Abstract
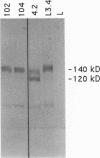
Two biological domains have been identified in the amino terminal region of the neural cell adhesion molecule (NCAM): a homophilic-binding domain, responsible for NCAM-NCAM interactions, and a heparin-binding domain (HBD). It is not known whether these two domains exist as distinct structural entities in the NCAM molecule. To approach this question, we have further defined the relationship between NCAM-heparin binding and cell adhesion. A putative HBD consisting of two clusters of basic amino acid residues located close to each other in the linear amino acid sequence of NCAM has previously been identified. Synthetic peptides corresponding to this domain were shown to bind both heparin and retinal cells. Here we report the construction of NCAM cDNAs with targeted mutations in the HBD. Mouse fibroblast cells transfected with the mutant cDNAs express NCAM polypeptides with altered HBD (NCAM-102 and NCAM-104) or deleted HBD (HBD-) at levels similar to those of wild-type NCAM. Mutant NCAM polypeptides purified from transfected cell lines have substantially reduced binding to heparin and fail to promote chick retinal cell attachment. Furthermore, whereas a synthetic peptide that contains both basic amino acid clusters inhibits retinal-cell adhesion to NCAM-coated dishes, synthetic peptides in which either one of the two basic regions is altered to contain only neutral amino acids do not inhibit this adhesion. These results confirm that this region of the NCAM polypeptide does indeed mediate not only the large majority of NCAM's affinity for heparin but also a significant portion of the cell-adhesion-mediating capability of NCAM.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akeson R. A., Wujek J. R., Roe S., Warren S. L., Small S. J. Smooth muscle cells transiently express NCAM. Brain Res. 1988 Sep;464(2):107–120. doi: 10.1016/0169-328x(88)90003-4. [DOI] [PubMed] [Google Scholar]
- Barthels D., Santoni M. J., Wille W., Ruppert C., Chaix J. C., Hirsch M. R., Fontecilla-Camps J. C., Goridis C. Isolation and nucleotide sequence of mouse NCAM cDNA that codes for a Mr 79,000 polypeptide without a membrane-spanning region. EMBO J. 1987 Apr;6(4):907–914. doi: 10.1002/j.1460-2075.1987.tb04837.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardin A. D., Weintraub H. J. Molecular modeling of protein-glycosaminoglycan interactions. Arteriosclerosis. 1989 Jan-Feb;9(1):21–32. doi: 10.1161/01.atv.9.1.21. [DOI] [PubMed] [Google Scholar]
- Cole G. J., Akeson R. Identification of a heparin binding domain of the neural cell adhesion molecule N-CAM using synthetic peptides. Neuron. 1989 Feb;2(2):1157–1165. doi: 10.1016/0896-6273(89)90182-7. [DOI] [PubMed] [Google Scholar]
- Cole G. J., Burg M. Characterization of a heparan sulfate proteoglycan that copurifies with the neural cell adhesion molecule. Exp Cell Res. 1989 May;182(1):44–60. doi: 10.1016/0014-4827(89)90278-4. [DOI] [PubMed] [Google Scholar]
