Abstract
The level of vitamin D-binding protein (DBP) is increased in the cerebrospinal fluid of patients with Alzheimer's disease (AD), suggesting a relationship with its pathogenesis. In this study, we investigated whether and how DBP is related to AD using several different approaches. A pull-down assay and a surface plasmon resonance binding assay indicated direct interactions between purified DBP and amyloid beta (Aβ), which was confirmed in the brain of AD patients and transgenic AD model mice by immunoprecipitation assay and immunohistochemical double-staining method. Moreover, atomic force microscopic examination revealed that DBP reduced Aβ aggregation in vitro. DBP also prevented Aβ-mediated death in cultured mouse hippocampal HT22 cell line. Finally, DBP decreased Aβ-induced synaptic loss in the hippocampus and rescued memory deficits in mice after injection of Aβ into the lateral ventricle. These results provide converging evidence that DBP attenuates the harmful effects of Aβ by a direct interaction, and suggest that DBP is a promising therapeutic agent for the treatment of AD.
Keywords: Alzheimer's disease, amyloid beta, vitamin D-binding protein, cytotoxicity, neurodegeneration
Alzheimer's disease (AD) is one of the most common age-related neurological disorders. Although the causative factors underlying its pathogenesis have still not been fully elucidated, it is widely thought that amyloid beta (Aβ) peptide plays a pivotal role.1 AD-related brain damage is primarily caused by Aβ, the levels of which are strongly correlated with the extent of neurodegeneration and severity of memory deficits.2, 3 Aβ is normally present in soluble form in the plasma and cerebrospinal fluid (CSF) and aggregates and accumulates as amyloid plaques in the brain of AD patients, which has been suggested to be a critical event in the pathogenesis of AD.2 Currently, many strategies are available for the treatment of AD, and anti-Aβ therapy, which includes immunization against Aβ and treatment with amyloid-binding agents, is expected to be the most effective approach.4 CSF and plasma have been reported to contain several circulating Aβ-binding proteins, such as transthyretin (TTR), apolipoprotein E (apoE), cystatin C, and albumin, which have been shown to influence Aβ-mediated pathology,5, 6, 7, 8 and have attracted much interest. Several studies have demonstrated that Aβ-sequestering proteins may reduce the pathogenesis of AD by binding to Aβ, preventing its aggregation, and subsequently inhibiting Aβ-induced cytotoxicity. These findings suggest that Aβ-binding proteins could play a key role in the pathogenesis of AD and might constitute possible therapeutic agents for its treatment.
Vitamin D-binding protein (DBP), with a molecular weight of approximately 58 kDa, belongs to the albumin gene family and is primarily synthesized in the liver.9 It is a secretory protein that has been detected in the plasma and CSF at markedly higher concentrations than its ligands.10, 11 The nomenclature of DBP has changed several times in the past six decades because of the different biological actions that it controls, and has been known as group-specific component globulin, DBP, and DBP-macrophage-activating factor.12 DBP contains binding sites for several proteins and molecules, including vitamin D, C5a, actin, fatty acids, and endotoxin, suggesting that it is a multifunctional protein. The primary function of DBP is to act as a transport protein for vitamin D and its metabolites. In addition, it exerts several other important physiological functions, including fatty acid transport, macrophage modulation, osteoclast activation, and chemotaxis.13 One of the most important roles of DBP is to scavenge extracellular toxic actin, which is released into the extracellular space after cell death: DBP interacts with and sequesters monomeric actin and inhibits its polymerization.13 Similarly, gelsolin, another Aβ-binding protein, also interacts with and eliminates actin. A growing body of evidence suggests that DBP has excellent therapeutic potential for vitamin D-deficient conditions, cancer, liver disease, trauma, and osteopetrosis. It is currently undergoing clinical trials in these diseases, which aim to inhibit the growth of various types of cancer, protect against vitamin D intoxication, and correct skeletal defects in osteopetrosis by upregulating the oxidative metabolism in mutant cells and increasing the number of osteoclasts.10, 12, 13
Many studies have reported that changes in DBP concentration are linked to the pathology of several diseases, including hepatic failure, diabetes, and multiple sclerosis.10, 12, 13, 14 Recently, Gressner et al.11 reported that intrathecal synthesis of DBP is increased in patients with severe neurodegeneration, including AD, and suggested that upregulated DBP may act as an actin scavenger in neurodegenerative diseases. Several other studies also have shown that the level of DBP in CSF is increased in AD patients and suggested that this plays a role in the pathology of AD.15, 16 However, the functions of extracellular DBP in AD have not yet been elucidated.
These findings prompted us to hypothesize that DBP could play an important role in the pathogenesis of AD. In this study, we investigated whether DBP interacted directly with Aβ1–42 and inhibited its fibrilization, resulting in the suppression of Aβ1–42-induced AD pathogenesis in vitro and in vivo. We found that DBP interacted directly with both the Aβ1–42 monomer and oligomer, resulting in a reduced aggregation of Aβ. DBP bound to and sequestrated Aβ1–42, and prevented neuronal death induced by Aβ1–42 monomer and oligomer in vitro, suggesting that it is an inhibitor of Aβ1–42 neurotoxicity. Moreover, we used mice administered Aβ oligomers (AβO) intraventricularly as a model to investigate the preventive potential role of DBP on AβO-triggered cognitive and neuropathological outcomes. We found that DBP improved AβO-associated impairment of cognitive function by inhibiting synaptic degeneration. Our results indicate that DBP has a preventive role in AD pathogenesis by directly binding to Aβ.
Results
DBP directly interacts with Aβ
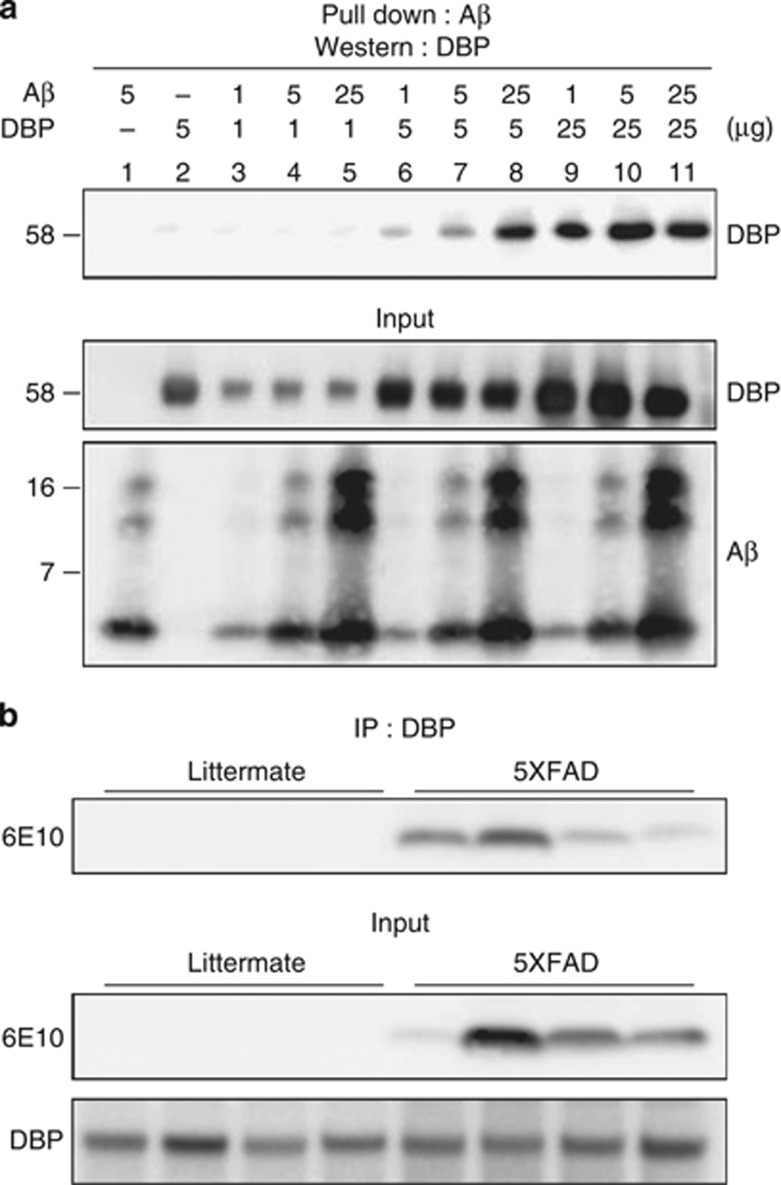
We investigated whether Aβ1–42 binds to DBP using pull-down assay and immunoprecipitation assay (IP) (Figure 1). We incubated biotinylated Aβ1–42 (1, 5, and 10 μg) with various concentrations of DBP (1, 5, and 10 μg) in test tubes. The specific interaction of DBP with Aβ was demonstrated by the pull-down assay, followed by immunoblot analysis with anti-DBP antibody. Lanes 6, 7, and 8 in the top panel of Figure 1a show the binding of DBP (5 μg) to different concentrations of Aβ1–42 (1, 5, and 10 μg), and indicating that this is dependent on the concentration of Aβ. The pulled-down amounts of DBP and biotin-Aβ1–42 are shown in the input panels of Figure 1a. We further tested whether DBP and Aβ have physiological interaction in vivo using AD animal model, 5 familial AD mutations (5XFAD) mice, which overexpresses Aβ in the brain.56 The cortex of 6-month-old 5XFAD mice was used for IP of DBP and Aβ. The specific binding of DBP with Aβ in brains of 5XFAD mice was showen by the IP, followed by immunoblot analysis with anti-Aβ-specific antibody, 6E10 (Figure 1b). No Aβ bands were detected in the littermate mice because these mice do not express human APP as well as Aβ, which is recognized by 6E10 antibody. These data indicated the direct interaction between DBP and Aβ in vitro and in vivo.
