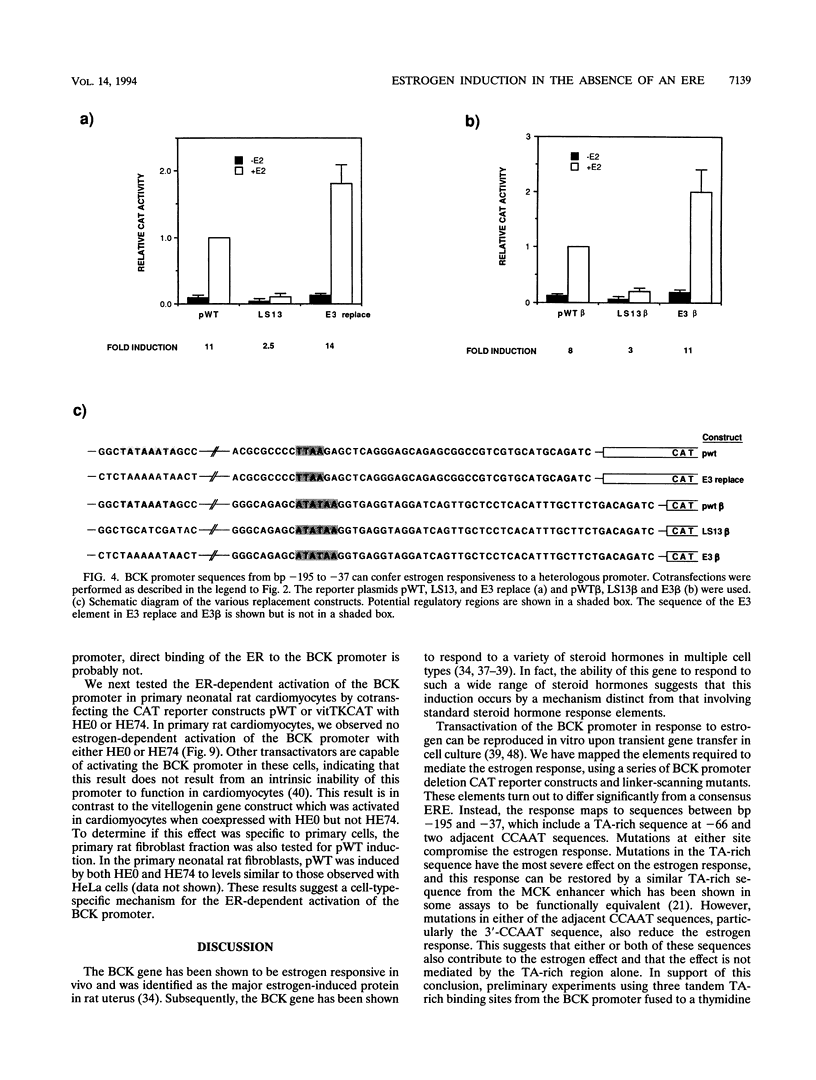
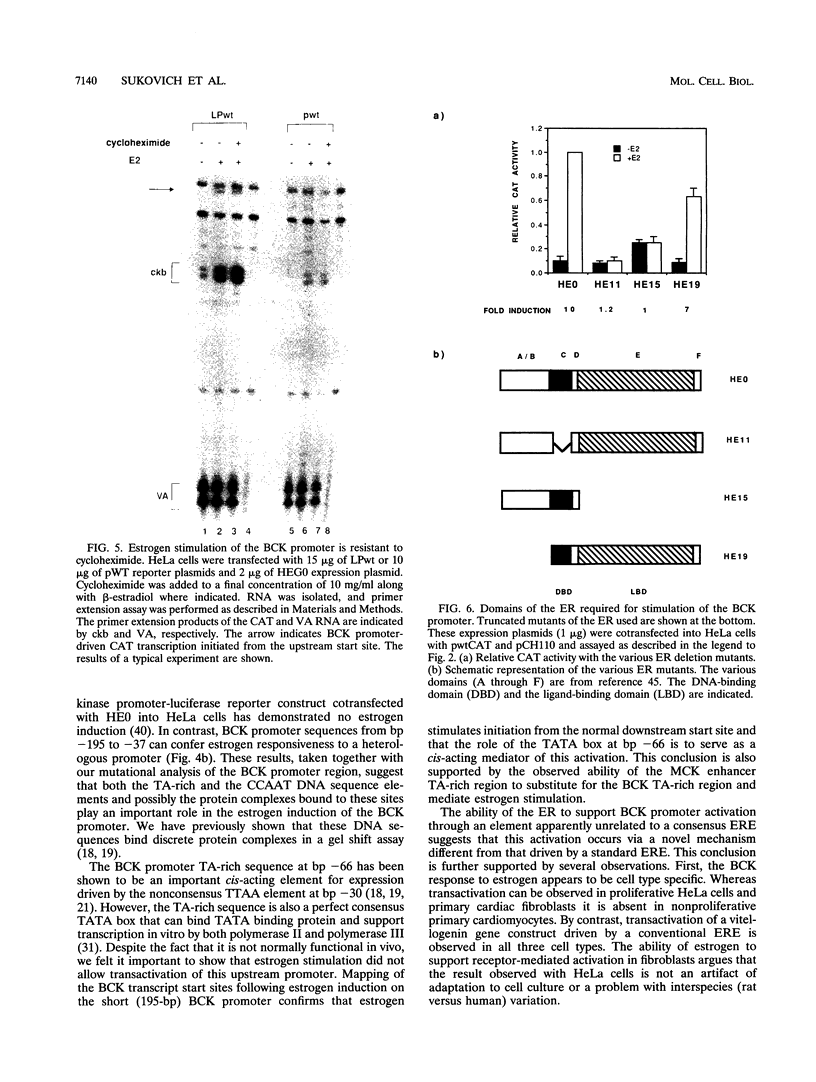
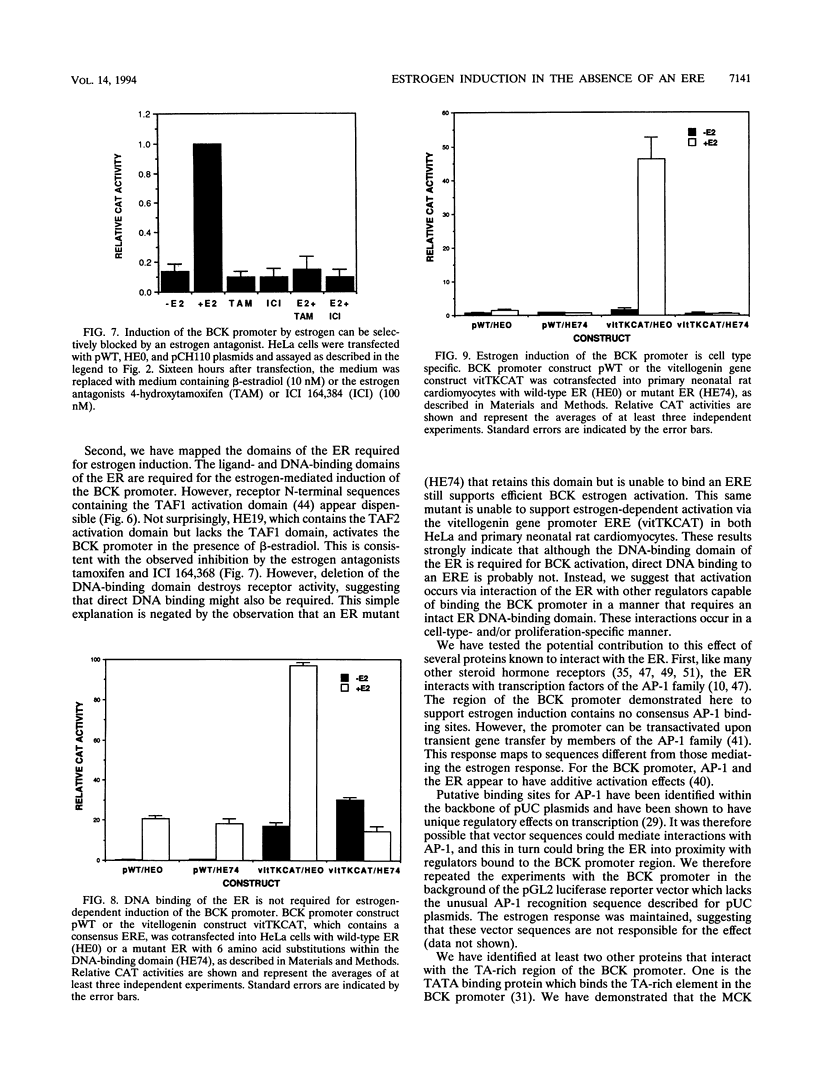
Abstract
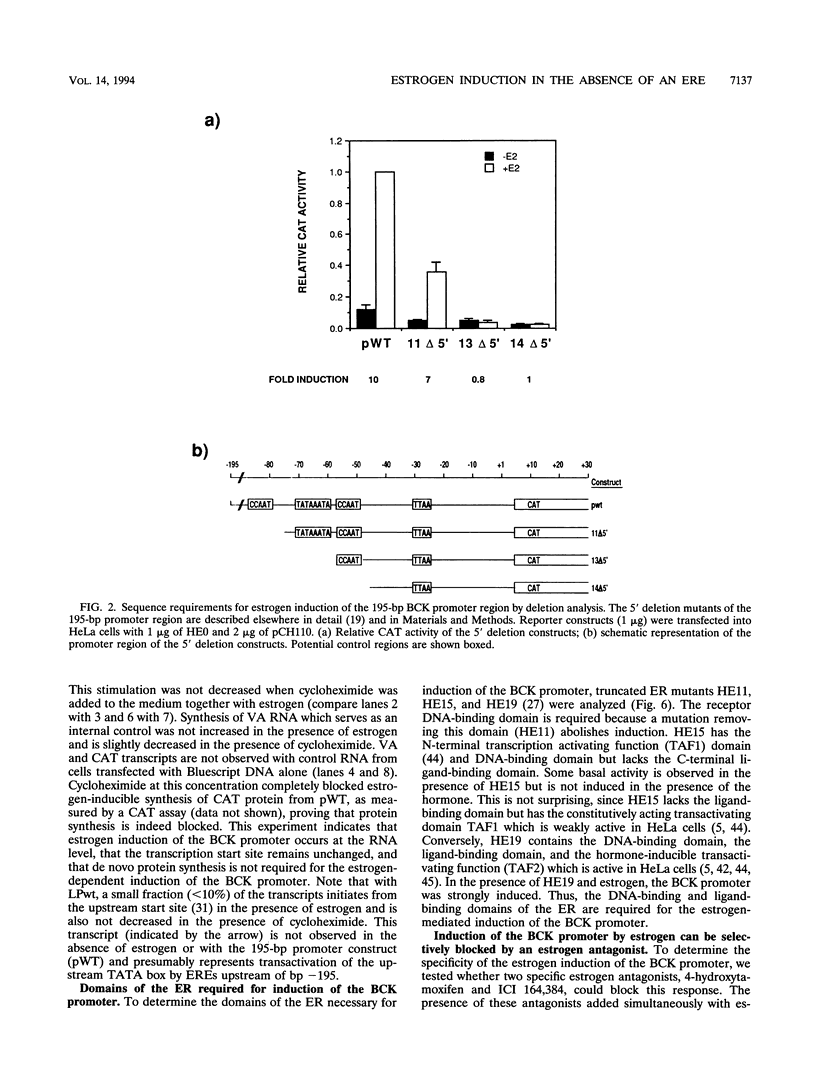
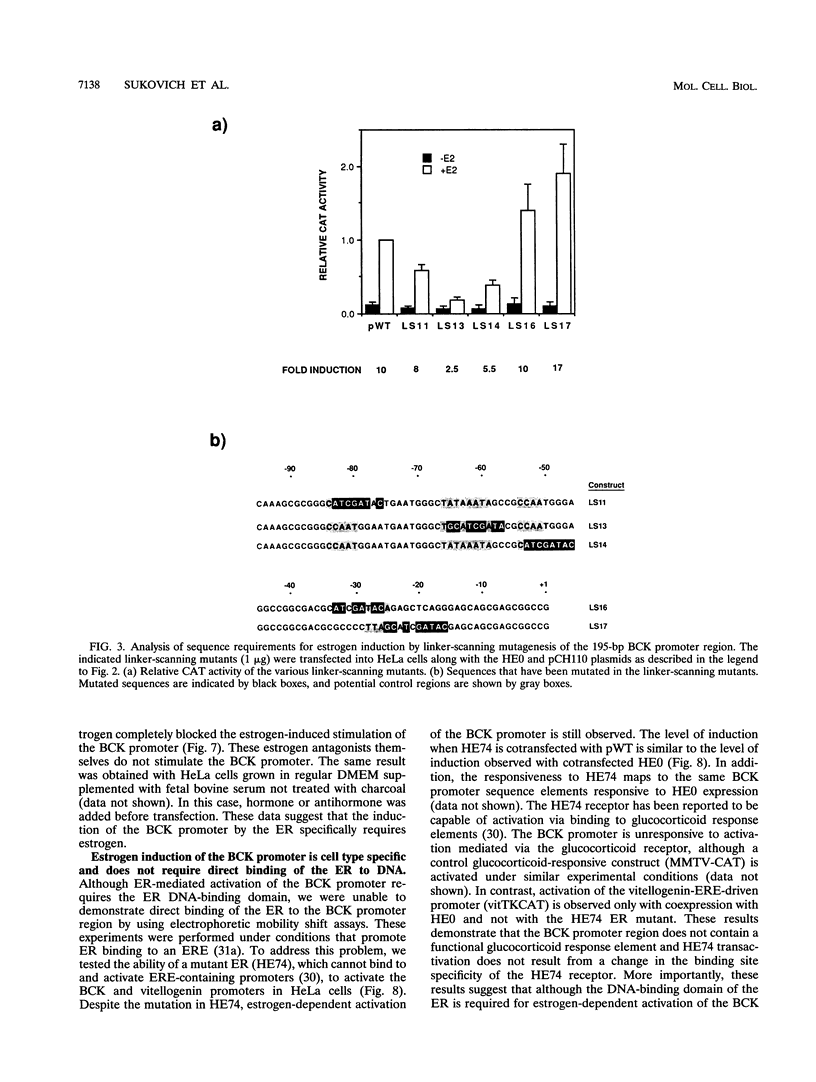
The estrogen receptor (ER) typically activates gene transcription by binding to estrogen-responsive elements (EREs). The brain creatine kinase (BCK) promoter is responsive to estrogen but contains no ERE-related sequence. To investigate the mechanism of estrogen induction, we have introduced the estrogen receptor into HeLa cells and primary rat cardiomyocytes and fibroblasts along with 195 bp of BCK promoter linked to a chloramphenicol acetyltransferase (CAT) reporter gene. A 10-fold stimulation of CAT activity was observed in the presence of beta-estradiol in both HeLa and rat primary fibroblasts, but no induction was observed in primary rat cardiomyocytes. In contrast, a control vitellogenin gene construct which contains a typical ERE was induced in an ER-dependent manner in all cell types studied. Estrogen induction in HeLa was not sensitive to cycloheximide and was blocked by the ER antagonists tamoxifen and ICI 164,384. Analysis of 5' deletion and linker-scanning mutations indicates sequences between bp -45 and -75 including a TA-rich sequence and a CCAAT sequence to be crucial for stimulation of the BCK promoter by the ER. BCK estrogen induction is dependent on the DNA-binding domain and transactivation domain TAF2 of the ER. However, direct DNA binding is probably not required. Taken together, these results suggest a novel mechanism for ER-mediated gene activation. This mechanism is consensus ERE independent and cell type specific and requires interactions between the ER and molecules capable of interacting with the BCK promoter TA-rich region.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beato M. Gene regulation by steroid hormones. Cell. 1989 Feb 10;56(3):335–344. doi: 10.1016/0092-8674(89)90237-7. [DOI] [PubMed] [Google Scholar]
