Abstract
Purpose
Recent results have demonstrated that myeloproliferative neoplasms (MPN) are strongly associated with constitutive activation of the JAK2 tyrosine kinase. However, JAK2 inhibitors currently approved or under development for treating myeloproliferative neoplasms do not selectively deplete the malignant clone, and the inhibition of activity of the drug target (JAK2) has not been rigorously evaluated in clinical studies. Therefore in this study we developed an in vitro assay to gain insight into how effectively JAK2 activity is inhibited in patient samples.
Experimental Design
We treated primary cells from normal donors and patients with MPN with JAK2 inhibitors and measured phosphorylation of downstream targets STAT5 and STAT3 by flow cytometry. Obtained results were next correlated with JAK2 V617F allele burden and plasma cytokines level.
Results
We observed a dose-dependent decrease in pSTAT5 and pSTAT3 in ex vivo treated granulocytes. However, phosphorylation of STAT3 and STAT5 in cells from patients with myelofibrosis was significantly less inhibited when compared to cells from patients with polycythemia vera, essential thrombocytosis, and normal donors. Sensitivity to inhibition did not correlate with JAK2 V617F clonal burden. Mixing studies using plasma from patients with myelofibrosis did not transfer resistance to sensitive cells. Likewise, no single cytokine measured appeared to account for the observed pattern of resistance. Taken together these observations suggest that there are cell intrinsic mechanisms that define a priori resistance to JAK2 inhibition in myelofibrosis, and the lesion is localized upstream of STAT3 and STAT5.
Keywords: JAK2 inhibition, myeloproliferative neoplasms, biomarkers, phospho-specific flow cytometry
Introduction
Polycythemia vera (PV), essential thrombocythemia (ET), and myelofibrosis (MF) are myeloproliferative neoplasms (MPN) unified by the constitutive activation of the JAK2 pathway, conferred most commonly by a point mutation in the pseudokinase domain of JAK2 (JAK2V617F)(1–4). The identification of this mutation has prompted wide interest in targeting JAK2 for therapeutic benefit. Myelofibrosis, whether primary or evolved from PV or ET, has a variable but overall poor prognosis. Clinical investigation of JAK2 inhibitors has focused on myelofibrosis, with some unexpected results: while reduction in spleen size and amelioration of constitutional symptoms are frequent and can be dramatic, only modest changes in mutant clonal burden have been observed and complete responses by any criteria are exceedingly rare(5–7).
Notably, validation of inhibition of activity of the drug target (JAK2) has never been rigorously evaluated in clinical studies. Janus Kinase (JAK) family proteins are non-receptor tyrosine kinases that associate with cytokine receptors. Cytokine engagement with receptor leads to the phosphorylation of JAK2 and the recruitment and phosphorylation of cytoplasmic signal transducers and activators of transcription (STAT3 and 5). Upon activation, phosphorylated STAT3 and STAT5 (pSTAT3 and pSTAT5) form homodimers and translocate to the nucleus to activate transcription. In MPN, JAK2 with a V617F substitution is constitutively active, and confers cytokine independence/hypersensitivity to hematopoietic tissues. Small molecule tyrosine kinase inhibitors have been shown to be active in both preclinical and late-phase clinical testing, with analyses of cytokine arrays and measurement of JAK2V617F clonal burden used as potential biomarkers for response(6, 8). In order to study the efficacy of JAK2 inhibitors to inhibit JAK signaling in primary patient cells, we used phospho-specific flow cytometry on whole peripheral blood from patients with MPN and normal controls to measure phosphorylation of STAT3 and STAT5, the canonical targets of JAK2. We hypothesized that this measurement would be a feasible and informative pharmacodynamic assay for measuring JAK2 signaling in patients on clinical trials and that differences in signaling might be observed across the spectrum of disease.
MATERIALS AND METHODS
Peripheral blood samples
Peripheral blood samples from patients and normal donors were collected, annotated and stored through an institutional review board approved hematologic diseases tissue bank. Previously collected, anonymous samples were obtained from the Stem Cell and Xenograft Core facility at the University of Pennsylvania.
Reagents
JAK2 inhibitors: CEP701 (lestaurtinib) was provided by Cephalon Oncology (Frazer, PA) or purchased (LC laboratories, Woburn, MA). CYT387 and INCB18424 were synthesized by ChemieTek (Indianapolis, IN) and kindly provided by Dr Ross Levine (Memorial Sloan-Kettering Cancer Center, New York, NY). Antibodies were purchased from BD Bioscience (San Jose, CA) and R&D Systems (Minneapolis, MN).
Flow cytometry
Whole blood samples were fixed and permeabilized using a formaldehyde/triton/methanol method (9, 10) and analyzed by flow cytometry. Briefly, 1003L of whole blood was exposed to varying concentrations of inhibitor for 15 minutes at 37°C and then stimulated for 20 minutes at 37°C with 10ng/mL GM-CSF (for pSTAT5 measurement) or 100ng/mL G-CSF (for pSTAT3 measurement). After stimulation samples were fixed with 4% formaldehyde for 10min at room temperature, and permeabilized with 0.1% Triton X-100 for 15 minutes at 37°C. Next samples were washed twice in cold PBS supplemented with 4% BSA, treated with cold 100% methanol to enhance epitope availability, and stored at −20°C. Prior to analysis samples were washed twice in a cold PBS supplemented with 4% BSA and then incubated with directly labeled antibodies at room temperature for 30 minutes in the dark. Data were acquired on a BD FACSCalibur using CellQuest Pro software, and analyzed using FlowJo version 9.3.1. For each patient sample, untreated mean fluorescence intensity (MFI) of pSTAT3 and pSTAT5 was subtracted from treated samples and MFI for cytokine-stimulated samples was set at 100%. Persistence of pSTAT3 and pSTAT5 in the presence of inhibitor (X) was calculated as the ratio of mean fluorescence values of gated events relative to cytokine-stimulated sample; inhibition is described as 100 – X. Statistical analysis was performed using Graph Pad Prism 5.
