Abstract
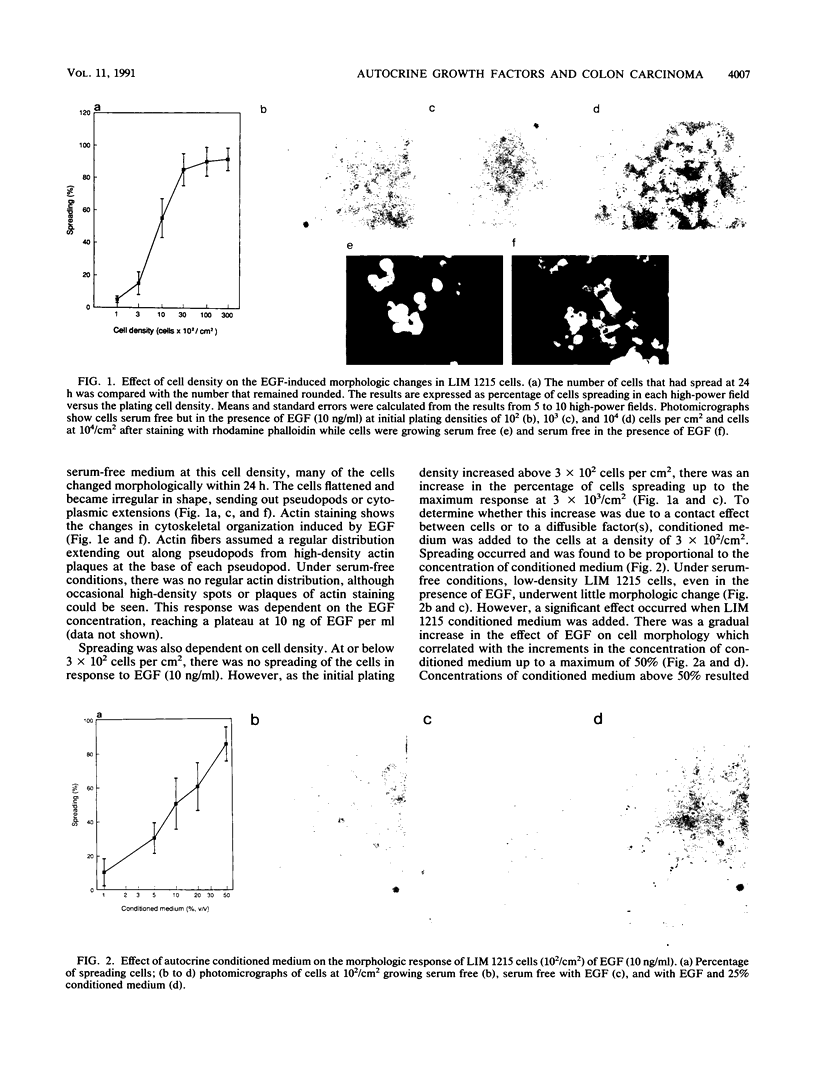
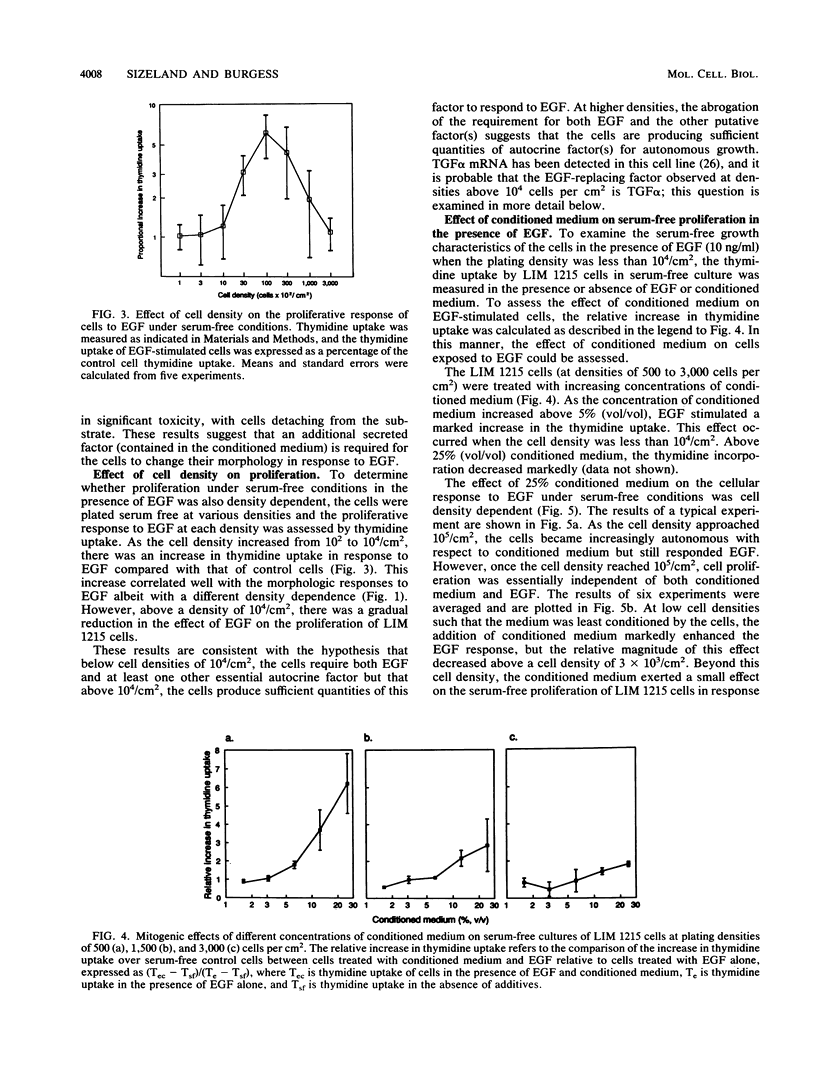
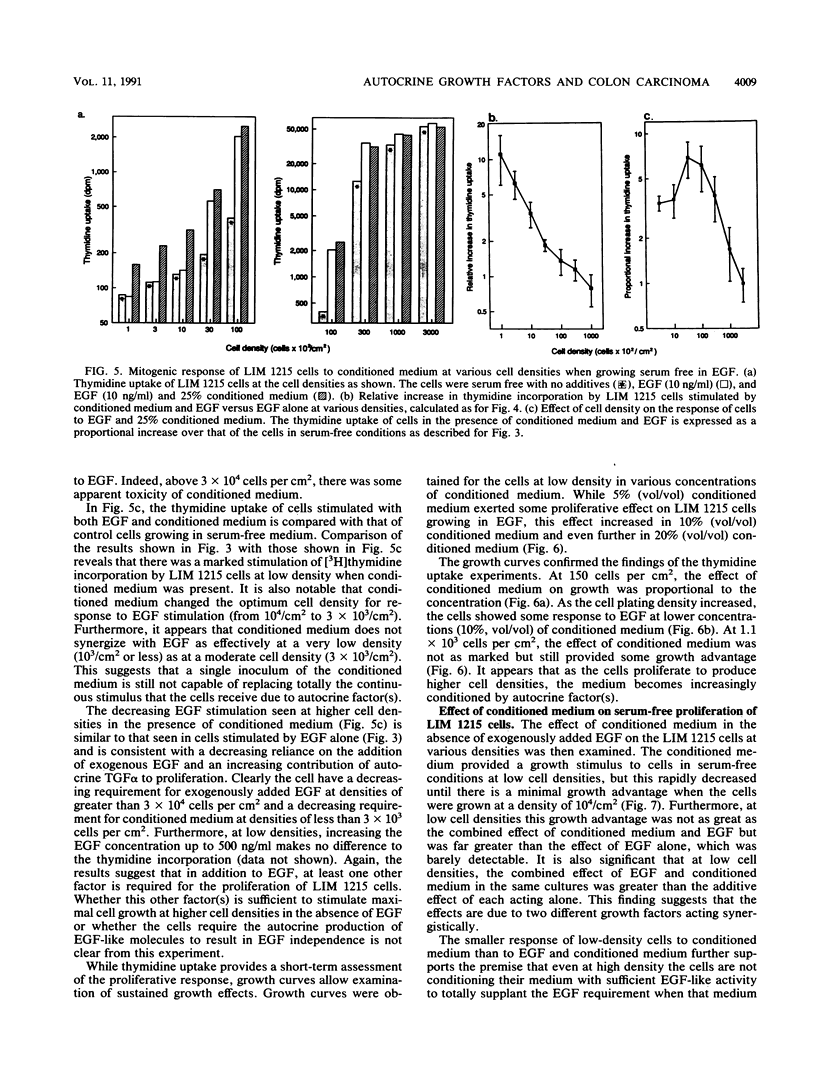
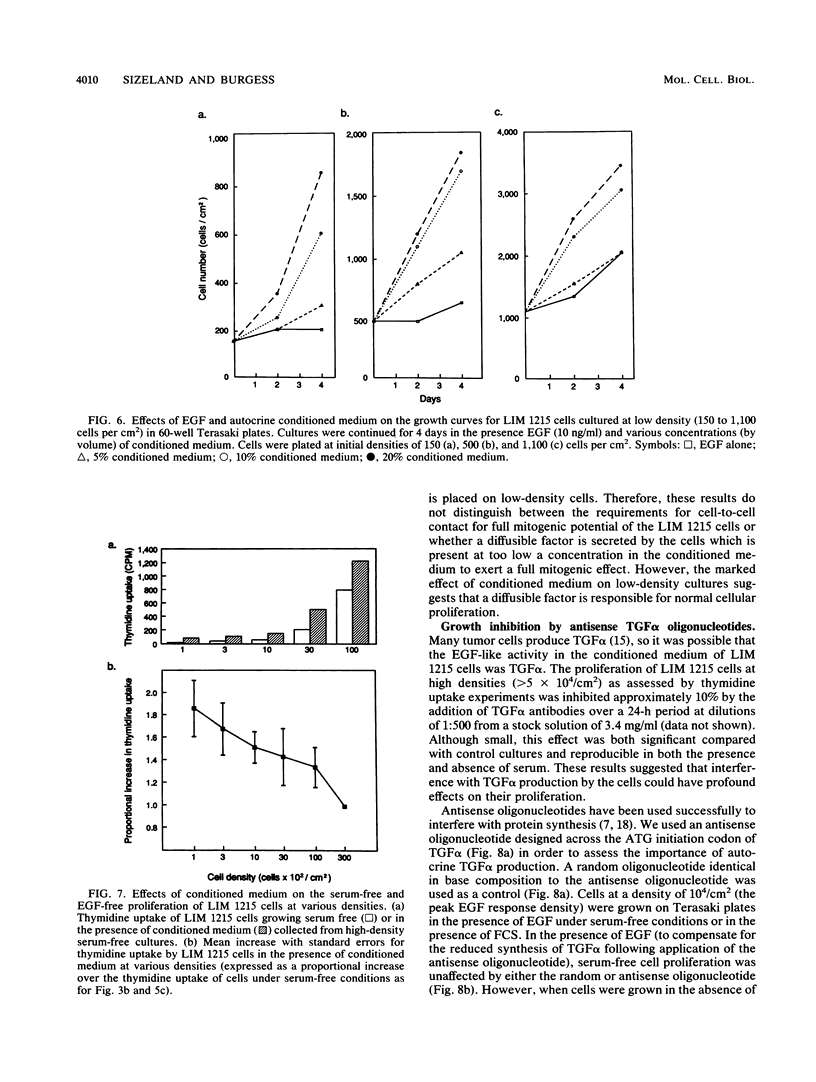
The role of autocrine growth factors in tumor cell growth has been difficult to prove. Our results indicate that more than one autocrine factor is required for the autonomous growth of the LIM 1215 colonic carcinoma cell line. Furthermore, the morphologic changes induced by epidermal growth factor (EGF) are also density dependent and appear to require a synergistic autocrine factor. The serum-free proliferation of the colonic carcinoma cell line LIM 1215 depends on cell density and the presence of EGF (A. Sizeland, S. Bol, and A.W. Burgess, Growth Factors 4:129-143, 1991). At cell densities below 10(4)/cm2, conditioned medium (from cells at a density of 10(5)/cm2) was required for the cells to elicit a mitogenic response to exogenous EGF. At higher cell densities (10(5)/cm2), the cells were independent of both exogenous EGF and conditioned medium. In addition, the EGF receptor was found to be phosphorylated on tyrosine in LIM 1215 cells proliferating at high density, suggesting that the autocrine production of transforming growth factor alpha (TGF alpha) and subsequent ligation to the EGF receptor was occurring. The proliferation of cells at high density was partly inhibited by TGF alpha antibodies but was almost completely inhibited by an antisense oligonucleotide to TGF alpha. The antisense inhibition could be overcome by the addition of EGF, indicating that the effect of the antisense TGF alpha oligonucleotide was on the production of autocrine TGF alpha. LIM 1215 cells were also observed to undergo morphologic changes (spreading and actin cable organization) in response to EGF. These changes were density dependent, but they occurred with a cell density dependence different from that of the proliferative response. These results suggest two possibilities: that the morphologic changes and proliferative responses have different sensitivities to the autocrine factors or that the actions of the autocrine factors are mediated through different signal transduction pathways.