- Benfield P. A., Graf D., Korolkoff P. N., Hobson G., Pearson M. L. Isolation of four rat creatine kinase genes and identification of multiple potential promoter sequences within the rat brain creatine kinase promoter region. Gene. 1988 Mar 31;63(2):227–243. doi: 10.1016/0378-1119(88)90527-6. [DOI] [PubMed] [Google Scholar]
- Berry M., Metzger D., Chambon P. Role of the two activating domains of the oestrogen receptor in the cell-type and promoter-context dependent agonistic activity of the anti-oestrogen 4-hydroxytamoxifen. EMBO J. 1990 Sep;9(9):2811–2818. doi: 10.1002/j.1460-2075.1990.tb07469.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berthois Y., Katzenellenbogen J. A., Katzenellenbogen B. S. Phenol red in tissue culture media is a weak estrogen: implications concerning the study of estrogen-responsive cells in culture. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2496–2500. doi: 10.1073/pnas.83.8.2496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Cserjesi P., Olson E. N. Myogenin induces the myocyte-specific enhancer binding factor MEF-2 independently of other muscle-specific gene products. Mol Cell Biol. 1991 Oct;11(10):4854–4862. doi: 10.1128/mcb.11.10.4854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eustice D. C., Feldman P. A., Colberg-Poley A. M., Buckery R. M., Neubauer R. H. A sensitive method for the detection of beta-galactosidase in transfected mammalian cells. Biotechniques. 1991 Dec;11(6):739-40, 742-3. [PubMed] [Google Scholar]
- Gaub M. P., Bellard M., Scheuer I., Chambon P., Sassone-Corsi P. Activation of the ovalbumin gene by the estrogen receptor involves the fos-jun complex. Cell. 1990 Dec 21;63(6):1267–1276. doi: 10.1016/0092-8674(90)90422-b. [DOI] [PubMed] [Google Scholar]
