Abstract
In 2008, one of the worst public health crises occurred in the state of Nevada, where authorities discovered up to 63,000 patients were potentially exposed to hepatitis C infection, largely due to substandard infection control and other negligent practices at two endoscopy clinics in Las Vegas. In the subsequent grand jury proceedings that followed, it was discovered that several clinic employees not only participated in these egregious practices, but doctors, nurses, and other health care professionals witnessed yet failed to report these incidents, largely due to fears of whistleblower retaliation. In response, the Nevada state legislature attempted to strengthen whistleblower protection laws, but it remains unclear if such laws actually protect employees who attempt to report patient safety concerns. As the push for quality patient outcomes becomes more prominent with health care reform, whistleblower concerns must be effectively addressed to ensure that health care professionals can report patient safety concerns without fear of retaliation.
Keywords: nurses, nursing, whistleblower, whistleblowing, hepatitis C, Nevada, outbreak, infection control, Desai, nurse anesthetists, public health, retaliation
As a new graduate nurse fresh out of nursing school, Anne Yost was nervous her first day on the job at the Endoscopy Center of Southern Nevada, a gastroenterology surgical center in Las Vegas [1]. What made Yost even more anxious was the fact that two of the nurses who were assigned to train her expected Yost to pre-chart on patients (enter vital signs and other information before having seen the patient), in violation of standard nursing practice. “I didn’t want to document on a patient before I had actually seen them,” Yost said [2]. Besides pre-charting, Yost was also asked to document inaccurate times when patients had been assessed and to sign off on charting that was filled out by other nurses. “I would ask why the paperwork was already filled out and the nurse training me would say that was done so the turning (patient turnaround time) could be done quicker” [3].
Cutting corners and high patient volume were the priorities at the center, where Yost was also instructed to chart that all of the patients were in good condition, whether they were or not. “Every time it was documented exactly the same way, that they were in good health” [4]. Yost quickly realized she was not going to fit into this type of work environment so, after just 3 days, she quit. When Yost attempted to report her concerns to the Nevada State Board of Nursing (NSBN), she was told that she would have to name specific nurses who participated in the false documentation practices. Since she was a new employee, Yost only remembered the first names of nurses at the center, not last names. “They said they couldn’t process it any further without somebody to point the finger at” [5].
Seven months later, the center became the focus of one of most egregious public health crises in U.S. history, where up to 63,000 patients were potentially exposed to blood-borne pathogens, including hepatitis C [6]. The exposures were attributed to unsafe injection practices and other failed infection control procedures at the endoscopy center [7]. At least nine patients ultimately contracted hepatitis C due to these unsafe practices, and one patient died [8]. In the grand jury proceedings that followed, it was revealed that doctors, nurses, and other clinic employees were aware of the unsafe practices at the clinic [9]. Yet, with the exception of Yost, none of these employees attempted to report the incidents to authorities, mostly due to fears of losing their jobs [10]. Consequently, these dangerous practices were not discovered until months later, when local officials identified an unusual cluster of hepatitis C cases that originated from the endoscopy center [6]. Since whistleblower retaliation fears delayed reporting of the incidents leading to the outbreak, this case raises serious concerns regarding whistleblower retaliation in the health care environment, especially when employer directives jeopardize patient safety.
Background
This case study is developed from publicly available documents, including websites, government documents, grand jury transcripts, and Internet news reports. The case should not be taken as a definitive account of the legal issues, since a good deal of the material comes from the transcripts of grand jury hearings over several days [9]. The evidentiary standard for grand jury proceedings is not as rigorous as criminal trials, and there is no cross-examination [11].
The target of the grand jury probe was Dr. Depak Desai, a gastrointestinal specialist who owned and operated a number of endoscopy centers in Las Vegas, Nevada [12]. In 2007, the year before the hepatitis outbreak occurred, Dr. Desai made $6.8 million and his physician partners made another $3.4 million in profits [13]. According to grand jury testimony, much of those profits were generated due to Dr. Desai’s willingness to disregard basic health care standards, including patient safety [9]. Dr. Desai employed salaried physicians and certified registered nurse anesthetists (CRNAs) whose billings went into a central account, which he controlled [14]. While nurse anesthetist billings also went into this account, the nurses did not participate financially beyond their salaries [15].
