Abstract
Allergic asthma is associated with Th2-mediated inflammation. Several flavonoids were isolated from Glycyrrhiza uralensis, one of the herbs in the anti-asthma herbal medicine intervention, ASHMI. The aim of this investigation was to determine whether Glycyrrhiza uralensis flavonoids have inhibitory effects on memory Th2 responses in vitro, and antigen induced Th2 inflammation in vivo. The effects of three Glycyrrhiza uralensis flavonoids on effector memory Th2 cells, D10.G4.1 (D10 cells), were determined by measuring Th2 cytokine production. Isoliquiritigenin, 7, 4’-dihydroxyflavone (7, 4’-DHF) and liquiritigenin significantly suppressed IL-4 and IL-5 production in a dose dependent manner, 7, 4’-DHF being most potent. It was also evaluated for effects on D10 cell proliferation, GATA-3 expression and IL-4 mRNA expression, which were suppressed, with no loss of cell viability. Chronic treatment with 7, 4’-DHF in a murine model of allergic asthma not only significantly reduced eosinophilic pulmonary inflammation, serum IgE levels, IL-4 and IL-13 levels, but also increased IFN-γ production in lung cell cultures in response to antigen stimulation.
Keywords: Glycyrrhiza uralensis, flavonoids, D10. G 4.1, Th2 cytokines, GATA-3, Murine model of asthma
INTRODUCTION
Asthma, a chronic inflammatory airway disease, is one of the most common respiratory diseases in the United States. About 25 million Americans have asthma, and 300 million people are affected worldwide (American Academy of Allergy, 2010). Asthma is the leading childhood chronic disease, affecting 9.6% of American children (American Academy of Allergy, 2010). This disease involves activation of many inflammatory cell types, and abnormal levels of multiple cytokines (Finkelman et al., 2010). There is substantial evidence to indicate that polarized (effector memory) Th2 cells secretion of Th2 cytokines following repeated antigen encounter are the major driving forces behind chronic allergic asthma (Finkelman et al., 2010; Larche et al., 2003; Salek-Ardakani et al., 2003). Th2 polarized cells with a CD44+CD62L-CCR7- phenotype in mice and CD45RO+CCR7- in man, termed “effector memory” CD4 T cells, are endowed with immediate effector functions (Blander et al., 2003; Dutton et al., 1998; Messi et al., 2003; Reinhardt et al., 2001; Sallusto et al., 1999). IL-4, generated by CD4+ Th2 effector cells, is essential for Th2 cell differentiation and production of allergen-specific IgE (Epstein, 2006). IL-5, secreted by activated T cells, is critical in regulating differentiation, maturation and activation of eosinophils. IL-5 also enhances IL-4-induced IgE production (Pene et al., 1988). IL-13 performs many of the same biological functions as IL-4 (Wynn, 2003). GATA-3 plays a critical role in Th2 cell differentiation, Th2 cytokine expression and memory (Barnes, 2008). GATA-3 regulates Th2 cytokine expression not only at the transcription level by direct binding to the promoter of IL-4, IL-5, and IL-13 genes, but also by involvement in remodeling of the chromatin structures (Barnes, 2008). GATA-3 mRNA expression was found to be significantly increased in airways of asthmatics, and the number of T cells in bronchoalveolar lavage fluids (BALF) expressing GATA-3 transcripts was correlated significantly with airway hyperresponsiveness (Nakamura et al., 1999).
Although progress in our understanding of the pathogenesis of asthma and allergic diseases has significantly increased in the past two decades, it is still very difficult to control severe asthma. Most asthmatic children still suffer acute exacerbations of asthma, and relapses of asthma after therapy is withdrawn is common (Akinbami et al., 2009; Sheth et al., 2005). In addition, most asthma and allergy drugs, such as corticosteroids and long-acting β-agonists, have undesirable side effects, and overuse can lead to worsening of disease (Sheth et al., 2005). Thus, there is an urgent need to develop novel approaches for treatment of asthma. Traditional Chinese Medicines (TCM) has a long history of treating asthma in Asia. Glycyrrhiza uralensis, common name licorice, is widely used in traditional Chinese Medicine for its properties of relieving cough and reducing asthma symptoms (Zheng, 2000). In 1998, Glycyrrhiza uralensis was reported to decrease the airway responsiveness in asthma patients (Wang et al., 1998). A recently developed Glycyrrhiza uralensis -containing formula, Anti-asthma Herbal Medicine Intervention (ASHMI), has been shown to improve lung function and reduce airway hyperresponsiveness (AHR) in allergic asthma (Wen et al., 2005). This formula abolished both the early-phase and the late-phase airway responses in an animal model (Zhang et al., 2010).
The major chemical constituents in Glycyrrhiza uralensis are triterpenoids and flavonoids (Zhang and Ye, 2009). Pharmacological activities of several Glycyrrhiza uralensis derived compounds have been demonstrated over the last three decades. Licochalcone A has been reported to exhibit anti-inflammatory activity (Cui et al., 2008). Glycyrrhizin, one of the major triterpenoids, has been reported to reduce airway inflammation in a mouse model (Ram et al., 2006). However, there has been no previous report of effects of Glycyrrhiza uralensis flavonoids on Th2 responses, the key pathological mechanisms of asthma. We previously isolated 5 flavonoids from Glycyrrhiza uralensis, and compared the effects of these compounds to that of glycyrrhizin on secretion of eotaxin-1 by human fetal lung fibroblasts (HFL-1) (Jayaprakasam et al., 2009). The flavonoids liquiritigenin, isoliquiritigenin, and 7, 4’-dihydroxyflavone (7, 4’-DHF) had greater efficacy in suppression of eotaxin secretion than did glycyrrhizin (Jayaprakasam et al., 2009). Flavonoids are widely distributed in foods, fruits, vegetables, and beverages. Most flavonoids are safe for human use. Flavonoids have been demonstrated to have diverse clinically useful actions: anti-inflammatory, antineoplastic, antithrombogenic, antiosteoporotic, and antiviral activities (Nijveldt et al., 2001).
Considering the anti-inflammatory effect on eotaxin secretion and safety of these three flavonoids isolated from Glycyrrhiza uralensis, we hypothesized that these flavonoids may have anti-Th2 inflammatory effects in vitro and in vivo and be potential candidates for clinical use. To test this possibility, we tested the effects of liquiritigenin, isoliquiritigenin, and 7, 4’-DHF on effector memory Th2 cells (D10.G.4.1), a classical Th2 clone, and in a mouse model of allergic asthma.
METHODS
Reagents and mice
ASHMI and Glycyrrhiza uralensis water extract were manufactured by the Sino-Lion Pharmaceutical Company (a GMP(Good Manufacturing Practice) certified facility) in Weifang, China as described previously (Kelly-Pieper K et al., 2009). Composition, quality control and chemical analyses have been described in detail previously (Kelly-Pieper K et al., 2009; Zhang et al., 2010) following FDA Guidance for Botanical Product Industry, Chemistry, Manufacturing, and Control Data (The US Food and Drug Administration (FDA) and Center for Drug Evaluation and Research, 2004).
Glycyrrhiza uralensis flavonoids, isoliquiritigenin, 7,4’-DHF, and liquiritigenin, were isolated as previously described (Jayaprakasam et al., 2009). HPLC grade acetonitrile, formic acid and methanol were purchased from Fisher Scientifics (Pittsburgh, PA). Conalbumin (CA) was purchased from Sigma-Aldrich (St Louis, MO).
