Abstract
Mucins are high-molecular-weight glycoproteins, which are heavily decorated with a large number of O-linked oligosaccharides and a few N-glycan chains, linked to a protein backbone. The protein backbone is called mucin core protein or MUC apomucins. MUC expression is down-regulated or up-regulated in malignant neoplasms. These alterations of MUC apomucins, which are regulated by MUC genes, are associated with carcinogenesis and malignant potentials of cancers. MUC expression during human fetal intrahepatic bile duct (IBD) development has been studied only once, and there has been only one histochemical study of mucins in human fetal IBD development. The author herein immunohistochemically investigated the expression of MUC1, MUC2, MUC5AC, and MUC6, and histochemically investigated carbohydrate component of mucins in human fetal cholangiocytes with the use of 32 human fetal livers of various gestational ages. MUC1 is a transmembranous apomucin, while MUC2, MUC5AC and MUC6 are secretory apomucins. Under normal conditions, MUC1 (polymorphic epithelial mucin) is present mainly in the pancreatic epithelium. MUC2 (goblet cell mucin) is mainly located in goblet cells. MUC5AC (gastric foveolar mucin) and MUC6 (pyloric gland-type mucin) are located in the stomach. In the present study, the processes of the human IBD development could be categorized into four stages; ductal plate (DP), remodeling DP, remodeled DP, and mature IBDs. The author identified that MUC1 was present in ductal plate (DP), remodeling DP, remodeled DP, and mature IBD in human fetal livers. MUC5AC and MUC6 were present only in the DP. MUC5AC and MUC6 were absent in remodeling DP, remodeled DP, and mature IBD in human fetal livers. No expression of MUC2 was seen throughout the fetal IBD development. Histochemically, no carbohydrate component of mucins were seen in the remodeling DP and remodeled DP, while neutral and acidic mucins (carboxylated and sulfated mucins) were seen in mature IBD in human fetal livers. The DP showed frequently neutral mucins and less frequently acidic mucins (carboxylated and sulfated mucins residues). These findings suggest that the DP cells have MUC1, MUC5AC and MUC6, and that remodeling DP, remodeled DP, and mature IBDs have MUC1, but not MUC5AC and MUC6. The presence of neutral and acidic carbohydrates in DP suggests that these carbohydrates of mucin are attached to the MUC5AC and MUC6 mucin core proteins. Although the implications are unclear, the expression of these MUC apomucins and their carbohydrate residues are associated with normal development of IBDs in human fetal livers.
Keywords: Ductal plate, human fetal liver, intrahepatic bile duct development, mucins, MUC apomucins, histochemistry, immunohistochemistry
Introduction
The author has investigated the fetal development of intrahepatic bile ducts (IBDs) in humans [1-16]. The similar studies of fetal development of IBDs in humans have been reported by Desmet’s group [17-20] and Gerber’s group [21-23]. The author’s studies [1-16] and other studies [17-23] have revealed that the IBDs are derived from fetal ductal plate (DP) which is a double layered cylindrical structures located in the interface between hepatoblasts and portal mesenchyme [1-23]. The DP undergoes remodeling, giving rise to the emergence of future IBDs. The remnants of DP disappear by apoptosis [7]. The remodeled DP further give rise to mature IBDs resembling adult IBDs [1-23]. The author demonstrated that intrahepatic peribiliary glands, which were discovered by the author [24-36], are also derived from DP in human livers [1,5]. The author also proved that pancreatic acinar cells clusters may be derived from remodeling DP and remodeled DP in human fetal livers [1,5,6]. The author demonstrated that the normal development of the human fetal IBDs involves many molecules and molecular mechanisms including apoptosis, apoptosis-related proteins, DP cell proliferation, pancreatic digestive enzymes such as α-amylase, trypsinogen and lipase, some proteinases including matrix metalloproteinases and tissue inhibitors of matrix metalloproteinases, peribiliary vascular plexus, carbohydrate structures of many glycoproteins, MUC apomucin expression, expression of cytokeratin (CK), E-cadherin-catenin systems, double-stranded RNA-activated protein kinase, midkine, truncated midkine, type IV collagen, laminin, tenascin, trypsin, chymotrypsin, transforming growth factor-α and its receptor, and pancreatic amylase mRNA [1-16].
The developmental failures of these human fetal IBDs of DP give rise to the persistence of biliary structures in the postnatal human livers. Such structures are called DP malformations (DPM) or hepatobiliary fibropolycystic disease, which is seen in congenital hepatic fibrosis, polycystic diseases (adult and infantile) of the liver and kidneys, congenital biliary atresia, von-Meyenburg complex, and Caroli’s disease [17-20,37-42]. DP is also seen in ductular reactions [43-45] and DPM in some liver hamartoma [44] and cholangiocarcinoma [46]. The DP in ductular reaction of focal nodular hyperplasia interestingly may express KIT [45], a receptor of stem cell factor [47].
Mucins are high-molecular-weight glycoproteins, which are heavily decorated with a large number of O-linked oligosaccharides and a few N-glycan chains, linked to a protein backbone [48]. The protein backbone is called mucin core protein or MUC [48]. At present, at least 20 MUC have been identified [49] in humans. Mucins are now classified into secreted mucins and transmembrane mucins [48,49]. Secreted mucins form mucin gels, and are composed of MUC2, MUC5AC, MUC5B, MUC6, MUC7, MUC8, MUC9 and MUC19, while transmembrane mucins consist of MUC1, MUC3A, MUC3B, MUC4, MUC11, MUC12, MUC13, MUC15, MUC16, MUC17, MUC20 and MUC21. These transmembranous MUCs are located in the cell membrane and do not form mucin gels [48,49]. These MUC proteins are encoded by various MUC genes [48,49]. Mucins play an important role in the protection, local innate immunity, and lubrication of mucosal surface of various organs. Mucins are also involved in the pathogenesis of benign and malignant diseases of secretory epithelial cells [48,49]. It is well known that MUC expression is down-regulated or up-regulated in most malignant neoplasms of various organs [48,49]. These alterations of MUC apomucins, which are regulated by MUC genes, are thought to be associated with carcinogenesis and malignant potentials of cancer, though the mechanisms remain unclear [50-52].
Among the MUC apomucins, MUC1, MUC2, MUC5AC and MUC6 are representative. MUC1 is a transmembranous apomucin, and present dominantly in pancreatic epithelium [48,49]. MUC1 is also called “polymorphic epithelial mucin (PEM)”. MUC2 is a secretory apomucin, and present mainly in goblet cells of small intestine, large intestine, and bronchus. MUC2 is also called “goblet cell mucin”. MUC5AC is a secretory apomucin, and is seen mainly in gastric foveolar cells. MUC5AC is also called “gastric foveolar mucin”. MUC6 is also a secretory mucin, and is found largely in pyloric glands of the stomach, duodenal Brunner’s glands, and esophageal glands. MUC6 is also termed “pyloric gland-type mucin” [48-52].
MUC apomucin expression has been studied largely in pathogenesis of carcinogenesis of carcinomas of various organs [48-52]. There has been only one study of the expression of MUC apomucins during human fetal IBD development [13]. There is only one study on the carbohydrates side chain structures of mucins in human fetal IBD development [2]. In addition, there have been no comprehensive studies on mucins using mucin histochemical techniques. The author herein investigated the expression of MUC1, MUC2, MUC5AC, and MUC6 in human fetal IBD development. The author also herein report the expression mucins performed by conventional mucin stains.
Materials and methods
The author collected 32 human fetal livers at various hospitals. They were abortions (spontaneous and artificial), intrauterine fetal death, and autopsies. The gestational ages (weeks) of the 32 fetal livers were as follows: 7, 8, 9 (n=2), 10 (n=3), 11 (n=2), 12 (n=3), 13 (n=2), 14 (n=2), 15 (n=2), 16 (n=2), 17, 18, 19, 21, 23, 24, 26, 29, 30, 36, 38, and 40 week. The sex was unclear. Informed consent was obtained from each mother. The fetal liver specimens thus obtained were immediately fixed in formalin and embedded in paraffin. Many 3μm thin sections were cut, and they were subjected to hematoxylin and eosin (HE) stain, mucus histochemistry, and immunohistochemistry.
