Abstract
Selenium (Se) is an essential human micronutrient with critical roles in immune functioning and antioxidant defence. Estimates of dietary Se intakes and status are scarce for Africa although crop surveys indicate deficiency is probably widespread in Malawi. Here we show that Se deficiency is likely endemic in Malawi based on the Se status of adults consuming food from contrasting soil types. These data are consistent with food balance sheets and composition tables revealing that >80% of the Malawi population is at risk of dietary Se inadequacy. Risk of dietary Se inadequacy is >60% in seven other countries in Southern Africa, and 22% across Africa as a whole. Given that most Malawi soils cannot supply sufficient Se to crops for adequate human nutrition, the cost and benefits of interventions to alleviate Se deficiency should be determined; for example, Se-enriched nitrogen fertilisers could be adopted as in Finland.
Selenium is an essential micronutrient for humans1, with 25 genes expressing selenoproteins in the human genome2. These include iodothyronine deiodinases, thioredoxin reductases and glutathione peroxidases (GPx) which have critical roles in thyroid function, redox homeostasis and antioxidant defence and can be compromised by Se deficiency1,2,3. Selenium deficiency also affects immune responses and is linked to lower CD4+ T cell counts, disease progression and mortality among individuals infected with HIV-1 (ref. 4). Several biomarkers are used to define human Se status, including its concentration in blood fractions and urine1. Whole-plasma GPx3 activity saturates at ~100 μg Se L−1, corresponding to habitual Se intakes of ~1 μg Se kg−1 body mass d−1, and this relationship is used by many expert bodies to set dietary recommendations of 25–75 μg Se person−1 d−1 at an individual level1. Few studies have been conducted on Se nutrition in Southern Africa, although Se deficiency is probably widespread based on intake data and extrapolation. For example, intakes of 17 μg Se d−1 were reported for adults in rural Burundi5 and 15–21 μg Se d−1 for children in rural areas of Zomba District, Malawi6. The latter study corresponds to low plasma Se status (typically <60 μg L−1) among adults in this area7,8,9. In a recent spatial survey of soil and maize grain in Malawi, >90% of the population were estimated to consume <7.5 μg Se person−1 d−1 from maize grain10. Maize provides >50% of dietary energy supply in Malawi based on retail-level food balance sheets11 and household12 surveys, but contributes more in some groups. On calcareous soils classified as Eutric Vertisols, grain Se concentrations were >10-fold higher than on other soil types10 due to increased soil-to-crop Se transfer (Figure 1). This is likely to be due mainly to greater stability of soluble Se(VI) species at high pH, i.e. the most plant-available form of Se13 and decreased strength of Se(IV) adsorption on soil colloids and differences in soil clay mineralogy10. However, soils of the Eutric Vertisols type comprise only 0.5% of the land area of Malawi and Se deficiency risk is likely to be high at a population level.
Figure 1. Soil-to-maize grain Se transfer.
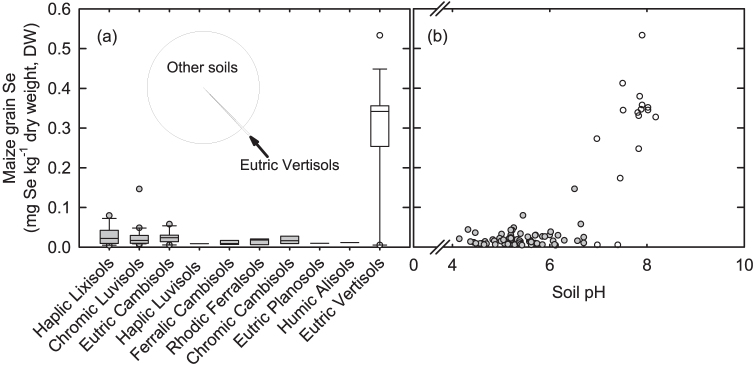
(a, b) Most Malawi soils (grey symbols) have low soil-to-maize Se transfer linked to soil type including a low soil pH, in contrast to Eutric Vertisols (open symbols). In (a) boxes represent 25 and 75%-iles with median lines; whiskers are 5 and 95%-iles; circles are outliers. Inset in (a), pie chart shows that Eutric Vertisols comprise <0.5% of all soil types in Malawi. Data redrawn from Chilimba et al. (2011; ref. 10).
The aims of this study were (1) to test whether spatial variation in soil-to-crop transfer of Se due to soil pH corresponds with Se intake and Se status in individuals, and (2) to determine the risk of dietary Se inadequacy from net food availability and food composition tables. Where Se deficiency risks are high, it may be feasible to adopt agricultural-based programmes to enhance the mineral composition of foodstuffs, for example, by enriching fertilisers with Se14,15,16.