Six-week-old female BALB/c mice were purchased from the Jackson Laboratory (Bar Harbor, ME). All animal experiments were conducted according to ethical guidelines of Mount Sinai School of Medicine, New York.
Th2 clone D10.G4.1 cell culture and in vitro treatment experiments
D10. G4.1 (D10 cells), an conalbumin (CA)-specific Th2 clone purchased from American Type Culture Collection (ATCC, Manassas, VA), was originally generated by Kaye et al. in 1983 (Kaye et al., 1983). D10 cells exhibit the effector memory CD4 T cell phenotype and functions (Li et al., 2000; Li et al., 1998; Srivastava et al., 2004). D10 cells produce robust Th2 cytokines upon CA stimulation. This Th2 clone has been widely used in numerous previous studies, including ours, to characterize the role of polarized Th2 cells in the pathogenesis of allergic reactions and to test various approaches to modulation of the Th2 phenotype (Li et al., 2000; Li et al., 1998; Srivastava et al., 2004; Szabo et al., 2000). The antigen presenting cells (APCs) were irradiated spleen cells from syngeneic AKR mice prepared according to the ATCC protocol and as described previously (Srivastava et al., 2004). In brief, splenocytes were prepared by gently mincing the spleens with 2 sterile slides (Fisher Scientific, Pittsburgh, PA), lysing of red blood cells with lysing buffer (ACK lysis buffer, Gibco, Grand Island, NY). After extensive washing, cells were suspended in complete cell culture medium (RPMI 1640 containing 10% FCS, 1% penicillin-streptomycin, and 10% rat growth factor) and irradiated at 393 rads/min for 12.7 mins for total 5000 rads.
D10 cells were co-cultured with APCs in complete culture media in the ratio of 1:5 and CA (100μg/mL). After three days, the cells were split into two flasks. Seven days later, D10 cells were collected. D10 cells (0.25 × 106) and APCs (1.25 × 106) were plated onto 24-well plates, stimulated with 50 μg/mL CA (Li et al., 1998) and cultured with or without flavonoids, isoliquiritigenin, 7,4’-DHF, or liquiritigenin at varying concentrations. After 72 hours, supernatants were collected and IL-4 and IL-5 levels were determined using an ELISA kit (BD Biosciences, San Diego, CA) according to the manufacturer’s instructions. The IC50 values were calculated using non-linear regression analysis by GraphPad Prism 4.01 (GraphPad Software Inc., San Diego, CA).
Analysis of Glycyrrhiza uralensis flavonoids by Liquid Chromatography-Mass Spectrometry (LC-MS)
Flavonoid analysis was accomplished using a Waters 2695 HPLC system (Waters Corporation, Milford, MA) coupled to a Waters’ Micromass LCT Premier mass spectrometer. 100 μL of 1.0 mg/mL of each flavonoid (isoliquiritigenin, liquiritigenin, 7, 4’-DHF) was added into 1.0 mL of ASHMI solution (33.34 mg/mL). The mixture was separated on an analytical Column: Zorbax® SB C18 (5.0μm, 4.6 × 150mm) (Agilent Technologies, Santa Clara, CA) with 0.1% formic acid as mobile phase A and acetonitrile with 0.1% formic acid as mobile phase B. A gradient program was used as follows: 0-75min, linear change from A-B (98:2, v/v) to A-B (52:48, v/v). A 15 min equilibration time was used before each HPLC run. The flow rate was set at 1.0 mL/min. The UV spectra were recorded between 200 and 460nm. The detection wavelength was set at 254nm.
Mass spectrometry was performed on a Time-of-Flight mass spectrometer (Micromass LCT Premier, Waters Corporation). The parameters were set as: Capillary Voltage: 3200 v; Cone Voltage: 15 v; Aperture I: 25 v; Desolvation Temperature: 300 °C; Source Temperature: 100 °C; Desolvation gas: 500 L/h; Nebulizing gas: 30 L/h; Ionization mode: Electrospray; Positive Ion Acquisition Range: m/z 50-1000. The results were collected and analyzed by Empower and Masslynx software.
D10 cell Proliferation assay
Cell proliferation was assessed by [3H] thymidine incorporation. 0.05× 106 D10 cells and 0.25× 106 APCs in 0.2mL were seeded in the wells of a 96 well plate in the presence or absence of 7,4’-DHF at 6.25 μg/mL with or without CA (50 μg/mL). The plates were incubated at 37°C 5% CO2 for 72 hours. 1 μCi/mL [3H]-Thymidine was then added into each well. The cells were incubated for another 18 hr and harvested. The amount of radioactivity incorporated was counted in a β-scintillation counter. The results are expressed as counts per minute (cpm).
D10 Cell Viability assay
Cell viability was evaluated using MTT assay and trypan blue exclusion assay. D10 Cells were seeded in 96-well-plates as described above. Cells were treated with 7, 4’-DHF at 6.25 μg/mL with CA (50 μg/ml) stimulation and incubated at 37°C 5% CO2 for 24 hours. 20 μL of 1.0 mg/mL 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide (MTT) solution was added to each well and incubated with the cells for 4 hours until a purple precipitate was visible. The plate was then centrifuged at 1200 rpm for 10 mins. The supernatant was carefully removed without disturbing the cells. 200 μL of dimethylsulfoxide (DMSO) was then added into each well and the plate was left in the dark at room temperature for 2 hours. Absorbance was measured at a wavelength of 595nm. The percent viability was calculated with respect to the cells in medium only.
Western-blot analysis Assay of D10 cells GATA3 expression
GATA-3 expression was analyzed by Western blot analysis using a monoclonal anti-GATA-3 antibody (HG3-31, Santa Cruz Biotechnology, Santa Cruz, CA). In brief, 1× 107 D10 cells were incubated with 5× 107 APC and treated with 7, 4’-DHF (from 0.2 μg/mL to 6.25 μg/mL) for 72 hours. Cells were then collected and lysed. After denaturation at 95°C for 5 min, 20μg of total proteins were separated by 10% SDS-PAGE and transferred onto a nitrocellulose membrane. After blotting, the membranes were incubated with anti-GATA-3 (HG-31) antibody and β-actin, respectively. After washing three times to remove the primary antibody, the membranes were incubated with secondary antibody, horseradish peroxidase-conjugated anti-mouse IgG. The proteins were detected using ECL Western detection system (Amersham Pharmacia Biotech, Piscataway, NJ).
Extraction of RNA and real time reverse transcriptase-polymerase chain reaction (real time-PCR)
Total RNA was isolated from D10 cells treated with 7,4’-dihydroxyflavone using Trizol reagent (Life Technologies, Grand Island, NY) following the manufacture’s instruction. The total RNA concentrations were measured by triplicate optical density (OD) readings (Bio-Rad SmartSpect 3000; Bio-Rad, Hercules, CA). The reverse transcription was performed using the ImProm-II™ Reverse Transcriptase (Promega Corporation, Madison, WI) as described by the manufacturer’s instruction. The polymerase chain reaction was performed by using Maxima™ SYBR Green qPCR Master Mix (2X) kit (Fermentas, Glen Burnie, MD). PCR were started at 95°C for 10 mins followed by 40 cycles for IL-4 and Gusb (housekeeping gene). The temperature profile of each cycle is: 95 °C for 15 seconds, at 60 °C for 30 seconds, and at 72 °C for 30 seconds. The following primers were used: IL-4 forward: TCCTCACAGCAACGAAGAAC; IL-4 reverse: TGCAGCTCCATGAGAACACT; Gusb forward: AGTATGGAGCAGACGCAATC; Gusb reverse: CTCTCCGACCACGTATTCTT. All primers were synthesized by Sigma-Aldrich Corporation (St. Louis, MO).