- Cole G. J., Glaser L. A heparin-binding domain from N-CAM is involved in neural cell-substratum adhesion. J Cell Biol. 1986 Feb;102(2):403–412. doi: 10.1083/jcb.102.2.403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cole G. J., Schubert D., Glaser L. Cell-substratum adhesion in chick neural retina depends upon protein-heparan sulfate interactions. J Cell Biol. 1985 Apr;100(4):1192–1199. doi: 10.1083/jcb.100.4.1192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham B. A., Hemperly J. J., Murray B. A., Prediger E. A., Brackenbury R., Edelman G. M. Neural cell adhesion molecule: structure, immunoglobulin-like domains, cell surface modulation, and alternative RNA splicing. Science. 1987 May 15;236(4803):799–806. doi: 10.1126/science.3576199. [DOI] [PubMed] [Google Scholar]
- Davis G. E., Klier F. G., Engvall E., Cornbrooks C., Varon S., Manthorpe M. Association of laminin with heparan and chondroitin sulfate-bearing proteoglycans in neurite-promoting factor complexes from rat schwannoma cells. Neurochem Res. 1987 Oct;12(10):909–921. doi: 10.1007/BF00966313. [DOI] [PubMed] [Google Scholar]
- Dixit V. M., Hennessy S. W., Grant G. A., Rotwein P., Frazier W. A. Characterization of a cDNA encoding the heparin and collagen binding domains of human thrombospondin. Proc Natl Acad Sci U S A. 1986 Aug;83(15):5449–5453. doi: 10.1073/pnas.83.15.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelman G. M. Morphoregulatory molecules. Biochemistry. 1988 May 17;27(10):3533–3543. doi: 10.1021/bi00410a001. [DOI] [PubMed] [Google Scholar]
- Erickson H. P., Bourdon M. A. Tenascin: an extracellular matrix protein prominent in specialized embryonic tissues and tumors. Annu Rev Cell Biol. 1989;5:71–92. doi: 10.1146/annurev.cb.05.110189.000443. [DOI] [PubMed] [Google Scholar]
- Frelinger A. L., 3rd, Rutishauser U. Topography of N-CAM structural and functional determinants. II. Placement of monoclonal antibody epitopes. J Cell Biol. 1986 Nov;103(5):1729–1737. doi: 10.1083/jcb.103.5.1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorman C. M., Merlino G. T., Willingham M. C., Pastan I., Howard B. H. The Rous sarcoma virus long terminal repeat is a strong promoter when introduced into a variety of eukaryotic cells by DNA-mediated transfection. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6777–6781. doi: 10.1073/pnas.79.22.6777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. Molecular biology of fibronectin. Annu Rev Cell Biol. 1985;1:67–90. doi: 10.1146/annurev.cb.01.110185.000435. [DOI] [PubMed] [Google Scholar]
- Kadmon G., Kowitz A., Altevogt P., Schachner M. The neural cell adhesion molecule N-CAM enhances L1-dependent cell-cell interactions. J Cell Biol. 1990 Jan;110(1):193–208. doi: 10.1083/jcb.110.1.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kouzi-Koliakos K., Koliakos G. G., Tsilibary E. C., Furcht L. T., Charonis A. S. Mapping of three major heparin-binding sites on laminin and identification of a novel heparin-binding site on the B1 chain. J Biol Chem. 1989 Oct 25;264(30):17971–17978. [PubMed] [Google Scholar]
