Abstract
In obligate symbioses, the host’s survival relies on the successful acquisition and maintenance of symbionts. Symbionts can either be transferred from parent to offspring via direct inheritance (vertical transmission) or acquired anew each generation from the environment (horizontal transmission). With vertical symbiont transmission, progeny benefit by not having to search for their obligate symbionts, and, with symbiont inheritance, a mechanism exists for perpetuating advantageous symbionts. But, if the progeny encounter an environment that differs from that of their parent, they may be disadvantaged if the inherited symbionts prove suboptimal. Conversely, while in horizontal symbiont acquisition host survival hinges on an unpredictable symbiont source, an individual host may acquire genetically diverse symbionts well suited to any given environment. In horizontal acquisition, however, a potentially advantageous symbiont will not be transmitted to subsequent generations. Adaptation in obligate symbioses may require mechanisms for both novel symbiont acquisition and symbiont inheritance. Using denaturing-gradient gel electrophoresis and real-time PCR, we identified the dinoflagellate symbionts (genus Symbiodinium) hosted by the Red Sea coral Stylophora pistillata throughout its ontogenesis and over depth. We present evidence that S. pistillata juvenile colonies may utilize both vertical and horizontal symbiont acquisition strategies. By releasing progeny with maternally derived symbionts, that are also capable of subsequent horizontal symbiont acquisition, coral colonies may acquire physiologically advantageous novel symbionts that are then perpetuated via vertical transmission to subsequent generations. With symbiont inheritance, natural selection can act upon the symbiotic variability, providing a mechanism for coral adaptation.
Introduction
Obligate mutualistic symbioses are ubiquitous on earth and play pivotal roles in many ecosystems [1], [2]. By definition, in obligate mutualisms, the host must possess symbionts in order to survive. If a host secures the perpetuation of obligate symbionts by directly transferring symbionts to the offspring (vertical transmission) [3], progeny encountering an environment that differs from that of their parent may be disadvantaged by hosting a suboptimal symbiont. On the other hand, if a host releases aposymbiotic progeny that must acquire symbionts from the environment (horizontal transmission), progeny may acquire symbionts that are beneficial in a new environment [4]. As partner fidelity is not absolute in horizontal transmission, strong partner choice and symbiont sexual recombination can allow mutualisms to persist and evolve in systems with horizontal transmission [2], [5], [6]. In horizontal transmission however, since subsequent offspring do not inherit the symbionts, advantageous symbionts may or may not be acquired again, leaving each generation to potentially gamble with the continuation of a beneficial symbiosis.
Adaptation, that is maintained via natural selection and subsequent evolution, may be required for species to survive in a changing environment, but neither vertical nor horizontal symbiont acquisition strategies alone provide a mechanism for the adaptation of an obligate symbiosis via symbiont partner change. Vertical transmission provides a means for the perpetuation of symbionts, but offers no mechanism for the acquisition of novel symbionts. In contrast, horizontal transmission provides a mechanism for acquiring novel symbionts but limited means for perpetuating the novel symbionts. Combining both acquisition strategies may provide a mechanism for adaptation, but evidence of the same host species utilizing both transmission modes is rare. Phylogenetic analyses of specific obligate prokaryote-insect [7], [8] and prokaryote-marine invertebrate [9], [10] symbioses reveal predominant vertical symbiont transmission punctuated by infrequent horizontal symbiont acquisition. We investigated whether both symbiont transmission modes could occur in a eukaryote-invertebrate obligate symbiosis.
Reef building corals have an obligatory mutualism with dinoflagellate algae (genus Symbiodinium), which provides a nutritional foundation for host metabolism [11] and calcification [12] making them fundamental components of coral reef ecosystems. In the Symbiodinium genus, species remain largely unresolved, limiting classification to membership within nine Symbiodinium clades (named A–I) and multiple types within each clade [13], [14]. Symbiodinium can exhibit different physiologies in response to variations in light and temperature [15]–[17]. Consequently, the same host can display different physiologies based on the Symbiodinium found within it [18].
Regardless of whether Symbiodinium are acquired horizontally or vertically, adult corals exhibit extremely stable and specific mutualisms with Symbiodinium [19]–[24]. In contrast, larvae (planulae) and/or juveniles of coral species with horizontal Symbiodinium acquisition can acquire non-parental symbionts [25]–[27]. While the juvenile stage may be key in establishing novel symbioses, no studies to date have demonstrated coral juveniles capable of successfully maintaining a novel symbiont type into adulthood [28], nor have they provided a mechanism for the perpetuation of a novel symbiont to subsequent generations.
Contrary to horizontal symbiont acquisition, vertical transmission is often regarded as a “closed” system that precludes symbiont diversity in all life stages [3], [20], [29]–[32] but see [33]. Regarding vertical symbiont transmission as a closed system may explain why the symbiont identity in planulae and juveniles of coral species with vertical symbiont transmission has not been determined. Only recently has the symbiont identities in eggs of one coral species with vertical symbiont transmission been documented [34]. Planulae and/or juvenile corals with maternally derived symbionts may be capable of subsequent horizontal acquisition, which would facilitate diversity. Importantly, the inheritance of symbionts via vertical transmission would perpetuate the novel symbiosis if it increased the fitness of current and subsequent host generations. Therefore, deciphering the symbiont acquisition strategies utilized by corals throughout ontogenesis is key to understanding corals’ ability, or lack thereof, to alter their symbionts based on the environmental conditions of the habitat in which they grow.
