Abstract
Context:
The safety of insulin in the treatment of type 2 diabetes mellitus (T2DM) has recently undergone scrutiny.
Objective:
The objective of the study was to characterize the risk of adverse events associated with glucose-lowering therapies in people with T2DM.
Design and Setting:
This was a retrospective cohort study using data from the UK General Practice Research Database, 2000–2010.
Patients:
Patients comprised 84 622 primary care patients with T2DM treated with one of five glucose-lowering regimens: metformin monotherapy, sulfonylurea monotherapy, insulin monotherapy, metformin plus sulfonylurea combination therapy, and insulin plus metformin combination therapy. There were 105 123 exposure periods.
Main Outcome Measures:
The risk of the first major adverse cardiac event, first cancer, or mortality was measured. Secondary outcomes included these individual constituents and microvascular complications.
Results:
In the same model, and using metformin monotherapy as the referent, the adjusted hazard ratio (aHR) for the primary end point was significantly increased for sulfonylurea monotherapy (1.436, 95% confidence interval [CI] 1.354–1.523), insulin monotherapy (1.808, 95% CI 1.630–2.005), and insulin plus metformin (1.309, 95% CI 1.150–1.491). In glycosylated hemoglobin/morbidity subgroups, patients treated with insulin monotherapy had aHRs for the primary outcome ranging from 1.469 (95% CI 0.978–2.206) to 2.644 (95% CI 1.896–3.687). For all secondary outcomes, insulin monotherapy had increased aHRs: myocardial infarction (1.954, 95% CI 1.479–2.583), major adverse cardiac events (1.736, 95% CI 1.441–2.092), stroke (1.432, 95% CI 1.159–1.771), renal complications (3.504, 95% CI 2.718–4.518), neuropathy (2.146, 95% CI 1.832–2.514), eye complications (1.171, 95% CI 1.057–1.298), cancer (1.437, 95% CI 1.234–1.674), or all-cause mortality (2.197, 95% CI 1.983–2.434). When compared directly, aHRs were higher for insulin monotherapy vs all other regimens for the primary end point and all-cause mortality.
Conclusions:
In people with T2DM, exogenous insulin therapy was associated with an increased risk of diabetes-related complications, cancer, and all-cause mortality. Differences in baseline characteristics between treatment groups should be considered when interpreting these results.
The primary aim of diabetes management is the achievement and maintenance of normoglycemia (1). The importance of glucose control in reducing vascular outcomes is well known (2). However, the proliferation of new diabetes therapies and a spotlight on the safety profile of once-common therapies have led to debate about what the optimum target level of control should be (3–5).
Current guidelines recommend metformin monotherapy after the failure of diet and exercise in type 2 diabetes (6). If blood glucose levels become difficult to maintain on metformin monotherapy, it is recommended that a sulfonylurea be added to the regimen or, in the case of contraindication, a dipeptidyl peptidase-4 inhibitor or a thiazolidinedione. After this, the guidelines become more complex, attempting to balance glucose control with other factors such as weight gain and hypoglycemia, reflecting the limited evidence comparing different regimens.
Exogenous insulin is one of the most established blood-glucose-lowering therapies, and its use in people with type 2 diabetes mellitus (T2DM) has grown markedly over recent years (7), consequent upon findings from the UK Prospective Diabetes Study and the availability of analog insulins that have increased comfort with insulin initiation and titration. Indeed, we have recently estimated that, in the United Kingdom, the number of people with T2DM treated with insulin increased from 37 000 in 1991 to 277 000 in 2010 (Holden, SE, unpublished data). Early insulization has recently been recommended in guidelines from the American Diabetes Association and European Association for the Study of Diabetes (8), but the risk–benefit profile of exogenous insulin in the management of people with T2DM has also undergone scrutiny (9–12).
Initial concerns regarding the utility of insulin in T2DM emerged from epidemiological data characterizing the mortality impact of insulin from a large, population-based study in Canada, in which a dose–response relationship was observed between insulin use and all-cause mortality (13). The investigators reported a three-fold increase in mortality in the highest-exposure group. Similarly, in an epidemiological study of people with T2DM treated in UK primary care, those treated with insulin had a 50% increased mortality compared with those managed with a combination of metformin plus sulfonylurea (14). Other large-scale observational studies have reported a 2.5-fold increased risk of cardiac events in insulin-treated people with T2DM (15, 16). Insulin therapy has been related to increased mortality in people with T2DM complicated by advanced heart failure (17).
Potential mechanisms that may account for any relationship between exogenous insulin and adverse vascular outcomes in people with T2DM include cardiac arrhythmia leading to sudden death as a consequence of hypoglycemia (18), inflammation (19), and coagulation (20). Simultaneously, the link between insulin therapy and cancer is under considerable scrutiny (21, 22).
In view of these considerations, our objective was to compare the clinical outcomes of people with T2DM treated with insulin with those in patients treated with other glucose-lowering regimens.
Materials and Methods
Ethics statement
Studies using the General Practice Research Database (GPRD) are covered by ethics approval granted by the Trent Multicentre Research Ethics Committee (reference 05/MRE04/87). This study was granted GPRD Independent Scientific Advisory Committee approval (ISAC 11-17).
Design
The study used a retrospective-cohort design using data from the GPRD (replaced by the Clinical Practice Research Datalink from March 2012), a longitudinal, anonymized research database derived from nearly 600 primary care practices in the United Kingdom (23). The GPRD database contained records for more than 10 million people, of whom approximately 5 million were actively registered and could be followed up prospectively. Available data included demographics, medical history (including diagnoses and health contacts), clinical investigation results, and prescriptions. Diagnostic information in GPRD was recorded using the Read code classification, a UK general-practice standard. For the purposes of this study, data were selected from January 1, 2000, to December 31, 2010.
Patient selection
All patients were required to be incident cases based on a wash-in period of at least 180 days between the subject's registering at the practice and their first record of diabetes (either first diabetes diagnosis or first glucose-lowering prescription).