Figure 1.
DBP interacts directly with Aβ in vitro and in vivo. Interaction between DBP and Aβ using the pull-down assay and IP. (a) Various concentrations of synthetic Aβ1–42 (1, 5, and 10 μg) were mixed with DBP from human plasma (1, 5, and 10 μg) and kept at 4 °C for 12 h before pull down with anti-Aβ antibody. The assay yielded a fragment of approximately 58 kDa binding to Aβ (top panel). Input panels represent western blots for DBP and Aβ in the samples used. Molecular mass is shown on the left in kDa. (b) IP of DBP and Aβ in the cortex of 5XFAD mice. No Aβ bands were detected in the littermate mice; n=4 each cases.
DBP binds to both monomeric and oligomeric forms of Aβ1–42
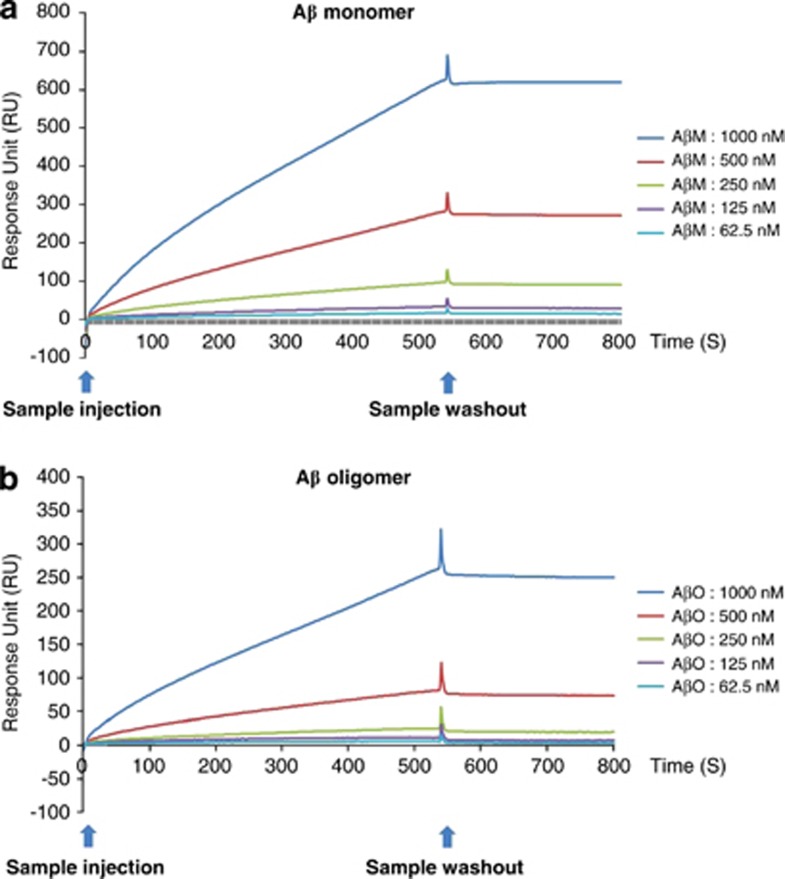
Using surface plasmon resonance (SPR) experiments, we examined the direct binding between DBP and various concentrations of monomeric and oligomeric Aβ1–42. This SPR method has been used for the highly sensitive real-time measurement of interactions between Aβ1–42 in the flow phase and DBP immobilized on the sensor chip. To study the concentration-dependent binding of Aβ1–42 to DBP, SPR experiments were conducted with various concentrations of monomeric and oligomeric Aβ1–42 (62.5, 125, 250, 500, and 1000 nM). The measurements indicated that the association of Aβ1–42 with DBP is dependent on the concentration of Aβ1–42 (Figure 2). These data demonstrated that DBP binds directly to both monomeric and oligomeric Aβ1–42with a KD of 1.265 and 108.5 nM, respectively, indicating that DBP has a higher affinity for the monomeric than for the oligomeric form of Aβ1–42.
Figure 2.
Both monomeric and oligomeric Aβ dose-dependently bind to DBP. The response curves in the SPR experiment after binding of monomeric (a) and oligomeric (b) Aβ1–42 to immobilized DBP on the sensor chip. SPR response using Biacore X-100 was elicited by the DBP indicated with Aβ concentrations of 62.5-1000 nM. RU, response unit.
DBP is accumulated in the amyloid plaques in vivo
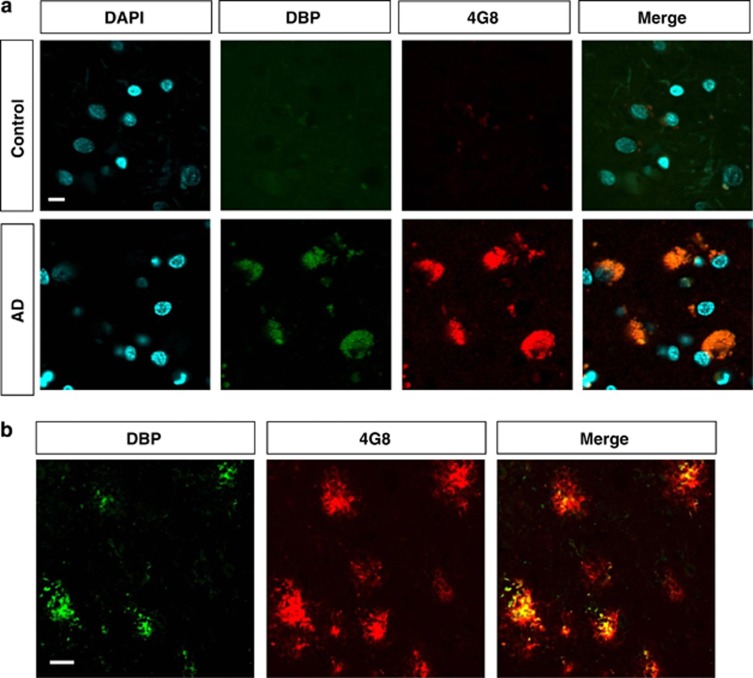
As many Aβ-binding proteins, such as TTR, apoE, apoJ, and apoA1, are known to accumulate with Aβ in amyloid deposits5, 17 and the level of DBP in CSF is increased in AD patients,11, 15, 16 we examined whether DBP is present in the amyloid plaques of brains from AD patients and transgenic animal model of AD. We double-stained human AD and control brain with antibodies against Aβ (4G8) and DBP. The immunoreactivity of DBP colocalizes with 4G8-stained plaques in the superior temporal cortex of AD patients' brains but not in the control brains (Figure 3a). Using the same antibodies, we also stained the brains of 6-month-old 5XFAD mice and observed that DBP coincides with Aβ in the frontal cortex of 5XFAD mice (Figure 3b). Consistent with a previous report using same DBP antibody (H300: sc-32899; Santa Cruz Biotechnology, Santa Cruz, CA, USA),18 DBP-expressing cells were detected in the hypothalamus of 6-month-old 5XFAD mice, demonstrating the specificity of DBP immunoreactivity (Supplementary Figure 1). Aβ accumulation was not detectable in the hypothalamus of 6-month-old 5XFAD mice. These data showed that DBP is present in amyloid deposits of AD brains, implying that endogenously produced DBP can interact with endogenous Aβ in vivo.
Figure 3.
Colocalization of DBP and Aβ accumulation in AD brain. (a) Immunoreactivity of DBP and Aβ in the superior temporal cortex of human AD and control brains. No amyloid plaques were detected in the control human brains. DBP was colocalized with the amyloid plaques in brain of AD patients. Scale bar=10 μm. (b) In the cortex of 6-month-old 5XFAD mice, immunoreactivity against DBP was detected in the center of 4G8-stained plaques. Scale bar=20 μm.
