Abstract
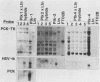
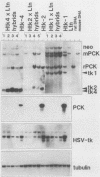
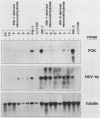
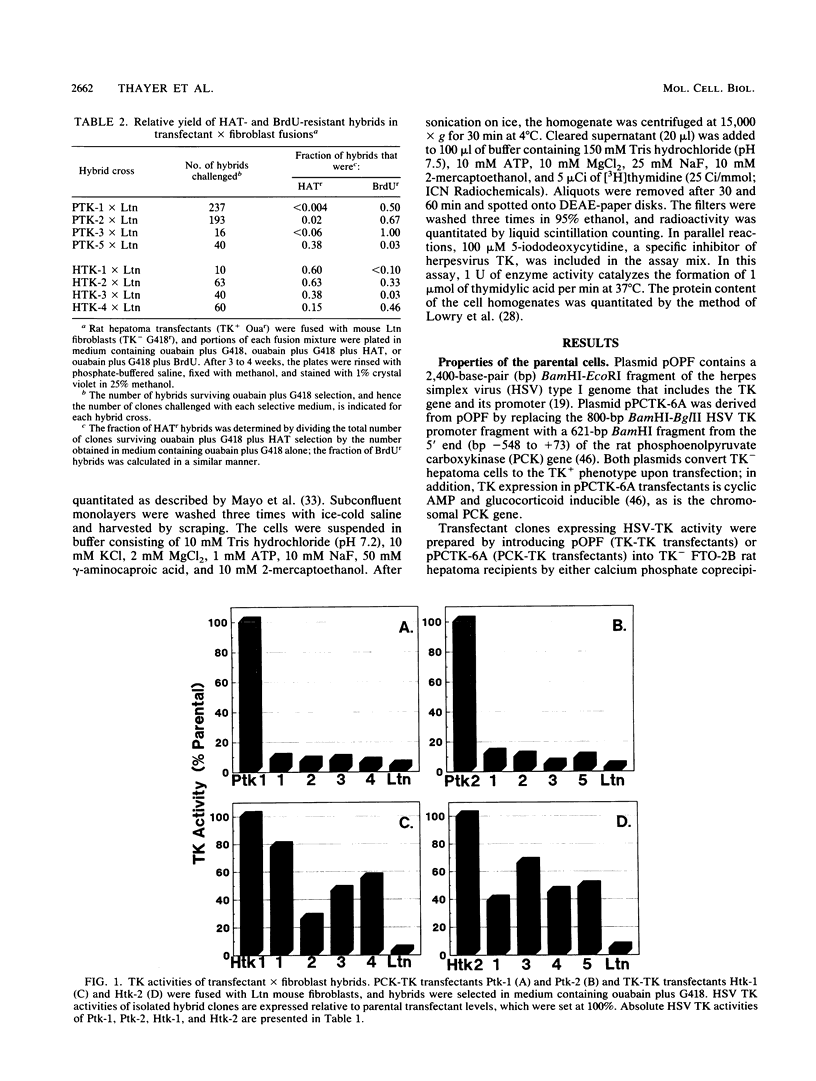
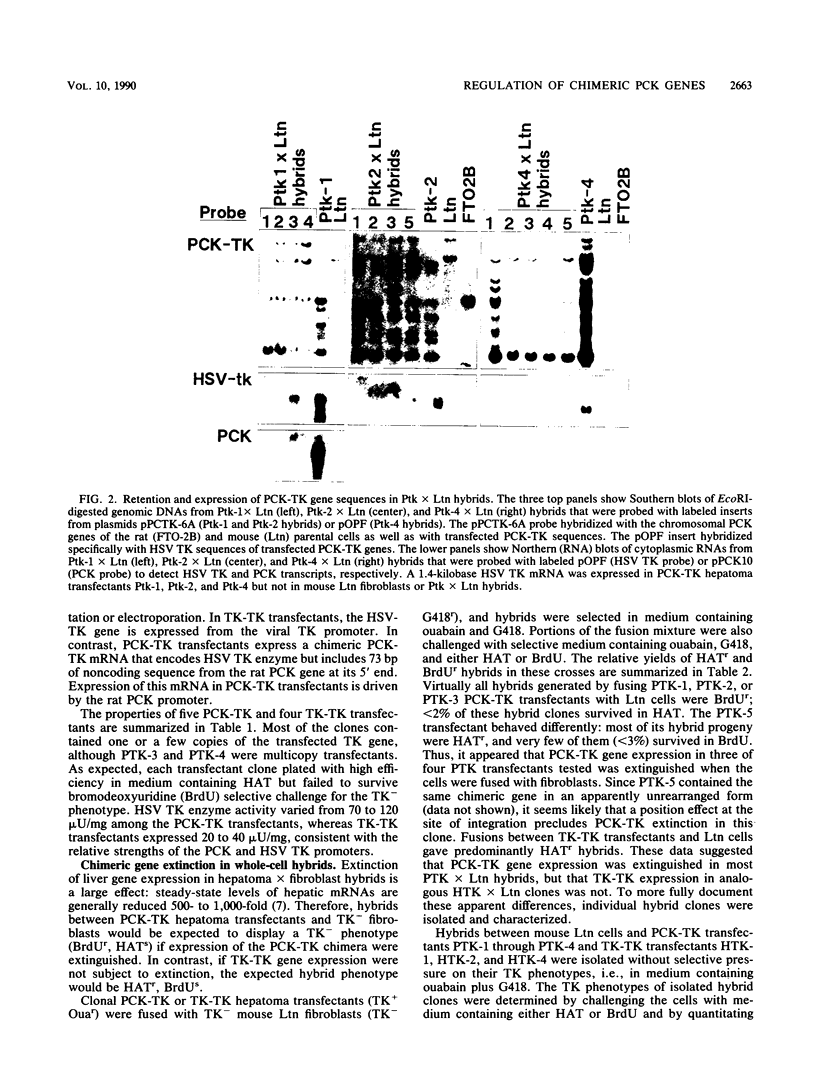
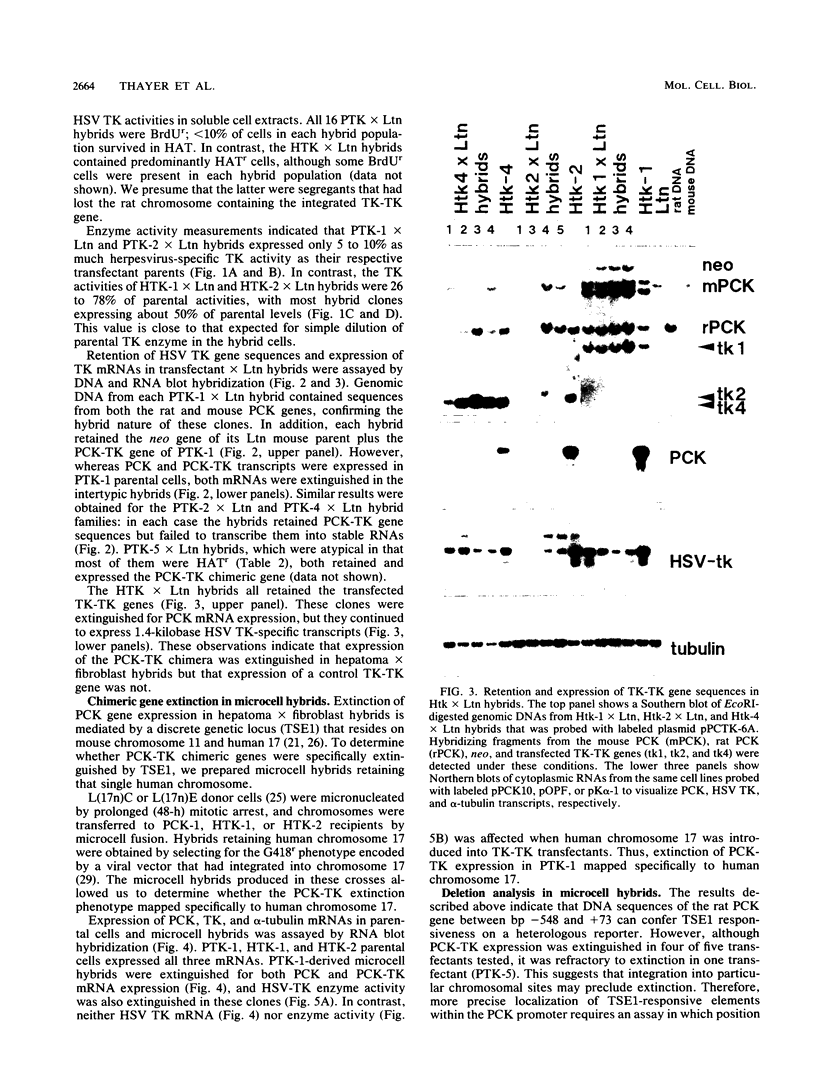
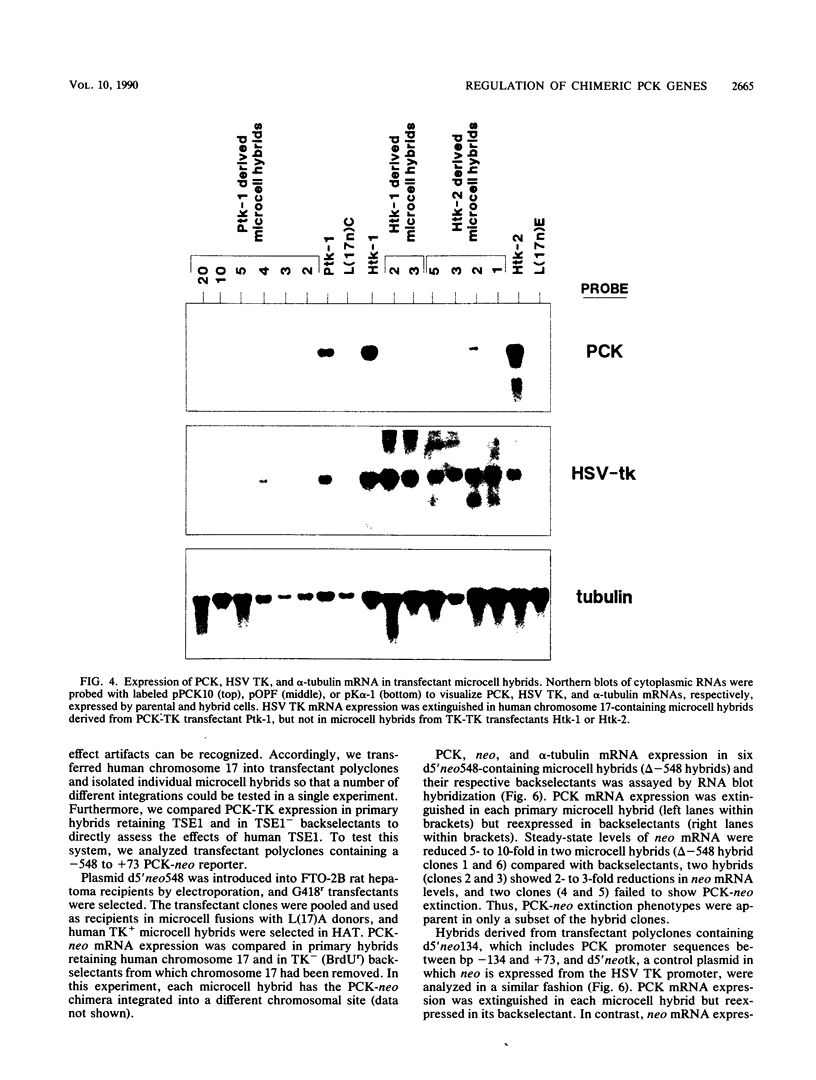
Extinction of phosphoenolpyruvate carboxykinase (PCK) gene expression in hepatoma x fibroblast hybrids is mediated by a trans-acting genetic locus designated tissue-specific extinguisher 1 (TSE1). To identify PCK gene sequences required for extinction, hepatoma transfectants expressing PCK-thymidine kinase (TK) chimeric genes were fused with TK- fibroblasts and PCK-TK expression in the resulting hybrids was monitored. Expression of a PCK-TK chimera containing PCK sequences between base pairs -548 and +73 was extinguished in four of five hepatoma transfectants tested, although hybrids derived from one transfectant clone failed to extinguish PCK-TK expression. In contrast, crosses between hepatoma transfectants expressing the herpesvirus TK gene from its own promoter and TK- fibroblasts produced TK+ hybrids; extinction of the transfected TK gene was not observed. Thus, rat PCK gene sequences between base pairs -548 and +73 are sufficient for tissue-specific extinction in hybrid cells. Extinction of PCK-TK gene expression in transfectant microcell hybrids mapped specifically to human chromosome 17, the site of human TSE1.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Banerji J., Olson L., Schaffner W. A lymphocyte-specific cellular enhancer is located downstream of the joining region in immunoglobulin heavy chain genes. Cell. 1983 Jul;33(3):729–740. doi: 10.1016/0092-8674(83)90015-6. [DOI] [PubMed] [Google Scholar]
- Baniahmad A., Muller M., Steiner C., Renkawitz R. Activity of two different silencer elements of the chicken lysozyme gene can be compensated by enhancer elements. EMBO J. 1987 Aug;6(8):2297–2303. doi: 10.1002/j.1460-2075.1987.tb02504.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blau H. M., Chiu C. P., Webster C. Cytoplasmic activation of human nuclear genes in stable heterocaryons. Cell. 1983 Apr;32(4):1171–1180. doi: 10.1016/0092-8674(83)90300-8. [DOI] [PubMed] [Google Scholar]
