Abstract
The goals of this study were to determine if the muscle contributions to vertical and fore-aft acceleration of the mass center differ between crouch gait and unimpaired gait and if these muscle contributions change with crouch severity. Examining muscle contributions to mass center acceleration provides insight into the roles of individual muscles during gait and can provide guidance for treatment planning. We calculated vertical and fore-aft accelerations using musculoskeletal simulations of typically-developing children and children with cerebral palsy and crouch gait. Analysis of these simulations revealed that during unimpaired gait the quadriceps produce large upward and backward accelerations during early stance, whereas the ankle plantarflexors produce large upward and forward accelerations later in stance. In contrast, during crouch gait, the quadriceps and ankle plantarflexors produce large, opposing fore-aft accelerations throughout stance. The quadriceps force required to accelerate the mass center upward was significantly larger in crouch gait than in unimpaired gait and increased with crouch severity. The gluteus medius accelerated the mass center upward during midstance in unimpaired gait; however, during crouch gait the upward acceleration produced by the gluteus medius was significantly reduced. During unimpaired gait the quadriceps and ankle plantarflexors accelerate the mass center at different times, efficiently modulating fore-aft accelerations. However, during crouch gait, the quadriceps and ankle plantarflexors produce fore-aft accelerations at the same time and the opposing fore-aft accelerations generated by these muscles contribute to the inefficiency of crouch gait.
Keywords: cerebral palsy, crouch gait, induced acceleration analysis, muscle, simulation
Introduction
Many individuals with cerebral palsy walk in a crouch gait pattern characterized by excess hip and knee flexion [1]. Walking in a crouched posture can lead to joint pain [2] and bone deformities [3] and is inefficient [4,5]. Understanding how muscles accelerate the mass center during crouch gait may provide insight about the underlying mechanics and inefficiencies associated with this gait pattern.
Previous studies have documented how muscles accelerate the mass center during unimpaired gait [6–8] and have shown that the same muscles that accelerate the mass center upward also modulate fore-aft acceleration [9]. Examining muscle contributions to mass center acceleration can provide insight into the role of individual muscles and the control strategy used during gait. During unimpaired gait, the vasti and gluteus maximus accelerate the mass center upward and backward in early stance, the gluteus medius accelerates the mass center upward in mid stance, and the ankle plantarflexors accelerate the mass center upward and forward in late stance [6–8]. This coordinated muscle activity uses different muscle groups at different periods of stance to efficiently modulate vertical and fore-aft mass center accelerations.
How muscles modulate vertical and fore-aft accelerations of the mass center during crouch gait and how these accelerations change with crouch severity is not well understood. Previous work has shown that similar muscles accelerate the mass center upward during the single-limb stance phase of both mild crouch gait and unimpaired gait [10]. However, the role of muscles during the double support phase, when fore-aft accelerations are largest, is not known for crouch gait. The ability of muscles such as the gluteus medius and soleus to extend the hip and knee [11] and accelerate the mass center upward [12] has been shown to decrease in a crouched posture; however, the effect of crouch severity on muscle contributions to mass center acceleration has not been investigated. Alterations in the ability of muscles to generate vertical and fore-aft accelerations of the mass center may change the strategy used to accelerate the mass center during crouch gait and contribute to the inefficiency of this gait pattern.
The goals of this study were to determine whether: (1) the magnitude and timing of muscle contributions to vertical and fore-aft accelerations differ between crouch gait and unimpaired gait, and (2) the muscle contributions to vertical and fore-aft accelerations change with crouch severity. To calculate muscle contributions to mass center accelerations we created three-dimensional musculoskeletal simulations of gait for typically-developing children and children with cerebral palsy and varying degrees of crouch gait (Fig. 1). Examining how individual muscles contribute to vertical and fore-aft accelerations can elucidate the mechanics of crouch gait and inform treatment strategies.
Figure 1.
Musculoskeletal model of an individual with cerebral palsy and crouch gait. Vertical and fore-aft accelerations of the mass center were calculated by analyzing muscle-driven simulations.
