Abstract
Background. Hepatitis C virus (HCV) transmission among people who inject drugs remains a challenging public health problem. We investigated the risk of HCV transmission by analyzing the direct association of HCV with filters, water to dilute drugs, and water containers.
Methods. Experiments were designed to replicate practices by people who inject drugs and include routinely used injection equipment. HCV stability in water was assessed by inoculation of bottled water with HCV. Viral association with containers was investigated by filling the containers with water, inoculating the water with HCV, emptying the water, and refilling the container with fresh water. Transmission risk associated with drug preparation filters was determined after drawing virus through a filter and incubating the filter to release infectious particles.
Results. HCV can survive for up to 3 weeks in bottled water. Water containers present a risk for HCV transmission, as infectious virions remained associated with water containers after washing. Physical properties of the water containers determined the degree of HCV contamination after containers were refilled with water. HCV was also associated with filter material, in which around 10% of the viral inoculum was detectable.
Conclusions. This study demonstrates the potential risk of HCV transmission among injection drug users who share water, filters, and water containers and will help to define public health interventions to reduce HCV transmission.
Keywords: Hepatitis C virus, transmission, injecting drug users, paraphernalia
Hepatitis C virus (HCV) infection is considered a major public health problem, occurring in about 160 million people worldwide, representing 2.3% of the human population [1]. HCV infection causes acute and chronic liver diseases, including chronic hepatitis, cirrhosis, and hepatocellular carcinoma [2]. HCV infection is the most frequent indication for liver transplantation [3]. Different patient isolates are grouped into 7 genotypes and >100 subtypes within the genus Hepacivirus of the family Flaviviridae. Current treatment, a combination of polyethylene glycol–conjugated interferon alfa and ribavirin, is only effective in a fraction of patients and associated with severe side effects [4]. Because of the extreme genetic variability and high mutation rate of the virus, a prophylactic or therapeutic vaccine is so far not available. Currently, there are several direct-acting antiviral drugs in clinical development and 2 specific protease inhibitors, telaprevir and boceprevir, were licensed in 2011. However, these drugs will complement rather than replace current treatment regimens and are prone to rapidly inducing the emergence of viral resistance.
Transmission of HCV occurs via direct blood-to-blood contact. Risk factors to date are injection drug use, blood transfusions, accidental needlestick injuries, and other parental exposures, including nosocomial transmissions [5–8]. In the case of tattooing, piercing, acupuncture, and medical interventions, application of insufficient sterilized instruments can lead to transmission of the virus. The probability that HCV infections are attributed to sexual intercourse with HCV-positive partners is quiet low. Vertical transmission from mother to child prior to or during birth occurs in ≤4% of cases [9].
HCV infections among people who inject drugs (PWID) is a worldwide problem, and a recent study reported that globally about 10 million PWID have been infected with the virus [10]. Current estimates suggest that >60% of newly acquired infections occur in individuals who have injected drugs [5, 11]. In line with these results, seroprevalence of HCV among PWID is high, varying from 40% to 90%, and the incidence may be 20–40 infections per 100 person-years [12–15]. HCV contamination of injection equipment may occur at various stages of the drug preparation process, which involves multiple steps and different pieces of equipment. HCV cross-transmission between PWID is mainly associated with sharing syringes and drug preparation equipment, such as cookers, filters, water, and water containers [14, 16–20]. In the past 2 decades, the successful implementation of harm reduction programs has led to a decline in the prevalence of syringe sharing, but the shared use of other injection paraphernalia persists and contributes to extremely high transmission rates [12, 17, 21, 22].
For better understanding and prevention of HCV transmission among PWID, experimental systems simulating real-life conditions and quantifying risk are highly relevant. However, the adequate assessment of transmission risks and mechanisms between PWID has been difficult to study experimentally because of the lack of appropriate cell culture model systems and animal models that can be used to analyze HCV infectivity and replication. This roadblock has been overcome with the establishment of an HCV cell culture system based on the Japanese fulminant hepatitis (JFH1) HCV isolate, which reproduces the complete viral replication cycle in vitro [23–25]. This infection system was recently used to analyze the environmental stability of HCV and its susceptibility to chemical biocides in liquid suspensions and on inanimate surfaces [26, 27]. Two studies also evaluated the survival of HCV in contaminated syringes and on cookers in simulated drug injection scenarios [27, 28], but the stability and association of HCV with other drug preparation equipment has not yet been addressed so far.