Results
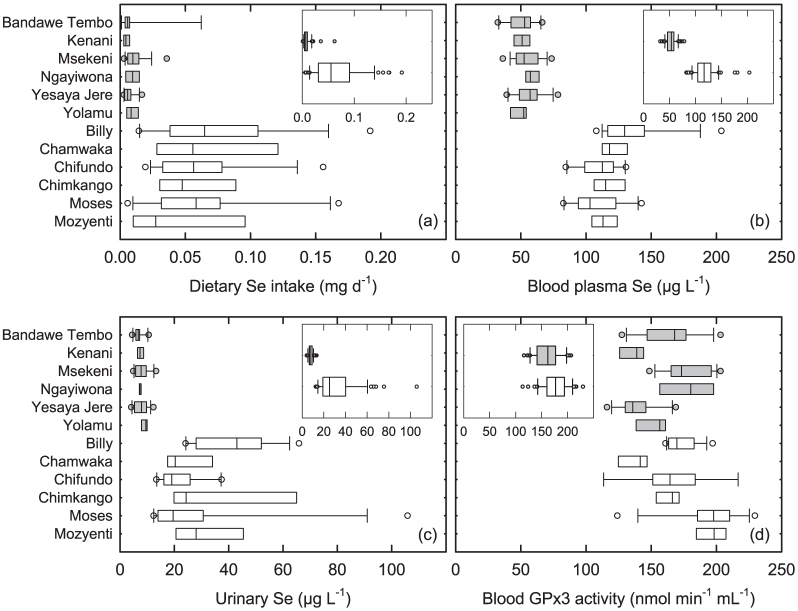
Soil pH markedly affected dietary Se intake and biomarkers of Se status in Malawi, as predicted from spatial surveys of maize10. Women from villages with acid soils in Zombwe Extension Planning Area (EPA) had median dietary Se intake of 6.5 μg Se d−1 (standard deviation (SD) 9.4, range 1.1–62.3 μg Se d−1, n = 56) from all dietary sources including water (Figure 2a; Supplementary Tables 1, 2). Selenium intake was eight-fold higher in villages with proximal calcareous soils in Mikalango EPA (median 55.3 μg Se d−1, SD 44.9, range 5.8–192 μg Se d−1, n = 58). Plasma Se concentration in Zombwe EPA (median 53.7 μg L−1, SD = 9.7, range 32.3–78.4, n = 60) was less than half of those in Mikalango EPA (median 117 μg L−1, SD = 22.5, range 82.6–204, n = 60; Figure 2b). Urine Se concentration in Zombwe EPA (median 7.3 μg L−1, SD = 2.0, range 4.1–13.3, n = 59) was one third that of Mikalango EPA (median 25.3 μg L−1, SD = 18.9, range 12.4–106, n = 56; Figure 2c). Variation between EPAs was much greater than that between villages within an EPA for dietary Se intake and plasma and urine Se concentrations. EPA + village terms explained 43 + 2, 79 + 4 and 46 + 9% of the total variation in dietary Se intake, plasma and urine Se concentrations, respectively. Differences between EPAs were highly significant for dietary Se intake and plasma and urine Se concentrations (P≪0.001). Variation between villages within specific EPAs also occurred for plasma (P = 0.004) and urine (P = 0.021) Se concentration, but not for dietary Se intake (P = 0.943). GPx3 activity also differed between EPAs (P = 0.002). However, only 8% of the total variation in GPx3 activity was explained by EPA, whereas 46% of the variation occurred between villages within an EPA (Figure 2d). Thus, plasma GPx3 activity in Zombwe EPA (median 162 nmol min−1 mL−1, SD 24.1, range 116–207, n = 60) was lower than in Mikalango EPA (median 177 nmol min−1 mL−1, SD 25.6, range 113–230, n = 53). Residual variation was due to individual-level variation within villages. Given that approximately half of the variation in dietary Se intake, urine Se concentration and plasma GPx3 activity occurred among individual volunteers, it seems that plasma Se concentration is the most robust biomarker of Se status in these settings. Urine Se concentration will be more sensitive to short-term variation in Se intake than blood plasma Se concentration, whereas GPx3 may also vary due to oxidative stress conditions1. However, urine Se concentration measurements (Figure 3) could provide an effective non-invasive method for identifying Se deficiency risk in populations, once cut-off points to assess the severity of Se deficiency have been established, as is used routinely for iodine17,18.
Figure 2. Dietary Se intake and biomarkers of Se status in adult females in Malawi.
(a) Dietary Se intake, (b) blood plasma Se concentration, (c) urine Se concentration and (d) blood plasma glutathione peroxidase 3 (GPx3) activity. Grey bars represent six villages from Zombwe Extension Planning Area (EPA) with low pH soils; open bars represent six villages from Mikalango EPA with high pH soils. Insets show these villages grouped by EPA. Boxes are 25 and 75%-iles with median lines; whiskers are 5 and 95%-iles; circles are outliers.
Figure 3. Selenium biomarkers as functions of dietary Se intake among adult females in Malawi.
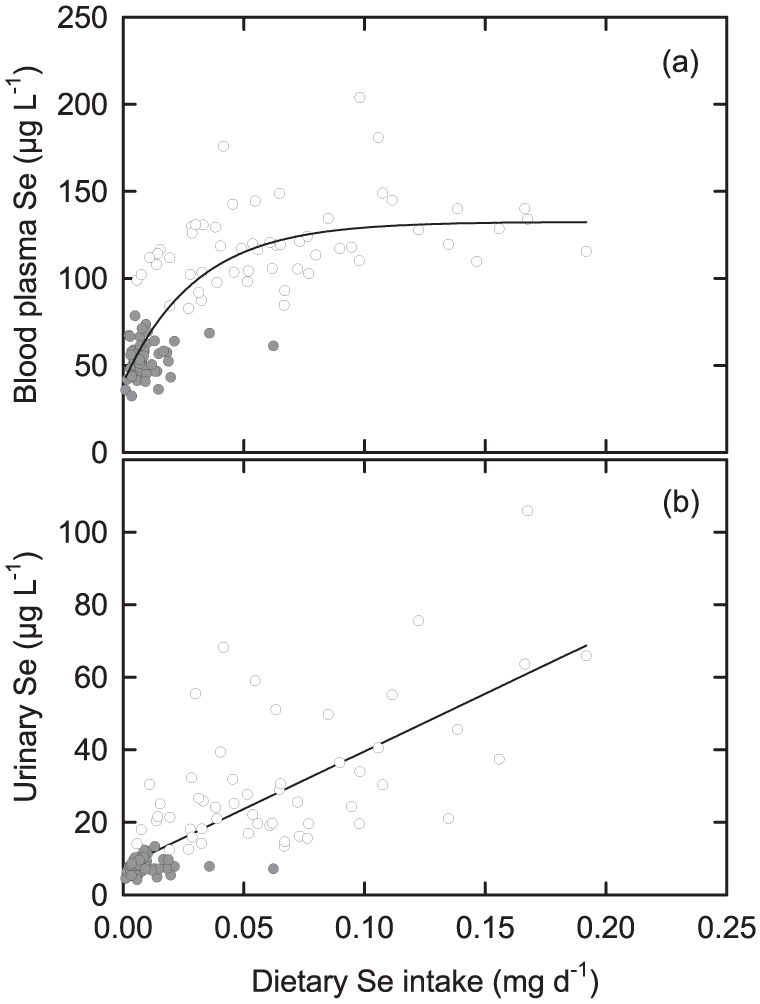
(a) Blood plasma Se concentration and (b) urine Se concentration. Grey circles are adults from villages with low pH soils; open circles are adults from villages with high pH soils. Regression lines are (a) y = 39.9 + (92.6*(1 − (3.2*10−15)x)), r2 = 0.648, P < 0.0001; (b) y = 7.8 + (318*x), r2 = 0.5707, P < 0.0001.
