Summary
Background and objectives
β-trace protein (βTP), β2-microglobulin (β2M), and cystatin C (CysC) have advantages over creatinine for estimating GFR and prognosis. This study compares the distribution of all four markers in the general population and their associations with possible determinants of GFR.
Design, setting, participants, & measurements
βTP and β2M were measured in 7596 participants (aged ≥12 years) of the Third National Health and Nutrition Examination Survey (1988–1994). βTP and β2M concentrations and the proportion of persons with elevated (≥99th percentile for young healthy participants) βTP (≥0.81 mg/L), β2M (≥2.80 mg/L), standardized CysC (≥1.03 mg/L), and creatinine (≥1.2 mg/dl for men and ≥1.0 mg/dl for women) were compared across demographic and clinical factors.
Results
Elevated βTP, β2M, and CysC showed stronger associations with age than elevated serum creatinine, the prevalence of elevated levels reaching 47%, 44%, 58%, and 26%, respectively, by age 80 years. βTP, CysC, and creatinine were higher in men but β2M was not associated with sex. Mexican Americans had lower βTP, β2M, CysC, and creatinine compared with non-Hispanic whites. Hypertension and higher C-reactive protein were associated with elevations in all markers, whereas non-Hispanic black race, body mass index, diabetes, smoking status, triglycerides, HDL cholesterol, and education were not associated in a consistent manner across the different markers.
Conclusions
βTP, β2M, CysC, and creatinine differ in their associations with demographic and clinical factors, suggesting variation in their non-GFR determinants. Future studies should examine these markers with measured GFR to determine their diagnostic and prognostic utility.
Introduction
β-trace protein (βTP) and β2-microglobulin (β2M) are low molecular mass serum proteins that are increasingly viewed as useful for estimating GFR and predicting prognosis in kidney disease, cardiovascular outcomes, and death, independently of GFR (1–3). Recent studies have shown that βTP and β2M are better predictors of adverse health outcomes than creatinine and are potentially as good as cystatin C (CysC) (4). It is now appreciated that the use of a combination of filtration markers leads to more accurate estimates of GFR than single markers (5), and as in other fields, it is expected that a combination of markers will lead to more accurate predictions of prognosis (6). However, as we have learned from creatinine and CysC, knowledge of their non-GFR determinants in the general population will be necessary for optimal use.
Determinants of the serum concentrations in βTP and β2M have not yet been studied in the general population. βTP, a 168 amino acid glycoprotein (7), originates from the choroid plexus of the central nervous system (8). β2M, a subunit of the major histocompatibility class I molecule, is produced by all nucleated cells (9). Like CysC, βTP and β2M are considered candidates for the assessment of GFR because they are produced at a constant rate, are freely filtered by the glomerulus, and are then reabsorbed and almost completely metabolized by the proximal tubule with no appreciable entry into the circulation (10–12). There is some evidence that cerebrospinal fluid leaks (13) and corticosteroid use (14) affect βTP, whereas rare inflammatory conditions affect β2M (15–18).
The challenge of measuring GFR in large population studies and the almost complete absence of urinary excretion of these markers make it difficult to study the relative contributions of GFR and non-GFR determinants to their serum concentrations. As such, examining four filtration markers in the general population presents an opportunity to infer demographic and clinical factors that may be associated with variation in underlying GFR. Thus, the objectives of this study were to describe the distributions of βTP and β2M in the US population, to identify factors associated with elevations in βTP and β2M, to compare these distributions and associations with those of standardized creatinine and CysC, and to distinguish factors that may be associated with GFR versus non-GFR determinants of all four filtration markers.
Materials and Methods
Study Population
Data for this study were derived from the Third National Health and Nutrition Examination Survey (NHANES III), a large, cross-sectional study conducted by the National Center for Health Statistics (NCHS) using a complex multistage sampling design. Interviews, physical examinations, and laboratory measurements were obtained from 18,722 individuals aged ≥12 years (19). Stored serum samples from a subset of NHANES III participants were used for the measurement of nontraditional markers of GFR. Details of this study design were previously described (20,21). Briefly, we selected the following: all participants aged ≥60 years because decreased GFR is common in this group (n=5248), all participants with elevated creatinine >1.189 mg/dl in men and >0.997 mg/dl in women (n=305), and a 25% random sample of participants aged 12–59 years (n=3293). This design minimized costs while retaining power and generalizability. Sample weights were re-calculated based on the total number of samples that were not missing CysC (86%; n=7596). Among the original CysC subsample, there were 7529 (99.1%) participants with a βTP measurement and 7534 (99.2%) participants with a β2M measurement.
Primary Outcome Measurements
In 2009, we measured βTP and β2M in stored serum samples collected in 1988–1994, using particle-enhanced immunonephelometric assays (N Latex β-trace protein assay and N Latex β-2 microglobulin assay; Dade Behring, Deerfield, IL). Both βTP and β2M are reported to be robust to freeze-thaw cycles (22). Interassay coefficients of variation (CVs) for the βTP assay and the β2M assay were 5.7% (mean 0.594 mg/L) and 2.7% (mean 1.757 mg/L), respectively. Creatinine was measured using a modified kinetic Jaffe reaction, traceable to higher-order isotope dilution mass spectrometry methods, and the CV ranged from 1.2% (mean 1.00 mg/dl) to 1.6% (mean 3.84 mg/dl) (23). CysC was measured using a particle-enhanced immunonephelometric assay (20) and was calibrated to standardized CysC (5). Its CV ranged from 4.9% (mean 1.90 mg/L) to 5.1% (mean 0.97 mg/L) (20,21). All serum specimens underwent at least one thaw cycle.
Definition of Elevated Filtration Markers and Decreased GFR
Cutoffs for elevated βTP (≥0.81 mg/L), β2M (≥2.80 mg/L), and CysC (≥1.03 mg/L) were defined as the 99th percentile in participants aged 20–39 years who did not have hypertension or diabetes (diagnosed or undiagnosed) (20). Creatinine cutoffs were sex specific, corresponding to the study design, with cutoffs of ≥1.2 mg/dl for men and ≥1.0 mg/dl for women. This is approximately equal to the 99th percentile in either sex in participants aged 20–39 years without diabetes and hypertension. The cutoff for decreased estimated GFR using creatinine was <60 ml/min per 1.73m2, computed from the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation (24).
