Abstract
Pseudomonas aeruginosa has developed a complex cell-to-cell communication system that relies on low-molecular weight excreted molecules to control the production of its virulence factors. We previously characterized the transcriptional regulator MvfR, that controls a major network of acute virulence functions in P. aeruginosa through the control of its ligands, the 4-hydroxy-2-alkylquinolines (HAQs)—4-hydroxy-2-heptylquinoline (HHQ) and 3,4-dihydroxy-2-heptylquinoline (PQS). Though HHQ and PQS are produced in infected animals, their ratios differ from those in bacterial cultures. Because these molecules are critical for the potency of activation of acute virulence functions, here we investigated whether they are also produced during human P. aeruginosa acute wound infection and whether their ratio is similar to that observed in P. aeruginosa-infected mice. We found that a clinically relevant P. aeruginosa isolate produced detectable levels of HAQs with ratios of HHQ and PQS that were similar to those produced in burned and infected animals, and not resembling ratios in bacterial cultures. These molecules could be isolated from wound tissue as well as from drainage liquid. These results demonstrate for the first time that HAQs can be isolated and quantified from acute human wound infection sites and validate the relevance of previous studies conducted in mammalian models of infection.
1. Introduction
Emerging multi-drug resistant organisms are major contributors to morbidity and mortality following burn injury [1]. Pseudomonas aeruginosa infections are common following burns, often presenting as wound infections [2–4]. Current prophylactic measures intended to prevent these infections in burn patients consist of isolation and use of strict sterile techniques combined with rapid surgical wound closure. Although these prevention efforts can be effective under ideal circumstances, they often fail due to insufficient nursing, anesthesia, and critical care management resources, insufficient donor sites for immediate wound closure, or spread of infection from the surrounding environment during the initial first aid provided [5]. Treatment of Pseudomonas wound infections, along with the frequently associated shock and respiratory failure, can involve complex fluid and ventilator management, definitive surgical debridement, and advanced wound closure technologies. The use of antimicrobial agents such as colistin (polymyxin E), tailored to navigate the most recent antibiotic resistance pattern, has been useful.
Clearly, new innovations for the prevention and treatment of Pseudomonas wound infections in burn patients are needed. Ongoing basic research efforts have made strides in dissecting the pathogenesis of Pseudomonas infections using various models of infections in plants, flies, and mice [6–10]. P. aeruginosa coordinates the expression of multiple virulence factors using a complex regulatory communication system, namely, Quorum Sensing (QS), which has been shown to control approximately 10% of P. aeruginosa genes [11, 12]. QS is a cell-density-dependent phenomenon based on the release of low-molecular weight molecules that coordinate gene expression in a cell population [11, 13, 14]. In response to population density, P. aeruginosa produces and secretes such molecules, some of which act as specific chemical signals that control the production of virulence factors mediating acute infection.
A decade ago, we identified the QS transcriptional regulator MvfR (multiple virulence factors regulator) [9]. The MvfR protein controls the expression of most of P. aeruginosa's acute virulence-associated factors [15] as well as the biosynthesis of more than 60 excreted anthranilic acid (AA) low-molecular weight derivatives, most of which belong to the 4-hydroxy-2-alkylquinolines (HAQs) family [12, 13, 16]. HAQ expression is subject to positive autoregulation, with two HAQs, namely, 4-hydroxy-2-heptylquinoline (HHQ) [17] and 3,4-dihydroxy-2-heptylquinoline (PQS) [18], functioning as MvfR ligands. These HAQs, in concert with MvfR, positively regulate the transcription of the pqsABCD [19] and phnAB [9] operons, which encode HAQ biosynthetic enzymes of these molecules [9, 15, 19]. PQS and HHQ are produced in vitro and in vivo in acute models of infection [9, 11, 15, 19]. We showed previously that PQS is more potent than HHQ in inducing MvfR binding to the pqsA promoter as well as inducing pqsA-E transcription in pqsA::pqsH P. aeruginosa mutant [19]. The relative potency of HHQ versus PQS in inducing MvfR ligand-binding domain precipitation suggests that these molecules induce different MvfR conformational changes; a difference in conformation effects could subsequently results in different binding affinities to the same MvfR target promoters, and differences in MvfR-regulated gene transcription.