Origins of the Case
The Southern Nevada Health District (SNHD), like all other state departments of health, collects routine surveillance data on infections, among other things [16]. On January 2, 2008, the Department of Health identified an unusual cluster of new hepatitis C cases and discovered the infected patients all had contact with one endoscopy clinic as well as another clinic owned by Dr. Desai [6]. Not having the resources to conduct a full epidemiologic investigation, the health department enlisted the help of the Centers for Disease Control (CDC), which investigates such outbreaks [7]. The CDC sent an investigative team and, after a thorough study including observations at the endoscopy site, performed genetic testing that tracked the infections to the endoscopy center [7]. The Executive Summary of the SNHD Report concluded:
The total cost of this outbreak investigation, response and testing to the community was estimated to be between $16 million and $21 million. Each of the 63,000 possible patient exposures identified in this investigation were [sic] entirely preventable, and would not have occurred if clinical staff had adhered to well-established, safe and common sense injection practices [6].
Apart from the direct evidence of hepatitis C transmission, observations made by the CDC investigators flagged personnel policies, clinical protocols, and other practices common at the center that raised legal concerns [7]. Based on these findings, the District Attorney’s Office eventually filed criminal charges against Dr. Desai and two nurse anesthetists, Ronald Lakeman and Keith Mathahs [17]. A grand jury was convened, and testimony was taken from a large number of witnesses, including nurses, nurse anesthetists, physicians, billing clerks, and other employees [9]. Several witnesses were granted “use immunity,” meaning that they would not be prosecuted in the immediate case but the immunity would not protect them from legal action from other authorities such as state licensing boards [18].
Cost Cutting At Patient Expense
According to grand jury testimony, some of the more nefarious cost-cutting practices condoned by Dr. Desai included use of contaminated endoscopes [19], re-use of fecal contaminated gowns [20], re-use of disposable bite blocks [21] and biopsy equipment [22] as well as instructions to staff members to avoid using gloves unnecessarily [23]. In an extreme cost-cutting measure, Dr. Desai even instructed nurses to cut Chux under pads in half [24], even though this is an inexpensive item that costs only 19 cents per Amazon.com rates [25].
In one of Dr. Desai’s most egregious practices, he instructed one nurse anesthetist to sedate a patient who failed to comply with nothing-by-mouth instructions, in which the patient is instructed not to ingest food or beverages before the procedure [26]. After witnessing the patient drinking in the waiting room, nurse anesthetist Ann Marie Lobiondo feared the patient would aspirate, so she refused to administer the anesthesia [27]. During anesthesia, patients lose control of their airway, and stomach contents may move from the esophagus into the lungs, potentially causing aspiration pneumonia. “It’s a bad thing,” Lobiondo testified. “You can wind up in the hospital or on a ventilator for that” [27].
Yet Dr. Desai and his office manager, Tonya Rushing, insisted that Lobiondo anesthetize the patient anyway [27]. “I absolutely refused and said I wasn’t going to do it,” Lobiondo testified. “They insisted. I said, well, then, I’m going to have to leave the facility” [27]. When Lobiondo did leave, Rushing followed her out to the parking lot, where the office manager threatened to call Dr. Desai’s lawyers [27]. Nevertheless, Lobiondo did leave, and she never returned to the center again [27].
Infection from Vial Reuse
While there were many practices at the center that endangered patients’ lives, what ultimately caused the infection of the nine hepatitis C patients was the re-use of anesthetic propofol vials [7]. Propofol (implicated in the death of Michael Jackson [28]) is an anesthetic medication known to harbor bacterial growth, which is why the manufacturer as well as the Food and Drug Administration (FDA) recommend that vials be discarded immediately after they are used for individual patients [29].
However, in order to cut costs and also enhance patient turnover by keeping patient sedation to a minimum, Dr. Desai insisted that the nurse anesthetists use as little propofol as possible [30]. Dr. Desai also instructed the nurse anesthetists to re-use the propofol vials so no medication would be wasted [31]. The nurse anesthetists for the most part complied [32]. Some nurses even re-used syringes [33]. The outbreak was traced to two patients known to have contracted hepatitis C [6], in which the nurse anesthetists drew up propofol and injected it into the port, withdrawing a small amount of blood (Figure 1). The nurses then discarded the needles but used the same syringe to inject propofol from the same vial into the next patient [7].