Determination of Potential Risk Factors
We examined covariates associated with CKD. Race and ethnicity were grouped as non-Hispanic white, non-Hispanic black, Mexican American, and other, although the “other” category was not presented due to inherent heterogeneity and small sample size (25). Education (<12 years, ≥12 years), current smoking status (yes/no), and obesity (body mass index [BMI] <30 kg/m2, ≥30 kg/m2) were dichotomized. BMI was also analyzed as a continuous variable (scaled to per 5 kg/m2). Hypertensive status (yes/no) was defined as a physician-diagnosis of hypertension, hypertension medication use, or a mean systolic or diastolic BP ≥140 mmHg or ≥90 mmHg, respectively. Diabetes status (yes/no) was based on a fasting plasma glucose of ≥126 mg/dl (≥7.0 mmol/L) or a nonfasting glucose of ≥200 mg/dl (≥11.0 mmol/L) or self-reported diagnosis by a physician (independent of pregnancy), use of insulin, or use of diabetes medication. HDL cholesterol was defined as a continuous variable (per 10-mg/dl increase) for logistic regression, and as a dichotomous variable (<40 mg/dl for men and <50 mg/dl for women) to estimate study population prevalence. Triglycerides were log-transformed to normalize its distribution. C-reactive protein (CRP) was divided into three categories (<0.22 mg/dl, 0.22–1 mg/dl, >1 mg/dl). The lower limit of detection for CRP was 0.22 mg/dl. Urine albumin was measured by a solid-phase fluorescent immunoassay, and urine creatinine was measured using the modified kinetic rate Jaffe method. Urinary albumin/creatinine ratio (ACR) was expressed in milligrams per gram (26,27). Additional data collection details, including assay precision estimates, are available in the NHANES field manual published online (19).
Statistical Analyses
The method used to generate sample weights was described in detail elsewhere (20). Briefly, the original sampling weights were modified to account for both missing creatinine and CysC using standard methods approved by the NCHS. These weights were utilized in all of our analyses and SEMs for estimates were obtained using the Taylor series (linearization) method (28). Analyses were performed using Stata11.1 software (StataCorp LP, College Station, TX).
Means and proportions were estimated by age group, and weighted percentiles of βTP and β2M were estimated using the subset of healthy individuals aged 20–39 years. Filtration markers were transformed (−1/βTP and −1/β2M) to model GFR. We used logistic regression models to examine the associations of demographic and clinical factors with elevated βTP and β2M concentrations, overall (≥20 years) and stratified by age group (20–59 years, ≥60 years). These models were applied to CysC and creatinine for comparison. This analysis was performed in participants aged ≥60 years, reflecting subsample design as well as a population with decreased GFR. The analysis was also conducted in participants aged 20–59 years. Participants aged <20 years were excluded due to differences in NHANES III covariate definitions among youth as well as lack of data on education. Our models were adjusted for age, sex, race/ethnicity, BMI, hypertension, diabetes, smoking, CRP, log-transformed triglycerides, HDL cholesterol, and education. We also repeated this analysis adjusting for estimated GFR using creatinine both in place of and in addition to age, sex, and race/ethnicity. The weighted median and 5th and 95th percentiles of 1/βTP and 1/β2M were evaluated by age, sex, and race/ethnicity to model GFR across subgroups and to normalize their distributions. Continuous associations between markers and age were evaluated using linear splines with knots every 10 years. Pearson correlation coefficients were calculated between βTP, β2M, CysC, and creatinine. The proportion of the US population aged ≥12 years with an elevated serum concentration of each filtration marker was plotted by age along with the proportion of decreased estimated GFR using creatinine to observe age-related trends.
Informed consent was obtained from all participants. Protocols for the conduct and implementation of this study were approved by the institutional review boards of both the NCHS and the Johns Hopkins Bloomberg School of Public Health.
Results
Population characteristics are shown in Table 1. The mean βTP and β2M concentrations in the overall US population were 0.58 mg/L (95% confidence interval, 0.57, 0.59) and 1.88 mg/L (95% confidence interval, 1.85, 1.92). Percentiles by age and sex showed βTP levels being higher in men than women (P<0.01), with details by age, marker, and ethnicity shown in Supplemental Figures 1–3. The correlation between βTP and β2M was high (0.85), as were the correlations with CysC (0.73 and 0.82), whereas correlations with creatinine were lower (0.65 and 0.68).