We determined whether horizontal symbiont acquisition could occur in a coral host with vertical symbiont transmission. The coral Stylophora pistillata (Pocilloporidae) broods and releases planulae with vertically transmitted Symbiodinium [35]. S. pistillata is widely distributed throughout the Indo-Pacific and Red Sea [36], and is among the most abundant reef building corals in the Gulf of Eilat, Red Sea [35]. In the Gulf of Eilat, S. pistillata adult colonies host two distinct Symbiodinium clades. Shallow water colonies (<17 m) associate with clade A Symbiodinium [37], [38] (type A1 [39]), while congeners sampled in deep-water harbor symbionts of clade C [38] (e.g. type C72 at 20–30 m [5]).
We examined the Symbiodinium genetic identity in S. pistillata adults, their released planulae, and juvenile colonies, in shallow and deep depths, using techniques capable of detecting both abundant and possible low-level symbiont populations. We determined whether shallow and deep-water adult S. pistillata colonies hosted previously undetected low-levels of the second Symbiodinium clade found in S. pistillata adults. We also identified the Symbiodinium inherited by the planulae. Due to physiological differences between Symbiodinium, which symbiont(s) the progeny inherit may affect their survivorship in different habitats [34], [38]. Additionally, we looked at the Symbiodinium naturally occurring in juveniles at both shallow and deep depths. If S. pistillata juvenile colonies can acquire symbionts from the environment their dual mode of symbiont acquisition may enable rapid adaptation.
Materials and Methods
Sample Collection
Samples were collected from a reef in front of the Interuniversity Institute for Marine Sciences in Eilat (IUI), Gulf of Eilat (Aqaba), Red Sea (29° 30′ N, 34° 56′ E). Stylophora pistillata colonies were haphazardly collected from both shallow (2–6 m) and deep (24–26 m) water habitats in May, June, and/or July of 2009–2011 using scuba. S. pistillata were collected from three distinct age classes: adult colonies (∼15–30 cm width), juveniles colonies (∼0.5–2.8 cm width), and pelagic planulae. Field collection of animals complied with a permit issued by the Israel Nature and National Parks Protection Authority.
From each adult colony, a branch piece of approximately 2 cm in length was collected. Additionally, from each adult colony sampled in 2009 and 2010, spawned planulae were collected using planula collection nets [40]. No planulae were collected in 2011. Entire juvenile S. pistillata colonies were haphazardly collected from both depths in July of 2010. All samples were immediately frozen at −80°C or preserved in 95–100% ethanol for DNA analysis.
DNA Extraction, Amplification, and Denaturing Gradient Gel Electrophoresis
Genomic DNA was extracted from each adult and juvenile coral fragment [39], [41]. Reagent volumes were reduced 10 fold for planula extraction to accommodate the small sample volume [42]. The internal transcribed spacer 2 region of the ribosomal DNA was amplified using the ITSinfor2 and ITS2CLAMP primers developed by LaJeunesse and Trench [43] and amplification conditions described by LaJeunesse [44].
The PCR amplified ITS2 product was electrophoresed on an 8% polyacrylamide denaturing gradient gel (45–80% urea-formamide gradient) at a constant temperature (60°C) for 13 hours at 120 V [20]. Resulting denaturing gradient gel electrophoresis (DGGE) gels were stained with SYBR Green I nucleic acid gel stain (Invitrogen) for at least 20 minutes. Adult corals were run on the same gel with a maximum of 17 of their released planulae. Informative bands were excised [5] and sequenced on an Applied Biosystems 3730 capillary sequencer. For Symbiodinium type identification, sequence chromatographs were analyzed manually using Geneious (version 5.3.6) and compared to GenBank submissions.
Real-time PCR
Real-time PCR was used to evaluate possible low-levels of Symbiodinium that fell below the detection limit of DGGE. S. pistillata in the Red Sea has only been reported to associate with clade A in shallow water and clade C in deep water [37], [38]. Consequently, clade A (SymA28S) and C (SymC28S) Symbiodinium specific primer pairs were used to target the 28S ribosomal region [45].
Prior to running assays, primer optimizations and standard curves were run to confirm optimal primer and DNA concentrations. All samples were run in triplicate on an Applied Biosystems 7300 real-time PCR system. A 25 µl reaction included 0.1–2 ng/µl of DNA, Power SYBR green PCR mastermix (Applied Biosystems), and either 450 nM or 150 nM of clade A or C primers, respectively. Plates were run following standard amplification conditions [46] with a dissociation curve [47]. In addition to positive and negative controls, standard curves were run on each plate in duplicate over five, three-fold serial dilutions from 0.33 ng/µl to 0.004 ng/µl.
In order to accurately compare samples, strict values were set to consistently define a positive reaction. The cycle-threshold (CT) represents the PCR cycle at which sample fluorescence surpasses a fixed threshold limit. By comparing the standard curves run on each plate, a fixed threshold value was created based on average automatic software threshold settings for each primer pair [47]. As each primer pair was considered individually, the clade C primer pair had a fixed threshold of 0.57 while the clade A primer pair had a fixed threshold of 1.30.