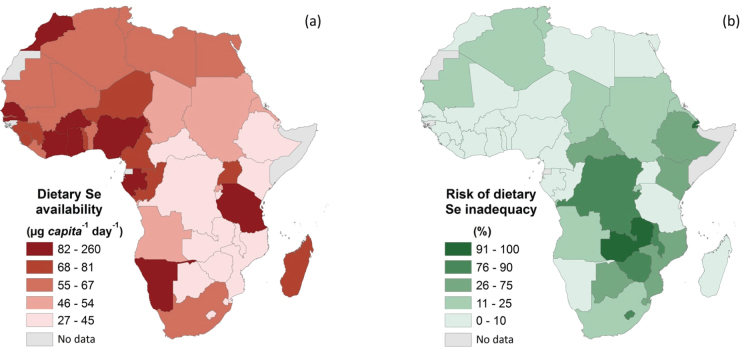
We estimated per capita supply of dietary Se available for human consumption in 46 African countries using an approach described previously for magnesium19, by integrating Food and Agriculture Organization (FAO) Food Balance Sheets (FBSs) for 2007, and a food Se composition table (Supplementary Table 3). Risk of inadequate Se intake was estimated using an EAR cut-point method19,20. Mean Se supply in 2007 for Africa was 71 μg person−1 d−1, ranging from 27 μg d−1 in Djibouti to 264 μg d−1 in Ghana (Figure 4; Supplementary Table 4). Selenium supply tended to be lower in Southern and Mid Africa. The risk of inadequate Se intake in Africa, is therefore 22% overall representing 230 M people (Figure 4). It must be stressed that the use of FBSs and food Se composition tables to estimate Se supply (i.e. and thereby infer intake and deficiency risks) must be conducted with great caution. For example, Se concentrations of fresh coconut in this analysis is from a single West Africa food composition table21 which reports a very high value of 810 μg 100 g−1 FW. This value is much higher than that reported for coconut by the US Department of Agriculture22 of 10.1 μg 100 g−1 FW. So in Ghana, for example, which has a FBS value for 2007 of 7.3 kg coconut person−1 y−1, the estimated supply of Se from coconut could be either 162 or 2 μg Se person−1 d−1 depending on which source is used. Here, we elected to use the higher value, as it was more geographically relevant, but we note that our decision may underestimate the risk of Se deficiency. In a recent study of Se intakes of children (ages 12–15) in 3 residential care orphanages in Ghana from duplicate diets23, the mean dietary intakes of Se were 58, 82, 92 μg Se person−1 d−1. These are less than the supply figure of 264 μg Se person−1 d−1 derived from FBSs for Ghana, but interestingly, are greater than reported Se intakes in Malawi in our study. So whilst Se supply is likely be lower than suggested in Figure 4 for countries where coconut consumption is high, an approach based on FBSs and food composition tables can still identify populations at higher risks of deficiency and also guide efforts to improve food composition tables.
Figure 4. Mapping dietary Se availability in Africa.
(a) Mean dietary Se availability and (b) estimated risk of inadequate Se intake based on US Estimated Average Requirement (EAR) of 45 and 23 μg Se person−1 d−1 for those aged >10 and <10 yr, respectively, and 49 and 59 μg Se person−1 d−1 for pregnant and lactating women, respectively39. Data are calculated from national-level food balance sheets for 46 African countries in 2007 and regional food composition tables using methods described previously19.
Discussion
These new biomarker measurements and analyses of per capita Se supply indicate that policies to alleviate Se deficiency should be considered alongside other micronutrient intervention programmes in Southern Africa. However, despite the known roles of Se in many communicable and non-communicable diseases1, it is not yet possible to assess the health/economic impacts of Se deficiency at population scale due to a lack of suitable framework and input data. In contrast, frameworks have been established for other micronutrient deficiencies (e.g. iron, iodine, zinc and vitamin A) which co-exist in developing countries. These affect growth, immune function and cognitive development, cause disease and premature death, and constrain economic growth. For Malawi, we estimate that zinc deficiency (ZnD) leads to an annual loss of 6,500 “disability-adjusted life years” (DALYs), i.e. person-years lost to disability and shortened life, per million population (>99,900 DALYs in total), and >3,800 instances of child mortality per year using established methodologies24,25. This is higher than the 2,750, 2,020 and 1,660 DALYs estimated to be lost due to ZnD per million population in India, Honduras and Nicaragua, respectively24. These data suggest that ZnD alone imposes an economic burden on Malawi of ~$100 m yr−1.