In this study, we investigated the risk of transmission of HCV among PWID by analyzing the direct association of the virus with different drug preparation equipment. These results contribute to a better understanding of the mechanisms by which HCV is transmitted among this population. Moreover, our data highlight the risk of standard drug preparation procedures in the drug community and therefore should help to improve healthcare educational strategies aimed at reducing transmission via this route.
METHODS
Plasmids and Viruses
The plasmid pFK-Jc1 has been described recently [29]. Construct JcR-2a is a monocistronic reporter virus derived from the Jc1 chimera. The gene encoding the Renilla luciferase (RLuc) is fused N-terminally with the 16 N-terminal amino acids of the core protein and C-terminally with the foot-and-mouth disease virus (FMDV) 2A peptide coding region to allow proteolytic release of the RLuc protein from the HCV polyprotein [30].
Cell Culture
Huh7.5 cells were cultured in Dulbecco's modified Eagle's medium (Invitrogen) with 10% fetal bovine serum, 1 × nonessential amino acids (Invitrogen), 100 μg/mL streptomycin (Invitrogen), and 100 IU/mL penicillin (Invitrogen).
In Vitro Transcription, Electroporation, and Production of Cell Culture–Derived HCV
Infectious HCV particles were produced as described previously [26]. Briefly, Jc1 or JcR-2a plasmid DNA was linearized and transcribed into RNA, which was then electroporated into naive Huh7.5 cells. Virus-containing culture fluids were harvested after 48 or 72 hours by filtering fluids through a filter with a pore size of 0.45 μm. For determination of viral infectivity, cell-free supernatants were used to infect Huh7.5 target cells.
Determination of HCV Infectivity
Titers of infectious virus were determined by using a limiting dilution assay on Huh7.5 cells, with a few minor modifications, and the median tissue culture infectious dose (TCID50) was determined as described [23]. For determination of JcR-2a reporter activity, infected cells were washed with phosphate-buffered saline (PBS) and lysed in 35 µL of lysis buffer (Promega, Mannheim, Germany). Renilla luciferase activity was measured after addition of the luciferase substrate (1 µmol/L of coelenterazin; P.J.K., Kleinblittersdorf, Germany; in PBS) with 20 µL of sample for 1 second in a luminometer (Lumat LB9507), as described previously [30].
RNA Quantification by Quantitative Reverse Transcription Polymerase Chain Reaction (qRT-PCR)
RNA from cell culture supernatants was isolated using a Nucleo Spin RNAII Kit (Macherey/ Nagel, Düren, Germany). Two microliters of RNA sample was used for qRT-PCR analysis, using a Light Cycler 480 device (Roche, Mannheim, Germany). HCV-specific RT-PCRs were conducted in duplicate, using a 1-step RT-PCR LightCycler 480 RNA master hydrolysis probes kit (Roche) and the following HCV-specific probe (TIB Molbiol, Berlin, Germany) and primers (MWG-Biotech, Martinsried, Germany): A-195, 5-6FAM (6-carboxy-fluorescein)-AAA GGA CCC AGT CTT CCC GGC AAT T-TAMRA (tetra-chloro-6-carboxy-fluorescein)-3; S-146, 5-TCT GCG GAA CCG GTGAGT A-3; and A-219, 5-GGG CAT AGA GTG GGT TTA TCC A-3. Reactions were performed as described elsewhere [31].
Experimental Procedure to Assess HCV Stability in Drinking Water
To test the stability of HCV in drinking water over time, cell culture–grown Jc1 viral particles with a titer of about 106 TCID50/mL were spiked at different concentrations into 100 mL of drinking water in plastic containers (EUCO, Hamburg, Germany) and incubated for several days at room temperature. At indicated time points, 1-mL sample were collected and used to inoculate naive Huh7.5 cells. Infectivity and RNA copy numbers were determined as described above.
Experimental Procedure to Investigate HCV Association With Different Water Containers
To assess the association of HCV with different water containers, water containers made out of plastic (EUCO), aluminum (Red Bull, Austria), or glass (Coca-Cola, Berlin, Germany) were filled with 100 mL of standard drinking water (EUCO) and spiked with 40 µL cell culture–grown Jc1 virus with a titer of about 106 TCID50/mL. A 1 mL sample was collected (virus spike) and the remaining water was discarded after about 30 seconds. The same containers were filled with 100 mL of virus-free drinking water (EUCO) and directly tested for infectivity after concentration using centricons at 3500 × g for 20 minutes (Centricon Plus-70; Millipore, Carrigtwohill, County Cork, Ireland). Infectivity and RNA copy numbers were determined as described above.
