Abstract
The objective of this study was to determine the optimal assessment of arterial stiffness that relates to diastolic dysfunction. Forty-one patients had measurements of brachial-ankle pulse wave velocity (baPWV), carotid-femoral pulse wave velocity (cfPWV), ankle brachial index (ABI), pulse pressure (PP), and augmentation index (AIx). Diastolic dysfunction was evaluated by echocardiographic indices of the ratio of the peak early diastolic mitral valve velocity and the peak late diastolic velocity (E/A ratio), left atrial diameter, and left atrial volume indexes. There was a significant (P < 0.05) correlation between baPWV and E/A ratio with an inverse relationship indicating that higher arterial stiffness was associated with greater diastolic dysfunction. In contrast, there was no significant correlation between E/A ratio and cfPWV, PP, ABI, or AIx. After multivariate analysis, the relationship between baPWV and E/A ratio remained significant (P < 0.05), independent of age and systolic blood pressure (BP). There were no correlations between any index of vascular stiffness and left atrial dimension or volume. In summary, baPWV correlates with diastolic dysfunction, independent of a patient's age and BP and is a better indicator of diastolic dysfunction than other indicators of arterial stiffness. baPWV has the utility of infering the presence of left ventricular diastolic dysfunction.
1. Introduction
Compromised left ventricular diastolic function represents an ongoing cardiovascular challenge from the perspective of its detection, assessment, and management [1, 2]. The early detection of diastolic dysfunction has garnered increasing attention because it predicts the development of heart failure [3, 4]. Currently, the primary imaging modality for assessing left ventricular diastolic function is Doppler echocardiography although there are controversies in the precision of its assessment [5–8]. It can require the integration of transmitral flow, pulmonary venous flow, mitral annular motion, myocardial deformation, and cardiac structure. This complex measurement of diastolic function can be reduced to several common elements—the assessment of transmitral flow velocity and evaluation of cardiac structure which includes left atrial size [1, 8]. The ratios of peak early diastolic mitral flow filling velocity (E) and of the peak late mitral diastolic filling velocity (A), E/A ratio, characterize the severity and stage of the diastolic dysfunction [9]. In diastolic dysfunction, the E/A ratio decreases as E velocity is reduced and A velocity is increased [9]. Because ageing alters the E/A ratio, age adjustment is necessary [6]. Another component of the assessment of diastolic dysfunction is left atrial (LA) dimension. LA size or volume has been proposed as a simple noninvasive assessment of the degree of LV diastolic dysfunction [10]. LA size is increased, and LA emptying is decreased in patients with diastolic heart failure [11].
While the role of arterial stiffness in prediction of cardiovascular events is well documented [12–15], the capacity of arterial stiffness to alter left ventricular diastolic function is less certain. Arterial stiffness is hypothesized to produce LV diastolic dysfunction through increased cardiac after load [16, 17] and reduced coronary perfusion; the latter is hypothesized to produce LV diastolic dysfunction through decreased diastolic blood pressure [18]. The concept that systolic arterial loading can impair subsequent diastolic function, however, was not substantiated by investigators using the more direct assessment of myocardial relaxation by examining LV pressure decay during early diastole or tau [19]. In addition, the proposition that arterial stiffness impairs left ventricular relaxation and subsequent diastolic dysfunction has had mixed clinical evidence, with some studies supporting the presence of an association [20], some are equivocal [21], while other studies found no relationship [22]. Some of the studies that reported a relationship between arterial stiffness and diastolic dysfunction did not adjust for factors such as age [20] that affect arterial stiffness and diastolic dysfunction independently.
Variations in reports may in part be due to differences in techniques for the measurement of arterial stiffness. Pulse wave velocity (PWV) is a validated method to quantify arterial stiffness [23] and can be measured in different arterial segments, with brachial-ankle PWV (baPWV) and carotid-femoral PWV (cfPWV) being the most common ones. Augmentation index (AIx), pulse pressure (PP), and increased ankle brachial index (ABI) are also indices of arterial stiffness. AIx has traditionally been considered to represent increased wave reflection [24], while more recently increased AIx has been attributed to a reduction in the reservoir function—compliance properties of the aorta and other elastic arteries [25]. Increased arterial stiffness produces an increase in systolic blood pressure and reduction in diastolic blood pressure which is easily recognized by an increase in pulse pressure [26]. Increased ABI, occurring in the absence of peripheral arterial disease, indicates a greater augmentation in systolic arterial pressure as the pulse wave moves the longer distance to the ankle arteries compared to the shorter distance to the brachial arteries, and it is due to an increase in arterial stiffness [27]. Which of these indices of arterial stiffness, if any, best identifies a relationship with diastolic dysfunction needs to be determined?
The objective of this study was to determine whether arterial stiffness is related to left ventricular diastolic dysfunction and to determine which of four indices of arterial stiffness—baPWV, cfPWV, ABI, or pulse pressure—correlated best with an echocardiographic assessment of diastolic dysfunction.
2. Methods
Patients from a cardiology clinic at a University teaching hospital who met the following criteria were studied. Men or women over the age of 45 years that agreed to participate in the study were included. Patients were only included if they had a satisfactory echocardiographic assessment without significant aortic or mitral valve diseases or impairment in left systolic ventricular function identified on echocardiographic assessment.
