Abstract
Dilated cardiomyopathies (DCM) show remarkable variability in their age of onset, phenotypic presentation, and clinical course. Hence, disease mechanisms must exist that modify the occurrence and progression of DCM, either by genetic or epigenetic factors that may interact with environmental stimuli. In the present study, we examined genome-wide cardiac DNA methylation in patients with idiopathic DCM and controls. We detected methylation differences in pathways related to heart disease, but also in genes with yet unknown function in DCM or heart failure, namely Lymphocyte antigen 75 (LY75), Tyrosine kinase-type cell surface receptor HER3 (ERBB3), Homeobox B13 (HOXB13) and Adenosine receptor A2A (ADORA2A). Mass-spectrometric analysis and bisulphite-sequencing enabled confirmation of the observed DNA methylation changes in independent cohorts. Aberrant DNA methylation in DCM patients was associated with significant changes in LY75 and ADORA2A mRNA expression, but not in ERBB3 and HOXB13. In vivo studies of orthologous ly75 and adora2a in zebrafish demonstrate a functional role of these genes in adaptive or maladaptive pathways in heart failure.
Keywords: biomarker, dilated cardiomyopathy, DNA methylation, epigenetics, heart failure
INTRODUCTION
Idiopathic dilated cardiomyopathy (DCM) is a frequent heart muscle disease with an estimated prevalence of 1:2500 (Karkkainen & Peuhkurinen, 2007). The progressive nature of the disorder is responsible for nearly 50,000 hospitalizations and 10,000 deaths per year in the US alone and is the main cause for heart transplantation in young adults (Dec & Fuster, 1994). Overall, the incidence of the disease has continually increased over the past years and it was recognized that DCM has a substantial genetic contribution (Grunig et al, 1998). It is estimated that about 30–40% of all DCM cases show familial aggregation and until now more than 40 different genes were found to cause genetic DCM (Meder & Katus, 2012). However, since the course of even monogenetic DCM is highly variable, genetic modifiers are thought to have an important influence on phenotypic characteristics and outcome (Friedrichs et al, 2009; Villard et al, 2011). Accordingly, several studies have now identified common genetic polymorphisms that are associated with DCM or heart failure (Friedrichs et al, 2009; Villard et al, 2011).
Disease modification through epigenetic alterations has been convincingly demonstrated for a number of diseases (Feinberg & Tycko, 2004; Jones & Baylin, 2002). In the cardiovascular system, histone modifications and chromatin remodelling are thought to direct adaptive as well as maladaptive molecular pathways in cardiac hypertrophy and failure (Montgomery et al, 2007), and DNA methylation was found to be responsible for the hypermutability of distinct cardiac genes (Meurs & Kuan, 2011). Furthermore, recent studies have highlighted potential interplay between environmental factors and the disease phenotype by epigenetic mechanisms (Herceg & Vaissiere, 2011; Jirtle & Skinner, 2007). However, the knowledge about the impact of epigenetic alterations on the disease phenotype in human patients is still very limited.
The present study investigated for the first time the impact of genome-wide cardiac DNA methylation on human DCM in patients. We identified several candidate genes with altered methylation status and replicated these findings in an independent cohort of DCM patients and controls. Using gene expression analysis and zebrafish as an in vivo model, we could furthermore show that appropriate mRNA levels of LY75 and ADORA2A are important for unconstrained cardiac function.
RESULTS
DNA methylation is altered in patients with DCM
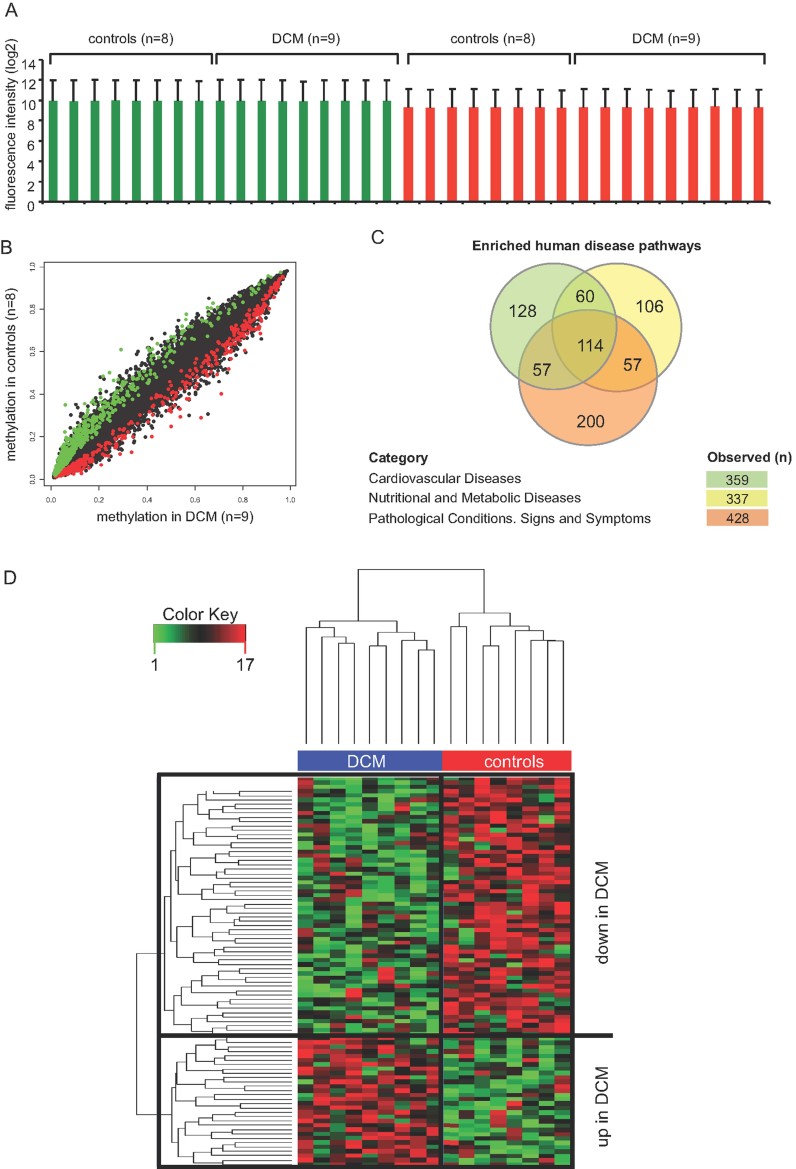
We performed two-staged, funnel-like DNA methylation mapping in non-ischaemic, idiopathic DCM patients and controls (Table 1). In the screening stage, we assessed genome-wide DNA methylation levels of CpG islands (CGIs) using the Infinium HumanMethylation 27 platform. We first extracted 1000 ng of genomic DNA from LV biopsies from 10 DCM patients and 10 controls. After methylation profiling, 17 datasets passed the stringent quality filter criteria, exemplarily shown by reaching highly similar bead color signal intensities (Fig 1A). Fig 1B shows a correlation plot of the 27,578 individual methylation sites for all further analysed patients and controls. While the degree of methylation for most CpG sites is highly correlated between the two groups, we detected several CGIs that are hypo- (green dots) or hyper-methylated (red dots) in DCM compared to the controls (unadjusted p-value <0.05).
Table 1.
Study sample characteristics of the screening and replication cohorts
| Cohort | N | Female (%) | Age (years) | LV-EF (%) |
|---|---|---|---|---|
| Screening | ||||
| DCM cases | 9 | 33 | 57 ± 5.4 | 27 ± 7.5 |
| Controls | 8 | 38 | 42 ± 14 | 60 ± 4.0 |
| Replication | ||||
| DCM cases | 30 | 37 | 58 ± 14 | 25.6 ± 8.4 |
| Controls | 28 | 46 | 57 ± 12 | 61.5 ± 5.3 |
For the control patients: Female, female donor status of transplanted heart; LV-EF, left ventricular ejection fraction.
Figure 1. Detection of DNA methylation patterns.
- Bar graphs showing quantile normalized bead colour signals (green and red) from methylation measurements from all patients and controls of the screening stage included in the further analysis. Signal quality was highly similar over all DCM and control samples. Error bars indicate standard deviation.
- Correlation plot showing the percentage of CpG methylation in controls versus DCM patients, resulting in an overall very high correlation. The coloured signals that are furthest away from the bisecting line show significantly hyper- (red) and hypo-methylated (green) CpGs in DCM patients.
- Gene-set enrichment analysis for NIA human disease pathways. The area-proportional Venn diagram shows that methylation changes in cardiovascular disease genes are significantly enriched together with the overlap of the other indicated gene sets.
- Cluster analysis for genes with known expression in the human heart and significantly altered methylation. The colour code used for the heatmap is shown in the upper left corner, values range from 1 (sample with the lowest methylation for the considered genes) to 17 (sample with the highest methylation for the considered genes).
We used the comprehensive screening datasets in a gene set enrichment analysis (GSEA) and identified within the top three (ranked by p values) enriched disease categories provided by the National Institute of Aging (NIA) the disease pathways ‘cardiovascular disease’, ‘metabolic disease’ and ‘pathological conditions’, all associated with DCM and with considerable overlaps between each other (Fig 1C). To detect patterns of genes with differential methylation in the screening cohorts, we carried out a clustering approach on genes with known abundant expression in the human heart (http://c-it.mpi-bn.mpg.de). From the annotated 2018 individual genes, 1858 (92.1%) were covered by the applied Infinium assay. Since the degrees of methylation for the genes were not normally distributed, we used two-tailed Wilcoxon rank-sum test to compute a significance value for each gene, which resulted in a total of 90 genes surpassing statistical significance (p-value <0.05). While about one-third showed increased methylation in DCM patients, approximately two-thirds were significantly less methylated. Fig 1D gives a graphical representation of differential methylation patterns by using hierarchical clustering on the Euclidian distance. The heat map shows the patterns of higher- and lower methylated genes in DCM patients and controls.
