Abstract
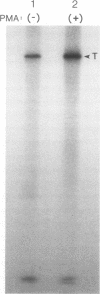
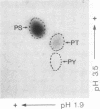
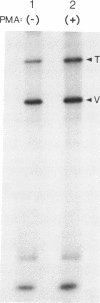
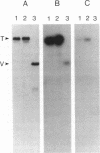
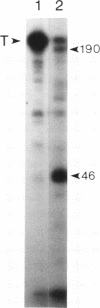
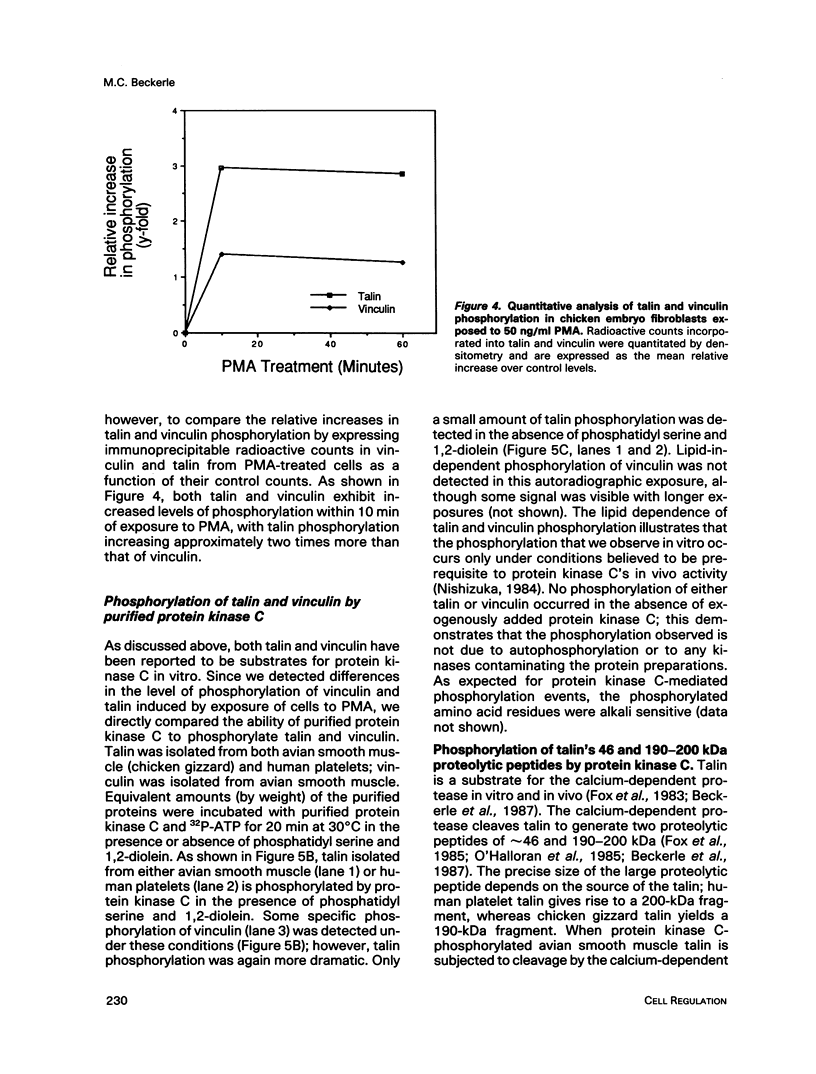
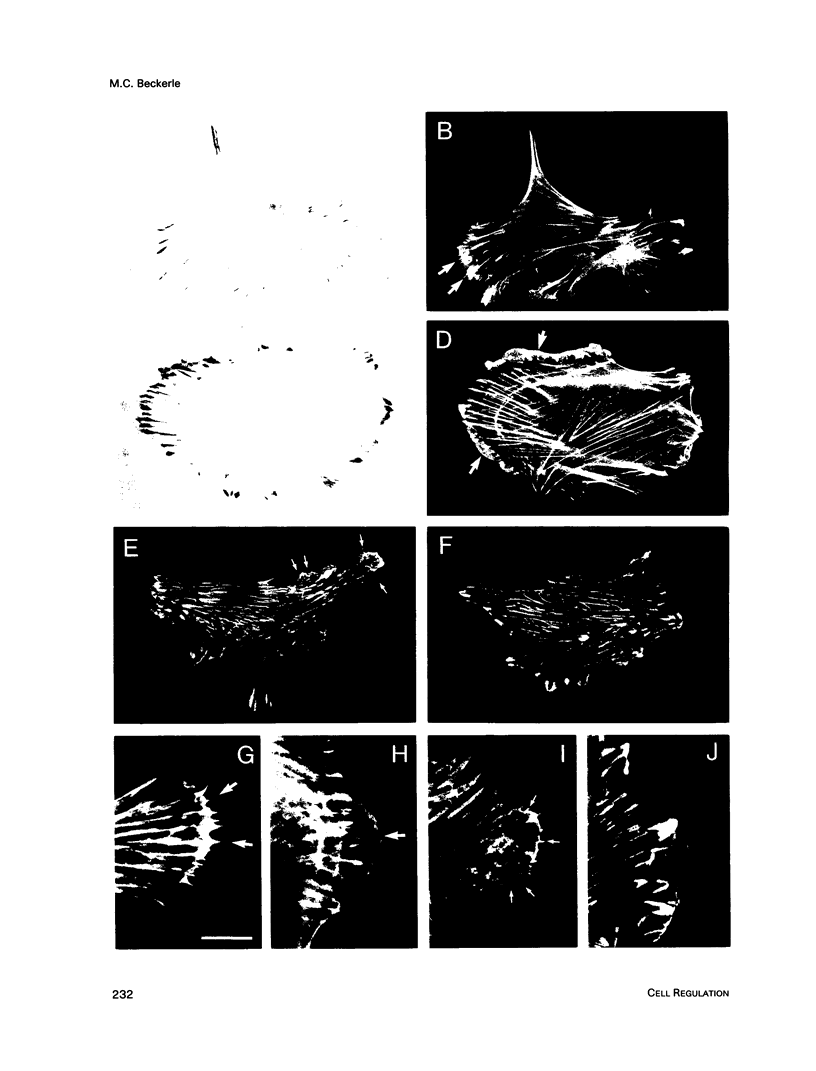
Talin is a high molecular weight phosphoprotein that is localized at adhesion plaques. We have found that talin phosphorylation increases 3.0-fold upon exposure of chicken embryo fibroblasts to the tumor-promoting phorbol ester, phorbol 12-myristate 13-acetate. Talin isolated from tumor promoter-treated cells is phosphorylated on serine and threonine residues. Vinculin, a 130 kDa talin-binding protein, also exhibits increased phosphorylation in vivo in response to tumor promoter, but to a lesser degree than does talin. Because tumor-promoting phorbol esters augment protein kinase C activity, we have compared the ability of purified protein kinase C to phosphorylate talin and vinculin in vitro. Both talin and vinculin were found to be substrates for protein kinase C; however, talin was phosphorylated to a greater extent than was vinculin. Cleavage of protein kinase C-phosphorylated talin by the calcium-dependent protease (Type II) revealed that while both the resulting 190-200 and 46 kDa proteolytic peptides were phosphorylated, the majority of label was contained within the 46-kDa fragment. Although incubation of chicken embryo fibroblasts with tumor-promoting phorbol ester induces a dramatic increase in talin phosphorylation, we detected no change in the organization of stress fibers and focal contacts in these cells. Exposure of the cells to tumor promoter did, however, result in a loss of actin and talin-rich cell surface elaborations that resemble focal contact precursor structures.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beckerle M. C., Burridge K., DeMartino G. N., Croall D. E. Colocalization of calcium-dependent protease II and one of its substrates at sites of cell adhesion. Cell. 1987 Nov 20;51(4):569–577. doi: 10.1016/0092-8674(87)90126-7. [DOI] [PubMed] [Google Scholar]
- Beckerle M. C. Identification of a new protein localized at sites of cell-substrate adhesion. J Cell Biol. 1986 Nov;103(5):1679–1687. doi: 10.1083/jcb.103.5.1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beckerle M. C., O'Halloran T., Burridge K. Demonstration of a relationship between talin and P235, a major substrate of the calcium-dependent protease in platelets. J Cell Biochem. 1986;30(3):259–270. doi: 10.1002/jcb.240300307. [DOI] [PubMed] [Google Scholar]