A noninvasive measure of vascular stiffness was performed as previously outlined [27, 28]. PWV and augmentation index were measured on an Omron Colin VP1000/2000 (Ill, USA). Briefly, the subject was examined while resting in the supine position, with electrocardiogram electrodes placed on both wrists and a microphone for detecting heart sounds placed on the left edge of the sternum. Blood pressure cuffs, which were connected to the device, were placed around both arms and ankles. After the participants rested in the supine position, the right carotid pulse wave and femoral artery contours were recorded simultaneously by placement of a transducer over these arteries. Using the carotid pressure waveform, the augmentation index (AIx) was calculated as the difference between the second and the first systolic shoulder and was expressed as a percentage of the carotid pulse contour. PWV was calculated as the ratio of the distance in meters from the brachial artery to ankle or carotid to femoral artery and the transit time in seconds, measured from the basal level of one pulse to the other either baPWV or cfPWV.
Cardiac structure and function were assessed according to a standardized protocol. Two-dimensional echocardiograms were performed. Parasternal long- and short-axes views were used to determine left ventricular end-diastolic and end-systolic measurements and wall thickness dimensions and left atrial diameter according to the recommendations of the American Society of Echocardiography [29]. Diastolic function was determined using Doppler assessment of mitral inflow velocity specifically peak early and late diastolic mitral flow velocities. Left atrial volume was measured using the biplane area-length method determined from the apical 4 chamber view. Left atrial volume was indexed by body surface area. All measurements were recorded by an ultrasonographer unaware of the clinical condition of the participants.
All numeric data are expressed as mean values (±SD), and categorical data are present at percentages. Univariate and multivariate regression analyses were conducted using PASW Statistics 18. Statistical significance was set at P < 0.05.
3. Results
The study population consisted of 41 adult patients (mean age 68 ± 11 years, 66% male) of whom 73% had hypertension and 12% had diabetes mellitus (Table 1). The echocardiographic data indicate that the group had a normal systolic function with an ejection fraction of 63.3 ± 5%. There was a significant correlation between baPWV and E/A ratio (Figure 1). The relationship was negative, indicating a decreasing E/A ratio with increases in baPWV. None of the other indices of arterial stiffness, cfPWV, ABI, or Aix, correlated significantly with E/A ratio (Table 2). Measures of left atrial size, left atrial diameter (LAD) and left atrial volume index (LAVI), did not correlate significantly with any index of arterial stiffness. LAD correlated significantly with age. The number of subjects without hypertension is small and precluded meaningful separate analysis.
Table 1.
Clinical characteristics of the study population.
| Number of subjects | 41 |
|---|---|
| Age (years) | 68 ± 11 |
| Gender (% men) | 66 |
| BMI (kg/m2) | 26.7 ± 3.6 |
| BSA (m2) | 1.84 ± 0.22 |
|
| |
| Hypertension (%) | 73.2 |
| Dyslipidemia (%) | 48.8 |
| Diabetes (%) | 12.2 |
| CAD (%) | 22.0 |
|
| |
| Medications | |
| β-blockers (%) | 34.1 |
| ACEIs (%) | 36.6 |
| ARBs (%) | 7.3 |
| CCBs (%) | 24.4 |
| Statins (%) | 43.9 |
| Aspirin (%) | 53.7 |
| Antiplatelets (%) | 14.6 |
| Diuretics (%) | 19.5 |
|
| |
| Heart rate (bpm) | 67.3 ± 12.8 |
| SBP (mm Hg) | 138.5 ± 18.1 |
| DBP (mm Hg) | 78.8 ± 8.4 |
| PP (mm Hg) | 59.7 ± 14.2 |
|
| |
| baPWV (cm/s) | 1814 ± 415 |
| cfPWV (cm/s) | 1610 ± 500 |
| AIx (%) | 20.0 ± 18.3 |
| ABI | 1.14 ± 0.12 |
|
| |
| LAD (mm) | 36.8 ± 6.5 |
| LAVI (ml/m2) | 33.5 ± 11.7 |
| E (cm/s) | 79.6 ± 26.0 |
| A (cm/s) | 76.0 ± 25.5 |
| E/A | 1.15 ± 0.54 |
| Age < 60: 1.3 ± 0.4 | |
| Age ≥ 60: 1.1 ± 0.6 | |
| LVEF (%) | 63.3 ± 5.0 |
| LVM (g) | 151.2 ± 36.6 |
| LVMI (g/m2) | 80.3 ± 21.0 |
| LVDD (mm) | 45.0 ± 5.1 |
| LVSD (mm) | 28.4 ± 5.5 |
| IVST (mm) | 10.0 ± 1.4 |
| PWT (mm) | 9.7 ± 1.2 |
BMI: body mass index, BSA: body surface index, CAD: coronary artery disease, ACEIs: angiotensin-converting enzyme inhibitors, ARBs: angiotensin II receptor antagonists, CCBs: calcium channel blockers, SBP: systolic blood pressure, DBP: diastolic blood pressure, PP: pulse pressure, ABI: ankle-brachial index, Aix: augmentation index, cfPWV: carotid-femoral pulse wave velocity, baPWV: brachial-ankle pulse wave velocity, LAD: left atrial diameter, LAVI: left atrial volume index, E: peak early diastolic filling velocity, A: peak late diastolic filling velocity, E/A: E/A ratio; LVEF: left ventricular ejection fraction, LVM: left ventricular mass, LVMI: left ventricular mass index, LVDD: left ventricular end-diastolic diameter, LVSD: left ventricular end-systolic diameter, IVS: interventricular septal end-diastolic thickness, and PWT: posterior wall end-diastolic thickness.