In addition to a fixed threshold, we determined a cutoff CT value to consistently define a positive versus negative reaction. In competitive mixed clade trials [46], the target clade amplified consistently when it composed at least 0.9% of the total DNA template, setting conservative cutoff CT values of 34 and 32 for the clade A and C primers, respectively. Samples were considered positive if the average CT value was equal to or less than the cutoff CT.
Results
The Symbiodinium types present in S. pistillata samples collected from shallow (2–6 m) and deep (24–26 m) water habitats were identified using DGGE (Figure 1). Subsequent sequencing of the dominant bands confirmed that all shallow-water adults and their planulae (N = 266) contained Symbiodinium type A1. All deep-water adults and their planulae (N = 268) contained Symbiodinium type C72 (Figure 1). DGGE, however, cannot detect Symbiodinium that constitute less than 5–10% of the total symbiont population [20]. Thus, we also used real-time PCR to detect potential low-level Symbiodinium types [45]–[47].
Figure 1. DGGE gel of Symbiodinium types present in Stylophora pistillata adults (Ad) and released planulae (Pl).

Samples were collected from both shallow (Sh, 2–6 m) and deep (Dp, 24–26 m) water. In all cases, the planula DGGE fingerprints were identical to that of their maternal colony. The sequence of the upper dominant band in the DGGE fingerprint from shallow water samples was identical to Symbiodinium type A1 (accession AF333505), while the lower dominant band showed a 1 bp difference from A1, indicating S. pistillata hosts an A1 variant. All deep-water samples hosted type C72 (accession AY765407). Symbiodinium A1 and C72 standards (Std) were run on every gel.
In adult S. pistillata, the presence of low-level symbionts varied with depth. Low-level Symbiodinium were not detected in any shallow water adult colonies, but low-levels of clade A Symbiodinium were detected in some of the deep-water adults analyzed (Table 1). In 2009, all adult deep-water colonies sampled hosted only clade C, but in 2010, the majority of deep-water adult colonies sampled contained low-levels of clade A in addition to the abundant clade C symbionts. In 2011, only one of the sampled deep-water colonies hosted clade A at low-levels (Table 1).
Table 1. Abundant and low-level Symbiodinium clades in Stylophora pistillata.
| Year | Depth | Life Stage | N | Abundant Clade | Low-level Clade |
| 2009 | Shallow | Adult | 10 | A (10) | – |
| Planula | 28 | A (28) | – | ||
| Deep | Adult | 10 | C (10) | – | |
| Planula | 9 | C (9) | – | ||
| 2010 | Shallow | Adult | 9 | A (9) | – |
| Planula | 134 | A (134) | – | ||
| Juvenile | 25 | A (25) | C (1) | ||
| Deep | Adult | 10 | C (10) | A (7) | |
| Planula | 107 | C (107) | – | ||
| Juvenile | 25 | C (5); A (11) | – | ||
| A & C (9)* | |||||
| 2011 | Shallow | Adult | 10 | A (10) | – |
| Deep | Adult | 10 | C (10) | A (1) |
Symbiodinium clades present in shallow and deep-water S. pistillata adults, planulae, and juvenlies analyzed (N) with real time PCR. Numbers in parentheses denote the number of samples containing a given clade.
Samples that contained both A and C at levels detectable by DGGE, hence both clades were listed as abundant.
All planulae processed, from both depths, hosted a single Symbiodinium clade with no detectable low-levels of the second clade (Table 1). Even maternal colonies hosting both abundant and low-level Symbiodinium released planulae without the low-level symbiont. Using one-sample proportion tests, we tested a series of statistical null hypotheses to determine the lowest proportion of released planulae that may contain low-level Symbiodinium to determine if the lack of detection of additional symbionts was due to under-sampling. We were able to reject the null hypothesis that ≥2% of planulae released from shallow water adults may contain low-levels of clade C Symbiodinium (N = 161, df = 1, χ2 = 3.286, p-value = 0.035). Similarly, we rejected the null hypothesis that ≥3% of planulae released from deep-water adults may contain low-levels of clade A Symbiodinium (N = 116, df = 1, χ2 = 3.588, p-value = 0.029). Consequently, our sampling of planulae is most likely representative of what naturally occurs on the reef. Furthermore, based on the DGGE analysis, none of the planulae analyzed had a symbiont composition that differed from their parent.
Some juvenile S. pistillata colonies harbored a mixture of symbionts, in stark contrast to planulae, which only contained the abundant parental symbiont clade. Similar to the results for shallow water adult colonies, the majority (24/25) of juveniles collected in shallow water hosted only clade A Symbiodinium. One juvenile collected in shallow water, however, hosted both clade A and low-levels of C simultaneously. Of the 25 juveniles collected in deep-water, only five individuals solely contained clade C Symbiodinium. Nine other deep-water juveniles hosted both clades C and A Symbiodinium (Table 1). Eleven of the deep-water juveniles analyzed in the present study solely contained clade A Symbiodinium, with no detectable traces of clade C (Table 1), which is the dominant symbiont hosted by deep-water adult colonies.