DBP inhibits the aggregation and oligomerization of Aβ1–42
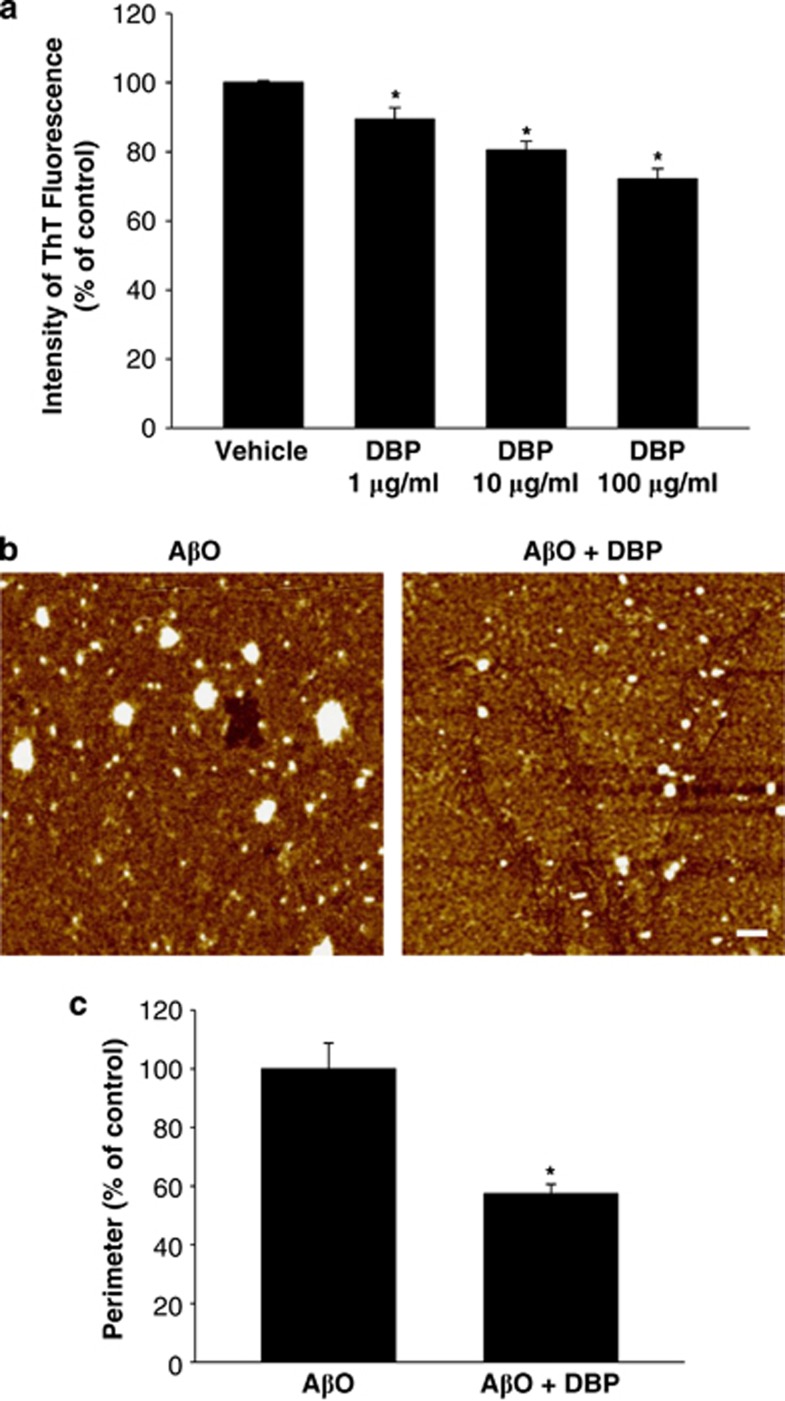
Several Aβ-binding proteins have been reported to be negative regulators of Aβ polymerization.8, 19, 20, 21 Using thioflavin T (ThT) fluorescence assay, we assessed whether Aβ-binding DBP could affect the Aβ aggregation. DBP (1, 10, and 100 μg/ml) treatment showed the dose-dependent inhibition of aggregation of monomeric Aβ1–42 (10 μM), as demonstrated by reduced ThT fluorescence intensity (Figure 4a). To further study the inhibitory effects of DBP on Aβ1–42 polymerization, we examined the Aβ1–42 oligomerization by quantifying the oligomer size using atomic force microscopy (AFM). Analysis of AFM images showed that the initial monomeric Aβ1–42 (10 μM) spontaneously polymerized into larger globular aggregates over 24 h. The size and morphology of oligomeric Aβ1–42 particles were similar to those reported previously.22 When DBP (100 μg/ml) was incubated with monomeric Aβ1–42, its formation was significantly attenuated (Figure 4b). Quantitative data revealed that the average perimeter of particles in the samples supplemented with DBP was markedly reduced compared with that of control samples incubated without DBP (Figure 4c). These results suggest that DBP interacts with and/or sequestrates Aβ monomers and oligomers, resulting in a reduction in Aβ1–42 aggregation and oligomerization.
Figure 4.
DBP inhibits the aggregation of Aβ. (a) Fluorescence emission intensity of ThT in the presence or absence of DBP. The extent of aggregation of monomeric Aβ1–42 peptides on incubation with vehicle and DBP (1, 10, and 100 μg/ml) was examined using ThT assay. Data are the mean±S.E.M. *P<0.05 versus vehicle-treated group. (a and b) The aggregation of Aβ peptides (white particles) on incubation with and without DBP for 24 h was imaged using AFM. (b) Representative AFM images of oligomeric Aβ1–42 are shown. (c) The average perimeter of detectable oligomeric Aβ1–42 particles in each sample was quantified by Image J software (National Institutes of Health, Bethesda, MD, USA). The differences in aggregation of monomeric Aβ1–42 are represented as mean±S.E.M. *P<0.05 versus AβO- and vehicle-treated group. Scale bar=20 nm.
DBP attenuates Aβ-induced neuronal cell death in vitro
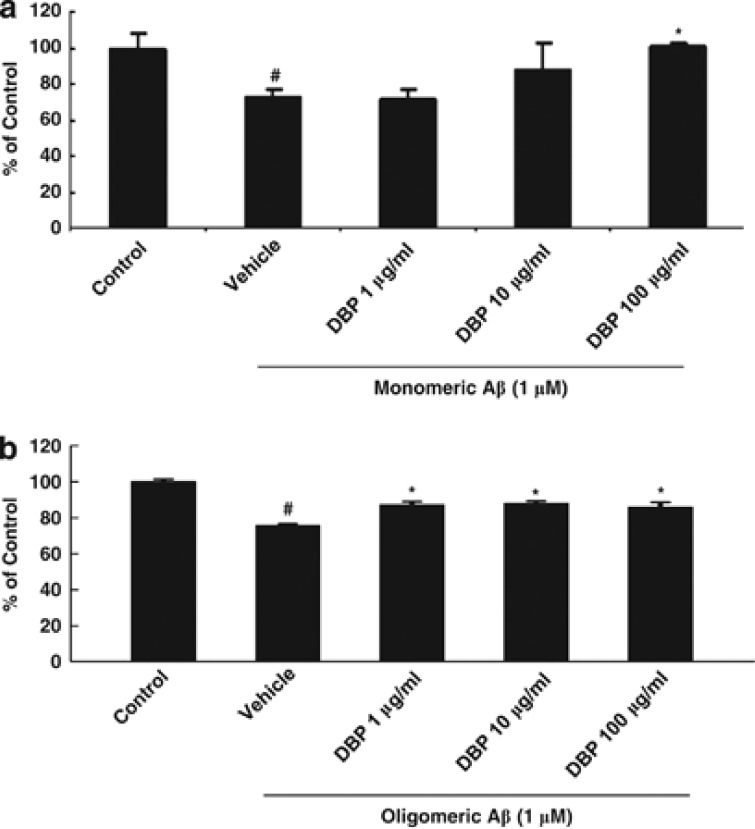
Neuronal loss and atrophy are one of the hallmarks of and cause of memory deficits in AD in humans.23, 24 Recent studies showed that Aβ-binding heat-shock proteins, which prevent Aβ aggregation and oligomerization, inhibit Aβ-induced neuronal death.19, 25 To elucidate the effect of DBP on the cytotoxicity of Aβ, we examined whether DBP influences Aβ-mediated neuronal death using the MTT assay, MTS assay, and DAPI staining. Treatment of the mouse hippocampal cell line, HT22, with both monomeric (Figure 5a and Supplementary Figure 2) and oligomeric (Figure 5b) forms of Aβ significantly reduced the cell viability within 24 h. The exposure of HT22 cells to DBP (1, 10, and 100 μg/ml) 30 min before the addition of various concentrations of Aβ significantly inhibited Aβ-induced neuronal death (Figure 5 and Supplementary Figure 2). Treatment with DBP alone did not show any significant effects (data not shown). Our data suggest that binding of DBP to both monomeric and oligomeric Aβ leads to the suppression of Aβ toxicity in mouse hippocampal HT22 cell line.
Figure 5.