The histological examination was done with HE stain. Mucins were investigated by mucin histochemical stains including mucicarmine, diastase-periodic acid-Schiff (d-PAS), PAS, Alcian blue (AB) at pH2.5 and pH1.0, and combined d-PAS and AB at pH2.5, as previously reported [24,27].
A cytokeratin (CK) immunohistochemical study was performed to identify biliary cells which are positive for CK7, CK8, CK18 and CK19 and hepatocellular precursor cells which are positive for CK8 and CK18 [1,17-19]. Thus, CK immunostaining was performed to differentiated primitive human fetal cholangiocytes from primitive human hepatocytes [1,17-19].
An immunohistochemical study was performed with the use of Dako Envision method (Dako Corp, Glostrup, Denbmark) as previously described [53,54]. The antibodies used were as follows: MUC1 (clone Ma695, Novocastra Laboratories, NewCastle Upon Tyne, UK; diluation= 1:100), MUC2 (clone Ccp58, Novocastra; diluation=1:100), MUC5AC (clone CLH2, Novocastra; dilution=1:200), MUC6 (clone CLH5, Novocastra; dilution=1:200), CK7 (clone OV-TL, Dako Corp, dilution=1:200), CK8 (Clone 35bH11, Dako Corp, dilution=1:150), CK18 (clone DC10, Dako Corp, dilution=1:200), and CK19 (clone BA17, Dako Corp, dilution=1:100). Microwave pretreatment was performed by each immunohistochemical run.
Results
Histologically, the processes of the human IBD development can be categorized into four stages; DP, remodeling DP, remodeled DP, and mature IBDs. The DP was composed of single or double-layered cylindrical structures located in the interface between primitive hepatocytes (hepatoblast) and mesenchyme of the portal tracts (Figure 1A-C). The remodeling DP showed disappearance of some cells of the DP and development of tubular structures of future bile ducts, which was moving into the portal mesenchyme (Figure 1D). The remodeled DP was characterized by almost disappearance of DP and the tubular structures moved into the portal mesenchyme (Figure 1E). The mature IBDs were mature duct similar to adult IBDs (Figure 1F). These presses progressed from the hilar IBDs to peripheral IBDs. The periods of these processes in the portal tracts near the hepatic hilus were as follows: the DP stages, 7-10 gestational weeks; the remodeling DP stage 11-15 gestational weeks; the remodeled DP stage, 16-24 gestational weeks; the mature IBDs stage, 26-40 weeks. However, these stage periods differed from case to case as well as from portal tract to another. Interestingly, differentiation of intrahepatic peribiliary glands and pancreatic acinar cells from the remodeling and remodeled DP were seen frequently and occasionally in the hilar areas, respectively.
Figure 1.
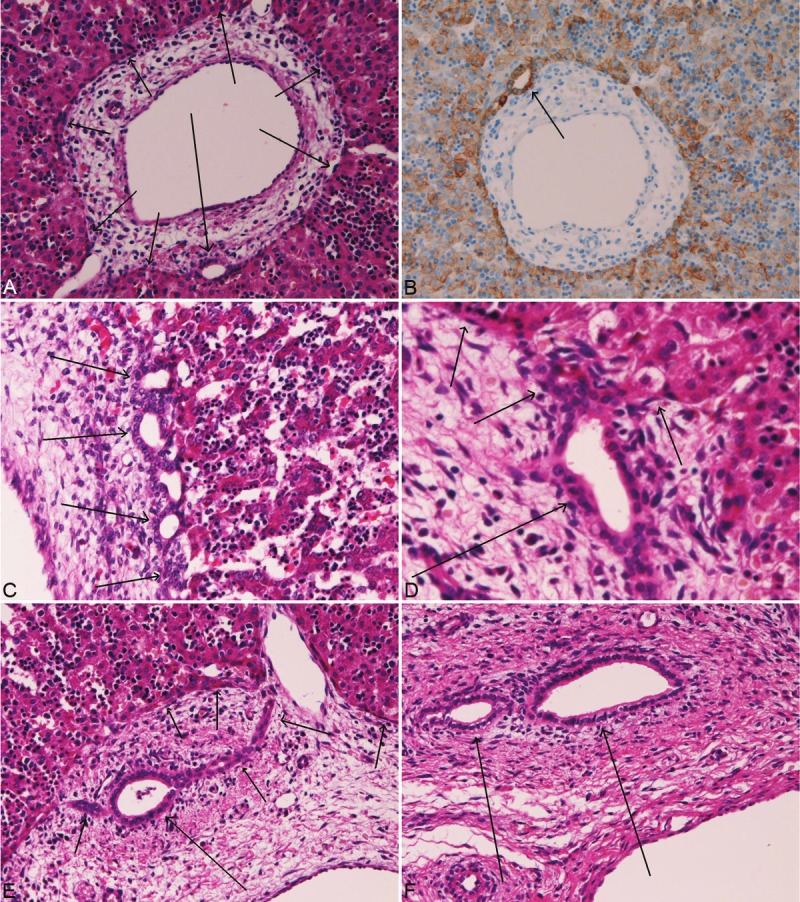
The HE histologies of ductal plate and intrahepatic biliary cells in human fetal livers. The ductal plate (A, short arrows) is composed of single or double-layered cylindrical structures located in the interface between hepatoblast and mesenchyme of the portal tract. A tubular structure (long arrow) of future bile duct is seen in the ductal plate. Immunostaining of cytokeratin 18 highlights the ductal plate (B) and a tubular formation of future bile duct (B, arrow) in the ductal plate. The ductal plate is composed of single or double layered cuboidal cells (arrows) of beaded appearances. The remodeling ductal plate shows disappearance of some cells of the ductal plate (D, short arrows) and development of tubular structures of future bile ducts, which is moving into the portal mesenchyme (D, long arrow). The remodeled ductal plate is characterized by almost disappearance of ductal plate (E, short arrows) and the tubular structure (E, long arrow) moves from the ductal plate (E, short arrows) into the portal mesenchyme (E, long arrow). The mature intrahepatic bile ducts are mature ducts similar to adult intrahepatic bile ducts (F, arrows). A, C, D, and E: HE stain. A: x40. C, D, and E: x200. F: x300.
Mucin immunohistochemistry showed that the cells of DP (Figure 2A and 2B), remodeling DP (Figure 2C and 2D), and remodeled DP (Figure 2E and 2F) were not stained by mucicarmine, PAS, AB at pH2.5, and AB at pH1.0. However, the DP showed magenta products in d-PAS and combined d-PAS/AB at pH2.5 (Figure 2G and 2H). In contrast, the mature bile ducts were positive with all of these techniques (Figure 2I and 2J), indicating the presence of neutral, carboxylated and sulfated mucins. Of particular interest, the cells of DP occasionally positive for acidic mucins as revealed by AB at pH 2.5, AB at pH 1.0, and combined d-PAS/AB pH2.5 (Figure 2K).
Figure 2.