Because HAQs govern and dictate infection course, they may be of use as indicators of infection stage. Moreover, interfering with these molecules could be a means by which to control Pseudomonas infections. The production of Pseudomonas HAQs has yet to be studied in human wounds. Therefore, here, we examined for the first time whether clinically important Pseudomonas specimens, derived from an active burn wound infection in a human patient, produce and excrete detectable levels HAQs in situ.
2. Materials and Methods
2.1. Human Samples
Permission for use of excess tissue and wound drainage material for the purposes of this study and permission for medical record review were obtained from the hospital institutional review board. The samples were obtained from a discarded resected infected burn wound on the patient right leg. Three types of specimen were collected: (i) one containing necrotic muscle with fat; (ii) one wet and oozing sample with pus and liquefied fat mixed together with some saline solution from the operative field; finally (iii) drainage liquid collected out from the wound.
2.1.1. Histology
Wound biopsies were performed for the purposes of diagnosis and therapy at the time of surgery in the course of the patient's treatment. The biopsy specimens were fixed in formalin, blocked in paraffin, and stained with hematoxylin and eosin. Wound drainage and resected tissues in excess of the amount required for the biopsy were used in this analysis.
2.1.2. HAQ Determination
The analyzed samples were obtained from excess material. Tissues were weighed, cut into small pieces, and then homogenized in 50% methanol. Whenever appropriate, liquid was mixed 1 : 1 with 50% methanol. The quantification of HAQs was performed by liquid chromatography/mass spectrometry (LC/MS) as described elsewhere [20], using HHQ-D4 and PQS-D4 as internal standards. Positive electrospray was employed in MRM mode with 2 × 10−3 mTorr argon and as the collision gas and 30 V of energy to quantify HAQs by LC/MS. The following ion transitions were used: HHQ 244>159, HHQ-D4 248>163, HQNO 260>159, PQS 260>175, and PQS-D4 264>179.
3. Results and Discussion
3.1. Case Report
A 17-year-old previously healthy male touched a high voltage wire in Honduras. He sustained burns to 58% of his total body surface area (TBSA), including partial thickness burns to his face, both arms, back, and buttocks, with full thickness injury to his right lower leg and medial left thigh. His initial treatment at two different hospitals in Honduras included right lower extremity fasciotomies and multiple dressing changes. He received clindamycin, meropenem, and amikacin for an unknown period of time. He was transferred to the Shriners Boston Burns Hospital 9 days after sustaining the original injury.
Upon admission to Shriners Hospital, the patient was awake and alert but mildly febrile and tachycardic. There was a strong odor from his dressings. He was taken immediately to the operating room for debridement and stabilization. His lower extremity wounds were grossly infected with deep necrotic wounds to the right lower extremity where eschar had been partially resected. Biopsies showed diffuse inflammatory infiltrate and intramuscular rod-shaped bacteria (Figure 1(a)). Cultures from the right lower extremity revealed three P. aeruginosa isolates, a pan-resistant Acinetobacter baumannii, Klebsiella pneumoniae, Clostridium species, and Candida krusei. Initially, the Pseudomonas was susceptible to aztreonam, ciprofloxacin, levofloxacin, piperacillin/sulbactam, colistin; and it was resistant to amikacin, cefepime, gentamycin, meropenem, ticarcillin, tobramycin, and imipenem. Pseudomonas specimens from the central line tip that the patient came in with were also grown in culture. The patient was treated initially with vancomycin and meropenem as well as topical mafenide acetate. He underwent serial operative debridements over the next month. The wounds were eventually closed with autografts. He was discharged after a 2.5-month-long hospitalization.
Figure 1.
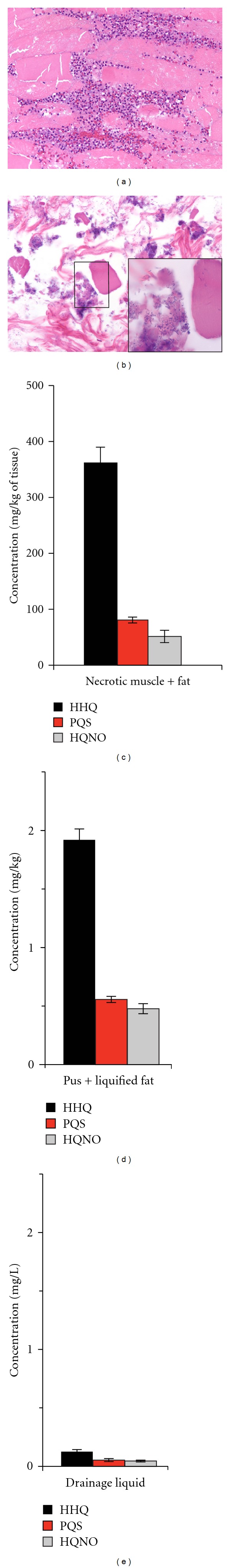
High levels of HAQs are produced in P. aeruginosa infected burn wounds. (a) Hematoxylin and eosin stained sections of right lower extremity biopsy specimen demonstrating active deep infection with myonecrosis and inflammatory infiltrate (10x) (b) Oil immersion view of the same specimen demonstrating myonecrosis with proliferating rods. Note the presence of intramuscular bacteria (inset). HHQ, PQS and HQNO levels measured in two operative specimens, necrotic muscle with fat (c) and pus and liquefied fat (d); and from the collected drainage liquid (e) obtained from an infected burn wound on the patient's right leg. Notably, HAQs were detected in the drainage liquid at a magnitude similar to that encountered in bacterial broth cultures.