JAK2 V617F Allele burden
Mononuclear cells and granulocytes were separated by density using Ficoll-Paque (Amersham Bioscience). The genomic DNA from granulocytes was isolated from cells using Gentra Purgene Blood Kit from Qiagen (Valencia, CA). JAK2V617F allele burden was measured using a quantitative RT-PCR assay adapted from Nussenzveig et al. (11) to detect the G→T substitution at position 1849 in exon 14 that employs an allele specific (wild-type vs. mutant) primer with specificity enhanced by mismatch and a locked nucleic acid modification. Allele frequency was calculated as described by Germer et al (12).
Determination of cytokines level in plasma from patients and normal donors
Plasma samples were obtained by centrifugation of whole peripheral blood at 600g for 10 minutes and stored at −80°C. Batched frozen samples were analyzed by the Human Immunology Core at the University of Pennsylvania using a Luminex instrument (Austin, TX) and the Milliplex MAP Human cytokine 9-plex from Millipore.
Statistics
One-way ANOVA and t test were used to analyze differences between patient and normal donor samples. Correlations were calculated using the Spearman correlation coefficient. All calculations were made using GraphPad Prism 5.
RESULTS
Phosphorylation of STAT5 and STAT3 is detectable with cytokine stimulation and is inhibited in the presence of CEP701
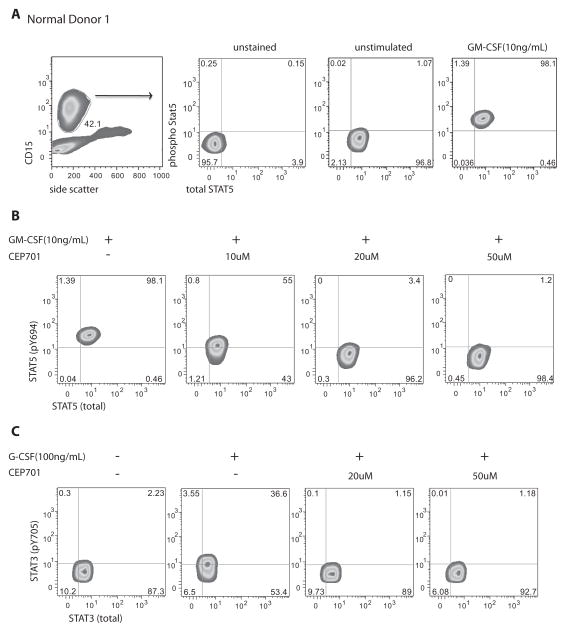
In order to more precisely validate target and to define biologic response to treatment with JAK2 inhibitors in patients with MPN, we investigated the level of target inhibition in peripheral blood samples collected from 29 patients with MPN (Table 1) and 6 normal donors. We measured phosphorylated STAT3 and 5 (pSTAT3 and pSTAT5) as biomarkers for JAK2 inhibition. In our initial studies we did not observe constitutive basal phosphorylation of STAT3 or STAT5 in any samples evaluated (Fig. 1A and Supplementary Fig. S1A, p=0.48). Therefore, we incorporated cytokine activation into the assay. Unmanipulated whole blood from normal donors or patients was stimulated with GM-CSF or G-CSF known to activate phosphorylation of STAT5 or STAT3, respectively in human myeloid cells(13, 14). Activation was measured in granulocytes by gating on CD15+ cells. There were no differences in cytokine responsiveness across disease and normal samples. The total level of intracellular STAT5 was measured to ensure adequate permeabilization of cells and did not change with stimulation or inhibition (Supplementary Fig. S1B–C).
Table 1.