Experimental Procedure to Investigate Association of HCV with Drug Preparation Filters
To assess the association of HCV with a drug preparation filter, JcR-2a reporter virus with a volume of 800 µL and a titer of about 105 TCID50/mL was drawn through a cigarette filter (OCB, Edeka, Hamburg, Germany), using a syringe (Braun, Melsungen, Germany). Contaminated filters were stored for 0, 3, 6, 12, 24, or 48 hours at room temperature, with or without standard household foil (Edeka, Hamburg, Germany). Residual infectivity was then assessed by incubation of filters in 1 mL of cell culture medium for 5 minutes at 37°C under shaking. Infectivity was determined by infection of naive Huh7.5 cells and subsequent measurement of luciferase reporter activity, as described above.
RESULTS
Survival of HCV in Bottled Drinking Water
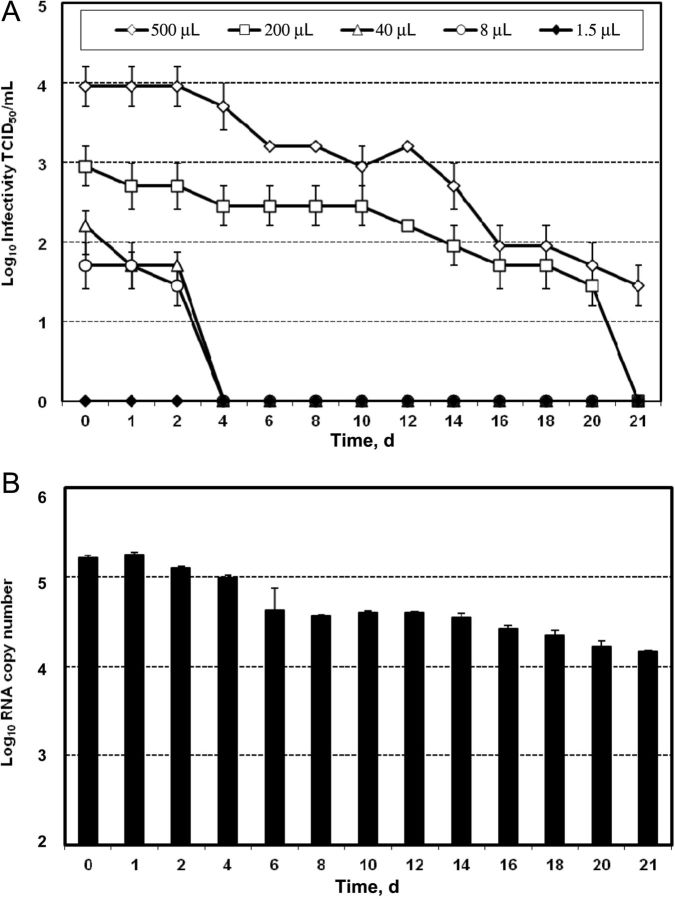
Epidemiologic reports clearly show that water used to mix drug solutions and to rinse equipment is shared among PWID and sometimes reused, indicating that water presents a high-risk source for viral cross-transmission of equipment [14, 18, 19, 32]. To evaluate the transmission risk via this route, we tested the stability of HCV in water. A total of 100 mL of drinking water in plastic containers was contaminated with HCV and incubated for several days at room temperature. Different doses of Jc1 virus, ranging from 500 µL to 1.5 µL, with a titer of 106 TCID50/mL were used to mimic different degrees of contamination and variable viral loads observed in patients. As depicted in Figure 1A, at the highest dose of 500 µL of virus (1:200 dilution), infectivity was reduced 10-fold after 10 days. We could still measure infectious HCV with a titer of 30 TCID50/mL after 21 days of incubation, demonstrating the stability of infectious HCV for >3 weeks in water under these experimental conditions. A viral dose of 200 µL showed a similar decay of infectivity, reaching undetectable levels after 21 days. Lower viral concentrations with an initial viral load of about 100 TCID50/mL resulted in a loss of infectious particles after 4 days of incubation, while for the lowest concentration of 1.5 µL of Jc1 suspension, no infectivity could be detected at any given time point. Similar viral decays could be observed with genotype 3–derived viral particles (data not shown). Since several studies that addressed the risk of HCV transmission are based on the detection of HCV RNA by qRT-PCR, we quantified the RNA copies over time in the samples spiked with 200 µL of virus (Figure 1B). The RNA copy numbers were only slightly reduced after several days and were clearly detectable after 21 days, with 104 viral genomes observed at this time point, demonstrating no correlation between the detection of genomic RNA and viral infectivity.