- Gauthier-Rouvière C., Basset M., Blanchard J. M., Cavadore J. C., Fernandez A., Lamb N. J. Casein kinase II induces c-fos expression via the serum response element pathway and p67SRF phosphorylation in living fibroblasts. EMBO J. 1991 Oct;10(10):2921–2930. doi: 10.1002/j.1460-2075.1991.tb07842.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorman C. M., Moffat L. F., Howard B. H. Recombinant genomes which express chloramphenicol acetyltransferase in mammalian cells. Mol Cell Biol. 1982 Sep;2(9):1044–1051. doi: 10.1128/mcb.2.9.1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gossett L. A., Kelvin D. J., Sternberg E. A., Olson E. N. A new myocyte-specific enhancer-binding factor that recognizes a conserved element associated with multiple muscle-specific genes. Mol Cell Biol. 1989 Nov;9(11):5022–5033. doi: 10.1128/mcb.9.11.5022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green S., Chambon P. Nuclear receptors enhance our understanding of transcription regulation. Trends Genet. 1988 Nov;4(11):309–314. doi: 10.1016/0168-9525(88)90108-4. [DOI] [PubMed] [Google Scholar]
- Herbomel P., Bourachot B., Yaniv M. Two distinct enhancers with different cell specificities coexist in the regulatory region of polyoma. Cell. 1984 Dec;39(3 Pt 2):653–662. doi: 10.1016/0092-8674(84)90472-0. [DOI] [PubMed] [Google Scholar]
- Hobson G. M., Mitchell M. T., Molloy G. R., Pearson M. L., Benfield P. A. Identification of a novel TA-rich DNA binding protein that recognizes a TATA sequence within the brain creatine kinase promoter. Nucleic Acids Res. 1988 Sep 26;16(18):8925–8944. doi: 10.1093/nar/16.18.8925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hobson G. M., Molloy G. R., Benfield P. A. Identification of cis-acting regulatory elements in the promoter region of the rat brain creatine kinase gene. Mol Cell Biol. 1990 Dec;10(12):6533–6543. doi: 10.1128/mcb.10.12.6533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horlick R. A., Benfield P. A. The upstream muscle-specific enhancer of the rat muscle creatine kinase gene is composed of multiple elements. Mol Cell Biol. 1989 Jun;9(6):2396–2413. doi: 10.1128/mcb.9.6.2396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horlick