A diagnosis of T2DM was defined by one or more of the following decision rules: 1) more than one diagnosis recorded exclusively for T2DM; 2) prescription of two or more differing classes of oral antidiabetic drug; or 3) a diagnostic code indicative of T2DM (regardless of a conflicting diagnosis of type 1 or nonspecific diabetes) plus a prescription for an oral antidiabetic drug. Patients with diagnoses of both type 1 and type 2 diabetes in their patient record and an age at diagnosis of less than 35 years were defined as type 1 diabetes and therefore excluded.
Glucose-lowering regimen cohorts
Patients were defined by treatment cohorts based on the criteria of a minimum exposure of 180 days on the same glucose-lowering regimen. Glucose-lowering regimens were grouped in the following cohorts: metformin monotherapy, sulfonylurea monotherapy, insulin monotherapy, metformin plus sulfonylurea combination therapy, and insulin plus metformin combination therapy. Patients could be included in multiple cohorts if they switched regimens during the study period.
Clinical end points
In addition to death, six clinical outcomes were defined by the Read code recorded within the patient record (see Supplemental Appendix 1, published on The Endocrine Society's Journals Online web site at http://jcem.endojournals.org). These were: 1) MACE (major adverse cardiac event), defined as nonfatal myocardial infarction (MI), nonfatal ischemic or hemorrhagic stroke, or cardiovascular death; 2) cancer, including solid tumors and hematological forms; 3) visual deterioration, including blindness, retinopathy, and glaucoma; 4) diabetic neuropathy; and 5) renal failure. The primary end point of this study was the occurrence of the first of the three major clinical outcomes: all-cause mortality, incident cancer, or MACE. Cases with a prior history of large-vessel disease (LVD) and/or cancer were excluded.
The secondary end points of the study were the individual components of the primary end point and the microvascular complications. For each analysis, patients with a prior history of the relevant morbidity were excluded. In addition, for the analysis of renal impairment, patients with baseline serum creatinine above the normal range were excluded.
Statistical analysis
Outcome risk was compared using Cox proportional hazards models from the index date to the date of an outcome event. Cases were censored either at the date of switching to an alternative glucose-lowering regimen or at the person's final recorded data. For each outcome, the adjusted hazard ratio is given with the 95% confidence interval (CI). The proportional hazards assumption was tested by examining the Pearson correlation between Schoenfeld residuals and the rank of survival time for cases that progressed to an event. Baseline differences between cohorts were assessed by univariate analysis appropriate to the distribution of the parameter; however, differences were expected but accounted for using multivariate models. For the main analyses, metformin monotherapy was selected as the reference glucose-lowering regimen. Two additional analyses were performed. The first compared all therapies to insulin monotherapy for the primary and secondary end points. The second compared insulin to metformin for patients stratified by tertiles of baseline glycosylated hemoglobin (HbA1c) and baseline morbidity as measured by the Charlson comorbidity index (24) defined as low (Charlson index = 1) or high (Charlson index >1).
Model adjustment
The following baseline covariates were identified a priori and included in all models: age, gender, systolic blood pressure, HbA1c, total cholesterol, serum creatinine, body mass index, smoking status, other risk-factor management (antihypertensive, lipid-lowering, and antiplatelet therapy), duration of diabetes, prior history of cancer, LVD, microvascular disease, number of contacts with the general practitioner in the year prior to the index date, and the Charlson comorbidity index.
Analysis was performed using multiple imputation for missing values using logistic and linear regression as appropriate to the target variable.
Results
Subjects and baseline characteristics
There were 105 123 relevant, glucose-lowering regimen exposure periods for 84 622 individual cases, with a total of 298 530 person-years' follow-up. Baseline characteristics are characterized in Table 1. There were expected differences between the groups in terms of demographics and baseline characteristics. Patients treated with sulfonylurea monotherapy were older [68.1 years compared with 61.9 years overall (P < .001)] and a greater proportion had a serum creatinine level above 130 μmol/L [17.2% vs 4.5% overall (P < .001)]. Those treated with insulin either as monotherapy or in combination with metformin had higher baseline HbA1c (9.5% and 9.6%, respectively, compared with 8.7% overall). There were also differences in baseline morbidity, with subjects treated with sulfonylurea monotherapy and insulin monotherapy having a greater prevalence of prior LVD (18.9% and 22.3%, respectively, vs 13.1% overall).
Table 1.
