Abstract
Klebsiella pneumoniae-associated pathology is largely mediated by neutrophilic inflammation. In this study, we administered Klebsiella pneumoniae to experimental guinea pig groups and the ELR-CXC chemokine antagonist CXCL8(3–72), ceftazidime, and dexamethasone to different groups, respectively. After 24 h, we assessed the animal's pulmonary inflammatory levels, including gross histopathology, airway neutrophilia, lung myeloperoxidase levels, expressions of CXCL8 and TNF, and airway bacterial loads. Compared with ceftazidime and dexamethasone treatments, the administration of the ELR-CXC chemokine antagonist CXCL8(3–72) alone was more effective than other methods, although it did not markedly attenuate the bacterial load. These results suggest new methods for the treatment of Klebsiella pneumoniae pathology.
1. Introduction
Klebsiella pneumoniae, a human intestinal flora, is the second most common cause of bacteremia which is frequently associated with hospital-acquired infections such as malignancy, cirrhosis, biliary tract disorders, and diabetes mellitus (DM) [1]. It accounts for a significant proportion of hospital-acquired urinary tract, pneumonias, and soft tissue infections, partly because of the frequent spreading of these organisms in hospital environments [2]. Pyogenic brain abscess, meningitis, and endophthalmitis, which are related to metastatic infections, are the most important characteristics of Klebsiella pneumoniae infections [1]. Lipopolysaccharide (LPS) is considered to be a major virulence determinant of Klebsiella, and mutations leading to lipid A anchor of its LPS are conditionally lethal and result in the enhancement of lethality [3]. Multidrug resistant Klebsiella pneumoniae organisms can cause nosocomial outbreaks, which are called extended-spectrum-beta-lactamase- (ESBL-) producer bacteria. The incidence of ESBL-producing strains has been steadily increasing over the past years thus limiting our therapeutic options because of the increase of drug resistance [4]. New and alternative measures are required to control further Klebsiella-associated hospital infections and their multidrug resistance. Chemokines and chemokines receptors are attractive pharmacological target for the treatment. They are secreted locally or in paracrine and autocrine fashions by leucocytes and tissue cells. They activate a G protein-coupled receptor which is involved in the attraction of leukocytes. The CXC chemokines (alpha chemokines) are classified into a subgroup dependent on the presence of the Glu-Leu-Arg (ELR) motif. This motif is conserved in all natural ligands of CXCR1, and CXCR2, and is essential for their activation by chemokines. Neutrophil recruitment is regulated by ELR-CXC and their activation is induced by the engagement of CXCR1 and/or CXCR2 [5]. CXCL8 (interleukin; (IL-8)), the prototypical ELR-CXC chemokine, is frequently detected in microbial infections, including Klebsiella infections [6]. CXCR1 acts as a receptor for CXCL8 and CXCL6 (granulocyte chemoattractant protein-2), while CXCR2 also binds with other ELR-CXC chemokines including those chemokines highlighted earlier. Compared to other chemokines, CXCL8 has a high affinity at these receptors [5]. ELR-CXC chemokines and other inflammatory mediators are mainly expressed by endothelial cells following bacterial activation in the airways [7, 8]. CXCR2 alone can produce a reduction in neutrophil infiltration, whilst CXCR1, and CXCR2, in combination, can regulate the neutrophil response to ELR chemokines. J. R. Gordon reformed human CXCL8 cDNA. They mutated lysine to arginine in the 11th position and glycine to praline in the 31th position. Then they got high-binding affinity without chemotactic activity product, named CXCL8(3–72) K11R/G31P (G31P in abbreviation). G31P is highly effective in blocking neutrophil recruitment into both microbial and nonmicrobial inflammatory responses [8–10].
2. Materials and Methods
2.1. Reagents and Bacteria
The following reagents were purchased: myeloperoxidase test kit (Nanjing, China), TRIzol RNA extraction kit, RT-PCR kit, and agarose (Dalian Takara Bio Inc., Dalian, China).The Klebsiella pneumoniae strain was donated by Ms. J. Wang of the First Affiliated Hospital of Dalian Medical University Laboratory. The concentration of bacteria was 109 cfu/mL (UV at Abs600). CXCL8, K11R/G31P chemokines were expressed and purified in our laboratory [8].