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ambesi-Impiombato F. S., Parks L. A., Coon H. G. Culture of hormone-dependent functional epithelial cells from rat thyroids. Proc Natl Acad Sci U S A. 1980 Jun;77(6):3455–3459. doi: 10.1073/pnas.77.6.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anzano M. A., Rieman D., Prichett W., Bowen-Pope D. F., Greig R. Growth factor production by human colon carcinoma cell lines. Cancer Res. 1989 Jun 1;49(11):2898–2904. [PubMed] [Google Scholar]
- Anzano M. A., Roberts A. B., Meyers C. A., Komoriya A., Lamb L. C., Smith J. M., Sporn M. B. Synergistic interaction of two classes of transforming growth factors from murine sarcoma cells. Cancer Res. 1982 Nov;42(11):4776–4778. [PubMed] [Google Scholar]
- Anzano M. A., Roberts A. B., Smith J. M., Sporn M. B., De Larco J. E. Sarcoma growth factor from conditioned medium of virally transformed cells is composed of both type alpha and type beta transforming growth factors. Proc Natl Acad Sci U S A. 1983 Oct;80(20):6264–6268. doi: 10.1073/pnas.80.20.6264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bade E. G., Feindler S. Liver epithelial cell migration induced by epidermal growth factor or transforming growth factor alpha is associated with changes in the gene expression of secreted proteins. In Vitro Cell Dev Biol. 1988 Feb;24(2):149–154. doi: 10.1007/BF02623892. [DOI] [PubMed] [Google Scholar]
- Bade E. G., Nitzgen B. Extracellular matrix (ECM) modulates the EGF-induced migration of liver epithelial cells in serum-free, hormone-supplemented medium. In Vitro Cell Dev Biol. 1985 Apr;21(4):245–248. doi: 10.1007/BF02620936. [DOI] [PubMed] [Google Scholar]