DBP rescues HT22 cells from cell death induced by both monomeric and oligomeric Aβ. HT22 cells were pretreated with different concentrations of DBP (1, 10, and 100 μg/ml), followed by administration of Aβ monomer (a) and oligomer (b) for 24 h. Using the MTT assay, the viability of HT22 cells exposed to both monomeric (a) and oligomeric (b) forms of Aβ was evaluated by MTT reduction. Data are the mean±S.E.M. #P<0.05 versus vehicle-treated control cells; *P<0.05 versus Aβ- and vehicle-treated cells.
DBP prevents AβO-induced memory impairment in vivo
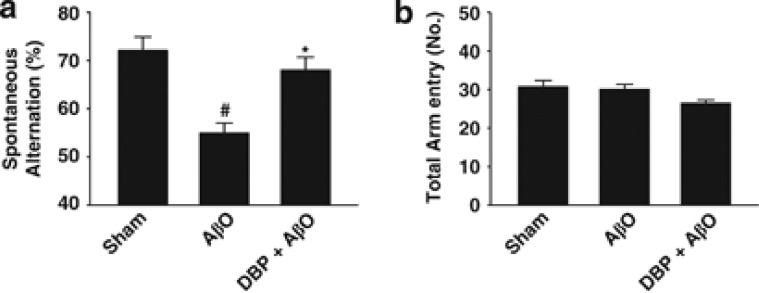
To study the effect of DBP on Aβ toxicity in vivo, we examined the effect of DBP on Aβ-induced cognitive dysfunction in mice. AβO, a major cause of memory impairment in AD,26 was used to disrupt memory function in mice. Previous reports have shown that Aβ-binding proteins or agents reduce the impairment of cognitive function in models of AD.5, 27 We therefore tested whether injection of DBP into the brain inhibits AβO-induced cognitive impairment. To analyze cognitive function and cognitive task, the Y-maze test was performed. In the Y-maze test, spontaneous alternation was recorded to evaluate spatial and short-term memory, and showed that AβO-injected mice had a lower percentage of spontaneous alternation than sham-operated controls (Figure 6a). Moreover, the AβO-induced reduction in levels of spontaneous alternation was significantly inhibited by intracerebroventricular injection of 20 μg DBP (Figure 6a). We observed no significant difference in the total number of arm entries during the Y-maze test in any of the groups, suggesting similar levels of motor function and exploratory activity (Figure 6b). Taken together, the cognitive test shows that central injection of DBP, which binds to AβO, attenuates AβO-induced impairment of cognitive function.
Figure 6.
Injection of DBP into the brain prevents memory deficits following intracerebroventricular injection of AβO. Cognitive function was assessed using a Y-maze task 6 days after stereotaxic surgery. The percentage of spontaneous alternation (a) and the numbers of arm entries (b) in the Y-maze test were estimated. Values are expressed as the mean±S.E.M. (n=10). *P<0.05 versus the AβO-treated group; #P<0.05 versus the sham-operated group (compared by analysis of variance (ANOVA) and the post hoc Fisher's least significant difference (LSD) test).
DBP prevents AβO-mediated synaptic degeneration in the hippocampus
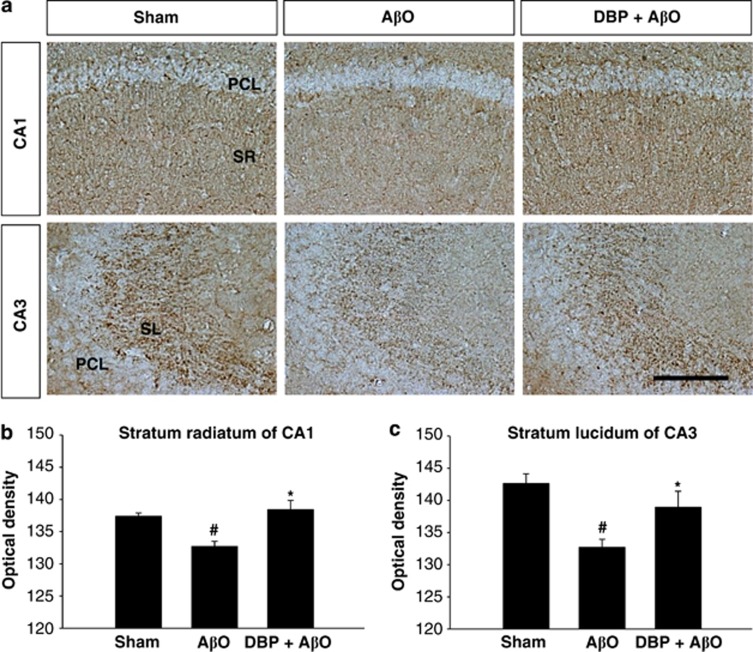
In AD, memory deficits are strongly correlated with levels of expression of synaptic proteins28, 29 and hippocampal synaptic density.30 Recent studies have shown that AβO binds to dendritic spines and induces synaptic degeneration.31 We therefore hypothesized that DBP directly interacts with AβO, blocks its subsequent binding to synaptic target structures in the hippocampus, and prevents its synaptotoxicity. We therefore determined whether DBP inhibits AβO-mediated synaptic loss. After immunostaining with antibody to synaptophysin (Figure 7) and postsynaptic density protein 95 (PSD-95) (Supplementary Figure 3), optical density was measured to compare synaptic density. Quantification of immunoreactivity for synaptophysin and PSD-95 demonstrated that its expression was significantly decreased in both the CA1 stratum radiatum and the CA3 stratum lucidum of the hippocampus in AβO-injected mice compared with the sham-operated group (Figure 7 and Supplementary Figure 3). Centrally administered DBP markedly attenuated the AβO-mediated reduction in synaptic density in the CA1 and CA3 subfields of the hippocampus (Figure 7 and Supplementary Figure 3). These data indicate that Aβ-binding DBP acts as an inhibitor of the synaptic toxicity induced by AβO.
Figure 7.
Administration of DBP inhibits the loss of synaptophysin in brains injected with AβO. (a) Representative hippocampal images of synaptophysin-stained brain. Note that mice injected with DBP plus AβO showed a higher expression level of synaptophysin in CA1 and CA3 subfields than those injected with AβO only. Quantification of the average intensity of synaptophysin immunoreactivity in the stratum radiatum of CA1 (b) and the stratum lucidum of CA3 (c) (n=10). *P<0.05 versus the AβO-treated group; #P<0.05 versus the sham-operated group. PCL, pyramidal cell layer. Scale bar=100 μm.
Discussion
Although the level of DBP in CSF has been known to be increased in AD patients, and several lines of evidence have suggested that DBP plays a role in AD,11, 15, 16, 32 there is no direct evidence of whether or how DBP affects AD-related pathology. Our data clearly show that DBP interacts directly with Aβ and reduces the formation of aggregated Aβ. Moreover, it protects HT22 cells from Aβ-induced death in vitro. Interestingly, central injection of DBP improves the cognitive dysfunction induced by AβO and protects against the reduction in hippocampal synaptic density in mice injected intracerebroventricularly with AβO. These results suggest that DBP has a functional role in binding to and/or sequestering Aβ and has multiple therapeutic effects on AD by inhibiting the formation of toxic AβO and protecting against the neurodegeneration and memory impairment induced by Aβ toxicity.
DBP is known to have a variety of physiological functions, including the transport of vitamin D, fatty acid transport, immune modulation, osteoclast activation, and chemotaxis,13 which are mediated by binding to various proteins, such as actin, lipoprotein and vitamin D, fatty acids, and endotoxin. In this study, we observed that DBP from human plasma and CSF interacted with Aβ, suggesting that DBP possibly acts as an Aβ-transporter protein in the circulation. To our knowledge, this is the first study to demonstrate that DBP plays a role in Aβ transport or Aβ binding. We propose that the major function of DBP is to bind to and sequester Aβ and form DBP–Aβ complexes under physiological conditions. The binding sites for several proteins have been identified within DBP: vitamin D – between residues 35 and 49; C5a – between residues 126 and 175; and actin – between residues 373 and 403. More detailed studies are needed to determine the Aβ-binding domain in DBP and the DBP-binding region in Aβ.
Soluble Aβ is a natural metabolic product, and is present in the plasma and CSF of normal subjects and AD patients.33 Because it is a crucial factor in the pathogenesis of AD, targeting Aβ is therefore an effective therapeutic strategy, such as by Aβ vaccination or anti-Aβ immunotherapy. It has been suggested that only free or unbound Aβ can participate in the formation of toxic aggregates.8 Therefore, some Aβ-binding molecules and proteins may play a role in the prevention of AD pathogenesis. CSF and plasma contain several circulating Aβ-binding proteins, such as TTR, apoE, cystatin C, heat-shock proteins, and albumin, which have been suggested to be therapeutic in AD.5, 6, 7, 8, 25 Endogenous Aβ-binding proteins have been shown to (1) sequester Aβ, resulting in an increase in the fraction of bound Aβ and a decrease in the level of free Aβ, (2) inhibit the aggregation and polymerization of Aβ, thus preventing amyloid plaque formation, and (3) suppress Aβ-induced cytotoxicity.5, 6, 7, 8 DBP is an abundant protein in both plasma and CSF,10, 11, 34 and our results show that, similar to other known circulating proteins that sequester Aβ, secretory DBP binds to Aβ and reduces Aβ-mediated pathology. We have therefore discovered a novel endogenous Aβ-binding protein with therapeutic potential.