It is of course feasible to diversify diets in order to increase Se intake, for example, by increased consumption of Se-rich foodstuffs (e.g. including animal-based foodstuffs). However, such approaches are challenging when baseline Se intakes are low and where there is a lack of purchasing power for foods rich in Se, as in many low-income countries. Short- and medium-term policies to alleviate iron, iodine, zinc, and vitamin A deficiencies have been adopted in Malawi and elsewhere, including dietary iron supplements and salt iodisation26,27, although such programmes are not used for Se. Longer-term crop-based approaches (biofortification) are also underway to increase iron, zinc and vitamin A intakes in the region28,29, although the breeding potential to biofortify crops with Se via conventional breeding is low14,29. A public health precedent to alleviate dietary Se inadequacy was set in Finland in 1984 following the introduction of Se-enriched fertilisers (agronomic biofortification), which has continued to date14,16. Selenium fertilisation has successfully increased the Se concentrations of Finnish foods and dietary Se intakes and status16. All types of cereal grains, and all grain fractions are easily enriched with Se when Se(VI) forms are added to fertilisers14,15,16,30,31. Fertiliser-based strategies also avoid the significant lead-times required for crop breeding programmes and distribution of new varieties. Agronomic biofortification is therefore feasible in a Southern African context where inorganic fertilisers are used. Malawi has operated a Farm Input Subsidy Programme (FISP) since 2005/06 whereby fertiliser vouchers are distributed to farmers at a village scale through the national extension service system32. The FISP is therefore a potential public health intervention route if fertilisers are enriched with Se during production15. However, there are knowledge gaps which require further research and capacity building in the nutrition, agriculture and economics sectors to ascertain the cost/benefit of this approach, including an assessment of the ongoing costs of Se supply and monitoring of health outcomes. Another agronomic approach may be to use lime applications to increase soil-to-grain Se transfers but this approach has not yet been tested to our knowledge. However, a liming strategy could reduce the plant-availability of other essential micronutrients such as iron and zinc.
Methods
Ethical approval
Ethical approval was obtained from the National Health Sciences Research Committee, Malawi (NHSRC reference #784), prior to commencing the study.
Site selection
Two Extension Planning Areas (EPAs) in Malawi were studied: Zombwe EPA in Mzuzu Agricultural Development Division (ADD) in the north and Mikalango EPA in Shire Valley ADD in the south. Zombwe EPA is characterised by low pH soils (median 5.2, n = 11), particularly Haplic Lixisols (70% of EPA area), which have a low soil-to-maize transfer of Se and median grain Se concentration of 22 μg Se kg−1 fresh weight (FW)10. Mikalango EPA is characterised by areas of calcareous Eutric Vertisols (median pH = 7.8, n = 16; 10% of EPA area) with a high soil-to-grain transfer of Se and a median grain Se concentration of 342 μg Se kg−1 FW10. Six villages were selected from each EPA for participant recruitment. In Zombwe EPA, villages were located in Zombwe I Section: Bandawe Tembo, Kenani, Msekeni, Ngayiwona, Yesaya Jere, Yolamu. In Mikalango EPA, villages were located in Nyamichimba Section: Billy, Chamwaka, Chifundo, Chimkango, Moses, Mozyenti.
Participant selection
In total, a sample of 120 apparently healthy adult females aged 18–50 years were recruited to participate in the study, from six villages per EPA, most of whom were subsistence farmers. Only one volunteer per family was recruited to avoid multiple sampling of participants with similar dietary patterns. Exclusion criteria for participants included known pregnancy, smokers, those diagnosed with chronic physical or mental disease or long term illness requiring treatment, acute or chronic infection, those taking regular prescribed medicine including oral contraceptive pills, those taking regular dietary supplements and those who had lived in the study area for less than six months. Sensitisation visits were held in September 2010 by the scientific team and Extension Planning Officers from the Malawi Ministry of Agriculture and Food Security, and in March 2011 by the Ministry of Health and Traditional Village Authorities. Informed consent of all volunteers was obtained prior to the collection of the duplicate diet composites and tissue samples by a trained member of the Lilongwe University of Agriculture and Natural Resources study team. No payment was made for participation or the use of tissue samples. Participants were reimbursed at cost-price for the food and beverages in the duplicate diet composites taken for analyses.
Socio-economic status of participants
Background data were collected using questionnaires administered by trained interviewers in the homes. A socio-economic status (SES) index was developed for each participant based on categories used earlier in the Nutrition Collaborative Research Support Program (NCRSP33), and in a later study of pregnant women in rural Malawi34. Information collected included house quality, sanitation, water source, household size, occupation and schooling. The index has a maximum theoretical numerical value of 14 (high SES). The numerical value is based on derived numerical equivalents calculated for each of the string variables in STATA (StataCorp LP, College Station, Texas, USA). The SES index and background data on participants are given in Supplementary Table 1.
Duplicate diet composites
Duplicate diet composites were collected over a full day (24 h) for each participant. Food records and diet composites samples were collected between 13 and 16 March 2011 in Mikalango EPA, and between 20 and 23 March 2011 in Zombwe EPA. Research assistants (RAs) were recruited to reside in the household of each participant to weigh and sample all food and beverage items (including drinking water and snack items consumed away from the household) during the sampling date using a 750 mL polyethylene jug and a kitchen scale accurate to ~1 g. For each food and beverage item consumed, an exact duplicate sample was weighed and collected in a double-lined trace-element-free sealable polyethylene bag. Composites were placed in a cooler box with ice packs, transferred to a central laboratory within 24 h of collection, and blended to a homogeneous slurry using a domestic blender. Approximately 50 mL of homogenate was subsampled using a trace-element-free pipette (7017, Corning, Costar, Amsterdam, The Netherlands) and this was divided into two trace-element-free universal tubes (Bibby Sterilin, Stone, Staffordshire, UK). Homogenate subsamples were frozen at −20°C. Weighed food samples were recorded (data not shown) to provide details on the composite diet samples containing animal products, to comply with import licence requirements.
