Abstract
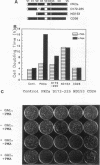
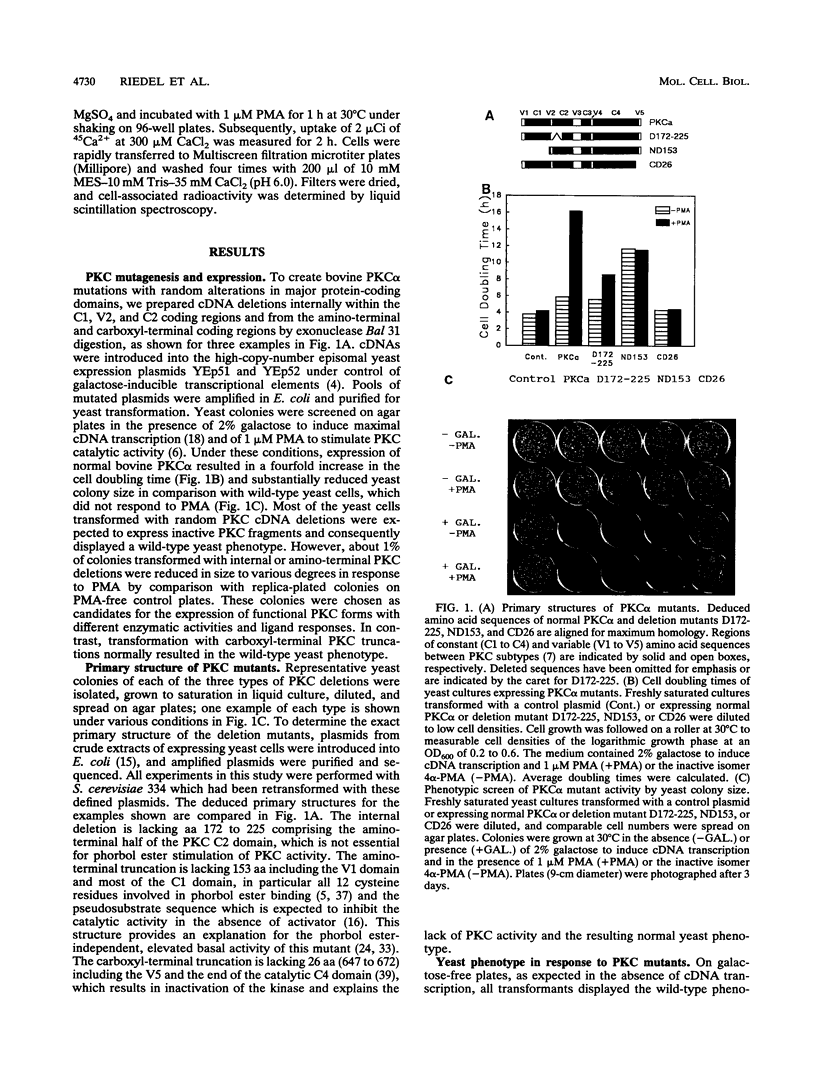
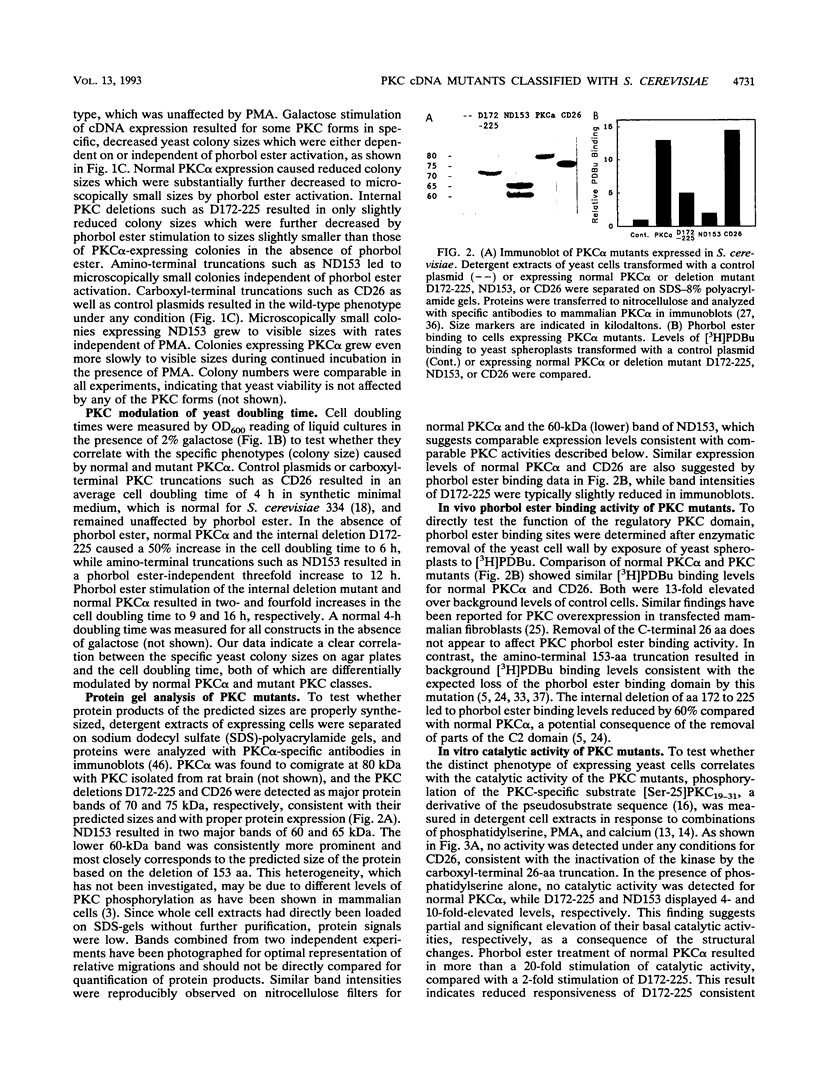
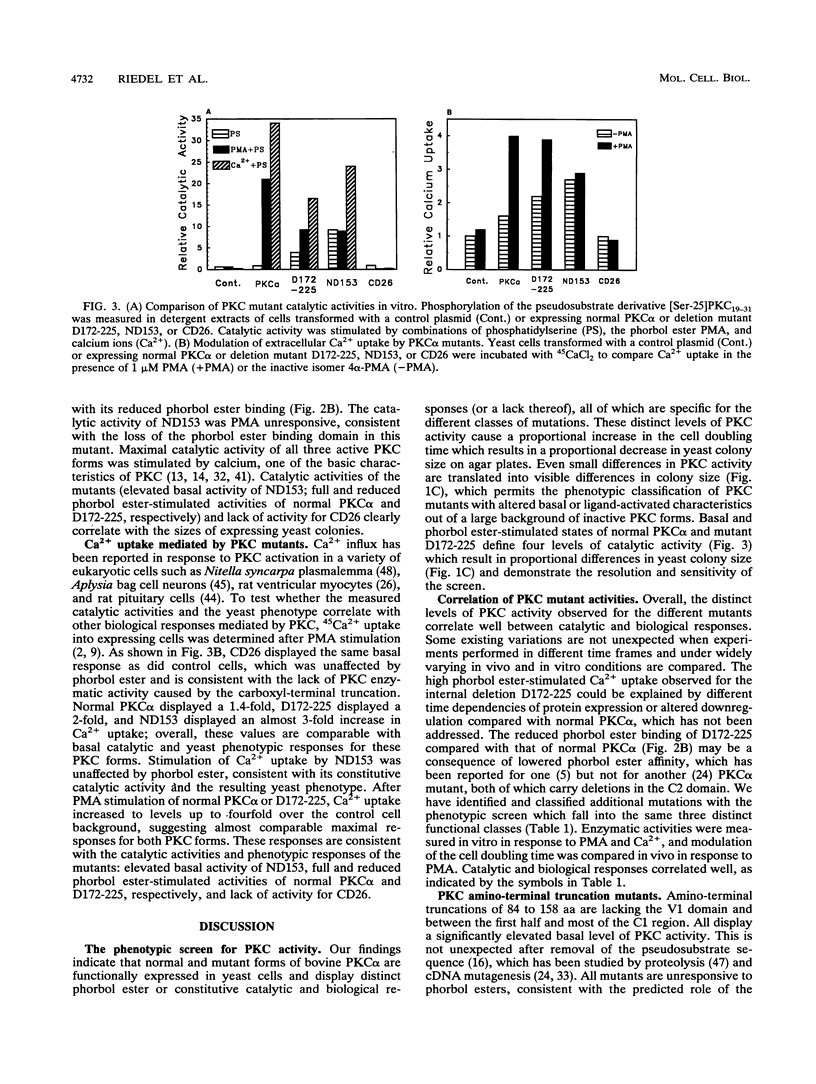
The phorbol ester receptor protein kinase C (PKC) gene family encodes essential mediators of eukaryotic cellular signals. Molecular dissection of their mechanisms of action has been limited in part by the lack of random mutagenesis approaches and by the complexity of signaling pathways in mammalian cells which involve multiple PKC isoforms. Here we present a rapid screen which permits the quantification of mammalian PKC activity phenotypically in the yeast Saccharomyces cerevisiae. Bovine PKC alpha cDNA is functionally expressed in S. cerevisiae. This results in a phorbol ester response: a fourfold increase in the cell doubling time and a substantial decrease in yeast colony size on agar plates. We have expressed pools of bovine PKC alpha cDNAs mutagenized by Bal 31 deletion of internal, amino-terminal, or carboxyl-terminal sequences and have identified three classes of mutants on the basis of their distinct yeast phenotypes. Representatives of each class were analyzed. An internal deletion of amino acids (aa) 172 to 225 displayed ligand-dependent but reduced catalytic activity, an amino-terminal truncation of aa 1 to 153 displayed elevated and ligand-independent activity, and a carboxyl-terminal 26-aa truncation (aa 647 to 672) lacked activity under any conditions. Additional mutations confirmed the distinct functional characteristics of these classes. Our data show that deletion of the V1 and C1 regions results in elevated basal catalytic activity which is still Ca2+ responsive. Internal deletions in the V2 and C2 regions do not abolish phorbol ester or Ca2+ regulation of PKC activity, suggesting that most of the C2 domain is not essential for phorbol ester stimulation and most of the regulatory domain is dispensable for Ca2+ regulation of PKC activity. These distinct activities od the PKC mutants correlate with a specific and proportional yeast phenotype and are quantified on agar plates by yeast colony size. This provides a phenotypic screen which is suitable to identity rare, randomly altered but active mammalian PKC mutants. It quantifies their catalytic and biological activities in response to PKC activators or inhibitors for a systematic mapping of PKC structure and function or PKC-drug interaction.