Patient characteristics
| ID # | Registry ID | Diagnosis | Age | Sex | Race | WBC (THO/uL) | % Blasts | HGB (g/dL) | PLT (THO/uL) | Cytogenetics | Treatment History |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2211 | R1267 | Post-PV Myelofibrosis | 74 | M | W | 9.8 | 0 | 9.5 (t) | 49 | 46,XY [16] | PB, E |
| 2212 | R0602 | Polycythemia Vera | 56 | F | W | 4.6 | 0 | 14.6 | 356 | 46,XX [26] | PB, HU |
| 2222 | R1243 | Primary Myelofibrosis | 68 | F | W | 5.2 | 0 | 8.5 (t) | 222 | 46,XX [22] | THAL/PRED |
| 2223 | R1092 | Essential Thombocytosis | 82 | F | W | 8.7 | 0 | 12.8 | 600 | 46,XX [20] | HU, ANAG, |
| 2273 | R0682 | Essential Thombocytosis | 78 | M | W | 6.5 | 0 | 12.6 | 402 | 46,XY [8] | HU, |
| 2283 | R0633 | Primary Myelofibrosis | 46 | F | W | 4.1 | 0 | 9.1 | 245 | 46,XX[20] | ANAG, |
| 2294 | R1300 | Primary Myelofibrosis | 65 | F | W | 5.5 | 0 | 8.9 | 129 | 46,XX [20] | ES |
| 2310 | R0973 | RARS-T | 67 | F | AA | 12.8 | 1 | 8.4 | 1961 | 46,XX [20] | HU, ANAG, IFN, E |
| 2316 | R1231 | Primary Myelofibrosis | 67 | M | W | 13.7 | 5 | 10.7 | 123 | 46,XY [20] | ES |
| 2317 | R1312 | Polycythemia Vera | 78 | F | W | 13.1 | 0 | 14.8 | 346 | ND | PB, H |
| 2327 | R1319 | Polycythemia Vera | 41 | M | W | 4.2 | 0 | 14.4 | 171 | ND | PB, AS |
| 2328 | R1320 | Essential Thombocytosis | 46 | M | W | 5 | 0 | 13.4 | 320 | 46,XY [20] | HU |
| 2340 | R1231 | Primary Myelofibrosis | 67 | M | W | 11.6 | 3 | 9.3 | 93 | 46,XY [20] | ES |
| 2341 | R1325 | Primary Myelofibrosis | 66 | M | W | 18.1 | 0 | 10.1 | 1183 | 46,XY [21] | ESA, H |
| 2343 | R1322 | Polycythemia Vera | 56 | M | W | 12.1 | 0 | 18.2 | 443 | ND | PB, A |
| 2359 | R1182 | Essential Thombocytosis | 73 | F | AA | 4.9 | 0 | 13.3 | 721 | 46,XX [20] | HU, |
| 2369 | R0633 | Primary Myelofibrosis | 47 | F | W | 4.1 | 2 | 9.7 | 217 | ND | ANAG, |
| 2370 | R1269 | Primary Myelofibrosis | 58 | F | W | 9.5 | 0 | 13.4 | 89 | ND | none |
| 2371 | R1332 | Essential Thombocytosis | 40 | M | W | 13.7 | 0 | 17.1 | 608 | 46,XY [20] | AS |
| 2469 | R0682 | Essential Thombocytosis | 78 | M | W | 9.4 | 0 | 13.2 | 550 | 46,XY [20] | HU, |
| 2470 | R1362 | Polycythemia Vera | 45 | F | W | 4.4 | 0 | 12.8 | 368 | 46,XX [25] | ANAG, A |
| 2479 | R1308 | AML (from Post-PV MF) | 62 | M | AA | 121.9 | 35 | ND (t) | 91 | complex* | PB, HU, A |
| 2487 | R0670 | Post-PV Myelofibrosis | 57 | F | W | 4.7 | 2 | 9.6 | 133 | complex** | PB, HU, ASA, I |
| 2517 | R1378 | Primary Myelofibrosis | 63.1 | M | W | 7.6 | 0 | 12.3 | 110 | 46,XY [20] | ANAG, A |
| 2521 | R1236 | Primary Myelofibrosis | 81.8 | F | W | 5.1 | 22 | 10.6 | 87 | 46,XX [20] | HU, E |
| 3013 | R1550 | Polycythemia Vera | 43 | M | W | 4.3 | 0 | 12.7 | 227 | 46,XY [20] | HU, IFN, P |
| 3014 | R0639 | Essential Thombocytosis | 60.5 | F | W | 7 | 0 | 11.5 | 1040 | ND | ANAG, H |
| 3016 | R1589 | Polycythemia Vera | 59 | M | AA | 6.8 | 0 | 15 | 304 | 46,XY [20] | ANAG, H |
| 2518 | R1319 | Polycythemia Vera | 42 | M | W | 3.1 | 0 | 12.6 | 122 | ND | ASA, P |
ND - no data; POS - positive but unknown percentage; HSCT - hematopoietic stem cell transplantation; IFN - interferon; PB - phlebotomy; HU - hydroxyurea; ASA- aspirin; ESA - erythropoietin/darbopoietin;
47,XY,+der(9)t(1;9)(q21;q21)[1]/47, idem,del(5)(q21q34)[19];
46,XX+der(9)t(1;9)(q12;q21)[20]
Figure 1. Measurement of STAT5 and STAT3 phosphorylation in normal peripheral blood granulocytes.
Panel A shows the gating strategy for isolating granulocytes based upon forward/side scatter properties and expression of CD15 antigen. Total (x-axis) and phospho- (y axis) STAT5 (panel B) and STAT3 (panel C); detection of total STAT3 or STAT5 served as an internal control for permeabilization. Panels B and C represent target inhibition in response to exposure to CEP701 at 10, 20 and 50μM doses.
We next used this assay to study the efficacy of JAK2 inhibitors (Fig. 1B–C). For these studies, blood from normal donors (ND), patients with ET, PV and patients with the more advanced disease, myelofibrosis (MF, either primary or evolved from PV or ET) was first exposed to inhibitor, primarily lestaurtinib (CEP701) at a concentration of 10– 50μM and then stimulated with GM-CSF or G-CSF to activate phosphorylation of STAT5 (Fig. 1B) or STAT3 (Fig. 1C), respectively. The range of inhibitor concentration was based upon mean steady state plasma concentrations of 7.7 – 12 μM (range 1 –40 μM) previously described in treated patients for CEP701(15), and from published pharmacokinetic information on achievable concentrations, when available, for INCB18424 and CYT387(16). We observed a dose-dependent decrease in cytokine activated pSTAT3 and pSTAT5 in CD15+ granulocytes from normal donors when treated ex vivo with CEP701 (Fig. 1B–C).