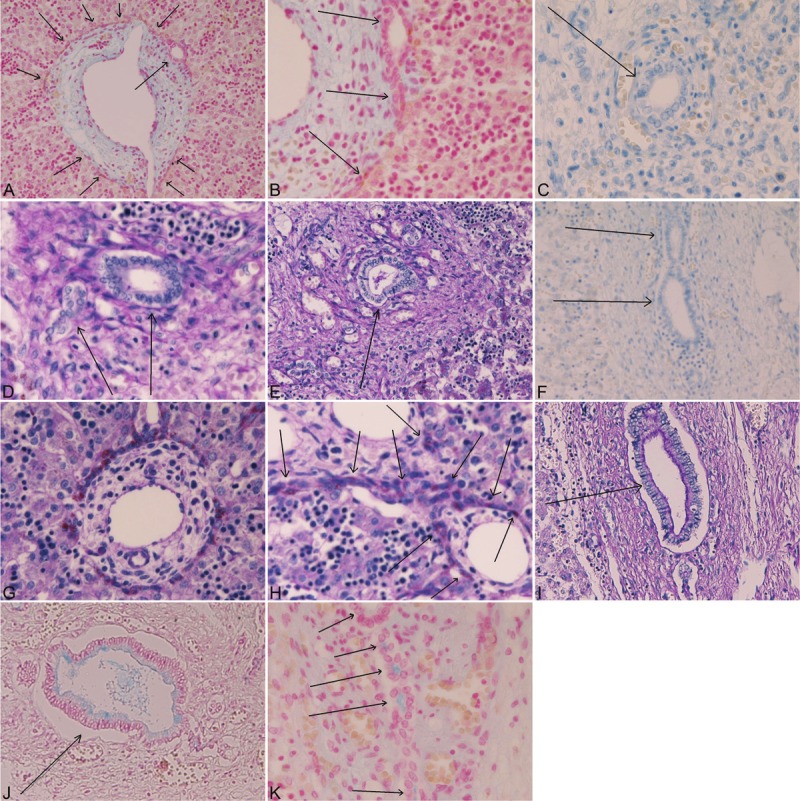
Mucin immunohistochemistry in biliary cells in human fetal livers. The cells of ductal plate (A, B, arrows) are negative for mucins in these figures. The tubular formations of future bile ducts are indicated by two arrows. The cells of remodeling ductal plate indicated by arrows (C, D) are negative for mucins. The cells of remodeled ductal plate indicated by arrows (E, F) are negative for mucins. The ductal plate cells show magenta products in d-PAS and combined d-PAS/AB at pH2.5 (G, H, arrows). The mature intrahepatic bile ducts (arrows) are positive for neutral and acidic mucins (I, J). The cells of ductal plate are occasionally positive for acidic mucinous (K, arrows) in AB techniques (K, arrows). A, B: AB at pH 2.5 stain; A: x40; B: x100. C: mucicarmine stain, x200. D: combined d-PAS/AB at pH2.5 stain, x200. E: d-PAS technique, x200. F: mucicarmine technique, x200. G: d-PAS technique, x100. H: d-PAS technique, x400. I: d-PAS technique, x100. J: AB at pH2.5 technique, x100. K: AB at pH2.5 technique, x400.
Immunohistochemistry for cytokeratin revealed that the cells of DP, remodeling DP, remodeled DP, and mature IBDs were strongly positive for CK8 (Figure 3A) and CK18 (Figure 3B), while they were weakly positive for CK7 (Figure 3C) and CK19 (Figure 3D). Of particular interest, CK7 and CK19, both of cholangiocytic lineage antigens, were very faintly expressed in the periportal primitive hepatocytes near the DP (Figure 3C and 3D).
Figure 3.
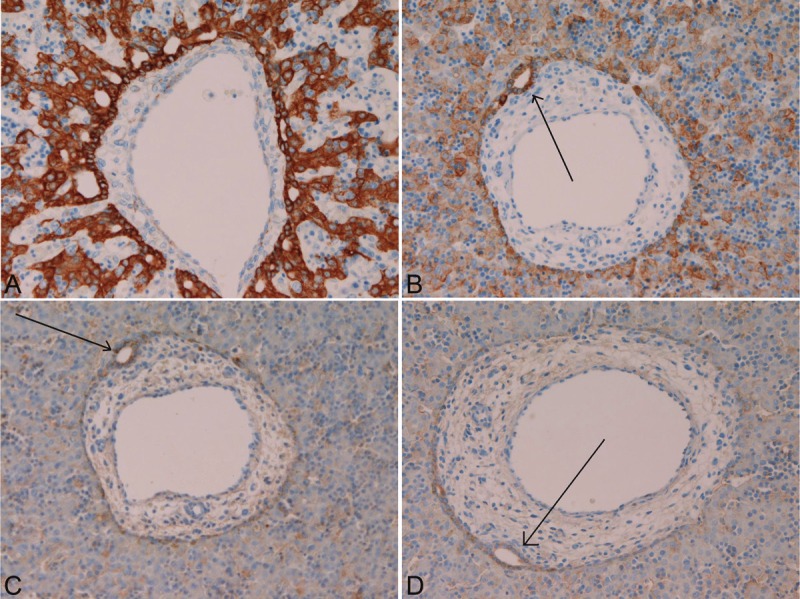
Immunohistochemistry for cytokeratins in human fetal livers. The cells of ductal plate (A, B, C, and D), remodeling ductal plate, remodeled ductal plate, and mature intrahepatic bile ducts are strongly positive for cytokeratin 8 (A) and 18 (B), while they were weakly positive for cytokeratin 7 (C) and cytokeratin 19 (D). A, B, C, and D: x100. Arrows in B, C and D indicate tubular formations of future bile duct in the ductal plate of human fetal livers.
Immunohistochemistry of MUC apomucins showed that MUC1 was expressed in the DP (Figure 4A), remodeling DP (Figure 4B), remodeled DP (Figure 4C), and mature bile ducts (Figure 4D). MUC2 was always negative in the human fetal cholangiocytes. MUC5AC was expressed in the DP (Figure 5A), but not in the remodeling DP (Figure 5B), the remodeled DP (Figure 5C), and mature bile ducts (Figure 5D). MUC6 was expressed in the DP (Figure 6A), but not in the remodeling DP (Figure 6B), the remodeled DP (Figure 6C), and mature bile ducts (Figure 6D). The expression pattern of these MUC apoproteins were mainly luminal staining, but occasionally cytoplasmic staining, particularly in the DP.
Figure 4.
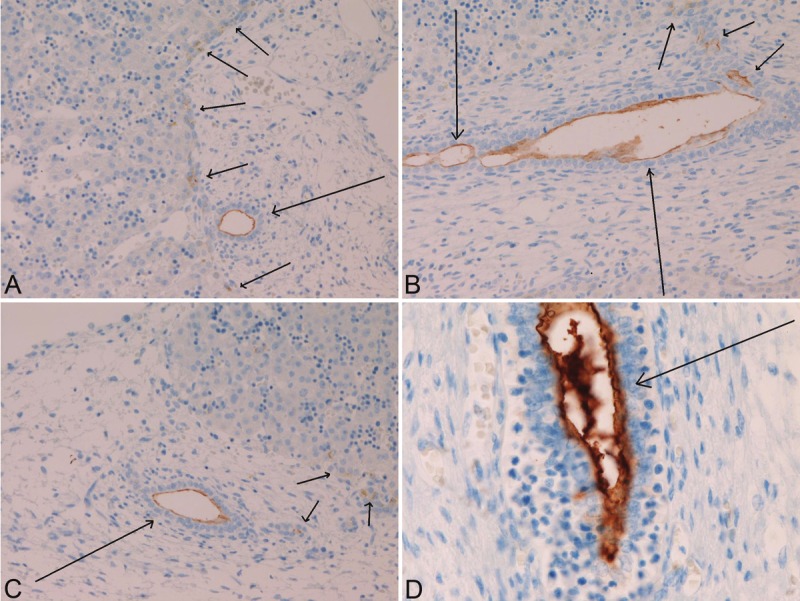
Immunohistochemistry of MUC1 apomucin in the biliary element of human fetal livers. MUC1 is always expressed in cells of ductal plate (A, short arrows). The tubular formation of future bile duct is indicated by long arrows in A. MUC1 is positive in cells of ductal plate remodeling stages (A). Ductal plate cells (short arrows) and migrating future duct element (long arrow) (B). MUC 1 is positive in cells of remodeled stage of the ductal plate (C, arrow). The mature bile duct (arrow) is strongly positive for MUC1.
Figure 5.
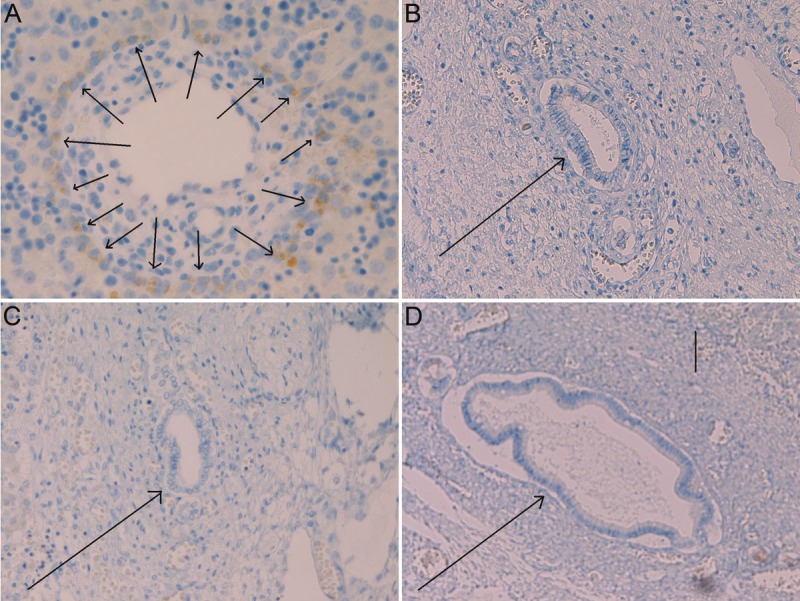
Immunohistochemistry of MUC5AC apomucin in the biliary element of human fetal livers. MUC5AC is expressed in cells of the ductal plate (A, arrows), but MUC5AC is negative for cells of ductal plate remodeling stages (B, arrow), is negative for cells of remodeled stage of ductal plate (C, arrow), and is negative for mature bile duct (D, arrow) in human fetal livers. A: x350. B: x 200. C: x 150. D: x80.