One set of frozen diet composite subsamples (n = 115) was transported to the UK on dry ice for Se analyses, and the remaining set was retained in Malawi. Subsamples were freeze-dried in their original universal tubes with muslin caps. Subsample volumes were recalculated to correspond to the loss of mass on freeze-drying plus the residual mass of solid material, assuming that the dry food had a material density of 1.0 and was wholly in suspension. The mean volume of each subsample was 24.7 mL: typically 20–23 mL and 1–4 g dry material. Freeze-dried diet composites were manually ground to homogenise the samples and crush seeds etc. Approximately 0.3 g of each sample was microwave-digested in 3.0 mL of 70% Trace Analysis Grade (TAG) HNO3, 2.0 mL H2O2 and 3.0 mL milli-Q water (18.2 MΩ cm; Fisher Scientific UK Ltd, Loughborough, UK). Elemental analysis was by inductively coupled plasma-mass spectrometry (ICP-MS; X-SeriesII, Thermo Fisher Scientific Inc., Waltham, MA, USA). Acid digests were analysed by ICP-MS (i) using Collision Cell Technology with Kinetic Energy Discrimination (CCT-KED) mode for Na, Mg, K, Ca, Al, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, As, Rb, Sr, Mo, Cd, Cs, Ba, Pb and U (using Sc, Ge, Rh and Ir internal standards), and (ii) in H2-reaction cell mode for 78Se alone (using Ge and Rh internal standards) with 2% methanol added to samples to maximise sensitivity for Se determination. Three certified reference materials (CRMs) from NIST (National Institute of Standards and Technology, Gaithersburg, MD, USA) were included: NIST 1573a (tomato leaves, certified value 0.054 mg Se kg−1 dry weight, DW, 123% recovery), NIST 1577c (bovine liver, 2.031 mg Se kg−1 DW, 96% recovery) and NIST 1567a (wheat flour, 1.1 mg Se kg−1 DW, 100% recovery). In addition, four inter-lab flour samples supplied by Dave Hart, Institute of Food Research, UK yielded an average recovery of 102%. Operational blanks (n = 10) were run to determine practical Limit of Detection (LOD; 3*SD) and Limit of Quantification (LOQ, 10*SD) values for Se of 0.0018 and 0.0059 mg kg−1 DW, respectively, in composite diet samples. These and subsequent data collected from individuals were analysed by ANOVA with ‘EPA/Village’ as a treatment factor and no blocking terms using GenStat (15th Edition, VSN International, Hemel Hempstead, UK).
Blood and urine samples
Whole blood was collected from consenting volunteers by trained staff from the Community Health Sciences Unit, Malawi. Samples were collected on Saturday 12 March 2011 in Mikalango EPA and Saturday 19 March 2011 in Zombwe EPA. Each volunteer was seated, their skin cleaned with alcohol at the site of the antecubital vein, and their arm restricted with a tourniquet for <1 minute. Blood was drawn into two 7 mL trace-element-free blue-top evacuated tubes (NH119 I.U. ref 367735, Becton Dickinson UK Ltd, Plymouth, UK). Disposable polyethylene powder-free gloves were used for all blood handling steps. The tourniquet was loosened during blood collection and before the needle was removed from the arm. Immediately after the blood was collected the tube was inverted 10 times to mix the contents before placing the samples in a cooler box with ice packs for transport to the nearest laboratory for processing (Zombwe EPA, Mzuzu Central Hospital; Mikalango EPA, Zomba District Hospital). For one of the 7 mL samples, plasma was separated by centrifugation at 1500 g for 10 min and transferred into trace-element-free tubes (n = 4 aliquots for each sample) and frozen at −80°C. Plasma samples were transported to the UK on dry ice for Se analysis using ICP-MS, and to determine Se-dependent glutathione peroxidase activity, as described below. The other 7 mL samples were used for whole-blood analyses (data not shown). Casual urine (i.e. samples unrelated to eating/sleeping patterns) samples were collected on the same day as the blood sampling. These were collected in sterile pre-labelled tubes (30 mL sterile universal tubes, Bibby Sterilin, UK) and placed in a cooler with ice before transfer to the nearest laboratory for processing. Two 8 mL subsamples were frozen at −20°C and transported to the UK using dry ice for ICP-MS analysis.
Selenium concentration in blood plasma was determined by ICP-MS in H2-cell mode. Samples and standards (SPEX CertiPrep Inc., Metuchen, NJ, USA) were diluted 1-in-20 in 1% TAG HNO3 containing 0.1% of a non-ionic surfactant (‘Triton X-100’ + ‘antifoam-B’, Sigma-Aldrich Company Ltd., Dorset, UK) and 2% methanol. Internal standards containing Ir (5 μg L−1), Rh (10 μg L−1), Ga (25 μg L−1) and Sc (50 μg L−1) were included. Calibrations were in the range 0–50 μg Se L−1. Quality control CRMs included Seronorm™-1 (107 μg Se L−1, 109% recovery), Seronorm™-2 (163 μg Se L−1, 102% recovery) (Sero AS, Billingstad, Norway) and UTAK-66816 Lot 7528 (certified value 115 μg Se L−1, 106% recovery) (UTAK Labs Inc., Valencia, CA, USA) CRMs. Sample blanks (n = 10) were run to determine LOD and LOQ values of 0.055 and 0.182 μg L−1, respectively, for blood plasma. Urine Se was determined by ICP-MS in H2-cell mode for samples diluted 1-in-10 with 2% HNO3 and calibrated using Seronorm™-1 (certified value 58.6 μg Se L−1) as a matrix-matched calibration standard. Urine assays were corrected for creatinine by dividing urine Se concentration (μg L−1) by creatinine concentration (g L−1). Sample blanks (2% HNO3; n = 10) were run to determine LOD and LOQ values of 0.095 and 0.316 μg L−1, respectively, for urine. Creatinine was analysed using a commercial kit on a RX IMOLA Chemistry Analyzer (Randox Laboratories, Belfast, UK) according to the manufacturer's instructions.