Baseline Characteristics
| Parameter | All Subjects | Metformin | Sulfonylurea | Metformin + Sulfonylurea | Insulin | Insulin + Metformin |
|---|---|---|---|---|---|---|
| Number of persons, n (%) | 105 123 | 58 532 (53.4) | 16 218 (14.8) | 23 049 (21.0) | 3944 (3.6) | 3380 (3.1) |
| Follow-up, total (mean), y | 298 530 (2.8) | 163 790 (2.8) | 44 712 (2.8) | 65448 (2.8) | 12 842 (3.3) | 11 738 (3.5) |
| Age, mean (median), ya | 61.9 (12.8) | 60.7 (12.6) | 68.1 (12.6) | 61.5 (12.1) | 61.3 (14.5) | 57.6 (11.9) |
| Males, n (%) | 59 378 (56.5) | 32 700 (55.9) | 8817 (54.4) | 13 840 (60.0) | 2083 (52.8) | 1938 (57.3) |
| HbA1c, mean (SD), %b | 8.7 (1.9) | 8.5 (1.9) | 8.7 (2.1) | 9.0 (1.8) | 9.5 (2.2) | 9.6 (1.8) |
| SBP prior, mean (SD), mm Hgb | 138.8 (17.9) | 139.3 (17.5) | 139.6 (19.8) | 137.9 (17.3) | 134.6 (19.9) | 136.2 (17.0) |
| Smoked ever, n (%) | 62 969 (59.9) | 34 862 (59.6) | 9283 (57.2) | 14 189 (61.6) | 2444 (62.0) | 2191 (64.8) |
| TC prior, mean (SD), mmol/Lb | 4.9 (1.3) | 5.0 (1.3) | 5.0 (1.3) | 4.6 (1.2) | 4.7 (1.3) | 4.6 (1.2) |
| Weight male, mean (SD), kgb | 93.8 (18.5) | 96.6 (18.2) | 84.0 (15.7) | 93.6 (18.2) | 86.8 (18.9) | 96.5 (18.9) |
| Weight female, mean (SD), kgb | 82.2 (18.1) | 85.0 (17.8) | 72.1 (15.9) | 81.8 (17.5) | 76.8 (18.4) | 85.1 (17.6) |
| DM duration, mean (SD), yc | 2.3 (3.0) | 1.5 (2.3) | 2.1 (3.0) | 3.5 (3.1) | 5.2 (4.3) | 5.6 (3.8) |
| LVD prior, n (%)d | 13 742 (13.1) | 6491 (11.1) | 3070 (18.9) | 2816 (12.2) | 878 (22.3) | 487 (14.4) |
| Cancer, n (%) | 8693 (8.3) | 4373 (7.5) | 1926 (11.9) | 1766 (7.7) | 394 (10.0) | 234 (6.9) |
| Vision problems prior, n (%) | 24 170 (23.0) | 13 409 (22.9) | 4027 (24.8) | 5122 (22.2) | 880 (22.3) | 732 (21.7) |
| Creatinine >130 μmol/L prior, n (%)e | 4765 (4.5) | 1151 (2.2) | 2198 (17.2) | 634 (3.1) | 684 (21.1) | 98 (3.3) |
| Prior antihypertensives, n (%) | 72 686 (69.1) | 39 521 (67.5) | 11 348 (70.0) | 16 422 (71.2) | 2852 (72.3) | 2543 (75.2) |
| Prior lipid-lowering drugs, n (%) | 56 408 (53.7) | 29 376 (50.2) | 7285 (44.9) | 14 807 (64.2) | 2448 (62.1) | 2492 (73.7) |
| Prior antiplatelet drugs, n (%) | 45 196 (43.0) | 22 691 (38.8) | 7415 (45.7) | 11 156 (48.4) | 2063 (52.3) | 1871 (55.4) |
| Prior aspirin in previous year, n (%) | 44 594 (42.4) | 22 406 (38.3) | 7293 (45.0) | 11 032 (47.9) | 2020 (51.2) | 1843 (54.5) |
| Charlson Index, mean (SD) | 1.8 (1.2) | 1.6 (1.1) | 2.1 (1.5) | 1.9 (1.2) | 2.4 (1.6) | 2.0 (1.3) |
| GP contacts prior year, mean (SD) | 11.7 (10.1) | 10.9 (9.4) | 12.7 (11.1) | 11.7 (9.7) | 16.8 (13.2) | 14.0 (10.9) |
Abbreviations: DM, diabetes mellitus; GP, general practitioner; SBP, systolic blood pressure; TC, total cholesterol.
At index date (treatment initiation).
Nearest value to index date.
Duration of diabetes mellitus from presentation to index date.
Clinically emergent large-vessel disease before the index date (defined by Action to Control Cardiovascular Risk in Diabetes trial criteria).
Any record of a serum creatinine test result greater than 130 μmol/L prior to the index date; proportion refers to numbers with valid creatinine measurement.
Crude event rates
The number of recorded events and the crude rates for the selected outcomes are listed in Table 2. Overall, the mortality rate was 22.2 deaths per 1000 person-years, whereas the rates for MACE and incident cancer were 10.0 and 15.0 events per 1000 person-years, respectively. The rate for the primary end point was 35.6 events per 1000 person-years. For specific regimens, crude mortality rates were notably higher for sulfonylurea monotherapy (50.7 deaths per 1000 person-years) and insulin monotherapy (46.0 deaths per 1000 person-years). This was also true for the primary end point, with respective rates of 67.7 and 58.9 events per 1000 person-years.
Table 2.
Crude Event Frequency and Event Rate per 1000 Person-Years, by Glucose-Lowering Regimen
| Parameter | Primary End Point | All-Cause Mortality | MACEa | Cancerb | Eye Complicationsc | Neuropathy | Renal Complications | Combined Microvascular |
|---|---|---|---|---|---|---|---|---|
| All subjects | ||||||||
| Events | 8252 | 6630 | 2558 | 4029 | 8190 | 3633 | 1050 | 10 322 |
| Rate | 35.6 | 22.2 | 10.0 | 15.0 | 41.4 | 12.8 | 3.4 | 52.5 |
| Metformin monotherapy | ||||||||
| Events | 5929 | 2434 | 1936 | 2581 | 3637 | 1690 | 263 | 4515 |
| Rate | 36.6 | 14.9 | 11.6 | 15.6 | 32.6 | 10.7 | 1.5 | 42.0 |
| Metformin + sulfonylurea | ||||||||
| Events | 1626 | 1152 | 538 | 818 | 2250 | 825 | 149 | 1716 |
| Rate | 31.4 | 17.6 | 9.5 | 13.8 | 52.7 | 13.4 | 2.2 | 65.1 |
| Insulin + metformin | ||||||||
| Events | 281 | 184 | 87 | 173 | 396 | 247 | 28 | 2562 |
| Rate | 30.8 | 15.7 | 8.8 | 16.1 | 53.8 | 23.5 | 2.3 | 63.7 |
| Sulfonylurea monotherapy | ||||||||
| Events | 2088 | 2269 | 624 | 805 | 1387 | 579 | 435 | 585 |
| Rate | 67.7 | 50.7 | 17.6 | 20.9 | 48.6 | 13.6 | 9.6 | 88.2 |
| Insulin monotherapy | ||||||||
| Events | 518 | 591 | 164 | 231 | 520 | 292 | 175 | 464 |
| Rate | 58.9 | 46.0 | 16.7 | 20.3 | 66.4 | 25.9 | 13.5 | 70.2 |
Nonfatal MI, nonfatal ischemic or hemorrhagic stroke, or cardiovascular death.