- Burridge K., Connell L. A new protein of adhesion plaques and ruffling membranes. J Cell Biol. 1983 Aug;97(2):359–367. doi: 10.1083/jcb.97.2.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burridge K., Mangeat P. An interaction between vinculin and talin. Nature. 1984 Apr 19;308(5961):744–746. doi: 10.1038/308744a0. [DOI] [PubMed] [Google Scholar]
- Chen W. T., Greve J. M., Gottlieb D. I., Singer S. J. Immunocytochemical localization of 140 kD cell adhesion molecules in cultured chicken fibroblasts, and in chicken smooth muscle and intestinal epithelial tissues. J Histochem Cytochem. 1985 Jun;33(6):576–586. doi: 10.1177/33.6.3889142. [DOI] [PubMed] [Google Scholar]
- Collier N. C., Wang K. Purification and properties of human platelet P235. A high molecular weight protein substrate of endogenous calcium-activated protease(s). J Biol Chem. 1982 Jun 25;257(12):6937–6943. [PubMed] [Google Scholar]
- Cooper J. A., Sefton B. M., Hunter T. Detection and quantification of phosphotyrosine in proteins. Methods Enzymol. 1983;99:387–402. doi: 10.1016/0076-6879(83)99075-4. [DOI] [PubMed] [Google Scholar]
- Croall D. E., DeMartino G. N. Comparison of two calcium-dependent proteinases from bovine heart. Biochim Biophys Acta. 1984 Aug 14;788(3):348–355. doi: 10.1016/0167-4838(84)90048-7. [DOI] [PubMed] [Google Scholar]
- Damsky C. H., Knudsen K. A., Bradley D., Buck C. A., Horwitz A. F. Distribution of the cell substratum attachment (CSAT) antigen on myogenic and fibroblastic cells in culture. J Cell Biol. 1985 May;100(5):1528–1539. doi: 10.1083/jcb.100.5.1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David-Pfeuty T., Singer S. J. Altered distributions of the cytoskeletal proteins vinculin and alpha-actinin in cultured fibroblasts transformed by Rous sarcoma virus. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6687–6691. doi: 10.1073/pnas.77.11.6687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeClue J. E., Martin G. S. Phosphorylation of talin at tyrosine in Rous sarcoma virus-transformed cells. Mol Cell Biol. 1987 Jan;7(1):371–378. doi: 10.1128/mcb.7.1.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox J. E., Goll D. E., Reynolds C. C., Phillips D. R. Identification of two proteins (actin-binding protein and P235) that are hydrolyzed by endogenous Ca2+-dependent protease during platelet aggregation. J Biol Chem. 1985 Jan 25;260(2):1060–1066. [PubMed] [Google Scholar]