R. A., Hobson G. M., Patterson J. H., Mitchell M. T., Benfield P. A. Brain and muscle creatine kinase genes contain common TA-rich recognition protein-binding regulatory elements. Mol Cell Biol. 1990 Sep;10(9):4826–4836. doi: 10.1128/mcb.10.9.4826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz K. B., McGuire W. L. Nuclear mechanisms of estrogen action. Effects of estradiol and anti-estrogens on estrogen receptors and nuclear receptor processing. J Biol Chem. 1978 Nov 25;253(22):8185–8191. [PubMed] [Google Scholar]
- Ignar-Trowbridge D. M., Nelson K. G., Bidwell M. C., Curtis S. W., Washburn T. F., McLachlan J. A., Korach K. S. Coupling of dual signaling pathways: epidermal growth factor action involves the estrogen receptor. Proc Natl Acad Sci U S A. 1992 May 15;89(10):4658–4662. doi: 10.1073/pnas.89.10.4658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwaki K., Sukhatme V. P., Shubeita H. E., Chien K. R. Alpha- and beta-adrenergic stimulation induces distinct patterns of immediate early gene expression in neonatal rat myocardial cells. fos/jun expression is associated with sarcomere assembly; Egr-1 induction is primarily an alpha 1-mediated response. J Biol Chem. 1990 Aug 15;265(23):13809–13817. [PubMed] [Google Scholar]
- Kenyon G. L., Reed G. H. Creatine kinase: structure-activity relationships. Adv Enzymol Relat Areas Mol Biol. 1983;54:367–426. doi: 10.1002/9780470122990.ch6. [DOI] [PubMed] [Google Scholar]
- Klein-Hitpass L., Schorpp M., Wagner U., Ryffel G. U. An estrogen-responsive element derived from the 5' flanking region of the Xenopus vitellogenin A2 gene functions in transfected human cells. Cell. 1986 Sep 26;46(7):1053–1061. doi: 10.1016/0092-8674(86)90705-1. [DOI] [PubMed] [Google Scholar]
- Kumar V., Chambon P. The estrogen receptor binds tightly to its responsive element as a ligand-induced homodimer. Cell. 1988 Oct 7;55(1):145–156. doi: 10.1016/0092-8674(88)90017-7. [DOI] [PubMed] [Google Scholar]