- Becker D., Meier C. B., Herlyn M. Proliferation of human malignant melanomas is inhibited by antisense oligodeoxynucleotides targeted against basic fibroblast growth factor. EMBO J. 1989 Dec 1;8(12):3685–3691. doi: 10.1002/j.1460-2075.1989.tb08543.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blay J., Brown K. D. Epidermal growth factor promotes the chemotactic migration of cultured rat intestinal epithelial cells. J Cell Physiol. 1985 Jul;124(1):107–112. doi: 10.1002/jcp.1041240117. [DOI] [PubMed] [Google Scholar]
- Bringman T. S., Lindquist P. B., Derynck R. Different transforming growth factor-alpha species are derived from a glycosylated and palmitoylated transmembrane precursor. Cell. 1987 Feb 13;48(3):429–440. doi: 10.1016/0092-8674(87)90194-2. [DOI] [PubMed] [Google Scholar]
- Böhmer R. M., Burgess A. W. An autocrine activity capable of substituting for serum in cell cultures. Growth Factors. 1989;1(4):347–356. doi: 10.3109/08977198909000258. [DOI] [PubMed] [Google Scholar]
- Coffey R. J., Jr, Goustin A. S., Soderquist A. M., Shipley G. D., Wolfshohl J., Carpenter G., Moses H. L. Transforming growth factor alpha and beta expression in human colon cancer lines: implications for an autocrine model. Cancer Res. 1987 Sep 1;47(17):4590–4594. [PubMed] [Google Scholar]
- Coffey R. J., Jr, Shipley G. D., Moses H. L. Production of transforming growth factors by human colon cancer lines. Cancer Res. 1986 Mar;46(3):1164–1169. [PubMed] [Google Scholar]
- Cuttitta F., Carney D. N., Mulshine J., Moody T. W., Fedorko J., Fischler A., Minna J. D. Bombesin-like peptides can function as autocrine growth factors in human small-cell lung cancer. 1985 Aug 29-Sep 4Nature. 316(6031):823–826. doi: 10.1038/316823a0. [DOI] [PubMed] [Google Scholar]
- De Larco J. E., Preston Y. A., Todaro G. J. Properties of a sarcoma-growth-factor-like peptide from cells transformed by a temperature-sensitive sarcoma virus. J Cell Physiol. 1981 Oct;109(1):143–152. doi: 10.1002/jcp.1041090116. [DOI] [PubMed] [Google Scholar]
- Derynck R., Roberts A. B., Winkler M. E., Chen E. Y., Goeddel D. V. Human transforming growth factor-alpha: precursor structure and expression in E. coli. Cell. 1984 Aug;38(1):287–297. doi: 10.1016/0092-8674(84)90550-6. [DOI] [PubMed] [Google Scholar]