3.2. Assessment of HAQs
P. aeruginosa acute burn wound infection is characterized by the presence of a diffuse inflammatory infiltrate related to muscle necrosis (Figure 1(a)), together with intracellular rode shaped bacteria (Figure 1(b) + inset). Bacteria-induced muscle necrosis and the bacteria's ability to invade muscle cells are two phenotypic characteristics that strongly rely on the coordinated expression of several bacterial virulence factors, many of which are under the control of MvfR [17]. We thus sought to determine the major MvfR ligands that are produced during burn wound infection in humans. As shown in Figures 1(c) and 1(d), we detected HHQ, PQS, and HQNO in whole muscle tissue as well as in drainage liquid. Interestingly, HHQ levels were higher than PQS levels. This result corroborates the ratios of these molecules that we measured previously in muscle biopsies from burned and P. aeruginosa infected mice [17], and are in sharp contrast to ratios measured in bacterial cell cultures [12, 13, 21], indicating that the “in vitro” bacterial culture setting does not reflect accurately the in vivo burn wound situation. Thus, in agreement with our previous results in burned and infected animals [17], we found that HHQ is produced at high levels in human burn wounds and not fully converted into PQS. PQS is converted from its precursor HHQ by the enzyme PqsH [22], whose activity is regulated by the homoserine lactone regulator LasR [22]. The low PQS levels observed in both mouse and human infected tissues might be due to reduced PqsH activity in vivo. Nevertheless, we have shown that PQS is fully dispensable in that pqsH-mutant cells, which produce HHQ but completely lack PQS, display normal MvfR-dependent gene expression and virulence in burned and infected mice [17]. Conversely, PQS is required for full production of the phenazine pyocyanin [17].
4. Conclusions
The findings presented here show for the first time that HAQs are produced during acute P. aeruginosa infections in human burn wounds. Moreover, these findings demonstrate that HAQs cell-to-cell communication molecules can be measured directly from an infected patient and that the ratio between the various HAQ molecule types resembles that found in burned and infected mice but not that produced in bacterial cell cultures. These results validate the use of burn mouse models in studies of QS-dependent P. aeruginosa acute infection. The fact that these QS low-molecular-weight molecules are detected also in drainage liquid collected from surgical sites is of the utmost importance for several reasons: (i) it demonstrates that these small molecules can affect large area of wounds and that in humans the bacteria can communicate beyond their immediate surroundings; (ii) it indicates that these molecules might affect other pathogens in polymicrobial infections [23, 24], such as in the presently reported case; (iii) it supports the hypothesis that targeting virulence and QS with selective anti-QS inhibitors may diminish infections caused by multi-drug resistant bacteria [10, 25].
The ability to accurately follow bacterial pathogenesis is an important prerequisite for the development of antivirulence agents aimed at controlling the course of infection. In this context, QS is a suitable drug target for new antimicrobials because it controls bacterial pathogenesis and it is evolutionarily conserved among pathogenic bacteria [26, 27]. Conventional antibiotics target a narrow spectrum of activities that are essential for bacterial growth. Pharmacological targeting of nonessential functions such as virulence factor production may decrease the emergence of resistance since bacterial survival is not directly challenged or subjected to selection pressure. For instance, we showed that pharmacologic inhibition of the MvfR regulon greatly reduced P. aeruginosa virulence in mice [10].