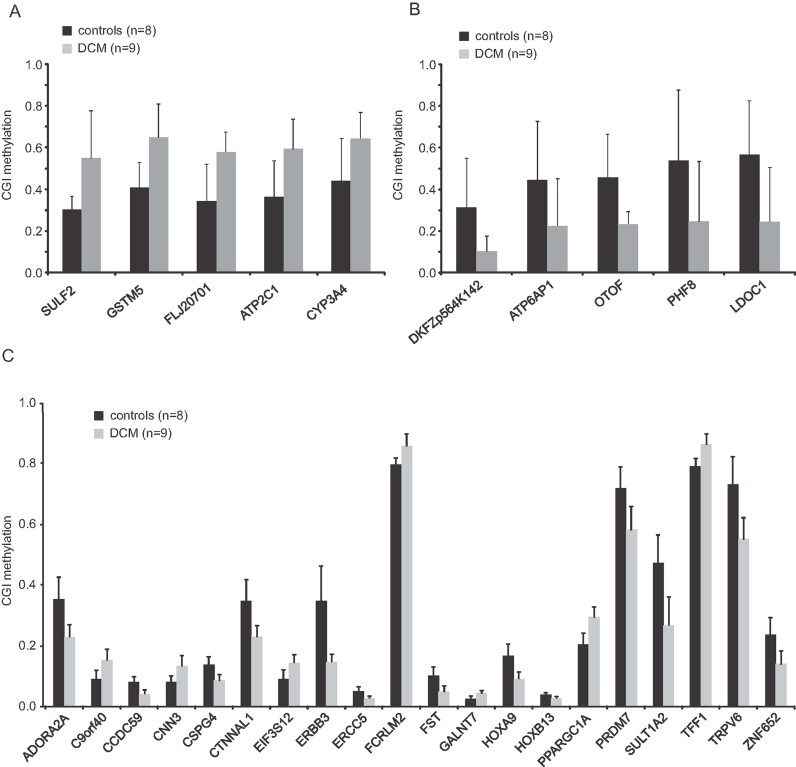
For the replication stage in an independent cohort, methylation patterns of single genes were used to define candidates for a mass spectrometry-based fine-mapping. Fig 2A and B shows the five most hyper- and hypomethylated genes, while Fig 2C details the 20 most significantly dys-methylated genes of the screening stage. The final selection was based on unadjusted p values or absolute methylation difference, CGI localization, capability to design specific assay probes, and known expression in the heart.
Figure 2. Differentially methylated genes in DCM patients.
- A, B. Bar graphs showing the degree of methylation of CGIs of the screening cohort (n = 9 DCM patients; n = 8 controls). The five genes with the largest increase in methylation in DCM patients are shown in (A) and genes with the largest decrease in methylation are displayed in (B).
- C. The 20 genes with the most significant methylation changes in the screening phase. Error bars indicate standard deviation.
Validation of aberrant DNA methylation in DCM
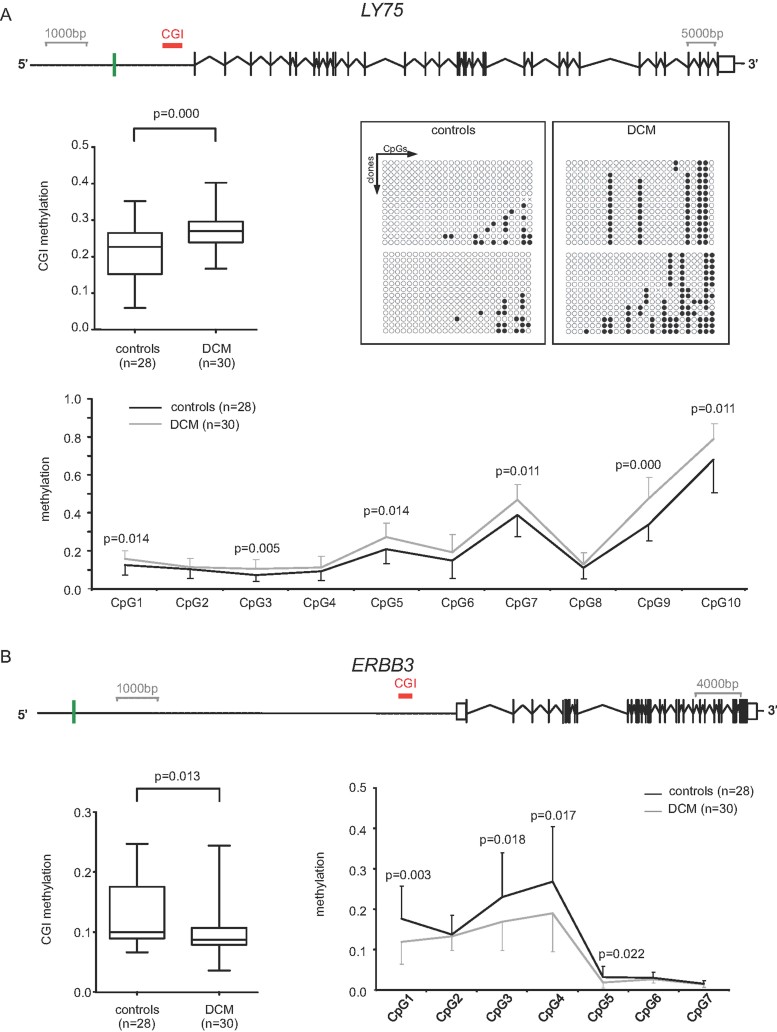
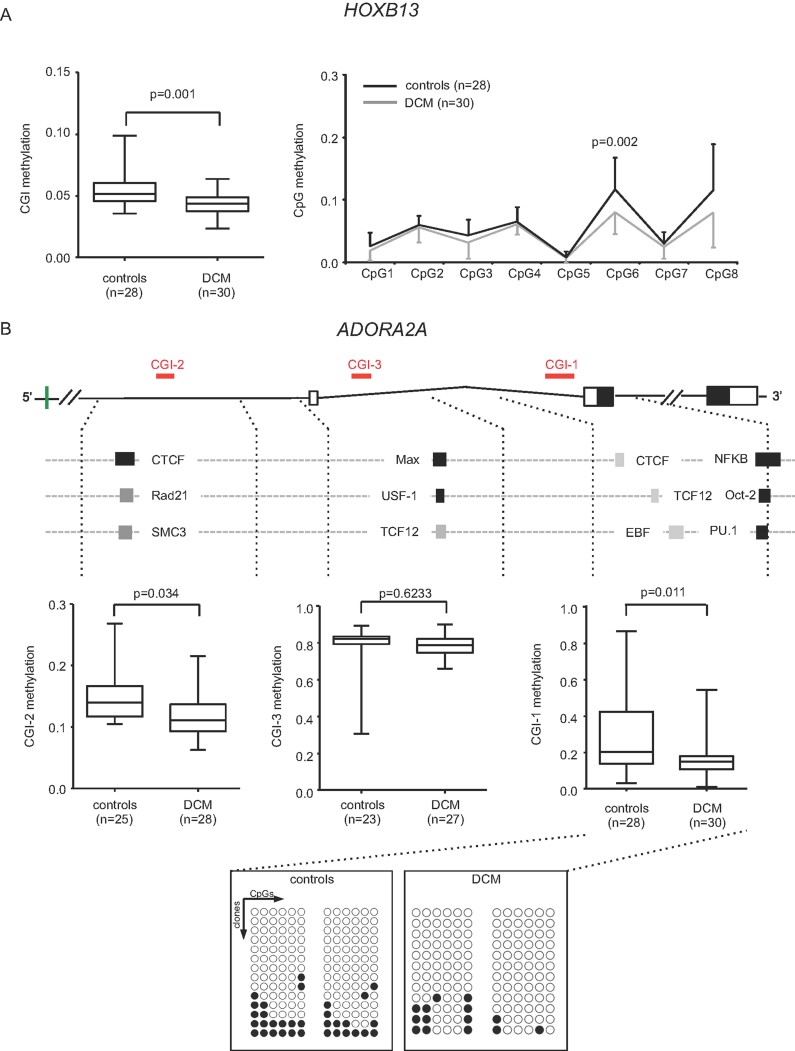
As denoted above, we carried out an independent replication and fine mapping of the selected genes in a larger cohort of 30 idiopathic DCM patients and 28 controls. All selected candidates were fine-mapped by using MassARRAY (Ehrich et al, 2005). For each gene, several CpGs were retrieved and their methylation status quantified. From 20 candidate genes, 12 showed the same direction of altered methylation between the screening and the replication stage and four of them reached statistical significance, namely LY75 (p = 0.000), ERBB3 (p = 0.013), HOXB13 (p = 0.001), and ADORA2A (p = 0.011). Figs 3 and 4 present the mean methylation changes of the replicated genes. Additionally, methylation of individual CpGs is displayed for LY75 and ERBB3 (Fig. 3), HOXB13 (Fig. 4) and ADORA2A (Supporting Information Fig 1). Interestingly, ADORA2A showed significantly altered methylation throughout all tested CpGs, while ERBB3 or LY75 showed methylation alterations in a subset of the CpG nucleotides, possibly resulting in different functional consequences. Supporting Information Fig 2 gives the mean methylation of the remaining investigated CGIs. Since epigenetic marks may be also dependent on the gender, we additionally matched the gender ratio of cases and controls of the replication cohort to the ratio of the screening cohort, leading to significance of the same CGIs shown above. This is also true when matching females and males 1:1.
Figure 3. MassARRAY-based validation of differential methylation in LY75 and ERBB3.
DCM patients show significantly increased DNA methylation in LY75 (A), while ERBB3 is significantly lower methylated (B). The schemas above the methylation graphs represent the tested CGI (red lines), in relation to the predicted transcription start-site (green lines), the exons (black bars) and alternatively spliced exons (white boxes) of the genes.
- The box and whiskers plot (min to max) on the upper left side represents the mean CpG methylation measured by MassARRAY, the individual CpG methylation is shown at the bottom. The pattern of CpG methylation in multiple clones is shown on the upper right insert.
- The box and whiskers plot (min to max) on the left side represents the mean CpG methylation measured by MassARRAY, the individual CpG methylation is shown on the right.
Figure 4. MassARRAY-based validation of differential methylation in HOXB13 and ADORA2A.
DCM patients show significantly decreased DNA methylation in HOXB13 (A) and ADORA2A (B).
- The box and whiskers plot (min to max) on the left side represents the mean CpG methylation measured by MassARRAY, the individual CpG methylation is shown on the right.
- The scheme above the graph represents the tested CGI (red line), in relation to the predicted transcription start site (green line), the exons (black boxes) and alternatively spliced exons (white boxes) of adora2a. Dashed lines indicate the 1500 bp up- and downstream region of the CGIs wherein the listed transcription factor binding sites are found. Boxplots below show the mean methylation in DCM and controls at the corresponding CGI. The pattern of CpG methylation as assessed by bisulphite-sequencing in multiple clones is shown for CGI-1 at the bottom of the figure.
In addition to MassARRAY, we applied bisulphite sequencing for LY75 and ADORA2A to fine-map and technically replicate our results. To do so, we generated 14 LY75 and 12 ADORA2A clones from two DCM patients and two controls each, and sequenced them before and after treatment with bisulphite (Fig 3A and B; black circles = methylated CpG, white circles = unmethylated CpG). Bisulphite sequencing confirmed the corresponding MassARRAY data and, hence, demonstrated reliability of the latter technique.