Figure 1.
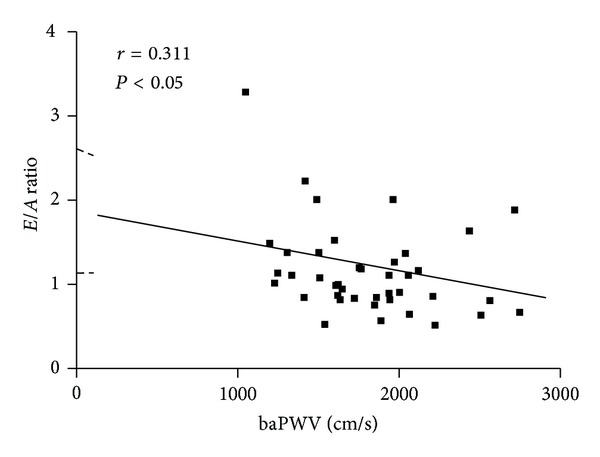
The relationship between baPWV and E/A ratio.
Table 2.
Univariate correlates between echocardiographic variables and indices of arterial stiffness.
| E/A | LAd | LAVI | ||||
|---|---|---|---|---|---|---|
| r | P | r | P | r | P | |
| baPWV | −0.311* | 0.04 | 0101 | 0.52 | 0.008 | 0.96 |
| cfPWV | −0.040 | 0.11 | 0.119 | 0.45 | 0.213 | 0.18 |
| PP | −0.152 | 0.34 | 0.260 | 0.10 | 0.238 | 0.13 |
| ABI | 0.156 | 0.32 | −0.096 | 0.55 | 0.135 | 0.40 |
| AIx | −0.044 | 0.78 | 0.048 | 0.76 | −0.098 | 0.54 |
| Age | −0.180 | 0.26 | 0.313* | 0.04 | 0.210 | 0.18 |
| SBP | −0.170 | 0.28 | 0.144 | 0.36 | 0.108 | 0.50 |
r: pearson correlation coefficient; other abbreviations as in Table 1.
*Indicates P < 0.05.
Because baPWV and cfPWV comprise some of the same elements of arterial stiffness, the relationship between baPWV and cfPWV was explored by a linear correlation analysis. There was a significant relationship between the two factors with an r value of 0.717 or r 2 value of 0.514 (Figure 2). The line of identity demonstrates that baPWV exceeds cfPWV on a consistent basis. baPWV was 17% greater than cfPWV.
Figure 2.
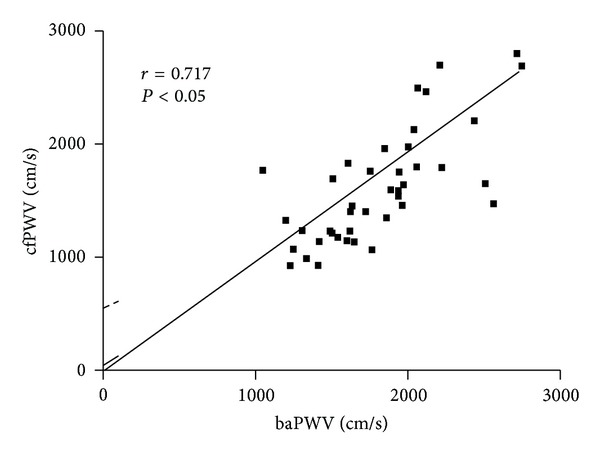
The relationship between baPWV and cfPWV.
Age was positively correlated with baPWV (P < 0.001) and cfPWV (P < 0.001). There were also significant positive correlations between systolic blood pressure (SBP) and both baPWV (P = 0.044) and cfPWV (P = 0.002). Pulse pressure was significantly correlated with cfPWV (P = 0.007), but IT did not correlate significantly with baPWV or AIx. There was a significant positive correlation between age and pulse pressure (P = 0.015).
The relationship between age and indices of aortic stiffness necessitates the use of multivariate analysis in order to consider the potential effect of age on the relationship between aortic stiffness and E/A ratio. A significant association between baPWV and E/A ratio persisted after adjusting for age, SBP, and PP in a multiple regression analysis (Table 3). Multivariate analysis was utilized, in case one of the variables was obscuring a relationship between cfPWV and E/A ratio. Age and SBP were significantly correlated with cfPWV in multivariate analysis. There was no significant relationship between cfPWV and E/A ratio after multiple regression analysis (Table 4).
Table 3.