Neutrophils from patients with MF are intrinsically resistant to JAK2 inhibitors
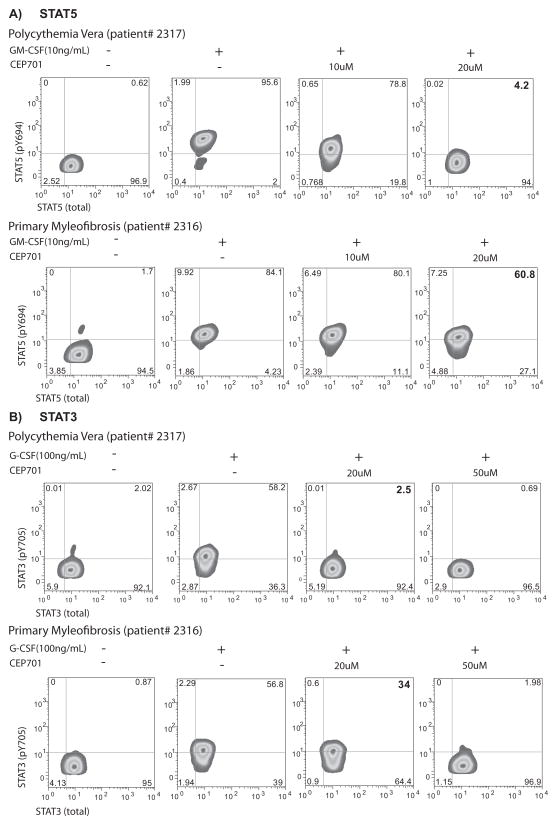
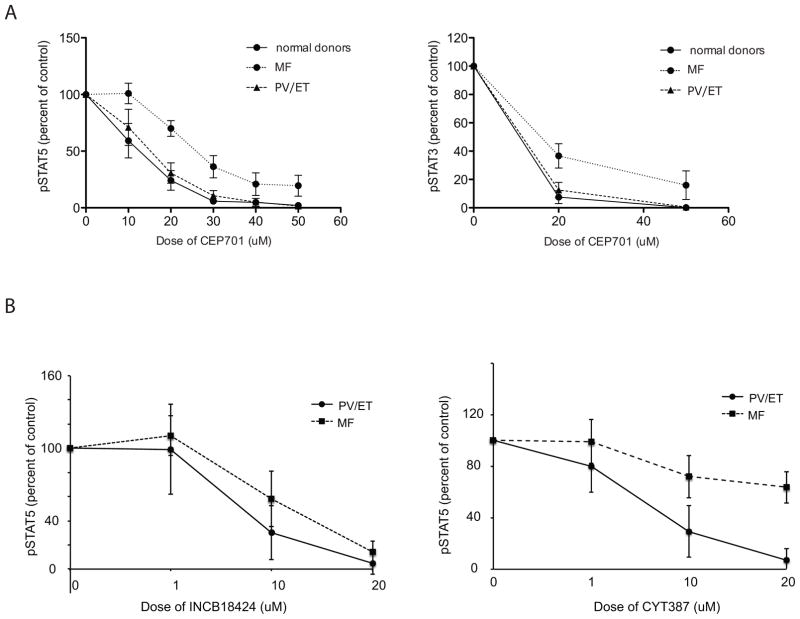
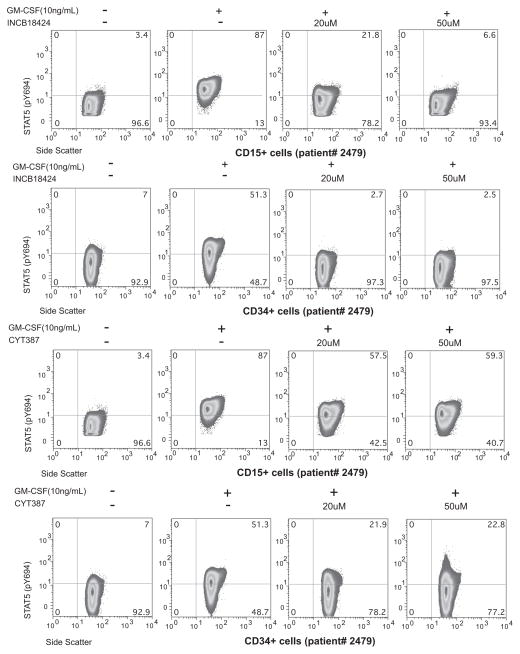
Next, we sought to measure response to treatment across MPN phenotypes. We found that the response to JAK2 inhibitor differs among MPN patients: while phosphorylation of STAT5 (Fig. 2A) and STAT3 (Fig. 2B) in both ET and PV was similar and nearly completely abrogated in the presence of 20 μM CEP701, pSTAT3 and pSTAT5 in MF samples was minimally inhibited (Fig. 2A–B). In aggregate across samples, the mean inhibition at 20μM in ET/PV samples as measured by pSTAT5 (69%), was significantly greater than that in MF samples (36%, p = 0.015; Fig. 3A and Supplementary Table S1). The response to inhibition in normal donors (76% inhibited) was comparable to that observed in ET and PV samples (pSTAT5, 68.9% inhibited p= 0.90). For pSTAT3, PV/ET samples were more sensitive (73% inhibited) compared to MF samples (60% inhibited) although this was not significant (p = 0.43). This suggests either that downstream signaling through STAT3 in MF samples is truly more responsive to inhibition than STAT5 or may reflect a more subtle difference not captured in this number of samples: we also noted a narrower dynamic range for pSTAT3 measurements (Supplementary Fig. S1B). Thus we observed that pSTAT5 and pSTAT3 can be measured in whole blood by phospho-flow cytometry in the presence of exogenous cytokine, and that pharmacologic inhibition is both dose- and MPN-subtype dependent. To extend this observation using two additional tyrosine kinase inhibitors, we repeated these studies with CYT387 and INCB18424, agents currently being investigated/approved for the treatment of MF. We found MF samples to be similarly less sensitive to inhibition with these compounds when compared to PV samples (Fig. 3B). MF samples exposed to 10 and 20μM CYT387 were significantly more resistant (28% and 36.4% inhibition of pSTAT5 respectively) then PV samples (74% and 93% inhibition, p= 0.003 for 10 μM; p=0.001 for 20 μM). Differences in response observed to INCB18424 were also significant (10 μM; MF samples were 42% inhibited while PV samples were 72% inhibited, p=0.025). Taken together, these results suggest that peripheral blood neutrophils from patients with MF were intrinsically resistant to JAK2 inhibitors. To begin to understand what, if any, coherence might exist between terminally differentiated neutrophils and more primitive progenitor compartments with respect to signaling response, we measured pSTAT5 in CD15+ and CD34+ cells exposed to CYT387 and INCB18424 from a patient with acute myeloid leukemia that had evolved from post-PV MF (Fig. 4). We found that CD34+ stimulation with GM-CSF results in a more heterogeneous pSTAT5 signal, likely reflecting differences in GM-CSF receptor expression in this compartment(17), and that the response to inhibition in CD34+ cells generally mirrors that in CD15+ cells.