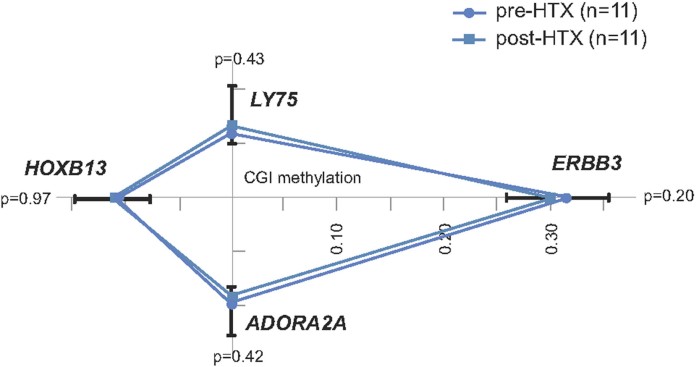
Next, to rule out a potential effect of the immunosuppressive medication received by the control subjects (Table 2) on the methylation of the investigated genes, we analysed the methylation patterns of genomic DNA from peripheral blood of 11 subjects drawn pre- and post-heart transplantation (HTX; mean timespan after HTX = 37 months under medication). As shown in Fig 5 we found highly comparable methylation levels (p = n.s.) of LY75, ADORA2A, ERBB3 and HOXB13 CGIs in pre- and post-HTX, indicating that the observed methylation differences in DCM are not due to methylation changes in the controls receiving immunosuppressive medication.
Table 2.
Immunosuppressive medication of control individuals
| Medication | N | % |
|---|---|---|
| Tacrolimus | 19 | 54 |
| Mycophenolat-mofetil | 23 | 66 |
| Ciclosporin | 8 | 23 |
| Prednisolon | 11 | 31 |
| Everolimus | 14 | 40 |
All control individuals received one or more immunosuppressives.
Figure 5. CpG island methylation of control subjects before and after heart transplantation.
The line diagram shows the highly similar degree of methylation of DNA derived from peripheral blood of control subjects pre- (light blue) and post- (dark blue) heart transplantation (n = 11) for LY75, ADORA2A, ERBB3 and HOXB13. Error bars indicate standard deviation.
Differential gene expression and functional evaluation in vivo suggest contribution of LY75 and ADORA2A to DCM
DNA methylation is often correlated with changes in the accessibility of DNA to transcriptional activators, enhancers, or repressors. Hence, we first studied the impact of DNA methylation at the LY75, ERBB3, HOXB13, and ADORA2A loci on their gene expression by quantitative PCR (q-PCR) in controls, mild DCM (NYHA class II) and moderate to severe DCM (NYHA class III–IV).
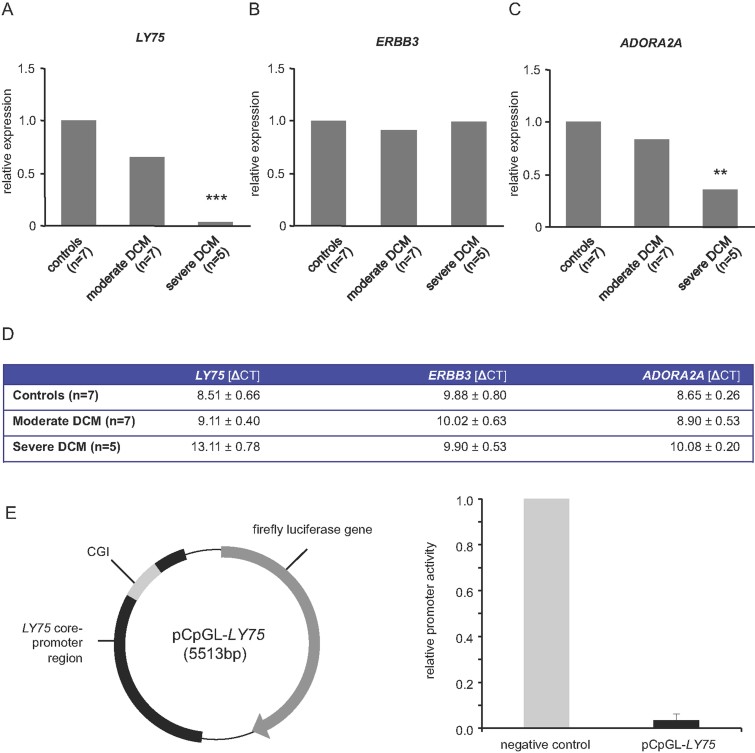
For ADORA2A, we found a positive relationship of gene expression with methylation (relative expression 0.33; p = 0.002), while ERBB3 did not show significant alterations in cardiac expression levels (Fig 6B–D). HOXB13 transcripts could not be PCR amplified in LV biopsies from patients and controls. In case of LY75, the hypermethylated CpG island is relatively close to the transcriptional start site (distance 1395 bp), whereas the distance between the CGIs in ERBB3 (7518 bp) and ADORA2A (14,329 bp) is markedly larger (Figs 3 and 4). Since we also observed a strong reduction in LY75 expression in myocardial tissue (relative expression 0.04; p = 0.001) in patients with DCM (Fig 6A and D), we asked if increased promoter methylation is directly responsible for decreased transcriptional activity. Hence, we performed a luciferase promoter assay with methylated and unmethylated LY75 promoters. The CpG-free control reporter vector pCpGL-CMV/EF1 served as a negative control. As shown in Fig 6E, we found a strong reduction in promoter activity after in vitro methylation, supporting the functional relationship between LY75 hypermethylation and reduced expression in human DCM.
Figure 6. mRNA expression of genes with altered methylation status and gene promoter analysis.
- A–C. Bar graphs showing relative mRNA expression levels of LY75, ERBB3 and ADORA2A in mild and moderate-severe DCM in comparison to controls. (A) LY75 expression is strongly reduced (relative expression = 0.04), while (B) ERBB3 mRNA levels are not differentially regulated. (C) ADORA2A is also significantly down-regulated in DCM (relative expression = 0.37).
- D. Given are the mean delta-CT values (±SEM) of LY75, ERBB3, and ADORA2A in the different groups (controls, moderate DCM, severe DCM). The reference is based on the mean of the three housekeeper genes: GAPDH, RPL13, β-actin.
- E. Left: Schema of the vector construct to measure LY75 promoter activity without and after treatment with the methylase SssI. Right: Relative promoter activity (mean of three technical replicates ± SD) showing the strongly reduced activity in LY75 after methylation, whereas the negative control does not show significant differences.
Since we found a rather unusual positive relationship of ADORA2A gene expression and methylation, we first sought to identify the predominant isoform of ADORA2A in the human myocardium. By PCR, we found that the longer ADORA2A transcripts ENST00000337539 and ENST00000541988 are highly expressed in the heart. However isoform ENST00000417596, which is highly expressed in peripheral blood, is not significantly expressed in the heart. We then investigated potential repressor regions in the close vicinity of the tested CGIs. For the detection of transcription factor binding sites, the ENCODE transcription factor ChIP-seq data was used. We found for the aberrantly methylated CGI-1 two potential repressor sites (CTCF and NFKB), for aberrantly methylated CGI-2 one repressor site (CTCF) and for the unaltered CGI-3 one repressor site (Max). Interestingly, both hypomethylated CGIs (1 and 2) carry CTCF binding sites, which were recently identified as epigenetic key regulators in various other diseases.
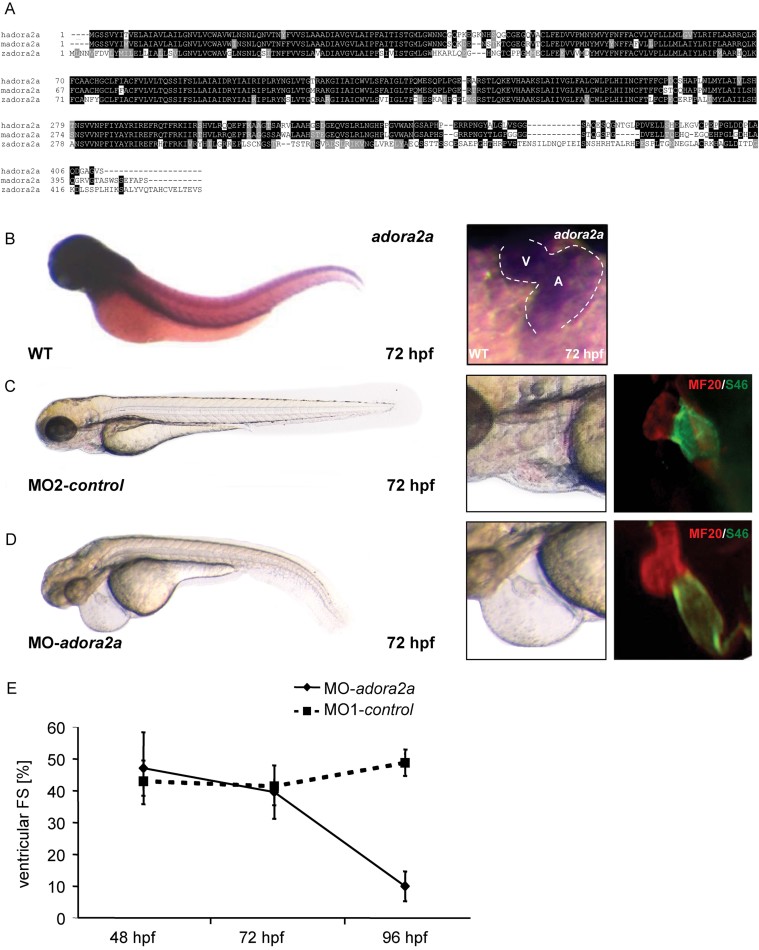
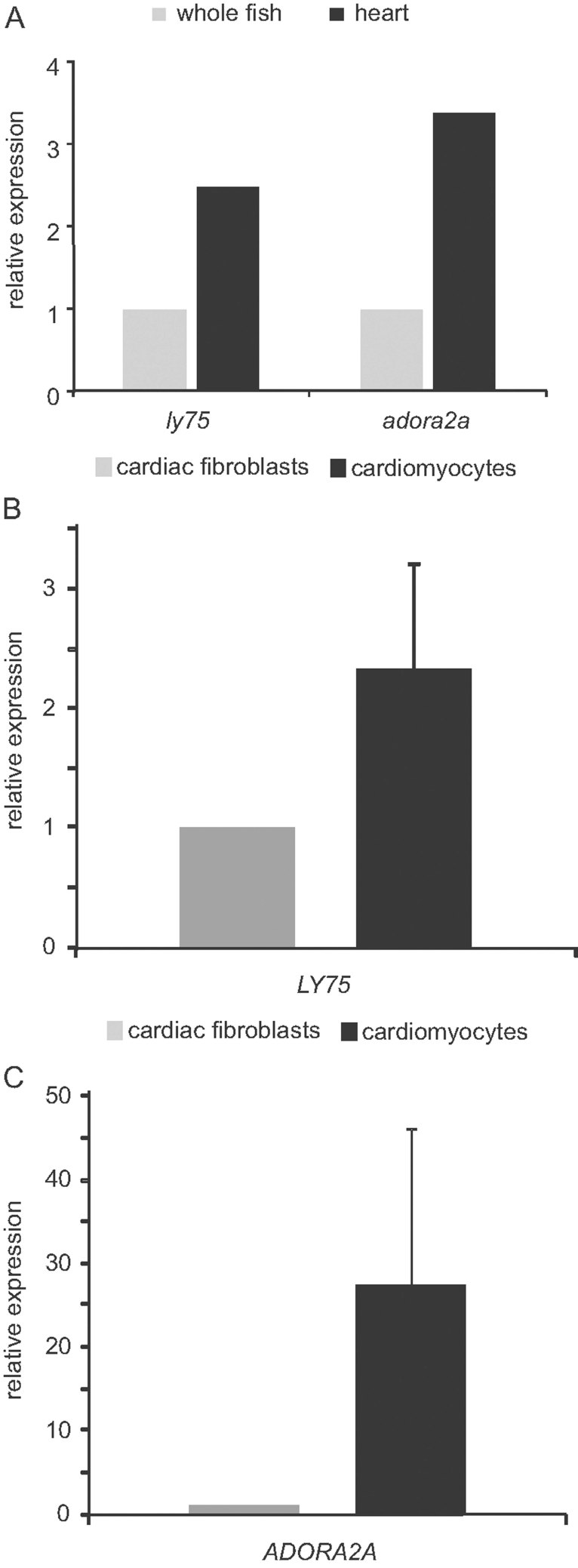
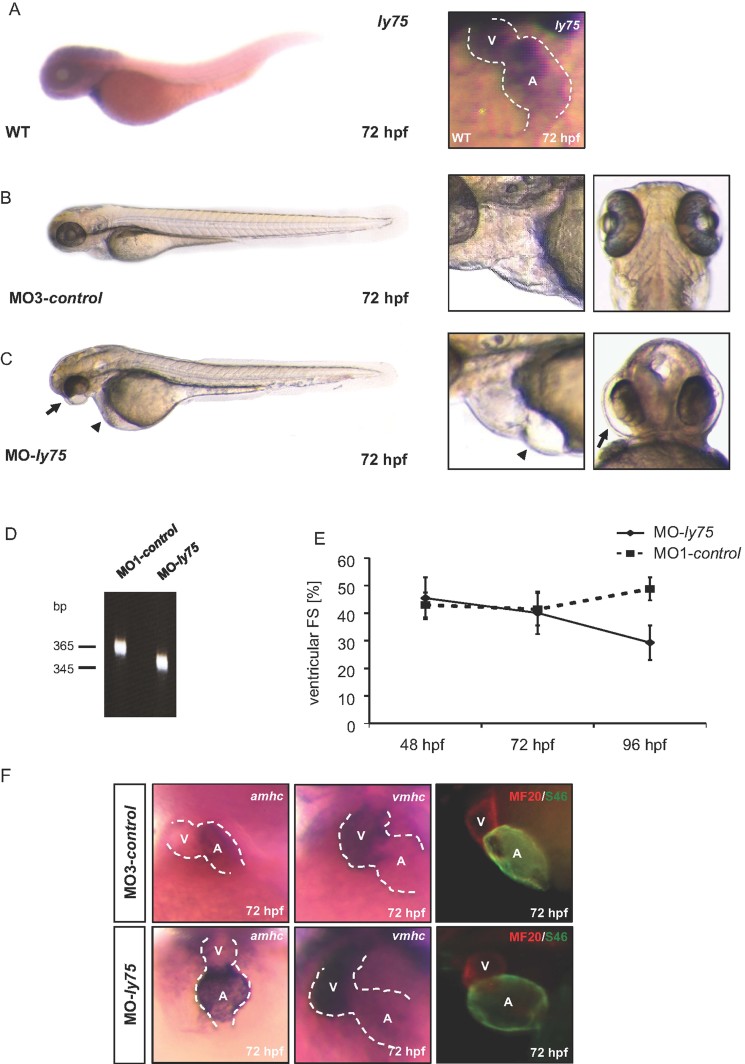
