Abstract
The metabolism of pre-implantation embryos is far from being understood. In human embryos, the two major obstacles are the scarcity of material, for obvious ethical reasons, and complete absence of a relevant in vivo control model. Over-extrapolation from animal species to human systems adds to the complexity of the problem. Removal of some metabolites from media has been proposed, such as glucose and essential amino acids, on the basis of their pseudo “toxicity”. In contrast, addition of some compounds such as growth factors has been proposed in order to decrease apoptosis, which is a natural physiologic process. These suggestions reflect the absence of global knowledge, and in consequence mask reality. Some aspects of metabolism have been ignored, such as lipid metabolism. Others are seriously underestimated, such as oxidative stress and its relationship to imprinting/methylation, of paramount importance for genetic regulation and chromosomal stability. It has become increasingly obvious that more studies are essential, especially in view of the major extension of ART activities worldwide.
Keywords: Human embryo, Metabolism, Imprinting, Oxidative stress, Culture medium
Introduction
The goal of in-vitro fertilization (IVF) and embryo culture is to provide high quality embryos capable of continued development and implantation that will result in the birth of healthy babies. The early studies of pre-implantation embryo in vitro culture were conducted in animal systems [1–6], and considerable progress has been made since these were undertaken. During the early 70s, we and other teams [7] began to design and define new, more complex culture media, which were based on the composition of genital tract secretions [8]. During the initial stages of zygote formation and early cleavage divisions, only a minimal level of RNA transcription takes place (SRY, [9]): the viability of a mature oocyte after ovulation is dependent upon the presence of a stored pool of proteins and/or mRNA transcripts. After completing fertilization, the newly fertilised oocyte undergoes a process of activation, which modifies its metabolism. Pronuclear fusion during syngamy provides the zygote with a diploid complement of chromosomes, and the first mitotic division takes place. Early embryo cleavage then involves intense DNA replication and cell division in the absence of growth, and the accompanying metabolic requirements must be provided by the ovarian pool of stored maternal reserves. During these initial stages of development, there must be an effective mechanism for DNA repair in order to correct decays linked to oocyte and sperm DNA [10, 11]. An estimated 1.5 to two million repair operations are performed during the first cell cycle, and maternal reserves of mRNA, proteins, organelles, etc. are required to support and direct these processes. The maternal oocyte storage pools must be capable of supporting the metabolic pathways of early embryo development prior to activation of the zygote genome by coding for all of the enzymes required, at the correct timing and in the appropriate equilibrium. The cycle during which the zygote genome is activated (Zygotic Gene Activation, ZGA) is always the longest cell cycle of pre-implantation development [12]: any delay at this time will result in a decrease in the level of mRNA below a critical threshold, and without appropriate ZGA the mammalian embryo fails to develop further. This crucial transition takes place during the 4–8 cell stage in human embryos. Polyadenylated maternal RNA transcripts are depleted, and new embryonic mRNAs are transcribed.
Although maternal oocyte storage pools are obviously important, the environment of the embryo is also critical. Culture conditions have a direct impact on transcription and translation [13–15]; embryonic metabolism is depressed during in vitro culture, protein turnover is accelerated [14] and mitochondrial function is usually impaired. The imprinting process may also be defective due to mis-regulated methylation activity [16]. Suboptimal culture conditions have frequently been observed to decrease cell numbers and jeopardize human embryo viability. Therefore, for many years embryos have routinely been transferred at early “cleavage stages”, in order to minimize the time spent in vitro. An ineluctable statement must be emphasized here: in the majority of cases, incorporation of a molecule is less energy and time consuming than its synthesis. The ‘free floating’ embryo has to manage in the culture medium in vitro, with active uptake of sugars, amino acids, and nucleic acid precursors, as well as passive transport, especially of lipids, which are associated mainly with albumin in vitro. The culture medium must also be equilibrated and fulfill the requirements for specific metabolites, available in the appropriate ratios. Culture media rarely reflect the in vivo situation, failing to take into account the efficacy of transport systems into the oocyte and early embryo. This neglected aspect has led to the emergence of aberrant proposals, such as a theory surrounding the role of essential amino acids in pre implantation development (the essential amino acid methionine and effects on imprinting). In this review, we will summarize current biochemical knowledge, based mainly on animal studies—bearing in mind that evidence from animal systems cannot always be extrapolated to human embryo physio-biochemistry. Moreover, interactions between different classes of metabolites i.e. sugars, lipids, amino acids are not necessarily the same in differing animal systems. We will draw attention to new information that has been elucidated through the study of oocyte mRNA content, especially concerning important basic metabolic pathways, ROS-linked decays and protection against DNA damage.
Physico-chemical parameters, electrolytes and embryo metabolism
A wide range of osmolarity allows the fertilization process and the development of the pre-implantation embryo [5, 17]. Physiological osmolarity surrounding the embryo in vivo, i.e. tubal fluid is 280–290 milliOsmols/Kg. A value of 260 mOsm/Kg is often observed for commercial culture media, based on the “mouse embryo assay”, which is not necessarily a rational idea [12, 18]. This lowering of osmolarity results in an increase in the transfer of water into the pre-implantation embryo. In contrast, such a low osmolarity decreases blastocyst formation in bovine [19]. Apart from an increase or decrease in the exchange of metabolites, the real impact of sub-physiologically low osmolarity on embryo metabolism is difficult to analyse, and is unforeseeable in the short or medium term. Endogenous pH is regulated by a bicarbonate buffer equilibrated in vivo via carbonic anhydrase and CO2 circulation, and in vitro by the CO2 atmosphere associated with culture medium bicarbonate [2, 8, 20]. CO2 is used as a metabolite, incorporated for the synthesis of the pyrimidine bases thymine, cytosine and uracil. There is no doubt that this pathway is used: cytoplasmic carbamoyl phosphate synthase (CPS) 2, involved in the first step of pyrimidine synthesis, is highly expressed in the oocyte, at 25X background expresssion. CO2 is also incorporated in the ureogenesis pathway involving ornthine, citrulline and arginine: mitochondrial CPS1 is expressed at a level even higher than CPS2, at 35X background level. CPS1 is involved in the elimination of ammonia (NH3) and is energy (ATP) consuming. In vivo O2 concentration differs from that in vitro: tubal fluid contains 7 % O2, whereas embryos are currently cultured in 18 % O2 (5 % CO2 in air). However, there is a current trend towards culturing embryos in a 5 % O2 atmosphere, although this is still a controversial issue. According to Gomes Sobrinho et al. [21] “it seems too early to conclude that low O2 has an effect on IVF outcome, at least in humans”. In vivo red-Ox potential is −0.1 mV, due to the presence of reducing substances and dynamic regulation in the embryonic environment [22]. It should also be noted that culture media spontaneously generate free radicals and ROS even under conditions of low oxygen concentration [23], dependent upon the composition of the culture media; therefore it is difficult to determine what is linked to oxygen tension and/or to “protective” quality of the culture media. Iron (Fe3+) and copper cations appear to arrest embryo development, encouraging free radical formation (Haber-Weiss and Fenton reactions)—therefore penicillamine or the poly-amino carboxylic acid EDTA, Ethylene di-amine tetra-acetic acid, are beneficial as chelating agents of toxic divalent cations in early stage culture media. EDTA is commonly added to IVF culture media in order to reduce generation of reactive oxygen species, but its physiological role is far from being understood, especially as Ca++ and Mg++ are also added to these media. EDTA is also a free radical scavenger. A suggestion that EDTA might be deleterious after genome activation and should be removed in the second phase of culture has been shown to be invalid. Zinc is an essential trace element that is required during mammalian developmental processes, acting as a co-factor for at least 200 enzymes, including carbonic anhydrase (generating HCO3). It is involved in regulating methylation processes via recycling of homocysteine (vide infra re vitamins), and prevents oxidative stress by capturing superoxide and hydroxyl radicals through its involvement in metallothioneins (MT) and Zn superoxide dismutase. It is the second most abundant transition metal after iron, and prevents the toxicity of iron and copper. Zinc is rarely/never added to culture media; its bioavailability is weak/nil unless it is introduced in a chelated state.