The ThT assay and AFM analysis showed that DBP inhibits oligomerization of Aβ, and may bind to a site or sites that are crucial for Aβ polymerization and/or aggregation. Some residues in Aβ peptides are crucial for Aβ aggregation, fibrilization, and conformational change.35, 36 Because of its Aβ-binding function, DBP has a physiological significance in the pathogenesis of AD, but the contribution of the DBP–Aβ interaction required elucidation. We first determined that DBP can bind to Aβ and inhibit Aβ oligomerization, and can influence Aβ-mediated neuronal death in vitro. Using the cell viability and toxicity assays, we found that DBP has a protective effect on Aβ-induced death of mouse hippocampal HT22 cell line (Figure 5). One possible explanation for this effect is that DBP binds to monomeric Aβ species, and blocks the formation of the highly toxic oligomeric species (Figures 2a, 4, and 5a). Second, DBP–AβO binding might mask regions that are required for its interaction with cellular targets and inhibit further aggregation or growth of AβO (Figures 2b, 4, and 5b). Third, DBP might sequester AβO and subsequently reduce its binding to receptors and uptake (Figures 2b and 5b).
In behavioral and histological studies, we demonstrated the protective effects of DBP on AβO-induced memory impairment and neurodegeneration in vivo (Figures 6 and 7). Intracerebroventricular injection of AβO is known to result in memory deficits and synaptic loss in rodents,37 and mice administered AβO in our study had cognitive impairment and a reduction in synaptic density in the hippocampus as shown by the Y-maze test and immunohistochemistry of synaptic markers, respectively. Interestingly, these cognitive and synaptic deficits were significantly improved by administration of DBP. The inhibiting effects of Aβ-binding DBP on cognitive impairment is consistent with a previous study in which TTR, an Aβ-binding protein, attenuated Aβ-mediated deficits in cognitive function in the APP23 transgenic mouse model of AD.5 Because memory and synaptic deficits in AD are mainly caused by soluble AβO, the protective effects of DBP in a mouse model of AD constructed by intracerebroventricular injection of AβO can probably be attributed to the sequestration of AβO and subsequent blockage of its binding to the synapse, resulting in the inhibition of AβO-associated pathology. However, no significant neuronal loss was detected in the mice injected with AβO (data not shown).
The identification of changes in the level of circulating proteins in the plasma and CSF of AD patients supports the association between these proteins and the pathogenesis of AD. In AD patients, the concentration of circulating Aβ-binding proteins, such TTR, β-trace, cystatin C, apoE, and albumin, is significantly altered compared with non-demented subjects.32, 38, 39 Many studies have also shown alterations in the concentration of DBP in the CSF of AD patients, who demonstrated upregulation of DBP expression.11, 15, 16 Based on these reports, Aβ-binding proteins, including DBP, might provide effective diagnostic tools or biomarkers for AD.32, 38, 39 It has been suggested that upregulation of neuroprotective and Aβ-sequestering proteins, such as ADNP and cystatin C, in response to AD represents an intrinsic compensatory response that may counteract the progression of the disease.40, 41 We propose that the imbalance in the expression level or failure (or deficit) in the Aβ-chaperone activity of Aβ-sequestering proteins, including DBP, may lead to the development of sporadic late-onset AD.
In addition to its Aβ-binding effects, DBP may be relevant to numerous other pathological mediators of AD. There are several lines of evidence that an increase in extracellular actin in the CSF, vitamin D deficiency, neuroendothelial disruption, and immune dysfunction are commonly associated with AD pathology.42, 43, 44, 45 Interestingly, DBP is known to act as an actin scavenger, a vitamin D transporter, a growth factor for endothelial cells, an immunmodulator, and a macrophage activator.10, 46 Therefore, it is possible that DBP may improve the synaptic loss and inhibit memory deficits through the regulation of other pathological mediators of AD. There are no DBP-positive cells in the extrahypothalamic areas, including the cortex (Figure 3) and hippocampus (data not shown). Since DBP is a secreted protein, DBP produced in hypothalamic neurons (Supplementary Figure 1) might have functional effects in brain areas with more direct relevance to AD.
Although we showed that DBP has preventive effects on AβO-mediated pathology in vivo and DBP directly binds to Aβ and inhibits aggregation in vitro, there is no direct evidence to elucidate whether DBP affects Aβ aggregation in vivo. Further work, such as generation of new DBP transgenic mouse models, will be needed to determine if DBP actually influences Aβ metabolism or Aβ aggregation in vivo.
In conclusion, we have demonstrated for the first time that the direct binding of DBP to Aβ suppresses Aβ-related pathology in vitro and in vivo. Our results suggest that increasing the levels of DBP may be a promising therapeutic strategy for AD.
Materials and Methods
Pull-down assay
Various concentrations (1, 5, and 25 μg) of biotin-Aβ1–42 (American Peptide, Sunnyvale, CA, USA) and DBP from human plasma (Sigma, St. Louis, MO, USA) were mixed in binding buffer (50 mM Tris, pH 7.5, 300 mM NaCl, 0.05% NP-40) and incubated at 4 °C for 12 h to allow the formation of Aβ–DBP complexes. The samples were then incubated with streptavidin-Sepharose beads (GE Healthcare, Piscataway, NJ, USA) in a rotating shaker at 4 °C for 2 h. Subsequently, proteins were boiled in 2 × SDS sample buffer at 100 °C and were loaded onto 15% Tris-glycine gel, followed by analysis with western blot. DBP (58 kDa) bound to Aβ was immunodetected using a rabbit anti-human DBP polyclonal antibody (1 : 1000; Santa Cruz Biotechnology, Santa Cruz, CA, USA). We also found immunopositive Aβ bands using an anti-Aβ monoclonal antibody 6E10 (Covance, Emeryville, CA, USA).
SPR spectroscopy
The SPR protocol for the study of real-time direct binding of Aβ and DBP was essentially similar to that described previously47, 48 and was performed using a Biacore X-100 (GE Healthcare). The CM5 sensor chip (GE Healthcare) was preactivated by a mixture of N-hydroxysuccinimide and N-ethyl-N'-(dimethylaminopropyl) carbodiimide. DBPs (20 μg/ml; Sigma) were immobilized separately by N-ethyl-N'-(dimethylaminopropyl) carbodiimide –N-hydroxysuccinimide amine coupling on the CM5 sensor chip. As a control, the same volume of HBS-EP buffer was applied to the chip surface and the unreached sites were blocked by injection of ethanolamine. To analyze the binding kinetics, different concentrations of Aβ diluted in HBS-EP buffer were injected onto the sensor chip for 540 s at 10 μl/min, and the response unit (RU) was then recorded at 25 °C. After injection of the analyte was stopped, HBS-EP buffer was poured over the chip for 700 s at 10 μl/min to allow the bound analytes to dissociate from the immobilized DBP, and dissociation curves were obtained. The RU elicited by injecting HBS-EP buffer alone was used as the control. Biacore X-100 software (GE Healthcare) was used to record the changes in RU and to plot the binding curve. The curves obtained from the SPR experiments were analyzed and the dissociation equilibrium constant (KD) of Aβ to immobilized DBP was calculated using kinetic evaluation software. The dissociation constant KD (M) was derived from the equation, KD=kd/ka, where kd and ka are dissociation and association rate constants, respectively.
Preparation of monomeric and oligomeric Aβ1–42 solutions and aggregation assay using AFM and ThT assay
To obtain a solution of Aβ1–42 monomers, lyophilized Aβ1–42 (American Peptide) was dissolved in 1,1,1,3,3,3-hexafluoroisopropanol (Sigma-Aldrich, St. Louis, MO, USA), evaporated, re-dissolved in dimethyl sulfoxide (DMSO) (Sigma-Aldrich), and then diluted in PBS (pH 7.4) to yield a final concentration of 1 mM. Solutions of oligomeric Aβ1–42 were prepared as described previously.49 To prepare the oligomer solution, monomeric Aβ1–42 peptides were incubated with PBS at 4 °C for 24 h and immediately stored in aliquots at −80 °C. To examine the effect of DBP on oligomeric Aβ1–42 formation, peptide solutions containing Aβ1–42 monomers (10 μM) with and without DBP (100 μg/ml) were incubated at 4 °C for 24 h. DBP-free samples were treated with the same volume of distilled water. The resulting oligomers were examined by AFM (PARK Systems Inc., Suwon, South Korea) as described previously.48 To estimate oligomer size using AFM, peptide solutions were aliquoted from reaction mixtures and immediately spotted onto freshly cleaved micas. The micas were then rinsed with water two times, and dried in air. AFM images were examined to quantify the size of oligomeric Aβ1–42, the average perimeter of detectable white particles in each image, using Image J program (National Institutes of Health). The effect of DBP on aggregation of monomeric Aβ1–42 was examined using ThT assay as described previously.50 The solutions of monomeric Aβ1–42 (5 μl of 100 μM in DMSO) and 5 μl of DBP (1, 10, and 100 μg/ml in PBS) or PBS were added to 40 μl PBS at pH 7.4. The resulting mixture was incubated for 2 h at room temperature (RT). To the resulting mixture, 150 μl of ThT solution (5 μM in 50 mM glycine–NaOH at pH 8.5) was added. The ThT fluorescence was measured at 490 nm with excitation at 450 nm in a Tecan Infinite M200 Fluorescence Microplate Reader (Männendorf, Switzerland).