Figure 1.
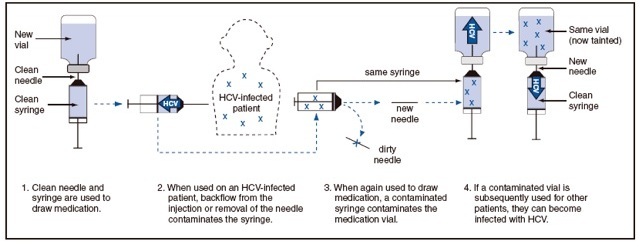
Unsafe Injection Practices at a Nevada Endoscopy Clinic Which Likely Led to Spread of Hepatitis C Infection in 2007 (Courtesy: Centers for Disease Control).
Fraudulent Billing
Besides poor infection control practices, the nurse anesthetists also falsified records of their time so Dr. Desai could fraudulently double and even triple bill insurance companies for anesthesia time spent on procedures [34]. Anesthesia is billed in “base” units for the procedure type and then time units in increments of 15 minutes [35]. Nevertheless, nearly all procedures in Dr. Desai’s clinics were billed for 31 to 32 minutes [36], even though grand jury testimony revealed that Dr. Desai typically spent a maximum of only 7 minutes for a colonoscopy and 2 minutes for an upper endoscopy [37].
Yet Dr. Desai insisted that the CRNAs bill at least 31 minutes for these procedures [40]. Why did the nurses comply with Dr. Desai’s instructions? Nurse anesthetist Vincent Sagendorf testified that fraudulent practice was necessary in order to keep his job [38]. “I knew it was wrong, but I had quit my job in California. I had a wife and I had house payments and I needed that job” [10]. Other nurses who worked for Dr. Desai also feared retaliation and job loss. As nurse anesthetist Lobiondo told the grand jury, she constantly felt that her job was in jeopardy if she did not cooperate with Dr. Desai’s demands. “He was very intimidating and he was brutal” [39].
One of Dr. Desai’s partners, Dr. Clifford Carroll, also found Dr. Desai intimidating. Carroll fired nurse anesthetist Linda Hubbard after she continued to re-use propofol vials even after public health investigators had linked the hepatitis outbreak to the re-used vials [40]. Carroll testified that he saw Hubbard save the propofol vial from one patient and then, with a new syringe and a new needle, go back into that same propofol vial for the next patient. Carroll immediately fired Hubbard, telling her, “I watched you do exactly what we told you not to do … You’re going to be terminated right now” [40]. Yet, Dr. Desai eventually overruled Carroll’s decision, rewarding Hubbard with paid leave and a new job at another endoscopy center [40].
Fallout
When the hepatitis C outbreak became public in 2008, authorities received numerous reports that nurses and other health care professionals failed to report patient safety concerns and even participated in unsafe practices at the center [41]. Five CRNAs voluntarily surrendered their licenses [42], and 22 registered nurses (RNs) and licensed practical nurses (LPNs) were investigated by the Nevada State of Board Nursing (NSBN) [43]. According to NSBN Executive Director Debra Scott (personal communication June 6, 2012), the board has not taken any public disciplinary action against any of the nurses with pending open investigations related to the hepatitis crisis [44]. “We are being very cautious because of the numerous civil and criminal cases still pending,” Scott says. “Not to mention that much of the evidence that we have subpoenaed is still held by other entities which are not willing to provide it to us” [44].
While the investigations are still pending, the Nevada legislature commissioned a survey of RNs conducted by University of Nevada researcher Lisa Black, which found that 34 percent of nurses surveyed were aware of harmful patient situations, yet 44 percent of those respondents did not report their concerns due to workplace retaliation fears [43]. Thirty-eight percent of the respondents said they did not report patient safety concerns “because they didn’t think anything would come of the report” [43]. In response, the Nevada legislature strengthened its whistleblower protection law, which now creates a rebuttable presumption that workers are victims of retaliation if they are fired or subject to adverse employment action within 60 days of reporting unsafe practices [45]. The statute also allows workers to receive court-ordered damages, including attorney’s fees [45].
