Abstract
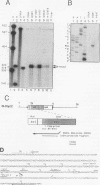
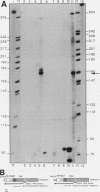
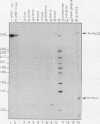
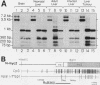
The woodchuck intronless proto-oncogene N-myc2 was initially discovered as a frequent target site for hepadnavirus integration in hepatocellular carcinoma. N-myc2 possesses characteristics of a functional retroposon derived from the woodchuck N-myc gene. We have investigated the regulatory signals governing N-myc2 expression and found that a short promoter, including a variant TATA box and potential binding sites for several transcription factors, is localized in the N-myc2 sequences homologous to the 5' untranslated region of the second N-myc exon. The corresponding region in the intron-containing woodchuck N-myc gene also exhibited promoter activity in transient transfection assays. The high evolutionary conservation of these sequences in mammalian N-myc genes suggests that they contain a cryptic N-myc promoter which may be unmasked in the particular context provided by the N-myc2 retroposon. Although N-myc2, like the woodchuck N-myc gene, contributes to an extended CpG island and was found constitutively hypomethylated, it presents a highly restricted expression pattern in adult animals. Whereas the intron-containing N-myc gene is expressed at low levels in different tissues, N-myc2 mRNA was detected only in brain tissue, raising questions about the functional significance of the maintenance of a second N-myc gene in the woodchuck genome.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akeson R., Bernards R. N-myc down regulates neural cell adhesion molecule expression in rat neuroblastoma. Mol Cell Biol. 1990 May;10(5):2012–2016. doi: 10.1128/mcb.10.5.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babiss L. E., Friedman J. M. Regulation of N-myc gene expression: use of an adenovirus vector to demonstrate posttranscriptional control. Mol Cell Biol. 1990 Dec;10(12):6700–6708. doi: 10.1128/mcb.10.12.6700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhandari B., Roesler W. J., DeLisio K. D., Klemm D. J., Ross N. S., Miller R. E. A functional promoter flanks an intronless glutamine synthetase gene. J Biol Chem. 1991 Apr 25;266(12):7784–7792. [PubMed] [Google Scholar]
- Brosius J. Retroposons--seeds of evolution. Science. 1991 Feb 15;251(4995):753–753. doi: 10.1126/science.1990437. [DOI] [PubMed] [Google Scholar]
- Corcoran L. M., Adams J. M., Dunn A. R., Cory S. Murine T lymphomas in which the cellular myc oncogene has been activated by retroviral insertion. Cell. 1984 May;37(1):113–122. doi: 10.1016/0092-8674(84)90306-4. [DOI] [PubMed] [Google Scholar]
- DePinho R. A., Hatton K. S., Tesfaye A., Yancopoulos G. D., Alt F. W. The human myc gene family: structure and activity of L-myc and an L-myc pseudogene. Genes Dev. 1987 Dec;1(10):1311–1326. doi: 10.1101/gad.1.10.1311. [DOI] [PubMed] [Google Scholar]
- DePinho R. A., Legouy E., Feldman L. B., Kohl N. E., Yancopoulos G. D., Alt F. W. Structure and expression of the murine N-myc gene. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1827–1831. doi: 10.1073/pnas.83.6.1827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DePinho R. A., Schreiber-Agus N., Alt F. W. myc family oncogenes in the development of normal and neoplastic cells. Adv Cancer Res. 1991;57:1–46. doi: 10.1016/s0065-230x(08)60994-x. [DOI] [PubMed] [Google Scholar]
- Eilers M., Schirm S., Bishop J. M. The MYC protein activates transcription of the alpha-prothymosin gene. EMBO J. 1991 Jan;10(1):133–141. doi: 10.1002/j.1460-2075.1991.tb07929.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fourel G., Tiollais P., Buendia M. A. Nucleotide sequence of the woodchuck N-myc gene (WN-myc1). Nucleic Acids Res. 1990 Aug 25;18(16):4918–4918. doi: 10.1093/nar/18.16.4918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fourel G., Trepo C., Bougueleret L., Henglein B., Ponzetto A., Tiollais P., Buendia M. A. Frequent activation of N-myc genes by hepadnavirus insertion in woodchuck liver tumours. Nature. 1990 Sep 20;347(6290):294–298. doi: 10.1038/347294a0. [DOI] [PubMed] [Google Scholar]
- Hiller S., Breit S., Wang Z. Q., Wagner E. F., Schwab M. Localization of regulatory elements controlling human MYCN expression. Oncogene. 1991 Jun;6(6):969–977. [PubMed] [Google Scholar]