Figure 2. Relative resistance to inhibition of STAT5 and STAT3 phosphorylation in myelofibrosis.
Representative flow cytometry plots show level of inhibition of STAT5 (A) and STAT3 (B) phosphorylation after exposure to CEP701 for patients respectively diagnosed with PV (top panels) and Primary MF (bottom panels).
Figure 3.
A) CD15+ cells from myelofibrosis patients are significantly more resistant to JAK2 inhibition than cells from patients with PV, ET and normal controls. Graphs show molecular response to various doses of JAK2 inhibitor - CEP701 in normal donors compared to PV/ET and MF patient samples. Molecular response to the drug is presented as a percent of STAT5 (left panel) and STAT3 (right panel) phosphorylation remaining after exposure to CEP701. Mean fluorescence from cells stimulated with GM-CSF for pSTAT5 or G-CSF for pSTAT3 minus background (mean fluorescence of unstimulated cells) was set as a 100%.
B) Resistance to inhibition can be generalized to additional JAK2 inhibitors under clinical development. Response to CYT387 (right panel) and INCB18424 (left panel) are plotted as the percent STAT5 phosphorylation after exposure to JAK2 inhibitors. Mean fluorescence intensity from cells stimulated with GM-CSF for pSTAT5 minus background (mean fluorescence of unstimulated cells) was set as a 100%.
Figure 4. Coherence between terminally differentiated and progenitor signaling response.
Measurement of pSTAT3 and pSTAT5 in CD15+ and CD34+ cells exposed to INCB18424 (top panels) and CYT387 (bottom panels) from a patient with acute myeloid leukemia evolved from post-PV myelofibrosis.
The pattern of resistance is not correlated with JAK2 V617F allele burden in MF
To gain insight into a mechanism by which STAT3 and STAT5 remain phosphorylated even in the presence of upstream JAK2 inhibition, we investigated whether we could detect a correlation between the frequency of JAK2 V617F mutant allele burden in samples from patients with the mutation and response to drug. Overall we found no relationship between allele frequency and level of target inhibition in tested samples (Table 2 and Supplementary Fig. S2). Likewise, a similar (and near complete) pattern of resistance was observed in a patient with MF who was negative for the JAK2 V617F mutation. Thus, the mutant to wild-type ratio of JAK2 does not appear to directly account for the observed pattern of resistance. We did note that in cells from patients without MF, there indeed appeared to be a significant direct correlation with higher levels of JAK2 V617F and persistent signaling (p = 0.04) although we interpret this cautiously, given the numbers of samples (n= 2) from patients with lower allele burdens.
Table 2.
Level of mutant JAK2 V617F allele frequency in granulocytes does not correlate with response to inhibition of phosphoSTAT5
| Stratified byJAK2 V617F allele freq | Mean allele freq (%) | Mean % pSTAT5 AT 20μM | n |
|---|---|---|---|
|
All patients with JAK2 V617 mutation
| |||
| < 50% | 17 | 49.6 | 5 |
| > 50% | 74 | 48.6 | 11 |
|
| |||
|
Polycythemia Vera/Essential Thrombocytosis
| |||
| < 50% | 31 | 4.05 | 2 |
| > 50% | 75 | 35.74 | 7 |
|
| |||
|
Myelofibrosis
| |||
| < 50% | 8 | 80.03 | 3 |
| > 50% | 73 | 71.02 | 4 |
Plasma components do not confer resistance to JAK2 inhibition
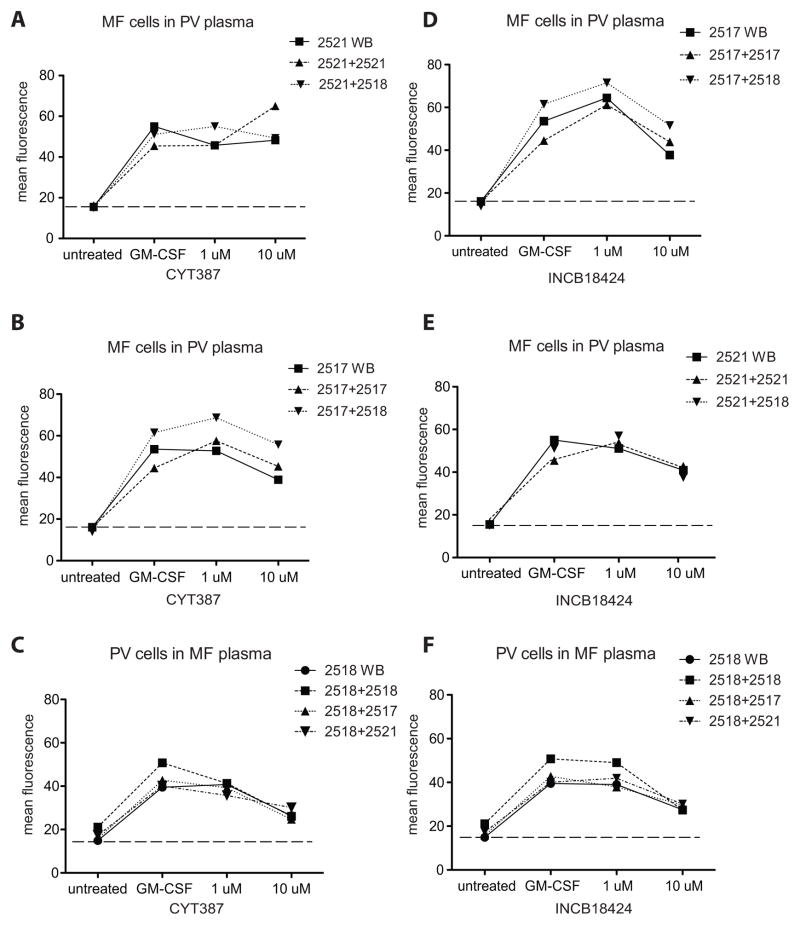
We next investigated the possibility that plasma components might contribute to or confer resistance of MF samples to JAK2 inhibitors. We mixed cells from a PV patient (sample# 2518) with plasma from MF patients (samples #: 2517 and 2521) and evaluated inhibition of pSTAT5 (Fig. 5A–F). For a given patient, response to CYT387 or INCB18424 as measured by mean pSTAT5 fluorescence followed a similar trend regardless of plasma source, which suggested that neither resistance to inhibition nor sensitivity to treatment was conferred by plasma components. We also analyzed basal cytokines levels in plasma samples from MPN samples and normal donors. Plasma cytokines that signal through JAK2 and/or are known to be altered in myelofibrosis were measured. No meaningful differences that could directly explain the differences in response were observed. For most of the nine evaluated cytokines (MIP-1β, TNF-α, IL-6, GM-CSF, IFN-γ, G-CSF, IFN-α and Rantes) the observed concentration in MPN patients was higher than that in normal donors, consistent with other reports (6, 18)(Supplementary Fig. S3). In particular, the basal plasma levels of the two cytokines also used exogenously in our experiments (G- and GM-CSF) were plotted against response to inhibitor, and there was no significant correlation between basal GM-CSF levels and response to inhibitor (p = 0.18, Supplementary Fig. S3B–D). These observations thus fail to demonstrate a soluble cell extrinsic factor that explains JAK2 inhibitor resistance in granulocytes from patients with MF.