- Briggs M. R., Kadonaga J. T., Bell S. P., Tjian R. Purification and biochemical characterization of the promoter-specific transcription factor, Sp1. Science. 1986 Oct 3;234(4772):47–52. doi: 10.1126/science.3529394. [DOI] [PubMed] [Google Scholar]
- Cepko C. L., Roberts B. E., Mulligan R. C. Construction and applications of a highly transmissible murine retrovirus shuttle vector. Cell. 1984 Jul;37(3):1053–1062. doi: 10.1016/0092-8674(84)90440-9. [DOI] [PubMed] [Google Scholar]
- Chen T. R. In situ detection of mycoplasma contamination in cell cultures by fluorescent Hoechst 33258 stain. Exp Cell Res. 1977 Feb;104(2):255–262. doi: 10.1016/0014-4827(77)90089-1. [DOI] [PubMed] [Google Scholar]
- Chin A. C., Fournier R. E. A genetic analysis of extinction: trans-regulation of 16 liver-specific genes in hepatoma-fibroblast hybrid cells. Proc Natl Acad Sci U S A. 1987 Mar;84(6):1614–1618. doi: 10.1073/pnas.84.6.1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin A. C., Fournier R. E. Tse-2: a trans-dominant extinguisher of albumin gene expression in hepatoma hybrid cells. Mol Cell Biol. 1989 Sep;9(9):3736–3743. doi: 10.1128/mcb.9.9.3736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Church G. M., Gilbert W. Genomic sequencing. Proc Natl Acad Sci U S A. 1984 Apr;81(7):1991–1995. doi: 10.1073/pnas.81.7.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan N. J., Dobner P. R., Fuchs E. V., Cleveland D. W. Expression of human alpha-tubulin genes: interspecies conservation of 3' untranslated regions. Mol Cell Biol. 1983 Oct;3(10):1738–1745. doi: 10.1128/mcb.3.10.1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson R. L. Gene expression in somatic cell hybrids. Annu Rev Genet. 1974;8:195–218. doi: 10.1146/annurev.ge.08.120174.001211. [DOI] [PubMed] [Google Scholar]
- Favaloro J., Treisman R., Kamen R. Transcription maps of polyoma virus-specific RNA: analysis by two-dimensional nuclease S1 gel mapping. Methods Enzymol. 1980;65(1):718–749. doi: 10.1016/s0076-6879(80)65070-8. [DOI] [PubMed] [Google Scholar]