Since LY75 and ADORA2A were not previously known to be involved in DCM or heart failure pathogenesis and both showed significant downregulation in the myocardium of DCM patients, we investigated their functional roles by gene knockdown in zebrafish embryos (Dahme et al, 2009). We identified orthologous sequences for human LY75 and ADORA2A by BLAST searches against the zebrafish Genbank database. Protein sequence identity between the zebrafish and human version was 61% for adora2a (Fig 9A) and 37% for ly75 (Supporting Information Fig 3). Similar to the human situation, we found adora2a and ly75 to be highly expressed in the zebrafish heart using q-PCR and RNA antisense in situ hybridization (Figs 7–9). Additionally, as shown in cultured neonatal rat cells, both genes are higher expressed in cardiomyocytes than in cardiofibroblasts (Fig 7B and C). Hence, to recapitulate the downregulation of both genes as observed in the human heart, we inactivated them in zebrafish embryos by injection of Morpholino-modified antisense oligonucleotides directed against the splice donor site of ly75 or the translational start-site of adora2a. While control-injected zebrafish (standard control Morpholino as well as scrambled control Morpholinos) embryos did not show any obvious phenotype, splice site Morpholino-mediated knockdown of ly75 resulted in partial skipping of exon 8 (Fig 8D) and a consecutive frameshift that predictably leads to a premature stop of protein translation. As a result, ly75-morphants developed late-onset heart failure with dilation of the atrium (Fig 8C) and reduced ventricular contractility beginning at the 96 h developmental stage (FS = 46 ± 5% at 48 hpf and 29 ± 4% at 96 hpf; Fig 8E, Supporting Information Movie 1). Additionally, ly75-morphants showed a pronounced detachment and oedema of the skin especially noticeable in the eye and head region (Fig 8C). MO-adora2a-injected embryos also developed severe heart failure with progressively decreasing ventricular contractility as measured by fractional shortening (Fig 9E, Supporting Information Movie 2). In detail, the ventricular contractility of adora2a-morphants decreased from 47 ± 11% at 48 hpf to 39 ± 8% at 72 hpf. By 96 hpf, both heart chambers became almost silent. Occasionally, we also observed atrial fibrillation in adora2a-morphants and embryos developed excessive pericardial effusion and precardial blood congestion as a consequence of the reduced cardiac function. For both, ly75- and adora2a-morphants, we saw no alterations in molecular chamber specification and expression of atrial and ventricular myosin heavy chain genes (Figs 8 and 9 and Supporting Information Fig 4).
Figure 9. Knockdown of adora2a leads to severe heart failure in zebrafish.
- A. Amino acid sequence alignments of human (h), mouse (m) and zebrafish (z) ADORA2A demonstrating its high cross-species homology. Black boxes indicate amino acid identity, grey boxes indicate amino acids with similar chemical properties.
- B. Adora2A is significantly expressed in the zebrafish heart.
- C, D. Lateral view of MO2-control (C), and MO-adora2a (D) injected embryos at 72 hpf.
- E. After injection of MO-adora2a, 66% of morphant embryos develop progressive heart failure and deterioration in ventricular contractility measured by fractional shortening at the indicated developmental stages. Error bars indicate standard deviation.
Figure 7. ly75 and adora2a tissue and cell-type expression.
- A. Relative expression levels of ly75 and adora2a quantified by q-PCR in 40 whole zebrafish embryos (grey bars) and 200 isolated zebrafish embryonic hearts (black bars).
- B, C. Expression of ly75 (B) and adora2a (C) in neonatal rat cardiomyocytes and cardiofibroblasts (n = 3 biological replicates). Error bars indicate standard deviation.
Figure 8. ly75 is essential to maintain cardiac contractility. hpf, hours post fertilization; amhc, atrial myosin heavy-chain; vmhc, ventricular myosin heavy-chain; A, atrium; V, ventricle. Error bars indicate standard deviation.
- A. mRNA antisense in situ hybridization showing that ly75 is significantly expressed in the zebrafish heart.
- B, C. Lateral views of MO3-control (B) and MO-ly75 (C) injected embryos at 72 hpf.
- D, E. After depletion of ly75, which can be monitored by cDNA splice-analysis (D), zebrafish hearts show reduced contractile force (E) and precardial blood congestion (arrowhead) as sign of manifest heart failure. Ly75-morphants also show noticeable skin detachment most pronounced in the head/eye region (C).
- F. Molecular chamber definition is not impaired in ly75-morphants compared to the control-injected zebrafish as demonstrated by regular amhc, vmhc, MF20 and S46 expression.
DISCUSSION
Epigenetic mechanisms are increasingly recognized as causes and modulators of human disease. Most studies conducted so far have focused on cancer and yet only few have investigated the role of epigenetic mechanisms, such as DNA methylation, in cardiovascular disease (Movassagh et al, 2011). This is surprising since epigenetic mechanisms are thought to control key adaptive and maladaptive processes such as cardiac hypertrophy, fibrosis and failure (Backs et al, 2006, 2008). Here, we investigated DNA methylation patterns on a genome-wide level in myocardium from patients with idiopathic DCM and functionally unaffected hearts of patients who had received heart transplantation. We found and confirmed aberrant DNA methylation alterations in a number of CGIs, suggesting that DNA methylation is associated with cardiac function and may modulate phenotypic characteristics of idiopathic DCM.
There are several epigenetic mechanisms in eukaryotes and some have already been linked to DCM or heart failure. Firstly, chromatin modifications through ATP-dependent enzymes from, e.g. the SWI/SNF genes enable the cell to regulate the expression of distinct gene programs in organ development and adaptation (Ho & Crabtree, 2010). Secondly, histone modifiers such as histone acetyltransferases (HATs) or histone deacetylases (HDACs) are known to play a key role during development and in maintenance of cardiac function (Backs et al, 2011; Montgomery et al, 2008; Zhou et al, 2011). Cardiac specific loss of HDAC1 and HDAC2 for example results in cardiac arrhythmias and heart failure (Montgomery et al, 2007), while loss of HDAC3 function leads to cardiac hypertrophy (Montgomery et al, 2008). Thirdly, short non-coding microRNAs (miRNAs) broadly influence gene expression (Humphreys et al, 2005) and are increasingly recognized as valuable diagnostic (Keller et al, 2011; Meder et al, 2011a, b) and potential therapeutic targets (Gambari et al, 2011). DCM caused by deregulation of miRNAs such as miRNA-133a has been recently described in animal models (Chen et al, 2008; Meder et al, 2008; van Rooij et al, 2007). The current study now adds DNA methylation as a novel mechanism in the pathogenesis of DCM and heart failure.