In conclusion, here we demonstrated that QS signaling molecules can be measured directly from an infected patient and that the ratio between the various HAQ molecule types resembles that found in burned and infected mice, demonstrating the importance of animal models in the evaluation of the efficacy of new drugs. These results also strengthen the validity of the hypothesis that QS inhibition may be an excellent target to prevent or halt multi-drug resistant P. aeruginosa infections.
Acknowledgments
This work was supported by the Shriners Hospitals for Children Research Grants no. 87700 and no. 87100 and National Institute of Health grant AI063433 to LGR. R. Hazen was supported by a Shriners Hospitals for Children Research Postdoctoral Fellowship (no. 8494). Y. A. Que was supported by Swiss National Science Foundation/Swiss Medical Association (FMH) Grant no. PASMP3-123226 and a grant from the SICPA Foundation. The funding agencies had no role in study design, data collection and analysis, decision to publish, or preparation of the paper. Y. A. Que and R. Hazan contributed equally to the work.
References
- 1.Branski LK, Al-Mousawi A, Rivero H, Jeschke MG, Sanford AP, Herndon DN. Emerging infections in burns. Surgical Infections. 2009;10(5):389–397. doi: 10.1089/sur.2009.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pereira C, Murphy K, Herndon D. Outcome measures in burn care: is mortality dead? Burns. 2004;30(8):761–771. doi: 10.1016/j.burns.2004.05.012. [DOI] [PubMed] [Google Scholar]
- 3.Cumming J, Purdue GF, Hunt JL, O’Keefe GE. Objective estimates of the incidence and consequences of multiple organ dysfunction and sepsis after burn trauma. Journal of Trauma. 2001;50(3):510–515. doi: 10.1097/00005373-200103000-00016. [DOI] [PubMed] [Google Scholar]
- 4.Calhoun JH, Murray CK, Manring MM. Multidrug-resistant organisms in military wounds from Iraq and Afghanistan. Clinical Orthopaedics and Related Research. 2008;466(6):1356–1362. doi: 10.1007/s11999-008-0212-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sheridan RL, Ryan CM, Pasternack MS, Weber JM, Tompkins RG. Flavobacterial sepsis in massively burned pediatric patients. Clinical Infectious Diseases. 1993;17(2):185–187. [PubMed] [Google Scholar]
- 6.Rahme LG, Stevens EJ, Wolfort SF, Shao J, Tompkins RG, Ausubel FM. Common virulence factors for bacterial pathogenicity in plants and animals. Science. 1995;268(5219):1899–1902. doi: 10.1126/science.7604262. [DOI] [PubMed] [Google Scholar]
- 7.Apidianakis Y, Rahme LG. Drosophila melanogaster as a model host for studying Pseudomonas aeruginosa infection. Nature Protocols. 2009;4(9):1285–1294. doi: 10.1038/nprot.2009.124. [DOI] [PubMed] [Google Scholar]
- 8.Starkey M, Rahme LG. Modeling Pseudomonas aeruginosa pathogenesis in plant hosts. Nature Protocols. 2009;4(2):117–124. doi: 10.1038/nprot.2008.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cao H, Krishnan G, Goumnerov B, Tsongalis J, Tompkins R, Rahme LG. A quorum sensing-associated virulence gene of Pseudomonas aeruginosa encodes a LysR-like transcription regulator with a unique self-regulatory mechanism. Proceedings of the National Academy of Sciences of the United States of America. 2001;98(25):14613–14618. doi: 10.1073/pnas.251465298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lesic B, Lépine F, Déziel E, et al. Inhibitors of pathogen intercellular signals as selective anti-infective compounds. PLoS Pathogens. 2007;3(9):1229–1239. doi: 10.1371/journal.ppat.0030126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hazan R, He J, Xiao G, et al. Homeostatic interplay between bacterial cell-cell signaling and iron in virulence. PLoS Pathogens. 2010;6(3, article e1000810) doi: 10.1371/journal.ppat.1000810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Heeb S, Fletcher MP, Chhabra SR, Diggle SP, Williams P, Cámara M. Quinolones: from antibiotics to autoinducers. FEMS Microbiology Reviews. 2011;35(2):247–274. doi: 10.1111/j.1574-6976.2010.00247.