- Fougère C., Ruiz F., Ephrussi B. Gene dosage dependence of pigment synthesis in melanoma x fibroblast hybrids (hamster cells-mouse fibroblast-DOPA-oxidase-irradiation). Proc Natl Acad Sci U S A. 1972 Feb;69(2):330–334. doi: 10.1073/pnas.69.2.330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fournier R. E. A general high-efficiency procedure for production of microcell hybrids. Proc Natl Acad Sci U S A. 1981 Oct;78(10):6349–6353. doi: 10.1073/pnas.78.10.6349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillies S. D., Morrison S. L., Oi V. T., Tonegawa S. A tissue-specific transcription enhancer element is located in the major intron of a rearranged immunoglobulin heavy chain gene. Cell. 1983 Jul;33(3):717–728. doi: 10.1016/0092-8674(83)90014-4. [DOI] [PubMed] [Google Scholar]
- Goodbourn S., Burstein H., Maniatis T. The human beta-interferon gene enhancer is under negative control. Cell. 1986 May 23;45(4):601–610. doi: 10.1016/0092-8674(86)90292-8. [DOI] [PubMed] [Google Scholar]
- Gopalakrishnan T. V., Thompson E. B. A method for enucleating cultured mammalian cells. Exp Cell Res. 1975 Dec;96(2):435–439. doi: 10.1016/0014-4827(75)90282-7. [DOI] [PubMed] [Google Scholar]
- Grosveld F. G., Lund T., Murray E. J., Mellor A. L., Dahl H. H., Flavell R. A. The construction of cosmid libraries which can be used to transform eukaryotic cells. Nucleic Acids Res. 1982 Nov 11;10(21):6715–6732. doi: 10.1093/nar/10.21.6715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadonaga J. T., Tjian R. Affinity purification of sequence-specific DNA binding proteins. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5889–5893. doi: 10.1073/pnas.83.16.5889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Killary A. M., Fournier R. E. A genetic analysis of extinction: trans-dominant loci regulate expression of liver-specific traits in hepatoma hybrid cells. Cell. 1984 Sep;38(2):523–534. doi: 10.1016/0092-8674(84)90507-5. [DOI] [PubMed] [Google Scholar]