- Lagenaur C., Lemmon V. An L1-like molecule, the 8D9 antigen, is a potent substrate for neurite extension. Proc Natl Acad Sci U S A. 1987 Nov;84(21):7753–7757. doi: 10.1073/pnas.84.21.7753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lander A. D., Fujii D. K., Reichardt L. F. Laminin is associated with the "neurite outgrowth-promoting factors" found in conditioned media. Proc Natl Acad Sci U S A. 1985 Apr;82(7):2183–2187. doi: 10.1073/pnas.82.7.2183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawler J., Hynes R. O. The structure of human thrombospondin, an adhesive glycoprotein with multiple calcium-binding sites and homologies with several different proteins. J Cell Biol. 1986 Nov;103(5):1635–1648. doi: 10.1083/jcb.103.5.1635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lessard J. L., Carlton D., Rein D. C., Akeson R. A solid-phase assay for antiactin antibody and actin using protein-A. Anal Biochem. 1979 Apr 1;94(1):140–149. doi: 10.1016/0003-2697(79)90803-0. [DOI] [PubMed] [Google Scholar]
- Martin G. R., Timpl R. Laminin and other basement membrane components. Annu Rev Cell Biol. 1987;3:57–85. doi: 10.1146/annurev.cb.03.110187.000421. [DOI] [PubMed] [Google Scholar]
- Moran N., Bock E. Characterization of the kinetics of neural cell adhesion molecule homophilic binding. FEBS Lett. 1988 Dec 19;242(1):121–124. doi: 10.1016/0014-5793(88)80998-0. [DOI] [PubMed] [Google Scholar]
- Nybroe O., Moran N., Bock E. Equilibrium binding analysis of neural cell adhesion molecule binding to heparin. J Neurochem. 1989 Jun;52(6):1947–1949. doi: 10.1111/j.1471-4159.1989.tb07283.x. [DOI] [PubMed] [Google Scholar]
- Obara M., Kang M. S., Yamada K. M. Site-directed mutagenesis of the cell-binding domain of human fibronectin: separable, synergistic sites mediate adhesive function. Cell. 1988 May 20;53(4):649–657. doi: 10.1016/0092-8674(88)90580-6. [DOI] [PubMed] [Google Scholar]
- Perris R., Paulsson M., Bronner-Fraser M. Molecular mechanisms of avian neural crest cell migration on fibronectin and laminin. Dev Biol. 1989 Nov;136(1):222–238. doi: 10.1016/0012-1606(89)90144-9. [DOI] [PubMed] [Google Scholar]
- Pierschbacher M. D., Ruoslahti E. Cell attachment activity of fibronectin can be duplicated by small synthetic fragments of the molecule. Nature. 1984 May 3;309(5963):30–33. doi: 10.1038/309030a0. [DOI] [PubMed] [Google Scholar]
- Poltorak M., Sadoul R., Keilhauer G., Landa C., Fahrig T., Schachner M. Myelin-associated glycoprotein, a member of the L2/HNK-1 family of neural cell adhesion molecules, is involved in neuron-oligodendrocyte and oligodendrocyte-oligodendrocyte interaction. J Cell Biol. 1987 Oct;105(4):1893–1899. doi: 10.1083/jcb.105.4.1893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodman J. S., Akeson R. A new antigen common to the rat nervous and immune systems: II. Molecular characterization. J Neurosci Res. 1981;6(2):179–192. doi: 10.1002/jnr.490060204. [DOI] [PubMed] [Google Scholar]