- Lissoos T. W., Beno D. W., Davis B. H. 1,25-Dihydroxyvitamin D3 activates Raf kinase and Raf perinuclear translocation via a protein kinase C-dependent pathway. J Biol Chem. 1993 Nov 25;268(33):25132–25138. [PubMed] [Google Scholar]
- Lopez G., Schaufele F., Webb P., Holloway J. M., Baxter J. D., Kushner P. J. Positive and negative modulation of Jun action by thyroid hormone receptor at a unique AP1 site. Mol Cell Biol. 1993 May;13(5):3042–3049. doi: 10.1128/mcb.13.5.3042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mader S., Kumar V., de Verneuil H., Chambon P. Three amino acids of the oestrogen receptor are essential to its ability to distinguish an oestrogen from a glucocorticoid-responsive element. Nature. 1989 Mar 16;338(6212):271–274. doi: 10.1038/338271a0. [DOI] [PubMed] [Google Scholar]
- Mitchell M. T., Benfield P. A. Two different RNA polymerase II initiation complexes can assemble on the rat brain creatine kinase promoter. J Biol Chem. 1990 May 15;265(14):8259–8267. [PubMed] [Google Scholar]
- Pentecost B. T., Mattheiss L., Dickerman H. W., Kumar S. A. Estrogen regulation of creatine kinase-B in the rat uterus. Mol Endocrinol. 1990 Jul;4(7):1000–1010. doi: 10.1210/mend-4-7-1000. [DOI] [PubMed] [Google Scholar]
- Pollock R., Treisman R. Human SRF-related proteins: DNA-binding properties and potential regulatory targets. Genes Dev. 1991 Dec;5(12A):2327–2341. doi: 10.1101/gad.5.12a.2327. [DOI] [PubMed] [Google Scholar]
- Reiss N. A., Kaye A. M. Identification of the major component of the estrogen-induced protein of rat uterus as the BB isozyme of creatine kinase. J Biol Chem. 1981 Jun 10;256(11):5741–5749. [PubMed] [Google Scholar]
- Schüle R., Rangarajan P., Kliewer S., Ransone L. J., Bolado J., Yang N., Verma I. M., Evans R. M. Functional antagonism between oncoprotein c-Jun and the glucocorticoid receptor. Cell. 1990 Sep 21;62(6):1217–1226. doi: 10.1016/0092-8674(90)90397-w. [DOI] [PubMed] [Google Scholar]