Cell cultures and cell death assay
To study the effect of DBP on Aβ toxicity, we used mouse hippocampal HT22 cells (donated by Dr. David Schubert at Salk Institute, La Jolla, CA, USA) exposed to Aβ as an in vitro model of AD. The cells were incubated with 1 or 5 μM Aβ1–42 for 24 h in the presence or absence of DBP (1, 10, and 100 μg/ml). DBP peptides were pretreated 30 min before the addition of Aβ1–42. The degree of cell viability or cell survival was measured using the MTT assay, MTS assay, and DAPI staining as described previously.51, 52, 53 The relative reduction of MTT and MTS was quantified by measuring optical density at 560 nm using a plate reader (Bio-Tek Instruments, Winooski, VT, USA). DAPI-stained cells are counted using Image-Pro Plus Version 6.0 (Media Cybernetics, Bethesda, MD, USA). The MTT assay, MTS assay, and DAPI staining were performed in triplicate and independently repeated two times. Data are presented as a percentage of control, vehicle-treated cells.
Stereotaxic injection of AβO and DBP into mouse brains
Male C57BL/6 mice (22–25 g, 8 weeks of age) were used for all of the experiments. They were given access to water and food ad libitum and were housed in a controlled environment at a constant temperature (23±1 °C) with a 12 h light/dark cycle. All animal procedures were carried out in accordance with the Principle of Laboratory Animal Care (NIH Publication No. 85-23, revised 1985) and the Animal Care and Use Guidelines of Kyung Hee University (Seoul, Korea). The AβO-injected animal model of AD was constructed according to a previous method.22 All stereotaxic surgical procedures were performed on anesthetized mice (mixture of Zoletil 50 (Virbac, Carros, France) and Rompun (Bayer Korea, Seoul, Korea), 3 : 1 ratio, 1 ml/kg intraperitoneally) that were mounted in a stereotaxic apparatus (myNeuroLab, St. Louis, MO, USA). Soluble AβO (10 μM in 3 μl) was injected into the lateral ventricle (−0.6 mm anterior–posterior (AP), 1.2 mm medial–lateral (ML), and −2 mm dorsal–ventral (DV)), relative to the bregma using a Mouse Brain Atlas,54 at a rate of 0.5 μl/min. Sham-operated groups were administered with the same volume of saline. To study the effects of DBP in AβO-injected mice, 3 μl DBP (20 μg) were also injected into the lateral ventricle (−0.4 mm AP, 1.2 mm ML, and −2 mm DV) 20 min before administration of AβO. Control animals were injected in parallel with saline.
Behavioral memory impairment test
Spontaneous alternation Y-maze was performed as described previously.22, 55 Short-term spatial recognition memory was investigated using the Y maze 6 days after stereotaxic surgery. The Y maze is composed of three arms with equal angles between the arms. The floor and walls of the maze were made of dark opaque polyvinyl plastic. Mice were placed in one arm, and were allowed to explore the maze freely. The alternation and the total number of arm entries (locomotor activity) were counted manually for each mouse during an 8-min session. Alternation was defined as consecutive entries into all three arms without repetition. The percentage of alternations was calculated as follows: ((the number of alternations)/(the total number of arms entered–2)) × 100.
Immunohistochemistry and quantification of immunoreactivity
For histological examination of the brain, mice were killed 8 days after stereotaxic surgery. Animals were anesthetized with a mixture of Zoletil and Rompun solution (3 : 1 ratio, 1 ml/kg intraperitoneally) and were then transcardially perfused with a cold solution of 4% paraformaldehyde in PBS. The brains were dissected and postfixed for 20 h in the same fixative solution before being immersed in a cryoprotectant solution of 30% sucrose in PBS. The brain tissues were cut into 30-μm-thick coronal sections with a cryostat (Leica, Nussloch, Germany) at −25 °C and stored at 4 °C. Four brain sections from each mouse were taken from the region between −1.3 and −2 mm relative to the bregma.54 The free-floating brain sections were then pretreated with 1% hydrogen peroxide for 15 min to inhibit endogenous peroxidase. Tissue sections were then incubated at 4 °C overnight with mouse anti-synaptophysin antibody (1 : 200; Sigma-Aldrich) or rabbit anti-PSD-95 antibody (1 : 1000; Abcam, Cambridge, MA, USA). Subsequently, the sections were incubated with biotinylated horse anti-mouse IgG antibody or biotinylated goat anti-rabbit IgG antibody (1 : 200; Vector Laboratories, Burlingame, CA, USA) and avidin–biotin–peroxidase complex solution and then visualized with 3,3′-diaminobenzidine as the chromogen.
To quantify immunoreactivity, the images were processed and analyzed using ImageJ program. The analysis was performed blindly in both hemispheres of four brain sections per animal. To measure the optical density of synaptophysin and PSD-95 immunoreactivity, the region of interest – the stratum radiatum of CA1 and the stratum lucidum of CA3 – was manually outlined and the average optical densities were acquired in the captured 8-bit grayscale images.
Double immunolabeling for DBP and Aβ in the brain of AD patients and 5XFAD mice
Human brain samples were obtained from Boston University Alzheimer's Disease Center and followed guidelines by Boston University School of Medicine. The detail information of brain tissues is described in Supplementary Table 1. Transgenic mice with 5XFAD used in this study were purchased from The Jackson Laboratory (Bar Harbor, ME, USA). These mice overexpressing Aβ peptide express both mutant human amyloid precursor protein (695) (with the Swedish mutation (K670N, M671L), Florida mutation (I716V), and London mutation (V717I)) and human presenilin 1, harboring two FAD mutations (M146L and L286V).56 Male animals were killed at 6 months of age (n=4) and brain tissue was prepared as described above.
To detect the immunofluorescence of Aβ and DBP in human and mouse brain tissue, free-floating sections were pretreated with 70% formic acid for 20 min and then incubated with mouse anti-human Aβ, biotin-labeled 4G8 (1 : 3000; Covance, Princeton, NJ, USA), and rabbit anti-human DBP (1 : 100; Santa Cruz Biotechnology) overnight at 4 °C. Following extensive washes in PBS, the sections were incubated with Alexa 594-conjugated streptavidin (1 : 500; Molecular Probes, Eugene, OR, USA) and goat anti-rabbit Alexa 488 (1 : 500; Molecular Probes, Eugene, OR, USA), respectively, for 1 h at RT. The signals were observed and analyzed on a confocal laser scanning microscope (FluoView FV10i; Olympus, Center Valley, PA, USA).
IP for DBP and Aβ in the brain of 5XFAD mice
Transgenic mice with 5XFAD were used in this study (Jackson laboratory).56 Male animals were killed at 6 months of age (n=4) and the brain cortex region was used for IP. Brain tissues were lysed in RIPA buffer (50 mM Tris-HCl, pH 7.5, 150 mM sodium chloride, 0.5% sodium deoxycholate, 0.1% SDS, 1% Triton X-100, 2 mM EDTA) containing protease inhibitors (Sigma-Aldrich), and lysates were centrifuged at 17 000 × g for 15 min. Equal amounts of protein were precipitated with the anti-DBP antibodies at 4 °C overnight on a rotator. Protein A/G agarose beads (Santa Cruz Biotechnology) were added to each sample and incubated at 4 °C for 1 h. IPs were collected by centrifugation and then washed four times with the same buffer. The agarose beads were resuspended in 6 μl of 4 × sample buffer and incubated at 95 °C for 10 min to release the proteins. After a pulse spin, the supernatants were loaded on SDS-PAGE for western blotting.
Statistical analysis
The significance of differences between the groups was defined by one-way analysis of variance, followed by the Fisher's LSD post hoc test or Student's t-test using SigmaStat for Windows Version 3.10 (Systat Software Inc., Point Richmond, CA, USA). Differences were considered to be significant for values of P<0.05. All values are shown as mean±standard error of the mean (S.E.M.).