Figure 1.
Stability of hepatitis C virus (HCV) in bottled water. A, Cell culture-grown Jc1 viral particles with a titer of 106 median tissue culture infective doses (TCID50)/mL were spiked at different concentrations into 100 mL of drinking water in plastic containers and incubated for several days at room temperature. At indicated time points, samples were collected and used to inoculate naive Huh7.5 cells. Infectivity was determined by a limiting dilution assay, and viral titers are displayed as TCID50. A representative experiment of 3 independent repetitions is shown with SDs of infection replicates. B, HCV RNA at the respective time points of the 200 µL virus spike was isolated and quantified by real-time reverse transcription polymerase chain reaction. A representative experiment of 3 independent repetitions is shown with SDs of the means.
Association Between Infectious HCV Particles and Different Water Containers
In the next set of experiments, we explored whether infectious HCV particles were associated with water container material if the same container was reused in the injection process. In addition, we analyzed whether the physical properties of different water containers routinely used (and reused) in the drug community influence the degree of HCV contamination on reuse. To this end, water containers made out of plastic, aluminum, or glass were filled with 100 mL of drinking water and spiked with 40 µL of Jc1 virus with a titer of 106 TCID50/mL. A 1-mL sample was collected from the 100 mL water inoculated with virus termed virus-spike, and the remaining water was discarded. The same containers were subsequently refilled with 100 mL of virus-free drinking water, and water containers were tested for associated infectivity by concentration of the complete water volume termed recovery concentrated. As expected, the virus spike titer was comparable for all containers around 103 TCID50/mL, roughly correlating with the 1:2500 dilution of the viral stock solution in the water (Figure 2A). Importantly, for the plastic container, low viral amounts could be recovered from the material, indicating that HCV was associated with the plastic material. For the aluminum can, even higher viral recovery titers were observed, showing an association of virus with about 1% from the viral input. In contrast, no residual infectivity could be detected in the glass container, implying that this material exhibited the lowest association with HCV particles. Longer incubation times (up to 1 hour) of the virus inoculum in the different water containers did not significantly affect the recovery rates (data not shown). Determination of the RNA copy numbers in the respective samples revealed no clear correlation of RNA to infectivity, but the highest number of viral genome copies was detected for the aluminum container, which also showed the highest viral titers in the recovery sample (Figure 2B). For the glass container, RNA copy numbers were lower in the recovery sample but still detectable.
Figure 2.
Association of hepatitis C virus (HCV) with different container materials. A, Three water containers of different materials were filled with 100 mL of standard drinking water and spiked with 40 µL of cell culture–grown Jc1 virus. A 1-mL sample was collected (virus spike), and the remaining water was discarded. The same containers were filled with 100 mL of drinking water and tested for associated infectivity by concentration of the complete liquid suspension in centricons (recovery concentrated). After inoculation of viral samples on naive Huh7.5 cells, infectivity was determined by a limiting dilution assay, and viral titers are displayed as median tissue culture infective doses (TCID50). A representative experiment of 3 independent repetitions is shown with SDs of infection replicates. B, HCV RNA of the respective samples was isolated and quantified by real-time reverse transcription polymerase chain reaction. A representative experiment of 3 independent repetitions is shown with SDs of mean values.