Figure 5. Plasma components do not alter sensitivity to JAK2 inhibitor.
Effect of plasma components on response to CYT387 (panels A–C) or INCB18424 (panels D–F) as measured by STAT5 phosphorylation and presented as MFI. Peripheral blood cells from two MF patients (2521, panel A and 2517, panel B) were treated ex vivo with GM-CSF in the presence or absence of CYT387, as whole, unperturbed blood (WB), as plasma-depleted blood reconstituted with native plasma (e.g. 2521+2521) or with PV patient (2518) plasma of equivalent volume (e.g. 2521+2518). Likewise, PV peripheral blood was exposed to CYT387 (panel C) or INCB18424 (panel F) as whole blood, native reconstituted blood, or with plasma from either MF patient (2518+2517 and 2518+2521). MF blood cells (2517, panel D and 2521, panel E) were treated with GM-CSF +/− INCB18424 in native plasma or PV plasma.
DISCUSSION
Our findings demonstrate that in myelofibrosis, cell intrinsic properties bestow resistance to JAK2 inhibition, although the mechanism remains unclear. We found that overall PV and ET samples are sensitive to inhibition by multiple investigational inhibitors, with responses overall comparable to those of normal donors. By contrast, in cells from patients with treatment-naïve myelofibrosis, STAT5 remained phosphorylated despite treatment with inhibitors, suggesting either incomplete upstream inhibition of JAK2 and/or alternative inputs to STAT5 (and possibly STAT3) that are specific to myelofibrosis, typically a more advanced and genetically complex disease. Our studies did not support a role for specific cell-extrinsic soluble factors in plasma as major mediators of resistance, nor could we correlate the clonal burden of JAK2 V617F with the resistance phenotype, suggesting that additional acquired features specific to myelofibrosis mediated the attenuated response.
Our signaling studies focused largely on mature, terminally differentiated neutrophils from patients, an experimental design conceived with the intent of developing a feasible peripheral blood-based pharmacodynamic assay for measuring response to treatment. But whether our observation in neutrophils extends to signaling in more primitive, disease-initiating compartments is also of great interest. Our exploratory studies of CD34+ blasts and neutrophils from a patient with acute leukemia evolved from post-PV myelofibrosis demonstrated a response that mirrors the response in granulocytes, supporting the possibility that a signaling phenotype carries through to terminal differentiation, but additional studies are needed to draw major conclusions about signaling responses throughout differentiation. Of note, other studies have confirmed that granulocytes from patients with myelofibrosis are derived largely from the malignant clone(19). While our studies cannot directly distinguish signaling within clonally derived granulocytes versus those derived from the pre-malignant stem cell pool, the signaling patterns in myelofibrosis tended to be relatively uniform, and would be expected to represent, at least proportionally, clonally-derived cells. By contrast, clonal dominance is not the predominant pattern in PV and ET, and this may explain the overlapping signaling phenotype between PV, ET, and normal donors(19). While we found no direct correlation between JAK2 V617F allele burden and resistance in MF, we found that samples from ET or PV patients with higher JAK2 V617F appeared to be less sensitive to treatment than samples with lower allele burdens. Taken together one wonders whether clonal dominance in a bulk population might influence probability of individual cell signaling.