Discussion
In obligate symbioses, in which the symbiont is vertically transmitted from parent to offspring, the offspring are guaranteed to receive the obligate symbiont. Vertical symbiont transmission in itself, however, is not necessarily straightforward. If different parents within a species host different symbionts, or if the same parent hosts multiple genetically distinct symbionts, progeny may inherit all or any one of the symbionts. In the case of Stylophora pistillata, we detected low-level symbionts in some adult colonies, indicating the potential for diverse Symbiodinium combinations in planulae. Which symbiont(s) the offspring inherit may vary between and even within a single parent. In addition, if progeny that inherit symbionts can later acquire symbionts from the environment, the symbiont variation within the coral species may further increase. If the symbionts differ physiologically, then which of the various symbionts the progeny acquire (either from a parental or environmental source) may affect their fitness.
The Symbiodinium genetic identity in coral species with vertical symbiont transmission has not been investigated extensively and we are aware of only one study that determined the Symbiodinium inherited in eggs of the coral Montipora capitata in Hawai’i [34]. While only approximately 35% of coral species vertically transmit Symbiodinium, these coral species belong to several widely distributed, dominant coral genera, e.g. Porites, Montipora, and Pocillopora [48]. Consequently, investigating the symbiont identity throughout ontogenesis in the numerous ecologically dominant coral species with vertical symbiont transmission is ecologically relevant to understanding coral–algal symbioses and coral reefs in general. It is equally important to determine if species with vertical Symbiodinium transmission are capable of symbiont acquisition from the environment.
Our results corroborate previous Symbiodinium identification in adult S. pistillata in the Gulf of Eilat, whereby adult S. pistillata colonies host two different Symbiodinium clades as a function of depth [37], [38]. Several coral species, with horizontal or vertical symbiont transmission, host different Symbiodinium, either at the same depth or over a depth gradient [15], [49], [50]. By employing molecular techniques with finer resolution, it has been demonstrated that, in some of these species, colonies can host one Symbiodinium type at abundant levels, in addition to a second symbiont type present at low-levels [51]. Using real-time PCR, we uncovered that the S. pistillata adult colonies sampled in shallow water only hosted clade A Symbiodinium, while colonies in deep-water could harbor low-levels of clade A in addition to the abundant levels of clade C Symbiodinium.
The symbiont depth zonation observed in S. pistillata in the Gulf of Eilat, at both abundant and low-levels, may be due to symbiont niche partitioning [4]. Clades A and C Symbiodinium hosted by adult S. pistillata colonies display differential responses to both elevated temperature and irradiance [38], [52]. Adult colonies hosting clade C are less resilient to thermal stress than colonies hosting clade A [38]. Additionally, cell size and chlorophyll content differ between Symbiodinium types A1 and C72 hosted by shallow and deep-water S. pistillata colonies, respectively [38]. These physiological differences may explain why in the present study, type C72 Symbiodinium appears mostly limited to S. pistillata in deeper water while type A1 is capable of surviving in colonies at both depths. The presence of clade A Symbiodinium in deep-water adults varied from individual to individual. Since we did not repeatedly sample the same colonies, we cannot ascertain whether low-levels of clade A are hosted permanently or transiently in some deep-water adult colonies. Hosting transient symbionts at abundant and low-levels, even those not known to associate with a given host, has been documented in temporal studies [20], [46], [53].
Regardless of whether S. pistillata maternal colonies hosted only one Symbiodinium clade, or had low-levels of the other Symbiodinium clade, all the planulae, from both depths, were released with only the abundant maternal Symbiodinium type. On the other hand, Padilla-Gomiño et al. [34] reported several instances in which Montipora capitata eggs harbored both parental and non-parental symbiont types. Although the techniques employed by Padilla-Gomiño et al. [34] may overestimate symbiont diversity [54], clade level differences were noted, with some of the non-parental symbiont clades and types known to associate with M. capitata in Hawai’i. As M. capitata vertically transmits Symbiodinium, the authors raised three hypotheses to explain these results, including environmental contamination, sampling bias, and the potentiality of horizontal symbiont acquisition in M. capitata eggs [34].
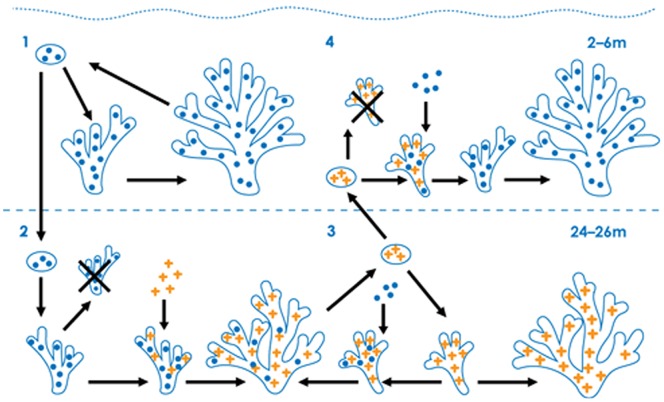
Our study provides evidence for the possibility of horizontal symbiont acquisition in a species with vertical symbiont transmission since S. pistillata planulae contained only one Symbiodinium type while some of the juvenile colonies, at both shallow and deep depths, harbored mixed symbioses. The incongruity between the symbionts in adult S. pistillata, the planulae and juveniles is likely explained by events occurring during the juvenile phase, and we present four plausible scenarios that may lead to the symbiont depth zonation observed in adult S. pistillata colonies. For example, if planulae released from shallow water colonies, with their clade A Symbiodinium complement, settle and metamorphose in deep-water, the resulting juveniles will initially contain clade A Symbiodinium (Figure 2). Two possible, not mutually exclusive, scenarios may then follow, both leading to the observed adult S. pistillata symbiosis with abundant clade C Symbiodinium in deep-water. First, juveniles that continue to maintain only clade A Symbiodinium may not survive to adulthood (Figure 2). Alternatively, if clade A containing juveniles in deep-water horizontally acquire clade C Symbiodinium, clade C may outcompete clade A and become the abundant symbiont present in adulthood (Figure 2). We may have witnessed a snapshot of this process in the nine juveniles that contained both Symbiodinium clades simultaneously. Similar scenarios may apply to shallow water S. pistillata (Figure 2).