- Hsu T., Möröy T., Etiemble J., Louise A., Trépo C., Tiollais P., Buendia M. A. Activation of c-myc by woodchuck hepatitis virus insertion in hepatocellular carcinoma. Cell. 1988 Nov 18;55(4):627–635. doi: 10.1016/0092-8674(88)90221-8. [DOI] [PubMed] [Google Scholar]
- Ingvarsson S., Asker C., Axelson H., Klein G., Sümegi J. Structure and expression of B-myc, a new member of the myc gene family. Mol Cell Biol. 1988 Aug;8(8):3168–3174. doi: 10.1128/mcb.8.8.3168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaddurah-Daouk R., Greene J. M., Baldwin A. S., Jr, Kingston R. E. Activation and repression of mammalian gene expression by the c-myc protein. Genes Dev. 1987 Jun;1(4):347–357. doi: 10.1101/gad.1.4.347. [DOI] [PubMed] [Google Scholar]
- Kadesch T., Berg P. Effects of the position of the simian virus 40 enhancer on expression of multiple transcription units in a single plasmid. Mol Cell Biol. 1986 Jul;6(7):2593–2601. doi: 10.1128/mcb.6.7.2593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato G. J., Barrett J., Villa-Garcia M., Dang C. V. An amino-terminal c-myc domain required for neoplastic transformation activates transcription. Mol Cell Biol. 1990 Nov;10(11):5914–5920. doi: 10.1128/mcb.10.11.5914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katoh K., Sawai S., Ueno K., Kondoh H. Complete nucleotide sequence and exon-intron boundaries of the 5' non-coding region of the mouse N-myc gene. Nucleic Acids Res. 1988 Apr 25;16(8):3589–3589. doi: 10.1093/nar/16.8.3589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kedzierski W., Porter J. C. A novel non-enzymatic procedure for removing DNA template from RNA transcription mixtures. Biotechniques. 1991 Feb;10(2):210–214. [PubMed] [Google Scholar]
- Kohl N. E., Legouy E., DePinho R. A., Nisen P. D., Smith R. K., Gee C. E., Alt F. W. Human N-myc is closely related in organization and nucleotide sequence to c-myc. Nature. 1986 Jan 2;319(6048):73–77. doi: 10.1038/319073a0. [DOI] [PubMed] [Google Scholar]
- Lenardo M., Rustgi A. K., Schievella A. R., Bernards R. Suppression of MHC class I gene expression by N-myc through enhancer inactivation. EMBO J. 1989 Nov;8(11):3351–3355. doi: 10.1002/j.1460-2075.1989.tb08497.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarrey J. R., Thomas K. Human testis-specific PGK gene lacks introns and possesses characteristics of a processed gene. Nature. 1987 Apr 2;326(6112):501–505. doi: 10.1038/326501a0. [DOI] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mount S. M. A catalogue of splice junction sequences. Nucleic Acids Res. 1982 Jan 22;10(2):459–472. doi: 10.1093/nar/10.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Möröy T., Etiemble J., Trépo C., Tiollais P., Buendia M. A. Transcription of woodchuck hepatitis virus in the chronically infected liver. EMBO J. 1985 Jun;4(6):1507–1514. doi: 10.1002/j.1460-2075.1985.tb03810.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onclercq R., Lavenu A., Cremisi C. Pleiotropic derepression of developmentally regulated cellular and viral genes by c-myc protooncogene products in undifferentiated embryonal carcinoma cells. Nucleic Acids Res. 1989 Jan 25;17(2):735–753. doi: 10.1093/nar/17.2.735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson N. G., Pomponio R. J., Mutter G. L., Morton C. C. Testis-specific expression of the human MYCL2 gene. Nucleic Acids Res. 1991 Jun 11;19(11):3129–3137. doi: 10.1093/nar/19.11.3129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson M. O., McCarrey J. R., Simon M. I. Transcriptional regulatory regions of testis-specific PGK2 defined in transgenic mice. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8437–8441. doi: 10.1073/pnas.86.21.8437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rustgi A. K., Dyson N., Bernards R. Amino-terminal domains of c-myc and N-myc proteins mediate binding to the retinoblastoma gene product. Nature. 1991 Aug 8;352(6335):541–544. doi: 10.1038/352541a0. [DOI] [PubMed] [Google Scholar]
- Smale S. T., Baltimore D. The "initiator" as a transcription control element. Cell. 1989 Apr 7;57(1):103–113. doi: 10.1016/0092-8674(89)90176-1. [DOI] [PubMed] [Google Scholar]