OVA sensitization / challenge and 7, 4’-DHF treatment in murine model
To generate an allergic asthma model, female BALB/c mice were sensitized weekly intraperitoneally (i.p.) with 200μg OVA (TypeV, Sigma-Aldrich) absorbed on 2mg alum (Pierce Biotechnology Inc., Rockford, IL) in 0.4 mL phosphate-buffered saline for 2 weeks (Fig. 1). After the last sensitization, 5 intratracheal (i.t.) challenges were performed on days 14, 21, 28, 45 and 46 with 100μg OVA in 0.05mL of phosphate-buffered saline. To study the effect of 7, 4’-DHF on the Th2 responses in vivo in this asthma model, eight mice received 6.0 μg of 7, 4’-DHF in 1mL of 0.1% DMSO in water intragastrically (i.g.), divided into two feedings daily for one month beginning 24 hours after the first i.t. challenge. The dose of ASHMI used in previous mouse model studies was 9~10 mg per mouse per day based on a human equivalent dose (Zhang et al., 2010). Based on the percentage of 7, 4’-DHF in ASHMI as analyzed by LC-MS, 9 mg of ASHMI contains approximately 3.0 μg of 7, 4’-DHF. In a preliminary study, we tested 3.0 μg and 6.0 μg daily doses of 7, 4’-DHF and found that 3.0 μg showed minimal inhibitory effect on OVA-induced Th2 responses (data not shown). For this study, we treated 8 mice with a 6.0 μg daily dose of 7,4’-DHF (Fig.1). Six OVA sensitized / challenged mice receiving 0.5ml of 0.1% DMSO in water twice daily for one month served as sham control mice for the experiment. Six mice not sensitized / challenged or treated served as the naïve control group. Analysis was performed 2 days after the last i.t. challenge.
Figure 1. Mouse model protocol.
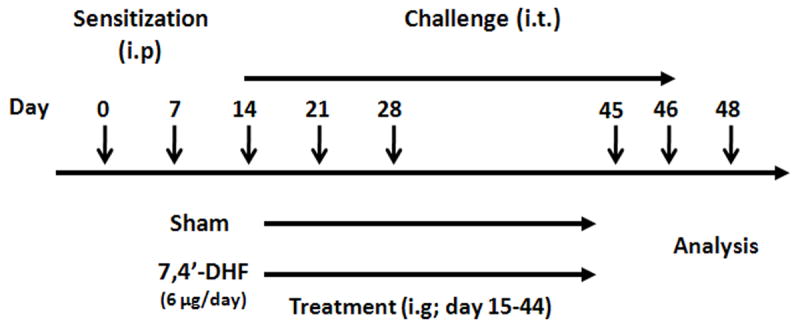
The protocol of sensitization, challenge, and treatment with 7, 4’-DHF is diagrammed. Mice were sensitized twice intraperitoneally (i.p.) and challenged intratracheally (i.t.) on day 14, 21, 28, 45 and 46 with 100μg OVA in 0.05mL of phosphate-buffered saline. Twenty-four hours after the first i.t. challenge, mice were treated with 6.0 μg of 7, 4’-DHF intragastrically or 0.1 % DMSO (Sham group) (i.g. twice daily).
Lung histology
The lungs were surgically removed following the collection of BALF. The left lower lobe was fixed in neutral buffered formaldehyde. Five micrometer paraffin sections were stained with hematoxylin and eosin (HE) for evaluation of inflammation and periodic acid-Schiff (PAS) for evaluation of goblet cells. The percentage of PAS -positive cells were determined (number of PAS-positive cells divided by the total epithelial cell number) in at least 6 randomly selected bronchioles per animal as previously described (Zhang et al., 2010).
Lung cell culture and cytokine measurement
Lungs from 4 mice / group were finely chopped, and digested in Hanks Balanced Salt Solution supplemented with 3 mg/mL collagenase (type V, Sigma Aldrich), 0.1 mg/mL DNase (Roche Applied Science, Indianapolis, IN) and 1% penicillin-streptomycin for 1hour in a tissue culture incubator (Salek-Ardakani et al., 2003). Single cell preparations were obtained by passing cells through a syringe several times. Thereafter the lung cells (4×106/ml) were cultured in RPMI-1640 complete media (RPMI 1640 supplemented with 10% heat-inactivated FBS, 1% L-glutamine, 50 μM 2-mercaptoethanol, 1% streptomycin-penicillin) either with OVA (50 μg/mL), concanavalin A (ConA; 2.5 μg/mL) or medium alone for 72 hours. IL-4, IL-5, IL-13 and IFN-γ levels in the supernatant were determined by ELISA (BD biosciences for all except IL-13; R & D systems).
Measurement of late-phase airway responsiveness and BAL cell differential counts
Forty-eight hours after the final i.t. challenge, late phase airway responsiveness to acetylcholine was measured as the time-integrated change in peak airway pressure (APTI) as previously described (Li et al., 1996). After APTI measurements, the lungs were lavaged and the total numbers of cells in the BALF were determined. Cytospin slides were prepared and differential counts were obtained by counting 500 cells/slide after staining with Hema 3 (Fisher Scientific, Pittsburgh, PA).
Measurement of serum OVA-specific IgE antibodies
Blood was collected after the APTI measurements and the separated sera were stored at -80°C until ELISA antibody measurements were performed as described previously (Zhang et al., 2010).
Statistical analysis
Statistical analyses were performed using one-way ANOVA, pair-wise and multiple-comparison testing between groups, as well as the Mann–Whitney rank sum test. A P-value≤0.05 was considered to be statistically significant. All statistical analyses were performed using Sigma Stat software (Systat Software Inc., Chicago, IL).
RESULTS
ASHMI formula contains isoliquiritigenin, 7, 4’-DHF, and liquiritigenin
Previously, we isolated 5 flavonoids from Glycyrrhiza uralensis, one of the herbal constituents in ASHMI (Jayaprakasam et al., 2009). Three of these flavonoids, isoliquiritigenin, 7,4’-DHF, and liquiritigenin (Fig. 2A) inhibited eotaxin-1 production by lung fibroblasts (Jayaprakasam et al., 2009). We used LC-MS to confirm that these compounds were present in the ASHMI extract and to estimate their concentrations using a method described previously (Fernand et al., 2008). 100 μL of 1.0 mg/mL of either isoliquiritigenin, 7, 4’-DHF, or liquiritigenin was added to 1.0 mL of a 33.34 mg/mL solution of ASHMI. These mixtures and ASHMI alone were loaded onto the LC-MS system. The added compounds produced peaks with retention times and molecular weights identical to three of the peaks in ASHMI. The peak areas of each compound in the mixtures and ASHMI were measured. The isoliquiritigenin peak area increased from 680 to 4572; the 7, 4’-DHF peak area increased from 320 to 3132; the liquiritigenin, peak area increased from 647 to 2307 (Fig. 2B). These results confirmed that ASHMI contained these three Glycyrrhiza uralensis flavonoids.
Figure 2. The presence in ASHMI of flavonoids isolated from Glycyrrhiza uralensis.