Including solid-tumor and hematological forms.
Including blindness, retinopathy, and glaucoma.
Adjusted relative outcome by glucose-lowering regimen
The likelihood of adverse events varied by outcome and by glucose-lowering regimen. Table 3 lists the overall adjusted hazard ratios (aHRs) for each end point for each regimen, and within the same Cox model using metformin monotherapy as the reference regimen. For mortality, there were increased aHRs for insulin monotherapy (aHR 2.197, 95% CI 1.983–2.434), sulfonylurea monotherapy (1.749, 95% CI 1.643–1.863), and insulin plus metformin combination therapy (1.344, 95% CI 1.148–1.575).
Table 3.
aHRs by Glucose-Lowering Regimen
| Parameter | Primary End Point |
All-Cause Mortality |
MACE |
Cancer |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| aHR | 95% CI | P Value | aHR | 95% CI | P Value | aHR | 95% CI | P Value | aHR | 95% CI | P Value | |||||
| Metformin monotherapy (referent) | 1.000 | 1.000 | 1.000 | 1.000 | ||||||||||||
| Sulfonylurea monotherapy | 1.436 | 1.354 | 1.523 | <.0001 | 1.749 | 1.643 | 1.863 | <.0001 | 1.392 | 1.251 | 1.549 | <.0001 | 1.097 | 1.004 | 1.199 | .0410 |
| Metformin + sulfonylurea | 1.024 | 0.963 | 1.090 | .4454 | 1.100 | 1.022 | 1.184 | .0111 | 1.095 | 0.982 | 1.221 | .1035 | 0.956 | 0.877 | 1.043 | .3120 |
| Insulin monotherapy | 1.808 | 1.630 | 2.005 | <.0001 | 2.197 | 1.983 | 2.434 | <.0001 | 1.736 | 1.441 | 2.092 | <.0001 | 1.437 | 1.234 | 1.674 | <.0001 |
| Insulin + metformin | 1.309 | 1.150 | 1.491 | <.0001 | 1.344 | 1.148 | 1.575 | .0002 | 1.217 | 0.963 | 1.539 | .1001 | 1.394 | 1.176 | 1.651 | .0001 |
| Eye Complications | Neuropathy | Renal Complications | Combined Microvascular | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Metformin monotherapy (referent) | 1.000 | 1.000 | 1.000 | 1.000 | ||||||||||||
| Sulfonylurea monotherapy | 1.026 | 0.960 | 1.097 | .4514 | 1.127 | 1.011 | 1.256 | .0303 | 2.632 | 2.198 | 3.151 | <.0001 | 1.097 | 1.033 | 1.165 | .0024 |
| Metformin + sulfonylurea | 1.208 | 1.143 | 1.278 | <.0001 | 1.214 | 1.102 | 1.337 | .0001 | 1.389 | 1.122 | 1.720 | .0026 | 1.214 | 1.153 | 1.279 | <.0001 |
| Insulin monotherapy | 1.171 | 1.057 | 1.298 | .0026 | 2.146 | 1.832 | 2.514 | <.0001 | 3.504 | 2.718 | 4.518 | <.0001 | 1.363 | 1.240 | 1.498 | <.0001 |
| Insulin + metformin | 1.181 | 1.055 | 1.321 | .0037 | 2.318 | 1.964 | 2.736 | <.0001 | 1.667 | 1.091 | 2.547 | .0182 | 1.326 | 1.196 | 1.470 | <.0001 |
For MACE, there were increased hazard ratios for all therapies vs metformin: sulfonylurea monotherapy (1.392, 95% CI 1.251–1.549), metformin plus sulfonylurea (1.095, 95% CI 0.982–1.221), insulin monotherapy (1.736, 95% CI 1.441–2.092), and insulin plus metformin (1.217, 95% CI 0.963–1.539). For MI, all therapies had higher aHRs: sulfonylurea monotherapy (1.648, 95% CI 1.402–1.938), insulin monotherapy (1.954, 95% CI 1.479–2.583), and insulin plus metformin (1.438, 95% CI 1.027–2.014). For cerebrovascular events, sulfonylurea (1.237, 95% CI 1.101–1.391), metformin plus sulfonylurea (1.130, 95% CI 1.006–1.269), and insulin monotherapy (1.432, 95% CI 1.159–1.771) all had increased aHRs vs metformin. For cancer, there were increased aHRs for sulfonylurea (1.097, 95% CI 1.004–1.199), insulin monotherapy (1.437, 95% CI 1.234–1.674), and insulin plus metformin (1.394, 95% CI 1.176–1.651).
For the primary end, point, there were significantly increased aHRs for sulfonylurea monotherapy (1.436, 95% CI 1.354–1.523), insulin monotherapy (1.808, 95% CI 1.630–2.005), and insulin plus metformin (1.309, 95% CI 1.150–1.491).
For visual end points, metformin plus sulfonylurea (1.208, 95% CI 1.143–1.278), insulin monotherapy (1.171, 95% CI 1.057–1.298), and insulin plus metformin (1.181, 95% CI 1.055–1.332) all had increased aHRs compared with metformin monotherapy. For the neuropathy end point, metformin plus sulfonylurea (1.214, 95% CI 1.102–1.337), insulin monotherapy (2.146, 95% CI 1.832–2.514), and insulin plus metformin (2.318, 95% CI 1.964–2.736) all had increased aHRs compared with metformin monotherapy. For renal end points, sulfonylurea (2.632, 95% CI 2.198–3.151), metformin and sulfonylurea (1.389, 95% CI 1.122–1.720), insulin monotherapy (3.504, 95% CI 2.718–4.518), and insulin plus metformin (1.667, 95% CI 1.091–2.547) all had increased aHRs compared with metformin monotherapy.