- Soares M. B., Schon E., Henderson A., Karathanasis S. K., Cate R., Zeitlin S., Chirgwin J., Efstratiadis A. RNA-mediated gene duplication: the rat preproinsulin I gene is a functional retroposon. Mol Cell Biol. 1985 Aug;5(8):2090–2103. doi: 10.1128/mcb.5.8.2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanton L. W., Bishop J. M. Alternative processing of RNA transcribed from NMYC. Mol Cell Biol. 1987 Dec;7(12):4266–4272. doi: 10.1128/mcb.7.12.4266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanton L. W., Schwab M., Bishop J. M. Nucleotide sequence of the human N-myc gene. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1772–1776. doi: 10.1073/pnas.83.6.1772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein J. P., Munjaal R. P., Lagace L., Lai E. C., O'Malley B. W., Means A. R. Tissue-specific expression of a chicken calmodulin pseudogene lacking intervening sequences. Proc Natl Acad Sci U S A. 1983 Nov;80(21):6485–6489. doi: 10.1073/pnas.80.21.6485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiyama A., Kume A., Nemoto K., Lee S. Y., Asami Y., Nemoto F., Nishimura S., Kuchino Y. Isolation and characterization of s-myc, a member of the rat myc gene family. Proc Natl Acad Sci U S A. 1989 Dec;86(23):9144–9148. doi: 10.1073/pnas.86.23.9144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiyama A., Miyagi Y., Shirasawa Y., Kuchino Y. Different usage of two polyadenylylation signals in transcription of the N-myc gene in rat tumor cells. Oncogene. 1991 Nov;6(11):2027–2032. [PubMed] [Google Scholar]
- Transy C., Fourel G., Robinson W. S., Tiollais P., Marion P. L., Buendia M. A. Frequent amplification of c-myc in ground squirrel liver tumors associated with past or ongoing infection with a hepadnavirus. Proc Natl Acad Sci U S A. 1992 May 1;89(9):3874–3878. doi: 10.1073/pnas.89.9.3874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanin E. F. Processed pseudogenes: characteristics and evolution. Annu Rev Genet. 1985;19:253–272. doi: 10.1146/annurev.ge.19.120185.001345. [DOI] [PubMed] [Google Scholar]
- Wei Y., Fourel G., Ponzetto A., Silvestro M., Tiollais P., Buendia M. A. Hepadnavirus integration: mechanisms of activation of the N-myc2 retrotransposon in woodchuck liver tumors. J Virol. 1992 Sep;66(9):5265–5276. doi: 10.1128/jvi.66.9.5265-5276.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu L., Morgenbesser S. D., DePinho R. A. Complex transcriptional regulation of myc family gene expression in the developing mouse brain and liver. Mol Cell Biol. 1991 Dec;11(12):6007–6015. doi: 10.1128/mcb.11.12.6007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerman K. A., Yancopoulos G. D., Collum R. G., Smith R. K., Kohl N. E., Denis K. A., Nau M. M., Witte O. N., Toran-Allerand D., Gee C. E. Differential expression of myc family genes during murine development. 1986 Feb 27-Mar 5Nature. 319(6056):780–783. doi: 10.1038/319780a0. [DOI] [PubMed] [Google Scholar]
- Zimmerman K., Legouy E., Stewart V., Depinho R., Alt F. W. Differential regulation of the N-myc gene in transfected cells and transgenic mice. Mol Cell Biol. 1990 May;10(5):2096–2103. doi: 10.1128/mcb.10.5.2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ar-Rushdi A., Nishikura K., Erikson J., Watt R., Rovera G., Croce C. M. Differential expression of the translocated and the untranslocated c-myc oncogene in Burkitt lymphoma. Science. 1983 Oct 28;222(4622):390–393. doi: 10.1126/science.6414084. [DOI] [PubMed] [Google Scholar]
- de Thé H., Chomienne C., Lanotte M., Degos L., Dejean A. The t(15;17) translocation of acute promyelocytic leukaemia fuses the retinoic acid receptor alpha gene to a novel transcribed locus. Nature. 1990 Oct 11;347(6293):558–561. doi: 10.1038/347558a0. [DOI] [PubMed] [Google Scholar]
- de Wet J. R., Wood K. V., DeLuca M., Helinski D. R., Subramani S. Firefly luciferase gene: structure and expression in mammalian cells. Mol Cell Biol. 1987 Feb;7(2):725–737. doi: 10.1128/mcb.7.2.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lohuizen M., Breuer M., Berns A. N-myc is frequently activated by proviral insertion in MuLV-induced T cell lymphomas. EMBO J. 1989 Jan;8(1):133–136. doi: 10.1002/j.1460-2075.1989.tb03357.x. [DOI] [PMC free article] [PubMed] [Google Scholar]