- Killary A. M., Lugo T. G., Fournier R. E. Isolation of thymidine kinase-deficient rat hepatoma cells by selection with bromodeoxyuridine, Hoechst 33258, and visible light. Biochem Genet. 1984 Apr;22(3-4):201–213. doi: 10.1007/BF00484224. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laimins L., Holmgren-König M., Khoury G. Transcriptional "silencer" element in rat repetitive sequences associated with the rat insulin 1 gene locus. Proc Natl Acad Sci U S A. 1986 May;83(10):3151–3155. doi: 10.1073/pnas.83.10.3151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamers W. H., Hanson R. W., Meisner H. M. cAMP stimulates transcription of the gene for cytosolic phosphoenolpyruvate carboxykinase in rat liver nuclei. Proc Natl Acad Sci U S A. 1982 Sep;79(17):5137–5141. doi: 10.1073/pnas.79.17.5137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leach R. J., Thayer M. J., Schafer A. J., Fournier R. E. Physical mapping of human chromosome 17 using fragment-containing microcell hybrids. Genomics. 1989 Aug;5(2):167–176. doi: 10.1016/0888-7543(89)90043-8. [DOI] [PubMed] [Google Scholar]
- Lem J., Chin A. C., Thayer M. J., Leach R. J., Fournier R. E. Coordinate regulation of two genes encoding gluconeogenic enzymes by the trans-dominant locus Tse-1. Proc Natl Acad Sci U S A. 1988 Oct;85(19):7302–7306. doi: 10.1073/pnas.85.19.7302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lem J., Fournier R. E. Assignment of the gene encoding cytosolic phosphoenolpyruvate carboxykinase (GTP) to Mus musculus chromosome 2. Somat Cell Mol Genet. 1985 Nov;11(6):633–638. doi: 10.1007/BF01534728. [DOI] [PubMed] [Google Scholar]
- Lugo T. G., Handelin B., Killary A. M., Housman D. E., Fournier R. E. Isolation of microcell hybrid clones containing retroviral vector insertions into specific human chromosomes. Mol Cell Biol. 1987 Aug;7(8):2814–2820. doi: 10.1128/mcb.7.8.2814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magnuson M. A., Quinn P. G., Granner D. K. Multihormonal regulation of phosphoenolpyruvate carboxykinase-chloramphenicol acetyltransferase fusion genes. Insulin's effects oppose those of cAMP and dexamethasone. J Biol Chem. 1987 Nov 5;262(31):14917–14920. [PubMed] [Google Scholar]
- Maniatis T., Goodbourn S., Fischer J. A. Regulation of inducible and tissue-specific gene expression. Science. 1987 Jun 5;236(4806):1237–1245. doi: 10.1126/science.3296191. [DOI] [PubMed] [Google Scholar]