Metformin vs insulin monotherapy subgroup analysis
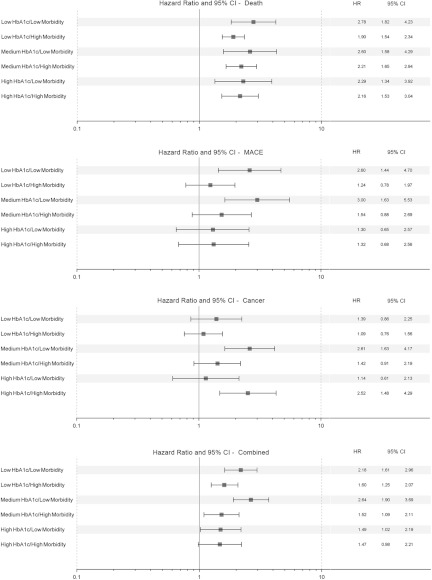
For each end point, aHRs for each of the six combinations of baseline HbA1c (low: ≤8.4%, medium: >8.4% and ≤10.2%, high: >10.2%) and morbidity (low: Charlson index 1, high: Charlson index >1) are illustrated in Figure 1. For mortality, the aHRs were increased across all six categories with a range from 1.897 [95% CI 1.541–2.336 (low HbA1c, high morbidity)] to 2.776 [95% CI 1.823–4.228 (low HbA1c, low morbidity)]. For the primary end point, all aHRs were significant with the exception of high morbidity, high HbA1c. The range was 1.469 [95% CI 0.978–2.206 (high HbA1c, high morbidity)] to 2.644 [95% CI 1.896–3.687 (medium HbA1c, low morbidity)].
Figure 1.
Adjusted hazard ratios for insulin monotherapy vs metformin monotherapy. Baseline HbA1c was as follows: low, ≤8.4; medium, >8.4 and ≤10.2; high, >10.2. Morbidity was as follows: low, Charlson index = 1; high, Charlson index > 1.
Insulin monotherapy vs all alternative glucose-lowering regimens
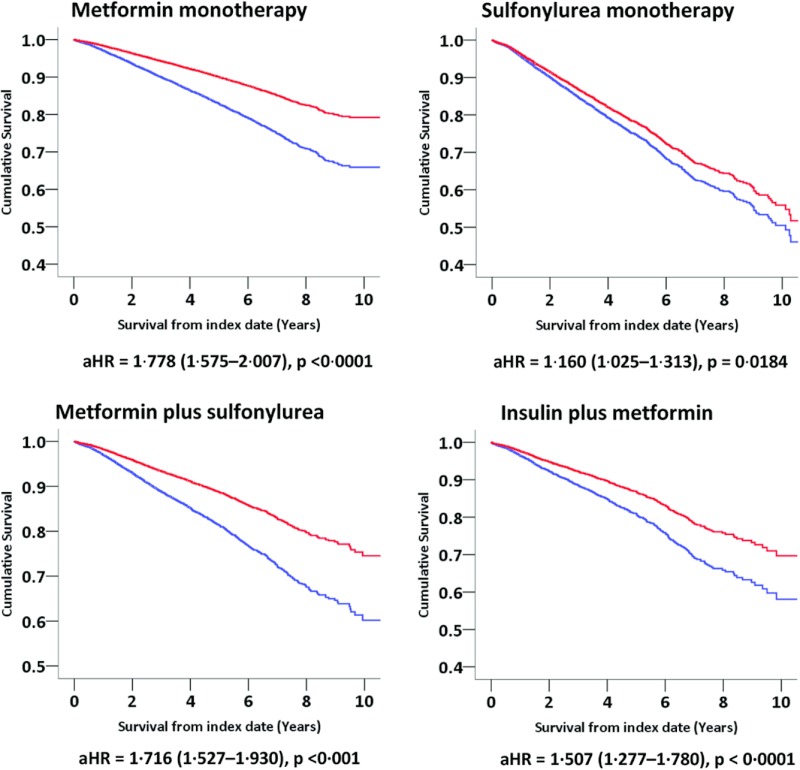
In order to address criticism that metformin monotherapy was used as the comparator regimen throughout, we have also provided for the primary endpoint a direct, adjusted comparison between each individual glucose-lowering regimen and insulin monotherapy (Figure 2). For the primary end point and after excluding the other regimens from the model (thus potentially different aHRs in the proportional hazards model), insulin monotherapy was consistently associated with increased risk compared with metformin monotherapy (1.778, 95% CI 1.575–2.007), sulfonylurea monotherapy (1.160, 95% CI 1.025–1.313), metformin plus sulfonylurea combination therapy (1.716, 95% CI 1.527–1.930), and insulin plus metformin combination therapy (1.507, 95% CI 1.277–1.780).
Figure 2.
Adjusted survival curves for each specific regimen vs insulin monotherapy for the primary end point. Blue line indicates insulin monotherapy. Red line indicates comparator. Model specification includes the following: age, gender, smoking history, prior primary care contacts, and Charlson index.
Discussion
This epidemiological study demonstrated an association for people with T2DM between treatment with exogenous insulin and worse outcomes for cardiovascular events, stroke, cancer, neuropathy, eye complications, renal disease, and all-cause mortality compared with patients treated with metformin monotherapy. Furthermore, sulfonylurea monotherapy resulted in increased hazard for a range of outcomes, although combination therapy with metformin attenuated risk to some extent. Insulin monotherapy resulted in worse outcome vs all other regimens for the primary end point and all-cause mortality.
These data are consistent with a variety of previous evidence observing worse outcomes in people with T2DM treated with insulin. For instance, there are data from large, population-based, observational studies that have found an association between increasing insulin use and serious events (13–17, 21, 22), although, to date, there has been a relative dearth of long-term clinical trial data evaluating the risk–benefit profile of exogenous insulin in T2DM.