Acquired resistance to targeted therapies has been well described(20) although classical escape mutations do not appear to be a major mechanism of resistance to JAK2 inhibitors. Perhaps unique features of this target – JAK2 is absolutely required for normal hematopoiesis – and the narrow therapeutic window as a consequence, lead to less selective pressure for outgrowth of resistant clones, so that classical gatekeeper mutations as seen in chronic myeloid leukemia would not be favored. Elegant experimental engineered ex vivo systems of chronic drug exposure have likewise demonstrated that resistance mutations do not account for persistent (or reactivated) downstream signaling in JAK2 V617F cells that grow despite the presence of inhibitor(21). Rather, signaling in trans of JAK2 by JAK1 (and TYK2) appears to mediate a novel mechanism of drug insensitivity that appears to be epigenetically determined at the JAK2 locus itself. Our data are distinguished from these in that cells have not been chronically exposed to drug but instead are derived from treatment naïve patients, and yet our observations may have similar underpinnings: myelofibrosis is increasingly recognized as an entity governed by epigenetic dysregulation that confers competitive advantage to the diseased stem cell compartment – an advantage with which JAK2 V617F itself may conspire(22, 23), but where the mutation alone cannot confer such an advantage(24).
We used pSTAT5 as a surrogate for activation of the JAK2 pathway. One limitation of this strategy is that we cannot exclude the possibility that JAK2 was appropriately inhibited in MF samples while STAT5 remained activated in response to other signaling pathways. We were not able to directly detect JAK2/phospho-JAK2 in primary MPN samples. Future studies using other strategies will address whether novel heterodimers can be detected in primary cells, and if present, whether they correlate with persistent activation of STAT3 and STAT5. Our methods also could not specifically address microenvironmental factors known to play an important role in drug response and resistance, that might carry through in progeny of diseased stem/progenitor cells(25). Of particular interest is whether terminally differentiated cell signaling in a clonal population faithfully recapitulates that of more primitive ancestors within the stem/progenitor cell compartment.
Supplementary Material
Statement of translational relevance.
The identification of JAK2 mutations in classical, Philadelphia-chromosome negative myeloproliferative neoplasms (MPN) suggested a target for small molecule therapy for MPN, with the hope of responses akin to those seen in chronic myeloid leukemia treated with imatinib. While subsequent studies of JAK2 inhibitors have proven they are therapeutic, responses have been unexpected, and do not in fact meaningfully deplete the clonal burden of disease. The mechanism(s) through which JAK2 inhibitors exert activity are incompletely understood, including how effectively they inhibit JAK2 kinase activity in blood cells. We directly measured downstream inhibition of STAT3 and STAT5 phosphorylation in whole blood and found that across disease subtypes, the response to inhibition varied, with myelofibrosis overall resistant. Our findings identify a pattern of resistance that is not in the classical “escape mutation” paradigm, and may begin to explain the attenuated clinical responses that have been observed in the clinic.
Acknowledgments
Financial support: Elizabeth Hexner: National Institutes of Health (K23-HL-093366-01A1); American Society of Hematology Scholar (fellow) in Clinical Translational Research.
We thank Joy Cannon of the Stem Cell and Xenograft Core at the University of Pennsylvania for coordination of samples. We also thank Gerald Wertheim MD, PhD for help with qPCR calculations. This study was supported by grants from the National Institutes of Health (K23-HL-093366-01A1), and the American Society of Hematology Scholar (fellow) in Clinical Translational Research to E.O.H.
Footnotes
Author Contributions
AK designed and performed experiments, interpreted data, and wrote the manuscript. EOH designed experiments, interpreted data, and wrote the manuscript. GJ performed experiments, interpreted data, and contributed to writing of the manuscript. MC contributed to the interpretation and editing of the manuscript.
Conflict of Interest: M.C. has previously received research funding from Cephalon Oncology. E.H., A.K., G.J. have no financial conflict of interest.