Figure 2. Schematic depicting potential scenarios (1–4) of symbiont inheritance and acquisition throughout Stylophora pistillata ontogenesis.
(1) Shallow water adults and planulae only host clade A Symbiodinium (•). Planulae settling in shallow water will become adults hosting clade A. (2) Some planulae from shallow water adults may settle in deep-water. These juveniles may horizontally acquire clade C Symbiodinium (+) while juveniles hosting only clade A may perish (×). (3) Deep-water adults abundantly host clade C Symbiodinium (potentially low-levels of clade A), and planulae only inherit clade C. Upon settlement in deep-water, juveniles will maintain clade C or horizontally acquire clade A. (4) Planulae from deep-water adults may settle in shallow water. Juveniles only hosting clade C may perish; horizontally acquiring clade A may facilitate survival to adulthood.
Horizontal symbiont acquisition in the juvenile phase offers the most parsimonious explanation for the presence of multiple Symbiodinium clades within several juvenile colonies and the lack of multiple symbiont clades within all planulae analyzed. Alternatively, very low-levels of background symbionts could be present in some planulae, which could explain the presence of multiple symbiont clades in some juveniles. The real-time PCR assay reliably detected a Symbiodinium clade comprising at least 1% of the total symbiont population. Given the large volume of planulae released from a single parental colony over the course of the spawning season, it is possible that a small number of planulae inherit multiple symbionts, although statistical analyses indicated that this is very unlikely (see results). Given the tools utilized, for this coral species, horizontal symbiont acquisition in addition to vertical symbiont transmission seems plausible, although it remains to be determined whether juveniles that acquire symbionts from the environment survive to adulthood.
The occurrence of both vertical and horizontal symbiont transmission modes within a single host species, although not necessarily within a single individual, has previously been inferred in studies on vertically transmitted prokaryotic symbionts. These studies detected phylogenetic evidence of horizontal symbiont transmission [7]–[10], [55], although the frequency and life stage of acquisition were not determined. By sampling members of a coral species throughout its ontogeny, we were able to investigate a mutualism with inherited eukaryotic symbionts that may also engage in horizontal symbiont acquisition.
Conclusions
The ecological and evolutionary implications of employing both modes of symbiont transmission are substantial. Horizontal acquisition of novel symbionts may be a means by which coral species can adapt to environmental changes [56]. Although sexual reproduction in Symbiodinium occurs relatively infrequently [39], [57], [58], new host-symbiont combinations can emerge that may lead to novel, advantageous, and potentially specific symbioses [5]. Horizontal symbiont acquisition in adult corals, however, may either not occur or may be transient [19], [20], [41], [46], [59]. In coral species with horizontal symbiont acquisition, the juvenile stage appears to be more flexible in acquiring symbionts not present in the adult population [25]–[28], [42], [60]. Thus, horizontal symbiont acquisition may allow an individual juvenile to obtain, and subsequently maintain, novel symbionts. In turn, this may increase the juvenile’s fitness and survival as it grows into an adult coral, potentially enabling short-term acclimation on an individual level.
Horizontal symbiont acquisition, however, does not provide a mechanism for the perpetuation of the novel symbionts in subsequent generations. Since symbionts are not transferred to the progeny, the advantageous symbionts will be lost each generation when the progeny must acquire symbionts anew. In contrast, if juvenile corals with vertically transmitted symbionts are capable of acquiring novel, advantageous Symbiodinium that are maintained into adulthood, subsequent vertical symbiont inheritance to their progeny would facilitate the maintenance of novel Symbiodinium over generations.
Vertical symbiont inheritance, punctuated with horizontal symbiont acquisition, provides an evolutionary mechanism for adaptation to environmental changes through the acquisition and maintenance of advantageous symbionts. S. pistillata, for example, may benefit from both modes of symbiont transmission. On the one hand, the progeny are equipped with inherited Symbiodinium, eliminating the risk of not obtaining their obligate symbionts. On the other hand, juveniles may acquire novel symbionts, potentially increasing their chances of survival in a new environment. If the horizontally acquired novel symbionts improve host fitness, and become abundant in the resulting adult coral, they will be transferred to the brooded progeny, thereby perpetuating the novel symbiosis. Natural selection can then act on the genetic variation in the symbiosis, potentially establishing novel host-symbiont combinations that may be advantageous during changing environmental conditions.