Association of HCV with Drug Preparation Filters
After mixing the drug with water, a filter is placed into the cooker or spoon. The drug solution is drawn up through the filter by a syringe to remove impurities from the solution and to prevent needle blockage on injection. The frequency with which filters are shared by PWID varies from 50% to 77%, and the practice of saving filters for times when no drugs are available has been described [13, 33]. To investigate the risk of viral transmission by shared contaminated drug preparation filters, HCV reporter viruses in a volume 800 µL were drawn through a filter using a syringe. It is reported that the most common volume used to prepare a bag of heroin is about 800 µL [34]. The contaminated filters were stored for several hours at room temperature, with or without foil. Infectivity associated with the filters was released by incubation of filters with cell culture medium at 37°C under shaking. Infectivity was measured by inoculation of naive Huh7.5 cells and determination of luciferase activity. As depicted in Figure 3A, if filters were directly incubated to release possible trapped virus, we observed that 10% of the initial concentration of infectious viral particles was associated with the filter. When filters were not wrapped in foil, HCV infectivity decreased over time, reaching background levels 24 hours after filtering of the virus. When the contaminated filters were wrapped in foil, a practice often done by PWID, a similar reduction in HCV infectivity during the first few hours could be noted (Figure 3B). However, after 24 and 48 hours, infectivity was still detectable, demonstrating the prolonged stability of HCV in foil-protected filters. These results show that HCV can be associated with filter material at around 10% of the viral input and that foil wrapping of filters preserved viral stability.
Figure 3.
Association of hepatitis C virus (HCV) with drug preparation filters. Viral suspensions of 800 µL were drawn through a cigarette filter, using a syringe and present the filtered output. Contaminated filters were stored for indicated time periods at room temperature without (A) or with (B) standard household foil. Associated infectious particles were released by incubation of filters in 1 mL of cell culture medium, infectious particles in filters were released by incubation of filters in 1 mL of cell culture medium for 5 minutes at 37°C under shaking. Infectivity was determined by infection of naive Huh7.5 cells and performance of a luciferase reporter assay. A representative experiment of 3 independent repetitions is shown with SDs of mean values.
DISCUSSION
In this study, we performed a comprehensive experimental analysis to assess the HCV transmission risk associated with sharing drug preparation equipment, using the recently developed cell culture system for HCV infection and replication. Until now, experimental assays to determine HCV survival and stability in the environment were not described or were performed with surrogate markers (eg, antigens, RNA, and enzyme activity) for the presence and absence of infectious particles. The cell culture infection system used in this work is based on human hepatoma cells and viruses generated in vitro, which might differ from in vivo hepatocytes and patient-derived particles in some aspects. However, given the recent progress that has been made in HCV research in which this system is used [26–28, 35], analysis of cell culture–derived HCV particles is considered a state-of-the-art method for determining HCV infectivity and replication.
Injection drug use is an important public health issue around the world, with recent global epidemiological data revealing that about 10 million PWID have been infected with HCV and that more PWID have anti-HCV than HIV antibodies [10]. Importantly, multiple studies show that sharing drug preparation equipment significantly contributes to HCV cross-contamination of equipment, facilitating transmission [14, 16–20]. To address this oversight, the direct stability and association of HCV with injection material other than syringes was investigated in this study. We found that, depending on the viral dose, HCV can survive in water for >3 weeks and might thereby contribute to ongoing transmission of HCV in PWID networks when water is reused or shared. These results are in line with data showing the stability of the virus in serum for 3–4 weeks [26]. In syringes, HCV survival was dependent on syringe type, time, and temperature and infectivity could even be detected for up to 63 days in high void volume tuberculin syringes [28]. The transmission route of hepatitis B virus (HBV) is very similar to that of HCV, and HBV was also shown to be quite stable in the environment. The infectivity of HBV in plasma could be detected for at least 1 week, although longer incubation times were not investigated [36]. HCV RNA copy numbers in the water did not directly correlate with viral infectivity as previously demonstrated [26]. While viral titers measured by qRT-PCR are based on the detection of a small fragment of the viral genome, infectivity titers describe the number of intact, functional viral units required for replication and disease transmission. For HCV, the ratio of genome copies to infectivity may depend on the HCV genotype and the phase of infection of the viremic donor and ranges from 1 to 1000. Animal experiments suggested that the minimum infective dose (ie, the dose required to infect 50% of recipients) for HCV is approximately 10 viral particles [37]. However, so far no clear correlation between TCID50 and chimpanzee infectious dose has been established for HCV.
When we assessed the risk of HCV cross-transmission for different water container materials, we observed that HCV had the highest association with an aluminum can and a plastic container, whereas no residual virus could be detected in a glass bottle. In general, it is important to note that the container material itself can be associated with infectious virus after reuse, if the old water is replaced with clean water. The can had probably the highest association with HCV among the evaluated water containers because more of the original HCV-inoculated water remained in the can before it was refilled, owing to the can's small opening. For plastic bottles, in general, small water drops cling to the bottle neck; this is observed to a much lesser extent in the necks of glass bottles. We propose that the association between infectious HCV particles and water containers reflects differences in the interaction between residual contaminated water and the type and material of the container.