The Diabetes and Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI)-1 and DIGAMI-2 trials examined outcome effects of different glucose-lowering regimens in people with T2DM after MI. In the DIGAMI-1 study (25), insulin-based regimens compared with other glucose-lowering management improved survival in patients with acute MI. However, the benefit of insulin was never confirmed in DIGAMI-2 (26). In this second study, patients with T2DM and MI were randomized to three different glucose-lowering strategies, with no significant differences in the total mortality or cardiovascular morbidity rates between glucose-lowering strategies. The interpretation of the discrepant outcomes between DIGAMI-1 and DIGAMI-2 was that the second study, unlike the first, had no difference in glucose control between the treatment arms. Indeed, there was a trend toward diverging mortality and morbidity rates to the benefit of patients without insulin treatment by the end of DIGAMI-2. A higher rate of nonfatal reinfarction and stroke occurred among patients receiving insulin. Furthermore, an observational analysis of long-term mortality in the DIGAMI-2 cohort has suggested that long-term, insulin-based therapy may be linked to not only an increase in nonfatal cardiovascular events but also to more noncardiovascular deaths (26).
Exogenous insulin is initiated with a view to improving glucose control and is usually commenced at a late stage in the natural history of the disease, with recent data illustrating a mean HbA1c at insulin initiation in Europe of approximately 10% (27). There is some evidence that tight glycemic control initiated soon after diagnosis is associated with long-term outcome benefits, but data from studies such as DIGAMI-2 and Action to Control Cardiovascular Risk in Diabetes (ACCORD) (28) suggest that undoubted improvement in glucose control using insulin may not offset related risks. The Outcome Reduction With Initial Glargine Intervention (ORIGIN) study has considered outcomes associated with early insulinization, comparing the impact of targeting a fasting plasma glucose less than 5.3 mmol/L with insulin glargine vs usual care over an estimated five-year treatment period in people with established cardiovascular disease coupled with glucose intolerance or early T2DM (29). This study found no difference in cardiovascular, mortality, or cancer outcomes between patients treated with insulin and those treated with standard care. However, of those patients treated according to standard care, 47% were treated with a regimen including sulfonylurea and 11% with a regimen including insulin.
There are multiple potential mechanisms that could link exogenous insulin with adverse outcomes such as cardiovascular end points and cancer in people with T2DM. Insulin initiation and titration result in weight gain in the region of 2 kg per 1% reduction achieved in HbA1c, an effect that may exacerbate both cancer and cardiovascular risk. Insulin is a growth factor known to have atherogenic and mitogenic effects, which may provide an adaptive advantage for malignant foci and potentiate the development of atherosclerotic vascular disease. Indeed, endogenous hyperinsulinemia has been linked to an increase in cancer incidence in people without diabetes (30), whereas epidemiological data have linked exogenous insulin therapy with an increased cancer incidence in people with T2DM (22). These events will then result in increased mortality risk.
One plausible explanation for the increased risk of microvascular events with insulin exposure observed here and in other populations (31) is the hemodynamic effect of insulin itself. Insulin is known to induce vasodilation (32) mediated via endogenous arteriolar nitric oxide pathways (33). These changes increase pulse pressure and decrease augmentation index (34), exposing the microvasculature to increased pulsatility, which causes irreversible mechanical damage leading to retinal and renal sequelae (31, 35). Furthermore, excessive insulin exposure may result in excessive nitric oxide production, resulting in oxidative stress (36).
There are various limitations inherent in our study. Follow-up in this study was relatively short (approximately three years), reflecting the duration that patients remained on a single therapeutic regimen. This may be less than the latency period for certain conditions to manifest as clinical events, particularly for some cancers.
Observational studies like this are susceptible to a number of common flaws (37), the one most relevant to this study being the possibility of confounding by indication. Because diabetes is a progressive disease, therapy choice will reflect this progression such that patients who are well controlled on first-line therapy, usually metformin, will remain on this therapy, whereas those who fail in the sense of increased levels of glycemia or development of complications will be treated more aggressively with the addition of other treatment options. Metformin is also contraindicated in patients with moderate renal disease. It is evident from the baseline characteristics (Table 1) that there are significant differences between the cohorts in terms of prior vascular morbidity (metformin 11.1% vs 18.9% and 22.3% for sulfonylureas and insulin, respectively) and other demographic and biochemical markers. Equally, patients treated with insulin had higher HbA1c than other groups, indicating increased insulin resistance, which would predict worse outcomes.
To partly address this issue, we have adjusted our models with a wide range of potentially confounding covariates, but these results must be considered with the caveat of residual confounding both within the candidate covariates and those factors that could not be represented within our models.
The use of routine data for research purposes raises questions over the accuracy of diagnostic and other information. Studies involving GPRD have tended to conclude that the quality of recording is good (38), but there remains potential for error. For the specific case of diabetes, there remains the possibility of misclassification between type 1 and type 2 diabetes. This is due to either nonspecific codes being used or type 2 patients being incorrectly recorded as type 1 and vice versa. This is compounded by the use of insulin-dependent diabetes and non-insulin-dependent diabetes as synonyms for type 1 and type 2 diabetes, respectively, being incorrectly applied. We have developed a series of decision rules to classify patients in whom there is some doubt over diabetes type using diagnostic, demographic, and therapy criteria, but we accept that there is still room for error. This error will not be random but likely biased toward those patients treated with insulin monotherapy.