Acknowledgments
We thank D. Goulet, J. Hoeksema, and B. Noonan for a critical reading of the manuscript; D. Goulet, R. Horwitz, G. Lavi, L. McMeekin, L. Mertens, and M. Polak and the staff of the Interuniversity Institute for Marine Sciences, Eilat for their help with sample collection and processing; S. Baerson and M. Mask for technical assistance with real-time PCR; and M. Charnley for generating Figure 2. Dedicated to Howard Lenhoff.
Funding Statement
Funding was provided by Sigma Xi Grants-In-Aid of Research, the Lerner Gray Memorial Fund of the American Museum of Natural History, the University of Mississippi Graduate School Council (KAB); an Israel Science Foundation grant (MF); and by the University of Mississippi College of Liberal Arts, the Office of Research and Sponsored Programs, and the National Science Foundation (IOS-0747205) (TLG). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Douglas AE (1995) The ecology of symbiotic micro-organisms. Advances in Ecological Research 26: 69–103. [Google Scholar]
- 2. Leigh EG Jr (2010) The evolution of mutualism. Journal of Evolutionary Biology 23: 2507–2528. [DOI] [PubMed] [Google Scholar]
- 3. Douglas AE (1998) Host benefit and the evolution of specialization in symbiosis. Heredity 81: 599–603. [Google Scholar]
- 4. Rowan R, Knowlton N (1995) Intraspecific diversity and ecological zonation in coral-algal symbiosis. Proceedings of the National Academy of Sciences of the United States of America 92: 2850–2853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. LaJeunesse TC (2005) “Species” radiations of symbiotic dinoflagellates in the Atlantic and Indo-Pacific since the Miocene-Pliocene transition. Molecular Biology and Evolution 22: 570–581. [DOI] [PubMed] [Google Scholar]
- 6. Sachs J, Skophammer R, Regus J (2011) Evolutionary transitions in bacterial symbiosis. Proceedings of the National Academy of Sciences of the United States of America 108: 10800–10807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Allen JM, Reed DL, Perotti MA, Braig HR (2007) Evolutionary relationships of Candidatus Riesia spp., endosymbiotic enterobacteriaceae living within hematophagous primate lice. Applied and Environmental Microbiology 73: 1659–1664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kikuchi Y, Fukatsu T (2003) Diversity of Wolbachia endosymbionts in heteropteran bugs. Applied and Environmental Microbiology 69: 6082–6090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Stewart FJ, Young CR, Cavanaugh CM (2008) Lateral symbiont acquisition in a maternally transmitted chemosynthetic clam endosymbiosis. Molecular Biology and Evolution 25: 673–687. [DOI] [PubMed] [Google Scholar]
- 10. Yang J, Sun J, Lee O, Wong Y, Qian P (2011) Phylogenetic diversity and community structure of sponge-associated bacteria from mangroves of the Caribbean Sea. Aquatic Microbial Ecology 62: 231–240. [Google Scholar]
- 11.Muscatine L (1990) The role of symbiotic algae in carbon and energy flux in reef corals. In: Dubinsky Z, editor. Ecosystems of the World: Coral Reefs. New York: Elsevier Scientific Publishing Company. 75–87.
- 12.Allemand D, Tambutté É, Zoccola D, Tambutté S (2011) Coral calcification, cells to reefs. In: Dubinsky Z, Stambler N, editors. Coral Reefs: An Ecosystem in Transition: Springer. 119–150.
- 13. Pochon X, Gates RD (2010) A new Symbiodinium clade (Dinophyceae) from soritid foraminifera in Hawai’i. Molecular Phylogenetics and Evolution 56: 492–497. [DOI] [PubMed] [Google Scholar]
- 14. Pochon X, Montoya-Burgos JI, Stadelmann B, Pawlowski J (2006) Molecular phylogeny, evolutionary rates, and divergence timing of the symbiotic dinoflagellate genus Symbiodinium . Molecular Phylogenetics and Evolution 38: 20–30. [DOI] [PubMed] [Google Scholar]
- 15. Iglesias-Prieto R, Beltran VH, LaJeunesse TC, Reyes-Bonilla H, Thome PE (2004) Different algal symbionts explain the vertical distribution of dominant reef corals in the eastern Pacific. Proceedings of the Royal Society B: Biological Sciences 271: 1757–1763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Rowan R (2004) Coral bleaching: thermal adaptation in reef coral symbionts. Nature 430: 742. [DOI] [PubMed] [Google Scholar]
- 17. Tchernov D, Gorbunov MV, de Vargas C, Yadav SN, Milligan AJ, et al. (2004) Membrane lipids of symbiotic algae are diagnostic of sensitivity to thermal bleaching in corals. Proceedings of the National Academy of Sciences of the United States of America 101: 13531–13535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Goulet TL, Cook CB, Goulet D (2005) Effect of short-term exposure to elevated temperatures and light levels on photosynthesis of different host-symbiont combinations in the Aiptasia pallida/Symbiodinium symbiosis. Limnology and Oceanography 50: 1490–1498. [Google Scholar]