- Sen A., Miller J. C., Reynolds R., Willerson J. T., Buja L. M., Chien K. R. Inhibition of the release of arachidonic acid prevents the development of sarcolemmal membrane defects in cultured rat myocardial cells during adenosine triphosphate depletion. J Clin Invest. 1988 Oct;82(4):1333–1338. doi: 10.1172/JCI113735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spatz M., Waisman A., Kaye A. M. Responsiveness of the 5'-flanking region of the brain type isozyme of creatine kinase to estrogens and antiestrogen. J Steroid Biochem Mol Biol. 1992 Mar;41(3-8):711–714. doi: 10.1016/0960-0760(92)90409-c. [DOI] [PubMed] [Google Scholar]
- Sömjen D., Kaye A. M., Binderman I. 24R,25-dihydroxyvitamin D stimulates creatine kinase BB activity in chick cartilage cells in culture. FEBS Lett. 1984 Feb 27;167(2):281–284. doi: 10.1016/0014-5793(84)80143-x. [DOI] [PubMed] [Google Scholar]
- Sömjen D., Weisman Y., Harell A., Berger E., Kaye A. M. Direct and sex-specific stimulation by sex steroids of creatine kinase activity and DNA synthesis in rat bone. Proc Natl Acad Sci U S A. 1989 May;86(9):3361–3365. doi: 10.1073/pnas.86.9.3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tasset D., Tora L., Fromental C., Scheer E., Chambon P. Distinct classes of transcriptional activating domains function by different mechanisms. Cell. 1990 Sep 21;62(6):1177–1187. doi: 10.1016/0092-8674(90)90394-t. [DOI] [PubMed] [Google Scholar]
- Tora L., Mullick A., Metzger D., Ponglikitmongkol M., Park I., Chambon P. The cloned human oestrogen receptor contains a mutation which alters its hormone binding properties. EMBO J. 1989 Jul;8(7):1981–1986. doi: 10.1002/j.1460-2075.1989.tb03604.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tora L., White J., Brou C., Tasset D., Webster N., Scheer E., Chambon P. The human estrogen receptor has two independent nonacidic transcriptional activation functions. Cell. 1989 Nov 3;59(3):477–487. doi: 10.1016/0092-8674(89)90031-7. [DOI] [PubMed] [Google Scholar]