Table 1.
Characteristics overall and by age group, US population (NHANES III 1988–1994)
| Characteristic | Age Group (yr) | Overall | |||
|---|---|---|---|---|---|
| 12–19 (n=719) | 20–39 (n=1675) | 40–59 (n=1149) | ≥60 (n=4053) | All (n=7596) | |
| Male | 50.8±3.5 | 49.3±2.1 | 48.7±2.5 | 42.8±0.8 | 48.1±1.4 |
| Race/ethnicity | |||||
| Non-Hispanic white | 69.4±3.6 | 71.7±2.7 | 78.6±2.3 | 84.0±1.5 | 75.6±2.0 |
| Non-Hispanic black | 15.5±2.1 | 12.6±1.4 | 10.4±1.1 | 8.3±0.8 | 11.6±1.0 |
| Mexican American | 8.3±1.0 | 7.0±0.9 | 4.3±0.6 | 2.3±0.2 | 5.5±0.5 |
| Other ethnicity | 6.8±1.9 | 8.8±1.6 | 6.8±1.6 | 5.4±1.0 | 7.3±1.2 |
| Education <12 yra | NA | 18.6±1.7 | 20.1±2.2 | 40.0±1.7 | 23.0±1.4 |
| Current smokinga | 10.7±2.2 | 36.4±2.2 | 27.5±2.2 | 14.8±1.0 | 26.2±1.1 |
| Obesity (BMI ≥30 kg/m2)a | 7.1±1.9 | 18.0±1.8 | 28.3±2.1 | 23.8±0.9 | 20.4±1.1 |
| Hypertensiona | 0.6±0.4 | 5.2±0.7 | 24.0±2.2 | 58.7±1.0 | 20.1±1.2 |
| Diabetes a | 0.1±0.1 | 1.0±0.3 | 6.5±1.1 | 12.6±0.7 | 4.6±0.5 |
| Low HDL cholesterol (<40 mg/dl for men; <50 mg/dl for women)a | 38.7±3.2 | 36.2±1.7 | 39.4±2.1 | 37.4±1.4 | 37.6±1.3 |
| Triglycerides (mg/dl)a | 83.7 | 99.6 | 130.1 | 143.3 | 112.1 |
| C-reactive protein (mg/dl)a | |||||
| <0.22 | 88.9±2.4 | 77.4±1.9 | 68.7±2.6 | 61.4±2.1 | 73.5±1.5 |
| 0.22–1 | 8.5±1.9 | 17.3±1.5 | 23.6±2.2 | 28.6±1.7 | 20.0±1.2 |
| >1 | 2.6±1.3 | 4.5±0.7 | 7.6±1.5 | 9.9±0.7 | 6.1±0.6 |
| Albumin/creatinine ratio (mg/g)a,b | 8.2 | 4.7 | 6.2 | 11.1 | 6.6 |
| Estimated GFR, creatinine (ml/min per 1.73 m2) | 135.6±0.94 | 113.1±0.77 | 96.9±0.63 | 74.5±0.42 | 104.4±0.69 |
| Creatinine (mg/dl)c | |||||
| Male | 0.77±0.01 | 0.92±0.01 | 0.93±0.01 | 1.06±0.01 | 0.92±0.01d |
| Female | 0.66±0.01 | 0.70±0.01 | 0.74±0.01 | 0.84±0.01 | 0.74±0.005d |
| Cystatin C (mg/L)c | |||||
| Male | 0.82±0.01 | 0.78±0.01 | 0.83±0.01 | 1.04±0.01 | 0.85±0.01d |
| Female | 0.74±0.01 | 0.72±0.01 | 0.78±0.01 | 1.02±0.01 | 0.80±0.01d |
| β-trace protein (mg/L)c | |||||
| Male | 0.60±0.01 | 0.54±0.01 | 0.56±0.01 | 0.77±0.01 | 0.59±0.01d |
| Female | 0.56±0.01 | 0.51±0.01 | 0.54±0.01 | 0.72±0.01 | 0.57±0.01d |
| β2-microglobulin (mg/L)c | |||||
| Male | 1.65±0.02 | 1.71±0.02 | 1.86±0.02 | 2.56±0.03 | 1.89±0.02e |
| Female | 1.54±0.02 | 1.62±0.02 | 1.85±0.04 | 2.60±0.04 | 1.88±0.02e |
Data are presented as mean ± SEM for continuous data and as a proportion ± SEM for categorical variables. A geometric mean (without SEM) is provided for albumin/creatinine ratio and triglycerides. SEMs for geometric means were not obtained because of complex survey design. Survey sample size is denoted by n. Creatinine may be converted from mg/dl to μmol/L by multiplying by 88.4. Triglycerides may be converted from mg/dl to mmol/L by multiplying by 0.0113. HDL may be converted from mg/dl to mmol/L by multiplying by 0.0259. NHANES III, Third National Health and Nutrition Examination Survey; BMI, body mass index; NA, not available.
Data from a small fraction of participants with unknown covariate distribution not shown.
Albumin/creatinine ratio was determined in a subset of participants that excluded pregnant or menstruating women (n=250).
Men (n=3611), women (n=3985).
P value for overall mean across sexes was <0.01.
P value for overall mean across sexes was 0.66.
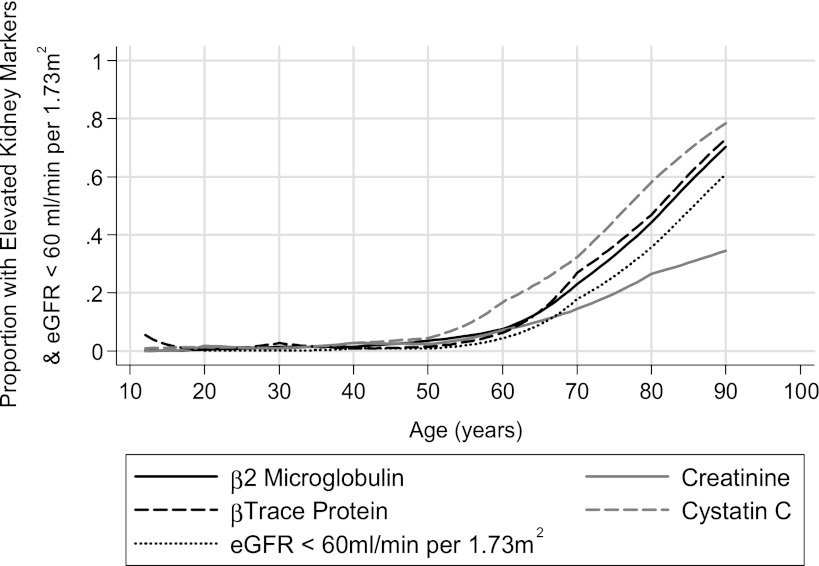
We compared the prevalence of elevated serum concentrations of each of the filtration markers and estimated GFR, using creatinine <60 ml/min per 1.73 m2 across all ages (Figure 1). CysC showed the sharpest incline in prevalence starting at about age 50 years, with βTP and β2M demonstrating a comparable rise but starting at around age 60 years. Although creatinine elevation also became more common starting at about age 50 years, its incline was far more gradual than for the other three markers. Estimated GFR from creatinine showed an intermediate behavior. At age 80 years, the proportions of the population with an elevated βTP, β2M, CysC, and creatinine were 47%, 44%, 58%, and 26% compared with 0.4%, 0.5%, 1.3%, and 1.7% at age 20 years.
Figure 1.
Proportion of the US population aged ≥12 years with an elevated filtration marker or CKD stage 3 by age. Elevated markers were β-trace protein (≥0.81 mg/L), β2-microglobulin (≥2.80 mg/L), standardized creatinine (men, ≥1.2 mg/dl; women, ≥1.0 mg/dl), or standardized cystatin C (≥1.03 mg/L). CKD stage 3 is defined as an estimated GFR <60 ml/min per 1.73 m2, estimated using the Chronic Kidney Disease Epidemiology Collaboration equation (24). The elevated markers or CKD stage 3 are independent definitions determined solely by the markers themselves or the estimated GFR (eGFR).