- 19. Goulet TL (2006) Most corals may not change their symbionts. Marine Ecology Progress Series 321: 1–7. [Google Scholar]
- 20. Thornhill D, LaJeunesse T, Kemp D, Fitt W, Schmidt G (2006) Multi-year, seasonal genotypic surveys of coral-algal symbioses reveal prevalent stability or post-bleaching reversion. Marine Biology 148: 711–722. [Google Scholar]
- 21. Thornhill DJ, Fitt WK, Schmidt GW (2006) Highly stable symbioses among western Atlantic brooding corals. Coral Reefs 25: 515–519. [Google Scholar]
- 22. Thornhill DJ, Xiang Y, Fitt WK, Santos SR (2009) Reef endemism, host specificity and temporal stability in populations of symbiotic dinoflagellates from two ecologically dominant caribbean corals. PLoS ONE 4: e6262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Stat M, Loh W, LaJeunesse T, Hoegh-Guldberg O, Carter D (2009) Stability of coral–endosymbiont associations during and after a thermal stress event in the southern Great Barrier Reef. Coral Reefs 28: 709–713. [Google Scholar]
- 24. LaJeunesse TC, Pettay DT, Sampayo EM, Phongsuwan N, Brown B, et al. (2010) Long-standing environmental conditions, geographic isolation and host–symbiont specificity influence the relative ecological dominance and genetic diversification of coral endosymbionts in the genus Symbiodinium. Journal of Biogeography 37: 785–800. [Google Scholar]
- 25. Gomez-Cabrera MdC, Ortiz JC, Loh WKW, Ward S, Hoegh-Guldberg O (2008) Acquisition of symbiotic dinoflagellates (Symbiodinium) by juveniles of the coral Acropora longicyathus . Coral Reefs 27: 219–226. [Google Scholar]
- 26. Little AF, van Oppen MJH, Willis BL (2004) Flexibility in algal endosymbioses shapes growth in reef corals. Science 304: 1492–1494. [DOI] [PubMed] [Google Scholar]
- 27. Rodriguez-Lanetty M, Krupp DA, Weis VM (2004) Distinct ITS types of Symbiodinium in clade C correlate with cnidarian/dinoflagellate specificity during onset of symbiosis. Marine Ecology Progress Series 275: 97–102. [Google Scholar]
- 28. Abrego D, Van Oppen MJH, Willis BL (2009) Highly infectious symbiont dominates initial uptake in coral juveniles. Molecular Ecology 18: 3518–3531. [DOI] [PubMed] [Google Scholar]
- 29. Harii S, Yasuda N, Rodriguez-Lanetty M, Irie T, Hidaka M (2009) Onset of symbiosis and distribution patterns of symbiotic dinoflagellates in the larvae of scleractinian corals. Marine Biology 156: 1203–1212. [Google Scholar]
- 30. Loh WKW, Loi T, Carter D, Hoegh GO (2001) Genetic variability of the symbiotic dinoflagellates from the wide ranging coral species Seriatopora hystrix and Acropora longicyathus in the Indo-West Pacific. Marine Ecology Progress Series 222: 97–107. [Google Scholar]
- 31. Weis VM, Reynolds WS, deBoer MD, Krupp DA (2001) Host-symbiont specificity during onset of symbiosis between the dinoflagellates Symbiodinium spp. and planula larvae of the scleractinian coral Fungia scutaria . Coral Reefs 20: 301–308. [Google Scholar]
- 32. Benayahu Y, Schleyer MH (1998) Reproduction in Anthelia glauca (Octocorallia: Xeniidae). II. Transmission of algal symbionts during planular brooding. Marine Biology 131: 433–442. [Google Scholar]
- 33. van Oppen MJH (2004) Mode of zooxanthella transmission does not affect zooxanthella diversity in acroporid corals. Marine Biology 144: 1–7. [Google Scholar]
- 34. Padilla-Gamiño JL, Pochon X, Bird C, Concepcion GT, Gates RD (2012) From parent to gamete: vertical transmission of Symbiodinium (Dinophyceae) ITS2 sequence assemblages in the reef building coral Montipora capitata . PLoS ONE 7: e38440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Rinkevich B, Loya Y (1979) The reproduction of the Red Sea coral Stylophora pistillata. I. Gonads and planulae. Marine Ecology Progress Series 1: 133–144. [Google Scholar]
- 36.Veron JEN (2000) Corals of the World; Stafford-Smith M, editor: Townsville, Australia: Australian Institute of Marine Science.
- 37. Lampert-Karako S, Stambler N, Katcoff DJ, Achituv Y, Dubinsky Z, et al. (2008) Effects of depth and eutrophication on the zooxanthella clades of Stylophora pistillata from the Gulf of Eilat (Red Sea). Aquatic Conservation 18: 1039–1045. [Google Scholar]
- 38.Winters G, Beer S, Ben Zvi B, Brickner I, Loya Y (2009) Spatial and temporal photoacclimation of Stylophora pistillata: zooxanthella size, pigmentation, location and clade. Marine Ecology Progress Series: 107–119.
