Abstract
Coccidioidomycosis (San Joaquin Valley fever) is a human respiratory disease caused by a soil-borne mold, and is recognized as an intransigent microbial infection by physicians who treat patients with the potentially life-threatening, disseminated form of this mycosis. Epidemiological studies based on surveys of skin-test reactivity of people who reside in the endemic regions of the Southwestern US have shown that at least 150,000 new infections occur annually. The clinical spectrum of coccidioidomycosis ranges from an asymptomatic insult to a severe pulmonary disease in which the pathogen may spread from the lungs to the skin, bones, brain and other body organs. Escalation of symptomatic infections and increased cost of long-term antifungal treatment warrant a concerted effort to develop a vaccine against coccidioidomycosis. This review examines recently reported strategies used to generate such a vaccine and summarizes current understanding of the nature of protective immunity to this formidable disease.
Keywords: Coccidioides, Coccidioidomycosis, Killed and live vaccines, Innate and adaptive immunity, CD4+ and CD8+ T cells, Epitope-based vaccine construct, Adjuvants
Introduction
Coccidioides immitis and C. posadasii are pathogenic molds which grow as saprobes in arid, alkaline soil of semidesert regions of the Southwestern US, Mexico, and parts of Central and South America. Inhalation of airborne infectious spores produced during the saprobic phase can result in the onset of a human respiratory disease known as coccidioidomycosis or San Joaquin Valley fever. Approximately 60% of these human exposures are asymptomatic infections, while 40% result in symptoms that range from a self-limited pulmonary, influenza-like illness to a relatively rare life-threatening, disseminated disease [1]. Coccidioides respiratory infections are a common cause of community-acquired pneumonia in both healthy individuals and immunocompromised patients who reside in endemic areas of the Southwestern US [2]. No person-to-person transmission of the pathogen occurs, except in cases of recipient-acquired infection from an organ donor during solid-organ transplantation [3•, 4]. Physicians are encouraged to employ appropriate diagnostic evaluation and prompt initiation of anti-Coccidioides therapy if donor-derived coccidioidal infection is a consideration [5]. Skin tests conducted in the 1950s showed that 50–70% of the population in the San Joaquin Valley of Southern California reacted to Coccidioides antigen, indicating prior exposure to the fungal pathogen or active disease [6]. There is a racial difference in the frequency of symptomatic primary infection. Dark-skinned races are more prone to severe primary illness and dissemination than Caucasians [7]. Clinical investigations have revealed that reactivation of coccidioidomycosis in people who initially have an asymptomatic coccidioidal infection can occur months to years after exposure to the pathogen during a visit to an endemic site [8•]. Major outbreaks of coccidioidomycosis continue to occur, particularly in California and Arizona [9, 10]. For example, a huge dust storm that swept through Phoenix on 5 July 2011 resulted in a significant rise in the number of symptomatic infections during the following 4-month period (http://www.vfce.arizona.edu/resources/pdf/SunenshineMD-AM_CME_Cocci_Epi_11_6_11.pdf). In fact, regions in the vicinity of the rapidly growing cities of Tucson and Phoenix account for about 60% of all cases of Coccidioides infections currently recorded in the US [11]. This is partly due to a decision made in 1997 to mandate the reporting of Valley Fever in Arizona [12]. Additional factors which contribute to the continued rise in the number of cases of this respiratory mycosis include an increase in frequency of persons with immunosuppressive conditions who reside in endemic regions, migration of immunologically naive individuals into hyperendemic areas, and aging of the population [13, 14]. San Joaquin Valley fever has long been a concern of the US military because many bases in the Southwest are located in regions known to harbor the soil-borne pathogen. Between 1942 and 1945 the military reported that approximately 4,000 men were infected with Coccidioides, resulting in 39 deaths [15]. Currently, more than 350,000 military service personnel are stationed in endemic regions.
A vaccine against Coccidioides infection would contribute significantly to the well-being of the approximately 30 million residents in the Southwestern US as well as the multitude of people who annually visit popular tourist sites in this region. The target population for this disease in endemic regions of Mexico and Latin America [16, 17, 18•] would also benefit from the availability of a human vaccine. A compelling argument for the feasibility of generating such a vaccine is based on the retrospective clinical observation that individuals who recover from a symptomatic coccidioidal infection remain skin-test positive, do not have recurrence of the disease and presumably have life-long acquired immunity to coccidioidomycosis [19, 20].