DNA methylation in single genes and its contribution to disease onset or progression has been demonstrated in studies for hypertrophic cardiomyopathy (Meurs & Kuan, 2011). Furthermore, Movassagh et al (Movassagh et al, 2011) reported distinct epigenomic features in explanted human failing hearts, highlighting a potential role of DNA methylation also in end-stage heart failure. However, feasibility of the used methyl-DNA precipitation and sequencing is restricted by the relatively large amounts of required tissue, which is not easily available from living patients. Furthermore, handling of the tissue and time points of DNA isolation were quite different in controls and patients. Tissue specimens from explanted hearts of end-stage heart failure patients may be altered due to tissue handling and could have undergone secondary alterations because of degradation, missing oxygenation, innervation and arrest of pumping function (Talens et al, 2010). Also, the authors did not replicate their important findings, which were obtained in four patients, in an independent series of unrelated samples and by an alternative methodology. Therefore, we used a different approach, confirmed our findings in a large replication cohort with two alternative methodologies and applied functional studies to underscore the relevance of our results.
Since availability of appropriate myocardial tissue from living patients is limited, studies on epigenetic alterations in patients with heart disease are rare. As a primary referral centre for cardiomyopathies, we could enroll a high number of patients and controls in this study. Our controls are not completely healthy, but had previously received heart transplantation. Hence, potential confounders include patient's medication and medical history. Importantly, on the functional level all controls had an unconstrained cardiac function as a prerequisite and we could apply the exactly same standardized procedure to sample and process left ventricular biopsies from DCM patients and controls. However, all controls received immunosuppressive medications to prevent organ rejection. This medication might have an influence on at least some genes, e.g. glucocorticoids exert effects on DNA methylation of Fkbp5 (Yang et al, 2012). As shown above, we found unchanged methylation levels for the tested genes in control subjects before and after heart transplantation, showing that immunosuppressives do not, at least in whole peripheral blood, impact on CGI DNA methylation of LY75, ADORA2A, ERBB3 and HOXB13. However, it still cannot be completely ruled out that the medication exerts a different effect on cardiac tissue, which is a limitation of the current study.
Tissue samples are composed of different cell types, which might change during disease progression. In the present study, we have paid special attention to patient selection and the process of tissue sampling. Consequently, this resulted in high homogeneity of overall methylation patterns as shown above. In a study by Grzeskowiak et al it was demonstrated that differences in gene expression of LV myocardial biopsies from controls and idiopathic DCM patients were almost exclusively due to genes expressed in cardiomyocytes and that the contribution of the second most common cell type, fibroblasts, is rather low and mostly unchanged during DCM (Grzeskowiak et al, 2003). The increased fibrosis observed in human DCM, for instance, seems to be mainly due to extracellular collagen deposition rather than an increase of cardiofibroblasts. Our in vivo experiments clearly underline that reduced expression of LY75 and ADORA2A in cardiomyocytes leads to heart failure as observed in our patients. Together, this approach allowed us to study epigenetic mechanisms and their functional consequences without the confounding background of organ hypoxia and apoptosis in explanted hearts of deceased patients.
Our global methylation analyses identified cardiac disease pathways and were complemented by extensive validation studies applying the highly precise MassARRAY and bisulphite sequencing techniques (Flotho et al, 2009; Ghoshal et al, 2010). We found 12 out of 20 genes showing the same direction of dys-methylation as found in the screening stage, and four reached statistical significance. This has at least three reasons: (i) we expected a high false positive rate due to the relatively small screening cohorts in contrast to the number of measured epigenetic features, (ii) we performed the replication measurements with a completely independent methodology, and (iii) we carried out these analyses in independent patients and controls. Therefore, the genes identified here represent consistently validated targets that show an epigenetic mode of regulation.
The deciphering of the functional implications of changes in DNA methylation is difficult but of pivotal importance. Since in many cases changes in CpG methylation are correlated with the accessibility of DNA to transcription factors and polymerases, measurements of mRNA levels represent an appropriate methodology. This was shown by using different model organisms such as fish (Mirbahai et al, 2011), mice (Tryndyak et al, 2011), as well as human tissues (Movassagh et al, 2010). In the present study, we find that methylation of novel cardiac genes is altered in patients with DCM. Hence, it can be hypothesized that the dys-methylated and dys-regulated genes also exert a functional role in the heart. The receptor tyrosine-protein kinase ERBB3 belongs to the membrane-bound epidermal growth factor receptor (EGFR) family. Functionally it is implicated in SOX10-mediated neural crest and early heart development (Prasad et al, 2011). Although we could not observe changes in ERBB3 transcript levels, the significant changes in DNA methylation could be a susceptibility factor mediated through yet unknown pathways, e.g. by exerting effects on DNA stability, ERBB3 isoform expression or histone binding. LY75, a collagen-binding mannose family receptor, is transcriptionally controlled by the interleukin-6 receptor IL6Rα and mediates antigen uptake and presentation in a clathrin-dependent manner (Giridhar et al, 2011; Tel et al, 2011). For DCM, a correlation of IL-6 levels and cross-linked type I collagen was found, potentially implicating a role of IL-6/LY75 signalling in cardiac remodelling (Timonen et al, 2008). For LY75, the dys-methylated CpGs reside within a classical CpG island covering exon 1 as well as part of the 5′ upstream region. Its transcriptional start site is predicted in very close vicinity (1395 bp). Our findings of significantly increased DNA methylation together with strongly reduced LY75-mRNA levels in DCM patients suggested a functional role. While it is to our knowledge not possible to recreate exactly the same methylation patterns in vitro as seen in the primary tissue, we could investigate here the functional consequence of global LY75 promoter methylation, suggesting a direct link between methylation and transcriptional activity. We also knocked down ly75 in the zebrafish model and found not only cardiac dysfunction in ly75-ablated embryos, but also a noticeable skin detachment phenotype, potentially due to disturbed collagen production. The adenosine receptor A2A (ADORA2A) is a member of the G protein-coupled receptor family and is highly abundant in neurons of basal ganglia, T lymphocytes, platelets and vasculature, but also shows high expression in myocardium as shown above. In the heart, activation of ADORA2A enhances cAMP production through αGs proteins (Sommerschild & Kirkeboen, 2000) and overexpression results in increased contractility and sarcoplasmic reticulum Ca2+ uptake (Hamad et al, 2010) as well as cardioprotection in ischemia and reperfusion damage (Urmaliya et al, 2009). Surprisingly, although ADORA2A methylation was reduced, we found mRNA levels to be significantly down-regulated. Such a positive relationship was described for other genes and is not fully understood in every case (Zhang et al, 2006). Our CGI annotation was based on the criteria by Takai and Jones (Takai & Jones, 2002), which define a CpG island as a nucleotide sequence of 200 bp or greater in length or with a GC content of 50% or more, or a ratio of observed versus expected CpGs of 0.6 or higher. In case of ADORA2A, the tested CGIs only met the second criterion and may therefore not be classical CGIs. For such sites, one explanation can be that hyper-methylation cannot only impede the binding of transcriptional activators but also can affect repressors of transcription. We therefore performed an in silico analysis of the promoter region of ADORA2A and found two binding sites for the transcription factor CTCF (CCCTC-binding factor) in close vicinity to both significantly hypo-methylated CGIs, but not within the single unaffected CGI of ADORA2A. CTCF is a unique insulator-binding protein (Bell et al, 1999; Felsenfeld et al, 2004; Gerasimova & Corces, 2001; West et al, 2002), which can act as transcriptional repressor by blocking distinct enhancer regions (Bell et al, 1999; Filippova et al, 1996; Gaszner & Felsenfeld, 2006; Lobanenkov et al, 1990; Ohlsson et al, 2001). Intriguingly, CTCF sites emerge as central players in regulatory networks linking gene regulation with epigenetic modifications (Bell & Felsenfeld, 2000; Ohlsson et al, 2001). Accordingly, our observed decrease in ADORA2A expression might be a result of enhanced binding of CTCF repressors due to CGI hypomethylation, which needs to be investigated in subsequent studies.
The detection of epigenetic mechanisms in human heart disease represents an attractive option to identify and dissect completely novel pathomechanisms. The validated targets from this study that have shown functional relevance are most likely modifiers of DCM rather than being independent disease causes. Therefore, it is reasonable to investigate their potential as diagnostic and therapeutic targets specifically in DCM but also in heart failure due to other causes. In the cancer field, epigenetic drugs have already entered the clinical arena (e.g. decitabine or azacitidine in the treatment of myelodysplastic syndromes), and methylation patterns are used as biomarkers to subtype and stage various cancers as a critical step towards a more effective personalized care (Coppede, 2011; Litzow, 2011). Hence, the genes identified here may be relevant druggable targets in DCM and heart failure and may aid in disease classification or risk stratification.
MATERIALS AND METHODS
Patients and controls
The study was conducted in accordance with the principles of the Declaration of Helsinki. All participants of the study have given written informed consent and the study was approved by the relevant ethics committees. DCM was diagnosed according to the guidelines of the World Health Organization (WHO; Richardson et al, 1995). Inclusion criteria for DCM cases were the presence of reduced left ventricular systolic function (left ventricular ejection fraction <45% assessed by echocardiography) in the absence of relevant coronary artery disease (CAD) as determined by coronary angiography. Patients with valvular or hypertensive heart disease, history of myocarditis, regular alcohol consumption or cardio-toxic chemotherapy were also excluded. The control biopsy specimens were obtained from patients who had received heart transplantation. All patients of the control cohort had successful cardiac transplantation more than 6 months ago with normal systolic and diastolic function and no evidence for relevant vasculopathy as judged by coronary angiography. Furthermore, all controls showed freedom from relevant acute or chronic organ rejection.
Processing of left ventricular biopsies
Biopsy specimens were obtained from the apical part of the free left ventricular wall (LV) from DCM patients or cardiac transplant patients (controls) undergoing cardiac catheterization using a standardized protocol. Biopsies were washed with NaCl (0.9%) and immediately transferred and stored in liquid nitrogen until DNA or RNA was extracted. DNA was extracted using the DNeasy blood and tissue kit, total RNA using the RNeasy kit according to the manufacturer's protocol (Qiagen, Germany). RNA purity and concentration were determined using the Bioanalyzer 2100 (Agilent Technologies, Berkshire, UK) with a Eukaryote Total RNA Pico assay chip. Since a critical issue for the reliability of gene expression analysis is the quality of the RNA samples (Vermeulen et al, 2011), a RNA integrity number (RIN) >6 was defined as minimum requirement for further analyses.
DNA methylation profiling and fine-mapping
For measuring methylation profiles, we used the Infinium HumanMethylation 27 BeadChip assay from Illumina with 1000 ng DNA per sample. The procedure followed the manufacturers standard workflow, starting with the bisulphite treatment of the sample DNA leading to a conversion of unmethylated cytosins to uracil, while 5-methylcytosines remain unchanged. After amplification and fragmentation of the bisulphite-converted DNA, they were hybridized to the Infinium BeadChips. DNA methylation data for DCM patients and controls have been deposited in NCBI's Gene Expression Omnibus (Edgar et al, 2002) and are accessible through GEO Series accession number GSE42510 (http://www.ncbi.nlm.nih.gov/sites/entrez?db=gds&term=GSE42510[Accession]&cmd=search). Fig 1A represents quantile normalized bead color signals (green and red) from 9350 consecutive probes on all further analysed arrays.