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beggs J. D. Transformation of yeast by a replicating hybrid plasmid. Nature. 1978 Sep 14;275(5676):104–109. doi: 10.1038/275104a0. [DOI] [PubMed] [Google Scholar]
- Borbolla M., Peña A. Some characteristics of Ca2+ uptake by yeast cells. J Membr Biol. 1980 May 23;54(2):149–156. doi: 10.1007/BF01940568. [DOI] [PubMed] [Google Scholar]
- Borner C., Filipuzzi I., Wartmann M., Eppenberger U., Fabbro D. Biosynthesis and posttranslational modifications of protein kinase C in human breast cancer cells. J Biol Chem. 1989 Aug 15;264(23):13902–13909. [PubMed] [Google Scholar]
- Burns D. J., Bell R. M. Protein kinase C contains two phorbol ester binding domains. J Biol Chem. 1991 Sep 25;266(27):18330–18338. [PubMed] [Google Scholar]
- Castagna M., Takai Y., Kaibuchi K., Sano K., Kikkawa U., Nishizuka Y. Direct activation of calcium-activated, phospholipid-dependent protein kinase by tumor-promoting phorbol esters. J Biol Chem. 1982 Jul 10;257(13):7847–7851. [PubMed] [Google Scholar]
- Cigan A. M., Donahue T. F. Sequence and structural features associated with translational initiator regions in yeast--a review. Gene. 1987;59(1):1–18. doi: 10.1016/0378-1119(87)90261-7. [DOI] [PubMed] [Google Scholar]
- Coussens L., Parker P. J., Rhee L., Yang-Feng T. L., Chen E., Waterfield M. D., Francke U., Ullrich A. Multiple, distinct forms of bovine and human protein kinase C suggest diversity in cellular signaling pathways. Science. 1986 Aug 22;233(4766):859–866. doi: 10.1126/science.3755548. [DOI] [PubMed] [Google Scholar]
- Eilam Y., Othman M., Halachmi D. Transient increase in Ca2+ influx in Saccharomyces cerevisiae in response to glucose: effects of intracellular acidification and cAMP levels. J Gen Microbiol. 1990 Dec;136(12):2537–2543. doi: 10.1099/00221287-136-12-2537. [DOI] [PubMed] [Google Scholar]
- Fields F. O., Thorner J. Genetic suppression analysis of the function of a protein kinase C (PKC1 gene product) in Saccharomyces cerevisiae cell cycle progression: the SKCd mutations. Cold Spring Harb Symp Quant Biol. 1991;56:51–60. doi: 10.1101/sqb.1991.056.01.008. [DOI] [PubMed] [Google Scholar]