The Parasitic Cycle
Coccidioides undergoes a parasitic cycle which is unique amongst the medically important fungi [21]. The parasitic phase can be cultured in a defined glucose/salts medium [22]. Morphological features of the stages of development in vitro are identical to those observed in infected lungs of the mammalian host. Growth of the saprobic (mycelial) phase at 30°C on a solid agar medium results in production of a confluent layer of spores (each typically 2 × 4 μm). The latter are used to inoculate a defined glucose/salts liquid medium for initiation of the parasitic cycle. Cultures are grown in a shaking incubator at 39°C in the presence of 20% CO2. The cylindrical spores germinate by growing isotropically and transform into large, multinucleate round cells (spherules; 60–100 μm diameter), each of which initially contains a large central vacuole. The cytoplasm surrounding the vacuole becomes progressively compartmentalized as a result of multiple sites of ingrowth of the inner layer of the spherule wall. This process signals the differentiation of endospores that undergo rapid isotropic growth, expand into the space previously occupied by the central vacuole, and are ultimately released when the wall of the maternal spherule ruptures. A single spherule can produce hundreds of endospores which disseminate from original sites of pulmonary infection and give rise to the next generation of parasitic cells. The first generation of the parasitic cycle is completed in vitro after about 5 to 6 days of incubation.
Killed Vaccine Candidates
Early protection studies evaluated the use of a formalin-killed spherule (FKS) vaccine in murine models of coccidioidomycosis [23]. Mice were immunized by the intramuscular route with approximately 3.2 mg of a formalin-fixed mixture of immature and mature spherules and endospores. The vaccinated mice were shown to be fully protected against a potentially lethal respiratory challenge with the pathogen. However, in spite of its success as a protective reagent in four animal species, including primates, FKS failed as a candidate human vaccine in a phase 3 clinical trial [24]. The killed spherule vaccine did not demonstrate a significant reduction in the incidence or severity of coccidioidomycosis in humans. This may have been due, at least in part, to an intense inflammatory reaction at sites of vaccination which necessitated adoption of a suboptimal dose of FKS during the clinical trial.
Immunization of mice with heat-killed yeast (HKY) of Saccharomyces cerevisiae has been reported to protect against aspergillosis, coccidioidomycosis and candidiasis [25, 26, 27••]. Preliminary data have been reported indicating that this same vaccine also provides protection against cryptococcosis [28], which led the authors to speculate that the HKY of S. cerevisiae could serve as a panfungal vaccine. Subcutaneous immunization of CD-1 outbred mice with a total of 2.5 or 5.0 mg of the HKY vaccine delivered at 21, 14, and 7 days prior to intravenous challenge with 260 viable Coccidioides spores resulted in 50–70% survival to at least 28 days after challenge [27••]. Approximately 90% of the nonvaccinated control mice died by 16–18 days after intravenous challenge. The authors argued on the basis of previous murine studies [29] that the intravenous and natural intranasal routes of challenge would yield comparable survival curves. However, the degree of protection afforded by immunization with HKY was significantly less than that of the FKS vaccine, and inferior to that afforded by subcutaneous immunization with a soluble, aqueous, subcellular antigenic fraction derived from mechanically disrupted spherules [29]. The latter resulted in 100% survival of outbred mice over a period of more than 100 days after intranasal challenge [30]. Sterile immunity to coccidioidomycosis was not achieved with either the heat-killed or subfraction vaccine, and co-immunization of HKY with various adjuvants or interleukin 12 (IL-12) did not enhance protection [27••]. It was suggested that immunogenic cell wall glycans shared between Saccharomyces yeast and Coccidioides parasitic cells are major contributors to the protection afforded by the HKY vaccine, while antigenic protein and lipid homologs could also act together in the induction of the protective host response [28].