Figure 6.
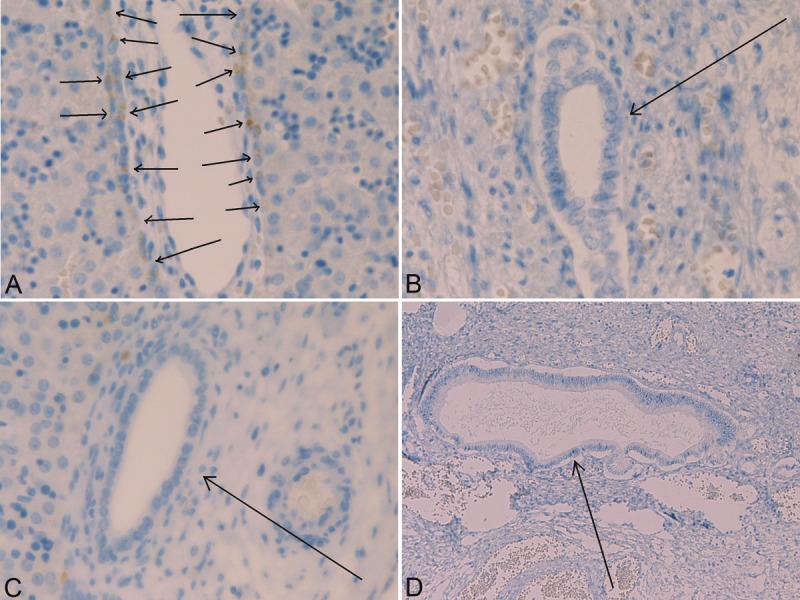
Immunohistochemistry of MUC6 apomucin in the biliary element of human fetal livers. MUC6 is expressed in cells of the ductal plate (A, arrows), but MUC6 is negative for cells of ductal plate remodeling stages (B, arrow), is negative for cells of remodeled stage of ductal plate (C, arrow), and is negative for mature bile duct (D, arrow) in human fetal livers. A: x350. B: x200. C: x150. D: x80.
Discussion
The present study was confirmed the presence of DP in the human fetal livers. The DP was composed of single or doubly-layers cylindrical structures located in the interface between primitive hepatocytes (hepatoblast) and portal mesenchyme. The author confirmed that biliary elements of future IBDs moved to mesenchyme and become mature IBDs. In contrast, the remnant cells of DP disappeared probably by apoptosis [7]. Although Carpentier et al [55] stressed that they never identified apoptosis by TUNEL method in mouse DP, the presence of apoptosis has been observed in human normal fetal livers, fetal liver of various animals, and diseased human fetal and infantile livers [7,56,57]. In the present study, it was found that the processes of IBD development in human fetus can be categorized into the following four stages: DP stage, remodeling DP stage, and remodeled DP stage, and mature IBDs stage. This categorization appears new in the human IBD development. The author also observed that the human IBD development started from the hilar portal areas to the peripheral portal tracts: the stages are early in IBD development in hilar portal tracts and late in IBD development in peripheral portal tracts. This appears a new finding. The author also demonstrated the gestational periods of the each process of IBD development in human. This is a new finding. The author observed that intrahepatic peribiliary glands were developed from the DP and remodeling DP. The author observed that this phenomenon always occurs in hilar portal tracts [1], but this phenomenon has not been described in other researchers. This report is the third reports of such a phenomenon [1,5]. Intrahepatic peribiliary glands were discovered by the author [24], and the author has reported pathologic alterations normal microstructure of the intrahepatic peribiliary glands [25-36]. The author wants to stress that intrahepatic peribiliary glands are actually derived from remodeling DP and remodeled DP near the hepatic hilus. There were many abnormalities of the intrahepatic peribiliary glands [25-36]. Although not recognized widely, there were also DP malformations involving the intrahepatic peribiliary glands. Such DP malformations of the peribiliary glands may be derived from the abnormal development of the peribiliary glands. The author also identified that pancreatic acinar cells developed from the remodeling DP and remodeled DP, although it occurrence was relatively infrequent. This phenomenon was discovered by the author [1,5,6], but has not been observed by other researchers. This is the third description [1,5,6]. Two reports by the author demonstrated that human adult livers occasionally contain areas of clusters of pancreatic acinar cells [31,58]. The author examined 1000 adult livers taking five sections from each liver, and founded that 50 cases (5%) contained pancreatic acinar cells which were confirmed by electron microscopy and immunohistochemical demonstration of pancreatic digestive enzymes [31]. In that study [31], the author used only five sections in the 1000 livers, suggesting that the percentage of the presence of pancreatic acinar cells is far higher than 5% reported by the author [31]. The ectopic pancreatic acinar cells never contained Langerhans islands [31], suggesting that the fetal differentiation of exocrine and endocrine pancreas is different from each another. The present findings highly suggest that pancreatic acinar cells in the normal adult livers develop from DP and matured in adult livers. The presence of pancreatic acinar cells in the human adult livers has been reported twice [58,59]. Wolf et al suggested that this phenomenon is metaplastic process [58]. However, the author believes that this phenomenon is derived from DP with pancreatic acinar differentiation. The occurrence of pancreatic-type carcinomas has been experienced. In such a phenomenon, it can be very probable that ectopic pancreatic acinar cells developed from the DP of human fetal livers undergo malignant transformation, thus giving rise to primary pancreatic carcinoma occurring in the liver. In any way, the author want to stress that pancreatic acinar cells are differentiated from remodeling DP and remodeled DP in human fetal livers.
CK is one of the intermediate filaments predominantly present in epithelial cells [60,61]. At least 20 well-defined subclasses of CK have been identified on the basis of the molecular weight and isoelectric pH value as reported by Moll [60,61]. Investigation of CK should be done with an aid of Moll’s CK catalogues [61]. In the human liver, there have been no comprehensive studies of CK examining all types of CKs of Moll’s catalogue. Most CK studies of the liver focused on CK7, CK8, CK18, and CK19, and other types of CK have not been examined. In the present study, the author also investigated only these four CKs. However, the author is now investigating at least 15 CK expressions in human fetal liver. In general, it is believed that cholangiocytes express CK7, CK8, CK18 and CK19, while hepatocytes express CK8 and CK18. Thus, CK7, CK8, CK18 and CK19 are markers of cholangiocytes, and CK8 and CK18 of the hepatocyte lineage. In the present study, human fetal cholangiocytes including DP expressed CK7, CK8, CK18, and CK19, and human fetal primitive hepatocytes expressed CK8 and CK18. The expressions of CK8 and CK18 in fetal cholangiocytes including DP were very strong, while the expressions of CK7 and CK19 were very weak. This phenomenon strongly suggests that most of CK in human fetal cholangiocytes including DP are CK8 and CK18, and that human fetal cholangiocytes have a small amount of CK7 and CK19. Of particular interest, the primitive hepatocytes near the portal tracts expressed biliary-type CKs, i.e, CK7 and CK19. Recently, Carpentier et al [55] suggested that mouse DP has potential to differentiate into periportal hepatocytes. The present findings of very weak expressions of CK7 and CK19 in the periportal primitive hepatocytes also support the suggestion of Carpentier et al [55]. Thus, the DP of human fetal livers may give rise to or differentiate into periportal hepatocytes. In the present study, the CK study was performed to identify cholangiocytes including DP in human fetal livers. In any way, the present study could identify DP and related cholangiocytes in the human fetal liver, and were extremely useful for the study of MUC apomucins and mucin histochemical studies. Van Eyken et al [17] reported that the primitive hepatocytes in the human fetal livers were consistently positive for CK8 and 18. They also demonstrated that the biliary cells expressed CK7 and CK19, in addition to CK8 and CK18, although CK19 appeared from the early process of human fetal IBD development while CK7 expression emerged relative late stage (after 20 gestational weeks) in the human fetal IBD development. Van Eyken et al [17] used frozen sections because they could not use reliable CK antibodies in the relatively old era. Now, many kinds of CK can be analyzed in reliable monoclonal antibodies which can be useful in formalin-fixed and paraffin-embedded sections. The present study showed strong expression of CK8 and CK18 in the primitive hepatocytes (hepatoblast) and primitive IBD cells in human fetal livers. The author also showed weak expression of CK7 and CK19 in the primitive IBD including DP in human fetal livers. Van Eyken et al [17] stressed that the expression of CK7 was not seen in early (<20 gestational weeks) human IBD development. However, the present study showed that the CK7 was expressed in early DP of 8 weeks and later weeks. This difference may be due to the different antibodies used. It seems that the data of present study is more reliable than those of Van Eyken et al [17]. Thus, the present findings suggest that CK7, CK8, CK18 and CK19 are expressed in the DP, remodeling DP, remodeled DP, and mature IBDs in human fetal livers.