Methods
Nine children with cerebral palsy were selected from a database of subjects who had undergone motion analysis at Gillette Children’s Specialty Healthcare. The selection criteria for the subjects included: (1) a diagnosis of spastic diplegic cerebral palsy, (2) a minimum knee flexion angle during stance greater than 15°, and (3) a tibial and femoral torsion deformity less than 30° [13]. We also required that the subjects did not use an assistive device during the motion analysis and had at least two consecutive force plate strikes. Nine subjects with crouch gait were divided into three groups based on minimum knee flexion angle during stance: 15 – 30° knee flexion was defined as mild crouch gait, 30 – 50° was defined as moderate crouch gait, and 50° or larger was defined as severe crouch gait (Table 1).
Table 1.
Subject characteristics (average ± standard deviation)
| N | N (with EMG) | Age (years) | Height (cm) | Mass (kg) | KFA* (deg) | |
|---|---|---|---|---|---|---|
| Unimpaired | 3 | 3 | 10 ± 3 | 144 ± 16 | 36 ± 9 | −2 ± 4 |
| Mild Crouch | 3 | 3 | 9 ± 1 | 124 ± 10 | 24 ± 4 | 18 ± 2 |
| Moderate Crouch | 3 | 1 | 11 ± 2 | 136 ± 6 | 43 ± 31 | 34 ± 2 |
| Severe Crouch | 3 | 2 | 14 ± 2 | 157 ± 12 | 41 ± 8 | 64 ± 20 |
KFA: Minimum knee flexion angle during stance
The typically-developing children were selected from a group of subjects who also visited Gillette Children’s Specialty Healthcare for motion analysis and whose gait has been previously simulated [7]. Previous studies demonstrated consistent contributions to vertical and fore-aft accelerations during gait in typically-developing children; thus, we chose three subjects with an age range similar to the subjects with crouch gait (Table 1).
Motion analysis data was collected using a 12-camera system (Vicon Motion Systems, Lake Forest, CA) and a standard marker measurement protocol [14]. Ground reaction forces were measured with four force plates (AMTI, Watertown, MA). All subjects walked barefoot at their self-selected speed. Surface electromyography (EMG) was recorded from the rectus femoris, hamstrings, gastrocnemius, and tibialis anterior (Motion Laboratory Systems, Baton Rouge, LA) for nine of the subjects. The nine subjects who had EMG data available included three typically-developing subjects, three mild crouch gait subjects, one moderate crouch gait subject, and two severe crouch gait subjects. The EMG signals were sampled at 1080 Hz, bandpass filtered between 20 and 400 Hz, rectified, and low-pass filtered at 10 Hz. The magnitude of the EMG signal was normalized for each muscle group from zero to one based on the minimum and maximum values observed during the motion analysis. Since EMG was not available for all subjects, we averaged the processed EMG signals for each group of subjects for comparison to simulated muscle activations.
To create dynamic musculoskeletal simulations of gait, we used a musculoskeletal model with 19 degrees of freedom and 92 musculotendon actuators (lower extremities from [15] and torso from [16]). The degrees of freedom in the model included three translations and three rotations of the pelvis, a ball-and-socket joint between the pelvis and the torso located at the third lumbar vertebrae, ball-and-socket joints at each hip, a custom joint with coupled translations and rotations at each knee, and a revolute joint at each ankle. This model has previously been used to model typically-developing children [7,9] and children with cerebral palsy [10,11,17]. The model was scaled according to anthropometric measurements for each subject using OpenSim [18]. We scaled the maximum isometric force of all muscles by height-squared [19].