C-type lectin receptors on the surface of dendritic and other antigen-presenting cells (APCs) of the mammalian host are known to play a key role as pattern recognition receptors (PRRs) through their detection of non-self glycans on microbial cells and have proved to be potent inducers and modulators of innate immunity to microbial infection [31, 32, 33••]. The ability of these receptors to bind fungal cell wall glycans, particularly β-glucan, correlates with initiation of a signaling cascade that culminates in the release of inflammatory cytokines and activation of protective cellular and humoral immunity [34, 35, 36•]. The C-type lectin receptors that have been shown to play key roles in the induction and modulation of cytokine production include Dectin-1, Dectin-2, mannose receptor, Mincle, DC-SIGN, and collectins. The last of these are secreted C-type lectins which bind directly to cell surface receptors such as CD14, TLR2, and TLR4 to modulate cytokine responses [36•]. A surprising observation of HKY-vaccinated and challenged mice was that in vitro recall stimulation of immune splenic lymphocytes isolated from these animals results in significant proliferation of CD3+ and CD8+ T cells but not CD4+ T cells [37]. Whether CD4+ T cells contribute significantly to the protective response in the HKY-vaccinated mouse model or are redundant is not clear. The killed yeast vaccine was also shown to induce production of high titers of antibody against Saccharomyces glucan and mannan [37]. Although a large body of evidence supports the role of T cells in adaptive immunity to Coccidioides infection [38••, 39], the mechanisms by which antibodies influence host protection against this respiratory mycosis are still not defined [40].
A Genetically Engineered Live Vaccine
For numerous bacterial and viral diseases in which natural infection confers protective immunity against reinfection, live attenuated vaccines have proved to be most successful in the generation of robust and durable immune defenses in the human host. Live vaccines typically elicit a broad spectrum of antigen-specific antibody responses by plasma cells as well as development of long-term T and B cell memory, thereby inducing optimal immunity by activation of many effectors that target a multitude of antigenic molecules of the pathogen [41]. The essential factor shared by live vaccine strains which have been promoted to clinical trial is that their disease-causing capacity is eliminated by genetic or technical manipulations, thus prohibiting reversion to the virulent state. A genetically defined, live attenuated strain of C. posadasii (ΔT) was generated which lost its ability to endosporulate in vivo but is able to elicit protective immunity to coccidioidomycosis (100% survival) in disease-susceptible mice [42]. Loss of reproductive capacity of the Coccidioides mutant was attributed to targeted disruption of two chitinase genes (CTS2 and CTS3). The combined expression of these genes in the parental strain was shown to be essential for endospore differentiation and reproduction of the pathogen both in vitro and in infected lung tissue. In contrast to FKS, this live vaccine shows only minimal, transient reactogenicity at sites of immunization in the mouse model. The vaccine strain persists as sterile parasitic cells and remains localized at sites of immunization for more than 6 weeks after completion of the vaccination protocol. Immunological studies of C57BL/6 and BALB/c mice immunized with the live ΔT vaccine against a potentially lethal Coccidioides lung infection have yielded new insights into the nature of vaccine immunity to coccidioidmycosis [42, 43•, 44•, 45].
Early Innate Immune Response to Coccidioides Infection
Host response to the initial phase of coccidioidal pneumonia is signaled by targeted lung infiltration of macrophages and polymorphonuclear leukocytes (PMNs) to sites of colonization by the parasitic cells. This is a pathological hallmark of bacterial and fungal infections of the respiratory system and is pivotal for host defense [46]. However, a peculiarity of coccidioidomycosis is that this pronounced phagocytic response to infection in mice is delayed after intranasal challenge with Coccidioides spores. Only when the spherules have ruptured and released their endospores do the phagocytic cell populations expand and migrate in a chemotaxis-like manner to the endosporulating parasitic cells (Fig. 1). The chemical signals produced by the host and/or pathogen which are responsible for the recruitment of innate cells to infection sites are unknown. Histopathological observations indicate that prior to the release of endospores little infiltration of inflammatory cells into the infected lungs occurs [43•] and PMNs fail to attack the spherules [47]. In vitro studies have also revealed that phagocytosis and fungicidal activity of PMNs declines during Coccidioides maturation from spores to mature parasitic cells, and then returns when endospores are released from the ruptured parasitic cells [47]. This apparent anergy during early stages of parasitic cell differentiation is still unexplained. The early recruitment of PMNs to the lungs of nonvaccinated mice occurs in the presence of sharply elevated concentrations of proinflammatory cytokines IL-1β, IL-1α, and TNF-α between 7 and 11 days after challenge [43•]. A prolonged, inflammatory response, which intensifies in nonvaccinated animals as the pathogen proliferates, contributes to tissue damage and exacerbates the course of disease. On the other hand, lung homogenates obtained from mice immunized with the live (ΔT) vaccine show a sharp decrease in concentrations of these and other proinflammatory cytokines by 11 days after challenge, which correlates with a reduction in neutrophil granulocyte number and a significant increase in lung infiltration of alveolar and tissue macrophages. The mechanism by which this downregulation occurs is still unknown. Eosinophils are recruited in progressively higher numbers to the lungs of nonvaccinated mice beginning at 11 days after challenge, but not in vaccinated mice. The presence of pulmonary eosinophilic microabscesses in Coccidioides-infected patients has been correlated with disseminated disease and a poor prognosis [48].