Investigation of the viral transmission risk with contaminated drug preparation filters revealed that up to 10% of the initial concentration of infectious viral particles was associated with the filter material. This association was time dependent and increased when the filters were wrapped in foil after contamination. Similar to other enveloped viruses, HCV seems to favor a liquid environment, because the foil protects the filters from drying out. We demonstrated in this study the high stability of HCV in water, which agrees with our previous observation that HCV survives much longer in suspension than in a dried environment [26, 27]. A recent study by Thibault et al described laboratory analysis of 620 pieces of injection material collected from PWID in France [38] in which HCV RNA was not detected in filters. However, only 10 filters were analyzed, and the HCV status of the PWID and the time at which the material was use were not recorded [38]. Limitations of our study are that simulation of HCV transmission by drug preparation equipment was performed under laboratory-controlled conditions that may not accurately reflect the natural transmission dynamics among PWID. Furthermore, in vivo hepatocytes and patient-derived particles might be slightly different, and not all HCV genotypes can be grown in vitro and therefore tested.
Recent studies report that sharing drug preparation equipment has not received much attention in public health research and prevention programs. Studies that analyzed the benefit of distributing sterile equipment to drug users have not been sufficiently conducted [18, 39]. HCV cross-transmission among PWID can be prevented if each PWID uses a single sterile container in which to heat drugs with sterile water, a sterile filter, and sterile syringes [22]. The findings described here should help to raise awareness and emphasize the risk of sharing drug preparation materials. Prevention messages and campaigns should be revised to notify PWID about the importance of eliminating all equipment-sharing practices, and supplying PWID with sterile equipment is recommended [32, 40].
In summary, we demonstrate that HCV can be directly associated with drug preparation equipment, like water, filters, and water containers. These findings add strong evidence to the high transmission rate of HCV among individuals who share these drug preparation materials and should help in the design of prevention strategies aimed at reducing HCV transmission.
Notes
Acknowledgments. We are grateful to Takaji Wakita and Jens Bukh, for JFH1 and J6CF isolates, respectively; Charles Rice, for Huh7.5 cells and the E9E10 monoclonal antibody; Heiner Wedemeyer, for support; Richard Brown, for critically reading the manuscript; and all members of the Department of Experimental Virology, Twincore, for helpful suggestions and discussions.
Disclaimer. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Drug Abuse or the National Institutes of Health.
Financial support. This work was supported by the DFG (grant STE 1954/1-1 to E. S.), the Helmholtz Centre for Infection Research (intramural young investigator award), the Helmholtz Association (grant SO-024 to T. P.), and the National Institute on Drug Abuse (award 1R01DA031597-01A1 to P. M.-G.).
Potential conflicts of interests. All authors: No reported conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Lavanchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect. 2011;17:107–15. doi: 10.1111/j.1469-0691.2010.03432.x. [DOI] [PubMed] [Google Scholar]
- 2.Hoofnagle JH. Hepatitis C: the clinical spectrum of disease. Hepatology. 1997;26:15S–20S. doi: 10.1002/hep.510260703. [DOI] [PubMed] [Google Scholar]
- 3.Seeff LB. Natural history of chronic hepatitis C. Hepatology. 2002;36:S35–46. doi: 10.1053/jhep.2002.36806. [DOI] [PubMed] [Google Scholar]
- 4.Manns MP, Wedemeyer H, Cornberg M. Treating viral hepatitis C: efficacy, side effects, and complications. Gut. 2006;55:1350–9. doi: 10.1136/gut.2005.076646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Alter MJ. Prevention of spread of hepatitis C. Hepatology. 2002;36:S93–8. doi: 10.1053/jhep.2002.36389. [DOI] [PubMed] [Google Scholar]