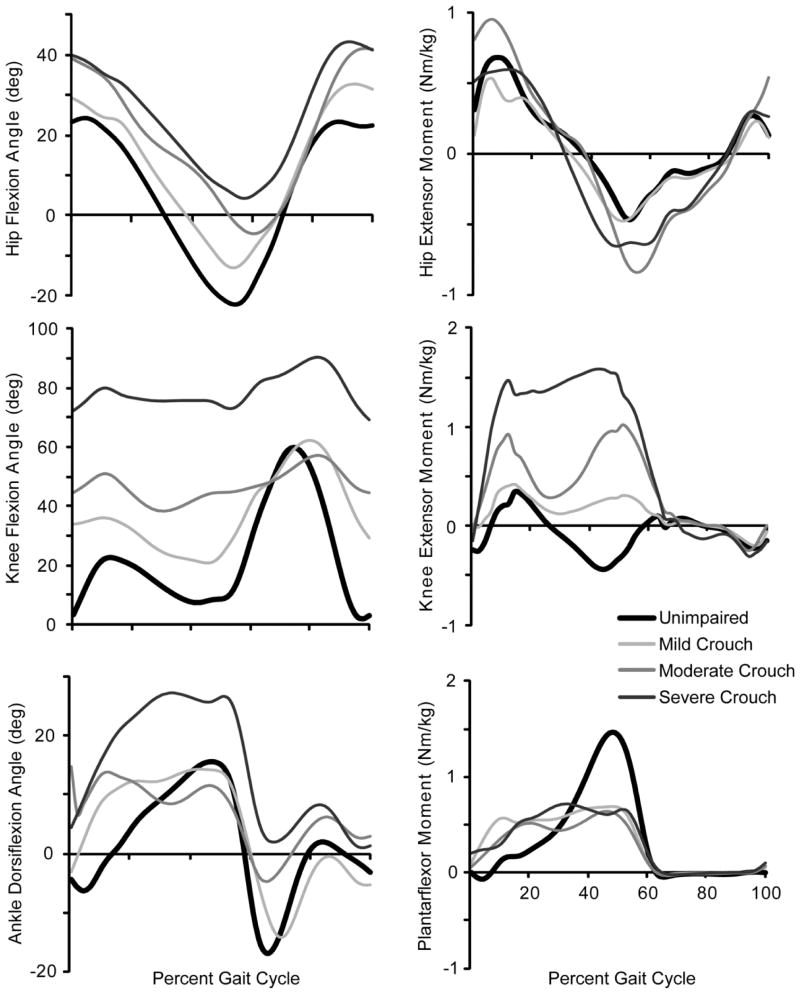
Inverse kinematics, which minimizes the difference between experimental marker trajectories and markers placed on the model, was used to calculate joint angles (Fig. 2A). Joint moments were calculated for each subject using inverse dynamics (Fig. 2B).
Figure 2.
Average hip, knee, and ankle flexion angles and moments during unimpaired gait and mild, moderate, and severe crouch gait. The joint moments are normalized by body mass (kg).
A dynamic simulation of one gait cycle was generated for each subject. The residual reduction algorithm (RRA) was used to reduce residuals at the pelvis [18]. Residuals are non-physiological forces and moments applied at the pelvis that balance dynamic inconsistencies resulting from experimental errors and modeling assumptions. RRA reduced these residuals by making small changes to measurements that may have experimental error including the position of the torso mass center (changes were less than 2 cm in magnitude) and the kinematics (changes were less than 2° for all joint angles). Using the adjusted model and kinematics determined from RRA, the computed muscle control algorithm (CMC) was used to estimate the muscle forces required to track each subject’s kinematics [20,21]. At each time step, CMC determines the distribution of muscle activations, such that model accelerations match the experimental accelerations for all degrees-of-freedom. The algorithm accounts for muscle activation and contraction dynamics, which includes the muscle force-length-velocity relationship. The distribution of muscle activations was determined by minimizing the sum of squared activations at each time step. The estimated muscle activations from CMC were qualitatively compared to the average EMG signal for each gait pattern; constraints on muscle excitations were used when the simulated muscle activity was inconsistent with the EMG signals.
An induced acceleration analysis was used to compute the contributions of individual muscles to vertical and fore-aft mass center accelerations [6,22–24] at each time point of a subject’s gait simulation. This analysis solves the model’s equations of motion, including foot-floor contact constraint equations, to determine the contributions of gravity, velocity, and each muscle force to the mass center acceleration. The contact constraint equations used in this analysis enforce a rolling without slipping constraint to model the foot-floor interaction [22].
Before addressing the research questions posed in this study, we evaluated the accuracy of the musculoskeletal simulations to ensure they recreated the dynamics of gait for the typically-developing children and children with cerebral palsy. The average residual forces and moments applied at the mass center of the pelvis were 9.7 N and 16.5 Nm, less than ten percent of bodyweight and body-weight*height, respectively, for all subjects. The on-off timing and magnitude of the simulated activations and electromyography were qualitatively similar (Supp. Fig. 1); however, there were some deviations, such as decreased quadriceps activity during early swing of unimpaired gait and increased gastrocnemius activity during swing of severe crouch gait. These differences had minimal effects on muscle contributions to vertical and fore-aft accelerations of the mass center, since these accelerations are generated predominately during stance.