Quantification of plasma GPx3 activity
Plasma glutathione peroxidase (GPx3) activity was quantified using a high throughput 96 well spectrophotometric enzyme assay35. All plasma samples were analysed in triplicate. The enzyme assay mixture contained 100 mM Tris-HCl pH 7.4, 3 mM glutathione, 0.25 mM NADPH, 1U glutathione reductase and 0.1% triton X100 with hydrogen peroxide as the substrate. The rate of decrease in absorbance at 340 nm was measured at 10 s intervals for 15 min at 37°C using a plate reader with kinetic capability (FLUOstar Omega plate reader, BMG Labtech, Germany). The GPx3 activities were calculated from the initial rates of reaction36, with one unit (U) defined as 1 nmol NADPH oxidised per minute.
Estimating dietary selenium supply from food balance sheets
Per capita supply of Se was estimated for all African countries as the product of food supply data available for human consumption at a retail level and food Se concentration using previously described methodology19. Food supply was based on Food Balance Sheet (FBS) data sourced from 46 Food and Agriculture Organization (FAO) member countries in Africa19. Food Se concentration data were sourced from published food composition tables21,22,37,38 and three food Se concentration databases were generated: East (E) Africa, Southern (S) Africa (also used for North (N) Africa) and West (W) Africa (also used for Mid (M) Africa). All Se concentration data are expressed as μg Se 100 g−1 fresh weight (FW) edible portion. Food Se concentration data, literature sources and best-fit FBS categories are shown in Supplementary Table 3. Per capita Se supply data were used to infer risk of dietary Se inadequacy using the EAR cut-point method19,20. The US Estimated Average Requirement (EAR) of 45 and 23 μg Se person−1 d−1 was used for those aged >10 and <10 yr, respectively, and 49 and 59 μg Se person−1 d−1 for pregnant and lactating women, respectively39, with a conservative inter-individual coefficient of variation (CV) of Se intake of 25%. As discussed previously19, the EAR cut-point method is highly sensitive to CV and also to food concentration data reported within the food composition tables. For example, Stadlymayr et al.21 reported 810 μg Se 100 g−1 FW edible portion in raw coconuts, resulting in high intake estimates for some West African countries (see Results).
Estimating disability-adjusted life years (DALYs) lost due to micronutrient deficiency in Malawi
It is not straightforward to determine the existing burden of disease due to Se deficiency in any country at present because an accepted framework for Se is lacking. However, it is possible to illustrate the impact of mineral deficiencies in Malawi using zinc deficiency (ZnD) as an example. Thus, the number of DALYs lost due to ZnD in Malawi was estimated using previously developed methods24,40,41,42. Population size was taken from World Bank sources43 and demographic data were obtained from the 2008 Population and Housing Census Results of the National Statistical Office of Malawi44; the crude birth rate was obtained from the 2008 Population and Housing Census Results Main Report45. Infant mortality rate was obtained from the 2008 Population and Housing Census Results Mortality Report46, while the under-five mortality rate was obtained from the 2012 Malawi Population Data Sheet47. Average remaining life expectancies were obtained from the World Health Organization life tables48, while stunting rates (height/age - 2 SD) were taken from the WHO Dataset on Child Growth and Malnutrition for Malawi49. The estimated number of episodes of diarrhoea and pneumonia in infants and children were taken from Stein et al.40 and confirmed by local expert opinion (AA Kalimbira, Lilongwe University of Agriculture and Natural Resources). Per capita income figures were taken from the World Development Indicators of the World Bank43; DALYs were valued in accordance with established methods50 and using a discount rate of 3%40,41. To convert DALYs lost to monetary terms, a standard figure for developing countries of US $1,000 was used which corresponds approximately to triple the annual per capita income in Malawi.
Author Contributions
R.H., S.D.Y., A.D.C.C., E.L.A., M.J.W., B.C., S.J.F.-T., R.S.G., A.A.K. and M.R.B. conceived the study. R.H., E.W.P.S., S.D.Y., E.J.M.J., E.L.A., J.G., D.K., R.S.G. and A.J.S. collected and analysed the data. All authors contributed to writing the manuscript.
Supplementary Material
Supplementary Tables
Acknowledgments
Author contributions to this study were funded primarily by a Partnership and Project Development Award (NE/I003347/1) from the UK Natural Environment Research Council (NERC), the UK Department for International Development (DFID) and the Economic and Social Research Council (ESRC) under the Ecosystems Services for Poverty Alleviation (ESPA) scheme. Additional funding was provided by the British Geological Survey and the University of Nottingham.