DNA methylation validation by MassARRAY and bisulphite sequencing
DNA methylation was validated by the MassARRAY technique as previously described (Ehrich et al, 2005). Briefly, 400–500 ng genomic DNA was chemically modified with sodium bisulfite using the EZ methylation kit (Zymo Research) according to the manufacturer's instructions. The bisulfite-treated DNA was PCR-amplified by primers designed to cover the Infinium probes that showed differential methylation at each locus. The amplicons were transcribed by T7 polymerase, followed by T-specific-RNAase-A cleavage. The digested fragments were quantified by MALDI-TOF-based technique. The primer sequences are given in Supporting Information Table 1. DNA methylation standards (0, 20, 40, 60, 80 and 100% methylated genomic DNA) were used to confirm the unbiased amplification of the amplicons. For LY75 and ADORA2A we additionally generated 14 and 12 clones, respectively, from two patients with DCM and two controls, each, and sequenced them before and after treatment with bisulfite. The methylation data was statistically analysed by Students t-test, or ANOVA, followed by Dunnett's multiple comparison.
Promoter luciferase assay
To assess the causative link of CGI methylation within the promoter region of LY75 with reduced mRNA expression observed in myocardial tissue, we performed a promoter luciferase assay essentially as described before (Smith et al, 2006). Therefore, we cloned a 1.6 kb large core fragment (Supporting Information Table 2) of the LY75 promoter region including the analysed CGI into SpeI/BamHI sites of the CpG-free luciferase vector pCpGL-basic. As negative control for the methylation effect we utilized the CpG-free control reporter vector pCpGL-CMV/EF1 (Klug & Rehli, 2006). Next, we treated both vectors with the methylase SssI (New England BioLabs) or leaved them untreated, respectively. The methylation was performed at 10 mM Tris, pH 7.9, 50 mM NaCl, 10 mM MgCl2, 1 mM DTT and 160 µM SAM at 37°C for 1 h (Blesa et al, 2008). Transfection of human HEK293A cells with equal amounts of the SssI-treated or untreated pCpG-LY75 or pCpGL-CMV/EF1 vectors was carried out using Lipofectamin (Invitrogen). Additionally, we co-transfected these cells with the pGL4.74 [hRluc/TK) Renilla control vector as a reference (Promega). After 48 h, cells were lysed and luciferase and Renilla activity measured according to manufactures protocol (Dual-Luciferase Reporter Assay System, Promega). Results are expressed as relative luciferase activity (background substracted and normalized to Renilla reference; three technical replicates) of the methylated versus the unmethylated constructs.
Promoter region prediction and transcription factor binding site analysis
We applied the Promoter 2.0 Prediction Software that predicts transcription start sites (TSS) of vertebrate Pol II promoters in genomic DNA to estimate the distance of the CGIs from the potential transcriptional start sites (Knudsen, 1999). For the detection of transcription factor binding sites, the ENCODE transcription factor ChIP-seq data was used. For each region (size of the CGI and 1500 bp up- and downstream), we selected in the maximum the three factors up- and three factors downstream with strongest observed binding.
Hierarchical clustering
To generate a graphical representation of the most significantly dys-methylated cardiac genes in the screening stage and to detect patterns among the respective genes, hierarchical clustering was applied. Prior to the clustering, the patients were ranked for each gene according to the degree of methylation, i.e. the patient with the lowest degree of methylation for a certain gene received a rank of 1, while the patient with the highest degree of methylation received a rank of 17, since 17 samples have met filter criteria for the screening stage. Then, for all samples and all genes the pair-wise Euclidian distances were computed and a bottom-up clustering on these distances was carried out. All computations were carried out using the statistical programming language R (Team-RDC, 2008).
Gene network analysis
To test whether genes with differential methylation patterns belong to a certain biological category, we carried out an unweighted gene set enrichment analysis (GSEA; Keller et al, 2007). In detail, we first sorted all genes according to the absolute value of the median distances between DCM patients and controls, generated a sorted list of genes where the most differentially methylated genes were on top while genes without differential methylation were located at the bottom of the list. If one gene was represented by multiple features on the biochip (different methylation sites), the median position of all replicates in the sorted list was computed. This sorted list was then used as input for GeneTrail (Backes et al, 2007). For each category, a significance value is computed by a dynamic programming approach (Keller et al, 2007) and all significance values of a certain category have been adjusted for multiple testing using Benjamini–Hochberg adjustment (Benjamini & Hochberg, 1995). The advantage of the applied cutoff-free biostatistical approach is that genes with p values of 0.049 and 0.051 are considered to be almost equally important in our approach while genes with larger values in the middle and at the end of the list are considered to be not of relevance. In contrast, classical over-representation analyses relying on cutoffs (usually 0.05) would consider genes with p-value of 0.049 of relevance while genes with p values of 0.051 would be considered to be non-relevant, having the same impact as genes with large p values.
The paper explained
PROBLEM:
Dilated cardiomyopathy (DCM) is one the most frequent heart muscle diseases. Although several factors including genetic mechanisms have been shown to cause DCM, we still find many cases unexplained and observe high phenotypic variability with respect to disease severity and prognosis. Epigenetic mechanisms are increasingly recognized as causes and modulators of human disease. Therefore, we studied genome-wide cardiac DNA methylation in DCM patients and controls to detect a possible epigenetic contribution to DCM.
RESULTS:
We detected distinct DNA methylation patterns in left ventricular heart tissue of DCM patients and reproduced the epigenetic mode of regulation for several genes with previously unknown function in DCM, namely Lymphocyte antigen 75 (LY75), Tyrosine kinase-type cell surface receptor HER3 (ERBB3), Homeobox B13 (HOXB13) and Adenosine receptor A2A (ADORA2A). The results were verified by alternative techniques in a well-phenotyped and large independent cohort of DCM patients and controls. Furthermore, we were able to show the functional relevance for the contribution of the identified genes in the pathogenesis of heart failure by using zebrafish as an in vivo model.
IMPACT:
Our results hint at a novel layer in the pathogenesis of DCM and heart failure and will likely impact on the development of novel biomarkers and future therapeutic strategies.
Transcriptomic analyses
Quantitative real-time PCR (q-PCR) was performed in order to measure expression of selected genes. Primers were designed using NCBI Primer-Blast and synthesized by Eurofins MWG Operon (Ebersberg, Germany). Sixty nanograms of total RNA extracted from biopsies of independent DCM patients (moderate: n = 7; severe: n = 5) and controls (n = 7) was reverse transcribed using SuperScript III first strand cDNA synthesis kit (Invitrogen). q-PCR was carried out according to standard protocols with the SYBR-Green method (Thermo Scientific) using an ABI 7000 system (ABI). Specificity of each primer-pair was monitored by dissociation curve analysis. Threshold cycle (CT) values were assessed in the exponential phase of amplification and the data were analysed using the delta-CT method. The mean value of the reference genes GAPDH, RPL13 and β-actin was used as a reference. To identify the predominant cardiac isoform of ADORA2A, we performed PCR with the following primer-pair for both isoforms (ENST00000337539 and ENST00000541988): 5′-CTGTGACATGGAGCAGGAGC-3′ and 5′-GCTGTCGTTTGCCATCGGCCT-3′. To evaluate the expression levels of ly75 and adora2a in the zebrafish heart and whole organism, we performed q-PCR with the following primer-pair for ly75: 5′-CATGGCCAGTTTCGATCCAT-3′ and 5′-CACCTGGGACTACACCTCCT-3′ and adora2a: 5′-TGCTGACCCAGAGCTCCATA-3′ and 5′-AGAGGCATCATCGCGATCTG-3′. Results are shown as relative expression values (normalization against the housekeeping gene elf1a). To analyze the expression of ly75 and adora2a in different cell types, we cultured neonatal rat cardiomyocytes and cardiofibroblasts for 5 days and proceeded as described above. q-PCR was performed using the primer pairs 18S RNA 5′-GGACATCTAAGGGCATCAC-3′ and 5′-CCTCCGACTTTCGTTCTTGA-3′; LY75 5′-CACGGTCTGATGAGCTGTGT-3′ and 5′-ACGAACTGCAACCTGACCAT-3′; ADORA2A 5′-CTGGTCCTCACGCAGAGTTC-3′ and 5′-GCGAAGGGCATCATTGCAAT-3′. The methylation data was statistically analysed by Students t-test.
Morpholino-mediated gene knockdown in zebrafish
The zebrafish experiments were performed under institutional approvals that conform to the Guide for the Care and Use of Laboratory Animals published by The US National Institute of Health (NIH Publication No. 85-23, revised 1996). Care and breeding of zebrafish (Danio rerio) were as described (Meder et al, 2011a, b). The following Morpholino-modified antisense oligonucleotides (GeneTools, USA) were designed: The ly75 (ENSDARG00000053113) Morpholino targets the splice-donor site of exon 3 (MO-ly75: 5′-GTGATGAAACGCACACCTCTCCTGA-3′; scrambled control MO3-control: 5′-GTCATGAAAGGCACACGTCTGCTCA-3′), while adora2a (ENSDARG00000033706) was targeted at its start-site (MO-adora2a: 5′-CATTGTTCAGCATGGTGAGGTCGCT-3′; scrambled control MO2-control: 5′-CATTCTTCACCATCGTGACGTGGCT-3′). Morpholinos or control oligonucleotides (MO-control) were injected into 1-cell stage embryos as described before (Meder et al, 2009). To confirm the efficiency of splice-site Morpholinos, we analysed their target region by cDNA splice-site analysis (Meder et al, 2011a, b).
Functional assessment and statistical analysis of cardiac function
Still images and high-resolution video microscopy movies were recorded and digitized with a Zeiss microscope/MCU II. The functional assessment of cardiac contractility was carried out as described before (Meder et al, 2009). Atrial and ventricular diameters were measured to calculate fractional shortening with the help of the zebraFS software (http://www.mederlab.com). Results are expressed as mean ± standard deviation. MF20/S46 stainings and mRNA antisense in situ hybridization was performed as previously described (Meder et al, 2009).
Acknowledgments
This work was supported by grants from the German Ministry of Education and Research (BMBF): NGFN-plus 01GS0836, NGFN-transfer 01GR0823, INSIGHT DCM, DZHK (‘Deutsches Zentrum für Herz-Kreislauf-Forschung’—German Centre for Cardiovascular Research), the University of Heidelberg (Innovationsfond FRONTIER), and the European Union (FP7 BestAgeing and INHERITANCE). Y. J. P. was supported by Roman Herzog research fellowship from Hertie Foundation and by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST; 2012018819).