The most important and main findings of the present study are expression status of MUC apomucins and histochemical analysis of mucins. As is well known, mucins are very high molecular weight glycoproteins which are very important in the protection, local innate immunity, and lubrication of mucosal surface of various organs. Mucins are also involved in the pathogenesis of benign and malignant diseases of secretory epithelial cells [48,49]. It is well known that MUC expression is down-regulated or up-regulated in most malignant neoplasms of various organs [48,49]. These alterations of MUC apomucins, which are regulated by MUC genes, are thought to be associated with carcinogenesis and malignant potentials of cancer, though the mechanisms remain unclear [50-52]. Mucins are structurally classified into the protein backbone called mucin core protein or MUC apomucins and the numerous glycosylated side chains [50-52]. The immunohistochemical study of MUC apomucins analyses the status of protein backbone, while mucin histochemistry the status of carbohydrate side chains of mucins. Thus, it is very reasonable to study the MUC apomucins and mucin histochemistry together.
In the liver, only a limited number of studies of a limited number of MUC profiles have been performed [62-66]. MUC apomucins consist of more than 20 MUC apomucin types [50-52]. However, the studies of MUC apomucins of the liver employed only a few MUC apomucins [62-66], and even the normal expression pattern of all the MUC apomucins has not been studied. According to Sasaki et al, who examined MUC1, MUC2, MUC3, MUC5 and MUC6, showed that only MUC3 is present in the biliary cells of normal adult livers [63-66]. The expressions of several MUC apomucins are known to alter in various hepatobiliary disease including cholangiocarcinoma, combined hepatocellular cholangiocarcinoma, cystic liver disease, viral hepatitis, primary biliary cirrhosis, extrahepatic biliary obstruction, biliary papillomatosis, intraductal papillary neoplasms of the biliary system, and hyperplastic, dysplastic, and neoplastic biliary epithelium of hepatolithiasis [62-66]. That is, biliary cells frequently show aberrant expression of some MUC apomucins in neoplastic, preneoplastic, inflammatory, autoimmune, cystic, hepatolithiasis, and cholangiocyte injury disease. These authors suggested the aberrant expression of a few MUC apomucins may be associated with carcinogenesis, malignant potential, tumor behaviors, bile duct injury, congenital IBD diseases, and degree of inflammation. However, this suggested association between MUC apomucins and liver disease remains unknown [62-66].
The MUC apomucins expression in the human fetal cholangiocytes including DP has been studied only once by Sasaki and the author [13]. We immunohistochemically studied the expression of MUC1, MUC2, MUC3, and MUC5/6 during IBD development, and found that MUC1 was consistently expressed mainly in the luminal side of remodeling DP, remodeled DP, and mature IBDs in human fetal livers. The MUC1 was absent in DP and adult IBDs [13]. In contrast, expressions of MUC2, MUC3 and MUC5/6 were consistently negative in human fetal livers [13]. The human postnatal and human adult livers, expression of MUC3 was seen, but expressions of MUC2 and MUC5/6 were absent in the human postnatal livers and adult livers [13]. Thus, class switch transition was recognized from MUC1 to MUC3 after the birth [13]. This characteristic transition was similar to the changes in the hepatocellular expression of alpha-fetoprotein and albumin during the perinatal period [13].
In the present study, the MUC apomucins profile showed that MUC1 (polymorphic epithelial mucin, transmembranous type) was expressed in the DP, remodeling DP, remodeled DP, and mature bile ducts. MUC2 (goblet cell mucin, secreted type) was always negative in the human fetal cholangiocytes. MUC5AC (gastic foveolar mucin, secreted type) was expressed in the DP, but not in the remodeling DP, the remodeled DP, and mature bile ducts. MUC6 (pyloric gland-type mucin, secreted mucin) was expressed in the DP, but not in the remodeling DP, the remodeled DP, and mature bile ducts. The expression pattern of these MUC apoproteins were mainly luminal staining, but occasionally cytoplasmic staining, particularly in the DP. These findings suggest that MUC2 apomucin is entirely absent from the cholangiocytes including DP in human fetal livers, and imply that the human fetal cholangiocytes do not show differentiation into goblet cells seen in the intestine and intestinal metaplasia of the stomach. It was particularly of interest, MUC1 apomucin was expressed in any stages of biliary developmental structures in human livers. MUC1 is seen even in the DP. The consistent presence of MUC1 may suggest that MUC1 apomucin may be associated with normal development of fetal cholangiocytes. The similar observations were once made by Sasaki and the author [13] using different antibodies. The present study of MUC1 apomucin is mildly different from our previous study [13], which showed no expression of MUC1 in the DP. The present study showed MUC1 in DP. The difference may be due to the different antibodies used. The antibodies of our present study were made by Dr. Kim YS [13]. Of particular interest in the current study is that MUC5AC and MUC6 are expressed in the DP, but not in the remodeling DP, remodeled DP, and mature IBDs. These are new findings. The data suggest that the MUC5AC and MUC6 apomucins may be associated with morphogenesis of DP, but not associated with remodeling and remodeled processes of the DP and mature IBD development in human fetus IBD development. The findings also suggest that the DP may have immunophenotypes similar to gastric foveolar cells and pyloric gland-type cells. In fact, the author reported that gastric foveolar type epithelium and gastric pyloric glands type epithelium were frequently found in proliferated biliary cells of hepatolithiasis [24,27,29]. Thus, it is apparent that the expression of MUC5AC and MUC6 in human fetal DP is associated with proliferation of biliary and peribiliary glands with gastric epithelial differentiation. It is very probable that the pronounced proliferation of biliary cells and peribiliary glands seen in hepatolithiasis may recapitulate the fetal DP phenotype, and they may be derived from hepatic stem/progenitor cells forming DP as demonstrated in mouse by Carpentier et al [55]. These findings seem very reasonable. The absence of MUC5AC and MUC6 in remodeling DP, remodeled DP, and mature IBDs in fetal liver show that these MUC apomucins expression is not associated with morphogenesis of primitive fetal cholangiocytes after the DP formation. The expression patterns of MUC5AC and MUC6 are different from those of our previous study [13], which showed no expression of MUC5/6 in the IBD development including DP in human fetus. This difference seems due to the different antibodies used. The antibodies of the four MUC apomucins are obtained from Novacastra Laboratory and their sensitivity and specificity are most reliable in the current era. Probably, the antibodies used in the current study are superior to those of our previous study [13]. However, the present study was limited, because, like other reports [13,62-66], only a several MUC apomucins were studied and the expression of other many MUC apomucins was unknown in fetal IBD development. The more comprehensive studies examining many MUC apomucins status in human fetal IBD development remain to be performed in future.