2.2. Guinea Pig Klebsiella pneumoniae Model
Six-week old guinea pigs of female gender and weighing about 250 g were obtained from the Dalian Medical University Laboratory Animal Center. All protocols followed the ethics guidelines regarding the treatment of animals used in experiments. We divided the animals into batches of five groups (n = 5 per group). The animals were anaesthetized with pentobarbital sodium (50 μg/kg). Group 1 guinea pigs were inoculated intraperitoneally with 109 cfu/mL of K. pneumoniae 200 μL (diluted with saline) and are referred to as the controls. Other three groups (from Group 1 to Group 4) were experimental groups which were given Klebsiella pneumoniae and different treatments (30 minutes before infection). Group 2 guinea-pigs were administered with G31P (500 μg/kg; i.p.), Group 3 with ceftazidime (200 mg/kg; s.c.), and Group 4 with dexamethasone (0.8 mg/kg; s.c.). Group 5 guinea-pigs were given saline only (s.c.). Animals were harvested 24 h after the bacterial/sham procedures. All experiments were repeated three times, with the depicted data being representative.
2.3. Myeloperoxidase Assay (MPO)
To determine myeloperoxidase (MPO) activity, BALF was incubated with o-dianisidine dihydrochloride in Hank's medium supplemented with 0.004% H2O2. The reaction was stopped by adding 1% NaN3, and MPO activity was determined by spectrophotometry(OD 460 nm).
2.4. Isolation of Total RNA from Lung Tissue
Lung tissue RNA was isolated using a TRIzol RNA purification kit, according to the supplier's instructions. Semiquantifications and purity assessments were performed by optical density (OD) measurements at 260 nm and 280 nm and samples stored at −80°C.
2.5. Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted with TRIzol reagent according to the manufacturer's protocol. Semi-quantifications and purity assessments were performed by optical density (OD) measurements at 260 nm and 280 nm. Total RNA was reverse transcribed into cDNA using an RNA PCR Kit (see the details in Table 1).
Table 1.
The sequence of primers and the length of amplification products.
| Name | Primer sequence | Length of product (bp) |
|---|---|---|
| β-actin | Forward 5-CGTAAGGACCTCTATGCCAACAC-3 Reverse 5-GACTCATCGTACTCCTGCTTGCT-3 |
246 |
| CXCL8 | Forward 5-TGCGATGCCAGTGTATTAAGATT-3 Reverse 5-CTCTTGAAGAACATGCTCACCAC-3 |
94 |
| TNF | Forward 5-CACTCATCTACTCCCAGGTCCTC-3 Reverse 5-ACTCTTGATGGCAGAGAGAAGGT-3 |
158 |
| IL-1α (CXCL1) | Forward 5-CTTCTGCCATTGACCATCTCTCT-3 Reverse 5-TCTGAGTTTTCGTAGGGATTCGG-3 |
126 |
bp: base pair; IL: interleukin; CXCL1: chemokine (C-X-C motif) ligand 1; TNF: tumor necrosis factor.
The first steps of the RT condition were 53°C for 30 min, 99°C for 5 min, and 5°C for 5 min. β-actin PCR reactions were performed as follows: initial denaturation at 94°C for 2 min, 30 cycles at 94°C for 30 s, 58°C for 30 s, and 72°C for 2 min, and a final extension at 72°C for 10 min. Other cytokines PCR reactions were performed as follows: initial denaturation at 94°C for 2 min, 30 cycles of 94°C for 30 s, 57°C for 30 s, 72°C for 2 min, and a final extension at 72°C for 10 min. The subsequent results were analysed using 1% agarose gel and a gelatinous imaging system in order to detect the integral absorbance (IOD).
2.6. Bronchoalveolar Lavage (BAL) Neutrophil Counting
Each guinea-pig was anesthetized by intraperitoneal injection of ketamine/xylazine. The trachea was exposed, and a bronchoalveolar lavage (BAL) was performed with 1 mL sterile PBS. BAL fluid (BALF) was subjected to 10-fold serial dilutions. Neutrophils were counted using a microscope.
2.7. Statistical Analyses
Statistical analysis of the data and group comparisons were made using a Student's t-test (two-tailed). Statistical significance was assigned with P ≤ 0.05. The results are expressed as the mean ± SEM.