Multivariate analysis for baPWV.
| Model | Unstandardized | Standardized | Sig | 95% Confidence | |||
|---|---|---|---|---|---|---|---|
| beta | S.E. | Beta | t | for beta | |||
| E/A | −455.5 | 215.065 | −.588 | −2.118 | .042 | −892.604 | −18.473 |
| Age | 26.9 | 4.959 | .751 | 5.418 | .000 | 16.791 | 36.947 |
| SBP | 21.5 | 6.315 | .937 | 3.401 | .002 | 8.644 | 34.310 |
| PP | −23.7 | 8.286 | −.811 | −2.854 | .007 | −40.489 | −6.810 |
Table 4.
Multivariate analysis for cfPWV.
| Model | Unstandardized | Standardized | Sig | 95% Confidence | |||
|---|---|---|---|---|---|---|---|
| Beta | S.E. | Beta | t | for beta | |||
| E/A | −21.990 | 278.229 | −.024 | −.079 | .937 | −587.418 | 543.438 |
| Age | 26.356 | 6.415 | .611 | 4.108 | .000 | 13.319 | 39.393 |
| SBP | 22.997 | 8.170 | .833 | 2.815 | .008 | 6.395 | 39.600 |
| PP | −18.876 | 10.720 | −.537 | −1.761 | .087 | −40.661 | 2.909 |
SBP is systolic blood pressure.
PP is pulse pressure.
4. Discussion
The major contribution of this study is the comparative analysis of different indices of arterial stiffness and their association with left ventricular diastolic dysfunction. We found that baPWV was significantly correlated with an indicator of diastolic dysfunction, and the association was independent of age and blood pressure. Importantly, baPWV was a better predictor of diastolic dysfunction than several other indices of arterial stiffness, namely, cfPWV, AIx, PP, or ABI.
Both baPWV and cfPWV are indicators of arterial stiffness, and each of them is related to cardiovascular morbidity and mortality [14, 15, 30–32]. With regards to diastolic dysfunction, recent investigations have mainly examined either baPWV [22, 33–37] or cfPWV [20, 38–41] in relation to E/A ratio. We compared baPWV and cfPWV and found a significant relationship between diastolic dysfunction and baPWV but not with cfPWV. It was not our purpose to compare baPWV versus cfPWV over the spectrum of cardiovascular diseases; we rather focused solely on the association with an index of left ventricular diastolic function and found a greater utility for baPWV. While both baPWV and cfPWV are indicators of arterial stiffness, baPWV is an indicator of the combination of central and peripheral arterial stiffness [31, 42–44]. Our study suggests that the effects of peripheral arterial stiffness on ventricular diastolic function are better represented by the greater arterial path measured by baPWV as compared to the centrally confined path of cfPWV. While baPWV and cfPWV are highly and significantly correlated with each other, the correlation is far from being identical. We found a r 2 of 0.513, indicating that only about 50% of the variance of one is accounted for by the other. This value is similar to the one reported by others (r 2 = 0.63) [42]. We found that baPWV was predominantly greater than cfPWV by 17%. This is consistent with another report that shows that, on average, baPWV was approximately 20% higher than cfPWV [42]. The higher value presumably reflects the additional contribution of “peripheral” (or muscular) arterial stiffness to central arterial stiffness [43]. baPWV recognizes a different vascular stiffness pathway, and, is one differently affected by interventions such as weight loss [45]. The association of baPWV with an increased risk of total cardiovascular events and all-cause mortality [15] may reflect the effect of baPWV on the heart.
Our finding that baPWV correlates with LV diastolic function is consistent with several studies [34, 35, 37] but they did not compare baPWV with a wide range of other measurements of arterial stiffness. Wang et al. compared PWV measurements in the brachial-ankle, carotid-femoral, heart-femoral, and heart-carotid segments among hypertensive patients and also found baPWV to be the best predictor of diastolic dysfunction amongst measurements of PWV [46]. However, they did not conduct a multivariate analysis adjusting for age and blood pressure [46]—two factors that influence diastolic cardiac function.
Age and systolic blood pressure are the two primary determinants of baPWV and cfPWV as we found and as has been reported previously [42]. Therefore, our measurements of PWV were adjusted for these two parameters. With advancing age, arterial stiffness increases due to changes in arterial elasticity and compliance, leading to elevations in systolic pressures and pulse pressure [47–49]. Prolonged elevations in blood pressure can also independently lead to similar changes in the vasculature that increases arterial stiffness [50, 51]. The associations of age, SBP, and PP with baPWV and cfPWV are confirmed in the present study. After including these factors in multivariate analysis, the negative relationship between baPWV and E/A ratio remained significant, and the baPWV relationship with mitral valve velocity measurements was independent of age, SBP, and PP.
We did not find any associations between indices of arterial stiffness and left atrial size assessment either by left atrial diameter or left atrial volume. While left atrial size has been purported to be a marker of chronic diastolic dysfunction and cardiovascular disease risk [52], previous data on the relationship of left atrial size and arterial stiffness have been limited and somewhat conflicting. One study reported no significant association between PWV or pulse pressure and left atrial size but an association between AIx and left atrial size [21]. Another study reported a significant correlation between cfPWV and left atrial diameter [39]. Brachial-ankle PWV and cfPWV correlated with left atrial diameter in patients with hypertension, end-stage renal disease, and heart failure [53]. One potential explanation for the differences between studies is the differences in the proportion of other factors that influence left atrial size in each of the study population. Left atrial size is larger in patients with hypertension and in the elderly as well as patients with reduced left ventricular systolic function [11, 22]. The proportion of individuals with each of these characteristics likely influenced the relationship between left atrial size and arterial stiffness. Our study consisted of individuals with normal LV systolic function.