References
- Fairweather-Tait S. J. et al. Selenium in human health and disease. Antioxid. Redox Signal. 14, 1337–1383 (2011). [DOI] [PubMed] [Google Scholar]
- Kryukov G. V. et al. Characterization of mammalian selenoproteomes. Science 300, 1439–1443 (2003). [DOI] [PubMed] [Google Scholar]
- Hurst R. et al. Establishing optimal selenium status: results of a randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 91, 923–931 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann P. R. & Berry M. J. The influence of selenium on immune responses. Mol. Nutr. Food Res. 52, 1273–1280 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benemariya H., Robberecht H. & Deelstra H. Daily dietary intake of copper, zinc and selenium by different population groups in Burundi, Africa. Sci. Total Environ. 136, 49–76 (1993). [DOI] [PubMed] [Google Scholar]
- Donovan U. M., Gibson R. S., Ferguson E. L., Ounpuu S. & Heywood P. Selenium intakes of children from Malawi and Papua New Guinea consuming plant-based diets. J. Trace Elem. Electrol. Health Dis. 6, 39–43 (1992). [PubMed] [Google Scholar]
- van Lettow M. et al. Micronutrient malnutrition and wasting in adults with pulmonary tuberculosis with and without HIV co-infection in Malawi. BMC Infect. Dis. 4, 61 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lettow M., West C. E., van der Meer J. W. M., Wieringa F. T. & Semba R. D. Low plasma selenium concentrations, high plasma human immunodeficiency virus load and high interleukin-6 concentrations are risk factors associated with anemia in adults presenting with pulmonary tuberculosis in Zomba district, Malawi. Eur. J. Clin. Nutr. 59, 526–532 (2005). [DOI] [PubMed] [Google Scholar]
- Gibson R. S., Bailey K. B., Ampong Romano A. B. & Thomson C. D. Plasma selenium concentration in pregnant women in two countries with contrasting soil selenium levels. J. Trace Elem. Med. Biol. 25, 230–235 (2011). [DOI] [PubMed] [Google Scholar]
- Chilimba A. D. C. et al. Maize grain and soil surveys reveal suboptimal dietary selenium intake is widespread in Malawi. Sci. Rep. 1, 72 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broadley M. R. et al. Dietary requirements for magnesium, but not calcium, are likely to be met in Malawi based on national food supply data. Int. J. Vitam. Nutr. Res. 82, 192–199. [DOI] [PubMed] [Google Scholar]
- Ecker O. & Qaim M. Analyzing nutritional impacts of policies: an empirical study for Malawi. World Dev. 39, 412–428 (2011). [Google Scholar]
- Li H.-F., McGrath S. P. & Zhao F. J. Selenium uptake, translocation and speciation in wheat supplied with selenate or selenite. New Phytol. 178, 92–102 (2008). [DOI] [PubMed] [Google Scholar]
- Broadley M. R. et al. Biofortification of U. K. food crops with selenium (Se). Proc. Nutr. Soc. 65, 169–181 (2006). [DOI] [PubMed] [Google Scholar]
- Chilimba A. D. C. et al. Agronomic biofortification of maize with selenium (Se) in Malawi. Field Crops Res. 125, 118–128 (2012). [Google Scholar]
- Alfthan G. et al. In Combating Micronutrient Deficiencies: Food-Based Approaches (eds Thompson B., & Amoroso L., eds. ) 312–337 (CAB International and Food and Agriculture Organization of the United Nations (FAO), Rome, Italy, 2011). [Google Scholar]
- Li M. & Eastman C. J. The changing epidemiology of iodine deficiency. Nat. Rev. Endocrinol. 8, 434–440 (2012). [DOI] [PubMed] [Google Scholar]
- Thomson C. D., Smith T. E., Butler K. A. & Packer M. A. An evaluation of urinary measures of iodine and selenium status. J. Trace Elem. Med. Biol. 10, 214–222 (1996). [DOI] [PubMed] [Google Scholar]
- Joy E. J. M. et al. Risk of dietary magnesium deficiency is low in most African countries based on food supply and composition data. Plant Soil. 10.1007/s11104-012-1388-z (2012). [Google Scholar]
- Wuehler S. E., Peerson J. M. & Brown K. H. Use of national food balance data to estimate the adequacy of zinc in national food supplies: methodology and regional estimates. Public Health Nutr. 8, 812–819 (2005). [DOI] [PubMed] [Google Scholar]
- Stadlmayr B. et al. Composition of Selected Foods from West Africa. (Food and Agriculture Organization, Rome, 2010). [Google Scholar]
- US Department of Agriculture, Agricultural Research Service (USDA-ARS). USDA National Nutrient Database for Standard Reference, Release 24. Nutrient Data Laboratory Home Page. Accessed online November 30, 2011: http://www.ars.usda.gov/nutrientdata (2011). [Google Scholar]
- Adotey D. K., Stibilj V., Serfor-Armah Y., Nyarko B. J. B. & Jaćimović R. Dietary supply of selenium for adolescents in three residential care orphanages in Southern Ghana. Sci. Total Environ. 410–411, 72–29 (2011). [DOI] [PubMed] [Google Scholar]
- Stein A. J. et al. Plant breeding to control zinc deficiency in India: how cost effective is biofortification? Public Health Nutr. 10, 492–501 (2007). [DOI] [PubMed] [Google Scholar]
- Stein A. J. The Potential of Biofortification of Rice, Beans, Cassava and Maize throughout Latin America. Evaluation Report for the International Food Policy Research Institute, Washington. Mimeo. Accessed online August 12, 2012: http://www.ajstein.de/cv/Stein_IFPRI_biofortificationLatAm.pdf (2010). [Google Scholar]
- Kalimbira A. A., MacDonald C. & Simpson J. R. The impact of an integrated community-based micronutrient and health programme on stunting in Malawian preschool children. Public Health Nutr. 13, 720–729 (2010). [DOI] [PubMed] [Google Scholar]