- Deuel T. F., Huang J. S., Huang S. S., Stroobant P., Waterfield M. D. Expression of a platelet-derived growth factor-like protein in simian sarcoma virus transformed cells. Science. 1983 Sep 30;221(4618):1348–1350. doi: 10.1126/science.6310754. [DOI] [PubMed] [Google Scholar]
- Gewirtz A. M., Calabretta B. A c-myb antisense oligodeoxynucleotide inhibits normal human hematopoiesis in vitro. Science. 1988 Dec 2;242(4883):1303–1306. doi: 10.1126/science.2461588. [DOI] [PubMed] [Google Scholar]
- Halper J., Moses H. L. Purification and characterization of a novel transforming growth factor. Cancer Res. 1987 Sep 1;47(17):4552–4559. [PubMed] [Google Scholar]
- Heldin C. H., Wasteson A., Westermark B. Growth of normal human glial cells in a defined medium containing platelet-derived growth factor. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6611–6615. doi: 10.1073/pnas.77.11.6611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J. S., Huang S. S., Deuel T. F. Transforming protein of simian sarcoma virus stimulates autocrine growth of SSV-transformed cells through PDGF cell-surface receptors. Cell. 1984 Nov;39(1):79–87. doi: 10.1016/0092-8674(84)90193-4. [DOI] [PubMed] [Google Scholar]
- Johnsson A., Betsholtz C., Heldin C. H., Westermark B. Antibodies against platelet-derived growth factor inhibit acute transformation by simian sarcoma virus. Nature. 1985 Oct 3;317(6036):438–440. doi: 10.1038/317438a0. [DOI] [PubMed] [Google Scholar]
- Kaighn M. E., Kirk D., Szalay M., Lechner J. F. Growth control of prostatic carcinoma cells in serum-free media: interrelationship of hormone response, cell density, and nutrient media. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5673–5676. doi: 10.1073/pnas.78.9.5673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan P. L., Anderson M., Ozanne B. Transforming growth factor(s) production enables cells to grow in the absence of serum: an autocrine system. Proc Natl Acad Sci U S A. 1982 Jan;79(2):485–489. doi: 10.1073/pnas.79.2.