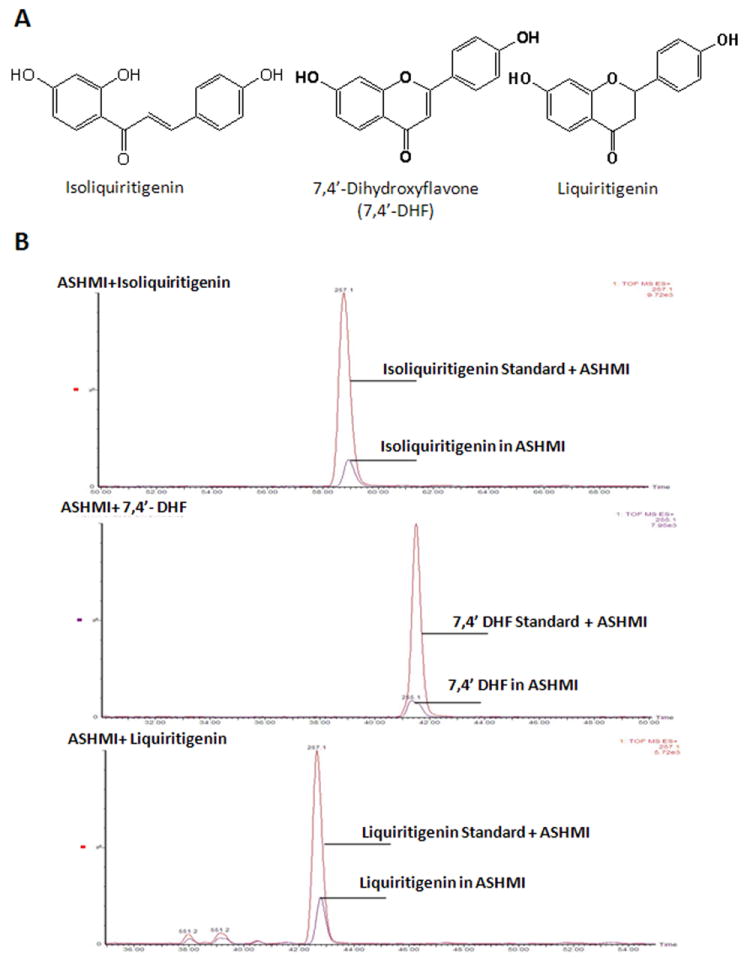
A. Chemical structures of flavonoids (isoliquiritigenin, 7, 4’-DHF, and liquiritigenin) isolated from Glycyrrhiza uralensis. B. Chromatogram of the peak intensities of each compound present in ASHMI and in ASHMI mixed with 100 μg of each compound. Purple peaks, each flavonoid in ASHMI; Red peaks, each flavonoid in the mixture of ASHMI and 100 μg pure flavonoid.
Quantitative analysis of each flavonoid in ASHMI was performed by calculating the ratio of peak area and the amount of each standard compound injected. ASHMI contained 0.052 % isoliquiritigenin, 0.034 % 7, 4’-DHF, and 0.116 % liquiritigenin.
Isoliquiritigenin, 7, 4’-DHF, and liquiritigenin inhibited Th2 cytokine synthesis
To determine whether the isolated flavonoids suppress Th2 cytokine synthesis, IL-4 and IL-5 levels in D10 cell culture supernatants following treatment with each flavonoid were determined by ELISA. CA-stimulated IL-4 (Fig. 3A) and IL-5 (Fig. 3B) production were markedly suppressed by all three flavonoids in a concentration-dependent manner without changing the cell viabilities (Fig. 3C). The IC50 (concentration producing 50% inhibition) values for each of the flavonoids are shown in Table 1. These results show that 7, 4’-DHF is the most potent Th2 inhibitor. The following studies, therefore, focused on 7, 4’-DHF.
Figure 3. IL-4 and IL-5 levels in culture supernatants of D10 cells treated with Glycyrrhiza uralensis flavonoids.
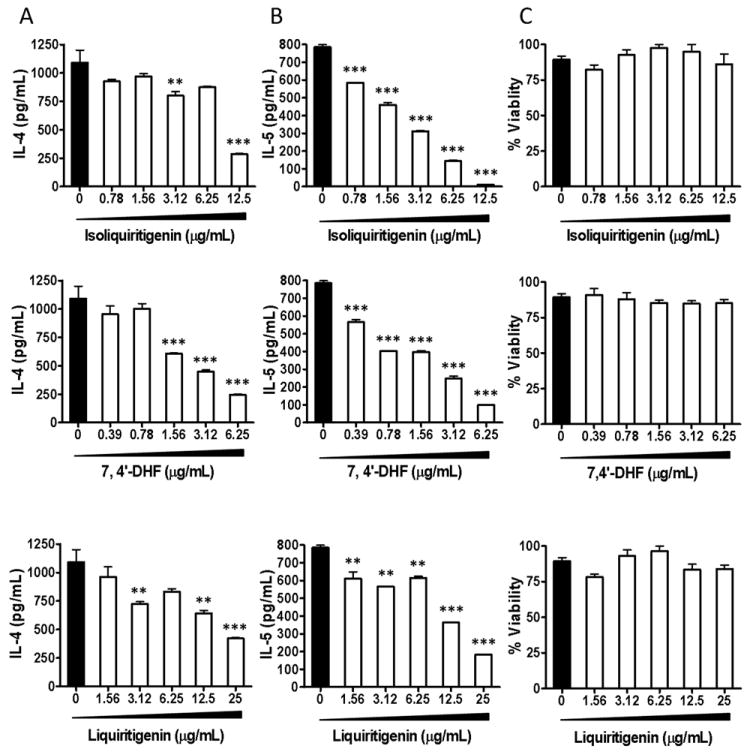
D10 cells (0.25 × 106) and APC (1.25 × 106) were stimulated with CA and incubated in the presence of either isoliquiritigenin, 7, 4’-DHF, or liquiritigenin at the concentrations shown. Supernatants were collected and analyzed by ELISA for IL-4 (A) and IL-5 (B) production. Cell viability was analyzed using trypan blue exclusion assay (C). ** p<0.05, *** p<0.001.
Table 1.
IC50 values of isoliquiritigenin, 7, 4’-DHF, and liquiritigenin against IL-4 and IL-5 production.
| Name | IC50 value (μg/mL) (mean ± SD)
|
|
|---|---|---|
| IL-4 | IL-5 | |
| Isoliquiritigenin | 9.0±2.8 | 2.0±0.2 |
| 7,4’-DHF | 2.3±1.0 | 1.1±0.1 |
| Liquiritigenin | 16.5±5.3 | 10.8±0.5 |
7, 4’-DHF inhibited memory Th2 cell proliferation in a non toxic manner
We next examined whether 7, 4’-DHF affected D10 cell proliferation. [3H]-Thymidine incorporation in D10 cells was significantly decreased in the presence of 6.25 μg/mL 7, 4’-DHF (Fig. 4A).
Figure 4. D10 cells proliferation, cell viability, GATA-3 protein and IL-4 mRNA expression in response to 7, 4’-DHF in vitro treatment.