Acknowledgments
This work was supported by grants from National Research Foundation 2012R1A2A1A01002881, World Class University-Neurocytomics (R32-10084), and the Korean National Institute of Health R&D Program Project (A092058), and supported, in part, by the Basic Research Program (2008-05943) and MRC at SNU (2011-0030738) by NRF.
Glossary
- DBP
vitamin D-binding protein
- TTR
transthyretin
- AD
Alzheimer's disease
- Aβ
amyloid beta
- AβO
Aβ oligomers
- CSF
cerebrospinal fluid
- IP
immunoprecipitation assay
- ThT
thioflavin T
- AFM
atomic force microscopy
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Cell Death and Differentiation website (http://www.nature.com/cdd)
Edited by N Bazan
Supplementary Material
References
- Selkoe DJ. Alzheimer's disease: a central role for amyloid. J Neuropathol Exp Neurol. 1994;53:438–447. doi: 10.1097/00005072-199409000-00003. [DOI] [PubMed] [Google Scholar]
- Sakono M, Zako T. Amyloid oligomers: formation and toxicity of Abeta oligomers. FEBS J. 2010;277:1348–1358. doi: 10.1111/j.1742-4658.2010.07568.x. [DOI] [PubMed] [Google Scholar]
- Cleary JP, Walsh DM, Hofmeister JJ, Shankar GM, Kuskowski MA, Selkoe DJ, et al. Natural oligomers of the amyloid-beta protein specifically disrupt cognitive function. Nat Neurosci. 2005;8:79–84. doi: 10.1038/nn1372. [DOI] [PubMed] [Google Scholar]
- Golde TE, Schneider LS, Koo EH. Anti-abeta therapeutics in Alzheimer's disease: the need for a paradigm shift. Neuron. 2011;69:203–213. doi: 10.1016/j.neuron.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buxbaum JN, Ye Z, Reixach N, Friske L, Levy C, Das P, et al. Transthyretin protects Alzheimer's mice from the behavioral and biochemical effects of Abeta toxicity. Proc Natl Acad Sci USA. 2008;105:2681–2686. doi: 10.1073/pnas.0712197105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeMattos RB, Cirrito JR, Parsadanian M, May PC, O'Dell MA, Taylor JW, et al. ApoE and clusterin cooperatively suppress Abeta levels and deposition: evidence that ApoE regulates extracellular Abeta metabolism in vivo. Neuron. 2004;41:193–202. doi: 10.1016/s0896-6273(03)00850-x. [DOI] [PubMed] [Google Scholar]
- Mi W, Pawlik M, Sastre M, Jung SS, Radvinsky DS, Klein AM, et al. Cystatin C inhibits amyloid-beta deposition in Alzheimer's disease mouse models. Nat Genet. 2007;39:1440–1442. doi: 10.1038/ng.2007.29. [DOI] [PubMed] [Google Scholar]
- Bohrmann B, Tjernberg L, Kuner P, Poli S, Levet-Trafit B, Naslund J, et al. Endogenous proteins controlling amyloid beta-peptide polymerization. Possible implications for beta-amyloid formation in the central nervous system and in peripheral tissues. J Biol Chem. 1999;274:15990–15995. doi: 10.1074/jbc.274.23.15990. [DOI] [PubMed] [Google Scholar]
- White P, Cooke N. The multifunctional properties and characteristics of vitamin D-binding protein. Trends Endocrinol Metab. 2000;11:320–327. doi: 10.1016/s1043-2760(00)00317-9. [DOI] [PubMed] [Google Scholar]
- Speeckaert M, Huang G, Delanghe JR, Taes YE. Biological and clinical aspects of the vitamin D binding protein (Gc-globulin) and its polymorphism. Clin Chim Acta. 2006;372:33–42. doi: 10.1016/j.cca.2006.03.011. [DOI] [PubMed] [Google Scholar]
- Gressner OA, Schifflers MC, Kim P, Heuts L, Lahme B, Gressner AM. Questioning the role of actinfree Gc-globulin as actin scavenger in neurodegenerative central nervous system disease: relationship to S-100B levels and blood-brain barrier function. Clin Chim Acta. 2009;400:86–90. doi: 10.1016/j.cca.2008.10.015. [DOI] [PubMed] [Google Scholar]
- Gomme PT, Bertolini J. Therapeutic potential of vitamin D-binding protein. Trends Biotechnol. 2004;22:340–345. doi: 10.1016/j.tibtech.2004.05.001. [DOI] [PubMed] [Google Scholar]
- Meier U, Gressner O, Lammert F, Gressner AM. Gc-globulin: roles in response to injury. Clin Chem. 2006;52:1247–1253. doi: 10.1373/clinchem.2005.065680. [DOI] [PubMed] [Google Scholar]
- Blanton D, Han Z, Bierschenk L, Linga-Reddy MV, Wang H, Clare-Salzler M, et al. Reduced serum vitamin d-binding protein levels are associated with type 1 diabetes. Diabetes. 2011;60:2566–2570. doi: 10.2337/db11-0576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdi F, Quinn JF, Jankovic J, McIntosh M, Leverenz JB, Peskind E, et al. Detection of biomarkers with a multiplex quantitative proteomic platform in cerebrospinal fluid of patients with neurodegenerative disorders. J Alzheimers Dis. 2006;9:293–348. doi: 10.3233/jad-2006-9309. [DOI] [PubMed] [Google Scholar]
- Zhang J, Sokal I, Peskind ER, Quinn JF, Jankovic J, Kenney C, et al. CSF multianalyte profile distinguishes Alzheimer and Parkinson diseases. Am J Clin Pathol. 2008;129:526–529. doi: 10.1309/W01Y0B808EMEH12L. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chauhan A, Pirttila T, Mehta P, Chauhan VP, Wisniewski HM. Effect of cerebrospinal fluid from normal and Alzheimer's patients with different apolipoprotein E phenotypes on in vitro aggregation of amyloid beta-protein. J Neurol Sci. 1996;141:54–58. doi: 10.1016/0022-510x(96)00123-2. [DOI] [PubMed] [Google Scholar]
- Jirikowski GF, Kaunzner UW, Dief Ael E, Caldwell JD. Distribution of vitamin D binding protein expressing neurons in the rat hypothalamus. Histochem Cell Biol. 2009;131:365–370. doi: 10.1007/s00418-008-0540-6. [DOI] [PubMed] [Google Scholar]
- Wilhelmus MM, Boelens WC, Otte-Holler I, Kamps B, de Waal RM, Verbeek MM. Small heat shock proteins inhibit amyloid-beta protein aggregation and cerebrovascular amyloid-beta protein toxicity. Brain Res. 2006;1089:67–78. doi: 10.1016/j.brainres.2006.03.058. [DOI] [PubMed] [Google Scholar]
- Ray I, Chauhan A, Wegiel J, Chauhan VP. Gelsolin inhibits the fibrillization of amyloid beta-protein, and also defibrillizes its preformed fibrils. Brain Res. 2000;853:344–351. doi: 10.1016/s0006-8993(99)02315-x. [DOI] [PubMed] [Google Scholar]
- Selenica ML, Wang X, Ostergaard-Pedersen L, Westlind-Danielsson A, Grubb A. Cystatin C reduces the in vitro formation of soluble Abeta1-42 oligomers and protofibrils. Scand J Clin Lab Invest. 2007;67:179–190. doi: 10.1080/00365510601009738. [DOI] [PubMed] [Google Scholar]
- Moon M, Choi JG, Nam DW, Hong HS, Choi YJ, Oh MS, et al. Ghrelin ameliorates cognitive dysfunction and neurodegeneration in intrahippocampal amyloid-beta1-42 oligomer-injected mice. J Alzheimers Dis. 2011;23:147–159. doi: 10.3233/JAD-2010-101263. [DOI] [PubMed] [Google Scholar]
- West MJ, Coleman PD, Flood DG, Troncoso JC. Differences in the pattern of hippocampal neuronal loss in normal ageing and Alzheimer's disease. Lancet. 1994;344:769–772. doi: 10.1016/s0140-6736(94)92338-8. [DOI] [PubMed] [Google Scholar]
- Tomiyama T, Matsuyama S, Iso H, Umeda T, Takuma H, Ohnishi K, et al. A mouse model of amyloid beta oligomers: their contribution to synaptic alteration, abnormal tau phosphorylation, glial activation, and neuronal loss in vivo. J Neurosci. 2010;30:4845–4856. doi: 10.1523/JNEUROSCI.5825-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilhelmus MM, de Waal RM, Verbeek MM. Heat shock proteins and amateur chaperones in amyloid-beta accumulation and clearance in Alzheimer's disease. Mol Neurobiol. 2007;35:203–216. doi: 10.1007/s12035-007-0029-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira ST, Klein WL. The Abeta oligomer hypothesis for synapse failure and memory loss in Alzheimer's disease. Neurobiol Learn Mem. 2011;96:529–543. doi: 10.1016/j.nlm.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Townsend M, Cleary JP, Mehta T, Hofmeister J, Lesne S, O'Hare E, et al. Orally available compound prevents deficits in memory caused by the Alzheimer amyloid-beta oligomers. Ann Neurol. 2006;60:668–676. doi: 10.1002/ana.21051. [DOI] [PubMed] [Google Scholar]