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malden L. T., Novak U., Burgess A. W. Expression of transforming growth factor alpha messenger RNA in the normal and neoplastic gastro-intestinal tract. Int J Cancer. 1989 Mar 15;43(3):380–384. doi: 10.1002/ijc.2910430305. [DOI] [PubMed] [Google Scholar]
- Marquardt H., Hunkapiller M. W., Hood L. E., Twardzik D. R., De Larco J. E., Stephenson J. R., Todaro G. J. Transforming growth factors produced by retrovirus-transformed rodent fibroblasts and human melanoma cells: amino acid sequence homology with epidermal growth factor. Proc Natl Acad Sci U S A. 1983 Aug;80(15):4684–4688. doi: 10.1073/pnas.80.15.4684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moses H. L., Branum E. L., Proper J. A., Robinson R. A. Transforming growth factor production by chemically transformed cells. Cancer Res. 1981 Jul;41(7):2842–2848. [PubMed] [Google Scholar]
- Muller W. J., Lee F. S., Dickson C., Peters G., Pattengale P., Leder P. The int-2 gene product acts as an epithelial growth factor in transgenic mice. EMBO J. 1990 Mar;9(3):907–913. doi: 10.1002/j.1460-2075.1990.tb08188.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nistér M., Libermann T. A., Betsholtz C., Pettersson M., Claesson-Welsh L., Heldin C. H., Schlessinger J., Westermark B. Expression of messenger RNAs for platelet-derived growth factor and transforming growth factor-alpha and their receptors in human malignant glioma cell lines. Cancer Res. 1988 Jul 15;48(14):3910–3918. [PubMed] [Google Scholar]
- Nusse R. The int genes in mammary tumorigenesis and in normal development. Trends Genet. 1988 Oct;4(10):291–295. doi: 10.1016/0168-9525(88)90172-2. [DOI] [PubMed] [Google Scholar]
- Ozanne B., Fulton R. J., Kaplan P. L. Kirsten murine sarcoma virus transformed cell lines and a spontaneously transformed rat cell-line produce transforming factors. J Cell Physiol. 1980 Oct;105(1):163–180. doi: 10.1002/jcp.1041050118. [DOI] [PubMed] [Google Scholar]