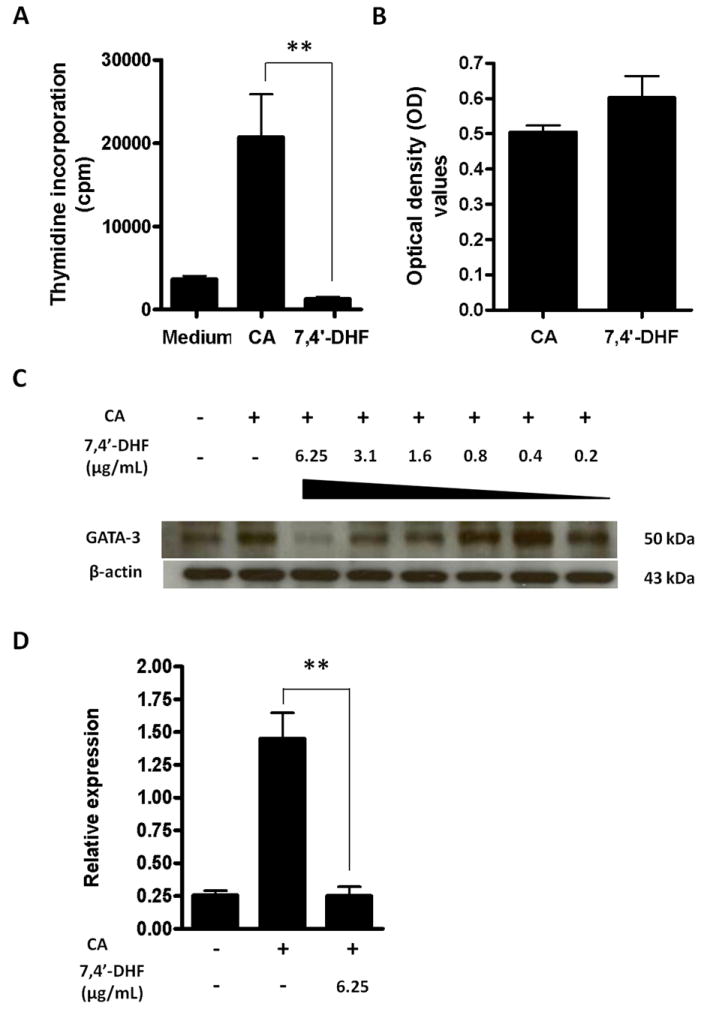
A. Cell proliferation ([3H] thymidine incorporation) assay in D10 cells with or without 7, 4’-DHF at 6.25 μg/mL. Experimental data are expressed as the mean ± SEM of four independent experiments; B. Cell viability assay of 7, 4’-DHF at 6.25 μg/mL was assessed by MTT (3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyltetrazolium bromide) assay. Results are expressed as optical density (OD); C. Western blot of GATA-3 in D10 cells in the presence of different concentrations of 7, 4’-DHF. GATA-3 expression was determined using monoclonal antibodies specific for GATA-3. D. Relative mRNA expression of IL-4 by D10 cells with treatment of 7, 4’-DHF. ** p<0.05.
In order to assess potential cyto-toxicity, antigen-primed D10 cells were treated with 7, 4’-DHF at 6.25 μg/mL for 24 hours and then incubated with MTT. The percent of viable cells was not different from the control group (Fig. 4B).
7, 4’-DHF suppressed memory Th2 cell transcription factor GATA-3 expression and decreased IL-4 mRNA levels in vitro
Since GATA-3 modulates Th2 cytokine production, and because ASHMI suppresses GATA-3 expression (unpublished data), we hypothesized that 7, 4’-DHF might also have this effect. D10 cells were stimulated with CA in the presence and absence of concentrations of 7, 4’-DHF ranging from 0.2 to 6.25 μg/mL. GATA-3 protein expression was markedly suppressed at 6.25 μg/mL and moderately suppressed at 3.1 and 1.6 μg/ml (Fig. 4C). To further determine the functional consequence of suppression of GATA-3 by 7, 4’-DHF, we investigated its effect on IL-4 mRNA levels. RNA was extracted from D10 cells stimulated with CA in the presence and absence of 7, 4’-DHF at 6.25 μg/mL. IL-4 mRNA expression was significantly reduced by 7, 4’-DHF (Fig. 4D, p<0.05).
7, 4’-DHF treatment reduced airway inflammation and the number of goblet cells
Histological analysis showed that sham treated mice present significant increases in the number of goblet cells compared with naïve mice, whereas only a few goblet cells were present in the airways of 7, 4’-DHF treated mice (Fig. 5A). The percentage of PAS-positive goblet cells among epithelial cells of 7, 4’-DHF treated mice was significantly lower than that of sham treated mice (Fig. 5B). Sham treated mice showed increased accumulation of inflammatory cells compared to naïve mice, while 7, 4’-DHF treatment reduced the number of inflammatory cells around the airways (Fig. 5A).
Figure 5. Lung histology.
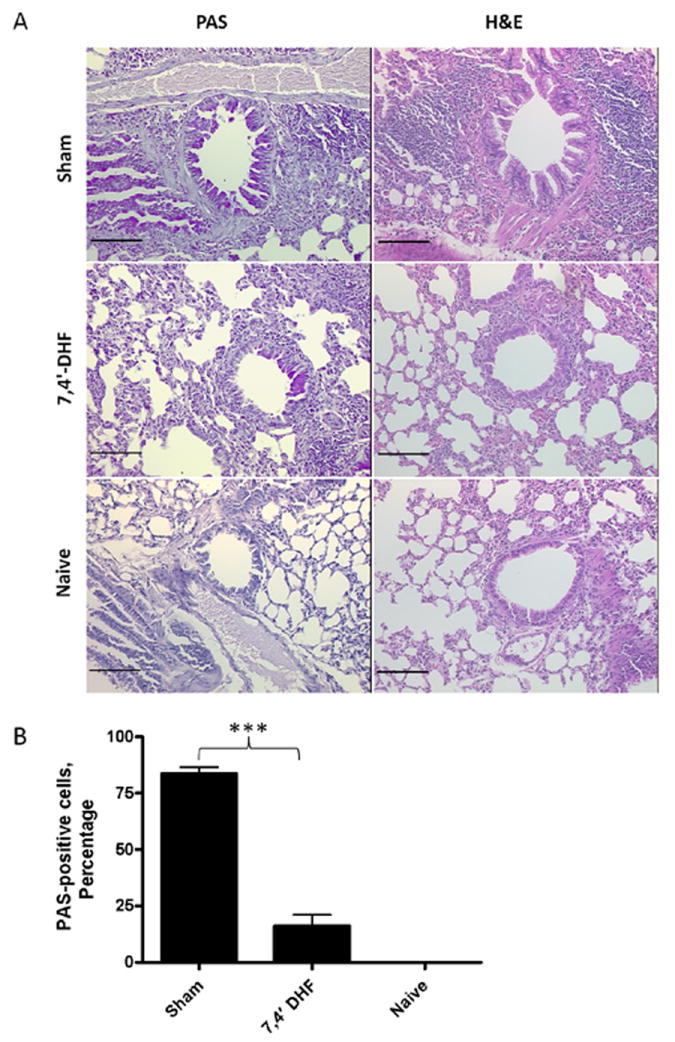
A. PAS and H&E stained sections of mouse airways. Bars=100μm. B. Percentage of PAS positive goblet cells in mouse airways. (*** p<0.001).