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Déziel E, Lépine F, Milot S, et al. Analysis of Pseudomonas aeruginosa 4-hydroxy-2-alkylquinolines (HAQs) reveals a role for 4-hydroxy-2-heptylquinoline in cell-to-cell communication. Proceedings of the National Academy of Sciences of the United States of America. 2004;101(5):1339–1344. doi: 10.1073/pnas.0307694100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ng WL, Bassler BL. Bacterial quorum-sensing network architectures. Annual Review of Genetics. 2009;43:197–222. doi: 10.1146/annurev-genet-102108-134304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Déziel E, Gopalan S, Tampakaki AP, et al. The contribution of MvfR to Pseudomonas aeruginosa pathogenesis and quorum sensing circuitry regulation: multiple quorum sensing-regulated genes are modulated without affecting IasRI, rhIRI or the production of N-acyl-L-homoserine lactones. Molecular Microbiology. 2005;55(4):998–1014. doi: 10.1111/j.1365-2958.2004.04448.x. [DOI] [PubMed] [Google Scholar]
- 16.Lépine F, Dekimpe V, Lesic B, et al. PqsA is required for the biosynthesis of 2,4-dihydroxyquinoline (DHQ), a newly identified metabolite produced by Pseudomonas aeruginosa and Burkholderia thailandensis. Biological Chemistry. 2007;388(8):839–845. doi: 10.1515/BC.2007.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xiao G, Déziel E, He J, et al. MvfR, a key Pseudomonas aeruginosa pathogenicity LTTR-class regulatory protein, has dual ligands. Molecular Microbiology. 2006;62(6):1689–1699. doi: 10.1111/j.1365-2958.2006.05462.x. [DOI] [PubMed] [Google Scholar]
- 18.McGrath S, Wade DS, Pesci EC. Dueling quorum sensing systems in Pseudomonas aeruginosa control the production of the Pseudomonas quinolone signal (PQS) FEMS Microbiology Letters. 2004;230(1):27–34. doi: 10.1016/S0378-1097(03)00849-8. [DOI] [PubMed] [Google Scholar]
- 19.Xiao G, He J, Rahme LG. Mutation analysis of the Pseudomonas aeruginosa mvfR and pqsABCDE gene promoters demonstrates complex quorum-sensing circuitry. Microbiology. 2006;152(6):1679–1686. doi: 10.1099/mic.0.28605-0. [DOI] [PubMed] [Google Scholar]
- 20.Lépine F, Milot S, Déziel E, He J, Rahme LG. Electrospray/mass spectrometric identification and analysis of 4-hydroxy-2-alkylquinolines (HAQs) produced by Pseudomonas aeruginosa . Journal of the American Society for Mass Spectrometry. 2004;15(6):862–869. doi: 10.1016/j.jasms.2004.02.012. [DOI] [PubMed] [Google Scholar]
- 21.Bredenbruch F, Nimtz M, Wray V, Morr M, Müller R, Häussler S. Biosynthetic pathway of Pseudomonas aeruginosa 4-hydroxy-2-alkylquinolines. Journal of Bacteriology. 2005;187(11):3630–3635. doi: 10.1128/JB.187.11.3630-3635.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gallagher LA, McKnight SL, Kuznetsova MS, Pesci EC, Manoil C. Functions required for extracellular quinolone signaling by Pseudomonas aeruginosa . Journal of Bacteriology. 2002;184(23):6472–6480. doi: 10.1128/JB.184.23.6472-6480.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mitchell G, Séguin DL, Asselin AE, et al. Staphylococcus aureus sigma B-dependent emergence of small-colony variants and biofilm production following exposure to Pseudomonas aeruginosa 4-hydroxy-2-heptylquinoline-N-oxide. BMC Microbiology. 2010;10, article 33 doi: 10.1186/1471-2180-10-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hoffman LR, Déziel E, D’Argenio DA, et al. Selection for Staphylococcus aureus small-colony variants due to growth in the presence of Pseudomonas aeruginosa . Proceedings of the National Academy of Sciences of the United States of America. 2006;103(52):19890–19895. doi: 10.1073/pnas.0606756104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rasmussen TB, Givskov M. Quorum-sensing inhibitors as anti-pathogenic drugs. International Journal of Medical Microbiology. 2006;296(2-3):149–161. doi: 10.1016/j.ijmm.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 26.Parker CT, Sperandio V. Cell-to-cell signalling during pathogenesis. Cellular Microbiology. 2009;11(3):363–369. doi: 10.1111/j.1462-5822.2008.01272.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smith RS, Iglewski BH. P. aeruginosa quorum-sensing systems and virulence. Current Opinion in Microbiology. 2003;6(1):56–60. doi: 10.1016/s1369-5274(03)00008-0. [DOI] [PubMed] [Google Scholar]