3. Results
3.1. ELR-CXC Chemokine Antagonism Significantly Decreases Airway Neutrophil Recruitment and Ameliorates Pulmonary Level Inflammation
ELR-CXC chemokine antagonists play an effective role in airway neutrophil recruitment and exert an inflammatory action. We assessed their pulmonary anti-inflammatory effects 24 hours after giving bacteria. We found the lungs to be pink, soft to touch, and with no evidence of inflammatory cell infiltration in normal animals. The lungs of the Klebsiella-treated animals presented with substantial pleural hemorrhagia, the airways were congested with inflammatory cells, and the lung parenchyma was edematous. The lungs of the G31P-treated group exhibited light pleural pathology, and the airways and lung parenchyma displayed mild edema with few inflammatory cells present.
3.2. Bronchoalveolar Lavage (BAL) Analyses by MPO Assay
Neutrophil analyses were carried out by determining MPO activity. Group-1 animals contained about 37% neutrophils, while Group-2 contained only background numbers of neutrophils (P > 0.05 versus the normal control animals). BAL fluid MPO levels mirrored these results. The G31P treatments significantly reduced the MPO levels to near baseline (P > 0.05 versus the normal control animals, see Figure 1). Meanwhile, compared with the control group, G31P markedly reduced the expression levels of the inflammatory mediators IL-1, CXCL8, and TNF. We thus observed a guinea pig model of aspiration pneumoniae [11] and amelioration of the neutrophil responses of pneumonic guinea-pigs following the administration of G31P, which did not affect their ability to resist a Klebsiella infection.
Figure 1.
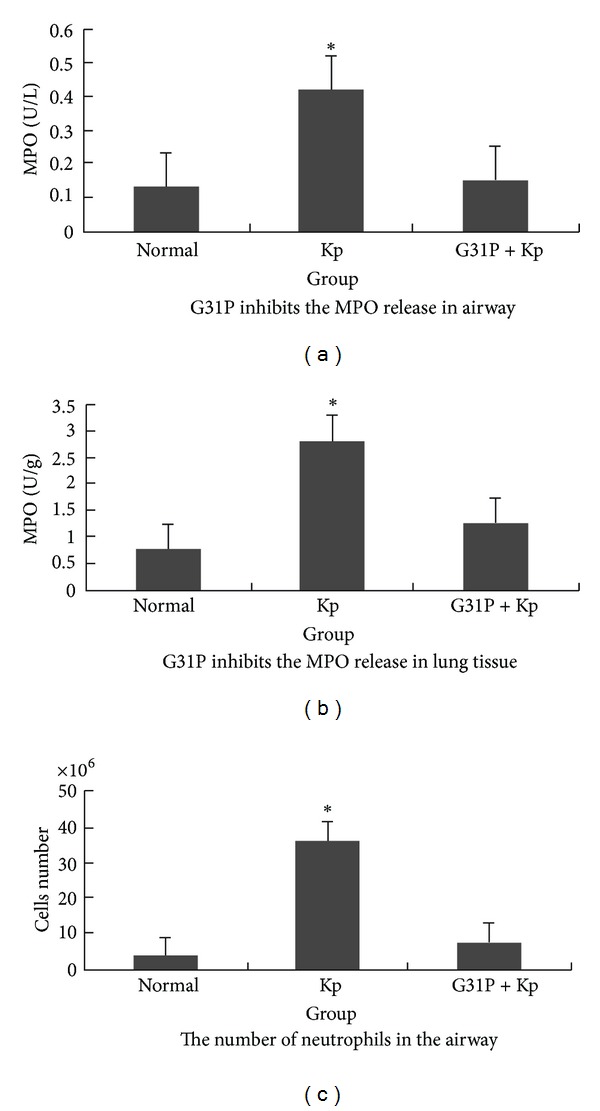
Treatment with the ELR-CXC chemokine antagonist G31P reduces pulmonary neutrophil responses in guinea pigs with Klebsiella pneumoniae. Guinea pigs were challenged with Klebsiella pneumoniae and treated with G31P. The airway neutrophil responses were assessed from differential counts of cells recovered by bronchoalveolar lavage (BAL). The airway (BAL fluid) levels of myeloperoxidase (MPO), as surrogate measurements of neutrophilic inflammation, were assessed using a chromogenic assay. The G31P treatments reduced each of these indicators of neutrophilic inflammation close to normal. This experiment was repeated three times; the data depicted are representative of each group.