Sugar and metabolic derivatives
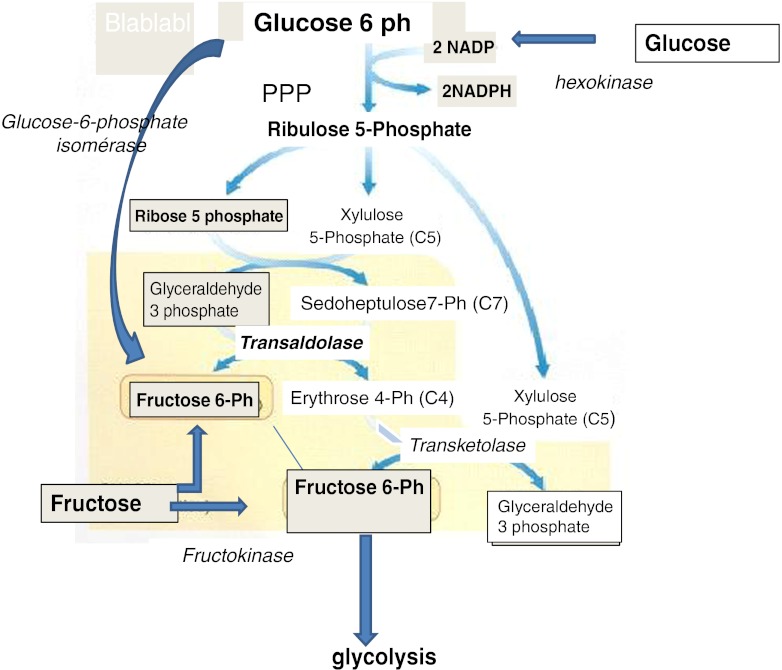
Metabolism of sugar is more complex than is often assumed. Active transport of glucose into the embryo was described in the late 60s [24]. Glucose and lactate are necessary for mouse embryo development in vitro [3]. Obviously, hexose metabolism is necessary for ATP (Adenosine tri phosphate) synthesis, the master molecule in embryo development: one mole of glucose generates from 30 to 36 mol of ATP. It has been suggested that glucose is toxic during in vitro culture before genomic activation, and that glucose and phosphate together may inhibit early embryo development [25, 26], an aspect of metabolism that remains controversial [27]. High levels of glucose may have a deleterious effect through an increase in free radical formation and impairment of mitochondrial function: this is particularly obvious in diabetic mammals [28] and probably in humans. High external concentrations of glucose in an excessively simple culture medium are toxic, rather than the glucose per se. The negative effect of glucose may be counterbalanced by the presence of a correct amino acid balance, i.e. the presence of sulfur amino acids and derivatives, neutralizing the ROS. In the bovine system, an excess of glucose increases apoptosis and induces shifts in sex ratio towards the female [29]: the female embryo has a more effective antiapoptotic mechanism than the male (XIAP, X-linked inhibitor of apoptosis). Indeed, an increase in monozygotic twinning (MZT) may be due to the quality of culture media/conditions and an excessively high level of glucose, rather than to prolonged culture time [30]. Glucose is present in female genital tract secretions at milli molar levels (from 0.5 to 3), depending on the animal species, and glucose transporters are present in the embryos; these observations are also true for human embryos. Lactate and pyruvate are also present, with the lactate/pyruvate ratio always 10–20x in favour of lactate. Pyruvate is an interesting compound, in that it acts not only as an energy source, but can also detoxify ammonia in the embryo through transamination to alanine, which can be exported from the cell. Complete removal of glucose from culture media is not beneficial: this approach was developed in CZB medium [26], with glucose replaced by glutamine and the lactate/pyruvate ratio increased to 100:1. However, these metabolites, even when added at high concentrations, are not sufficient: the early embryo rapidly needs hexoses for further development. Fructose is also present in the female genital tract, and replacing glucose with fructose alleviates the 2-cell block in outbred mice [31]. In general, pre-implantation embryos need glucose or fructose in order to enable further embryo viability post transfer [32]. The positive effect of fructose is related to a weak activity of Glucose-6-P isomerase (EC 5.3.1.9, [31]), which is weakly expressed in the human oocyte—in contrast to fructokinase, which is strongly expressed at 40X background level (2X the expression of Hexokinase2). Metabolism of glucose is oriented towards the pentose phosphate pathway (PPP), strongly up-regulated post fertilization at the time of pronuclear formation. The pentose phosphate pathway generates ribose5P and NADPH: one mole of Glucose 6 phosphate generates 1 mol of Ribose 5 phosphate + 2 mol of NADPH. NADPH is required for the majority of anabolic reactions, including lipid and nucleic acid synthesis, and is mandatory for regeneration of glutathione, the universal cell protector against ROS-linked decays. Oocyte glutathione is mandatory for sperm head swelling immediately after fertilization. Via methylene tetra-hydrofolate reductase, NADPH mediates methionine recycling from homocysteine, an inhibitor of methylation. NADPH is thus involved in the correct imprinting process (vide infra amino acids). Ribose phosphate, the ribose core of DNA, generates Fructose 6 phosphate through transaldolase, the essential link between the pentose phosphate pathway and glycolysis (Fig. 1). Transaldolase is one of the enzymes with highest expression in the oocyte, at 560X the background level. In conclusion, hexoses are necessary for pre-implantation development, but the equilibrium between sugars and other metabolic compounds is of paramount importance.
Fig. 1.
Interactions between pentose phosphate pathway (PPP) and Glycolysis. The PPP generates Ribose phosphate necessary for the nuclear material synthesis. Fructose (through Fructose6Ph), generated by transasldolase, or originating from external supply (female genital tract) seems to be more involved in glycolysis
Lipids
The lipid content of a pre-implantation embryo can be estimated as 55–60 nanograms. Lipid metabolism is often neglected: first of all because lipid solubility in vitro is not easy to manage, and in addition the analysis of lipids is a delicate process, especially in the case of fatty acids. The embryo needs lipids in very early stages, as lipid beta-oxidation is essential for meiotic resumption [33] and oocyte competence [34, 35], and lipids are then required immediately post fertilization. In terms of energy yields, one chain of fatty acid (palmitate) provides nearly three times more ATP, the fuel of early pre-implantation development, than one glucose molecule. Mitochondria are involved in the metabolism of lipids, and this requires the presence of carnitine as a catalytical molecule. The capacity of embryos to metabolize fatty acids was demonstrated in rabbit and mouse [36–38], indicating continued substrate oxidation from internal stores. Triglycerides are the most important class of lipids in embryos [39]. Immediately post fertilization the embryo can also synthesize phospholipids and cholesterol to a certain extent [40]. An endogenous supply seems mandatory: adding compactin or disogenin to block this cholesterol synthesis causes developmental arrest. The complexity of fatty acids increases during early embryonic development, with an increase in arachidonic acid (C20:4ω6) in particular (cow: [41]). In vitro the situation is complex: addition of lipids (through serum) increases embryo lipid content and upsets the equilibrium, but not necessarily in an appropriate manner, especially in consideration of viability after freezing. In any case, the amount of fat and its composition depends upon the lipids provided; embryo culture is performed in domestic animals without the addition of serum, for reasons of safety related to imprinting problems. The only lipids available are those that are bound to albumin, and we found that both saturated and unsaturated free fatty acids are present in largely sufficient quantities [41]. In humans, the situation seems more complicated: at least two important enzymes involved in carnitine synthesis (tri-methyl lysine hydroxylase and four gamma butyrobetaïne hydroxylase) are not expressed, whereas the enzymes involved in lipid beta oxidation are highly expressed [42]. This means that in vitro metabolism of lipids would appear to be impossible without the addition of carnitine; this feature may explain the improvement in blastocyst formation when carnitine is added in vitro [43]. Caprylic acid is the main lipid (fatty acid) bound to human serum albumin (HSA), due to methods of its preparation: in order to avoid any risk of bacterial and viral contamination, HSA is heated for several hours with caprylic acid and acetyl tryptophan, which may displace other fatty acids from the lipophilic albumin core. Overall, in the perspective of lipid metabolism, addition of carnitine to culture media for human embryos should not be overlooked. In fine, the use of an oil overlay is another aspect that can completely disturb “normal” embryo lipid metabolism: paraffin oil, supposedly considered to be an “inert” compound, may continuously “extract” some of the embryo (membrane) lipids, modifying the normal equilibrium and thus orienting lipid neo-syntheses incorrectly. This feature may be especially relevant when embryo culture is performed in very small microdrops.