- Mayo K. E., Warren R., Palmiter R. D. The mouse metallothionein-I gene is transcriptionally regulated by cadmium following transfection into human or mouse cells. Cell. 1982 May;29(1):99–108. doi: 10.1016/0092-8674(82)90094-0. [DOI] [PubMed] [Google Scholar]
- McGrane M. M., de Vente J., Yun J., Bloom J., Park E., Wynshaw-Boris A., Wagner T., Rottman F. M., Hanson R. W. Tissue-specific expression and dietary regulation of a chimeric phosphoenolpyruvate carboxykinase/bovine growth hormone gene in transgenic mice. J Biol Chem. 1988 Aug 15;263(23):11443–11451. [PubMed] [Google Scholar]
- Petersen D. D., Magnuson M. A., Granner D. K. Location and characterization of two widely separated glucocorticoid response elements in the phosphoenolpyruvate carboxykinase gene. Mol Cell Biol. 1988 Jan;8(1):96–104. doi: 10.1128/mcb.8.1.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson T. C., Killary A. M., Fournier R. E. Chromosomal assignment and trans regulation of the tyrosine aminotransferase structural gene in hepatoma hybrid cells. Mol Cell Biol. 1985 Sep;5(9):2491–2494. doi: 10.1128/mcb.5.9.2491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petit C., Levilliers J., Ott M. O., Weiss M. C. Tissue-specific expression of the rat albumin gene: genetic control of its extinction in microcell hybrids. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2561–2565. doi: 10.1073/pnas.83.8.2561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potter H., Weir L., Leder P. Enhancer-dependent expression of human kappa immunoglobulin genes introduced into mouse pre-B lymphocytes by electroporation. Proc Natl Acad Sci U S A. 1984 Nov;81(22):7161–7165. doi: 10.1073/pnas.81.22.7161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quinn P. G., Wong T. W., Magnuson M. A., Shabb J. B., Granner D. K. Identification of basal and cyclic AMP regulatory elements in the promoter of the phosphoenolpyruvate carboxykinase gene. Mol Cell Biol. 1988 Aug;8(8):3467–3475. doi: 10.1128/mcb.8.8.3467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Short J. M., Wynshaw-Boris A., Short H. P., Hanson R. W. Characterization of the phosphoenolpyruvate carboxykinase (GTP) promoter-regulatory region. II. Identification of cAMP and glucocorticoid regulatory domains. J Biol Chem. 1986 Jul 25;261(21):9721–9726. [PubMed] [Google Scholar]