- Webster N. J., Green S., Jin J. R., Chambon P. The hormone-binding domains of the estrogen and glucocorticoid receptors contain an inducible transcription activation function. Cell. 1988 Jul 15;54(2):199–207. doi: 10.1016/0092-8674(88)90552-1. [DOI] [PubMed] [Google Scholar]
- Weinstein L. S., Kats I., Spiegel A. M., Carter A. D. Characterization of the promoter of the human Gi2 alpha-subunit gene. Mol Endocrinol. 1990 Jul;4(7):958–964. doi: 10.1210/mend-4-7-958. [DOI] [PubMed] [Google Scholar]
- Weisz A., Rosales R. Identification of an estrogen response element upstream of the human c-fos gene that binds the estrogen receptor and the AP-1 transcription factor. Nucleic Acids Res. 1990 Sep 11;18(17):5097–5106. doi: 10.1093/nar/18.17.5097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu-Peng X. S., Pugliese T. E., Dickerman H. W., Pentecost B. T. Delineation of sites mediating estrogen regulation of the rat creatine kinase B gene. Mol Endocrinol. 1992 Feb;6(2):231–240. doi: 10.1210/mend.6.2.1569966. [DOI] [PubMed] [Google Scholar]
- Yang-Yen H. F., Chambard J. C., Sun Y. L., Smeal T., Schmidt T. J., Drouin J., Karin M. Transcriptional interference between c-Jun and the glucocorticoid receptor: mutual inhibition of DNA binding due to direct protein-protein interaction. Cell. 1990 Sep 21;62(6):1205–1215. doi: 10.1016/0092-8674(90)90396-v. [DOI] [PubMed] [Google Scholar]
- Yu Y. T., Breitbart R. E., Smoot L. B., Lee Y., Mahdavi V., Nadal-Ginard B. Human myocyte-specific enhancer factor 2 comprises a group of tissue-restricted MADS box transcription factors. Genes Dev. 1992 Sep;6(9):1783–1798. doi: 10.1101/gad.6.9.1783. [DOI] [PubMed] [Google Scholar]
- Zhang X. K., Wills K. N., Husmann M., Hermann T., Pfahl M. Novel pathway for thyroid hormone receptor action through interaction with jun and fos oncogene activities. Mol Cell Biol. 1991 Dec;11(12):6016–6025. doi: 10.1128/mcb.11.12.6016. [DOI] [PMC free article] [PubMed] [Google Scholar]