- 6.Esteban JI, Gomez J, Martell M, et al. Transmission of hepatitis C virus by a cardiac surgeon. N Engl J Med. 1996;334:555–60. doi: 10.1056/NEJM199602293340902. [DOI] [PubMed] [Google Scholar]
- 7.Macedo de Oliveira A, White KL, Leschinsky DP, et al. An outbreak of hepatitis C virus infections among outpatients at a hematology/oncology clinic. Ann Intern Med. 2005;142:898–902. doi: 10.7326/0003-4819-142-11-200506070-00007. [DOI] [PubMed] [Google Scholar]
- 8.Williams IT, Perz JF, Bell BP. Viral hepatitis transmission in ambulatory health care settings. Clin Infect Dis. 2004;38:1592–8. doi: 10.1086/420935. [DOI] [PubMed] [Google Scholar]
- 9.Yeung LT, King SM, Roberts EA. Mother-to-infant transmission of hepatitis C virus. Hepatology. 2001;34:223–9. doi: 10.1053/jhep.2001.25885. [DOI] [PubMed] [Google Scholar]
- 10.Nelson PK, Mathers BM, Cowie B, et al. Global epidemiology of hepatitis B and hepatitis C in people who inject drugs: results of systematic reviews. Lancet. 2011;378:571–83. doi: 10.1016/S0140-6736(11)61097-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shapshak P, Somboonwit C, Drumright LN, et al. Molecular and contextual markers of hepatitis C virus and drug abuse. Mol Diagn Ther. 2009;13:153–79. doi: 10.2165/01250444-200913030-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hagan H. Agent, host, and environment: hepatitis C virus in people who inject drugs. J Infect Dis. 2011;204:1819–21. doi: 10.1093/infdis/jir654. [DOI] [PubMed] [Google Scholar]
- 13.Hagan H, Pouget ER, Williams IT, et al. Attribution of hepatitis C virus seroconversion risk in young injection drug users in 5 US cities. J Infect Dis. 2010;201:378–85. doi: 10.1086/649783. [DOI] [PubMed] [Google Scholar]
- 14.Hagan H, Thiede H, Weiss NS, Hopkins SG, Duchin JS, Alexander ER. Sharing of drug preparation equipment as a risk factor for hepatitis C. Am J Public Health. 2001;91:42–6. doi: 10.2105/ajph.91.1.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lauer GM, Walker BD. Hepatitis C virus infection. N Engl J Med. 2001;345:41–52. doi: 10.1056/NEJM200107053450107. [DOI] [PubMed] [Google Scholar]
- 16.Hahn JA, Page-Shafer K, Lum PJ, et al. Hepatitis C virus seroconversion among young injection drug users: relationships and risks. J Infect Dis. 2002;186:1558–64. doi: 10.1086/345554. [DOI] [PubMed] [Google Scholar]
- 17.Koester S, Glanz J, Baron A. Drug sharing among heroin networks: implications for HIV and hepatitis B and C prevention. AIDS Behav. 2005;9:27–39. doi: 10.1007/s10461-005-1679-y. [DOI] [PubMed] [Google Scholar]
- 18.Strike C, Buchman DZ, Callaghan RC, et al. Giving away used injection equipment: missed prevention message? Harm Reduct J. 2010;7:2. doi: 10.1186/1477-7517-7-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thorpe LE, Ouellet LJ, Hershow R, et al. Risk of hepatitis C virus infection among young adult injection drug users who share injection equipment. Am J Epidemiol. 2002;155:645–53. doi: 10.1093/aje/155.7.645. [DOI] [PubMed] [Google Scholar]
- 20.Edlin BR, Carden MR. Injection drug users: the overlooked core of the hepatitis C epidemic. Clin Infect Dis. 2006;42:673–6. doi: 10.1086/499960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bluthenthal RN, Kral AH, Gee L, Erringer EA, Edlin BR. The effect of syringe exchange use on high-risk injection drug users: a cohort study. AIDS. 2000;14:605–11. doi: 10.1097/00002030-200003310-00015. [DOI] [PubMed] [Google Scholar]
- 22.Pouget ER, Hagan H, Des Jarlais DC. Meta-analysis of hepatitis C seroconversion in relation to shared syringes and drug preparation equipment. Addiction. 2012;107:1057–65. doi: 10.1111/j.1360-0443.2011.03765.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lindenbach BD, Evans MJ, Syder AJ, et al. Complete replication of hepatitis C virus in cell culture. Science. 2005;309:623–6. doi: 10.1126/science.1114016. [DOI] [PubMed] [Google Scholar]