- Scheff SW, DeKosky ST, Price DA. Quantitative assessment of cortical synaptic density in Alzheimer's disease. Neurobiol Aging. 1990;11:29–37. doi: 10.1016/0197-4580(90)90059-9. [DOI] [PubMed] [Google Scholar]
- Masliah E, Mallory M, Alford M, DeTeresa R, Hansen LA, McKeel DW, et al. Altered expression of synaptic proteins occurs early during progression of Alzheimer's disease. Neurology. 2001;56:127–129. doi: 10.1212/wnl.56.1.127. [DOI] [PubMed] [Google Scholar]
- Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, et al. Physical basis of cognitive alterations in Alzheimer's disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol. 1991;30:572–580. doi: 10.1002/ana.410300410. [DOI] [PubMed] [Google Scholar]
- Lacor PN, Buniel MC, Furlow PW, Clemente AS, Velasco PT, Wood M, et al. Abeta oligomer-induced aberrations in synapse composition, shape, and density provide a molecular basis for loss of connectivity in Alzheimer's disease. J Neurosci. 2007;27:796–807. doi: 10.1523/JNEUROSCI.3501-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finehout EJ, Franck Z, Choe LH, Relkin N, Lee KH. Cerebrospinal fluid proteomic biomarkers for Alzheimer's disease. Ann Neurol. 2007;61:120–129. doi: 10.1002/ana.21038. [DOI] [PubMed] [Google Scholar]
- Seubert P, Vigo-Pelfrey C, Esch F, Lee M, Dovey H, Davis D, et al. Isolation and quantification of soluble Alzheimer's beta-peptide from biological fluids. Nature. 1992;359:325–327. doi: 10.1038/359325a0. [DOI] [PubMed] [Google Scholar]
- Qin Z, Qin Y, Liu S. Alteration of DBP levels in CSF of patients with MS by proteomics analysis. Cell Mol Neurobiol. 2009;29:203–210. doi: 10.1007/s10571-008-9312-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petkova AT, Ishii Y, Balbach JJ, Antzutkin ON, Leapman RD, Delaglio F, et al. A structural model for Alzheimer's beta-amyloid fibrils based on experimental constraints from solid state NMR. Proc Natl Acad Sci USA. 2002;99:16742–16747. doi: 10.1073/pnas.262663499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinke AA, Gestwicki JE. Insight into amyloid structure using chemical probes. Chem Biol Drug Des. 2011;77:399–411. doi: 10.1111/j.1747-0285.2011.01110.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chambon C, Wegener N, Gravius A, Danysz W. Behavioural and cellular effects of exogenous amyloid-beta peptides in rodents. Behav Brain Res. 2011;225:623–641. doi: 10.1016/j.bbr.2011.08.024. [DOI] [PubMed] [Google Scholar]
- Han SH, Jung ES, Sohn JH, Hong HJ, Hong HS, Kim JW, et al. Human serum transthyretin levels correlate inversely with Alzheimer's disease. J Alzheimers Dis. 2011;25:77–84. doi: 10.3233/JAD-2011-102145. [DOI] [PubMed] [Google Scholar]
- Hansson SF, Andreasson U, Wall M, Skoog I, Andreasen N, Wallin A, et al. Reduced levels of amyloid-beta-binding proteins in cerebrospinal fluid from Alzheimer's disease patients. J Alzheimers Dis. 2009;16:389–397. doi: 10.3233/JAD-2009-0966. [DOI] [PubMed] [Google Scholar]
- Fernandez-Montesinos R, Torres M, Baglietto-Vargas D, Gutierrez A, Gozes I, Vitorica J, et al. Activity-dependent neuroprotective protein (ADNP) expression in the amyloid precursor protein/presenilin 1 mouse model of Alzheimer's disease. J Mol Neurosci. 41:114–120. doi: 10.1007/s12031-009-9300-x. [DOI] [PubMed] [Google Scholar]
- Tizon B, Ribe EM, Mi W, Troy CM, Levy E. Cystatin C protects neuronal cells from amyloid-beta-induced toxicity. J Alzheimers Dis. 19:885–894. doi: 10.3233/JAD-2010-1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merched A, Serot JM, Visvikis S, Aguillon D, Faure G, Siest G. Apolipoprotein E, transthyretin and actin in the CSF of Alzheimer's patients: relation with the senile plaques and cytoskeleton biochemistry. FEBS Lett. 1998;425:225–228. doi: 10.1016/s0014-5793(98)00234-8. [DOI] [PubMed] [Google Scholar]
- Pogge E. Vitamin D and Alzheimer's disease: is there a link. Consult Pharm. 2010;25:440–450. doi: 10.4140/TCP.n.2010.440. [DOI] [PubMed] [Google Scholar]
- Paris D, Patel N, DelleDonne A, Quadros A, Smeed R, Mullan M. Impaired angiogenesis in a transgenic mouse model of cerebral amyloidosis. Neurosci Lett. 2004;366:80–85. doi: 10.1016/j.neulet.2004.05.017. [DOI] [PubMed] [Google Scholar]
- Mattson MP. Oxidative stress, perturbed calcium homeostasis, and immune dysfunction in Alzheimer's disease. J Neurovirol. 2002;8:539–550. doi: 10.1080/13550280290100978. [DOI] [PubMed] [Google Scholar]
- Raymond MA, Desormeaux A, Labelle A, Soulez M, Soulez G, Langelier Y, et al. Endothelial stress induces the release of vitamin D-binding protein, a novel growth factor. Biochem Biophys Res Commun. 2005;338:1374–1382. doi: 10.1016/j.bbrc.2005.10.105. [DOI] [PubMed] [Google Scholar]
- Maezawa I, Hong HS, Liu R, Wu CY, Cheng RH, Kung MP, et al. Congo red and thioflavin-T analogs detect Abeta oligomers. J Neurochem. 2008;104:457–468. doi: 10.1111/j.1471-4159.2007.04972.x. [DOI] [PubMed] [Google Scholar]
- Maezawa I, Hong HS, Wu HC, Battina SK, Rana S, Iwamoto T, et al. A novel tricyclic pyrone compound ameliorates cell death associated with intracellular amyloid-beta oligomeric complexes. J Neurochem. 2006;98:57–67. doi: 10.1111/j.1471-4159.2006.03862.x. [DOI] [PubMed] [Google Scholar]
- Hong HS, Rana S, Barrigan L, Shi A, Zhang Y, Zhou F, et al. Inhibition of Alzheimer's amyloid toxicity with a tricyclic pyrone molecule in vitro and in vivo. J Neurochem. 2009;108:1097–1108. doi: 10.1111/j.1471-4159.2008.05866.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee KH, Shin BH, Shin KJ, Kim DJ, Yu J. A hybrid molecule that prohibits amyloid fibrils and alleviates neuronal toxicity induced by beta-amyloid (1–42) Biochem Biophys Res Commun. 2005;328:816–823. doi: 10.1016/j.bbrc.2005.01.030. [DOI] [PubMed] [Google Scholar]
- Bang OY, Hong HS, Kim DH, Kim H, Boo JH, Huh K, et al. Neuroprotective effect of genistein against beta amyloid-induced neurotoxicity. Neurobiol Dis. 2004;16:21–28. doi: 10.1016/j.nbd.2003.12.017. [DOI] [PubMed] [Google Scholar]
- Grohm J, Kim SW, Mamrak U, Tobaben S, Cassidy-Stone A, Nunnari J, et al. Inhibition of Drp1 provides neuroprotection in vitro and in vivo. Cell Death Differ. 2012;19:1446–1458. doi: 10.1038/cdd.2012.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang W, Dunlap JR, Andrews RB, Wetzel R. Aggregated polyglutamine peptides delivered to nuclei are toxic to mammalian cells. Hum Mol Genet. 2002;11:2905–2917. doi: 10.1093/hmg/11.23.2905. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Franklin KBJ.The Mouse Brain in Stereotaxic Coordinates2nd ednAcademic Press: San Diego, CA; 2001 [Google Scholar]
- Kim DH, Jeon SJ, Son KH, Jung JW, Lee S, Yoon BH, et al. The ameliorating effect of oroxylin A on scopolamine-induced memory impairment in mice. Neurobiol Learn Mem. 2007;87:536–546. doi: 10.1016/j.nlm.2006.11.005. [DOI] [PubMed] [Google Scholar]
- Moon M, Hong HS, Nam DW, Baik SH, Song H, Kook SY, et al. Intracellular amyloid-beta accumulation in calcium-binding protein-deficient neurons leads to amyloid-beta plaque formation in animal models of Alzheimer's disease. J Alzheimers Dis. 2012;29:615–628. doi: 10.3233/JAD-2011-111778. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.