There are 37 other states with similar whistleblower protection statutes [46], yet such remedies may not be very effective. For instance, a 2008 survey of 752 British nurses indicated that U.K. whistleblower protection statutes did not prevent employer retaliation [47]. In the British survey, 58 percent of the nurses felt nothing would be done and 46 percent felt they would experience retaliation if they reported patient safety concerns [47]. Similar findings were published in a study of the Sarbanes-Oxley Act, a federal statute intended to protect corporate whistleblowing employees [48]. Within 3 years of enactment, only 3.6 percent of Sarbanes-Oxley whistleblowers won relief through the initial administrative process set up by the law, and only 6.5 percent won their cases on appeal [48].
Illusion of Protection
There are several reasons why whistleblower protection laws may not be very effective. Legal remedies take time and frequently do not occur until years after the reprisals had begun [49]. In the meantime, whistleblowing employees may find themselves out of a job. “Nobody speaks out because they’ve seen that you can lose your job,” said one nurse who participated in a 2010 survey of whistleblowing employees [48]. This was the case for Anne Mitchell, a Texas registered nurse who was fired and criminally prosecuted in 2010 after she and another nurse reported a physician’s unsafe practices to the Texas Medical Board [50].
The Texas case marks the first time that nurses have been criminally prosecuted for reporting patient care concerns [43]. Although the prosecutor and the sheriff were eventually convicted of illegal activities in their pursuit of the whistleblowing nurses [49], other studies confirm nurses who report wrongdoing typically suffer severe consequences including demotion, reprimand and, even, threats [51]. “Results indicated there were severe professional reprisals if the nurse reported misconduct, but there were few professional consequences if the nurse remained silent” [51].
Nurses are not the only health care professionals who have suffered reprisals for whistleblowing activity. Dr. Eric Topol was demoted by the Cleveland Clinic after exposing cardiovascular safety concerns with the drug Vioxx, forcing Merck to withdraw the medication from the market [52]. Another doctor, Peter Rost, was fired by Pfizer in 2005 after blowing the whistle on the pharmaceutical giant’s marketing practices [53]. While Rost managed to launch a career out of his whistleblowing activities [54], most whistleblowers are not as fortunate. According to one survey of 233 whistleblowing employees, 60 percent lost their jobs, 17 percent experienced a decrease in salary, while 14 percent were forced to retire due to whistleblowing activities [55].
Even if they did not suffer financial harm, the vast majority of whistleblowers (82 percent) reported supervisory retaliation [55], which may include more subtle forms of intimidation such as spreading rumors, social ostracism, petty harassment, job re-assignments, as well as increased work demands [56]. Brian Martin, author of Illusions of Whistleblower Protection, writes that it is more difficult to prove whistleblower retaliation in these cases since it is easy to camouflage the changes as necessary due to organizational need [57]. “Ironically, it can be more difficult for the employee to deal with subtle undermining than with a more obvious attack such as demotion or dismissal” [56]. Along these lines, 26 percent of whistleblowing employees reported mental health as well as medical problems that they attributed to whistleblower retaliation [55].
Conclusions
Despite the proliferation of whistleblower protection statutes in recent years [46], perhaps it is not all that surprising that licensed health care professionals chose not to report the substandard infection control practices that caused the Nevada hepatitis C outbreak [9]. After all, whistleblower statutes cannot effectively address the immediate financial concerns associated with job loss and other forms of retaliation that inevitably occur once an employee chooses to blow the whistle [56]. Nevertheless, the Nevada hepatitis C case demonstrates the great quandary licensed health care professionals face in today’s environment. If employees “report unsafe practices, they risk losing their jobs,” Black writes. “If they don’t, they risk losing their licenses” [43].
As health care reform efforts are driving the need for more accurate reporting of patient safety concerns, whistleblower retaliation fears must be effectively addressed. Since statutes may fail to address the immediate effects of workplace retaliation, anonymous reporting systems may be a more effective solution. Health care professionals might be more inclined to report their patient safety concerns if their identities are protected and the risk of retaliation is minimized. In fact, the Veterans Administration encourages anonymous reporting through the Patient Safety Reporting System (PSRS), in which unidentified VA employees are allowed to report their concerns [57]. Within 2 years of implementing PSRS, the VA received more than 400 patient safety reports and issued 10 safety bulletins [58]. If similar anonymous reporting systems are widely adopted, perhaps this will encourage health care providers to report patient safety concerns without fear of workplace retaliation and job loss.