We performed multivariable logistic regression of factors associated with elevated levels of βTP, β2M, CysC, and creatinine (Table 2 and Figure 2). Among persons aged ≥20 years, after adjustment for all other factors examined, age, hypertension, and CRP were associated with elevated levels of all filtration markers. Consistent associations with all markers except creatinine were seen for HDL cholesterol and education, both in a protective direction. Other associations were less consistent across markers. Female sex and Mexican race/ethnicity were associated with a lower prevalence of elevations in βTP and creatinine but not significant associations with β2M and CysC. In contrast, triglycerides were associated with a higher prevalence of elevations in CysC and creatinine. Current smoking status was uniquely associated with a lower prevalence of creatinine elevations and diabetes was not associated with prevalence of elevations in any of the four markers. Two factors were associated in reverse directions with two markers. Non-Hispanic black race/ethnicity was associated with elevated concentrations of creatinine, but lower concentrations of βTP, and BMI was associated with lower concentrations of β2M, but elevations in CysC. Some differences in the patterns in strata of age 20–59 years versus ≥60 years are shown in Supplemental Tables 1–5 and Supplemental Figures 2–5. In general, adjustment for estimated GFR rather than age, sex, and race/ethnicity did not alter the above patterns (Table 3). The two exceptions were that both BMI and triglycerides were significantly associated with βTP and no longer associated with CysC after adjustment for estimated GFR. Similarly, adjustment for estimated GFR in addition to age, sex, and race/ethnicity yielded virtually the same results (Supplemental Table 6).
Table 2.
Adjusted ORs and 95% CIs of elevated serum β-trace protein (≥0.81 mg/L), elevated serum β2-microglobulin (≥2.80 mg/L), elevated standard cystatin C (≥1.03 mg/L), and elevated standard creatinine (men, ≥1.0 mg/dl; women, ≥1.2 mg/dl) in persons aged ≥20 years
| β-Trace Protein (≥0.81 mg/L) | β2-Microglobulin (≥2.80 mg/L) | Standard Cystatin C (≥1.03 mg/L) | Creatinine (Men, ≥1.0 mg/dl; Women, ≥1.2 mg/dl) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ≥20 yr (n=6682 [1220]) | ≥20 yr (n=6688 [1204]) | ≥20 yr (n=6742 [1680]) | ≥20 yr (n=6742 [966]) | |||||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | |
| Age (yr) | 1.09 | 1.06–1.12 | <0.01 | 1.09 | 1.07–1.11 | <0.01 | 1.10 | 1.08–1.12 | <0.01 | 1.06 | 1.05–1.07 | <0.01 |
| Female sex | 0.62 | 0.44–0.88 | <0.01 | 1.26 | 0.88–1.80 | 0.20 | 0.87 | 0.65–1.16 | 0.33 | 0.60 | 0.46–0.77 | <0.01 |
| Race/ethnicitya | ||||||||||||
| Non-Hispanic black | 0.52 | 0.37–0.73 | <0.01 | 1.04 | 0.78–1.40 | 0.79 | 0.76 | 0.53–1.07 | 0.11 | 3.09 | 2.54–3.75 | <0.01 |
| Mexican American | 0.56 | 0.37–0.84 | <0.01 | 0.82 | 0.57–1.18 | 0.28 | 0.85 | 0.48–1.49 | 0.56 | 0.44 | 0.31–0.62 | <0.01 |
| BMI (per 5 kg/m2) | 0.93 | 0.81–1.06 | 0.28 | 0.83 | 0.75–0.92 | <0.01 | 1.23 | 1.06–1.43 | 0.01 | 0.99 | 0.86–1.13 | 0.84 |
| Hypertension | 2.10 | 1.54–2.86 | <0.01 | 1.66 | 1.35–2.03 | <0.01 | 1.72 | 1.27–2.32 | <0.01 | 2.15 | 1.75–2.64 | <0.01 |
| Diabetes | 1.06 | 0.77–1.47 | 0.71 | 1.37 | 0.91–2.07 | 0.13 | 1.17 | 0.84–1.62 | 0.35 | 1.17 | 0.87–1.57 | 0.30 |
| Current smoker | 0.98 | 0.61–1.58 | 0.93 | 0.97 | 0.66–1.43 | 0.87 | 1.44 | 0.96–2.17 | 0.08 | 0.75 | 0.58–0.98 | 0.04 |
| CRP (mg/dl)b | ||||||||||||
| 0.22–1.0 | 1.30 | 0.95–1.79 | 0.10 | 1.75 | 1.32–2.30 | <0.01 | 1.13 | 0.86–1.48 | 0.39 | 1.18 | 0.95–1.46 | 0.13 |
| >1.0 | 2.12 | 1.37–3.28 | <0.01 | 4.10 | 2.65–6.33 | <0.01 | 2.49 | 1.60–3.86 | <0.01 | 1.65 | 1.08–2.53 | 0.02 |
| Triglycerides (mg/dl) | 0.84 | 0.56–1.27 | 0.41 | 0.98 | 0.66–1.46 | 0.93 | 1.50 | 1.08–2.09 | 0.02 | 1.70 | 1.32–2.20 | <0.01 |
| HDL (per 10 mg/dl) | 0.89 | 0.79–1.00 | 0.05 | 0.79 | 0.68–0.91 | <0.01 | 0.87 | 0.80–0.94 | <0.01 | 0.95 | 0.87–1.04 | 0.27 |
| Education ≥12 yr | 0.65 | 0.48–0.87 | <0.01 | 0.73 | 0.58–0.91 | 0.01 | 0.76 | 0.61–0.96 | 0.02 | 0.84 | 0.66–1.05 | 0.12 |
Numbers in brackets represent individuals with an elevated β-trace protein (≥0.81 mg/L) and elevated β2-microglobulin (≥2.80 mg/L). Triglycerides were analyzed on a logarithmic scale (ln). Triglycerides may be converted from mg/dl to mmol/L by multiplying by 0.0113. HDL may be converted from mg/dl to mmol/L by multiplying by 0.0259. OR, odds ratio; 95% CI, confidence interval; BMI, body mass index; CRP, C-reactive protein.
Reference group is non-Hispanic whites.
Reference group is individuals with CRP <0.22 mg/dl.
Figure 2.