Supporting Information is available at EMBO Molecular Medicine Online.
The authors declare that they have no conflict of interest.
Author contributions
BM, JH, KSF, YJP, DW, JDH, HAK, CP designed the study and experiments. JH, KSF, YJP, AB, SM, SaH, RN performed the experiments and analysed data together with AK, AML and BM. HAK, BM, BV, AD and JF provided samples. BM wrote the manuscript. JB, SF, NMW, DK, StH, DM, PE, JHS, HAK, CP, and TW edited the paper. FSH and EK analysed data.
Supplementary material
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer reviewed and may be re-organized for online delivery, but are not copy-edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
References
- Backes C, Keller A, Kuentzer J, Kneissl B, Comtesse N, Elnakady YA, Muller R, Meese E, Lenhof HP. GeneTrail—advanced gene set enrichment analysis. Nucleic Acids Res. 2007;35((web server issue)):W186–W192. doi: 10.1093/nar/gkm323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Backs J, Song K, Bezprozvannaya S, Chang S, Olson EN. CaM kinase II selectively signals to histone deacetylase 4 during cardiomyocyte hypertrophy. J Clin Invest. 2006;116:1853–1864. doi: 10.1172/JCI27438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Backs J, Backs T, Bezprozvannaya S, McKinsey TA, Olson EN. Histone deacetylase 5 acquires calcium/calmodulin-dependent kinase II responsiveness by oligomerization with histone deacetylase 4. Mol Cell Biol. 2008;28:3437–3445. doi: 10.1128/MCB.01611-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Backs J, Worst BC, Lehmann LH, Patrick DM, Jebessa Z, Kreusser MM, Sun Q, Chen L, Heft C, Katus HA, et al. Selective repression of MEF2 activity by PKA-dependent proteolysis of HDAC4. J Cell Biol. 2011;195:403–415. doi: 10.1083/jcb.201105063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bell AC, Felsenfeld G. Methylation of a CTCF-dependent boundary controls imprinted expression of the Igf2 gene. Nature. 2000;405:482–485. doi: 10.1038/35013100. [DOI] [PubMed] [Google Scholar]
- Bell AC, West AG, Felsenfeld G. The protein CTCF is required for the enhancer blocking activity of vertebrate insulators. Cell. 1999;98:387–396. doi: 10.1016/s0092-8674(00)81967-4. [DOI] [PubMed] [Google Scholar]
- Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B. 1995;57:289–300. [Google Scholar]
- Blesa JR, Hegde AA, Hernandez-Yago J. In vitro methylation of nuclear respiratory factor-2 binding sites suppresses the promoter activity of the human TOMM70 gene. Gene. 2008;427:58–64. doi: 10.1016/j.gene.2008.09.016. [DOI] [PubMed] [Google Scholar]
- Chen JF, Murchison EP, Tang R, Callis TE, Tatsuguchi M, Deng Z, Rojas M, Hammond SM, Schneider MD, Selzman CH, et al. Targeted deletion of Dicer in the heart leads to dilated cardiomyopathy and heart failure. Proc Natl Acad Sci USA. 2008;105:2111–2116. doi: 10.1073/pnas.0710228105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppede F. Epigenetic biomarkers of colorectal cancer: focus on DNA methylation. Cancer Lett. 2011 DOI 10.1016/j.canlet.2011.12.030. [Google Scholar]
- Dahme T, Katus HA, Rottbauer W. Fishing for the genetic basis of cardiovascular disease. Dis Model Mech. 2009;2:18–22. doi: 10.1242/dmm.000687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dec GW, Fuster V. Idiopathic dilated cardiomyopathy. N Engl J Med. 1994;331:1564–1575. doi: 10.1056/NEJM199412083312307. [DOI] [PubMed] [Google Scholar]
- Edgar R, Domrachev M, Lash AE. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res. 2002;30:207–210. doi: 10.1093/nar/30.1.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrich M, Nelson MR, Stanssens P, Zabeau M, Liloglou T, Xinarianos G, Cantor CR, Field JK, van den Boom D. Quantitative high-throughput analysis of DNA methylation patterns by base-specific cleavage and mass spectrometry. Proc Natl Acad Sci USA. 2005;102:15785–15790. doi: 10.1073/pnas.0507816102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg AP, Tycko B. The history of cancer epigenetics. Nat Rev Cancer. 2004;4:143–153. doi: 10.1038/nrc1279. [DOI] [PubMed] [Google Scholar]
- Felsenfeld G, Burgess-Beusse B, Farrell C, Gaszner M, Ghirlando R, Huang S, Jin C, Litt M, Magdinier F, Mutskov V, et al. Chromatin boundaries and chromatin domains. Cold Spring Harb Symp Quant Biol. 2004;69:245–250. doi: 10.1101/sqb.2004.69.245. [DOI] [PubMed] [Google Scholar]
- Filippova GN, Fagerlie S, Klenova EM, Myers C, Dehner Y, Goodwin G, Neiman PE, Collins SJ, Lobanenkov VV. An exceptionally conserved transcriptional repressor, CTCF, employs different combinations of zinc fingers to bind diverged promoter sequences of avian and mammalian c-myc oncogenes. Mol Cell Biol. 1996;16:2802–2813. doi: 10.1128/mcb.16.6.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flotho C, Claus R, Batz C, Schneider M, Sandrock I, Ihde S, Plass C, Niemeyer CM, Lubbert M. The DNA methyltransferase inhibitors azacitidine, decitabine and zebularine exert differential effects on cancer gene expression in acute myeloid leukemia cells. Leukemia. 2009;23:1019–1028. doi: 10.1038/leu.2008.397. [DOI] [PubMed] [Google Scholar]
- Friedrichs F, Zugck C, Rauch GJ, Ivandic B, Weichenhan D, Muller-Bardorff M, Meder B, El Mokhtari NE, Regitz-Zagrosek V, Hetzer R, et al. HBEGF, SRA1, and IK: three cosegregating genes as determinants of cardiomyopathy. Genome Res. 2009;19:395–403. doi: 10.1101/gr.076653.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gambari R, Fabbri E, Borgatti M, Lampronti I, Finotti A, Brognara E, Bianchi N, Manicardi A, Marchelli R, Corradini R. Targeting microRNAs involved in human diseases: a novel approach for modification of gene expression and drug development. Biochem Pharmacol. 2011;82:1416–1429. doi: 10.1016/j.bcp.2011.08.007. [DOI] [PubMed] [Google Scholar]
- Gaszner M, Felsenfeld G. Insulators: exploiting transcriptional and epigenetic mechanisms. Nat Rev Genet. 2006;7:703–713. doi: 10.1038/nrg1925. [DOI] [PubMed] [Google Scholar]
- Gerasimova TI, Corces VG. Chromatin insulators and boundaries: effects on transcription and nuclear organization. Annu Rev Genet. 2001;35:193–208. doi: 10.1146/annurev.genet.35.102401.090349. [DOI] [PubMed] [Google Scholar]
- Ghoshal K, Ghoshal K, Motiwala T, Claus R, Yan P, Kutay H, Datta J, Majumder S, Bai S, Majumder A, Huang T, et al. HOXB13, a target of DNMT3B, is methylated at an upstream CpG island, and functions as a tumor suppressor in primary colorectal tumors. PLoS ONE. 2010;5:e10338. doi: 10.1371/journal.pone.0010338. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Giridhar PV, Funk HM, Gallo CA, Porollo A, Mercer CA, Plas DR, Drew AF. Interleukin-6 receptor enhances early colonization of the murine omentum by upregulation of a mannose family receptor, LY75, in ovarian tumor cells. Clin Exp Metastasis. 2011;28:887–897. doi: 10.1007/s10585-011-9420-x. [DOI] [PubMed] [Google Scholar]
- Grunig E, Tasman JA, Kucherer H, Franz W, Kubler W, Katus HA. Frequency and phenotypes of familial dilated cardiomyopathy. J Am Coll Cardiol. 1998;31:186–194. doi: 10.1016/s0735-1097(97)00434-8. [DOI] [PubMed] [Google Scholar]
- Grzeskowiak R, Witt H, Drungowski M, Thermann R, Hennig S, Perrot A, Osterziel KJ, Klingbiel D, Scheid S, Spang R. Expression profiling of human idiopathic dilated cardiomyopathy. Cardiovasc Res. 2003;59:400–411. doi: 10.1016/s0008-6363(03)00426-7. [DOI] [PubMed] [Google Scholar]
- Hamad EA, Li X, Song J, Zhang XQ, Myers V, Funakoshi H, Zhang J, Wang J, Li J, Swope D. Effects of cardiac-restricted overexpression of the A(2A) adenosine receptor on adriamycin-induced cardiotoxicity. Am J Physiol Heart Circ Physiol. 2010;298:H1738–H1747. doi: 10.1152/ajpheart.00688.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herceg Z, Vaissiere T. Epigenetic mechanisms and cancer: an interface between the environment and the genome. Epigenetics. 2011;6:804–819. doi: 10.4161/epi.6.7.16262. [DOI] [PubMed] [Google Scholar]
- Ho L, Crabtree GR. Chromatin remodelling during development. Nature. 2010;463:474–484. doi: 10.1038/nature08911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Humphreys DT, Westman BJ, Martin DI, Preiss T. MicroRNAs control translation initiation by inhibiting eukaryotic initiation factor 4E/cap and poly(A) tail function. Proc Natl Acad Sci USA. 2005;102:16961–16966. doi: 10.1073/pnas.0506482102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jirtle RL, Skinner MK. Environmental epigenomics and disease susceptibility. Nat Rev Genet. 2007;8:253–262. doi: 10.1038/nrg2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones PA, Baylin SB. The fundamental role of epigenetic events in cancer. Nat Rev Genet. 2002;3:415–428. doi: 10.1038/nrg816. [DOI] [PubMed] [Google Scholar]
- Karkkainen S, Peuhkurinen K. Genetics of dilated cardiomyopathy. Ann Med. 2007;39:91–107. doi: 10.1080/07853890601145821. [DOI] [PubMed] [Google Scholar]
- Keller A, Backes C, Lenhof HP. Computation of significance scores of unweighted gene set enrichment analyses. BMC Bioinform. 2007;8:290–297. doi: 10.1186/1471-2105-8-290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keller A, Leidinger P, Bauer A, Elsharawy A, Haas J, Backes C, Wendschlag A, Giese N, Tjaden C, Ott K, et al. Toward the blood-borne miRNome of human diseases. Nat Methods. 2011;8:841–843. doi: 10.1038/nmeth.1682. [DOI] [PubMed] [Google Scholar]