- Kalimbira A. A., MacDonald C. & Simpson J. R. The impact of an integrated community-based micronutrient and health programme on anaemia in non-pregnant Malawian women. Public Health Nutr. 13, 1445–1452 (2010). [DOI] [PubMed] [Google Scholar]
- Bouis H. E., Hotz C., McClafferty B., Meenakshi J. V. & Pfeiffer W. H. Biofortification: A new tool to reduce micronutrient malnutrition. Food Nutr. Bull. 32, S31–S40 (2011). [DOI] [PubMed] [Google Scholar]
- White P. J. & Broadley M. R. Biofortification of crops with seven mineral elements often lacking in human diets - iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 182, 49–84 (2009). [DOI] [PubMed] [Google Scholar]
- Broadley M. R. et al. Selenium biofortification of high-yielding winter wheat (Triticum aestivum L.) by liquid or granular Se fertilisation. Plant Soil 332, 5–18 (2010). [Google Scholar]
- Hart D. J. et al. Selenium concentration and speciation in biofortified flour and bread: retention of selenium during grain biofortification, processing and production of Se-enriched food. Food Chem. 126, 1771–1778 (2011). [DOI] [PubMed] [Google Scholar]
- Dorward A. & Chirwa E. The Malawi agricultural input subsidy programme: 2005-6 to 2008-9. Int. J. Agric. Sustain. 9, 232–237 (2011). [Google Scholar]
- Kirksey A. et al. Relation of maternal zinc nutriture to pregnancy outcome and infant development in an Egyptian village. Am. J. Clin. Nutr. 60, 782–792 (1994). [DOI] [PubMed] [Google Scholar]
- Gibson R. S. & Huddle J.-M. Suboptimal zinc status in pregnant Malawian women: its association with low intakes of poorly available zinc, frequent reproductive cycling, and malaria. Am. J. Clin. Nutr. 67, 702–709 (1998). [DOI] [PubMed] [Google Scholar]
- Smith A. D. & Levander O. A. High-throughput 96-well microplate assays for determining specific activities of glutathione peroxidase and thioredoxin reductase. Methods Enzymol. 347, 113–121 (2002). [DOI] [PubMed] [Google Scholar]
- Paglia D. E. & Valentine W. N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70, 158–169 (1967). [PubMed] [Google Scholar]
- Donovan U. M., Gibson R. S., Ferguson E. L., Ounpuu S. & Heywood P. The selenium content of staple foods from Malawi and Papua New Guinea. J. Food Compos. Anal. 4, 329–336 (1991). [Google Scholar]
- Food Standards Agency. McCance and Widdowson's The Composition of Foods, Sixth Summary Edition. (Royal Society of Chemistry, Cambridge, 2002). [Google Scholar]
- Institute of Medicine (IoM). Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids: A Report of the Panel on Dietary Antioxidants and Related Compounds, Subcommittees on Upper Reference Levels of Nutrients and of Interpretation and Use of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. (National Academies Press, Washington DC, 2002). [Google Scholar]
- Stein A. J. et al. Analysing Health Benefits of Biofortified Staple Crops by Means of the Disability-Adjusted Life Years Approach: A Handbook Focusing on Iron, Zinc and Vitamin A. HarvestPlus Technical Monograph 4. Accessed online August 11, 2012: http://www.ifpri.org/publication/analyzing-health-benefits-biofortified-staple-crops-means-disability-adjusted-life-years (International Food Policy Research Institute, Washington DC, 2005). [Google Scholar]
- Stein A. J., Qaim M. & Nestel P. In Handbook of Disease Burdens and Quality of Life Measures (eds Preedy, V. & Watson, R. R.) 1151–1170 (Springer, Heidelberg, 2009). [Google Scholar]
- Meenakshi J. V. et al. How cost-effective is biofortification in combating micronutrient malnutrition? An ex ante assessment. World Develop. 38, 64–75 (2010). [Google Scholar]
- World Bank. World Development Indicators: Malawi, 2011. Accessed online August 11, 2012: http://data.worldbank.org/country/malawi (World Bank, Washington DC, 2012).
- National Statistical Office of Malawi. 2008 Population and Housing Census Results. Statistical Tables: Population Size and Composition. Accessed online August 11, 2012: http://www.nso.malawi.net/ (National Statistical Office of Malawi, Zomba, 2012). [Google Scholar]
- National Statistical Office of Malawi. 2008 Population and Housing Census Results. Main Report. Accessed online August 11, 2012: http://www.nso.malawi.net/ (National Statistical Office of Malawi, Zomba, 2012). [Google Scholar]
- National Statistical Office of Malawi. 2008 Population and Housing Census Results. Mortality Report. Accessed online August 11, 2012: http://www.nso.malawi.net/ (National Statistical Office of Malawi, Zomba, 2012). [Google Scholar]
- National Statistical Office of Malawi. Malawi Population Data Sheet 2012. Accessed online August 11, 2012: http://www.nso.malawi.net/ (National Statistical Office of Malawi, Zomba, 2012). [Google Scholar]
- World Health Organization. Global Health Observatory Data Repository: Life Tables, Malawi 2009. Accessed online August 11, 2012: http://apps.who.int/ghodata/?vid=720 (World Health Organization, Geneva, 2011). [Google Scholar]
- World Health Organization. Global Database on Child Growth and Malnutrition: Malawi. Data update: 13 January, 2012. Accessed online August 11, 2012: http://www.who.int/nutgrowthdb/database/countries/mwi/ (World Health Organization, Geneva, 2012). [Google Scholar]
- Stein A. J. & Qaim M. The human and economic cost of hidden hunger. Food Nutr. Bull. 28, 125–134 (2007). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Tables