To determine if the muscle contributions to vertical and fore-aft accelerations differed between unimpaired gait and crouch gait, we performed a student’s t-test with a significance level of 0.05. To determine if contributions to vertical and fore-aft accelerations changed with crouch severity we used a one-way ANOVA to compare mild, moderate, and severe crouch gait with a significance level of 0.05. To evaluate the relative timing of muscle contributions to fore-aft accelerations we used a covariance analysis which examined if major muscle groups, such as the ankle plantarflexors and vasti, were active at similar times during the gait cycle. To examine the relative magnitude of muscle contributions to fore-aft accelerations we calculated the area under the fore-aft acceleration versus gait cycle curves for major muscle groups. For both of these analyses, we compared unimpaired gait and crouch gait using a student’s t-test and mild, moderate, and severe crouch gait using a one-way ANOVA.
Results
The vasti (p = 0.03) and rectus femoris (p = 0.03) produced greater backward acceleration of the mass center and the gastrocnemius (p = 0.02) and soleus (p = 0.047) produced greater forward acceleration of the mass center during crouch gait than unimpaired gait (Fig. 3A). The forward acceleration produced by the soleus increased with crouch severity (p = 0.001). In contrast, the fore-aft accelerations produced by the gluteus medius (p < 0.001) and iliopsoas (p = 0.02) were significantly smaller in crouch gait than in unimpaired gait.
Figure 3.
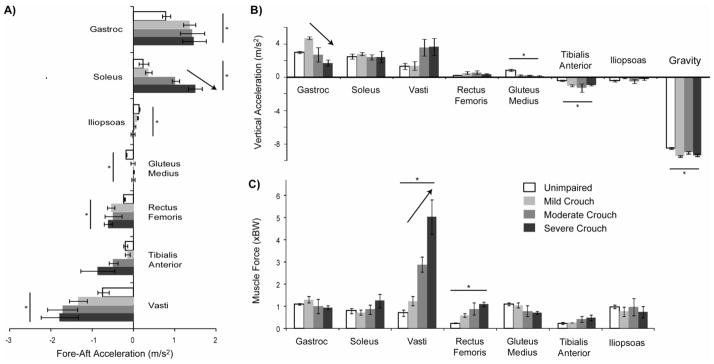
The average (A) fore-aft and (B) vertical accelerations of the mass center during stance produced by each muscle and (C) the average muscle force during stance normalized by body weight (BW). Error bars are ± 1 standard error. A ‘*’ indicates a significant difference (p < 0.05) in the student’s t-test comparing unimpaired gait and crouch gait. An arrow indicates a significant change with crouch severity (p < 0.05) from a one-way ANOVA comparing mild, moderate, and severe crouch gait. In (B), ‘Gravity’ indicates the acceleration of the mass center when only gravity is applied to the mass center. The vertical acceleration provided by skeletal alignment is equal to 9.81 m/s2 minus ‘Gravity.’
When only gravity was applied to the model, the downward acceleration of the mass center was greater (p = 0.001) in crouch gait than in unimpaired gait (Fig. 3B). In the erect posture of unimpaired gait, skeletal alignment supports a larger portion of body weight and, thus, the downward acceleration of the mass center when only gravity was applied was smaller than in a crouched posture. The contribution of the tibialis anterior to downward acceleration was greater during crouch gait than unimpaired gait; however, muscle contributions to downward acceleration of the mass center were small relative to gravity. The vasti, gastrocnemius, and soleus produced the largest upward accelerations of the mass center during crouch gait and unimpaired gait. The upward acceleration produced by the gluteus medius was significantly less during crouch gait than unimpaired gait (p < 0.001). The upward acceleration produced by the gastrocnemius decreased with crouch severity (p = 0.02).