With regards to carbohydrate side chains of mucins, the author has demonstrated that the carbohydrate residues of mucin glycoprotein were scant in the early stage of fetal IBD development, while they were abundant in the latest stage of human fetal IBD development [2]. Namely, the carbohydrate residues increased proportionally along with the process of IBD development in human fetus [2]. The author also examined the mucins profile in the fetal development of intrahepatic peribiliary glands with the same techniques as the present study [1]. The author showed remodeled DP and mature intrahepatic peribiliary glands in human fetal livers showed neutral, carboxylated, and sulfated mucins [1]. The DP and remodeling DP of fetal intrahepatic peribiliary glands did not show no mucins [1]. As is well known, mucins are histochemically classified into neutral mucins, carboxylated mucins, and sulfated mucins. PAS yield magenta color of glycogens and neutral mucins [1,24,27]. D-PAS indicates the presence of neutral mucins. AB at pH 2.5 and pH1.0 shows blue colors and demonstrate the presence of carboxylated mucins and sulfated mucins [1,24,27], respectively. Combined d-PAS/AB at pH2.5 shows both neutral mucins and carboxylated mucins and combined d-PAS/AB at pH1.2 neutral mucins and sulfated mucin. The mucicarmine stain shows so-called mucins of uncertain molecular characteristics, and this mucin stain is less sensitive and specific than other mucin stains [1,24,27].
In the present study, mucin immunohistochemistry showed that the cells of DP, remodeling DP, and remodeled DP were not stained by mucicarmine, PAS, AB at pH2.5, AB at pH1.0, and combined techniques. However, the DP showed magenta products in d-PAS and combined d-PAS/AB at pH2.5. These findings suggest that DP cells may contain neutral mucins, and that no mucins are present in remodeling DP and remodeled DP. Of particular interest in the present study, the cells of DP occasionally positive for acidic mucins (both carboxylated and sulfated mucins) as revealed by AB at pH 2.5, AB at pH 1.0, and combined d-PAS/AB pH2.5 and pH1.0. This finding suggests that DP has carboxylated and sulfated mucins in addition to neutral mucins. The author demonstrated that the human DP has MUC1, MUC5AC and MUC6 apomucins. Therefore, the mucin proteins of these MUC subtype are present in human fetal DP. Thus, the presence of neutral, carboxylated and sulfated mucin carbohydrate residues in the DP is very reasonably. In contrast, the author showed that the human remodeling DP and remodeled DP showed no secretory MUC apomucins (MUC5AC and MUC6). Therefore, the secreted mucin proteins of these MUC subtype are absent in human fetal IBD development. Thus, the absence of neutral, carboxylated and sulfated mucin carbohydrate residues in the remodeling DP and remodeled DP is very reasonable, because no corresponding mucin core proteins (MUC apomucins) were absent in these structures. In contrast, the mature bile ducts were positive with all of these techniques of mucin histochemistry. These findings suggest the presence of neutral, carboxylated and sulfated mucin residues of mucins in mature IBDs in human fetal livers. However, there were no expressions of secretory mucin core proteins (MUC5AC and MUC6) in mature IBDs in the fetal lever. This discrepancy was reasonably explained. Namely, probably, the mature IBD in fetus liver have other secretory apomucins with carbohydrate residues of neutral, carboxylated and sulfated mucins. This explanation is very reasonable. Finally, the author wants to stress that the increase of mucins detected by histochemical techniques indicates the gradually increased carbohydrate component of mucins in human fetal IBD development, as proved previously by the author [2]. In any way, much more studies using many MUC apomucins and development of new histochemical techniques remains to be performed.
In summary, the author herein immunohistochemically investigated the expression protein portions of mucins of MUC1, MUC2, MUC5AC, and MUC6, and histochemically investigated carbohydrate component of mucins in human fetal cholangiocytes with the use of 32 human fetal livers of various gestational ages. MUC1 is a transmembranous apomucin, while MUC2, MUC5AC and MUC6 are secretory apomucins. Under normal conditions, MUC1 (polymorphic epithelial mucin) is present in the pancreatic epithelium. MUC2 (goblet cell mucin) is located in goblet cells. MUC5AC (gastric foveolar mucin) and MUC6 (pyloric gland-type mucin) are located in the stomach. In the present study, the processes of the human IBD development could be categorized into four stages; ductal plate (DP), remodeling DP, remodeled DP, and mature IBDs. The author identified that MUC1 was consistently present in ductal plate (DP), remodeling DP, remodeled DP, and mature IBD in human fetal livers. MUC5AC and MUC6 were present only in the DP. MUC5AC and MUC6 were absent in remodeling DP, remodeled DP, and mature IBD in human fetal livers. No expression of MUC2 was seen throughout the fetal IBD development. Histochemically, no carbohydrate component of mucins were seen in the remodeling DP and remodeled DP, while neutral and acidic mucins (carboxylated and sulfated mucins) were seen in mature IBD in human fetal livers. The DP showed frequently neutral mucins and less frequently acidic mucins (carboxylated and sulfated mucins residue). These findings suggest that the DP cells have MUC1, MUC5AC and MUC6, and that remodeling DP, remodeled DP, and mature IBDs have MUC1, but not MUC5AC and MUC6. The presence of neutral and acidic carbohydrates in DP suggests that these carbohydrates of mucin are attached to the MUC5AC and MUC6 mucin core proteins. Although the implications are unclear, the expression of these MUC apomucins and their carbohydrate residues are associated with normal development of IBDs in human fetal livers.
Conflict of interest statement
The author has no conflict of interest.
References
- 1.Terada T, Nakanuma Y. Development of human intrahepatic peribiliary glands: Histological, keratin immunohistochemical and mucus histochemical analyses. Lab Invest. 1993;68:261–269. [PubMed] [Google Scholar]
- 2.Terada T, Nakanuma Y. Profiles of expression of carbohydrate chain structures during human intrahepatic bile duct development and maturation: a lectin-histochemical and immunohistochemical study. Hepatology. 1994;20:388–397. [PubMed] [Google Scholar]
- 3.Terada T, Kitamura Y, Nakanuma Y. Normal and abnormal development of the intrahepatic biliary system: A review. Tohoku J Exp Med. 1997;181:19–32. doi: 10.1620/tjem.181.19. [DOI] [PubMed] [Google Scholar]
- 4.Terada T, Ashida K, Kitamura Y, Matsunaga Y, Takashima K, Kato M, Ohta T. Expression of E-cadherin, alpha-catenin and beta-catenin during human intrahepatic bile duct development. J Hepatol. 1998;28:263–269. doi: 10.1016/0168-8278(88)80013-8. [DOI] [PubMed] [Google Scholar]
- 5.Terada T. Differentiation of intrahepatic peribiliary glands and pancreatic acinar cells from the remodeling ductal plate in human fetuses. Hepatology. 2012;56:2004–2005. doi: 10.1002/hep.25750. [DOI] [PubMed] [Google Scholar]
- 6.Terada T, Nakanuma Y. Expression of pancreatic enzymes (α-amylase, trypsinogen and lipase) during human liver development and maturation. Gastroenterology. 1995;108:1236–1245. doi: 10.1016/0016-5085(95)90225-2. [DOI] [PubMed] [Google Scholar]