- Rogers S. L., McCarthy J. B., Palm S. L., Furcht L. T., Letourneau P. C. Neuron-specific interactions with two neurite-promoting fragments of fibronectin. J Neurosci. 1985 Feb;5(2):369–378. doi: 10.1523/JNEUROSCI.05-02-00369.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruoslahti E. Fibronectin and its receptors. Annu Rev Biochem. 1988;57:375–413. doi: 10.1146/annurev.bi.57.070188.002111. [DOI] [PubMed] [Google Scholar]
- Ruoslahti E., Pierschbacher M. D. New perspectives in cell adhesion: RGD and integrins. Science. 1987 Oct 23;238(4826):491–497. doi: 10.1126/science.2821619. [DOI] [PubMed] [Google Scholar]
- Rutishauser U., Jessell T. M. Cell adhesion molecules in vertebrate neural development. Physiol Rev. 1988 Jul;68(3):819–857. doi: 10.1152/physrev.1988.68.3.819. [DOI] [PubMed] [Google Scholar]
- Sandrock A. W., Jr, Matthew W. D. Identification of a peripheral nerve neurite growth-promoting activity by development and use of an in vitro bioassay. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6934–6938. doi: 10.1073/pnas.84.19.6934. [DOI] [PMC free article] [PubMed] [Google Scholar]
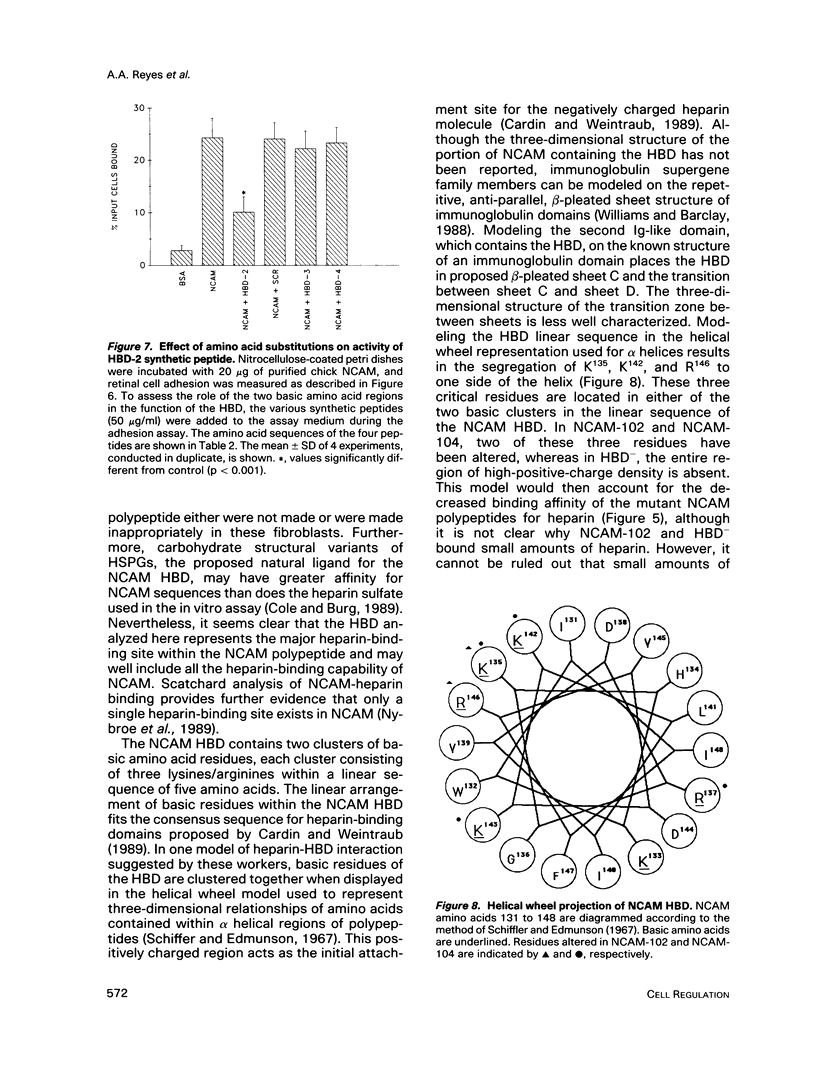
- Schiffer M., Edmundson A. B. Use of helical wheels to represent the structures of proteins and to identify segments with helical potential. Biophys J. 1967 Mar;7(2):121–135. doi: 10.1016/S0006-3495(67)86579-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Small S. J., Shull G. E., Santoni M. J., Akeson R. Identification of a cDNA clone that contains the complete coding sequence for a 140-kD rat NCAM polypeptide. J Cell Biol. 1987 Nov;105(5):2335–2345. doi: 10.1083/jcb.105.5.2335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith J. W., Knauer D. J. Ligand blotting with 125I-fluoresceinamine-heparin. Anal Biochem. 1987 Jan;160(1):105–114. doi: 10.1016/0003-2697(87)90619-1. [DOI] [PubMed] [Google Scholar]
- Suzuki S., Oldberg A., Hayman E. G., Pierschbacher M. D., Ruoslahti E. Complete amino acid sequence of human vitronectin deduced from cDNA. Similarity of cell attachment sites in vitronectin and fibronectin. EMBO J. 1985 Oct;4(10):2519–2524. doi: 10.1002/j.1460-2075.1985.tb03965.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeichi M. The cadherins: cell-cell adhesion molecules controlling animal morphogenesis. Development. 1988 Apr;102(4):639–655. doi: 10.1242/dev.102.4.639. [DOI] [PubMed] [Google Scholar]
- Van Doren K., Hanahan D., Gluzman Y. Infection of eucaryotic cells by helper-independent recombinant adenoviruses: early region 1 is not obligatory for integration of viral DNA. J Virol. 1984 May;50(2):606–614. doi: 10.1128/jvi.50.2.606-614.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. F., Barclay A. N. The immunoglobulin superfamily--domains for cell surface recognition. Annu Rev Immunol. 1988;6:381–405. doi: 10.1146/annurev.iy.06.040188.002121. [DOI] [PubMed] [Google Scholar]