Augmentation index was not associated with diastolic dysfunction in our study. This finding is consistent with two studies [21, 54] but contrasts with the findings of two another studies [55, 56]. While AIx is an indicator of arterial stiffness, pulse wave velocity more closely correlates with adverse cardiovascular outcomes [57]. We did not find any relationship between pulse pressure and diastolic function. Our findings are consistent with some previous data that say that PWV is superior to central and brachial pulse pressures for the detection of diastolic dysfunction in older adults with “preserved” LV ejection fraction [58].
Several possible limitations of this study should be considered. The patient population is relatively small. However, significant correlations were still found. Secondly, the changes in E/A ratio with the progression of diastolic dysfunction are such that the initial impairment of diastolic function is manifested as a decreased E/A ratio. In long standing disease, changes in ventricular and atrial structure occur, and the diastolic dysfunction may predominantly have a restrictive pattern, which is manifested as an increased E/A ratio [6]. Others however disagree with this postulated pattern of progression [8]. The use of decreasing E/A ratio as an indicator of diastolic dysfunction is appropriate in this study that consists of a study population without severe cardiac disease. Third, we focused mainly on E/A ratio as an indicator of diastolic dysfunction. Mitral valve velocities and myocardial tissue Doppler measurements can provide additional important data on the state of diastolic function. However, each of these measurements has unique limitations in their correlation with more precise measurements of diastolic function [8]. Importantly, E/A ratio remains a standard among the field of the detection of diastolic dysfunction. Fourth, subjects were on antihypertensive drugs that can impact on arterial stiffness and minimize the wave reflection thereby altering the relative strength of the relationship between different indices of arterial stiffness and diastolic function [59]. Fifth, we use the term arterial stiffness recognizing that this term may encompass abnormalities in separate measures of arterial compliance, distensibility, pressure augmentation, or wave reflection in different segments of the vasculature.
In summary, the present study shows that increased baPWV is associated with diastolic dysfunction, measured by E/A ratio. Age, SBP, and PP were major determinants of increased baPWV, but the relationship between baPWV and E/A ratio was independent of these clinical variables. Our findings show that baPWV is more closely correlated with E/A ratio than with other indices of arterial stiffness, namely cfPWV, AIx, ABI, or pulse pressure. The more extensive arterial pathway measured by baPWV in comparison to cfPWV may explain this relationship. These data suggest that measuring baPWV has the utility of infering or suspecting the presence of left ventricular diastolic dysfunction.
Conflict of Interests
The authors declare that they have no conflict of interests.
References
- 1.Borlaug BA, Paulus WJ. Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment. European Heart Journal. 2011;32(6):670–679. doi: 10.1093/eurheartj/ehq426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McMurray JJ, Adamopoulos S, Anker SD, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008. The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM) European Journal of Heart Failure. 2008;10(10):933–989. doi: 10.1016/j.ejheart.2008.08.005. [DOI] [PubMed] [Google Scholar]
- 3.Kane GC, Karon BL, Mahoney DW, et al. Progression of left ventricular diastolic dysfunction and risk of heart failure. Journal of the American Medical Association. 2011;306(6):856–863. doi: 10.1001/jama.2011.1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lam CSP, Lyass A, Kraigher-Krainer E, et al. Cardiac dysfunction and noncardiac dysfunction as precursors of heart failure with reduced and preserved ejection fraction in the community. Circulation. 2011;124(1):24–30. doi: 10.1161/CIRCULATIONAHA.110.979203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tschöpe C, Paulus WJ. Is echocardiographic evaluation of diastolic function useful in determining clinical care? Doppler echocardiography yields dubious estimates of left ventricular diastolic pressures. Circulation. 2009;120(9):810–819. doi: 10.1161/CIRCULATIONAHA.109.869628. [DOI] [PubMed] [Google Scholar]
- 6.Nagueh SF, Appleton CP, Gillebert TC, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography. Journal of the American Society of Echocardiography. 2009;22(2):107–133. doi: 10.1016/j.echo.2008.11.023. [DOI] [PubMed] [Google Scholar]