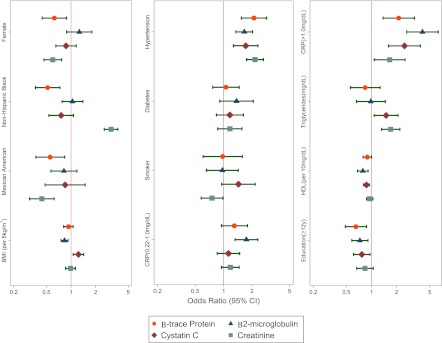
Odds of elevated filtration markers in participants aged ≥20 years. Odds ratios (95% CIs) of an elevated concentration of β-trace protein (≥0.81 mg/L), β2-microglobulin (≥2.80 mg/L), cystatin C (≥1.03 mg/L), and creatinine (men, ≥1.2 mg/dl; women, ≥1.0 mg/dl) after adjusting for age (per 1-year increase), female sex, non-Hispanic black race/ethnicity, Mexican American race/ethnicity, BMI per 5-kg/m2 increase, hypertension, diabetes, current smoking status, CRP from 0.22 to 1.0 mg/dl, CRP >1.0 mg/dl, triglycerides (mg/dl), HDL cholesterol (per 10-mg/dl increase), and education (≥12 years). Reference groups for categorical variables include male sex, non-Hispanic whites, no hypertension, no diabetes, no current smoking status, and an education <12 years. β-trace protein is depicted by a circle, β2-microglobulin is depicted by a triangle, cystatin C is depicted by a diamond, and creatinine is depicted by a square. The age covariate is not shown. BMI, body mass index; CRP, C-reactive protein; 95% CI, 95% confidence interval.
Table 3.
ORs and 95% CIs of elevated serum β-trace protein (≥0.81 mg/L), elevated serum β2-microglobulin (≥2.80 mg/L), and elevated serum cystatin C (≥1.03 mg/L) in persons aged ≥20 years adjusted for eGFR
| β-Trace Protein (≥0.81 mg/L) | β2-Microglobulin (≥2.80 mg/L) | Standard Cystatin C (≥1.03 mg/L) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| ≥20 yr (n=6682 [1220]) | ≥20 yr (n=6688 [1204]) | ≥20 yr (n=6742 [1680]) | |||||||
| OR | 95% CI | P | OR | 95% CI | P | OR | 95% CI | P | |
| eGFR creatinine | 0.92 | 0.90–0.93 | <0.01 | 0.92 | 0.91–0.94 | <0.01 | 0.91 | 0.90–0.92 | <0.01 |
| BMI (per 5 kg/m2 increase) | 0.80 | 0.68–0.95 | 0.01 | 0.74 | 0.66–0.82 | <0.01 | 1.10 | 0.96–1.26 | 0.15 |
| Hypertension | 1.99 | 1.37–2.90 | <0.01 | 1.70 | 1.36–2.14 | <0.01 | 1.71 | 1.18–2.46 | 0.01 |
| Diabetes | 1.03 | 0.67–1.59 | 0.88 | 1.51 | 0.92–2.47 | 0.10 | 1.48 | 0.96–2.28 | 0.08 |
| Current smoker | 0.89 | 0.55–1.44 | 0.64 | 0.82 | 0.54–1.24 | 0.34 | 1.35 | 0.91–2.01 | 0.13 |
| CRP (mg/dl)a | |||||||||
| 0.22–1.0 | 1.26 | 0.90–1.78 | 0.18 | 1.90 | 1.39–2.59 | <0.01 | 1.11 | 0.85–1.45 | 0.42 |
| >1.0 | 2.00 | 1.24–3.25 | 0.01 | 5.70 | 3.52–9.24 | <0.01 | 3.52 | 2.02–6.15 | <0.01 |
| Triglycerides (mg/dl) | 0.56 | 0.36–0.88 | 0.01 | 0.75 | 0.55–1.03 | 0.07 | 1.22 | 0.82–1.80 | 0.32 |
| HDL (per 10 mg/dl increase) | 0.82 | 0.72–0.94 | <0.01 | 0.80 | 0.71–0.90 | <0.01 | 0.84 | 0.76–0.92 | <0.01 |
| Education ≥12 yr | 0.56 | 0.41–0.76 | <0.01 | 0.60 | 0.47–0.77 | <0.01 | 0.59 | 0.46–0.76 | <0.01 |
Numbers in brackets represent individuals with an elevated β-trace protein (≥0.81 mg/L) and elevated β2-microglobulin (≥2.80 mg/L). Triglycerides were analyzed on a logarithmic scale (ln). Triglycerides may be converted from mg/dl to mmol/L by multiplying by 0.0113. HDL may be converted from mg/dl to mmol/L by multiplying by 0.0259. OR, odds ratio; 95% CI, confidence interval; eGFR, estimated GFR; BMI, body mass index; CRP, C-reactive protein.
Reference group is individuals with CRP <0.22 mg/dl.
Distributions of βTP and β2M in a young, healthy subgroup of participants aged 20–39 years without hypertension or diabetes are shown in Table 4. The differences in concentrations of βTP varied significantly by sex and by race/ethnicity, with βTP being greater among men versus women (P<0.01) and non-Hispanic whites versus non-Hispanic blacks and Mexican Americans, respectively (P<0.001 for both non-Hispanic blacks and Mexican Americans). Similar trends were noted for β2M concentrations. Median values were significantly greater among men than women (P of difference <0.001), and among non-Hispanic whites compared with non-Hispanic blacks (P of difference <0.01) and Mexican Americans (P of difference = 0.03).
Table 4.