Amino acids
Genital tract secretions contain all of the amino acids in vivo—they are mandatory for the synthesis of new proteins required during the first 3 days in human embryos, and these depend upon translation of polyadenylated mRNAs stored in the oocyte. The ratio between the different amino acids is probably more important than their overall quantity. Tubal secretions in most animals studied reveal similar aminograms. Any imbalance between amino acid concentrations reflects their capacity to be transported into the embryo as well their ability to act as organic osmolytes [44–46]. Some amino acids, especially glycine, the amino acid with the highest concentration in tubal secretions, have the ability to regulate embryonic endogenous osmolarity. They avoid the risk of ‘salting out’, i.e. precipitation of macromolecular compounds inside the cells due to an excessively high salt concentration. Moreover, there must be an equilibrium between the inorganic ions in order to facilitate their role as enzymatic cofactors, activators and even in signaling mechanisms. The differences observed in uptake/concentrations of amino acids has led to a theory that essential amino acids may be toxic: the differences are linked to their affinity for carrier molecules i.e. different amino acids compete for the same carriers. At the same millimolar concentrations, methionine almost completely prevents glycine entry into the embryo [47]. As mentioned [48], the concept of embryo-specific “essential” aminoacids is illogical, as it is not possible to determine exactly which amino acids are “essential” in embryo metabolism: interactions between amino acids and other metabolites are complicated and marginally understood ([49], Fig. 2). Assumption of “amino acid toxicity” is non-physiological, and may even be dangerous. This is especially true for sulfur aminoacids when applied in human (vide infra re methionine, imprinting and anti ROS protection). Moreover Brison et al. [50] were unable to define any difference between the uptake of essential and non-essential aminoacids. The idea of mimicking the amino acid composition of animal tubal fluid secretions was successfully developed in 1976 [8], and the media were shown to support human embryo culture in the absence of serum [51]. However, there is conflicting data regarding amino acid composition of human genital tract secretions [44, 52]. One of the two teams [44] describes aminograms that are more similar to that observed in animals. Large variations are observed, especially in the case of osmolyte aminoacids (taurine, glycine glutamine and alanine), which appear to be highly represented in vivo according to one report [44]. There are strong interactions between sugar metabolism and aminoacids, especially at the level of the Krebs cycle (Fig 2, [49]). Glutamine is a key amino acid in purine and pyrimidine synthesis, where it provides carbon atoms for de novo synthesis. Glutamine is stable in culture media under slightly alkaline conditions. Synthesis of 5′AMP from Ribose-5-P in embryonic cells requires two glutamine molecules, one glycine and one carbon originating from bicarbonate, as well as two Methyl tetrahydrofolate (vide infra re vitamins). Glycine, an important osmolyte, is present at millimolar concentration in tubal and uterine secretions. It can chelate Zn++ (four glycines for one Zn++) so that it is bioavailable for transport into embryonic cells. In fine, turnover of glycine is one of the markers of human embryo quality in vitro [50].
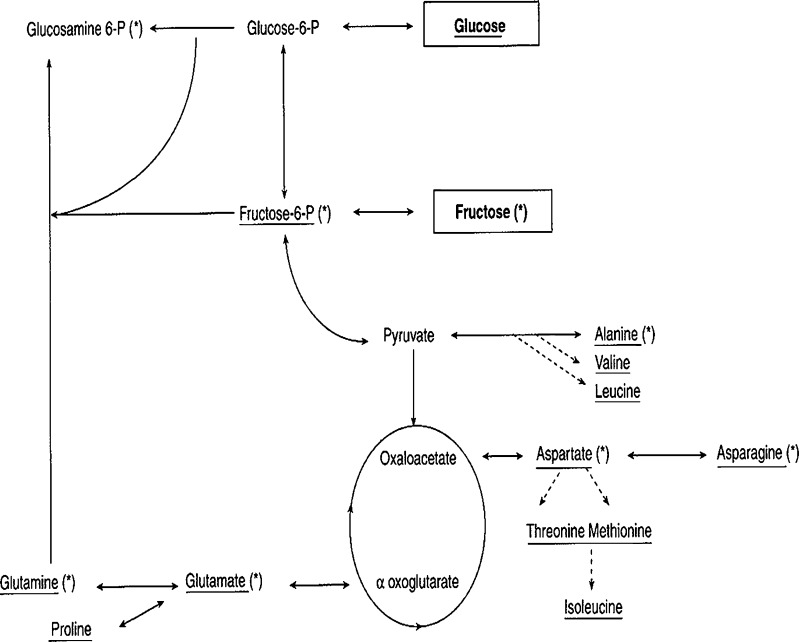
Fig. 2.
Major compounds originating from Fructose (*) and glucose and both(*) metabolism by bovine embryos. Labelling with 14C and determination of the aminio acids by ion exchange chromatography using ninhydrine post column staining (From Guyader Joly et al., Zygote 1996, [49] with permission). Fructose and glucose do not lead to the synthesis of the same aminoacids although a exactly similar pattern should have been observed
The sulfur amino acids cystine and methionine play a particularly important role:
-
Methylation/imprinting: maternal and paternal genomes must be complementary in order to ensure normal embryonic development. This is achieved via imprinting, a “dialogue” that takes place as early as the pronuclear stage, which involves variations in the methylation pattern of some genes. Examples of differential methylation include IGF2/IGF2-R [53], the maternal and the paternal X chromosome (Xist), H19 gene and a tumour proliferation suppressor. Disorganized imprinting may have harmful effects on early pre-implantation and late post-implantation development [54]. Imprinting occurs post fertilization in human embryos. Silent alleles are aberrantly expressed and hypomethylated in simplex media without amino acids in mouse embryos [55]. Methionine (Met), classified as an “essential amino acid” and thus omitted in some first phase sequential media formulations, is the major effector of methylation via S-Adenosyl methionine (Fig. 3): In fact, the balance between methionine other amino acids is of crucial significance. Methionine has such a high affinity for the transporters that if it is present in an excessively high concentration, it may prevent the uptake of other aminoacids, thus creating an imbalance in the endogenous pool. Met is incorporated by mouse, bovine and human embryos [47, 56], generating SAM: S-adenosyl methionine [56, 57]. SAM, apart from its role in methylation, is also a precursor of the polyamines spermine and spermidine. All of the enzymes involved in these synthetic pathways are very highly expressed, up to a level of 1000X background for ornithine decarboxylase (Fig. 4). The role of polyamines is not fully understood, but they bind to DNA and neutralize its acidic charges, playing a role in cell division.
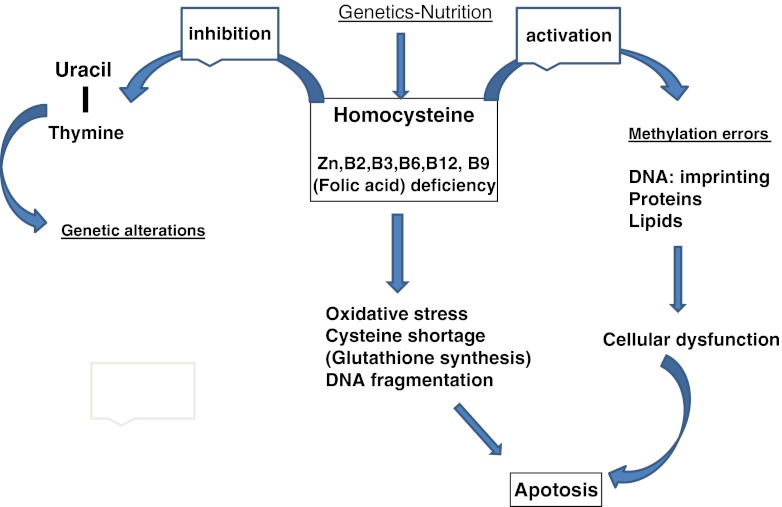
SAM and other methylating agents (Group B vitamins) are critical epigenetic regulators which can affect DNA stability, DNA and histone (H3) methylation, and imprinting [58–60]. Recycling of homocysteine (Hcy), i.e. the one-carbon cycle (Fig. 3), is of major importance as Hcy inhibits methylation [56]; it also competes for the same transporter as methionine for entry into the embryo [56]. B group vitamins and Zn are mandatory for homocysteine recycling (Vide infra, re vitamins). This aspect is especially important in human embryos, as the transsulfuration/cystathionine beta synthase pathway is poorly represented in the human oocyte [57]. In fine, homocysteine is both a cause and a consequence of oxidative stress [61, 62], and represents a link between oxidative stress and imprinting problems (Fig. 5).