- 7.Little WC, Oh JK. Echocardiographic evaluation of diastolic function can be used to guide clinical care. Circulation. 2009;120(9):802–809. doi: 10.1161/CIRCULATIONAHA.109.869602. [DOI] [PubMed] [Google Scholar]
- 8.Ogunyankin KO. Assessment of left ventricular diastolic function: the power, possibilities, and pitfalls of echocardiographic imaging techniques. Canadian Journal of Cardiology. 2011;27(3):311–318. doi: 10.1016/j.cjca.2010.12.042. [DOI] [PubMed] [Google Scholar]
- 9.Nishimura RA, Tajik AJ. Evaluation of diastolic filling of left ventricle in health and disease: doppler echocardiography is the clinician’s Rosetta Stone. Journal of the American College of Cardiology. 1997;30(1):8–18. doi: 10.1016/s0735-1097(97)00144-7. [DOI] [PubMed] [Google Scholar]
- 10.Simek CL, Feldman MD, Haber HL, Wu CC, Jayaweera AR, Kaul S. Relationship between left ventricular wall thickness and left atrial size: comparison with other measures of diastolic function. Journal of the American Society of Echocardiography. 1995;8(1):37–47. doi: 10.1016/s0894-7317(05)80356-6. [DOI] [PubMed] [Google Scholar]
- 11.Gottdiener JS, Kitzman DW, Aurigemma GP, Arnold AM, Manolio TA. Left atrial volume, geometry, and function in systolic and diastolic heart failure of persons ≥65 years of age (The Cardiovascular Health Study) The American Journal of Cardiology. 2006;97(1):83–89. doi: 10.1016/j.amjcard.2005.07.126. [DOI] [PubMed] [Google Scholar]
- 12.Vlachopoulos C, Aznaouridis K, O’Rourke MF, Safar ME, Baou K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: a systematic review and meta-analysis. European Heart Journal. 2010;31(15):1865–1871. doi: 10.1093/eurheartj/ehq024. [DOI] [PubMed] [Google Scholar]
- 13.Mattace-Raso FUS, Van Der Cammen TJM, Hofman A, et al. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation. 2006;113(5):657–663. doi: 10.1161/CIRCULATIONAHA.105.555235. [DOI] [PubMed] [Google Scholar]
- 14.Laurent S, Boutouyrie P, Asmar R, et al. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension. 2001;37(5):1236–1241. doi: 10.1161/01.hyp.37.5.1236. [DOI] [PubMed] [Google Scholar]
- 15.Vlachopoulos C, Aznaouridis K, Terentes-Printzios D, Ioakeimidis N, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with brachial-ankle elasticity index: a systematic review and meta-analysis. Hypertension. 2012;60(2):556–562. doi: 10.1161/HYPERTENSIONAHA.112.194779. [DOI] [PubMed] [Google Scholar]
- 16.Weber T, O’Rourke MF, Ammer M, Kvas E, Punzengruber C, Eber B. Arterial stiffness and arterial wave reflections are associated with systolic and diastolic function in patients with normal ejection fraction. American Journal of Hypertension. 2008;21(11):1194–1202. doi: 10.1038/ajh.2008.277. [DOI] [PubMed] [Google Scholar]
- 17.Borlaug BA, Melenovsky V, Redfield MM, et al. Impact of arterial load and loading sequence on left ventricular tissue velocities in humans. Journal of the American College of Cardiology. 2007;50(16):1570–1577. doi: 10.1016/j.jacc.2007.07.032. [DOI] [PubMed] [Google Scholar]
- 18.Guelen I, Mattace-Raso FUS, Van Popele NM, et al. Aortic stiffness and the balance between cardiac oxygen supply and demand: the Rotterdam Study. Journal of Hypertension. 2008;26(6):1237–1243. doi: 10.1097/HJH.0b013e3282fcc393. [DOI] [PubMed] [Google Scholar]
- 19.Starling MR, Montgomery DG, Mancini GBJ, Walsh RA. Load independence of the rate of isovolumic relaxation in man. Circulation. 1987;76(6):1274–1281. doi: 10.1161/01.cir.76.6.1274. [DOI] [PubMed] [Google Scholar]
- 20.Soldatos G, Jandeleit-Dahm K, Thomson H, et al. Large artery biomechanics and diastolic dysfunctionin patients with Type 2 diabetes. Diabetic Medicine. 2011;28(1):54–60. doi: 10.1111/j.1464-5491.2010.03146.x. [DOI] [PubMed] [Google Scholar]
- 21.Abhayaratna WP, Barnes ME, O’Rourke MF, et al. Relation of arterial stiffness to left ventricular diastolic function and cardiovascular risk prediction in patients ≥65 years of age. The American Journal of Cardiology. 2006;98(10):1387–1392. doi: 10.1016/j.amjcard.2006.06.035. [DOI] [PubMed] [Google Scholar]
- 22.Hsu PC, Lin TH, Lee CS, et al. Mismatch between arterial stiffness increase and left ventricular diastolic dysfunction. Heart and Vessels. 2010;25(6):485–492. doi: 10.1007/s00380-010-0021-4. [DOI] [PubMed] [Google Scholar]
- 23.Asmar R, Benetos A, Topouchian J, et al. Assessment of arterial distensibility by automatic pulse wave velocity measurement: validation and clinical application studies. Hypertension. 1995;26(3):485–490. doi: 10.1161/01.hyp.26.3.485. [DOI] [PubMed] [Google Scholar]