Update
Five victims of the hepatitis C outbreak lodged a class action suit against the pharmaceutical companies who produce propofol, alleging that supplying the medication in larger 50 ml vials rather than smaller 20 ml single dose vials encouraged reuse and thus transmission of infection [59]. These suits were eventually settled out of court [60]. Meanwhile, Dr. Desai surrendered his license [61], and his lawyer resisted trial for his client on the grounds that the physician was too ill and compromised to testify in his own behalf [62]. The court ultimately dismissed this line of reasoning, and the trial is tentatively scheduled to begin in May 2013 [63].
Abbreviations
- CDC
Centers for Disease Control
- CRNA
Certified Registered Nurse Anesthetist
- LPN
Licensed Practical Nurse
- NSBN
Nevada State of Board of Nursing
- RN
Registered Nurse
- SNHD
Southern Nevada Health District
- PSRS
Patient Safety Reporting System
- VA
Veterans Administration
Author contributions
This case study is the result of a collaboration between YSN Professor Emerita Donna Diers and YSN NMPL Master’s Candidate Elizabeth Leary. Ms. Leary contributed much of the writing and research for the article while Professor Diers provided critical guidance, editing, writing, and research assistance. No funding was provided for this article.
Author's note
Author Dr. Donna Diers died on February 23, 2013, before this issue went into press. This article was her idea, and she worked tirelessly to advance the practice of nursing.
References
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:50. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:27. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:28. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:33. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:44. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- Outbreak Investigation Team. Outbreak of Hepatitis C at Outpatient Surgical Centers: Public Health Investigation Report. Southern Nevada Health District [Internet] 2009. Dec, [cited 2012 Nov 21]. Available from: http://www.southernnevadahealthdistrict.org/download/outbreaks/final-hepc-investigation-report.pdf .
- Centers for Disease Control. Acute Hepatitis C Virus Infections Attributed of Unsafe Injection Practices at an Endoscopy Clinic — Nevada, 2007. MMWR Morb Mortal Wkly Rep. 2008;57(19):513–517. [PubMed] [Google Scholar]
- Rozyla L. Who Contracted Hepatitis C in Desai Clinic Dies. KLAS-TV [Internet] 2012. Jul 31, [cited 2012 Nov 21]. Available from: http://www.8newsnow.com/story/19161151/man-who-contracted-hep-c-in-desai-clinic-dies .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. Las Vegas Review Journal [Internet] 2010. Mar-May, [cited 2012 Nov 21]. pp. 1–9. Available from: http://www.lvrj.com/news/desai_grand_jury_transcripts.html .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:99. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- Ohio State Bar Association. How Does a Grand Jury Operate? [Internet] 2012. Nov 12, [cited 2012 Nov 22]. Available from: https://www.ohiobar.org/forpublic/resources/lawyoucanuse/pages/lawyoucanuse-86.aspx .
- Ball M, Haynes B. Clinic Owner Found Success. Las Vegas Review Journal [Internet] 2008. Feb 29, [updated 2008 Sept 26; cited 2012 Nov 21]. Available from: http://www.lvrj.com/news/16105787.html .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:128. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Mar 18;2:58. Available from: http://media.lvrj.com/documents/desai_transcript_vol2.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 21];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:129. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- Southern Nevada Health District. Mission: To Protect and Promote the Health, the Environment and the Well Being of Southern Nevada Residents and Visitors [Internet] 2012. [cited 2012 Nov 22]. Available from: https://www.southernnevadahealthdistrict.org/ .
- Clark County District Attorney. State of Nevada vs. Dipak Kantilala Desai, Ronald Ernest Lakeman, Keith J. Mathahs. District Court Clark County Nevada [Internet] 2010. [cited 2012 Nov 22]. Available from: http://media.lasvegassun.com/media/pdfs/blogs/documents/2010/06/04/desai0604.pdf/ .