3.3. ELR-CXC Chemokine Antagonism Is as Effective as Corticosteroid and Antibiotic Treatments
Either antagonizing the bacterial growth or dampening the inflammatory response pharmacologically has significant protective effects in the antimicrobial inflammatory response of bacterial pneumonias [12, 13]. We compared curative effects of different treatments, including G31P, the broad-spectrum β-lactam antibiotic ceftazidime (200 mg/kg), and dexamethasone (0.8 mg/kg/24 h). 24 h posttreatment, ceftazidime reduced inflammation levels but had no effect in eliminating the pulmonary Klesbiella load, pleural hemorrhagic consolidation (Figure 2), or CXCL8 and TNF responses (Figure 3). And it had marginal effects in preventing white cell infiltration (Figure 4). The dexamethasone treatment had no significant effects on airway Klebsiella loads, but it significantly improved pathologic damage (Figure 2), CXCL8 and TNF (Figure 3) levels, infiltration of inflammatory cells, and the lung MPO response (Figure 4). G31P treatment was more effective in reducing neutrophilic pathology (Figure 2) than antibiotic and corticosteroid treatments. CXCL8 and TNF levels reduced (Figure 3) airway inflammatory cell influx, MPO levels, and lung parenchymal MPO levels (Figure 4).
Figure 2.
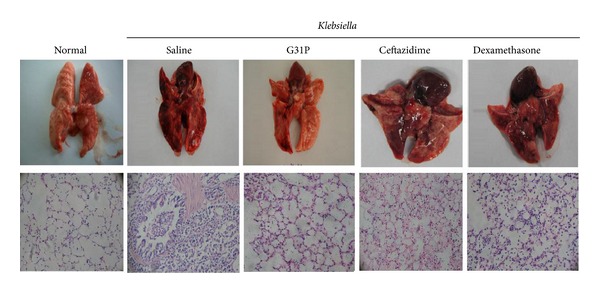
Comparison of different treatments for inflammatory pathology in guinea pigs with Klebsiella pneumoniae. The lungs of the control group were normal in gross appearance, while the lungs of the bacterial control group were markedly hemorrhagic and congested. The G31P-treated group was largely free of hemorrhagic consolidation and had greatly reduced pulmonary inflammation. The lungs of the saline controls exhibited a normal histological appearance, while the lungs of the bacterial control animals were markedly hemorrhagic and congested with red blood cells (RBC) and neutrophils (PMN).
Figure 3.
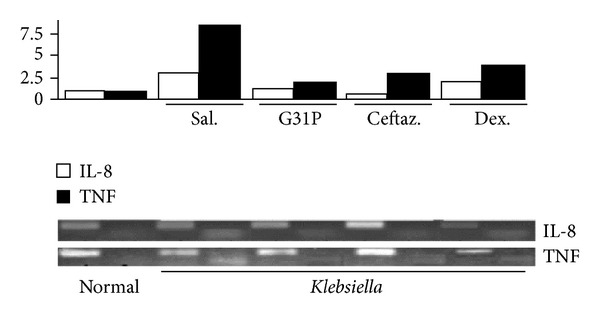
Impact of G31P, dexamethasone, or ceftazidime treatments on inflammatory pathology in guinea pigs with Klebsiella pneumoniae. Guinea-pigs were challenged with Klebsiella pneumonia, treated with G31P, dexamethasone, or ceftazamine, and then sacrificed 24 h later. Each of these treatments markedly reduced the expression levels of both mediators relative to the saline-treated animals. Each experiment was repeated three times; the data depicted are representative of each group.
Figure 4.
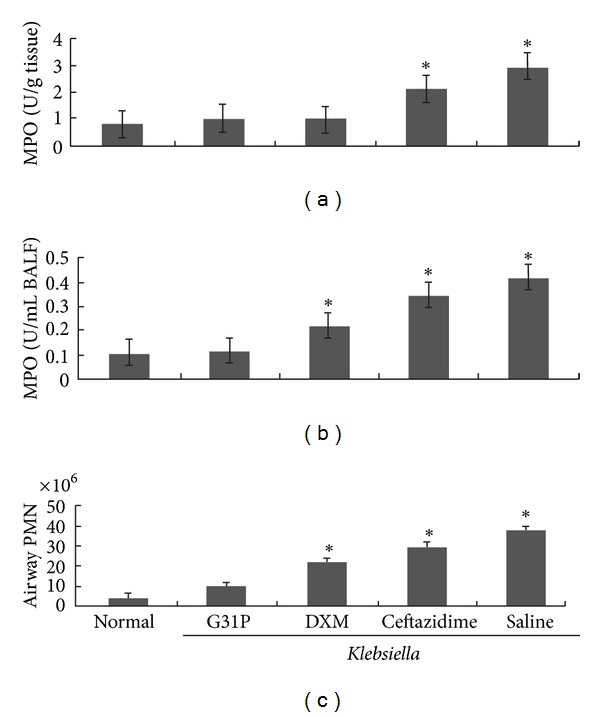
Impact of G31P, dexamethasone, or ceftazidime treatments on inflammatory pathology in guinea pigs with Klebsiella pneumoniae. In the G31P-treated group, PMN decreased significantly in BALF compared with the hormones, or antibiotics-treated group (*indicates P < 0.05). The data show that G31P, hormones, and antibiotics groups have a significant effect in the inhibition of MPO release from lung tissue (*indicates P < 0.05). G31P decreased significantly the release of MPO in lung tissue compared with the antibiotics group (*indicates P < 0.05). But there was no significant difference between the G31P and DXM groups.