- Flint A. J., Paladini R. D., Koshland D. E., Jr Autophosphorylation of protein kinase C at three separated regions of its primary sequence. Science. 1990 Jul 27;249(4967):408–411. doi: 10.1126/science.2377895. [DOI] [PubMed] [Google Scholar]
- Gescher A. Antiproliferative properties of phorbol ester tumour promoters. Biochem Pharmacol. 1985 Aug 1;34(15):2587–2592. doi: 10.1016/0006-2952(85)90552-0. [DOI] [PubMed] [Google Scholar]
- Hannun Y. A., Bell R. M. Rat brain protein kinase C. Kinetic analysis of substrate dependence, allosteric regulation, and autophosphorylation. J Biol Chem. 1990 Feb 15;265(5):2962–2972. [PubMed] [Google Scholar]
- Hannun Y. A., Loomis C. R., Bell R. M. Activation of protein kinase C by Triton X-100 mixed micelles containing diacylglycerol and phosphatidylserine. J Biol Chem. 1985 Aug 25;260(18):10039–10043. [PubMed] [Google Scholar]
- Hoffman C. S., Winston F. A ten-minute DNA preparation from yeast efficiently releases autonomous plasmids for transformation of Escherichia coli. Gene. 1987;57(2-3):267–272. doi: 10.1016/0378-1119(87)90131-4. [DOI] [PubMed] [Google Scholar]
- House C., Kemp B. E. Protein kinase C contains a pseudosubstrate prototope in its regulatory domain. Science. 1987 Dec 18;238(4834):1726–1728. doi: 10.1126/science.3686012. [DOI] [PubMed] [Google Scholar]
- Houslay M. D. 'Crosstalk': a pivotal role for protein kinase C in modulating relationships between signal transduction pathways. Eur J Biochem. 1991 Jan 1;195(1):9–27. doi: 10.1111/j.1432-1033.1991.tb15671.x. [DOI] [PubMed] [Google Scholar]
- Hovland P., Flick J., Johnston M., Sclafani R. A. Galactose as a gratuitous inducer of GAL gene expression in yeasts growing on glucose. Gene. 1989 Nov 15;83(1):57–64. doi: 10.1016/0378-1119(89)90403-4. [DOI] [PubMed] [Google Scholar]
- Hu K. Q., Backer J. M., Sahagian G., Feener E. P., King G. L. Modulation of the insulin growth factor II/mannose 6-phosphate receptor in microvascular endothelial cells by phorbol ester via protein kinase C. J Biol Chem. 1990 Aug 15;265(23):13864–13870. [PubMed] [Google Scholar]
- Huang K. P. Role of protein kinase C in cellular regulation. Biofactors. 1990 Jul;2(3):171–178. [PubMed] [Google Scholar]