- Thayer M. J., Fournier R. E. Hormonal regulation of TSE1-repressed genes: evidence for multiple genetic controls in extinction. Mol Cell Biol. 1989 Jul;9(7):2837–2846. doi: 10.1128/mcb.9.7.2837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss M. C., Chaplain M. Expression of differentiated functions in hepatoma cell hybrids: reappearance of tyrosine aminotransferase inducibility after the loss of chromosomes. Proc Natl Acad Sci U S A. 1971 Dec;68(12):3026–3030. doi: 10.1073/pnas.68.12.3026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss M. C., Sparkes R. S., Bertolotti R. Expression of differentiated functions in hepatoma cell hybrids: IX extinction and reexpression of liver-specific enzymes in rat hepatoma-Chinese hamster fibroblast hybrids. Somatic Cell Genet. 1975 Jan;1(1):27–40. doi: 10.1007/BF01538730. [DOI] [PubMed] [Google Scholar]
- Wigler M., Pellicer A., Silverstein S., Axel R., Urlaub G., Chasin L. DNA-mediated transfer of the adenine phosphoribosyltransferase locus into mammalian cells. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1373–1376. doi: 10.1073/pnas.76.3.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wynshaw-Boris A., Lugo T. G., Short J. M., Fournier R. E., Hanson R. W. Identification of a cAMP regulatory region in the gene for rat cytosolic phosphoenolpyruvate carboxykinase (GTP). Use of chimeric genes transfected into hepatoma cells. J Biol Chem. 1984 Oct 10;259(19):12161–12169. [PubMed] [Google Scholar]
- Wynshaw-Boris A., Short J. M., Loose D. S., Hanson R. W. Characterization of the phosphoenolpyruvate carboxykinase (GTP) promoter-regulatory region. I. Multiple hormone regulatory elements and the effects of enhancers. J Biol Chem. 1986 Jul 25;261(21):9714–9720. [PubMed] [Google Scholar]
- Yoo-Warren H., Monahan J. E., Short J., Short H., Bruzel A., Wynshaw-Boris A., Meisner H. M., Samols D., Hanson R. W. Isolation and characterization of the gene coding for cytosolic phosphoenolpyruvate carboxykinase (GTP) from the rat. Proc Natl Acad Sci U S A. 1983 Jun;80(12):3656–3660. doi: 10.1073/pnas.80.12.3656. [DOI] [PMC free article] [PubMed] [Google Scholar]