4. Discussion
Chemokines are small molecular proteins (8~10 KDa) that are important in the recruitment of leukocytes to extravascular tissues [6]. Although this is a normal physiological response during infection, excessive inflammation cells can cause tissue injury. In such cases chemokine antagonism can be used as a therapeutic strategy.
The ELR+ CXC chemokines include CXCL8, epithelial neutrophil activator 78 (ENA-78; CXCL5), granulocyte chemotactic protein-2 (GCP-2; CXCL6), neutrophil-activating protein-2 (NAP-2; CXCL7), and growth-related oncogene-α, -β, and -γ (GRO-α, -β, and -γ, resp. CXCL1, CXCL2, and CXCL3) [5]. These chemokines can bind to either CXCR1 or CXCR2, and recruit neutrophils into the local inflammatory response [2] in a complex multistep process [14–16]. The CXCR1 binds CXCL8 and GCP-2 with high affinity, while the CXCR2 binds CXCL8 with higher affinity, but other ELR-CXC chemokines at low affinity [5]. We previously engineered a human CXCL8 analogue (CXCL8(3-72) K11R/G31P; G31P) [17]. G31P antagonizes both CXCL1 and CXCL2 efficiently, but also desensitizes heterologous G protein-coupled receptors (GPCR) on cells that express CXCR1 or CXCR2 (i.e., the receptors for C5a, LTB4, and fMLP) [8, 17]. It has been reported that G31P serves as an effective anti-inflammatory agent in models of airway endotoxemia [9, 10, 18], aspiration pneumoniae [11], and ischemia-reperfusion injury [10, 19]. It has been reported previously that bacterial endotoxin-challenged bronchial epithelial cells secrete high levels of CXCL8 and CXCL5 and that these act as autocrine agonists for these cells [8], and others have documented that vascular endothelial and alveolar epithelial cells expression of CXCR2 plays an important role in pulmonary pathology in models of LPS-induced pneumoniae in mice [20, 21]. Specifically, G31P therapy reduced pulmonary expression of endogenous pyrogens (TNF and IL-6) as well as CXCL8, and this was associated with a dramatically reduced neutrophilic inflammatory response, but no change in the pulmonary loads of viable Klebsiella pneumoniae. Corticosteroids can be used effectively to dampen inflammation, but there is some discrepancy regarding whether they antagonize ELR-CXC chemokine responses [22–24] or not [25, 26] and thereby antagonize neutrophil responses. Nevertheless, the lung tissues of dexamethasone-treated mice contained reduced levels of TNF, CXCL8, and MPO and modestly reduced numbers of neutrophils relative to the saline-treated pneumonic animals, although the levels of each of these parameters still appeared to be somewhat higher than those seen in G31P-treated mice.
Aggarwal et al. reported a metastatic role of CXCL8, which promotes cell growth and metastasis. CXCL8 expression correlates with metastatic potential in human melanoma and ovarian cancer cells. It has been found to play an important role in astrocytomas, anaplastic astrocytomas, glioblastomas, central nervous system cervical carcinoma metastasis, brain tumors, gastric cardia adenocarcinoma (GCC), and esophageal squamous cell carcinoma [6]. G31P can be used as an antagonistic agent to treat the carcinoma by reducing the level of CXCL8. This study opens the future research aspect of ELR-CXC to treat these carcinomas.
In summary, our data indicate that the ELR-CXC antagonist G31P can effectively reduce Klebsiella pneumoniae pathology in a guinea-pig model. It is suggested that broad-spectrum ELR-CXC chemokine antagonism may be a useful adjunct in the therapeutic approach to treat Klebsiella pneumoniae in patients.