- Roberts A. B., Anzano M. A., Lamb L. C., Smith J. M., Frolik C. A., Marquardt H., Todaro G. J., Sporn M. B. Isolation from murine sarcoma cells of novel transforming growth factors potentiated by EGF. Nature. 1982 Feb 4;295(5848):417–419. doi: 10.1038/295417a0. [DOI] [PubMed] [Google Scholar]
- Roberts A. B., Anzano M. A., Wakefield L. M., Roche N. S., Stern D. F., Sporn M. B. Type beta transforming growth factor: a bifunctional regulator of cellular growth. Proc Natl Acad Sci U S A. 1985 Jan;82(1):119–123. doi: 10.1073/pnas.82.1.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts A. B., Lamb L. C., Newton D. L., Sporn M. B., De Larco J. E., Todaro G. J. Transforming growth factors: isolation of polypeptides from virally and chemically transformed cells by acid/ethanol extraction. Proc Natl Acad Sci U S A. 1980 Jun;77(6):3494–3498. doi: 10.1073/pnas.77.6.3494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakamoto H., Mori M., Taira M., Yoshida T., Matsukawa S., Shimizu K., Sekiguchi M., Terada M., Sugimura T. Transforming gene from human stomach cancers and a noncancerous portion of stomach mucosa. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3997–4001. doi: 10.1073/pnas.83.11.3997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlessinger J., Geiger B. Epidermal growth factor induces redistribution of actin and alpha-actinin in human epidermal carcinoma cells. Exp Cell Res. 1981 Aug;134(2):273–279. doi: 10.1016/0014-4827(81)90426-2. [DOI] [PubMed] [Google Scholar]
- Sizeland A., Bol S., Burgess A. W. The action of epidermal growth factor (EGF) is limited to specific phases of the cell cycle in an EGF dependent colonic cell line. Growth Factors. 1991;4(2):129–143. doi: 10.3109/08977199109000264. [DOI] [PubMed] [Google Scholar]
- Stern D. F., Hare D. L., Cecchini M. A., Weinberg R. A. Construction of a novel oncogene based on synthetic sequences encoding epidermal growth factor. Science. 1987 Jan 16;235(4786):321–324. doi: 10.1126/science.3492043. [DOI] [PubMed] [Google Scholar]