Initiation of all protein synthesis requires methionine, through met-tRNA.
Lack of sulphur amino acids leads to sulphur amino acid deprivation syndrome (SAAD). This syndrome induces programmed cell death independently of caspase activation [63]. Apoptosis induced by methionine restriction induces formation of nucleosomal DNA fragments and is mitochondria-dependent (mitochondrial metabolism is one of the main sources of free radical generation).
Cysteine participates in the synthesis of glutathione, the universal free radical scavenger of major importance in vivo [22]. De novo glutathione synthesis inside the embryo is important, as it is not transported into the embryo: therefore adding it to culture media has only a marginally positive effect. After uptake, methionine can be converted to cysteine via the transsulfuration pathway; however, this pathway is poorly expressed in the human oocyte (Fig. 6, [57]).
Cysteine is strongly involved in the synthesis of hypotaurine, another free radical scavenger, via the cysteine sulfinic acid pathway in the tubal and uterine environment in vivo [22, 64]. No expression of this pathway can be detected in the oocyte, but hypotaurine uptake is possible. Taurine, produced by hypotaurine oxidation, is also an important osmolyte [65], present at high concentrations (millimolar level) in human uterine secretions [44]. Incorporated by the early embryo, it may also act as a regulator of membrane phospholipid methylation.
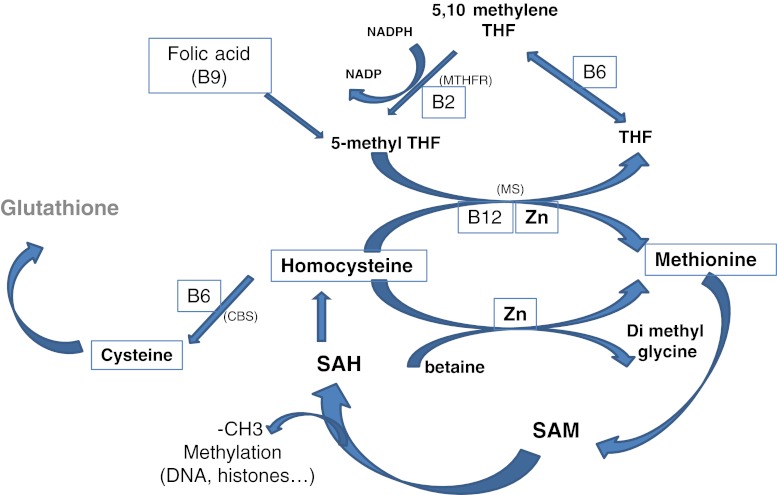
Fig. 3.
The one carbon cycle. Importance of group B vitamins and Zinc in Homocysteine recycling. Homocysteine can be recycled using three pathways; however the only one strongly expressed in the human oocyte is the MS pathway. The betaine-dimethyl glycine pathway is weakly expressed and the CBS pathway is totally absent during the pre-genomic activation period. SAH S-Adenosyl Homocysteine; SAM S-Adenosyl Methionine; THF tetrahydrofolate, CBS cystathionine beta synthase; MS Methionine synthase; MTHFR Methyltetrahydrofolate reductase Zn Zinc
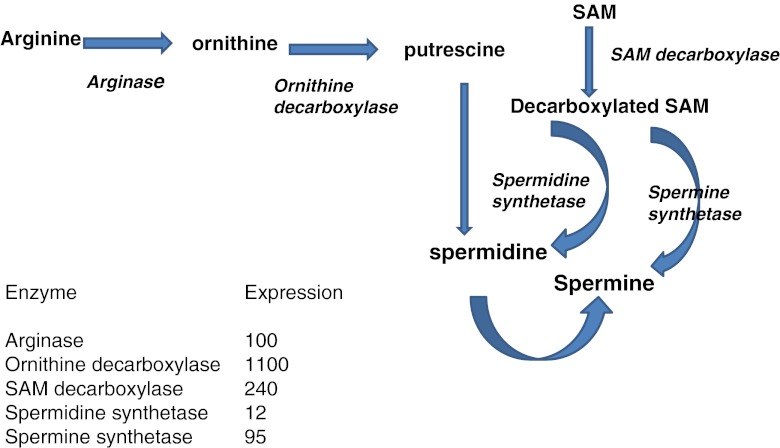
Fig. 4.
Polyamine synthesis and expression (X background signal) of enzymes involved. Spermidine is the condensation product of putrescine (originating from arginine metabolism and decarboxylated SAM). Then spermine will be formed from spermidine after condensation with decarboxylated SAM. The background signal (probe pair threshold ) value is 50. MicroArrays HG-U133,plus2. (For experimental procedure see [73]
Fig. 5.
Links between homocysteine, oxidative stress, imprinting and apoptosis (see [61, 62, 80])
Fig. 6.
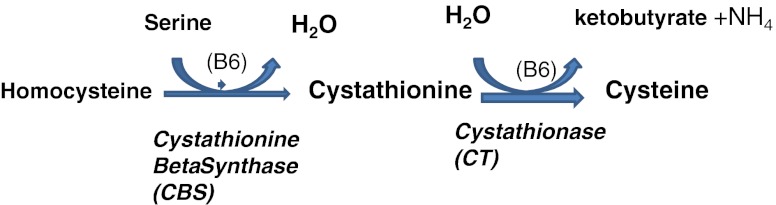
Cystathionine beta synthase pathway (conversion of homocysteine to cysteine): absent in human oocyte and early embryo
It has been assumed that continual culture in the presence of amino acids (especially glutamine) could reduce the viability of embryos as a consequence of ammonia formation resulting from spontaneous degradation of amino acids and/or their catabolism by the embryo [66]. In reality, the risk of ammonia toxicity is very low. Glutamine degradation is very weak in alkaline pH and less than 1 % of the ammonia formed in culture medium is retained. The salts formed, ammonium carbonate (NH4)2CO3 and bicarbonate (NH4HCO3) are highly unstable in a slightly alkaline pH, as in classical in vitro culture conditions. Their PKa is 7.2 at 25 ° C, and NH3 is released in the CO2 flux. Moreover, ammonia can also be removed by transamination of pyruvate to alanine. Alanine is then released into the culture medium [50], probably indicating an original dynamic system for removal of ammonia. The possibility that an ammonium gradient might be transitorily formed when embryo culture is performed in very small microdrops under oil cannot be excluded. Adding NH4Cl to culture media has recently been proposed as a model to mimic the toxicity of ammonium ions. However, NH4Cl is a highly stable acidic salt: this model does not reflect the physiological reality, and is therefore invalid. In fine, this poorly established concept has led to a reduction in the concentration of amino acids in embryo culture media relative to the in vivo environment [67] thus decreasing their “osmolyte protecting role” towards the embryo. As a consequence, osmolarity is decreased to 260 milli-osmoles in some commercial culture media, in order to counteract a possible negative effect of low amino acid concentrations (vide supra Physico-chemical parameters, electrolytes and embryo metabolism). However, this decreased amino acid concentration in culture media is likely to have a negative impact on some important embryo physiological processes such as imprinting [16, 68].