- 24.O’Rourke MF, Nichols WW. Aortic diameter, aortic stiffness, and wave reflection increase with age and isolated systolic hypertension. Hypertension. 2005;45(4):652–658. doi: 10.1161/01.HYP.0000153793.84859.b8. [DOI] [PubMed] [Google Scholar]
- 25.Davies JE, Baksi J, Francis DP, et al. The arterial reservoir pressure increases with aging and is the major determinant of the aortic augmentation index. American Journal of Physiology. 2010;298(2):H580–H586. doi: 10.1152/ajpheart.00875.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nichols WW, O'Rouke MF, Vlackopoulos C. McDonald's Blood Flow in Arteries: Theoretical, Experimental and Clinical Principles. London, UK: Hodder Arnold; 2011. [Google Scholar]
- 27.Rabkin SW, Chan SH, Sweeney C. Ankle-brachial index as an indicator of arterial stiffness in patients without peripheral artery disease. Angiology. 2012;63:150–154. doi: 10.1177/0003319711410307. [DOI] [PubMed] [Google Scholar]
- 28.Rabkin SW, Chan SH. Correlation of pulse wave velocity with left ventricular mass in patients with hypertension once blood pressure has been normalized. Heart International. 2012;7:p. e5. doi: 10.4081/hi.2012.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lang RM, Bierig M, Devereux RB, et al. Recommendations for chamber quantification: a report from the American Society of Echocardiography's guidelines and standards committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology. Journal of the American Society of Echocardiography. 2005;18(12):1440–1463. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 30.Turin TC, Kita Y, Rumana N, et al. Brachial-ankle pulse wave velocity predicts all-cause mortality in the general population: findings from the Takashima study, Japan. Hypertension Research. 2010;33(9):922–925. doi: 10.1038/hr.2010.103. [DOI] [PubMed] [Google Scholar]
- 31.Yamashina A, Tomiyama H, Takeda K, et al. Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertension Research. 2002;25(3):359–364. doi: 10.1291/hypres.25.359. [DOI] [PubMed] [Google Scholar]
- 32.Nakamura M, Yamashita T, Yajima J, et al. Brachial-ankle pulse wave velocity as a risk stratification index for the short-term prognosis of type 2 diabetic patients with coronary artery disease. Hypertension Research. 2010;33(10):1018–1024. doi: 10.1038/hr.2010.126. [DOI] [PubMed] [Google Scholar]
- 33.Usui Y, Takata Y, Inoue Y, et al. Severe obstructive sleep apnea impairs left ventricular diastolic function in non-obese men. Sleep Medicine. 2011;4:p. 4. doi: 10.1016/j.sleep.2010.09.014. [DOI] [PubMed] [Google Scholar]
- 34.Xu L, Jiang CQ, Lam TH, et al. Arterial stiffness and left-ventricular diastolic dysfunction: Guangzhou Biobank Cohort Study-CVD. Journal of Human Hypertension. 2011;25(3):152–158. doi: 10.1038/jhh.2010.44. [DOI] [PubMed] [Google Scholar]
- 35.Chung CM, Chu CM, Chang ST, et al. Quantification of aortic stiffness to predict the degree of left ventricular diastolic function. American Journal of the Medical Sciences. 2010;340(6):468–473. doi: 10.1097/MAJ.0b013e3181f0142c. [DOI] [PubMed] [Google Scholar]
- 36.Yambe M, Tomiyama H, Hirayama Y, et al. Arterial stiffening as a possible risk factor for both atherosclerosis and diastolic heart failure. Hypertension Research. 2004;27(9):625–631. doi: 10.1291/hypres.27.625. [DOI] [PubMed] [Google Scholar]
- 37.Masugata H, Senda S, Yoshikawa K, et al. Relationships between echocardiographic findings, pulse wave velocity, and carotid atherosclerosis in type 2 diabetic patients. Hypertension Research. 2005;28(12):965–971. doi: 10.1291/hypres.28.965. [DOI] [PubMed] [Google Scholar]
- 38.Soldatos G, Jandeleit-Dahm K, Thomson H, et al. Large artery biomechanics and diastolic dysfunctionin patients with type 2 diabetes. Diabetic Medicine. 2011;28(1):54–60. doi: 10.1111/j.1464-5491.2010.03146.x. [DOI] [PubMed] [Google Scholar]
- 39.Triantafyllidi H, Tzortzis S, Lekakis J, et al. Association of target organ damage with three arterial stiffness indexes according to blood pressure dipping status in untreated hypertensive patients. American Journal of Hypertension. 2010;23(12):1265–1272. doi: 10.1038/ajh.2010.156. [DOI] [PubMed] [Google Scholar]
- 40.Tsioufis C, Chatzis D, Dimitriadis K, et al. Left ventricular diastolic dysfunction is accompanied by increased aortic stiffness in the early stages of essential hypertension: a TDI approach. Journal of Hypertension. 2005;23(9):1745–1750. doi: 10.1097/01.hjh.0000174394.57644.69. [DOI] [PubMed] [Google Scholar]
- 41.Libhaber CD, Norton GR, Majane OHI, et al. Contribution of central and general adiposity to abnormal left ventricular diastolic function in a community sample with a high prevalence of obesity. The American Journal of Cardiology. 2009;104(11):1527–1533. doi: 10.1016/j.amjcard.2009.07.020. [DOI] [PubMed] [Google Scholar]