- Stoker M., Gherardi E., Perryman M., Gray J. Scatter factor is a fibroblast-derived modulator of epithelial cell mobility. Nature. 1987 May 21;327(6119):239–242. doi: 10.1038/327239a0. [DOI] [PubMed] [Google Scholar]
- Temin H. M. Studies on carcinogenesis by avian sarcoma viruses. 3. The differential effect of serum and polyanions on multiplication of uninfected and converted cells. J Natl Cancer Inst. 1966 Aug;37(2):167–175. [PubMed] [Google Scholar]
- Todaro G. J., De Larco J. E., Cohen S. Transformation by murine and feline sarcoma viruses specifically blocks binding of epidermal growth factor to cells. Nature. 1976 Nov 4;264(5581):26–31. doi: 10.1038/264026a0. [DOI] [PubMed] [Google Scholar]
- Walker F., Nicola N. A., Metcalf D., Burgess A. W. Hierarchical down-modulation of hemopoietic growth factor receptors. Cell. 1985 Nov;43(1):269–276. doi: 10.1016/0092-8674(85)90032-7. [DOI] [PubMed] [Google Scholar]
- Whitehead R. H., Macrae F. A., St John D. J., Ma J. A colon cancer cell line (LIM1215) derived from a patient with inherited nonpolyposis colorectal cancer. J Natl Cancer Inst. 1985 Apr;74(4):759–765. [PubMed] [Google Scholar]
- Wiegant F. A., Blok F. J., Defize L. H., Linnemans W. A., Verkley A. J., Boonstra J. Epidermal growth factor receptors associated to cytoskeletal elements of epidermoid carcinoma (A431) cells. J Cell Biol. 1986 Jul;103(1):87–94. doi: 10.1083/jcb.103.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yarden Y., Schreiber A. B., Schlessinger J. A nonmitogenic analogue of epidermal growth factor induces early responses mediated by epidermal growth factor. J Cell Biol. 1982 Mar;92(3):687–693. doi: 10.1083/jcb.92.3.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Larco J. E., Todaro G. J. Growth factors from murine sarcoma virus-transformed cells. Proc Natl Acad Sci U S A. 1978 Aug;75(8):4001–4005. doi: 10.1073/pnas.75.8.4001. [DOI] [PMC free article] [PubMed] [Google Scholar]