- Irie K., Takase M., Lee K. S., Levin D. E., Araki H., Matsumoto K., Oshima Y. MKK1 and MKK2, which encode Saccharomyces cerevisiae mitogen-activated protein kinase-kinase homologs, function in the pathway mediated by protein kinase C. Mol Cell Biol. 1993 May;13(5):3076–3083. doi: 10.1128/mcb.13.5.3076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito H., Fukuda Y., Murata K., Kimura A. Transformation of intact yeast cells treated with alkali cations. J Bacteriol. 1983 Jan;153(1):163–168. doi: 10.1128/jb.153.1.163-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwai T., Fujisawa N., Ogita K., Kikkawa U. Catalytic properties of yeast protein kinase C: difference between the yeast and mammalian enzymes. J Biochem. 1992 Jul;112(1):7–10. doi: 10.1093/oxfordjournals.jbchem.a123868. [DOI] [PubMed] [Google Scholar]
- Kaibuchi K., Fukumoto Y., Oku N., Takai Y., Arai K., Muramatsu M. Molecular genetic analysis of the regulatory and catalytic domains of protein kinase C. J Biol Chem. 1989 Aug 15;264(23):13489–13496. [PubMed] [Google Scholar]
- Knopf J. L., Lee M. H., Sultzman L. A., Kriz R. W., Loomis C. R., Hewick R. M., Bell R. M. Cloning and expression of multiple protein kinase C cDNAs. Cell. 1986 Aug 15;46(4):491–502. doi: 10.1016/0092-8674(86)90874-3. [DOI] [PubMed] [Google Scholar]
- Lacerda A. E., Rampe D., Brown A. M. Effects of protein kinase C activators on cardiac Ca2+ channels. Nature. 1988 Sep 15;335(6187):249–251. doi: 10.1038/335249a0. [DOI] [PubMed] [Google Scholar]
- Leach K. L., Powers E. A., McGuire J. C., Dong L., Kiley S. C., Jaken S. Monoclonal antibodies specific for type 3 protein kinase C recognize distinct domains of protein kinase C and inhibit in vitro functional activity. J Biol Chem. 1988 Sep 15;263(26):13223–13230. [PubMed] [Google Scholar]
- Lee K. S., Irie K., Gotoh Y., Watanabe Y., Araki H., Nishida E., Matsumoto K., Levin D. E. A yeast mitogen-activated protein kinase homolog (Mpk1p) mediates signalling by protein kinase C. Mol Cell Biol. 1993 May;13(5):3067–3075. doi: 10.1128/mcb.13.5.3067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K. S., Levin D. E. Dominant mutations in a gene encoding a putative protein kinase (BCK1) bypass the requirement for a Saccharomyces cerevisiae protein kinase C homolog. Mol Cell Biol. 1992 Jan;12(1):172–182. doi: 10.1128/mcb.12.1.