- 7.Terada T, Nakanuma Y. Detection of apoptosis and expression of apoptosis-related proteins during human intrahepatic bile duct development. Am J Pathol. 1995;146:67–74. [PMC free article] [PubMed] [Google Scholar]
- 8.Terada T, Okada Y, Nakanuma Y. Expression of matrix proteinases during human intrahepatic bile duct development: A possible role in biliary cell migration. Am J Pathol. 1995;147:1207–1213. [PMC free article] [PubMed] [Google Scholar]
- 9.Kato M, Shinozawa T, Kato S, Terada T. Divergent expression of midkine in the human fetal liver and kidney: an immunohistochemical analysis of developmental changes in hilar primitive bile ducts and hepatocytes. Liver. 2000;20:475–481. doi: 10.1034/j.1600-0676.2000.020006475.x. [DOI] [PubMed] [Google Scholar]
- 10.Kato M, Shinozawa T, Kato S, Terada T. Immunohistochemical localization of truncated midkine in developing human bile ducts. Histol Histopathol. 2003;18:129–134. doi: 10.14670/HH-18.129. [DOI] [PubMed] [Google Scholar]
- 11.Terada T, Nakanuma Y. Development of human peribiliary capillary plexus: A lectin-histochemical and immunohistochemical study. Hepatology. 1993;18:529–536. [PubMed] [Google Scholar]
- 12.Terada T, Nakanuma Y. Expression of tenascin, type IV collagen and laminin during human intrahepatic bile duct development and in intrahepatic cholangiocarcinoma. Histopathology. 1994;25:143–150. doi: 10.1111/j.1365-2559.1994.tb01570.x. [DOI] [PubMed] [Google Scholar]
- 13.Sasaki M, Nakanuma Y, Terada T, Kim YS. Biliary epithelial expression of MUC1, MUC2, MUC3, and MUC5/6 apomucins during human intrahepatic bile duct development and maturation: An immunohistochemical study. Am J Pathol. 1995;147:574–579. [PMC free article] [PubMed] [Google Scholar]
- 14.Terada T, Kato M, Horie S, Endo K, Kitamura Y. Expression of pancreatic alpha-amylase protein and messenger RNA on hilar primitive bile ducts and hepatocytes during human fetal liver organogenesis: an immunohistochemical and in situ hybridization study. Liver. 1998;18:313–319. doi: 10.1111/j.1600-0676.1998.tb00811.x. [DOI] [PubMed] [Google Scholar]
- 15.Terada T, Ukita Y, Ueyama J, Ohta T. Protein expression of double-stranded RNA-activated protein kinase (PKR) in intrahepatic bile ducts in normal adult livers, fetal livers, primary biliary cirrhosis, hepatolithiasis and intrahepatic cholangiocarcinoma. Liver. 2000;20:450–457. doi: 10.1034/j.1600-0676.2000.020006450.x. [DOI] [PubMed] [Google Scholar]
- 16.Terada T, Ohta T, Nakanuma Y. Expression of transforming growth factor-α and its receptor during human liver development and maturation. Virchows Archiv. 1994;424:669–675. doi: 10.1007/BF00195783. [DOI] [PubMed] [Google Scholar]
- 17.Van Eyken P, Sciot R, Callea F, Van der Steen K, Moerman P, Desmet VJ. The development of the intrahepatic bile ducts in man: a keratin immunohistochemical study. Hepatology. 1988;8:1586–1595. doi: 10.1002/hep.1840080619. [DOI] [PubMed] [Google Scholar]
- 18.Desmet VJ. Congenital disease of intrahepatic bile ducts: variations on the theme “ductal plate malformation”. Hepatology. 1992;16:1069–1083. doi: 10.1002/hep.1840160434. [DOI] [PubMed] [Google Scholar]
- 19.Desmet VJ. Intrahepatic bile duct under the lens. J Hepatol. 1985;1:545–559. doi: 10.1016/s0168-8278(85)80752-2. [DOI] [PubMed] [Google Scholar]
- 20.Roskams T, Desmet V. Embryology of extra- and intrahepatic bile ducts, the ductal plate. Anat Rec (Hoboken) 2008;291:628–635. doi: 10.1002/ar.20710. [DOI] [PubMed] [Google Scholar]
- 21.Shah KD, Gerber MA. Development of intrahepatic bile ducts in humans: immunohistochemical study using monoclonal cytokeratin antibodies. Arch Pathol Lab Med. 1989;113:1135–138. [PubMed] [Google Scholar]
- 22.Haruna Y, Saito K, Spaulding S, Nalesnik MA, Gerber MA. Identification of bipotential progenitor cells in human liver development. Hepatology. 1996;23:476–481. doi: 10.1002/hep.510230312. [DOI] [PubMed] [Google Scholar]
- 23.Shar KD, Gerber MA. Development of intrahepatic bile ducts in humans: possible role of laminin. Arch Pathol Lab Med. 1990;114:597–600. [PubMed] [Google Scholar]
- 24.Terada T, Nakanuma Y, Ohta G. Glandular elements around the intrahepatic bile ducts in man: their morphology and distribution in normal livers. Liver. 1987;7:1–8. doi: 10.1111/j.1600-0676.1987.tb00308.x. [DOI] [PubMed] [Google Scholar]
- 25.Terada T, Nakanuma Y. Solitary cystic dilation of the intrahepatic bile duct: Morphology of two autopsy cases and a review of the literature. Am J Gastroenterol. 1987;82:1301–1305. [PubMed] [Google Scholar]
- 26.Terada T, Takegoshi T, Doishita K, Nakanuma Y. Histological study of intrahepatic cavernous transformation in a patient with primary myelofibrosis and portal venous thrombosis. Virchows Arch A Pathol Anat Histopathol. 1988;412:339–345. doi: 10.1007/BF00750260. [DOI] [PubMed] [Google Scholar]
- 27.Terada T, Nakanuma Y. Morphological examination of intrahepatic bile ducts in hepatolithiasis. Virchows Arch A Pathol Anat Histopathol. 1988;413:167–176. doi: 10.1007/BF00749679. [DOI] [PubMed] [Google Scholar]
- 28.Terada T, Ishida F, Nakanuma Y. Vascular plexus around intrahepatic bile ducts in normal livers and portal hypertension. J Hepatol. 1989;8:139–149. doi: 10.1016/0168-8278(89)90001-9. [DOI] [PubMed] [Google Scholar]
- 29.Terada T, Nakanuma Y. Pathologic observations of intrahepatic peribiliary glands in 1,000 consecutive autopsy livers: IV. Hyperplasia of intramural and extramural glands. Hum Pathol. 1992;23:483–490. doi: 10.1016/0046-8177(92)90124-l. [DOI] [PubMed] [Google Scholar]
- 30.Terada T, Nakanuma Y. Innervation of intrahepatic bile ducts and peribiliary glands in normal livers, extrahepatic biliary obsrtruction and hepatolithiasis: an immunohistochemical study. J Hepatol. 1989;9:141–148. doi: 10.1016/0168-8278(89)90044-5. [DOI] [PubMed] [Google Scholar]
- 31.Terada T, Nakanuma Y, Kakita A. Pathologic observations of intrahepatic peribiliary glands in 1,000 consecutive autopsy livers: Heterotopic pancreas in the liver. Gastroenterology. 1990;98:1333–1337. doi: 10.1016/s0016-5085(12)90353-4. [DOI] [PubMed] [Google Scholar]
- 32.Terada T, Nakanuma Y. Pathological observations of intrahepatic peribiliary glands in 1,000 consecutive autopsy livers: II. A possible source of cholangiocarcinoma. Hepatology. 1990;12:92–97. doi: 10.1002/hep.1840120115. [DOI] [PubMed] [Google Scholar]
- 33.Terada T, Nakanuma Y. Pathological observations of intrahepatic peribiliary glands in 1,000 consecutive autopsy livers: III. Survey of necroinflammation and cystic dilatation. Hepatology. 1990;12:1229–1233. doi: 10.1002/hep.1840120525. [DOI] [PubMed] [Google Scholar]
- 34.Terada T, Kono N, Nakanuma Y. Immunohistochemical and immunoelectron microscopical analyses of α-amylase isozymes in the intrahepatic biliary epithelium and hepatocytes. J Histochem Cytochem. 1992;40:1627–1635. doi: 10.1177/40.11.1431051. [DOI] [PubMed] [Google Scholar]
- 35.Terada T, Nakanuma Y. Intrahepatic cholangiographic appearance simulating primary sclerosing cholangitis in hepatobiliary diseases: A postmortem cholangiographic and histolopathological study in 154 autopsy livers. Hepatology. 1995;22:75–81. [PubMed] [Google Scholar]