- 39. LaJeunesse TC (2001) Investigating the biodiversity, ecology, and phylogeny of endosymbiotic dinoflagellates in the genus Symbiodinium using the ITS region: In search of a “species” level marker. Journal of Phycology 37: 866–880. [Google Scholar]
- 40. Zakai D, Dubinsky Z, Avishai A, Caaras T, Chadwick NE (2006) Lunar periodicity of planula release in the reef-building coral Stylophora pistillata . Marine Ecology Progress Series 311: 93–102. [Google Scholar]
- 41. Goulet TL, Coffroth MA (2003) Stability of an octocoral-algal symbiosis over time and space. Marine Ecology Progress Series 250: 117–124. [Google Scholar]
- 42. Coffroth MA, Santos SR, Goulet TL (2001) Early ontogenetic expression of specificity in a cnidarian-algal symbiosis. Marine Ecology Progress Series 222: 85–96. [Google Scholar]
- 43. LaJeunesse TC, Trench RK (2000) Biogeography of two species of Symbiodinium (Freudenthal) inhabiting the intertidal sea anemone Anthopleura elegantissima (Brandt). Biological Bulletin 199: 126–134. [DOI] [PubMed] [Google Scholar]
- 44. LaJeunesse TC (2002) Diversity and community structure of symbiotic dinoflagellates from Caribbean coral reefs. Marine Biology 141: 387–400. [Google Scholar]
- 45. Yamashita H, Suzuki G, Hayashibara T, Koike K (2011) Do corals select zooxanthellae by alternative discharge? Marine Biology 158: 87–100. [Google Scholar]
- 46. LaJeunesse TC, Smith RT, Finney J, Oxenford H (2009) Outbreak and persistence of opportunistic symbiotic dinoflagellates during the 2005 Caribbean mass coral ‘bleaching’ event. Proceedings of the Royal Society B: Biological Sciences 276: 4139–4148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Correa A, McDonald M, Baker A (2009) Development of clade-specific Symbiodinium primers for quantitative PCR (qPCR) and their application to detecting clade D symbionts in Caribbean corals. Marine Biology 156: 2403–2411. [Google Scholar]
- 48. Baird AH, Guest JR, Willis BL (2009) Systematic and Biogeographical Patterns in the Reproductive Biology of Scleractinian Corals. Annual Review of Ecology, Evolution & Systematics 40: 551–571. [Google Scholar]
- 49. Baker AC (2003) Flexibility and specificity in coral-algal symbiosis: diversity, ecology, and biogeography of Symbiodinium . Annual Review of Ecology, Evolution & Systematics 34: 661–689. [Google Scholar]
- 50. Bongaerts P, Ridgway T, Sampayo E, Hoegh-Guldberg O (2010) Assessing the ‘deep reef refugia’ hypothesis: focus on Caribbean reefs. Coral Reefs 29: 309–327. [Google Scholar]
- 51. Jones AM, Berkelmans R, van Oppen MJ, Mieog JC, Sinclair W (2008) A community change in the algal endosymbionts of a scleractinian coral following a natural bleaching event: field evidence of acclimatization. Proceedings of the Royal Society B: Biological Sciences 275: 1359–1365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Reynolds JM, Bruns BU, Fitt WK, Schmidt GW (2008) Enhanced photoprotection pathways in symbiotic dinoflagellates of shallow-water corals and other cnidarians. Proceedings of the National Academy of Sciences of the United States of America 105: 13674–13678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Coffroth MA, Poland DM, Petrou EL, Brazeau DA, Holmberg JC (2010) Environmental symbiont acquisition may not be the solution to warming seas for reef-building corals. PLoS ONE 5: e13258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. LaJeunesse TC, Thornhill DJ (2011) Improved resolution of reef-coral endosymbiont (Symbiodinium) species diversity, ecology, and evolution through psbA non-coding region genotyping. PLoS ONE 6: e29013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Bright M, Bulgheresi S (2010) A complex journey: transmission of microbial symbionts. Nature Reviews Microbiology 8: 218–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Buddemeier RW, Baker AC, Fautin DG, Jacobs JR (2004) The adaptive hypothesis of bleaching In: Rosenberg E, editor. Coral Health and Disease Amsterdam: Springer-Verlag. 1–25.
- 57. Baillie BK, Belda BCA, Silvestre V, Sison M, Gomez AV, et al. (2000) Genetic variation in Symbiodinium isolates from giant clams based on random-amplified-polymorphic DNA (RAPD) patterns. Marine Biology 136: 829–836. [Google Scholar]
- 58. Santos SR, Taylor DJ, Kinzie RA III, Hidaka M, Sakai K, et al. (2002) Molecular phylogeny of symbiotic dinoflagellates inferred from partial chloroplast large subunit (23S)-rDNA sequences. Molecular Phylogenetics and Evolution 23: 97–111. [DOI] [PubMed] [Google Scholar]
- 59. LaJeunesse TC, Smith R, Walther M, Pinzón J, Pettay DT, et al. (2010) Host–symbiont recombination versus natural selection in the response of coral–dinoflagellate symbioses to environmental disturbance. Proceedings of the Royal Society B: Biological Sciences 277: 2925–2934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Abrego D, Van Oppen MJH, Willis BL (2009) Onset of algal endosymbiont specificity varies among closely related species of Acropora corals during early ontogeny. Molecular Ecology 18: 3532–3543. [DOI] [PubMed] [Google Scholar]