Percentiles of β-trace protein and β2-microglobulin distribution in persons aged 20–39 years without hypertension or diabetes mellitus overall, by sex, and by race/ethnicity, US population (1988–1994)
| Weighted Percentiles | n | |||||||
|---|---|---|---|---|---|---|---|---|
| 1st | 5th | 25th | 50th | 75th | 95th | 99th | ||
| β-trace protein (mg/L) | ||||||||
| Overall | 0.25 | 0.34 | 0.45 | 0.52 | 0.58 | 0.69 | 0.81 | 1513 |
| Male | 0.22 | 0.34 | 0.47 | 0.54 | 0.60 | 0.72 | 0.80 | 688 |
| Female | 0.25 | 0.34 | 0.44 | 0.50 | 0.55 | 0.66 | 0.96 | 825 |
| Non-Hispanic white | 0.30 | 0.38 | 0.47 | 0.53 | 0.59 | 0.70 | 0.91 | 437 |
| Male | NA | 0.38 | 0.48 | 0.56 | 0.61 | 0.72 | NA | 182 |
| Female | NA | 0.37 | 0.47 | 0.51 | 0.57 | 0.67 | NA | 255 |
| Non-Hispanic black | 0.22 | 0.31 | 0.42 | 0.47 | 0.52 | 0.64 | 0.76 | 502 |
| Male | NA | 0.31 | 0.42 | 0.49 | 0.56 | 0.66 | NA | 269 |
| Mexican American | 0.21 | 0.30 | 0.42 | 0.48 | 0.56 | 0.67 | 0.76 | 509 |
| Male | NA | 0.29 | 0.43 | 0.50 | 0.58 | 0.69 | NA | 243 |
| Female | NA | 0.31 | 0.40 | 0.47 | 0.54 | 0.64 | NA | 266 |
| β2-microglobulin (mg/L) | ||||||||
| Overall | 1.03 | 1.22 | 1.43 | 1.59 | 1.80 | 2.27 | 2.80 | 1514 |
| Male | 1.08 | 1.27 | 1.48 | 1.66 | 1.86 | 2.34 | 2.70 | 689 |
| Female | 1.03 | 1.19 | 1.40 | 1.54 | 1.74 | 2.27 | 2.92 | 825 |
| Non-Hispanic white | 1.04 | 1.26 | 1.46 | 1.62 | 1.81 | 2.27 | 2.82 | 438 |
| Male | NA | 1.31 | 1.49 | 1.68 | 1.86 | 2.21 | NA | 183 |
| Female | NA | 1.23 | 1.43 | 1.58 | 1.74 | 2.29 | NA | 255 |
| Non-Hispanic black | 1.03 | 1.13 | 1.35 | 1.51 | 1.75 | 2.21 | 2.84 | 502 |
| Male | NA | 1.19 | 1.37 | 1.54 | 1.79 | 2.42 | NA | 233 |
| Female | NA | 1.08 | 1.33 | 1.50 | 1.71 | 2.13 | NA | 269 |
| Mexican American | 1.03 | 1.14 | 1.37 | 1.54 | 1.8 | 2.34 | 2.97 | 509 |
| Male | NA | 1.17 | 1.42 | 1.60 | 1.82 | 2.32 | NA | 243 |
| Female | NA | 1.13 | 1.34 | 1.49 | 1.78 | 2.58 | NA | 266 |
There were insufficient numbers to generate sex- and race-specific estimates for the 1st and 99th percentiles. See text for comparison of mean serum concentrations. Sample size shows the number of participants used as the basis for the nationally representative estimates. NA, not available.
Discussion
This article is the first description of the distribution of serum concentrations of βTP and β2M in the US population and expands our understanding of their determinants. The comparisons with creatinine and CysC allow for inferences about factors that may be associated with GFR and non-GFR determinants of their serum concentrations, which cannot be evaluated directly in large population studies. In our study, older age, hypertension, and higher CRP were associated with elevations in all four filtration markers, consistent with lower GFR. In contrast, inconsistent associations across markers were observed for sex, black race/ethnicity, Mexican Americans, BMI, current smoking, triglycerides, HDL cholesterol, and education, suggesting more complex relationships, including associations with non-GFR determinants as well as with GFR.
In our study, serum concentrations of βTP and β2M began to rise in the fifth to sixth decade. This is consistent with prior reports that demonstrated minimal effects of age on βTP (29,30) or β2M (29) in younger adults (aged <22 years), but significant associations with age, particularly for β2M, in older adults (31–33). The positive associations of βTP and β2M with age were also seen with CysC and creatinine, which likely reflect a higher prevalence of decreased GFR with age (27). However, the proportions of older participants with elevations in CysC, βTP, and β2M were more pronounced than creatinine, possibly reflecting that the effects of muscle atrophy on creatinine generation in elderly individuals may be even more pronounced among individuals with CKD than among populations used to estimate the average age association for estimating GFR (34).
There is disagreement in the literature regarding the association of sex with βTP and β2M. Whereas some studies describe higher βTP concentrations in adult men compared with women (35), most studies have shown no sex differences in children (29,30). β2M has not been shown to differ by sex in youth (29), but some studies have reported higher β2M concentrations in adult men compared with women (33), whereas others report no sex differences (31,32). Our data suggest that men have higher concentrations of βTP than women aged <30 years and >50 years, whereas serum concentrations of β2M were generally greater in men aged <40 years. In contrast, we found that CysC and creatinine were greater in men at all ages. Although previous reports have also reported higher creatinine (36) and CysC in men (20), these findings have been inconsistent with measured GFR, which is known to be greater in men versus women (37). This illustrates the utility of a multi-marker approach to estimating GFR, in that one of the markers, β2M, seems to be less affected by sex-based, physiologic differences in marker generation that could be masking underlying differences in GFR. It also suggests the benefits of GFR estimating equations that remove known large non-GFR effects in order to produce an estimate that more specifically reflects kidney function. Estimates based on multiple markers can be combined or averaged to increase precision and can be compared to highlight similarities that are more likely to reflect GFR and differences that are more likely to reflect effects of the non-GFR determinants of the specific filtration markers.
Our data show inconsistent patterns for the relationship between race/ethnicity and βTP and β2M across ages. Non-Hispanic blacks had lower βTP and β2M at younger ages (<30 years), followed by increases during ages 50–70 years, and subsequent declines later in life. A similar trend was described previously for CysC (20) and may reflect incident low GFR at age 50 years before a decrease in survivorship at age ≥70 years. A similar pattern was not observed for creatinine, which exhibited higher values for non-Hispanic blacks versus whites regardless of age. This inconsistency reflects prior reports of higher muscle mass and creatinine production in non-Hispanic blacks (26,38–40). Our data also show that being Mexican American was associated with lower serum concentrations of βTP and β2M across all ages. Similar patterns have been described by others for CysC and creatinine (20,26,38), and may imply a higher level of GFR.