The forces produced by the vasti (p = 0.04) and rectus femoris (p = 0.01) were significantly greater in crouch gait than in unimpaired gait, and vasti force increased (p = 0.005) with crouch severity (Fig. 3C). The change in vasti force was larger than the change in upward and backward accelerations produced by the vasti because of a diminished capacity of the vasti to accelerate the mass center in a crouch posture (Supp. Fig. 2). For example, vasti force during severe crouch gait increased by 340% compared to unimpaired gait, but the upward acceleration produced by the vasti only increased by 184% because the upward acceleration produced per newton of muscle force of the vasti decreased by 54% in severe crouch gait.
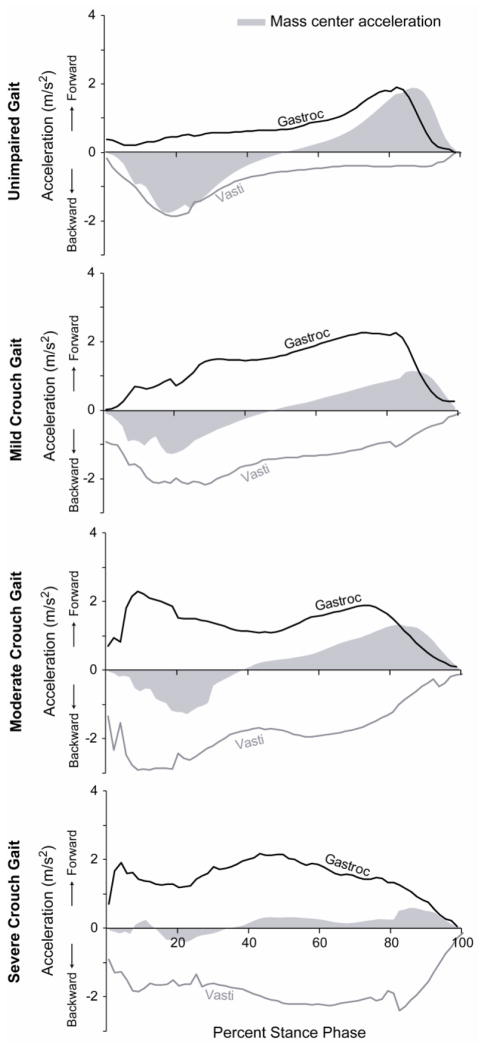
The relative timing and magnitude of the fore-aft accelerations generated by the vasti and gastrocnemius differed between crouch gait and unimpaired gait, as indicated by changes in the covariance and area under the fore-aft acceleration curves (Fig. 4). In unimpaired gait, the vasti accelerated the mass center upward and backward during early stance and the ankle plantarflexors accelerated the mass center upward and forward during late stance. The covariance of the fore-aft accelerations generated by the vasti and gastrocnemius during unimpaired gait was close to zero (−0.08 ± 0.03), indicating that these muscles were active at different times of the gait cycle. During crouch gait, the vasti and ankle plantarflexors both contributed to fore-aft accelerations throughout stance, resulting in large, opposing fore-aft accelerations. The covariance of the fore-aft accelerations generated by the vasti and gastrocnemius was −0.71 ± 0.14 during crouch gait, significantly different than unimpaired gait (p = 0.03), indicating that these muscles were active at the same time and accelerated the mass center in opposite directions. Additionally, the areas under the fore-aft acceleration curves of the vasti (p = 0.01) and gastrocnemius (p = 0.02) were significantly greater in crouch gait than in unimpaired gait due to these muscles’ sustained contributions to fore-aft accelerations during crouch gait.
Figure 4.
The fore-aft accelerations of the mass center produced by the vasti (gray line) and gastrocnemius (black line) during stance. The gray area shows the experimentally measured acceleration of the mass center (fore-aft ground reaction force normalized by body mass).
Discussion
As in unimpaired gait, two muscle groups, the quadriceps and ankle plantarflexors, were largely responsible for accelerating the mass center upward and modulating fore-aft acceleration during crouch gait. However, unlike unimpaired gait, these muscles contributed to mass center accelerations throughout stance and produced large, opposing fore-aft accelerations. During crouch gait, the ankle plantarflexors accelerated the mass center upward and forward while the vasti accelerated the mass center upward and backward throughout stance. In unimpaired gait, individuals modulate speed by increasing or decreasing quadriceps and ankle plantarflexor muscle activation proportionally across the gait cycle [7]. The sustained and opposing fore-aft accelerations generated by the quadriceps and ankle plantarflexors during crouch gait limit the effectiveness of this mechanism to modulate speed.