- Offices of the United States Attorneys. Transactional Immunity Distinguished. Title 9 Criminal Resource Manual 717 [Internet] 2012. [cited 2012 Nov 22]. Available from: http://www.justice.gov/usao/eousa/foia_reading_room/usam/title9/crm00717.htm .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 May 6;6:65. Available from: http://media.lvrj.com/documents/desai_transcript_vol6.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Mar 18;2:79. Available from: http://media.lvrj.com/documents/desai_transcript_vol2.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Apr 15;41:87. Available from: http://media.lvrj.com/documents/desai_transcript_vol41.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Apr 15;3:92. Available from: http://media.lvrj.com/documents/desai_transcript_vol3.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 May 27;9:47. Available from: http://media.lvrj.com/documents/desai_transcript_vol9.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Mar 11;1:98. Available from: http://media.lvrj.com/documents/desai_transcript_vol1.pdf . [Google Scholar]
- Amazon.com. Disposable Blue Under Pad Chux. Health and Personal Care [Internet] 2012. [cited 2012 Nov 22]. Available from: http://www.amazon.com/Disposable-Blue-Underpad-Chux-23x36/dp/B000FA05Y6 .
- Farlex. Nothing by Mouth (NPO). Medical Dictionary [Internet] 2012. [cited 2012 Nov 23]. Available from: http://medical-dictionary.thefreedictionary.com/nothing+by+mouth .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 May 6;6:26–27. Available from: http://media.lvrj.com/documents/desai_transcript_vol6.pdf . [Google Scholar]
- Levy RJ. The Michael Jackson Autopsy: Insights Provided by a Forensic Anesthesiologist. J Forensic Res. 2011;2:138. [Google Scholar]
- Food and Drug Administration. Information for Healthcare Professionals: Propofol. Drug Safety and Availability [Internet] 2009. Aug 28, [cited 2012 Nov 21]. Available from: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm125817.htm .
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Mar 11;6:71. Available from: http://media.lvrj.com/documents/desai_transcript_vol6.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:86. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 15;3a:53. Available from: http://media.lvrj.com/documents/desai_transcript_vol3a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 15;3:34. Available from: http://media.lvrj.com/documents/desai_transcript_vol3.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:92–106. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Mar 11;1:89. Available from: http://media.lvrj.com/documents/desai_transcript_vol1.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Mar 18;2:55. Available from: http://media.lvrj.com/documents/desai_transcript_vol2.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:92. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 23];Las Vegas Review Journal [Internet] 2010 Apr 22;4a:114. Available from: http://media.lvrj.com/documents/desai_transcript_vol4a.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 May 6;6:81. Available from: http://media.lvrj.com/documents/desai_transcript_vol6.pdf . [Google Scholar]
- State of Nevada Eighth Judicial District Court. Desai Grand Jury Transcripts. [cited 2012 Nov 22];Las Vegas Review Journal [Internet] 2010 Mar 18;2:73–75. Available from: http://media.lvrj.com/documents/desai_transcript_vol2.pdf . [Google Scholar]
- Sands L. Interim Report on the Endoscopy Center of South Nevada Hepatitis C Investigation. Memo [Internet] 2008. Mar 28, [cited 2012 Nov 23]. Available from: http://www.southernnevadahealthdistrict.org/download/outbreaks/interim-hepc-investigation-report.pdf .
- Wood D. CRNAs Surrender Licenses in Nevada Hep C Investigation. NurseZone.com [Internet] 2008. [cited 2012 Nov 24]. Available from: http://www.nursezone.com/nursing-news-events/more-news/CRNAs-Surrender-Licenses-in-Nevada-Hep-C-Investigation_26964.aspx .
- Black LM. Tragedy into Policy: A Quantitative Study of Nurses’ Attitudes toward Patient Advocacy Activities. Am J Nurs. 2011;111(6):26–35. doi: 10.1097/01.NAJ.0000398537.06542.c0. [DOI] [PubMed] [Google Scholar]
- Scott D. Message to Elizabeth Leary. 2012. Jun 6, 1 paragraph
- Blomstrom C. Legislative Report as of June, 2009. Nevada RNFormation: Official Publication of the Nevada Nurses Association [Internet] 2009. Aug, [cited 2012 Nov 23]. Available from: http://www.nursingald.com/Uploaded%5CNewsletterFiles%5CNV8_09.pdf .
- National Conference of State Legislatures. State Whistleblower Laws [Internet] 2009. Nov, [cited 2012 Nov 24]. Available from: http://www.ncsl.org/issues-research/labor/state-whistleblower-laws.aspx .