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin D. E., Bartlett-Heubusch E. Mutants in the S. cerevisiae PKC1 gene display a cell cycle-specific osmotic stability defect. J Cell Biol. 1992 Mar;116(5):1221–1229. doi: 10.1083/jcb.116.5.1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin D. E., Fields F. O., Kunisawa R., Bishop J. M., Thorner J. A candidate protein kinase C gene, PKC1, is required for the S. cerevisiae cell cycle. Cell. 1990 Jul 27;62(2):213–224. doi: 10.1016/0092-8674(90)90360-q. [DOI] [PubMed] [Google Scholar]
- Luo J. H., Kahn S., O'Driscoll K., Weinstein I. B. The regulatory domain of protein kinase C beta 1 contains phosphatidylserine- and phorbol ester-dependent calcium binding activity. J Biol Chem. 1993 Feb 15;268(5):3715–3719. [PubMed] [Google Scholar]
- Muramatsu M., Kaibuchi K., Arai K. A protein kinase C cDNA without the regulatory domain is active after transfection in vivo in the absence of phorbol ester. Mol Cell Biol. 1989 Feb;9(2):831–836. doi: 10.1128/mcb.9.2.831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishizuka Y. The molecular heterogeneity of protein kinase C and its implications for cellular regulation. Nature. 1988 Aug 25;334(6184):661–665. doi: 10.1038/334661a0. [DOI] [PubMed] [Google Scholar]
- Ogita K., Miyamoto S., Koide H., Iwai T., Oka M., Ando K., Kishimoto A., Ikeda K., Fukami Y., Nishizuka Y. Protein kinase C in Saccharomyces cerevisiae: comparison with the mammalian enzyme. Proc Natl Acad Sci U S A. 1990 Jul;87(13):5011–5015. doi: 10.1073/pnas.87.13.5011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliver F. J., de la Rubia G., Feener E. P., Lee M. E., Loeken M. R., Shiba T., Quertermous T., King G. L. Stimulation of endothelin-1 gene expression by insulin in endothelial cells. J Biol Chem. 1991 Dec 5;266(34):23251–23256. [PubMed] [Google Scholar]
- Ono Y., Fujii T., Igarashi K., Kuno T., Tanaka C., Kikkawa U., Nishizuka Y. Phorbol ester binding to protein kinase C requires a cysteine-rich zinc-finger-like sequence. Proc Natl Acad Sci U S A. 1989 Jul;86(13):4868–4871. doi: 10.1073/pnas.86.13.4868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paravicini G., Cooper M., Friedli L., Smith D. J., Carpentier J. L., Klig L. S., Payton M. A. The