A crouched posture increases the demand on muscles to accelerate the mass center upward due to a decrease in skeletal support and an increase in the knee extensor moment. The quadriceps and ankle plantarflexors produce the greatest upward acceleration of the mass center during crouch gait, but the gluteus medius’ ability to accelerate the mass center upward is significantly reduced compared to unimpaired gait [12]. During unimpaired gait, the gluteus medius accelerates the mass center upward in mid-stance during the transition from quadriceps to ankle plantarflexors activity. During crouch gait, the gluteus medius contribution to upward acceleration is reduced and a different strategy is needed to support the mass center in mid-stance. Furthermore, the upward acceleration produced per newton of muscle force decreases with crouch severity for both the quadriceps and ankle plantarflexors [11] and other muscles may need to be recruited to accelerate the mass center upward. Previous experimental studies have also reported increased quadriceps demand [25,26] and prolonged ankle plantarflexor activity during crouch gait [27].
Understanding how muscles contribute to vertical and fore-aft accelerations can assist in evaluating treatment options. Demand on the vasti and rectus femoris was greater during crouch gait and may contribute to fatigue. Strength training or other programs that improve the endurance of these muscles may improve the overall endurance of individuals with crouch gait. The ankle plantarflexors were also critical muscles for accelerating the mass center upward and forward during crouch gait and may be targets for strength training programs. Engsberg et al. (2006) reported an improvement in knee flexion during stance after strengthening the ankle plantarflexors [28]. Similarly, ankle foot orthoses may assist the ankle plantarflexors and improve the efficiency of crouch gait [29,30]. From a surgical perspective, weakening or reducing the force-generating capacity of the gastrocnemius or soleus by musculotendon lengthening, neuromuscular toxins, or other procedures could reduce an individual’s ability to accelerate their mass center upward or forward.
Simulation enables analysis of the accelerations produced by muscles; however, there are limitations to these methods. The models used in this study were scaled from adult models and may not reflect bone deformities or altered muscle physiology of individuals with cerebral palsy. We excluded subjects with significant bone deformities. However, measurements of how muscle physiology changes in individuals with cerebral palsy are needed to incorporate the effects of contracture, spasticity, and other pathologies. These changes in muscle physiology would not affect the direction of the vertical or fore-aft accelerations produced by each muscle, but could affect the relative magnitude of force produced by different muscles. Estimated muscle activations were compared to EMG; however, EMG data was available from a few muscles in a subset of the subjects and only from one of the subjects in the moderate crouch gait group. Although the estimated activations were similar to EMG during stance, when muscles predominately contribute to mass center accelerations, the EMG data was noisy and varied between subjects. Furthermore, to estimate muscle activations we minimized the sum of squared muscle activations, but recognize that other cost-functions may be more appropriate for simulating pathologic gait. EMG from more muscle groups and analysis of other cost-functions could improve future simulations. This study also had a limited number of subjects which may not be representative of the larger population and may not have provided the power to detect some differences in muscle contributions to mass-center accelerations. We have made the simulations included in this study freely available on-line for other researchers to evaluate and use for future research (https://simtk.org/home/crouchgait).
By creating musculoskeletal simulations of individuals with cerebral palsy and crouch gait, we have determined that a different strategy is used to accelerate the mass center during crouch gait. The quadriceps and ankle plantarflexors are the primary muscle groups that accelerate the mass center upward; however, sustained activity of these muscles during stance produces large, opposing fore-aft accelerations – like driving with your parking brake on. Future studies that examine the metabolic costs of crouch gait and explore why individuals adopt a crouch gait pattern will be critical for improving treatment and quality of life. Examining how individual muscles contribute to gait using musculoskeletal simulation provides a foundation for these future studies and a basis for clinicians and researchers to understand the underlying mechanics of gait pathologies.
Supplementary Material
Research Highligts.
We created simulations of crouch gait in individuals with cerebral palsy.
In crouch gait, the ankle plantarflexors and vasti accelerate the mass center up.
The ankle plantarflexors and vasti produce large, opposing fore-aft accelerations.