References
- 1.Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S, et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet. 2005;365:1054–61. doi: 10.1016/S0140-6736(05)71142-9. [DOI] [PubMed] [Google Scholar]
- 2.James C, Ugo V, Le Couedic JP, Staerk J, Delhommeau F, Lacout C, et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature. 2005;434:1144–8. doi: 10.1038/nature03546. [DOI] [PubMed] [Google Scholar]
- 3.Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR, et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005;352:1779–90. doi: 10.1056/NEJMoa051113. [DOI] [PubMed] [Google Scholar]
- 4.Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJ, et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell. 2005;7:387–97. doi: 10.1016/j.ccr.2005.03.023. [DOI] [PubMed] [Google Scholar]
- 5.Pardanani A, Gotlib JR, Jamieson C, Cortes JE, Talpaz M, Stone RM, et al. Safety and Efficacy of TG101348, a Selective JAK2 Inhibitor, in Myelofibrosis. J Clin Oncol. doi: 10.1200/JCO.2010.32.8021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Verstovsek S, Kantarjian H, Mesa RA, Pardanani AD, Cortes-Franco J, Thomas DA, et al. Safety and efficacy of INCB018424, a JAK1 and JAK2 inhibitor, in myelofibrosis. N Engl J Med. 2010;363:1117–27. doi: 10.1056/NEJMoa1002028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Verstovsek S, Mesa RA, Gotlib J, Levy RS, Gupta V, DiPersio JF, et al. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. N Engl J Med. 2012;366:799–807. doi: 10.1056/NEJMoa1110557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pardanani A, Gotlib JR, Jamieson C, Cortes JE, Talpaz M, Stone RM, et al. Safety and efficacy of TG101348, a selective JAK2 inhibitor, in myelofibrosis. J Clin Oncol. 2011;29:789–96. doi: 10.1200/JCO.2010.32.8021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chow S, Minden MD, Hedley DW. Constitutive phosphorylation of the S6 ribosomal protein via mTOR and ERK signaling in the peripheral blasts of acute leukemia patients. Exp Hematol. 2006;34:1183–91. doi: 10.1016/j.exphem.2006.05.002. [DOI] [PubMed] [Google Scholar]
- 10.Hedley DW, Chow S, Goolsby C, Shankey TV. Pharmacodynamic monitoring of molecular-targeted agents in the peripheral blood of leukemia patients using flow cytometry. Toxicologic pathology. 2008;36:133–9. doi: 10.1177/0192623307310952. [DOI] [PubMed] [Google Scholar]
- 11.Nussenzveig RH, Swierczek SI, Jelinek J, Gaikwad A, Liu E, Verstovsek S, et al. Polycythemia vera is not initiated by JAK2V617F mutation. Exp Hematol. 2007;35:32–8. doi: 10.1016/j.exphem.2006.11.012. [DOI] [PubMed] [Google Scholar]
- 12.Germer S, Holland MJ, Higuchi R. High-throughput SNP allele-frequency determination in pooled DNA samples by kinetic PCR. Genome research. 2000;10:258–66. doi: 10.1101/gr.10.2.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kimura A, Rieger MA, Simone JM, Chen W, Wickre MC, Zhu BM, et al. The transcription factors STAT5A/B regulate GM-CSF-mediated granulopoiesis. Blood. 2009;114:4721–8. doi: 10.1182/blood-2009-04-216390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang H, Nguyen-Jackson H, Panopoulos AD, Li HS, Murray PJ, Watowich SS. STAT3 controls myeloid progenitor growth during emergency granulopoiesis. Blood. 2010;116:2462–71. doi: 10.1182/blood-2009-12-259630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Levis M, Ravandi F, Wang ES, Baer MR, Perl A, Coutre S, et al. Results from a randomized trial of salvage chemotherapy followed by lestaurtinib for patients with FLT3 mutant AML in first relapse. Blood. 2011;117:3294–301. doi: 10.1182/blood-2010-08-301796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tyner JW, Bumm TG, Deininger J, Wood L, Aichberger KJ, Loriaux MM, et al. CYT387, a novel JAK2 inhibitor, induces hematologic responses and normalizes inflammatory cytokines in murine myeloproliferative neoplasms. Blood. 2010;115:5232–40. doi: 10.1182/blood-2009-05-223727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gibbs KD, Jr, Gilbert PM, Sachs K, Zhao F, Blau HM, Weissman IL, et al. Single-cell phospho-specific flow cytometric analysis demonstrates biochemical and functional heterogeneity in human hematopoietic stem and progenitor compartments. Blood. 2011;117:4226–33. doi: 10.1182/blood-2010-07-298232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fleischman AG, Aichberger KJ, Luty SB, Bumm TG, Petersen CL, Doratotaj S, et al. TNFalpha facilitates clonal expansion of JAK2V617F positive cells in myeloproliferative neoplasms. Blood. 2011;118:6392–8. doi: 10.1182/blood-2011-04-348144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Moliterno AR, Williams DM, Rogers O, Isaacs MA, Spivak JL. Phenotypic variability within the JAK2 V617F-positive MPD: Roles of progenitor cell and neutrophil allele burdens. Exp Hematol. 2008 doi: 10.1016/j.exphem.2008.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shah NP, Nicoll JM, Nagar B, Gorre ME, Paquette RL, Kuriyan J, et al. Multiple BCR-ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell. 2002;2:117–25. doi: 10.1016/s1535-6108(02)00096-x. [DOI] [PubMed] [Google Scholar]
- 21.Koppikar P, Bhagwat N, Kilpivaara O, Manshouri T, Adli M, Hricik T, et al. Heterodimeric JAK-STAT activation as a mechanism of persistence to JAK2 inhibitor therapy. Nature. 2012;489:155–9. doi: 10.1038/nature11303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu F, Zhao X, Perna F, Wang L, Koppikar P, Abdel-Wahab O, et al. JAK2V617F-mediated phosphorylation of PRMT5 downregulates its methyltransferase activity and promotes myeloproliferation. Cancer Cell. 2011;19:283–94. doi: 10.1016/j.ccr.2010.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dawson MA, Bannister AJ, Gottgens B, Foster SD, Bartke T, Green AR, et al. JAK2 phosphorylates histone H3Y41 and excludes HP1alpha from chromatin. Nature. 2009;461:819–22. doi: 10.1038/nature08448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li J, Spensberger D, Ahn JS, Anand S, Beer PA, Ghevaert C, et al. JAK2 V617F impairs hematopoietic stem cell function in a conditional knock-in mouse model of JAK2 V617F-positive essential thrombocythemia. Blood. 2010;116:1528–38. doi: 10.1182/blood-2009-12-259747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Houthuijzen JM, Daenen LG, Roodhart JM, Voest EE. The role of mesenchymal stem cells in anti-cancer drug resistance and tumour progression. British journal of cancer. 2012;106:1901–6. doi: 10.1038/bjc.2012.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.