7, 4’-DHF treatment inhibited Th2 responses and increased IFN-γ level in vivo
We next investigated whether 7, 4’-DHF inhibited Th2 responses in a murine model of asthma by measuring Th2 cytokine production in lung cell cultures in response to antigen stimulation. Lung cells isolated from sham-treated mice produced significantly higher IL-4, IL-5 and IL-13 levels following OVA stimulation than those from naïve mice (Fig. 6A-C, p<0.001). However, cultures of lung cells from 7, 4’-DHF-treated mice (6.0 μg/day) showed significantly reduced IL-4 and IL-13 levels, but not IL-5 levels, compared to cultured cells from sham-treated mice (Fig. 6A-C). IFN-γ production by lung cells from mice treated with 7, 4’-DHF treatment was elevated compared with the sham group [7, 4’-DHF vs. Sham, p<0.001] (Fig. 6D). Furthermore, 7, 4’-DHF-treated mice had significantly lower serum IgE levels than sham-treated mice (Fig. 6E).
Figure 6. The immunomodulatory effect of 7, 4’-DHF in vivo treatment (6.0 μg/day) on lung cell culture cytokines and serum IgE.
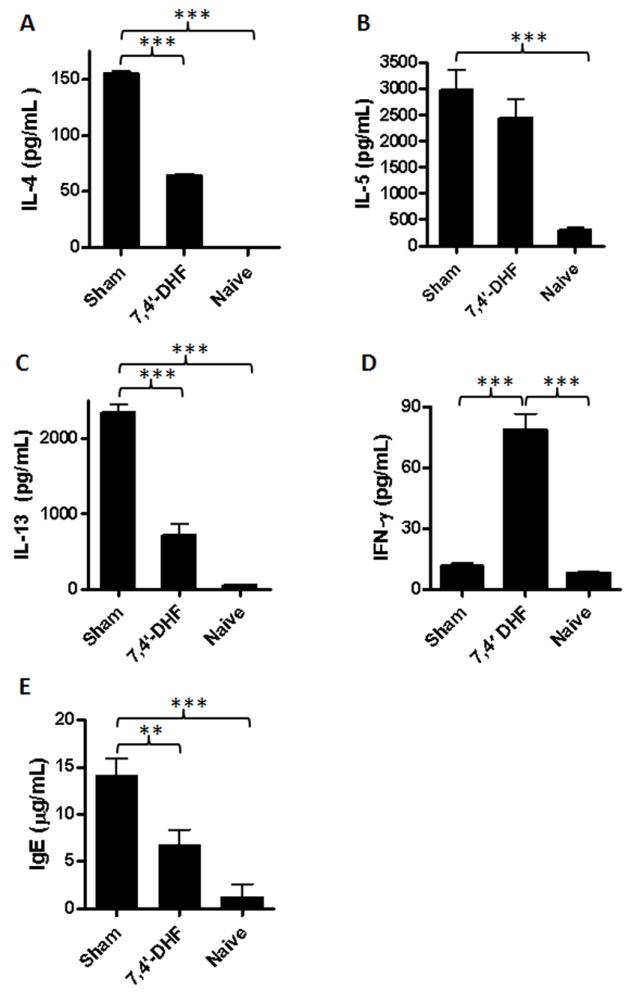
Pooled lung cells were cultured in complete RPMI media either with media alone, OVA or Con A and incubated for 72 hours at 37°C. The supernatants were analyzed for cytokines by ELISA. A. IL-4 (treated group (n=8) :63.98±1.75 pg/mL; sham group (n=6): 155.61±1.9 pg/mL); B. IL-5 (treated group: 2433.02 ± 349 pg/mL; sham group: 2975.66 ± 381 pg/mL); C. IL-13 (treated group: 721.25 ± 142 pg/mL; sham group: 2345.12 ± 104 pg/mL); D. IFN-γ (treated group: 78.91±13.35 pg/mL; sham group: 11.52±2.63pg/mL); E. OVA-specific IgE levels were significantly reduced in treated mice (n=4) compared to sham (n=5). ** p<0.05, *** p<0.001.
7, 4’-DHF treatment decreased antigen-induced pulmonary inflammation in an animal model
Sham-treated mice had an increased number of leukocytes and a higher percent of eosinophils in BALF, and higher APTI values as compared to naïve mice (Fig. 7A-C, p<0.005-0.001). Treatment with 7, 4’-DHF (6.0 μg/day) significantly reduced the percent of eosinophils compared to the sham treatment (p<0.001). There was also a trend of reduced total leukocyte count and APTI values compared to the sham-treated mice, but the difference did not reach statistical significance (p=0.058).
Figure 7. Effect of 7, 4’-DHF treatment (6.0 μg/day) on airway inflammation and AHR in a murine model of allergic asthma.
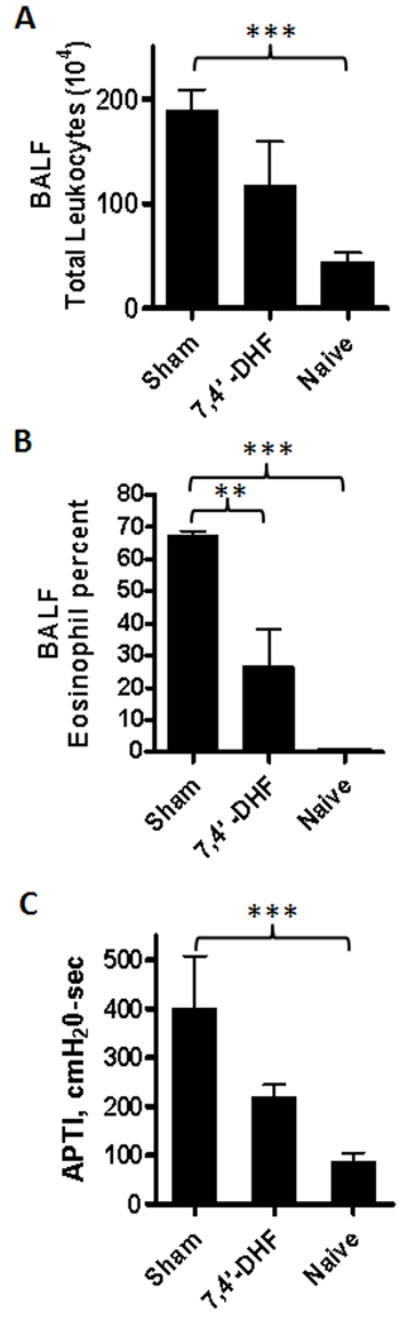
A. Total leukocytes in BALF; B. Eosinophil percent in BALF. In cytospun slides of BALF stained with Hema 3 kit, percent eosinophils were significantly reduced in treated group (n=8) compared to sham (n=6) (p= 0.016); C. Invasive Airway Pressure Time Index (APTI) values. APTI with acetylcholine challenge showed possible suppression of AHR in the treated group (n=8) (p= 0.059). **p<0.05, *** p<0.001.
DISCUSSION
Flavonoids, isoliquiritigenin, 7, 4’-DHF, and liquiritigenin, isolated from Glycyrrhiza uralensis, a component of ASHMI, dramatically suppressed the secretion of eotaxin-1 by human fetal lung fibroblasts (HFL-1) (Jayaprakasam et al., 2009). However, whether these three active compounds were extracted during the preparation of the ASHMI formula was unknown. Therefore, we first confirmed that ASHMI contained these three Glycyrrhiza uralensis flavonoids. By adding 100 μg of each flavonoid standard to ASHMI and loading the mixture onto LC-MS, we observed that the peak area of each flavonoid (identified by comparing the exact molecular weight and retention time) was increased. This method provided evidence of the presence and amount of each compound in ASHMI.