Precursors of DNA and RNA: purine and pyrimidine bases
It should be remembered that the pre-implantation embryo is able to synthesize these bases, utilizing CO2 from bicarbonate [69]—but this process has a high energy requirement. There are at least two reasons why the supply of nuclear bases is important: 1) as precursors for nucleic acid synthesis, since the amount of DNA is multiplied 8–16x before genomic activation. The requirement for bases is even more important for RNA synthesis, when there is an exponential increase linked to the first transcription waves at the beginning of maternal to zygotic transition (4–8 cells). 2) as “spare parts” for DNA repair, since a total of one to two million DNA repair operations are carried out on the male and female genomes at the time of fertilization and immediately afterwards [11]. Some repair operations involve removal of DNA adducts originating either from sperm [70] or oocyte [71], and others remove oxidized bases mainly, but not exclusively from the male genome [10]. There are more than 15 oxidized base products, the main one being 8 oxo Guanine. The mammalian genome contains CpG islands (300 to 3,000 base pairs in length), genomic regions with a high frequency of CpG sites; they are present close to, or within nearly one half of the promoter sites. Methylation occurs near the CpG islands or on the islands themselves. Any significant disturbance of these sites, linked to G oxidation, is expected to have a profound effect on gene expression. These CpG sites in DNA represent “sensitive” mutational hotspots [72]. Abasic sites and DNA strand breaks (also generated by oxidative stress) also need a supply of “clean” bases. The oocyte is well equipped to retrieve oxidized products from DNA, using several redundant mechanisms [11, 73], but the degraded products then have to be removed from the early embryo. This process sanitizes the endogenous pool, in order to avoid re-incorporation of the damaged bases: “Defective bricks build a defective house”. The pool of nucleotide sanitizing enzymes is highly expressed in oocytes (up to 100X background). In order to complete this process adequately, an exogenous supply of these molecules is recommended, especially since the early embryo is fully able to incorporate them from the media [74].
Vitamins
The role of vitamins during early development is not clear. First of all, lipid-soluble vitamins present an obstacle with regard to their solubility in culture media. Vitamins A and E are anti-oxidants; a biological function of vitamin E is to protect polyunsaturated fatty acids from oxidation in membranes. In bovine, these vitamins exert a positive effect on embryos in vitro [75]. Vitamin C is the natural anti-oxidant in follicular fluid, but it can be pro-oxidative when added to culture media, and may have a local denaturing effect on proteins [76]. Group B vitamins are mandatory (at least B2/6/9/12) for recycling of homocysteine (see Fig. 3). It was observed that B-group vitamins promote hamster embryo development in vitro [77], but other reports indicate that some of them could be inhibitory for early mouse embryos [78]. According to O’Neill [79] folic acid (B9) is mandatory as a methyl donor during early embryonic development, participating in at least in two pathways: synthesis of thymine and recycling of homocysteine. The folic acid cycle is linked to homocysteine recycling. All of the enzymes involved in these 2 cycles are highly expressed, at 20 and 500X the background signal [57]. There is one important exception: as previously mentioned, the human oocyte and the early preimplantation embryos are not able to use the trans-sulfuration pathway that allows direct conversion of homocysteine to cystine in two steps (Fig. 6). There is no expression of Cystathionine beta synthase, and CT (cystathionase) is very weakly expressed. This is another reason why a shortage of folic acid during very early embryogenesis must be avoided [80]. Moreover, the human oocyte has a high level of folate receptor 1 expression (300X background) as well as folate transporter member 1, also known as SLC19A1 (660X background), indicating a high level of trafficking around this molecule.
Macromolecules
Serum albumin is a macromolecule with a molecular weight of 65,000 KD (kilodaltons), containing 610 aminoacids. Along with transferrin, it is the most highly represented protein in the oviduct with antioxidant protecting roles. During the late 70s and early 80s, serum albumin replaced the use of serum in embryo culture [51], and in particular human serum albumin for human IVF. In the bovine system, there is evidence that serum is involved in the aetiology of large calf syndrome. Although the requirement for albumin may not be absolute, [81], it regulates oncotic pressure and also acts as a detergent through its capacity to bind lipids: this facilitates embryo manipulation, decreasing their capacity to stick to plastic. Albumin can enter the embryo directly, carrying numerous small molecules such as cathecholamines, amino acids and peptides into the embryo [82]. These peptides can probably be used as embryo “nutrition”, since they can be detached from the albumin core by salts. It is unlikely that albumin itself can be degraded.
The addition of Growth Factors in vitro remains controversial. Numerous growth factors (GFs) and interleukins (ILs), such as LIF (Leukemia inhibiting factor) and IL6 are present in the female genital tract, and corresponding receptors are also present on the surface of embryonic cells [83–85]. Moreover, when embryos are pooled in microdrops, external growth factors have been observed to improve embryo development to a certain extent, via an autocrine/paracrine regulation [86–88]. Growth factors that might potentially be added to embryo culture include platelet activating factor (PAF) and platelet-derived growth factor (PDGF). Cocultured cells also secrete interleukins and growth factors [85, 89, 90]. However it is also clear that growth factors act in both positive and negative synergy in order to produce a harmonious effect. Addition of a single growth factor is highly questionable: GF may reduce apoptosis, but apoptosis is a natural regulatory process, “destroying” cells with an abnormal chromosome status. There is a risk of improving the development of abnormal embryos through bypassing normal regulation “filters”. We have not yet reached the stage of being able to safely add single, or “mixtures” of growth factors.
Conclusions
Human “free floating” pre-implantation embryos are a unique feature in physiology, and our understanding of them is far from being complete. The obstacles to research include 1) scarcity of material available per embryo (around 55–60 nG of dry matter) 2) the number of embryos available for research, 3) complete absence of an appropriate corresponding model in vivo, 4) absence of a fully reliable animal model, as metabolic variations between animal models and human systems are evident. According to Patrizio and Sakkas [91], the yield in term of livebirths per oocyte retrieved can be estimated at around 5 %. However it must be emphasized clearly that culture medium is not solely responsible for this wastage. Culture in microdrops under oil in order to mimic the in vivo situation is risky: pollution of oil by hydrosoluble components has devastating consequences [92]. This type of culture does not mimic the in vivo situation, as dynamic evolution of the culture medium is not possible: embryonic lipids can be extracted by oil, with a resulting disruption of this aspect of embryo metabolism. With reference to the known interactions between lipid, sugar and aminoacid metabolism, this issue raises numerous questions. There is still room for improvement of culture media, with a basis in solid studies rather than unfounded statements. It is also important to avoid bypassing problems instead of solving them, eg: 1) the proposal of counteracting the so-called ‘toxic’ effects glucose or essential aminoacids, 2) reducing the amino acid concentration in order to avoid so-called “ammonia toxicity” and, as a consequence 3) decreasing osmolarity to a poorly physiological level of 250–260 milliosmolles/kG, allowing the mouse embryo assay to be passed 4) inhibiting apoptosis by the introduction of growth factor(s). The putative negative impact of these artifacts has to be taken into consideration, and merits attention on a mid/long term basis. Basic physiology and biochemistry may seem esoteric and tedious, but this remains the essential starting point in consideration of in vitro preimplantation development.
Footnotes
Capsule Knowledge of human embryo metabolism is complicated by the complete lack of an in vivo model. Metabolic variations between animal models and human systems are evident. We have attempted to add to current knowledge by interpreting information yielded via mRNA transcripts found in the oocyte. We have re-evaluated metabolism of lipids, glucose and amino acids, with a special focus on oxidative stress, imprinting and apoptosis, since certain physiological aspects have not previously been taken into account. More studies are essential in order to avoid introducing artifacts that bypass physiological processes and mask the biochemistry involved in real-time metabolism.
Contributor Information
Yves Ménézo, Email: yves.menezo@club-internet.fr.
Isabelle Lichtblau, Email: isalichtblau@gmail.com.
Kay Elder, Email: kay.elder@gmail.com.