Fig. 1.
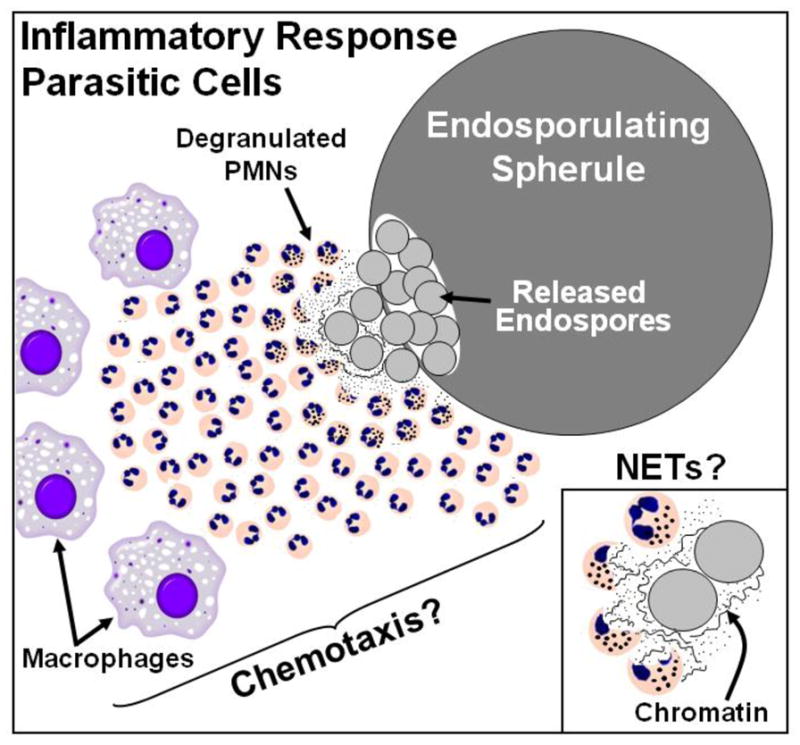
Polymorphonuclear leukocytes (PMNs) and macrophages in infected lung tissue target mature parasitic cells (spherules) of Coccidioides which have completed their reproductive phase (endosporulation), ruptured, and released their contents. Recruitment of innate immune cells to these sites of endospore release appears to occur in a chemotaxis-like manner. Formation of neutrophil extracellular traps (NETs) is associated with intense inflammatory response of PMNs to sites of microbial infection and results in degranulation, release of decondensed genomic DNA and ultimately to host cell death, a process referred to as NETosis. NETs have the capacity to trap and damage microbial pathogens and thereby contribute to host defense. NETS may also play a role in immunomodulation by activation or dampening of the inflammatory response. Appropriate conditions appear to exist for NET formation during PMN response to endosporulating spherules of Coccidioides
During the first 2 weeks after challenge, ΔT-vaccinated mice consistently show a 4 log10 or greater decrease in colony-forming units in lung homogenates compared to nonvaccinated mice. The latter are typically moribund at this stage of the disease [43•]. Nitric oxide (NO) produced by enzymatic activity of the inducible isoform of nitric oxide synthase and released from phagocytic cells has previously been thought to be an important first line of host defense against coccidioidal infection. However, Coccidioides as well as several other fungal pathogens that cause systemic mycoses of humans are capable of direct or indirect suppression of phagocyte secretion of NO [49–55]. Production of reactive oxygen species resulting from phagocytic NADPH oxidase (NOX2) activity represents another line of chemical defense of the mammalian host. Two independent studies have revealed that activated macrophages of Coccidioides isolated from either NOX2−/− knockout or wild-type mice show comparable killing efficiency [56, 57]. It is evident, at least based on these results of in vitro studies, that factors other than direct exposure of the fungal pathogen to reactive oxygen intermediates are responsible for the ability of phagocytes to kill the organism. Degranulation of PMNs results in secretion of an assortment of potentially fungicidal proteins. Myeloperoxidase released from azurophilic granules as well as NADPH oxidase secretion have been shown to contribute to the formation of neutrophil extracellular traps (NETs) [58, 59•], which consist largely of decondensed chromatin and antimicrobial proteins of dead PMNs at infection sites. NETs are induced by inflammation, lead to localized cell death (NETosis), attract phagocytes and stimulate them to trap and inhibit growth of a wide range of infectious microbes including fungi [60, 61], and may activate or dampen the inflammatory response by acting on innate PRRs [62]. NETs have been suggested to be particularly important in host defense against certain fungal pathogens, such as Coccidioides, which are difficult to phagocytose because of their large size (Fig. 1) [58]. This is an unexplored area of research of innate immune response to Coccidioides infection.