- 36.Terada T, Kida T, Nakanuma Y. Extrahepatic peribiliary glands express α-amylase isozymes, trypsin and pancreatic lipase: An immunohistochemical analysis. Hepatology. 1993;18:803–808. doi: 10.1002/hep.1840180409. [DOI] [PubMed] [Google Scholar]
- 37.Terada T, Nakanuma Y. Congenital biliary dilatation in autosomal dominant adult polycystic disease of the liver and kidneys. Arch Pathol Lab Med. 1988;112:1113–1116. [PubMed] [Google Scholar]
- 38.Jorgensen MJ. The ductal plate malformation. Acta Pathol Microbiol Scand Suppl. 1977;257:1–87. [PubMed] [Google Scholar]
- 39.Summerfield JA, Nagafuchi Y, Sherlock S, Cadafalch J, Scheuer PJ. Hepatobiliary fibropolycystic diseases: a clinical and histological review of 51 patients. J Hepatol. 1986;2:141–156. doi: 10.1016/s0168-8278(86)80073-3. [DOI] [PubMed] [Google Scholar]
- 40.Terada T, Moriki T. Monolobar ductal plate malformation disease of the liver. Pathol Int. 2010;60:407–12. doi: 10.1111/j.1440-1827.2010.02535.x. [DOI] [PubMed] [Google Scholar]
- 41.Terada T, Moriki T. Monolobar hepatobiliary fibropolycystic disease. Pathol Oncol Res. 2011;17:159–165. doi: 10.1007/s12253-010-9285-3. [DOI] [PubMed] [Google Scholar]
- 42.Nakanuma Y, Terada T, Ohta G, Matsubara F, Kurachi M. Caroli’s disease in congenital hepatic fibrosis and infantile polycystic disease. Liver. 1982;2:346–354. doi: 10.1111/j.1600-0676.1982.tb00833.x. [DOI] [PubMed] [Google Scholar]
- 43.Desmet VJ. Ductal plates in hepatic ductular reactions: hypothesis and implications. I. Types of ductular reaction reconsidered. Virchows Arch. 2011;458:251–259. doi: 10.1007/s00428-011-1048-3. [DOI] [PubMed] [Google Scholar]
- 44.Terada T. Hepatic nodular hamartoma containing liver cysts, ductal plate malformations and peribiliary glands. Hepatol Res. 2011;41:93–98. doi: 10.1111/j.1872-034X.2010.00746.x. [DOI] [PubMed] [Google Scholar]
- 45.Terada T. Projected focal nodular hyperplasia (FNH) of the liver with pronounced atypical ductular reaction resembling ductal plate and expressing KIT. Hepatol Res. 2012;42:721–726. doi: 10.1111/j.1872-034X.2012.00967.x. [DOI] [PubMed] [Google Scholar]
- 46.Nakanuma Y, Sato Y, Ikeda H, Harada K, Kobayashi M, Sano K, Uehara T, Yamamoto M, Arrizumi S, Park YN, Choi JH, Yu E. Intrahepatic cholangiocarcinoma with predominant “ductal plate malformation” pattern: a new subtype. Am J Surg Pathol. 2012;36:1629–1635. doi: 10.1097/PAS.0b013e31826e0249. [DOI] [PubMed] [Google Scholar]
- 47.Terada T. Mutations and protein expression of KIT and PDGFRA genes in ipsilateral testicular seminomas: an immunohistochemical and molecular genetic study. Appl Immunohistochem Mol Morphol. 2011;19:450–453. doi: 10.1097/PAI.0b013e31820d2872. [DOI] [PubMed] [Google Scholar]
- 48.Itoh Y, Kamata-Sakurai M, Denda-Nagai K, Nagai S, Tsuiji M, Ishii-Schrade K, Okada K, Goto A, Fukayama M, Irimura T. Identification and expression of human epiglycanin/MUC21: a novel transmembrane mucin. Glycobiology. 2008;18:74–83. doi: 10.1093/glycob/cwm118. [DOI] [PubMed] [Google Scholar]
- 49.Andrianifahanana M, Moniaux N, Batra SK. Regulation of mucin expression: mechanistic aspects and implications for cancer and inflammatory diseases. Biochim Biophys Acta. 2006;1765:189–222. doi: 10.1016/j.bbcan.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 50.Byrd JC, Bresalier RS. Mucins and mucin binding proteins in colorectal cancer. Cancer Metastasis Rev. 2004;23:77–99. doi: 10.1023/a:1025815113599. [DOI] [PubMed] [Google Scholar]
- 51.Kocer B, Soran A, Erdogan S, Karabeyoglu M, Yildirim O, Eroglu A, Bozkurt B, Cengiz O. Expression of MUC5AC in colorectal carcinoma and relationship with prognosis. Pathol Int. 2002;52:470–477. doi: 10.1046/j.1440-1827.2002.01369.x. [DOI] [PubMed] [Google Scholar]
- 52.Kocer B, Soran A, Erdogan S, Karabeyoglu M, Yildirim O, Eroglu A, Bozkurt B, Cengiz O. Expression of MUC5AC in colorectal carcinoma and relationship with prognosis. Pathol Int. 2002;52:470–477. doi: 10.1046/j.1440-1827.2002.01369.x. [DOI] [PubMed] [Google Scholar]
- 53.Terada T, Kawaguchi M, Furukawa K, Sekido Y, Osamura Y. Minute mixed ductal-endocrine carcinoma of the pancreas with predominant intraductal growth. Pathol Int. 2002;52:740–746. doi: 10.1046/j.1440-1827.2002.01416.x. [DOI] [PubMed] [Google Scholar]
- 54.Terada T, Kawaguchi M. Primary clear cell adenocarcinoma of the peritoneum. Tohoku J Exp Med. 2005;271:271–275. doi: 10.1620/tjem.206.271. [DOI] [PubMed] [Google Scholar]
- 55.Carpentier R, Suner RS, Van Hul N, Kopp JL, Beaudry JB, Cordi S, Antoniou A, Raynaud P, Lepreux S, Jacquemin P, Leclercq IA, Sander M, Lemaigre FP. Embryonic ductal plate cells give rise to cholangiocytes, periportal hepatocytes, and adult liver progenitor cells. Gastroenterology. 2011;141:1432–1438. doi: 10.1053/j.gastro.2011.06.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Funaki N, Sasano H, Shizawa S, Nio M, Iwami D, Ohi R, Nagura H. Apoptosis and cell proliferation in biliary atresia. J Pathol. 1998;186:429–433. doi: 10.1002/(SICI)1096-9896(199812)186:4<429::AID-PATH195>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 57.Sasaki H, Nio M, Iwami D, Funaki N, Sano N, Ohi R, Sasano H. E-cadherin, alpha-catenin and beta-catenin in biliary atresia: correlation with apoptosis and cell cycle. Pathol Int. 2001;51:923–932. doi: 10.1046/j.1440-1827.2001.01304.x. [DOI] [PubMed] [Google Scholar]
- 58.Wolf HK, Burchette JL Jr, Garcia JA, Michalopoulos G. Exocrine pancreatic tissue in human liver: a metaplastic process. Am J Surg Pathol. 1990;14:590–595. doi: 10.1097/00000478-199006000-00011. [DOI] [PubMed] [Google Scholar]
- 59.Kuo FY, Swanson PE, Yeh MM. Pancreatic acinar tissue in liver explants: a morpgologic and immunohistochemical study. Am J Surg Pathol. 2009;33:66–71. doi: 10.1097/PAS.0b013e31818c8482. [DOI] [PubMed] [Google Scholar]
- 60.Rosai J. Keratins. In: Rosai J, editor. Rosai and Ackermans Surgical Pathology. New York: V Mosby; 2004. pp. 55–56. [Google Scholar]
- 61.Moll R, Divo M, Langbein L. The human keratin: biology and pathology. Histochem Cell Biol. 2008;129:705–733. doi: 10.1007/s00418-008-0435-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Terada T, Ohta T, Sasaki M, Nakanuma Y, Kim YS. Expression of MUC apomucins in normal pancreas and pancreatic tumours. J Pathol. 1996;180:160–165. doi: 10.1002/(SICI)1096-9896(199610)180:2<160::AID-PATH625>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 63.Sasaki M, Nakanuma Y. Expression of mucin core protein of mammary type in primary liver cancer. Hepatology. 1994;20:1192–1197. [PubMed] [Google Scholar]
- 64.Sasaki M, Nakanuma Y. Abnormal expression of MUC1 apomucin and mature MUC1 mucin in biliary epithelial cells in various cystic liver diseases. Hepatology. 1996;24:539–43. doi: 10.1053/jhep.1996.v24.pm0008781320. [DOI] [PubMed] [Google Scholar]
- 65.Sasaki M, Nakanuma Y. Frequent expression of MUC1 apomucin on biliary epithelial cells of damaged small bile ducts in primary biliary cirrhosis and chronic viral hepatitis: an immunohistochemical study. Hepatology. 1996;23:1313–1317. doi: 10.1053/jhep.1996.v23.pm0008675144. [DOI] [PubMed] [Google Scholar]
- 66.Shibahara H, Tamada S, Higashi M, Goto M, Batra SK, Hollingsworth MA, Imai K, Yonezawa S. MUC4 is a novel prognostic factor of intrahepatic cholangiocarcinoma-mass forming type. Hepatology. 2004;39:220–229. doi: 10.1002/hep.20031. [DOI] [PubMed] [Google Scholar]