- Moberly R. Unfulfilled Expectations: An Empirical Analysis of Why Sarbanes-Oxley Whistleblowers Rarely Win. William and Mary Law Review. 2007;49(1):65,67. [Google Scholar]
- Jackson D, Peters K, Andrew S, Edenborough M, Halcomb E, Luck L. et al. Understanding Whistleblowing: Qualitative Insights from Nurse Whistleblowers. J Adv Nurs. 2010;66(10):2194–2201. doi: 10.1111/j.1365-2648.2010.05365.x. [DOI] [PubMed] [Google Scholar]
- Editorial Board. Justice Doesn’t Always Have Happy Ending. Statesman.com [Internet] 2011. Oct 6, [cited 2012 Nov 23]. Available from: http://www.statesman.com/news/news/opinion/justice-doesnt-always-have-happy-ending/nRf6t/ .
- Kennedy MS. Protectors in Need of Protection. Am J Nurs. 2011;111(6):7. doi: 10.1097/01.NAJ.0000398514.05649.82. [DOI] [PubMed] [Google Scholar]
- McDonald S, Ahern K. The Professional Consequences of Whistleblowing by Nurses. J Prof Nurs. 2000;16(6):313–321. doi: 10.1053/jpnu.2000.18178. [DOI] [PubMed] [Google Scholar]
- Abelson R, Saul S. Ties to Industry Cloud a Clinic’s Mission. The New York Times [Internet] 2005. Dec 17, Available from: http://www.nytimes.com/2005/12/17/business/17clinic.html .
- Rost P. The Whistleblower: Confessions of a Healthcare Hitman. 1st ed. Brooklyn: Soft Skull Press; 2006. [Google Scholar]
- Rost P. Peter Rost: Pharma Marketing Expert Witness, Medical Device Expert [Internet] 2012. [cited 2012 Dec 29]. Available from: http://peterrost.blogspot.com/ .
- Soeken K, Soeken D. A Survey of Whistleblowers: Their Stressors and Coping Strategies [Internet] 2012. [cited 2012 Dec 29]. Available from: http://www.whistleblower-net.de/pdf/Soeken.pdf .
- Martin B. Illusions of Whistleblower Protection. UTS Law Review [Internet] 2003. [cited 2012 Nov 23]. Available from: http://www.bmartin.cc/pubs/03utslr.html .
- National Aeronautics and Space Administration. Frequently Asked Questions. Patient Safety Reporting System [Internet] 2010. Feb 26, [cited 2012 Nov 23]. Available from: http://psrs.arc.nasa.gov/faq.html .
- The Commonwealth Fund. Case Study: NASA/VA Patient Safety Reporting System [Internet] 2005. Jan 20, [cited 2012 Nov 25]. Available from: http://www.commonwealthfund.org/Innovations/Case-Studies/2005/Jan/Case-Study--NASA-VA-Patient-Safety-Reporting-System.aspx .
- Ritter K. Jury: Drug Firms Liable in Vegas Hepatitis Case. The Seattle Times [Internet] 2011. Oct 6, [cited 2012 Nov 23]. Available from: http://seattletimes.com/html/health/2016430148_heptrial07.html .
- Haynes B. Drug Maker to Pay $285 Million to Settle Hepatitis Lawsuits. Las Vegas Review Journal [Internet] 2012. Feb 21, [cited 2012 Nov 23]. Available from: http://www.lvrj.com/news/propofol-maker-teva-to-pay-250-million-to-settle-nevada-lawsuits-139856843.html .
- Harasim P. Doctor Linked to Hepatitis C Outbreak Surrenders Medical License. Las Vegas Review Journal [Internet] 2010. Feb 25, [cited 2012 Nov 23]. Available from: http://www.lvrj.com/news/desai-surrenders-medical-license-85268132.html .
- McCabe F. Desai in Court to Begin Appeal of Competency Decision. Las Vegas Review Journal [Internet] 2011. Oct 11, [cited 2012 Nov 23]. Available from: http://www.lvrj.com/news/desai-in-court-to-begin-appeal-of-competency-decision-131539283.html .
- German J. Dr. Desai’s Federal Trial Delayed. Las Vegas Review Journal [Internet] 2012. Nov 12, [cited 2012 Nov 23]. Available from: http://www.lvrj.com/news/dr-desai-s-federal-trial-delayed-178950241.html .