This study represents the largest comparison of βTP, β2M, CysC, and creatinine to date. Furthermore, it was conducted in a well characterized, nationally representative sample of the US population, enabling us to determine normal levels of these markers. The major limitation is that we do not have measured GFR. Nevertheless, although NHANES III does not include measurements of GFR, the availability of questionnaires, physical examination measures, and laboratory specimens allows us to examine associations with a variety of factors that are generally not available in studies with measured GFR. In addition, due to its cross-sectional design, we cannot examine the temporal nature of the observed associations and differential mortality can distort associations. For example, not finding a higher prevalence of elevations of any of the filtration markers among patients with diabetes may reflect the high mortality among diabetic individuals, as well as possible canceling effects of hyperfiltration early in the course of diabetic kidney disease. Finally, we had only single measurements of βTP, β2M, CysC, and creatinine, and these measurements are known to vary within participants, which may contribute to differences in the observed risk factor associations (20,41).
In conclusion, this study describes the distributions of βTP and β2M in the general US population and provides a valuable comparison with creatinine and CysC. Knowledge of the demographic and clinical characteristics associated with these markers suggests that they have different non-GFR determinants. Consideration of these differences will be important for the appropriate interpretation of estimates of GFR or prognosis based on their serum concentration. Future studies should directly examine the relationship between βTP, β2M, and measured GFR, integrating them with CysC and creatinine to optimize estimation of GFR and their use in determining prognosis.
Disclosures
J.C. has consulted for Amgen and Merck and has an investigator-initiated grant from Amgen. L.A.I. has an investigator grant from Pharmalink and Gilead Inc.
Acknowledgments
This project was partially funded by Siemens and by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) (U01 DK067651). S.P.J. is supported in part by a National Heart Lung and Blood Institute cardiovascular epidemiology training grant (T32HL007024). L.A.I. is supported in part by an NIDDK grant (K23DK081017). Siemens had no role in the study design, data collection, analysis, interpretation of data, or report writing.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.08700812/-/DCSupplemental.
References
- 1.Aksun SA, Ozmen D, Ozmen B, Parildar Z, Mutaf I, Turgan N, Habif S, Kumanlioğluc K, Bayindir O: Beta2-microglobulin and cystatin C in type 2 diabetes: Assessment of diabetic nephropathy. Exp Clin Endocrinol Diabetes 112: 195–200, 2004 [DOI] [PubMed] [Google Scholar]
- 2.Viberti GC, Keen H, Mackintosh D: Beta 2-microglobulinaemia: A sensitive index of diminishing renal function in diabetics. Br Med J (Clin Res Ed) 282: 95–98, 1981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tangri N, Inker LA, Tighiouart H, Sorensen E, Menon V, Beck G, Shlipak M, Coresh J, Levey AS, Sarnak MJ: Filtration markers may have prognostic value independent of glomerular filtration rate. J Am Soc Nephrol 23: 351–359, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Astor BC, Shafi T, Hoogeveen RC, Matsushita K, Ballantyne CM, Inker LA, Coresh J: Novel markers of kidney function as predictors of ESRD, cardiovascular disease, and mortality in the general population. Am J Kidney Dis 59: 653–662, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Inker LA, Schmid CH, Tighiouart H, Eckfeldt JH, Feldman HI, Greene T, Kusek JW, Manzi J, Van Lente F, Zhang YL, Coresh J, Levey AS, CKD-EPI Investigators : Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med 367: 20–29, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ridker PM, Rifai N, Rose L, Buring JE, Cook NR: Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first cardiovascular events. N Engl J Med 347: 1557–1565, 2002 [DOI] [PubMed] [Google Scholar]
- 7.Hoffmann A, Nimtz M, Conradt HS: Molecular characterization of beta-trace protein in human serum and urine: A potential diagnostic marker for renal diseases. Glycobiology 7: 499–506, 1997 [DOI] [PubMed] [Google Scholar]
- 8.Olsson JE, Link H, Nosslin B: Metabolic studies on 125I-labelled beta-trace protein, with special reference to synthesis within the central nervous system. J Neurochem 21: 1153–1159, 1973 [DOI] [PubMed] [Google Scholar]
- 9.Ploegh HL, Orr HT, Strominger JL: Major histocompatibility antigens: The human (HLA-A, -B, -C) and murine (H-2K, H-2D) class I molecules. Cell 24: 287–299, 1981 [DOI] [PubMed] [Google Scholar]
- 10.Li W, Mase M, Inui T, Shimoda M, Isomura K, Oda H, Yamada K, Urade Y: Pharmacokinetics of recombinant human lipocalin-type prostaglandin D synthase/beta-trace in canine. Neurosci Res 61: 289–293, 2008 [DOI] [PubMed] [Google Scholar]
- 11.Vincent C, Revillard JP: Beta-2-microglobulin and HLA-related glycoproteins in human urine and serum. Contrib Nephrol 26: 66–88, 1981 [DOI] [PubMed] [Google Scholar]
- 12.Filler G, Bökenkamp A, Hofmann W, Le Bricon T, Martínez-Brú C, Grubb A: Cystatin C as a marker of GFR—history, indications, and future research. Clin Biochem 38: 1–8, 2005 [DOI] [PubMed] [Google Scholar]
- 13.Sampaio MH, de Barros-Mazon S, Sakano E, Chone CT: Predictability of quantification of beta-trace protein for diagnosis of cerebrospinal fluid leak: Cutoff determination in nasal fluids with two control groups. Am J Rhinol Allergy 23: 585–590, 2009 [DOI] [PubMed] [Google Scholar]
- 14.Abbink FC, Laarman CA, Braam KI, van Wijk JA, Kors WA, Bouman AA, Spreeuwenberg MD, Stoffel-Wagner B, Bökenkamp A: Beta-trace protein is not superior to cystatin C for the estimation of GFR in patients receiving corticosteroids. Clin Biochem 41: 299–305, 2008 [DOI] [PubMed] [Google Scholar]