Th2 cytokines play a critical role in orchestrating and perpetuating inflammation in asthmatic airways. Substantial evidence indicates that polarized (effector memory) Th2 cells produce robust quantities of Th2 cytokines in chronic allergic asthma following antigen re-exposure (Epstein, 2006; Salek-Ardakani et al., 2003). These cells often arise early in life and persist after repeated encounters with allergen. In a murine model of chronic asthma, these Th2 memory responses persisted for over a year (~ 50% of murine life expectancy) (Mojtabavi et al., 2002). D10 cells are classical effector memory Th2 cells. Previous research showed that ASHMI significantly inhibited IL-4 production by memory Th2 cells in a non-cytotoxic manner (Srivastava et al., 2004). We thus investigated the inhibitory effects of three flavonoids previously isolated from Glycyrrhiza uralensis on memory Th2 effector cells. Our in vitro study showed that isoliquiritigenin, 7, 4’-DHF, and liquiritigenin significantly suppressed production of Th2 cytokines, IL-4 and IL-5, by antigen stimulated D10 cells. 7, 4’-DHF had the highest potency and did not reduce cell viability of D10 cells at the highest concentration used. 7, 4’-DHF also inhibited D10 cell proliferation to antigen stimulation. GATA-3 is the key regulatory factor for Th2 cell differentiation, and is required for maintenance of the Th2 phenotype (Barnes, 2008). The presence of GATA-3 is also required for Th2 cell proliferation (Barnes, 2008). 7, 4’-DHF reduced GATA-3 levels in a concentration range similar to that which decreased Th2 cytokines. This finding is consistent with the hypothesis that 7, 4’-DHF inhibition of Th2 cytokine production and D10 cell proliferation is through alteration of GATA-3 levels.
Consistent with the in vitro findings, 7, 4’-DHF treatment (6.0 μg/day) of OVA challenged mice significantly inhibited IL-4 and IL-13 production by lung cells to recall OVA stimulation, as well as pulmonary eosinophilic inflammation. Th2-derived cytokines are key factors in IgE synthesis. We found that serum IgE levels were also significantly decreased by 7, 4’-DHF treatment in the murine model. A previous study found that ASHMI reduced serum Th2 cytokine levels, but not Th1 cytokines in asthmatic patients (Wen et al., 2005). In the murine model, ASHMI significantly reduced Th2 cytokines and increased Th1 cytokine production (Srivastava et al., 2010; Zhang et al., 2010). In this study, we also found that 7, 4’-DHF treatment of OVA challenged mice increased production of the Th1 cytokine, IFN-γ. These results demonstrated that like ASHMI, 7, 4’-DHF inhibits memory Th2 response without overall immune suppression. The effects of 7, 4’-DHF on Th2 responses and airway inflammation in the murine asthma model have not been previously reported.
ASHMI has been shown to significantly reduce IL-5 production in our murine model (Zhang et al., 2010). Although, 7, 4’-DHF reduced IL-5 production in vitro, its effects on IL-5 in vivo were not statistically significant, nor was its effect on AHR, despite the fact that the dose tested was double that present in effective doses of ASHMI.
It is not surprising that treatment with a single component of ASHMI, even at double the amount present in the whole herb extract, does not replicate the effect of ASHMI. First, although isoliquiritigenin and liquiritigenin were not as potent as 7, 4’-DHF in suppression of Th2 cytokine production by D10 cells in vitro, they did have activity and may themselves contribute to the effects of ASHMI. Second, ASHMI consists of Glycyrrhiza uralensis and two other herbs, Radix sophorae flavescentis and Ganoderma lucidum (Kelly-Pieper K et al., 2009). Extracts of these two herbs have also been shown to inhibit Th2 cytokine production in vivo (Chen and Lin, 2007; Wen et al., 2004). Third, we previously showed that ASHMI directly inhibited contraction of murine tracheal rings and increased production of the potent smooth muscle relaxant PGI2 by human bronchial smooth muscle cells (Zhang et al., 2010). This action appears to reside in Radix sophorae flavescentis (Data not published), and would contribute to reduction in AHR. Thus, other ASHMI components are required to produce the full in vivo therapeutic effect on asthma.
In summary, we, for the first time, demonstrated that three Glycyrrhiza uralensis flavonoids present in ASHMI, 7, 4’-DHF being the most potent, inhibited memory Th2 cells in vitro. 7, 4’-DHF reduced production of IL-4, IL-5, and cell proliferation without altering cell viability. This compound also strongly suppressed production of GATA-3, a key Th2 cell transcription factor. In a murine model, 7, 4’-DHF reduced some antigen induced Th2 responses including Th2 cytokine and antigen specific IgE production, and airway inflammation, while increasing IFN-γ.
Acknowledgments
Funding source: This work was supported by NIH/NCCAM center grants # P01 AT002647-01 01 to XM Li “Center for Chinese Herbal Therapy (CHT) for Asthma”, Project 1: Effects and mechanisms of Chinese herbal therapy in an asthma model and Project 3: Chemical and biological characteristics of Botanicals to Nan Yang.
Footnotes
Authors’ disclosure statement: Drs. Xiu-Min Li, Hugh Sampson and Ming Chun Wen are named inventors of a patent for the use of ASHMI (PCT/US05/08600 for ASHMI) and have shares of Herbal Spring LLC, which has exclusive rights to the patent. The other authors have no financial interests to disclose.