Ethical Approval
The project was approved by the Dalian Medical University ethics committee in China.
Conflict of Interests
The authors declare no conflict of interest.
Authors' Contribution
J. Wei and J. Peng have equally contributed to this paper.
Acknowledgments
This work is supported by Grants from the National Science Foundation of China (NSFC30772023) and the Natural Sciences and Engineering Research Council of Canada (J. R. Gordan). The authors appreciate the valuable comments from other members of their laboratories.
References
- 1.Tsai SS, Huang JC, Chen ST, et al. Characteristics of Klebsiella pneumoniae bacteremia in community-acquired and nosocomial infections in diabetic patients. Chang Gung Medical Journal. 2010;33(5):532–539. [PubMed] [Google Scholar]
- 2.Rashid T, Ebringer A. Ankylosing spondylitis is linked to Klebsiella—the evidence. Clinical Rheumatology. 2007;26(6):858–864. doi: 10.1007/s10067-006-0488-7. [DOI] [PubMed] [Google Scholar]
- 3.Vázquez MN, López de Castro JA. Similar cell surface expression of β2-microglobulin-free heavy chains by HLA-B27 subtypes differentially associated with ankylosing spondylitis. Arthritis and Rheumatism. 2005;52(10):3290–3299. doi: 10.1002/art.21284. [DOI] [PubMed] [Google Scholar]
- 4.Nordmann P, Cuzon G, Naas T. The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. The Lancet Infectious Diseases. 2009;9(4):228–236. doi: 10.1016/S1473-3099(09)70054-4. [DOI] [PubMed] [Google Scholar]
- 5.Baggiolini M. Chemokines in pathology and medicine. Journal of Internal Medicine. 2001;250(2):91–104. doi: 10.1046/j.1365-2796.2001.00867.x. [DOI] [PubMed] [Google Scholar]
- 6.Aggarwal BB, Shishodia S, Sandur SK, Pandey MK, Sethi G. Inflammation and cancer: how hot is the link? Biochemical Pharmacology. 2006;72(11):1605–1621. doi: 10.1016/j.bcp.2006.06.029. [DOI] [PubMed] [Google Scholar]
- 7.Pichavant M, Delneste Y, Jeannin P, et al. Outer membrane protein A from Klebsiella pneumoniae activates bronchial epithelial cells: implication in neutrophil recruitment. Journal of Immunology. 2003;171(12):6697–6705. doi: 10.4049/jimmunol.171.12.6697. [DOI] [PubMed] [Google Scholar]
- 8.Zhao X, Town JR, Li F, Zhang X, Cockcroft DW, Gordon JR. ELR-CXC chemokine receptor antagonism targets inflammatory responses at multiple levels. Journal of Immunology. 2009;182(5):3213–3222. doi: 10.4049/jimmunol.0800551. [DOI] [PubMed] [Google Scholar]
- 9.Gordon JR, Li F, Zhang X, Wang W, Zhao X, Nayyar A. The combined CXCR1/CXCR2 antagonist CXCL8(3–74)K11R/G31P blocks neutrophil infiltration, pyrexia, and pulmonary vascular pathology in endotoxemic animals. Journal of Leukocyte Biology. 2005;78(6):1265–1272. doi: 10.1189/jlb.0805458. [DOI] [PubMed] [Google Scholar]
- 10.Zhao X, Town JR, Yang A, et al. A novel ELR-CXC chemokine antagonist reduces intestinal ischemia reperfusion-induced mortality, and local and remote organ injury. Journal of Surgical Research. 2010;162(2):264–273. doi: 10.1016/j.jss.2009.04.047. [DOI] [PubMed] [Google Scholar]
- 11.Zhao X, Town JR, Li F, Li W, Zhang X, Gordon JR. Blockade of neutrophil responses in aspiration pneumonia via ELR-CXC chemokine antagonism does not predispose to airway bacterial outgrowth. Pulmonary Pharmacology and Therapeutics. 2010;23(1):22–28. doi: 10.1016/j.pupt.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 12.Moldoveanu B, Otmishi P, Jani P, et al. Inflammatory mechanisms in the lung. Journal of Inflammation Research. 2009;2:1–11. [PMC free article] [PubMed] [Google Scholar]