We used metformin monotherapy as the referent glucose-lowering regimen in the Cox models; however, all regimens were compared in adjusted analyses within the same models. In addition, we compared all therapies individually with insulin monotherapy as the referent. Our findings were generally consistent across a range of primary and individual outcomes. The source of these data (GPRD) is highly regarded, and the data are from real life, with almost 300 000 person-years' follow-up. GPRD is a proprietary data source so others can replicate this analysis. In general, the patterns elicited were consistent and the order of magnitude of the adjusted hazard ratios and the dispersion of their CIs often striking. Therefore, although this analysis is admittedly imperfect—like all epidemiological studies—these data should raise justifiable concerns in relation to the optimal use of exogenous insulin therapy in clinical practice.
Although these data may raise questions around the use of insulin in people with T2DM, data from large controlled clinical trials such as ACCORD (28) have demonstrated no specific adverse safety signal with respect to insulin therapy. However, the design of such studies, largely protocol-led and focusing on glycemic-control targets and multiple therapies, does not readily lend itself to the specific evaluation of adverse events that may be associated with a particular therapy.
Exogenous insulin is the longest-established blood-glucose-lowering therapy and produces large reductions in blood-glucose levels; it can be life-saving for patients that absolutely require insulin-replacement therapy, such as those with type 1 diabetes. Nevertheless, there is a clear need to review the way in which exogenous insulin is used in people with type 2 diabetes and to establish in detail the risk–benefit profile at differing stages of the natural history of the disease and in phenotypically different sub-groups.
Supplementary Material
Acknowledgments
We are grateful to Sara Jenkins-Jones for editorial assistance in revising the final drafts of this manuscript.
This work was supported by the European Foundation for the Study of Diabetes by means of a grant to C.D.P. and C.Ll.M. for a related study. No funder had any role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Disclosure Summary: C.J.C. is director of and C.Ll.M. and C.D.P. consult for Pharmatelligence, a research consultancy receiving funding from pharmaceutical companies. C.D.P. has consulted for the following manufacturers of diabetic pharmaceuticals: BMS, Lilly, Novo Nordisk, and Sanofi-Aventis. C.J.C. has received research grants from various health-related organizations including Abbott, Astellas, Diabetes UK, the Engineering and Physical Sciences Research Council, the European Association for the Study of Diabetes, Ferring, GSK, Lilly, the Medical Research Council, Medtronic, Merck, the National Health Service, Pfizer, Sanofi-Aventis, Shire, and Wyeth and consults for Amylin, Aryx, Astellas, Boehringer Ingelheim, BMS, Diabetes UK, Eisel, Ferring, GSK, Ipsen, Lilly, Medtronic, Merck, Pfizer, Sanofi-Aventis, Takeda, and Wyeth. M.E. and J.R.P. declare that they have no conflicts of interest with respect to this study.
Footnotes
- aHR
- Adjusted hazard ratio
- CI
- confidence interval
- DIGAMI
- Diabetes and Insulin-Glucose Infusion in Acute Myocardial Infarction
- GPRD
- General Practice Research Database
- HbA1c
- glycosylated hemoglobin
- LVD
- large-vessel disease
- MACE
- major adverse cardiac event
- MI
- myocardial infarction
- T2DM
- type 2 diabetes mellitus.
References
- 1. National Institute for Clinical Excellence Type 2 diabetes: the management of type 2 diabetes. 2008. http://guidance.nice.org.uk/CG66 Accessed August 8, 2012
- 2. Emerging Risk Factors Collaboration, Sarwar N, Gao P, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet. 2010;375:2215–2222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Yudkin JS, Richter B, Gale EA. Intensified glucose lowering in type 2 diabetes: time for a reappraisal. Diabetologia. 2010;53:2079–2085 [DOI] [PubMed] [Google Scholar]
- 4. European Medicines Agency 2010 European Medicines Agency update on ongoing benefit-risk review of Avandia, Avandamet and Avaglim. http://www.ema.europa.eu/docs/en_GB/document_library/Press_release/2010/07/WC500094981.pdf Accessed August 5, 2012
- 5. FDA 2010. Drug Safety Communication: Ongoing safety review of Actos (pioglitazone) and potential increased risk of bladder cancer after two years exposure. Available from URL http://www.fda.gov/Drugs/Drugsafety/ucm226214.htm Accessed August 5, 2012
- 6. Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycaemia in type 2 diabetes mellitus: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetologia. 2009;52:17–30 [DOI] [PubMed] [Google Scholar]
- 7. Currie CJ, Peters JR, Evans M. Dispensing patterns and financial costs of glucose-lowering therapies in the UK from 2000 to 2008. Diabet Med. 2010;27:744–752 [DOI] [PubMed] [Google Scholar]
- 8. American Diabetes Association Standards of medical care in diabetes. Diabetes Care. 2011;34:S11–S61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Rensing KL, Reuwer AQ, Arsenault BJ, et al. Reducing cardiovascular disease risk in diabetic persons with established macrovascular disease: can insulin be too much of a good thing? Diabetes Obes Metab. 2011;13:1073–1087 [DOI] [PubMed] [Google Scholar]
- 10. Nandish S, Bailon O, Wyatt J, Smith J, Stevens A, Lujan M, Chilton R. Vasculotoxic effects of insulin and its role in atherosclerosis: what is the evidence? Curr Atheroscler Rep. 2011;13:123–128 [DOI] [PubMed] [Google Scholar]
- 11. Lebovitz HE. Insulin: potential negative consequences of early routine use in persons with type 2 diabetes. Diabetes Care. 2011;34(suppl 2):s225–s230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Currie CJ, Johnson JA. The safety profile of exogenous insulin in people with type 2 diabetes: justification for concern. Diabetes Obes Metab. 2012;14:1–4 [DOI] [PubMed] [Google Scholar]