References
- 1.Whitten WK. Culture of tubal mouse ova. Nature. 1956;177:96. doi: 10.1038/177096a0. [DOI] [PubMed] [Google Scholar]
- 2.Brinster RL. Studies on the development of mouse embryos in vitro I the effect of osmolarity and hydrogen ion concentration. J Exp Zool. 1965;158:49–57. doi: 10.1002/jez.1401580105. [DOI] [PubMed] [Google Scholar]
- 3.Brinster RL. Studies on the development of mouse embryos in vitro II. The effect of energy sources. J Exp Zool. 1965;158:59–68. doi: 10.1002/jez.1401580106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brinster RL. Studies on the development of mouse embryos in vitro III the effect of fixed nitrogen source. J Exp Zool. 1965;158:69–77. doi: 10.1002/jez.1401580107. [DOI] [PubMed] [Google Scholar]
- 5.Brinster RL. In vitro culture of mammalian embryos. J Anim Sci. 1968;27(Suppl 1):1–14. [PubMed] [Google Scholar]
- 6.Whittingham DG. Culture of mouse ova. J Reprod Fertil Suppl. 1971;14:7–21. [PubMed] [Google Scholar]
- 7.Tervit HR, Whittingham D, Rowson LEA. Successful culture in vitro of sheep and cattle ova. J Reprod Fertil. 1972;20:493–497. doi: 10.1530/jrf.0.0300493. [DOI] [PubMed] [Google Scholar]
- 8.Ménézo Y. Milieu synthétique pour la survie et la maturation des gamètes et pour la culture de l’œuf fécondé. CR Acad Sci D Paris. 1976;282:1967–1970. [PubMed] [Google Scholar]
- 9.Ao A, Erickson RP, Winston RM, Handyside AH. Transcription of paternal Y-linked genes in the human zygote as early as the pronucleate stage. Zygote. 1994;2:281–7. doi: 10.1017/S0967199400002100. [DOI] [PubMed] [Google Scholar]
- 10.Lopes S, Jurisicova A, Casper RF. Gamete-specific DNA fragmentation in unfertilized human oocytes after intracytoplasmic sperm injection. Hum Reprod. 1998;13:703–8. doi: 10.1093/humrep/13.3.703. [DOI] [PubMed] [Google Scholar]
- 11.Ménézo Y, Dale B, Cohen M. DNA damage and repair in human oocytes and embryos: a review. Zygote. 2010;18:357–65. doi: 10.1017/S0967199410000286. [DOI] [PubMed] [Google Scholar]
- 12.Betteridge KJ, Rieger D. Embryo transfer and related techniques in domestic animals, and their implications for human medicine. Hum Reprod. 1993;8:147–67. doi: 10.1093/oxfordjournals.humrep.a137864. [DOI] [PubMed] [Google Scholar]
- 13.Ho Y, Doherty AS, Schultz RM. Mouse preimplantation embryo embryo development in vitro: effect of sodium concentration in culture media on RNA synthesis and accumulation and gene expression. Mol Reprod Dev. 1994;38:131–141. doi: 10.1002/mrd.1080380203. [DOI] [PubMed] [Google Scholar]
- 14.Jung T. Protein synthesis and degradation in non-cultured and in vitro cultured rabbit blastocysts. J Reprod Fertil. 1989;86:507–512. doi: 10.1530/jrf.0.0860507. [DOI] [PubMed] [Google Scholar]
- 15.Wrenzycki C, Herrmann D, Carnwath JW, Niemann H. Alterations in the relative abundance of gene transcripts in preimplantation bovine embryos cultured in medium supplemented with either serum or PVA. Mol Reprod Dev. 1999;53:8–18. doi: 10.1002/(SICI)1098-2795(199905)53:1<8::AID-MRD2>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 16.Hiura H, Okae H, Miyauchi N, Sato F, Sato A, Van De Pette M, John RM, Kagami M, Nakai K, Soejima H, Ogata T, Arima T. Characterization of DNA methylation errors in patients with imprinting disorders conceived by assisted reproduction technologies. Hum Reprod. 2012;27:2541–8. doi: 10.1093/humrep/des197. [DOI] [PubMed] [Google Scholar]
- 17.Naglee DL, Maurer RR, Foote RH. Effect of osmolarity on in vitro development of rabbit embryos in a chemically defined medium. Exp Cell Res. 1969;58:331–3. doi: 10.1016/0014-4827(69)90513-8. [DOI] [PubMed] [Google Scholar]
- 18.Ménézo YJ, Hérubel F. Mouse and bovine models for human IVF. Reprod Biomed Online. 2002;4:170–5. doi: 10.1016/S1472-6483(10)61936-0. [DOI] [PubMed] [Google Scholar]
- 19.Liu Z, Foote RH. Sodium chloride, osmolytes, and osmolarity effects on blastocyst formation in bovine embryos produced by in vitro fertilization (IVF) and cultured in simple serum-free media. J Assist Reprod Genet. 1996;13:562–8. doi: 10.1007/BF02066609. [DOI] [PubMed] [Google Scholar]
- 20.Kane MT. Bicarbonate requirements for culture of one-cell rabbit ova to blastocysts. Biol Reprod. 1975;12:552–5. doi: 10.1095/biolreprod12.5.552. [DOI] [PubMed] [Google Scholar]
- 21.Gomes Sobrinho DB, Oliveira JB, Petersen CG, Mauri AL, Silva LF, Massaro FC, Baruffi RL, Cavagna M, Franco JG., Jr IVF/ICSI outcomes after culture of human embryos at low oxygen tension: a meta-analysis. Reprod Biol Endocrinol. 2011;9:143–154. doi: 10.1186/1477-7827-9-143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guérin P, El Mouatassim S, Ménézo Y. Oxidative stress and protection against reactive oxygen species in the pre-implantation embryo and its surroundings. Hum Reprod Update. 2001;7:175–89. doi: 10.1093/humupd/7.2.175. [DOI] [PubMed] [Google Scholar]
- 23.Martín-Romero FJ, Miguel-Lasobras EM, Domínguez-Arroyo JA, González-Carrera E, Alvarez IS. Contribution of culture media to oxidative stress and its effect on human oocytes. Reprod Biomed Online. 2008;17:652–6. doi: 10.1016/S1472-6483(10)60312-4. [DOI] [PubMed] [Google Scholar]
- 24.Wales RG, Brinster RL. The uptake of hexoses by mouse pre-implantation embryos in vitro. J Reprod Fertil. 1968;15:415–22. doi: 10.1530/jrf.0.0150415. [DOI] [PubMed] [Google Scholar]
- 25.Schini, Bavister BD. Two-cell block to development of cultured hamster embryos is caused by phosphate and glucose. Biol Reprod. 1988;39:1183–92. doi: 10.1095/biolreprod39.5.1183. [DOI] [PubMed] [Google Scholar]
- 26.Chatot CL, Ziomek CA, Bavister BD, Lewis JL, Torres I. An improved culture medium supports development of random-bred 1-cell mouse embryos in vitro. J Reprod Fertil. 1989;86:679–88. doi: 10.1530/jrf.0.0860679. [DOI] [PubMed] [Google Scholar]
- 27.Biggers JD, McGinnis LK. Evidence that glucose is not always an inhibitor of mouse preimplantation development in vitro. Hum Reprod. 2001;16:153–163. doi: 10.1093/humrep/16.1.153. [DOI] [PubMed] [Google Scholar]
- 28.Pampfer S. Apoptosis in rodent peri-implantation embryos: differential susceptibility of inner cell mass and trophectoderm cell lineages—a review. Placenta. 2000;21(suppltA):S3–10. doi: 10.1053/plac.1999.0519. [DOI] [PubMed] [Google Scholar]
- 29.Jimenez A, Madrid-Bury N, Fernandez R, Pérez-Garnelo S, Moreira P, Pintado B, de la Fuente J, Gutiérrez-Adán A. Hyperglycemia-induced apoptosis affects sex ratio of bovine and murine preimplantation embryos. Mol Reprod Dev. 2003;65:180–7. doi: 10.1002/mrd.10286. [DOI] [PubMed] [Google Scholar]
- 30.Cassuto G, Chavrier M, Menezo Y. Culture conditions and not prolonged culture time are responsible for monozygotic twinning in human in vitro fertilization. Fertil Steril. 2003;80:462–463. doi: 10.1016/S0015-0282(03)00599-5. [DOI] [PubMed] [Google Scholar]
- 31.Ménézo Y, Khatchadourian C. Implication of glucose 6 phosphate isomerase (EC 5.3.1.9) activity in blocking segmentation of the mouse ovum at the 2 cell stage in vitro. CR Acad Sci. 1990;310:297–301. [PubMed] [Google Scholar]