- Fox J. E., Reynolds C. C., Phillips D. R. Calcium-dependent proteolysis occurs during platelet aggregation. J Biol Chem. 1983 Aug 25;258(16):9973–9981. [PubMed] [Google Scholar]
- Herman B., Pledger W. J. Platelet-derived growth factor-induced alterations in vinculin and actin distribution in BALB/c-3T3 cells. J Cell Biol. 1985 Apr;100(4):1031–1040. doi: 10.1083/jcb.100.4.1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horwitz A., Duggan K., Buck C., Beckerle M. C., Burridge K. Interaction of plasma membrane fibronectin receptor with talin--a transmembrane linkage. Nature. 1986 Apr 10;320(6062):531–533. doi: 10.1038/320531a0. [DOI] [PubMed] [Google Scholar]
- Hunter T., Sefton B. M. Transforming gene product of Rous sarcoma virus phosphorylates tyrosine. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1311–1315. doi: 10.1073/pnas.77.3.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izzard C. S. A precursor of the focal contact in cultured fibroblasts. Cell Motil Cytoskeleton. 1988;10(1-2):137–142. doi: 10.1002/cm.970100118. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lin Z. X., Eshleman J., Grund C., Fischman D. A., Masaki T., Franke W. W., Holtzer H. Differential response of myofibrillar and cytoskeletal proteins in cells treated with phorbol myristate acetate. J Cell Biol. 1989 Mar;108(3):1079–1091. doi: 10.1083/jcb.108.3.1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Litchfield D. W., Ball E. H. Phosphorylation of the cytoskeletal protein talin by protein kinase C. Biochem Biophys Res Commun. 1986 Feb 13;134(3):1276–1283. doi: 10.1016/0006-291x(86)90388-8. [DOI] [PubMed] [Google Scholar]
- Meigs J. B., Wang Y. L. Reorganization of alpha-actinin and vinculin induced by a phorbol ester in living cells. J Cell Biol. 1986 Apr;102(4):1430–1438. doi: 10.1083/jcb.102.4.1430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellstroöm K., Höglund A. S., Nistér M., Heldin C. H., Westermark B., Lindberg U. The effect of platelet-derived growth factor on morphology and motility of human glial cells. J Muscle Res Cell Motil. 1983 Oct;4(5):589–609. doi: 10.1007/BF00712117. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. Turnover of inositol phospholipids and signal transduction. Science. 1984 Sep 21;225(4668):1365–1370. doi: 10.1126/science.6147898. [DOI] [PubMed] [Google Scholar]