Acknowledgments
The authors thank the staff of the James R. Gage Center for Gait and Motion Analysis at Gillette Children’s Specialty Healthcare. This work was funded by NIH R01 HD033929, R-24 HD065690, U54GM072970, and an NSF Graduate Research Fellowship.
Footnotes
Conflict of Interest Statement
None of the authors had financial or personal conflict of interest with regard to this study. Original Article
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Wren TA, Rethlefsen S, Kay RM. Prevalence of specific gait abnormalities in children with cerebral palsy: influence of cerebral palsy subtype, age, and previous surgery. J Pediatr Orthop. 2005;25:79–83. doi: 10.1097/00004694-200501000-00018. [DOI] [PubMed] [Google Scholar]
- 2.Jahnsen R, Villien L, Aamodt G, Stanghelle JK, Holm I. Musculoskeletal pain in adults with cerebral palsy compared with the general population. J Rehabil Med. 2004;36:78–84. doi: 10.1080/16501970310018305. [DOI] [PubMed] [Google Scholar]
- 3.Graham H, Selber P. Musculoskeletal aspects of cerebral palsy. J Bone Joint Surg Br. 2003;85:157–66. doi: 10.1302/0301-620x.85b2.14066. [DOI] [PubMed] [Google Scholar]
- 4.Rose J, Gamble JG, Medeiros J, Burgos A, Haskell WL. Energy cost of walking in normal children and in those with cerebral palsy: comparison of heart rate and oxygen uptake. J Pediatr Orthop. 1989;9:276–9. [PubMed] [Google Scholar]
- 5.Waters RL, Mulroy S. The energy expenditure of normal and pathologic gait. Gait Posture. 1999;9:207–31. doi: 10.1016/s0966-6362(99)00009-0. [DOI] [PubMed] [Google Scholar]
- 6.Anderson FC, Pandy MG. Individual muscle contributions to support in normal walking. Gait Posture. 2003;17:159–69. doi: 10.1016/s0966-6362(02)00073-5. [DOI] [PubMed] [Google Scholar]
- 7.Liu MQ, Anderson FC, Schwartz MH, Delp SL. Muscle contributions to support and progression over a range of walking speeds. J Biomech. 2008;41:3243–52. doi: 10.1016/j.jbiomech.2008.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Neptune RR, Kautz SA, Zajac FE. Contributions of the individual ankle plantar flexors to support, forward progression and swing initiation during walking. J Biomech. 2001;34:1387–98. doi: 10.1016/s0021-9290(01)00105-1. [DOI] [PubMed] [Google Scholar]
- 9.Liu MQ, Anderson FC, Pandy MG, Delp SL. Muscles that support the body also modulate forward progression during walking. J Biomech. 2006;39:2623–30. doi: 10.1016/j.jbiomech.2005.08.017. [DOI] [PubMed] [Google Scholar]
- 10.Steele KM, Seth A, Hicks JL, Schwartz MS, Delp SL. Muscle contributions to support and progression during single-limb stance in crouch gait. J Biomech. 2010;43:2099–105. doi: 10.1016/j.jbiomech.2010.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hicks JL, Schwartz MH, Arnold AS, Delp SL. Crouched postures reduce the capacity of muscles to extend the hip and knee during the single-limb stance phase of gait. J Biomech. 2008;41:960–7. doi: 10.1016/j.jbiomech.2008.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Correa TA, Schache AG, Graham HK, Baker R, Thomason P, Pandy MG. Potential of lower-limb muscles to accelerate the body during cerebral palsy gait. Gait Posture. 2012;36:194–200. doi: 10.1016/j.gaitpost.2012.02.014. [DOI] [PubMed] [Google Scholar]
- 13.Hicks J, Arnold A, Anderson F, Schwartz M, Delp S. The effect of excessive tibial torsion on the capacity of muscles to extend the hip and knee during single-limb stance. Gait Posture. 2007;26:546–52. doi: 10.1016/j.gaitpost.2006.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Davis R, Ounpuu S, Tyburski D, Gage J. A gait analysis data collection and reduction technique. Hum Mov Sci. 1991;10:575–87. [Google Scholar]