- 32.Ludwig TE, Lane M, Bavister BD. Differential effect of hexoses on hamster embryo development in culture. Biol Reprod. 2001;64:1366–74. doi: 10.1095/biolreprod64.5.1366. [DOI] [PubMed] [Google Scholar]
- 33.Downs SM, Mosey JL, Klinger J. Fatty acid oxidation and meiotic resumption in mouse oocytes. Mol Reprod. Dev. 2009; 76:844–53. [DOI] [PMC free article] [PubMed]
- 34.Dunning KR, Akison LK, Russell DL, Norman RJ, Robker RL. Increased beta-oxidation and improved oocyte developmental competence in response to L-carnitine during ovarian in vitro follicle development in mice. Biol Reprod. 2011;85:548–55. doi: 10.1095/biolreprod.110.090415. [DOI] [PubMed] [Google Scholar]
- 35.Dunning KR, Cashman K, Russell DL, Thompson JG, Norman RJ, Robker RL. Beta-oxidation is essential for mouse oocyte developmental competence and early embryo development. Biol Reprod. 2010;83:909–18. doi: 10.1095/biolreprod.110.084145. [DOI] [PubMed] [Google Scholar]
- 36.Waterman RA, Wall RJ. Lipid interactions with in vitro development of mammalian zygotes. Gamete Res. 1988;21:243–54. doi: 10.1002/mrd.1120210306. [DOI] [PubMed] [Google Scholar]
- 37.Hillman N, Flynn TJ. The metabolism of exogenous fatty acids by preimplantation mouse embryos developing in vitro. J Embryol Exp Morphol. 1980;56:157–68. [PubMed] [Google Scholar]
- 38.Ferguson EM, Leese HJ. A potential role for triglyceride as an energy source during bovine oocyte maturation and early embryo development. Mol Reprod Dev. 2006;73:1195–201. doi: 10.1002/mrd.20494. [DOI] [PubMed] [Google Scholar]
- 39.Homa ST, Racowsky C, McGaughey RW. Lipid analysis of immature pig oocytes. J Reprod Fertil. 1986;77:425–434. doi: 10.1530/jrf.0.0770425. [DOI] [PubMed] [Google Scholar]
- 40.Flynn TJ, Hillman N. Lipid synthesis from [U14C]glucose in pre-implantation mouse embryos in culture. Biol Reprod. 1978;19:922–926. doi: 10.1095/biolreprod19.4.922. [DOI] [PubMed] [Google Scholar]
- 41.Ménézo Y, Renard JP, Delobel B, Pageaux JF. Kinetic study of fatty acid composition of day 7 to day 14 cow embryos. Biol Reprod. 1982;26:787–90. doi: 10.1095/biolreprod26.5.787. [DOI] [PubMed] [Google Scholar]
- 42.Montjean D, Entezami F, Lichtblau I, Belloc S, Gurgan T, Menezo Y. Carnitine content in the follicular fluid and expression of the enzymes involved in beta oxidation in oocytes and cumulus cells. J Assist Reprod Genet. 2012;29:1221–1225. doi: 10.1007/s10815-012-9855-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Abdelrazik A, El-Damen H, Badrawi R, Sharma A. Agarwal Introduction of a single step medium from fertilization through blastocyst stage by supplementation of the culture media with L-Carnitine. ASRM meeting, Washington DC; 2007. p. 312.
- 44.Casslén BG. Free amino acids in human uterine fluid. Possible role of high taurine concentration. J Reprod Med. 1987;32:181–184. [PubMed] [Google Scholar]
- 45.Dawson KM, Baltz JM. Organic osmolytes and embryos: substrates of the Gly and beta transport systems protect mouse zygotes against the effects of raised osmolarity. Biol Reprod. 1997;56:1550–8. doi: 10.1095/biolreprod56.6.1550. [DOI] [PubMed] [Google Scholar]
- 46.Baltz JM. Media composition: salts and osmolality. Methods Mol Biol. 2012;912:61–80. doi: 10.1007/978-1-61779-971-6_5. [DOI] [PubMed] [Google Scholar]
- 47.Khatchadourian C, Guillaud J, Menezo Y. Interactions in glycine and methionine uptake, conversion and incorporation into proteins in the preimplantation mouse embryo. Zygote. 1994;2:301–306. doi: 10.1017/S0967199400002124. [DOI] [PubMed] [Google Scholar]
- 48.Summers MC, Biggers JD. Chemically defined media and the culture of mammalian preimplantation embryos: historical perspective and current issues. Hum Reprod Update. 2003;9:557–82. doi: 10.1093/humupd/dmg039. [DOI] [PubMed] [Google Scholar]
- 49.Guyader-Joly C, Khatchadourian C, Ménézo Y. Comparative glucose and fructose incorporation and conversion by in vitro produced bovine embryos. Zygote. 1996;4:85–91. doi: 10.1017/S0967199400002951. [DOI] [PubMed] [Google Scholar]
- 50.Brison DR, Houghton FD, Falconer D, Roberts SA, Hawkhead J, Humpherson PG, Lieberman BA, Leese HJ. Identification of viable embryos in IVF by non-invasive measurement of amino acid turnover. Hum Reprod. 2004;19:2319–24. doi: 10.1093/humrep/deh409. [DOI] [PubMed] [Google Scholar]
- 51.Ménézo Y, Testart J, Perone D. Serum is not necessary in human in vitro fertilization, early embryo culture, and transfer. Fertil Steril. 1984;42:750–5. doi: 10.1016/s0015-0282(16)48202-6. [DOI] [PubMed] [Google Scholar]
- 52.Tay JI, Rutherford AJ, Killick SR, Maguiness SD, Partridge RJ, Leese HJ. Human tubal fluid: production, nutrient composition and response to adrenergic agents. Hum Reprod. 1997;12:2451–2456. doi: 10.1093/humrep/12.11.2451. [DOI] [PubMed] [Google Scholar]
- 53.De Groot N, Hochberg A. Gene imprinting during placental and embryonic development. Mol Reprod Dev. 1993;36:390–406. doi: 10.1002/mrd.1080360316. [DOI] [PubMed] [Google Scholar]
- 54.Goshen R, Ben Rafael Z, Gonik B, Lustig O, Tannos V, de-Groot N, Hochberg AA. The role of genomic imprinting in implantation. Fertil Steril. 1994;2:903–910. [PubMed] [Google Scholar]
- 55.Khosla S, Dean W, Reik W. Culture of pre-implantation embryos and its long-term effects on gene expression and phenotype. Hum Reprod Updat. 2001;7:419–427. doi: 10.1093/humupd/7.4.419. [DOI] [PubMed] [Google Scholar]
- 56.Ménézo Y, Khatchadourian C, Gharrib A, Hamidi J, Sarda N. Regulation of sadenosyl methionine synthesis in the mouse embryo. Life Sci. 1989;44:1601–1609. doi: 10.1016/0024-3205(89)90455-4. [DOI] [PubMed] [Google Scholar]
- 57.Benkhalifa M, Montjean D, Cohen-Bacrie P, Ménézo Y. Imprinting: RNA expression for ho mocysteine recycling in the human oocyte. Fertil Steril. 2010;93:1585–90. doi: 10.1016/j.fertnstert.2009.02.081. [DOI] [PubMed] [Google Scholar]
- 58.Niemitz EL, Feinberg AP. Epigenetics and assisted reproductive technology: a call for investigation. Am J Hum Genet. 2004;74:599–609. doi: 10.1086/382897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wolff GL, Kodell RL, Moore SR, Cooney CE. Maternal epigenetics and methyl supplements affect agouti gene expression in Avy/mice. FASEB J. 1998;12:949–957. [PubMed] [Google Scholar]
- 60.Xin Z, Tachibana M, Guggiari, et al. Role of histone methyltransferase G9a in CpG methylation of the Prader–Willi syndrome imprinting centre. J Biol Chem. 2003;278:14996–15000. doi: 10.1074/jbc.M211753200. [DOI] [PubMed] [Google Scholar]
- 61.Hoffman M. Hypothesis: hyperhomocysteinemia is an indicator of oxidant stress. Med Hypotheses. 2011;77:1088–93. doi: 10.1016/j.mehy.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 62.Ménézo Y, Mares P, Cohen M, Brack M, Viville S, Elder K. Autism, imprinting and epigenetic disorders: a metabolic syndrome linked to anomalies in homocysteine recycling starting in early life?? J Assist Reprod Genet. 2011;28:1143–5. doi: 10.1007/s10815-011-9645-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Lu S, Hoestje SM, Choo E, Epner DE. Induction of caspase dependant and independant apoptosis in response to methionine restriction. Int J Oncol. 2003;22:415–420. [PubMed] [Google Scholar]