- 13.Heit B, Colarusso P, Kubes P. Fundamentally different roles for LFA-1, Mac-1 and α 4-integrin in neutrophil chemotaxis. Journal of Cell Science. 2005;118(22):5205–5220. doi: 10.1242/jcs.02632. [DOI] [PubMed] [Google Scholar]
- 14.Ibbotson GC, Doig C, Kaur J, et al. Functional α4-integrin: a newly identified pathway of neutrophil recruitment in critically ill septic patients. Nature Medicine. 2001;7(4):465–470. doi: 10.1038/86539. [DOI] [PubMed] [Google Scholar]
- 15.Heit B, Colarusso P, Kubes P. Fundamentally different roles for LFA-1, Mac-1 and α 4-integrin in neutrophil chemotaxis. Journal of Cell Science. 2005;118(22):5205–5220. doi: 10.1242/jcs.02632. [DOI] [PubMed] [Google Scholar]
- 16.McDonald B, McAvoy EF, Lam F, et al. Interaction of CD44 and hyaluronan is the dominant mechanism for neutrophil sequestration in inflamed liver sinusoids. Journal of Experimental Medicine. 2008;205(4):915–927. doi: 10.1084/jem.20071765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao X, Town JR, Li F, Zhang X, Cockcroft DW, Gordon JR. ELR-CXC chemokine receptor antagonism targets inflammatory responses at multiple levels. Journal of Immunology. 2009;182(5):3213–3222. doi: 10.4049/jimmunol.0800551. [DOI] [PubMed] [Google Scholar]
- 18.Gordon JR, Zhang X, Li F, Nayyar A, Town J, Zhao X. Amelioration of pathology by ELR-CXC chemokine antagonism in a swine model of airway endotoxin exposure. Journal of Agromedicine. 2009;14(2):235–241. doi: 10.1080/10599240902845047. [DOI] [PubMed] [Google Scholar]
- 19.Zhao X, Town JR, Yang A, et al. A novel ELR-CXC chemokine antagonist reduces intestinal ischemia reperfusion-induced mortality, and local and remote organ injury. Journal of Surgical Research. 2010;162(2):264–273. doi: 10.1016/j.jss.2009.04.047. [DOI] [PubMed] [Google Scholar]
- 20.Fligiel SEG, Standiford T, Fligiel HM, et al. Matrix metalloproteinases and matrix metalloproteinase inhibitors in acute lung injury. Human Pathology. 2006;37(4):422–430. doi: 10.1016/j.humpath.2005.11.023. [DOI] [PubMed] [Google Scholar]
- 21.Reutershan J, Morris MA, Burcin TL, et al. Critical role of endothelial CXCR2 in LPS-induced neutrophil migration into the lung. Journal of Clinical Investigation. 2006;116(3):695–702. doi: 10.1172/JCI27009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Levine SJ, Larivee P, Logun C, Angus CW, Shelhamer JH. Corticosteroids differentially regulate secretion of IL-6, IL-8, and G-CSF by a human bronchial epithelial cell line. American Journal of Physiology. 1993;265(4, part 1):L360–L368. doi: 10.1152/ajplung.1993.265.4.L360. [DOI] [PubMed] [Google Scholar]
- 23.Tjandra K, Kubes P, Rioux K, Swain MG. Endogenous glucocorticoids inhibit neutrophil recruitment to inflammatory sites in cholestatic rats. American Journal of Physiology. 1996;270(5, part 1):G821–G825. doi: 10.1152/ajpgi.1996.270.5.G821. [DOI] [PubMed] [Google Scholar]
- 24.Nyhlén K, Linden M, Andersson R, Uppugunduri S. Corticosteroids and interferons inhibit cytokine-induced production of IL-8 by human endothelial cells. Cytokine. 2000;12(4):355–360. doi: 10.1006/cyto.1999.0557. [DOI] [PubMed] [Google Scholar]
- 25.Teoh KHT, Bradley CA, Gauldie J, Burrows H. Steroid inhibition of cytokine-mediated vasodilation after warm heart surgery. Circulation. 1995;92(9):II347–II353. doi: 10.1161/01.cir.92.9.347. [DOI] [PubMed] [Google Scholar]
- 26.Issa R, Xie S, Khorasani N, et al. Corticosteroid inhibition of growth-related oncogene protein-α via mitogen-activated kinase phosphatase-1 in airway smooth muscle cells. Journal of Immunology. 2007;178(11):7366–7375. doi: 10.4049/jimmunol.178.11.7366. [DOI] [PubMed] [Google Scholar]