- Klug M, Rehli M. Functional analysis of promoter CpG methylation using a CpG-free luciferase reporter vector. Epigenetics. 2006;1:127–130. doi: 10.4161/epi.1.3.3327. [DOI] [PubMed] [Google Scholar]
- Knudsen S. Promoter2.0: for the recognition of PolII promoter sequences. Bioinformatics. 1999;15:356–361. doi: 10.1093/bioinformatics/15.5.356. [DOI] [PubMed] [Google Scholar]
- Litzow MR. Novel therapeutic approaches for acute lymphoblastic leukemia. Hematol Oncol Clin North Am. 2011;25:1303–1317. doi: 10.1016/j.hoc.2011.09.019. [DOI] [PubMed] [Google Scholar]
- Lobanenkov VV, Nicolas RH, Adler VV, Paterson H, Klenova EM, Polotskaja AV, Goodwin GH. A novel sequence-specific DNA binding protein which interacts with three regularly spaced direct repeats of the CCCTC-motif in the 5′-flanking sequence of the chicken c-myc gene. Oncogene. 1990;5:1743–1753. [PubMed] [Google Scholar]
- Meder B, Laufer C, Hassel D, Just S, Marquart S, Vogel B, Hess A, Fishman MC, Katus HA, Rottbauer W. A single serine in the carboxyl terminus of cardiac essential myosin light chain-1 controls cardiomyocyte contractility in vivo. Circ Res. 2009;104:650–659. doi: 10.1161/CIRCRESAHA.108.186676. [DOI] [PubMed] [Google Scholar]
- Meder B, Keller A, Vogel B, Haas J, Sedaghat-Hamedani F, Kayvanpour E, Just S, Borries A, Rudloff J, Leidinger P, et al. MicroRNA signatures in total peripheral blood as novel biomarkers for acute myocardial infarction. Basic Res Cardiol. 2011a;106:13–23. doi: 10.1007/s00395-010-0123-2. [DOI] [PubMed] [Google Scholar]
- Meder B, Huttner IG, Sedaghat-Hamedani F, Just S, Dahme T, Frese KS, Vogel B, Kohler D, Kloos W, Rudloff J, et al. PINCH proteins regulate cardiac contractility by modulating integrin-linked kinase-protein kinase B signaling. Mol Cell Biol. 2011b;31:3424–3435. doi: 10.1128/MCB.05269-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meder B, Katus HA. Clinical and genetic aspects of hypertrophic and dilated cardiomyopathy. Internist (Berl) 2012;53:408–418. doi: 10.1007/s00108-011-2988-z. [DOI] [PubMed] [Google Scholar]
- Meder B, Katus HA, Rottbauer W. Right into the heart of microRNA-133a. Genes Dev. 2008;22:3227–3231. doi: 10.1101/gad.1753508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meurs KM, Kuan M. Differential methylation of CpG sites in two isoforms of myosin binding protein C, an important hypertrophic cardiomyopathy gene. Environ Mol Mutagen. 2011;52:161–164. doi: 10.1002/em.20596. [DOI] [PubMed] [Google Scholar]
- Mirbahai L, Yin G, Bignell JP, Li N, Williams TD, Chipman JK, et al. DNA methylation in liver tumorigenesis in fish from the environment. Epigenetics. 2011;6:1319–1333. doi: 10.4161/epi.6.11.17890. [DOI] [PubMed] [Google Scholar]
- Montgomery RL, Davis CA, Potthoff MJ, Haberland M, Fielitz J, Qi X, Hill JA, Richardson JA, Olson EN. Histone deacetylases 1 and 2 redundantly regulate cardiac morphogenesis, growth, and contractility. Genes Dev. 2007;21:1790–1802. doi: 10.1101/gad.1563807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montgomery RL, Potthoff MJ, Haberland M, Qi X, Matsuzaki S, Humphries KM, Richardson JA, Bassel-Duby R, Olson EN. Maintenance of cardiac energy metabolism by histone deacetylase 3 in mice. J Clin Invest. 2008;118:3588–3597. doi: 10.1172/JCI35847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Movassagh M, Choy MK, Goddard M, Bennett MR, Down TA, Foo RS, et al. Differential DNA methylation correlates with differential expression of angiogenic factors in human heart failure. PLoS ONE. 2010;5:e8564. doi: 10.1371/journal.pone.0008564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Movassagh M, Choy MK, Knowles DA, Cordeddu L, Haider S, Down T, Siggens L, Vujic A, Simeoni I, Penkett C, et al. Distinct epigenomic features in end-stage failing human hearts. Circulation. 2011;124:2411–2422. doi: 10.1161/CIRCULATIONAHA.111.040071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohlsson R, Renkawitz R, Lobanenkov V. CTCF is a uniquely versatile transcription regulator linked to epigenetics and disease. Trends Genet. 2001;17:520–527. doi: 10.1016/s0168-9525(01)02366-6. [DOI] [PubMed] [Google Scholar]
- Prasad MK, Reed X, Gorkin DU, Cronin JC, McAdow AR, Chain K, Hodonsky CJ, Jones EA, Svaren J, Antonellis A, et al. SOX10 directly modulates ERBB3 transcription via an intronic neural crest enhancer. BMC Dev Biol. 2011;11:40. doi: 10.1186/1471-213X-11-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson P, McKenna W, Bristow M, Maisch B, Mautner B, O'Connell J, Olsen E, Thiene G, Goodwin J, Gyarfas I, et al. Report of the (1995) World Health Organization/International Society and Federation of Cardiology Task Force on the Definition and Classification of cardiomyopathies. Circulation. 1996;93:841–842. doi: 10.1161/01.cir.93.5.841. [DOI] [PubMed] [Google Scholar]
- Smith LT, Sanders JZ, Kaiser RJ, Hughes P, Dodd C, Connell CR, Heiner C, Kent SB, Hood LE. Epigenetic regulation of the tumor suppressor gene TCF21 on 6q23-q24 in lung and head and neck cancer. Proc Natl Acad Sci USA. 2006;103:982–987. doi: 10.1073/pnas.0510171102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sommerschild HT, Kirkeboen KA. Adenosine and cardioprotection during ischaemia and reperfusion—an overview. Acta Anaesthesiol Scand. 2000;44:1038–1055. doi: 10.1034/j.1399-6576.2000.440903.x. [DOI] [PubMed] [Google Scholar]
- Takai D, Jones PA. Comprehensive analysis of CpG islands in human chromosomes 21 and 22. Proc Natl Acad Sci USA. 2002;99:3740–3745. doi: 10.1073/pnas.052410099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talens RP, Boomsma DI, Tobi EW, Kremer D, Jukema JW, Willemsen G, Putter H, Slagboom PE, Heijmans BT. Variation, patterns, and temporal stability of DNA methylation: considerations for epigenetic epidemiology. FASEB J. 2010;24:3135–3144. doi: 10.1096/fj.09-150490. [DOI] [PubMed] [Google Scholar]
- Team-RDC. R: A Language and Environment for Statistical Computing. Vienna, Austria: R Foundation for Statistical Computing; 2008. [Google Scholar]
- Tel J, Benitez-Ribas D, Hoosemans S, Cambi A, Adema GJ, Figdor CG, Tacken PJ, de Vries IJ. DEC-205 mediates antigen uptake and presentation by both resting and activated human plasmacytoid dendritic cells. Eur J Immunol. 2011;41:1014–1023. doi: 10.1002/eji.201040790. [DOI] [PubMed] [Google Scholar]
- Timonen P, Magga J, Risteli J, Punnonen K, Vanninen E, Turpeinen A, Tuomainen P, Kuusisto J, Vuolteenaho O, Peuhkurinen K. Cytokines, interstitial collagen and ventricular remodelling in dilated cardiomyopathy. Int J Cardiol. 2008;124:293–300. doi: 10.1016/j.ijcard.2007.02.004. [DOI] [PubMed] [Google Scholar]
- Tryndyak VP, Han T, Muskhelishvili L, Fuscoe JC, Ross SA, Beland FA, Pogribny IP. Coupling global methylation and gene expression profiles reveal key pathophysiological events in liver injury induced by a methyl-deficient diet. Mol Nutr Food Res. 2011;55:411–418. doi: 10.1002/mnfr.201000300. [DOI] [PubMed] [Google Scholar]
- Urmaliya VB, Church JE, Coupar IM, Rose'Meyer RB, Pouton CW, White PJ. Cardioprotection induced by adenosine A1 receptor agonists in a cardiac cell ischemia model involves cooperative activation of adenosine A2A and A2B receptors by endogenous adenosine. J Cardiovasc Pharmacol. 2009;53:424–433. doi: 10.1097/FJC.0b013e3181a443e2. [DOI] [PubMed] [Google Scholar]
- van Rooij E, Sutherland LB, Qi X, Richardson JA, Hill J, Olson EN. Control of stress-dependent cardiac growth and gene expression by a microRNA. Science. 2007;316:575–579. doi: 10.1126/science.1139089. [DOI] [PubMed] [Google Scholar]
- Vermeulen J, De Preter K, Lefever S, Nuytens J, De Vloed F, Derveaux S, Hellemans J, Speleman F, Vandesompele J. Measurable impact of RNA quality on gene expression results from quantitative PCR. Nucleic Acids Res. 2011;39:e63. doi: 10.1093/nar/gkr065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villard E, Perret C, Gary F, Proust C, Dilanian G, Hengstenberg C, Ruppert V, Arbustini E, Wichter T, Germain M. A genome-wide association study identifies two loci associated with heart failure due to dilated cardiomyopathy. Eur Heart J. 2011;32:1065–1076. doi: 10.1093/eurheartj/ehr105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West AG, Gaszner M, Felsenfeld G. Insulators: many functions, many mechanisms. Genes Dev. 2002;16:271–288. doi: 10.1101/gad.954702. [DOI] [PubMed] [Google Scholar]
- Yang X, Ewald ER, Huo Y, Tamashiro KL, Salvatori R, Sawa A, Wand GS, Lee RS. Glucocorticoid-induced loss of DNA methylation in non-neuronal cells and potential involvement of DNMT1 in epigenetic regulation of Fkbp5. Biochem Biophys Res Commun. 2012;420:570–575. doi: 10.1016/j.bbrc.2012.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Yazaki J, Sundaresan A, Cokus S, Chan SW, Chen H, Henderson IR, Shinn P, Pellegrini M, Jacobsen SE, et al. Genome-wide high-resolution mapping and functional analysis of DNA methylation in arabidopsis. Cell. 2006;126:1189–1201. doi: 10.1016/j.cell.2006.08.003. [DOI] [PubMed] [Google Scholar]
- Zhou B, Margariti A, Zeng L, Xu Q. Role of histone deacetylases in vascular cell homeostasis and arteriosclerosis. Cardiovasc Res. 2011;90:413–420. doi: 10.1093/cvr/cvr003. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.