osmotic integrity of the yeast cell requires a functional PKC1 gene product. Mol Cell Biol. 1992 Nov;12(11):4896–4905. doi: 10.1128/mcb.12.11.4896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker P. J., Coussens L., Totty N., Rhee L., Young S., Chen E., Stabel S., Waterfield M. D., Ullrich A. The complete primary structure of protein kinase C--the major phorbol ester receptor. Science. 1986 Aug 22;233(4766):853–859. doi: 10.1126/science.3755547. [DOI] [PubMed] [Google Scholar]
- Portillo F., Mazón M. J. Activation of yeast plasma membrane ATPase by phorbol ester. FEBS Lett. 1985 Nov 11;192(1):95–98. doi: 10.1016/0014-5793(85)80050-8. [DOI] [PubMed] [Google Scholar]
- Riedel H., Parissenti A. M., Hansen H., Su L., Shieh H. L. Stimulation of calcium uptake in Saccharomyces cerevisiae by bovine protein kinase C alpha. J Biol Chem. 1993 Feb 15;268(5):3456–3462. [PubMed] [Google Scholar]
- Simon A. J., Milner Y., Saville S. P., Dvir A., Mochly-Rosen D., Orr E. The identification and purification of a mammalian-like protein kinase C in the yeast Saccharomyces cerevisiae. Proc Biol Sci. 1991 Feb 22;243(1307):165–171. doi: 10.1098/rspb.1991.0027. [DOI] [PubMed] [Google Scholar]
- Stojilković S. S., Chang J. P., Izumi S., Tasaka K., Catt K. J. Mechanisms of secretory responses to gonadotropin-releasing hormone and phorbol esters in cultured pituitary cells. Participation of protein kinase C and extracellular calcium mobilization. J Biol Chem. 1988 Nov 25;263(33):17301–17306. [PubMed] [Google Scholar]
- Strong J. A., Fox A. P., Tsien R. W., Kaczmarek L. K. Stimulation of protein kinase C recruits covert calcium channels in Aplysia bag cell neurons. Nature. 1987 Feb 19;325(6106):714–717. doi: 10.1038/325714a0. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young S., Rothbard J., Parker P. J. A monoclonal antibody recognising the site of limited proteolysis of protein kinase C. Inhibition of down-regulation in vivo. Eur J Biochem. 1988 Apr 5;173(1):247–252. doi: 10.1111/j.1432-1033.1988.tb13991.x. [DOI] [PubMed] [Google Scholar]
- Zherelova O. M. Protein kinase C is involved in regulation of Ca2+ channels in plasmalemma of Nitella syncarpa. FEBS Lett. 1989 Jan 2;242(2):330–332. doi: 10.1016/0014-5793(89)80495-8. [DOI] [PubMed] [Google Scholar]