- 15.Delp SL, Loan JP, Hoy MG, Zajac FE, Topp EL, Rosen JM. An interactive graphics-based model of the lower extremity to study orthopaedic surgical procedures. IEEE Trans Biomed Eng. 1990;37:757–67. doi: 10.1109/10.102791. [DOI] [PubMed] [Google Scholar]
- 16.Anderson FC, Pandy MG. A Dynamic Optimization Solution for Vertical Jumping in Three Dimensions. Comput Methods Biomech Biomed Engin. 1999;2:201–31. doi: 10.1080/10255849908907988. [DOI] [PubMed] [Google Scholar]
- 17.Reinbolt J, Fox M, Arnold A, Delp SL. Importance of Preswing Rectus Femoris Activity in Stiff-Knee Gait. Journal of Biomechanics. 2008;41:2362–9. doi: 10.1016/j.jbiomech.2008.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Delp SL, Anderson FC, Arnold AS, Loan P, Habib A, John CT, Guendelman E, Thelen DG. OpenSim: open-source software to create and analyze dynamic simulations of movement. IEEE Trans Biomed Eng. 2007;54:1940–50. doi: 10.1109/TBME.2007.901024. [DOI] [PubMed] [Google Scholar]
- 19.Jaric S, Ugarkovic D, Kukolj M. Evaluation of methods for normalizing muscle strength in elite and young athletes. J Sports Med Phys Fitness. 2002;42:141–51. [PubMed] [Google Scholar]
- 20.Thelen DG, Anderson FC. Using computed muscle control to generate forward dynamic simulations of human walking from experimental data. J Biomech. 2006;39:1107–15. doi: 10.1016/j.jbiomech.2005.02.010. [DOI] [PubMed] [Google Scholar]
- 21.Thelen DG, Anderson FC, Delp SL. Generating dynamic simulations of movement using computed muscle control. J Biomech. 2003;36:321–8. doi: 10.1016/s0021-9290(02)00432-3. [DOI] [PubMed] [Google Scholar]
- 22.Hamner SR, Seth A, Delp SL. Muscle contributions to propulsion and support during running. J Biomech. 2010;43:2709–16. doi: 10.1016/j.jbiomech.2010.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zajac FE. Muscle and tendon: properties, models, scaling, and application to biomechanics and motor control. Crit Rev Biomed Eng. 1989;17:359–411. [PubMed] [Google Scholar]
- 24.Riley PO, Kerrigan DC. Kinetics of stiff-legged gait: induced acceleration analysis. IEEE Trans Rehabil Eng. 1999;7:420–6. doi: 10.1109/86.808945. [DOI] [PubMed] [Google Scholar]
- 25.Hsu AT, Perry J, Gronley JK, Hislop HJ. Quadriceps force and myoelectric activity during flexed knee stance. Clin Orthop Relat Res. 1993:254–62. [PubMed] [Google Scholar]
- 26.Perry J, Antonelli D, Ford W. Analysis of knee-joint forces during flexed-knee stance. J Bone Joint Surg Am. 1975;57:961–7. [PubMed] [Google Scholar]
- 27.Thomas SS, Moore C, Kelp-Lenane C, Norris C. Simulated gait patterns: the resulting effects on gait parameters, dynamic electromyography, joint moments, and physiological cost index. Gait Posture. 1996;4:100–7. [Google Scholar]
- 28.Engsberg JR, Ross SA, Collins DR. Increasing ankle strength to improve gait and function in children with cerebral palsy: a pilot study. Pediatr Phys Ther. 2006;18:266–75. doi: 10.1097/01.pep.0000233023.33383.2b. [DOI] [PubMed] [Google Scholar]
- 29.Lucareli PR, de Lima OM, Lucarelli JG, Lima FP. Changes in joint kinematics in children with cerebral palsy while walking with and without a floor reaction ankle-foot orthosis. Clinics (Sao Paulo) 2007;62:63–8. doi: 10.1590/s1807-59322007000100010. [DOI] [PubMed] [Google Scholar]
- 30.Rogozinski BM, Davids JR, Davis RB, 3rd, Jameson GG, Blackhurst DW. The efficacy of the floor-reaction ankle-foot orthosis in children with cerebral palsy. J Bone Joint Surg Am. 2009;91:2440–7. doi: 10.2106/JBJS.H.00965. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.