- 13. Gamble JM, Simpson SH, Eurich DT, Majumdar SR, Johnson JA. Insulin use and increased risk of mortality in type 2 diabetes: a cohort study. Diabetes Obes Metab. 2010;12:47–53 [DOI] [PubMed] [Google Scholar]
- 14. Currie CJ, Peters JR, Tynan A, et al. Survival as a function of HbA1c in people with type 2 diabetes: a retrospective cohort study. Lancet. 2010;375:481–489 [DOI] [PubMed] [Google Scholar]
- 15. Colayco DC, Niu F, McCombs JS, Cheetham TC. A1C and cardiovascular outcomes in type 2 diabetes: a nested case-control study. Diabetes Care. 2011;34:77–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Margolis DJ, Hoffstad O, Strom BL. Association between serious ischemic cardiac outcomes and medications used to treat diabetes. Pharmacoepidemiol Drug Safe. 2008;17:753–759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Smooke S, Horwich TB, Fonarow GC. Insulin-treated diabetes is associated with a marked increase in mortality in persons with advanced heart failure. Am Heart J. 2005;149:168–174 [DOI] [PubMed] [Google Scholar]
- 18. Nordin C. The case for hypoglycaemia as a proarrhythmic event: basic and clinical evidence. Diabetologia. 2010;53:1552–1561 [DOI] [PubMed] [Google Scholar]
- 19. Antoniades C, Tousoulis D, Marinou K, et al. Effects of insulin dependence on inflammatory process, thrombotic mechanisms and endothelial function, in patients with type 2 diabetes mellitus and coronary atherosclerosis. Clin Cardiol. 2007;30:295–300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Angiolillo DJ, Bernardo E, Ramírez C, et al. Insulin therapy is associated with platelet dysfunction in patients with type 2 diabetes mellitus on dual oral antiplatelet treatment. J Am Coll Cardiol. 2006;48:298–304 [DOI] [PubMed] [Google Scholar]
- 21. Bowker SL, Yasui Y, Veugelers P, Johnson JA. Glucose-lowering agents and cancer mortality rates in type 2 diabetes: assessing effects of time-varying exposure. Diabetologia. 2010;53:1631–1637 [DOI] [PubMed] [Google Scholar]
- 22. Currie CJ, Poole CD, Gale EAM. The influence of glucose-lowering therapies on cancer risk in type 2 diabetes. Diabetologia. 2009;52:1766–1777 [DOI] [PubMed] [Google Scholar]
- 23. Medicines and Healthcare Products Regulatory Agency The Clinical Practice Research Datalink (CPRD). 2012. http://www.cprd.com Accessed August 5, 2012
- 24. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis. 1987;40:373–383 [DOI] [PubMed] [Google Scholar]
- 25. Malmberg K, Norhammar A, Wedel H, Ryden L. Glycometabolic state at admission: important risk marker of mortality in conventionally treated patients with diabetes mellitus and acute myocardial infarction—long-term results from the Diabetes and Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) study. Circulation. 1999;99:2626–2632 [DOI] [PubMed] [Google Scholar]
- 26. Mellbin LG, Malmberg K, Norhammar A, Wedel H, Ryden L, for the DIGAMI 2 Investigators Prognostic implications of glucose-lowering treatment in persons with acute myocardial infarction and diabetes: experiences from an extended follow-up of the Diabetes Mellitus Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) 2 Study. Diabetologia. 2011;54:1308–1317 [DOI] [PubMed] [Google Scholar]
- 27. Liebl A, Jones S, Benroubi, et al. Clinical outcomes after insulin initiation in patients with type 2 diabetes: 6-month data from the INSTIGATE observational study in five European countries. Curr Med Res Opin. 2011;27:887–895 [DOI] [PubMed] [Google Scholar]
- 28. The Action to Control Cardiovascular Risk in Diabetes Study Group Effects of intensive glucose-lowering in type 2 diabetes. N Engl J Med. 2008;358:2545–2559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. ORIGIN Trial Investigators, Gerstein HC, Bosch J, et al. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367:319–328 [DOI] [PubMed] [Google Scholar]
- 30. Lipscombe LL, Goodwin PJ, Zinman B, McLaughlin JR, Hux JE. The impact of diabetes on survival following breast cancer. Breast Cancer Res Treat. 2008;109:389–395 [DOI] [PubMed] [Google Scholar]
- 31. Nkondi Mbadi AN, Longo-Mbenza B, Mvitu Muaka M, Mbungu FS, Lemogoum D. Relationship between pulse pressure, visual impairment and severity of diabetic retinopathy in sub-Saharan Africa. Mali Med. 2009;24:17–21 [PubMed] [Google Scholar]
- 32. Laakso M, Edelman SV, Brechtel G, Baron AD. Decreased effect of insulin to stimulate skeletal muscle blood flow in obese man. A novel mechanism for insulin resistance. J Clin Invest. 1990;85:1844–1852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Scherrer U, Randin D, Vollenweider P, Vollenweider L, Nicod P. Nitric oxide release accounts for insulin's vascular effects in humans. J Clin Invest. 1994;94:2511–2515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Westerbacka J, Wilkinson I, Cockcroft J, Utriainen T, Vehkavaara S, Yki-Järvinen H. Diminished wave reflection in the aorta. A novel physiological action of insulin on large blood vessels. Hypertension. 1999;33:1118–1122 [DOI] [PubMed] [Google Scholar]
- 35. Hashimoto J, Ito S. Central pulse pressure and aortic stiffness determine renal hemodynamics: pathophysiological implication for microalbuminuria in hypertension. Hypertension. 2011;58:839–846 [DOI] [PubMed] [Google Scholar]
- 36. Vadde Ramakrishna V, Jailkhani R. Oxidative stress in non-insulin-dependent diabetes mellitus (NIDDM) patients. Acta Diabetologica. 2008;45:41–46 [DOI] [PubMed] [Google Scholar]
- 37. ISPOR 2012 Good Outcomes Research Practices Index. http://www.ispor.org/workpaper/practices_index.asp Accessed August 5, 2012
- 38. Khan Nada F, Harrison Sian E, Rose Peter W. Validity of diagnostic coding within the General Practice Research Database: a systematic review. Br J Gen Pract. 2010;60:e128–e136 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.