- O'Halloran T., Beckerle M. C., Burridge K. Identification of talin as a major cytoplasmic protein implicated in platelet activation. Nature. 1985 Oct 3;317(6036):449–451. doi: 10.1038/317449a0. [DOI] [PubMed] [Google Scholar]
- O'Halloran T., Molony L., Burridge K. Purification and assay of vinculin, metavinculin, and talin. Methods Enzymol. 1986;134:69–77. doi: 10.1016/0076-6879(86)34076-x. [DOI] [PubMed] [Google Scholar]
- Otto J. J. Detection of vinculin-binding proteins with an 125I-vinculin gel overlay technique. J Cell Biol. 1983 Oct;97(4):1283–1287. doi: 10.1083/jcb.97.4.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker P. J., Stabel S., Waterfield M. D. Purification to homogeneity of protein kinase C from bovine brain--identity with the phorbol ester receptor. EMBO J. 1984 May;3(5):953–959. doi: 10.1002/j.1460-2075.1984.tb01913.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquale E. B., Maher P. A., Singer S. J. Talin is phosphorylated on tyrosine in chicken embryo fibroblasts transformed by Rous sarcoma virus. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5507–5511. doi: 10.1073/pnas.83.15.5507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schliwa M., Nakamura T., Porter K. R., Euteneuer U. A tumor promoter induces rapid and coordinated reorganization of actin and vinculin in cultured cells. J Cell Biol. 1984 Sep;99(3):1045–1059. doi: 10.1083/jcb.99.3.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sefton B. M., Hunter T., Ball E. H., Singer S. J. Vinculin: a cytoskeletal target of the transforming protein of Rous sarcoma virus. Cell. 1981 Apr;24(1):165–174. doi: 10.1016/0092-8674(81)90512-2. [DOI] [PubMed] [Google Scholar]
- Singer I. I. The fibronexus: a transmembrane association of fibronectin-containing fibers and bundles of 5 nm microfilaments in hamster and human fibroblasts. Cell. 1979 Mar;16(3):675–685. doi: 10.1016/0092-8674(79)90040-0. [DOI] [PubMed] [Google Scholar]
- Ways D. K., Dodd R. C., Gwynne J. T., Earp H. S. Lipoprotein modulation of the intracellular localization of protein kinase C and alteration of phorbol ester-stimulated differentiation in the human monoblastic U937 cell line. Cancer Res. 1986 Dec;46(12 Pt 1):6049–6053. [PubMed] [Google Scholar]
- Werth D. K., Pastan I. Vinculin phosphorylation in response to calcium and phorbol esters in intact cells. J Biol Chem. 1984 Apr 25;259(8):5264–5270. [PubMed] [Google Scholar]