- 42.Tanaka H, Munakata M, Kawano Y, et al. Comparison between carotid-femoral and brachial-ankle pulse wave velocity as measures of arterial stiffness. Journal of Hypertension. 2009;27(10):2022–2027. doi: 10.1097/HJH.0b013e32832e94e7. [DOI] [PubMed] [Google Scholar]
- 43.Sugawara J, Hayashi K, Yokoi T, et al. Brachial-ankle pulse wave velocity: an index of central arterial stiffness? Journal of Human Hypertension. 2005;19(5):401–406. doi: 10.1038/sj.jhh.1001838. [DOI] [PubMed] [Google Scholar]
- 44.Munakata M, Ito N, Nunokawa T, Yoshinaga K. Utility of automated brachial ankle pulse wave velocity measurements in hypertensive patients. American Journal of Hypertension. 2003;16(8):653–657. doi: 10.1016/s0895-7061(03)00918-x. [DOI] [PubMed] [Google Scholar]
- 45.Cooper JN, Buchanich JM, Youk A, et al. Reductions in arterial stiffness with weight loss in overweight and obese young adults: potential mechanisms. Atherosclerosis. 2012;223(2):485–490. doi: 10.1016/j.atherosclerosis.2012.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang CP, Hung WC, Yu TH, et al. Brachial-ankle pulse wave velocity as an early indicator of left ventricular diastolic function among hypertensive subjects. Clinical and Experimental Hypertension. 2009;31(1):31–43. doi: 10.1080/10641960802409796. [DOI] [PubMed] [Google Scholar]
- 47.Mitchell GF, Parise H, Benjamin EJ, et al. Changes in arterial stiffness and wave reflection with advancing age in healthy men and women: the Framingham Heart Study. Hypertension. 2004;43(6):1239–1245. doi: 10.1161/01.HYP.0000128420.01881.aa. [DOI] [PubMed] [Google Scholar]
- 48.Benetos A, Waeber B, Izzo J, et al. Influence of age, risk factors, and cardiovascular and renal disease on arterial stiffness: clinical applications. American Journal of Hypertension. 2002;15(12):1101–1108. doi: 10.1016/s0895-7061(02)03029-7. [DOI] [PubMed] [Google Scholar]
- 49.Vaitkevicius PV, Fleg JL, Engel JH, et al. Effects of age and aerobic capacity on arterial stiffness in healthy adults. Circulation. 1993;88(4):1456–1462. doi: 10.1161/01.cir.88.4.1456. [DOI] [PubMed] [Google Scholar]
- 50.Franklin SS. Arterial stiffness and hypertension: a two-way street? Hypertension. 2005;45(3):349–351. doi: 10.1161/01.HYP.0000157819.31611.87. [DOI] [PubMed] [Google Scholar]
- 51.O’Rourke M. Arterial stiffness, systolic blood pressure, and logical treatment of arterial hypertension. Hypertension. 1990;15(4):339–347. doi: 10.1161/01.hyp.15.4.339. [DOI] [PubMed] [Google Scholar]
- 52.Douglas PS. The left atrium: a biomarker of chronic diastolic dysfunction and cardiovascular disease risk. Journal of the American College of Cardiology. 2003;42(7):1206–1207. doi: 10.1016/s0735-1097(03)00956-2. [DOI] [PubMed] [Google Scholar]
- 53.Yu WC, Chuang SY, Lin YP, Chen CH. Brachial-ankle vs carotid-femoral pulse wave velocity as a determinant of cardiovascular structure and function. Journal of Human Hypertension. 2008;22(1):24–31. doi: 10.1038/sj.jhh.1002259. [DOI] [PubMed] [Google Scholar]
- 54.Nakae I, Matsuo S, Matsumoto T, Mitsunami K, Horie M. Augmentation index and pulse wave velocity as indicators of cardiovascular stiffness. Angiology. 2008;59(4):421–426. doi: 10.1177/0003319707306299. [DOI] [PubMed] [Google Scholar]
- 55.Ikonomidis I, Lekakis J, Papadopoulos C, et al. Incremental value of pulse wave velocity in the determination of coronary microcirculatory dysfunction in never-treated patients with essential hypertension. American Journal of Hypertension. 2008;21(7):806–813. doi: 10.1038/ajh.2008.172. [DOI] [PubMed] [Google Scholar]
- 56.Weber T, Auer J, O’Rourke MF, Punzengruber C, Kvas E, Eber B. Prolonged mechanical systole and increased arterial wave reflections in diastolic dysfunction. Heart. 2006;92(11):1616–1622. doi: 10.1136/hrt.2005.084145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Laurent S, Boutouyrie P. Recent advances in arterial stiffness and wave reflection in human hypertension. Hypertension. 2007;49(6):1202–1206. doi: 10.1161/HYPERTENSIONAHA.106.076166. [DOI] [PubMed] [Google Scholar]
- 58.Abhayaratna WP, Srikusalanukul W, Budge MM. Aortic stiffness for the detection of preclinical left ventricular diastolic dysfunction: pulse wave velocity versus pulse pressure. Journal of Hypertension. 2008;26(4):758–764. doi: 10.1097/HJH.0b013e3282f55038. [DOI] [PubMed] [Google Scholar]
- 59.Matsui Y, Eguchi K, O’Rourke MF, et al. Differential effects between a calcium channel blocker and a diuretic when used in combination with angiotensin II receptor blocker on central aortic pressure in hypertensive patients. Hypertension. 2009;54(4):716–723. doi: 10.1161/HYPERTENSIONAHA.109.131466. [DOI] [PubMed] [Google Scholar]