References
- Akinbami LJ, Moorman JE, Garbe PL, et al. Status of childhood asthma in the United States, 1980-2007. Pediatrics. 2009;123(Suppl 3):S131–S145. doi: 10.1542/peds.2008-2233C. [DOI] [PubMed] [Google Scholar]
- American Academy of Allergy AaI. Asthma statistics 2010 [Google Scholar]
- Barnes PJ. Role of GATA-3 in allergic diseases. Curr Mol Med. 2008;8:330–334. doi: 10.2174/156652408785160952. [DOI] [PubMed] [Google Scholar]
- Blander JM, Sant’Angelo DB, Metz D, et al. A pool of central memory-like CD4 T cells contains effector memory precursors. J Immunol. 2003;170:2940–2948. doi: 10.4049/jimmunol.170.6.2940. [DOI] [PubMed] [Google Scholar]
- Chen ML, Lin BF. Effects of triterpenoid-rich extracts of Ganoderma tsugae on airway hyperreactivity and Th2 responses in vivo. Int Arch Allergy Immunol. 2007;143:21–30. doi: 10.1159/000098222. [DOI] [PubMed] [Google Scholar]
- Cui Y, Ao M, Li W, et al. Anti-inflammatory activity of licochalcone A isolated from Glycyrrhiza inflata. Z Naturforsch C. 2008;63:361–365. doi: 10.1515/znc-2008-5-609. [DOI] [PubMed] [Google Scholar]
- Dutton RW, Bradley LM, Swain SL. T cell memory. Annu Rev Immunol. 1998;16:201–223. doi: 10.1146/annurev.immunol.16.1.201. [DOI] [PubMed] [Google Scholar]
- Epstein MM. Targeting memory Th2 cells for the treatment of allergic asthma. Pharmacol Ther. 2006;109:107–136. doi: 10.1016/j.pharmthera.2005.06.006. [DOI] [PubMed] [Google Scholar]
- Fernand VE, Dinh DT, Washington SJ, et al. Determination of pharmacologically active compounds in root extracts of Cassia alata L. by use of high performance liquid chromatography. Talanta. 2008;74:896–902. doi: 10.1016/j.talanta.2007.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelman FD, Hogan SP, Hershey GK, et al. Importance of cytokines in murine allergic airway disease and human asthma. J Immunol. 2010;184:1663–1674. doi: 10.4049/jimmunol.0902185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayaprakasam B, Doddaga S, Wang R, et al. Licorice flavonoids inhibit eotaxin-1 secretion by human fetal lung fibroblasts in vitro. J Agric Food Chem. 2009;57:820–825. doi: 10.1021/jf802601j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaye J, Porcelli S, Tite J, et al. Both a monoclonal antibody and antisera specific for determinants unique to individual cloned helper T cell lines can substitute for antigen and antigen-presenting cells in the activation of T cells. J Exp Med. 1983;158:836–856. doi: 10.1084/jem.158.3.836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly-Pieper K, Patil SP, Busse P, et al. Safety and tolerability of an antiasthma herbal formula (ASHMI™) in adult asthmatics: a randomized, double-blinded, placebo-controlled, dose escalation phase I study. Journal of Alternative and complementary Medicine. 2009;15:735–743. doi: 10.1089/acm.2008.0543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larche M, Robinson DS, Kay AB. The role of T lymphocytes in the pathogenesis of asthma. J Allergy Clin Immunol. 2003;111:450–463. doi: 10.1067/mai.2003.169. [DOI] [PubMed] [Google Scholar]
- Li B, Yu H, Zheng W, et al. Role of the guanosine triphosphatase Rac2 in T helper 1 cell differentiation. Science. 2000;288:2219–2222. doi: 10.1126/science.288.5474.2219. [DOI] [PubMed] [Google Scholar]
- Li XM, Chopra RK, Chou TY, et al. Mucosal IFN-gamma gene transfer inhibits pulmonary allergic responses in mice. J Immunol. 1996;157:3216–3219. [PubMed] [Google Scholar]
- Li XM, Schofield BH, Wang QF, et al. Induction of pulmonary allergic responses by antigen-specific Th2 cells. J Immunol. 1998;160:1378–1384. [PubMed] [Google Scholar]
- Messi M, Giacchetto I, Nagata K, et al. Memory and flexibility of cytokine gene expression as separable properties of human T(H)1 and T(H)2 lymphocytes. Nat Immunol. 2003;4:78–86. doi: 10.1038/ni872. [DOI] [PubMed] [Google Scholar]
- Mojtabavi N, Dekan G, Stingl G, et al. Long-lived Th2 memory in experimental allergic asthma. J Immunol. 2002;169:4788–4796. doi: 10.4049/jimmunol.169.9.4788. [DOI] [PubMed] [Google Scholar]
- Nakamura Y, Ghaffar O, Olivenstein R, et al. Gene expression of the GATA-3 transcription factor is increased in atopic asthma. J Allergy Clin Immunol. 1999;103:215–222. doi: 10.1016/s0091-6749(99)70493-8. [DOI] [PubMed] [Google Scholar]
- Nijveldt RJ, van NE, van Hoorn DE, et al. Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr. 2001;74:418–425. doi: 10.1093/ajcn/74.4.418. [DOI] [PubMed] [Google Scholar]
- Pene J, Rousset F, Briere F, et al. Interleukin 5 enhances interleukin 4-induced IgE production by normal human B cells. The role of soluble CD23 antigen. Eur J Immunol. 1988;18:929–935. doi: 10.1002/eji.1830180615. [DOI] [PubMed] [Google Scholar]
- Ram A, Mabalirajan U, Das M, et al. Glycyrrhizin alleviates experimental allergic asthma in mice. Int Immunopharmacol. 2006;6:1468–1477. doi: 10.1016/j.intimp.2006.04.020. [DOI] [PubMed] [Google Scholar]
- Reinhardt RL, Khoruts A, Merica R, et al. Visualizing the generation of memory CD4 T cells in the whole body. Nature. 2001;410:101–105. doi: 10.1038/35065111. [DOI] [PubMed] [Google Scholar]
- Salek-Ardakani S, Song J, Halteman BS, et al. OX40 (CD134) controls memory T helper 2 cells that drive lung inflammation. J Exp Med. 2003;198:315–324. doi: 10.1084/jem.20021937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sallusto F, Lenig D, Forster R, et al. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- Sheth A, Reddymasu S, Jackson R. Worsening of Asthma with Systemic Corticosteroids. J Gen Intern Med. 2005 doi: 10.1111/j.1525-1497.2005.00290.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srivastava K, Teper AA, Zhang TF, et al. Immunomodulatory effect of the anti-asthma Chinese herbal formula, MSSM-002 on Th2 cells. J Allergy Clin Immunol. 2004;113:268–276. doi: 10.1016/j.jaci.2003.10.062. [DOI] [PubMed] [Google Scholar]
- Srivastava K, Zhang T, Yang N, et al. Anti-Asthma Simplified Herbal Medicine Intervention-induced long-lasting tolerance to allergen exposure in an asthma model is interferon-gamma, but not transforming growth factor-beta dependent. Clin Exp Allergy. 2010 doi: 10.1111/j.1365-2222.2010.03545.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szabo SJ, Kim ST, Costa GL, et al. A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell. 2000;100:655–669. doi: 10.1016/s0092-8674(00)80702-3. [DOI] [PubMed] [Google Scholar]
- The US Food and Drug Administration (FDA), Center for Drug Evaluation and Research. Guidance for Industry Botanical Drug Products. 2004 Revised. [Google Scholar]
- Wang H, Chang B, Wang B. The effect of herbal medicine including astragalus membranaceus (fisch) bge, codonpsis pilosula and glycyrrhiza uralensis fisch on airway responsiveness. Zhonghua Jie He He Hu Xi Za Zhi. 1998;21:287–288. [PubMed] [Google Scholar]
- Wen MC, Huang CK, Srivastava KD, et al. Ku-Shen (Sophora flavescens Ait), a single Chinese herb, abrogates airway hyperreactivity in a murine model of asthma. J Allergy Clin Immunol (Abstract) 2004;113:218. [Google Scholar]
- Wen MC, Wei CH, Hu ZQ, et al. Efficacy and tolerability of anti-asthma herbal medicine intervention in adult patients with moderate-severe allergic asthma 23. J Allergy Clin Immunol. 2005;116:517–524. doi: 10.1016/j.jaci.2005.05.029. [DOI] [PubMed] [Google Scholar]
- Wynn TA. IL-13 effector functions. Annu Rev Immunol. 2003;21:425–56. doi: 10.1146/annurev.immunol.21.120601.141142. Epub;%2001 Dec;%19.: 425-456. [DOI] [PubMed] [Google Scholar]
- Zhang Q, Ye M. Chemical analysis of the Chinese herbal medicine Gan-Cao (licorice) J Chromatogr A. 2009;1216:1954–1969. doi: 10.1016/j.chroma.2008.07.072. [DOI] [PubMed] [Google Scholar]
- Zhang T, Srivastava K, Wen MC, et al. Pharmacology and immunological actions of a herbal medicine ASHMI on allergic asthma. Phytother Res. 2010;24:1047–1055. doi: 10.1002/ptr.3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng XY. Pharmacopoeia of the People’s Republic of China. English ed 2000 [Google Scholar]