- 64.Guérin P, Ménézo Y. Hypotaurine and taurine in gamete and embryo environments: de novo synthesis via the cysteine sulfinic acid pathway in oviduct cells. Zygote. 1995;3:333–43. doi: 10.1017/S0967199400002768. [DOI] [PubMed] [Google Scholar]
- 65.Dumoulin JC, van Wissen LC, Menheere PP, Michiels AH, Geraedts JP, Evers JL. Taurine acts as an osmolyte in human and mouse oocytes and embryos. Biol Reprod. 1997;56:739–44. doi: 10.1095/biolreprod56.3.739. [DOI] [PubMed] [Google Scholar]
- 66.Gardner DK, Lane M. Amino acids and ammonium regulate mouse embryo development in culture. Biol Reprod. 1993;48:377–85. doi: 10.1095/biolreprod48.2.377. [DOI] [PubMed] [Google Scholar]
- 67.Elhassan YM, Wu G, Leanez AC, et al. Amino acid concentrations in fluids from the bovine oviduct and uterus and in KSOM-based culture media. Theriogenology. 2001;55:1907–18. doi: 10.1016/S0093-691X(01)00532-5. [DOI] [PubMed] [Google Scholar]
- 68.Katari S, Turan N, Bibikova M, Erinle O, Chalian R, Foster M, Gaughan JP, Coutifaris C, Sapienza C. DNA methylation and gene expression differences in children conceived in vitro or in vivo. Hum Mol Genet. 2009;18:3769–78. doi: 10.1093/hmg/ddp319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Pike IL, Murdoch RN, Wales RG. The incorporation of carbon dioxide into the major classes of RNA during culture of the preimplantation mouse embryo. J Reprod Fertil. 1975;45:211–2. doi: 10.1530/jrf.0.0450211. [DOI] [PubMed] [Google Scholar]
- 70.Badouard C, Ménézo Y, Panteix G, Ravanat JL, Douki T, Cadet J, Favier A. Determination of new types of DNA lesions in human sperm. Zygote. 2008;16:9–13. doi: 10.1017/S0967199407004340. [DOI] [PubMed] [Google Scholar]
- 71.Zenzes MT, Puy LA, Bielecki R. Immunodetection of benzo[a]pyrene adducts in ovarian cells of women exposed to cigarette smoke. Mol Hum Reprod. 1998;4:159–65. doi: 10.1093/molehr/4.2.159. [DOI] [PubMed] [Google Scholar]
- 72.Wachsman JT. DNA methylation and the association between genetic and epigenetic changes: relation to carcinogenesis. Mutat Res. 1997;375:1–8. doi: 10.1016/S0027-5107(97)00003-1. [DOI] [PubMed] [Google Scholar]
- 73.Ménézo YJR, Russo G, Tosti E, El Mouatassim S, Benkhalifa M. Expression profile of genes coding for DNA repair in human oocytes using pangenomic microarrays, with a special focus on ROS linked decays. J Assist Reprod Genet. 2007;24:513–20. doi: 10.1007/s10815-007-9167-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Epstein CJ. Gene expression and macromolecular synthesis during pre-implantation embryonic development. Biol Reprod. 1975;12:82–105. doi: 10.1095/biolreprod12.1.82. [DOI] [PubMed] [Google Scholar]
- 75.Olson SE, Seidel GE., Jr Culture of in vitro-produced bovine embryos with vitamin E improves development in vitro and after transfer to recipients. Biol Reprod. 2000;62:248–252. doi: 10.1095/biolreprod62.2.248. [DOI] [PubMed] [Google Scholar]
- 76.Giustarini D, Dalle-Donne I, Colombo R, Milzani A, Rossi R. Is ascorbate able to reduce disulfide bridges? A cautionary note. Nitric Oxide. 2008;19:252–258. doi: 10.1016/j.niox.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 77.Bavister BD, Leibfried ML, Lieberman G. Development of preimplantation embryos of the golden hamster in a defined culture medium. Biol Reprod. 1983;28:235–247. doi: 10.1095/biolreprod28.1.235. [DOI] [PubMed] [Google Scholar]
- 78.Tsai FC, Gardner DK. Nicotinamide, a component of complex culture media, inhibits mouse embryo development in vitro and reduces subsequent developmental potential after transfer. Fertil Steril. 1994;61:376–382. doi: 10.1016/s0015-0282(16)56534-0. [DOI] [PubMed] [Google Scholar]
- 79.O’Neill C. Endogenous folic acid is essential for normal development of preimplantation embryos. Hum Reprod. 1998;13:1312–6. doi: 10.1093/humrep/13.5.1312. [DOI] [PubMed] [Google Scholar]
- 80.Ménézo Y, Entezami F, Lichtblau I, Belloc S, Cohen M, Dale B. Oxidative stress and fertility: incorrect assumptions and ineffective solutions? Zygote. 2012;12:1–11. doi: 10.1017/S0967199412000263. [DOI] [PubMed] [Google Scholar]
- 81.Caro CM, Trounson A. Successful fertilization, embryo development, and pregnancy in human in vitro fertilization (IVF) using a chemically defined culture medium containing no protein. J In Vitro Fert Embryo Transf. 1986;3:215–7. doi: 10.1007/BF01132806. [DOI] [PubMed] [Google Scholar]
- 82.Menezo Y, Khatchadourian C. Peptides bound to albumin. Life Sci. 1986;39:1751–1753. doi: 10.1016/0024-3205(86)90094-9. [DOI] [PubMed] [Google Scholar]
- 83.Croteau S, Menezo Y. Growth factors: from oocyte maturation to blastocyst. Contracept Fertil Sex. 1994;22:648–655. [PubMed] [Google Scholar]
- 84.Richter KS. The importance of growth factors for preimplantation embryo development and in-vitro culture. Curr Opin Obstet Gynecol. 2008;20:292–304. doi: 10.1097/GCO.0b013e3282fe743b. [DOI] [PubMed] [Google Scholar]
- 85.Ménézo YJ, Servy E, Veiga A, Hazout A, Elder K. Culture systems: embryo co-culture. Methods Mol Biol. 2012;912:231–247. doi: 10.1007/978-1-61779-971-6_14. [DOI] [PubMed] [Google Scholar]
- 86.Paria BC, Dey SK. Preimplantation embryo development in vitro: cooperative interactions among embryos and the role of growth factors. Proc Natl Acad Sci USA. 1990;87:4756–4760. doi: 10.1073/pnas.87.12.4756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gopichandran N, Leese HJ. The effect of paracrine/autocrine interactions on the in vitro culture of bovine preimplantation embryos. Reprod. 2006;131:269–67. doi: 10.1530/rep.1.00677. [DOI] [PubMed] [Google Scholar]
- 88.O’Neill C. Evidence for the requirement of autocrine growth factors for development of mouse—preimplantation embryos in vitro. Biol Reprod. 1997;56:229–237. doi: 10.1095/biolreprod56.1.229. [DOI] [PubMed] [Google Scholar]
- 89.Desai NN, Goldfarb JM. Growth factor/cytokine secretion by a permanent human endometrial cell line with embryotrophic properties. J Assist Reprod Genet. 1996;13:546–50. doi: 10.1007/BF02066606. [DOI] [PubMed] [Google Scholar]
- 90.Desai NN, Goldfarb J. Co-cultured human embryos may be subjected to widely different microenvironments: pattern of growth factor/cytokine release by Vero cells during the co-culture interval. Hum Reprod. 1998;13:1600–5. doi: 10.1093/humrep/13.6.1600. [DOI] [PubMed] [Google Scholar]
- 91.Patrizio P, Sakkas D. From oocyte to baby: a clinical evaluation of the biological efficiency of in vitro fertilization. Fertil Steril. 2009;91:1061–6. doi: 10.1016/j.fertnstert.2008.01.003. [DOI] [PubMed] [Google Scholar]
- 92.Menezo Y, Guerin P. Preimplantation embryo metabolism and embryo interaction with the in vitro environment. In: Elder K, Cohen J, editors. Human preimplantation Embryo evaluation and selection. London: Taylor-Francis; 2007. pp. 191–200. [Google Scholar]