- 15.Evrin PE, Wibell L: Serum 2-microglobulin in various disorders. Clin Chim Acta 43: 183–186, 1973 [DOI] [PubMed]
- 16.Michalski JP, Daniels TE, Talal N, Grey HM: Beta2 microglobulin and lymphocytic infiltration in Sjögren’s syndrome. N Engl J Med 293: 1228–1231, 1975 [DOI] [PubMed] [Google Scholar]
- 17.Bataille R, Magub M, Grenier J, Donnadio D, Sany J: Serum beta-2-microglobulin in multiple myeloma: Relation to presenting features and clinical status. Eur J Cancer Clin Oncol 18: 59–66, 1982 [DOI] [PubMed] [Google Scholar]
- 18.Maury CP, Helve T, Sjöblom C: Serum beta 2-microglobulin, sialic acid, and C-reactive protein in systemic lupus erythematosus. Rheumatol Int 2: 145–149, 1982 [DOI] [PubMed] [Google Scholar]
- 19.US Centers for Disease Control and Prevention: National Health and Nutrition Examination Survey III data files, documentation, and codebooks. Available at: http://www.cdc.gov/nchs/about/major/nhanes/nh3data.htm Accessed August 13, 2011
- 20.Köttgen A, Selvin E, Stevens LA, Levey AS, Van Lente F, Coresh J: Serum cystatin C in the United States: The Third National Health and Nutrition Examination Survey (NHANES III). Am J Kidney Dis 51: 385–394, 2008 [DOI] [PubMed] [Google Scholar]
- 21.US Centers for Disease Control and Prevention: National Health and Nutrition Examination Survey III Surplus Sera Laboratory Component: Cystatin C (Surplus Sera), 2007. Available at: ftp://ftp.cdc.gov/pub/Health_Statistics/NCHS/nhanes/nhanes3/27a/SSCYSTAT.pdf Accessed October 24, 2011
- 22.Juraschek SP, Coresh J, Inker LA, Rynders GP, Eckfeldt JH, Selvin E: The effects of freeze-thaw on β-trace protein and β2-microglobulin assays after long-term sample storage. Clin Biochem 45: 694–696, 2012 [DOI] [PMC free article] [PubMed]
- 23.Selvin E, Manzi J, Stevens LA, Van Lente F, Lacher DA, Levey AS, Coresh J: Calibration of serum creatinine in the National Health and Nutrition Examination Surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis 50: 918–926, 2007 [DOI] [PubMed] [Google Scholar]
- 24.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.National Center for Health Statistics : Plan and operations of the Third National Health and Nutrition Examination Survey, 1988-94 Series 1: Programs and collection procedures. Vital Health Stat 32: 1–407, 1994 [PubMed] [Google Scholar]
- 26.Coresh J, Astor BC, Greene T, Eknoyan G, Levey AS: Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis 41: 1–12, 2003 [DOI] [PubMed] [Google Scholar]
- 27.Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS: Prevalence of chronic kidney disease in the United States. JAMA 298: 2038–2047, 2007 [DOI] [PubMed] [Google Scholar]
- 28.National Center for Health Statistics : Analytic and Reporting Guidelines: The Third National Health and Nutrition Examination Survey, NHANES III (1988-94), Hyattsville, MD, National Center for Health Statistics, Centers for Disease Control and Prevention, 1996 [Google Scholar]
- 29.Bökenkamp A, Franke I, Schlieber M, Düker G, Schmitt J, Buderus S, Lentze MJ, Stoffel-Wagner B: Beta-trace protein—a marker of kidney function in children: “Original research communication-clinical investigation”. Clin Biochem 40: 969–975, 2007 [DOI] [PubMed] [Google Scholar]
- 30.Filler G, Witt I, Priem F, Ehrich JH, Jung K: Are cystatin C and beta 2-microglobulin better markers than serum creatinine for prediction of a normal glomerular filtration rate in pediatric subjects? Clin Chem 43: 1077–1078, 1997 [PubMed] [Google Scholar]
- 31.Norlund L, Fex G, Lanke J, Von Schenck H, Nilsson JE, Leksell H, Grubb A: Reference intervals for the glomerular filtration rate and cell-proliferation markers: Serum cystatin C and serum beta 2-microglobulin/cystatin C-ratio. Scand J Clin Lab Invest 57: 463–470, 1997 [DOI] [PubMed] [Google Scholar]
- 32.Parildar Z, Gulter C, Habif S, Mutaf I, Turgan N, Ozmen D, Bayindir O: Age and gender associated changes in cystatin C and beta2-microglobulin. Turk J Med Sci 32: 317–321, 2002 [Google Scholar]
- 33.Jovanović D, Krstivojević P, Obradović I, Durdević V, Dukanović L: Serum cystatin C and beta2-microglobulin as markers of glomerular filtration rate. Ren Fail 25: 123–133, 2003 [DOI] [PubMed] [Google Scholar]
- 34.Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, De Zeeuw D, Hostetter TH, Lameire N, Eknoyan G: Definition and classification of chronic kidney disease: A position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 67: 2089–2100, 2005 [DOI] [PubMed] [Google Scholar]
- 35.Donadio C, Lucchesi A, Ardini M, Donadio E, Giordani R: Serum levels of beta-trace protein and glomerular filtration rate—preliminary results. J Pharm Biomed Anal 32: 1099–1104, 2003 [DOI] [PubMed] [Google Scholar]
- 36.Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D, Modification of Diet in Renal Disease Study Group : A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Ann Intern Med 130: 461–470, 1999 [DOI] [PubMed] [Google Scholar]
- 37.Wesson L: Physiology of the Human Kidney, New York, Grune & Stratton, 1969 [Google Scholar]
- 38.Jones CA, McQuillan GM, Kusek JW, Eberhardt MS, Herman WH, Coresh J, Salive M, Jones CP, Agodoa LY: Serum creatinine levels in the US population: Third National Health and Nutrition Examination Survey. Am J Kidney Dis 32: 992–999, 1998 [DOI] [PubMed] [Google Scholar]
- 39.James GD, Sealey JE, Alderman M, Ljungman S, Mueller FB, Pecker MS, Laragh JH: A longitudinal study of urinary creatinine and creatinine clearance in normal subjects. Race, sex, and age differences. Am J Hypertens 1: 124–131, 1988 [DOI] [PubMed] [Google Scholar]
- 40.Goldwasser P, Aboul-Magd A, Maru M: Race and creatinine excretion in chronic renal insufficiency. Am J Kidney Dis 30: 16–22, 1997 [DOI] [PubMed] [Google Scholar]
- 41.Selvin E, Juraschek SP, Eckfeldt J, Levey AS, Inker LA, Coresh J: Within-person variability in kidney measures. Am J Kidney Dis 2013, in press [DOI] [PMC free article] [PubMed] [Google Scholar]