- 24.Wakita T, Pietschmann T, Kato T, et al. Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat Med. 2005;11:791–6. doi: 10.1038/nm1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhong J, Gastaminza P, Cheng G, et al. Robust hepatitis C virus infection in vitro. Proc Natl Acad Sci U S A. 2005;102:9294–9. doi: 10.1073/pnas.0503596102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ciesek S, Friesland M, Steinmann J, et al. How stable is the hepatitis C virus (HCV)? Environmental stability of HCV and its susceptibility to chemical biocides. J Infect Dis. 2010;201:1859–66. doi: 10.1086/652803. [DOI] [PubMed] [Google Scholar]
- 27.Doerrbecker J, Friesland M, Ciesek S, et al. Inactivation and survival of hepatitis C virus on inanimate surfaces. J Infect Dis. 2011;204:1830–8. doi: 10.1093/infdis/jir535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Paintsil E, He H, Peters C, Lindenbach BD, Heimer R. Survival of hepatitis C virus in syringes: implication for transmission among injection drug users. J Infect Dis. 2010;202:984–90. doi: 10.1086/656212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pietschmann T, Kaul A, Koutsoudakis G, et al. Construction and characterization of infectious intragenotypic and intergenotypic hepatitis C virus chimeras. Proc Natl Acad Sci U S A. 2006;103:7408–13. doi: 10.1073/pnas.0504877103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Reiss S, Rebhan I, Backes P, et al. Recruitment and activation of a lipid kinase by hepatitis C virus NS5A is essential for integrity of the membranous replication compartment. Cell Host Microbe. 2011;9:32–45. doi: 10.1016/j.chom.2010.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Steinmann E, Brohm C, Kallis S, Bartenschlager R, Pietschmann T. Efficient trans-encapsidation of hepatitis C virus RNAs into infectious virus-like particles. J Virol. 2008;82:7034–46. doi: 10.1128/JVI.00118-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mateu-Gelabert P, Learned J, Sandoval M, Friedman SR. National Development Research Institutes,; 2011. Staying safe: strategies to help injection drug users avoid acquiring and transmitting HIV and hepatitis C. Unpublished prevention manual. [Google Scholar]
- 33.Hunter GM, Donoghoe MC, Stimson GV, Rhodes T, Chalmers CP. Changes in the injecting risk behaviour of injecting drug users in London, 1990–1993. AIDS. 1995;9:493–501. [PubMed] [Google Scholar]
- 34.Scott J, Winfield A, Kennedy E, Bond C. Laboratory study of the effects of citric and ascorbic acids on injections prepared with brown heroin. Int J Drug Policy. 2000;11:417–22. doi: 10.1016/s0955-3959(00)00068-2. [DOI] [PubMed] [Google Scholar]
- 35.Gottwein JM, Bukh J. Cutting the gordian knot-development and biological relevance of hepatitis C virus cell culture systems. Adv Virus Res. 2008;71:51–133. doi: 10.1016/S0065-3527(08)00002-X. [DOI] [PubMed] [Google Scholar]
- 36.Bond WW, Favero MS, Petersen NJ, Gravelle CR, Ebert JW, Maynard JE. Survival of hepatitis B virus after drying and storage for one week. Lancet. 1981;1:550–1. doi: 10.1016/s0140-6736(81)92877-4. [DOI] [PubMed] [Google Scholar]
- 37.Shata MT, Tricoche N, Perkus M, et al. Exposure to low infective doses of HCV induces cellular immune responses without consistently detectable viremia or seroconversion in chimpanzees. Virology. 2003;314:601–16. doi: 10.1016/s0042-6822(03)00461-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thibault V, Bara JL, Nefau T, Duplessy-Garson C. Hepatitis C transmission in injection drug users: could swabs be the main culprit? J Infect Dis. 2011;204:1839–42. doi: 10.1093/infdis/jir650. [DOI] [PubMed] [Google Scholar]
- 39.Gillies M, Palmateer N, Hutchinson S, Ahmed S, Taylor A, Goldberg D. The provision of non-needle/syringe drug injecting paraphernalia in the primary prevention of HCV among IDU: a systematic review. BMC Public Health. 2010;10:721. doi: 10.1186/1471-2458-10-721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Friedman SR, Mateu-Gelabert P, Sandoval M, Hagan H, Des Jarlais DC. Positive deviance control-case life history: a method to develop grounded hypotheses about successful long-term avoidance of infection. BMC Public Health. 2008;8:94. doi: 10.1186/1471-2458-8-94. [DOI] [PMC free article] [PubMed] [Google Scholar]