The Protective T-cell Repertoire
Intranasally challenged C57BL/6 mice immunized with the same live (ΔT) vaccine as described above have been shown to mount a robust CD4+ T cell-mediated immune response to the respiratory infection, develop well-differentiated pulmonary granulomas in the infected lungs, reveal little to no evidence of inflammatory damage, and exhibit near clearance of the organism from lung tissue with minimal dissemination of the pathogen to extrapulmonary sites [42, 43•]. Vaccinated mice have also been shown to induce protective antifungal CD8+ T cell immunity in the absence of CD4+ T cells [63, 64], which raises the possibility that immunocompromised hosts lacking CD4+ T cells may still be protected against coccidioidomycosis by recruitment of the antifungal memory CD8+ T cell arm of acquired immunity [65••]. Results of numerous studies suggest that the T helper 1 (Th1) subpopulation of CD4+ T cells plays a pivotal role in protection against coccidioidomycosis [43•, 66]. Production of IFN-γ by Th1 cells activates phagocytes to kill the fungal pathogen and contributes to the development of granulomas at sites of tissue insult [67], while loss of IL-12 or IFN-γ secretion increases the severity of fungal infections [68, 69].
Activated Th17 cells have recently been recognized as essential constituents of the protective CD4+ T-cell repertoire [38••, 39, 40, 43•]. In fact, it is now evident that a vaccine against Coccidioides infection must induce a major response of Th17 cells during the early course of infection if it is to be effective [33••, 38••]. Several lines of evidence indicate that activation of the Th17 signal pathway plays a key role in vaccine immunity to coccidioidomycosis. For example, loss of function of IL-17 receptor A in IL-17 ra−/− mice immunized with the protective ΔT vaccine results in 60% reduction of their survival over 45 days after challenge compared to 100% survival of the vaccinated wild-type C57BL/6 mice [43•]. Activation of alveolar macrophages and PMNs by recombinant IL-17 in vitro has been shown to augment fungicidal capacity of the phagocytes, suggesting that antifungal Th17 cells contribute to protection by recruitment and activation of innate cells [38••]. IL-17 also influences PMN expansion and survival, and modulates neutrophil infiltration to infection sites via G-CSF and CXCL1/KC stimulation together with other members of the family of CXC-type chemokines [70]. A key modulating factor in Th1 and Th17 promotion of the inflammatory response to Coccidioides infection is the production of cytokines of the nine-member IL-10 family, which includes IL-10, IL-19, IL-20, IL-22. IL24, IL-26 and distantly related IL-28A, IL-28B and IL-29 [71•]. These chemical signals mainly participate in repression of excessive inflammatory response as well as remodeling and wound-healing activities. The dominant IL-10 cytokine appears to be primarily produced by CD4+ T cells in C57BL/6 mice immunized with the ΔT vaccine followed by intranasal challenge, and plays important roles in both prevention of inflammatory pathology and the differentiation of Th1 and Th17 effector memory cells [45]. The latter are essential for durable protection against coccidioidomycosis. Determination of the signal pathways responsible for these regulatory processes is a major focus of our current investigations of vaccine immunity to Coccidioides lung infection.
A T-cell Epitope-based Recombinant Protein Vaccine
Although the genetically engineered, sterile mutant strain (ΔT) of Coccidioides showed promise in mice as a live vaccine, its application to humans has anticipated problems. In spite of the apparent ability of a live vaccine to elicit and maintain long-term CD8+ T cell memory in the absence of CD4+ T cell help, individuals with underlying conditions that compromise their cell-mediated immune system could show severe reactogenicity to immunization with the vaccine. In addition, the unprecedented concept of immunization with a live fungal vaccine would be expected to raise serious skepticism amongst recruits in a clinical trial. Nevertheless, the knowledge gained from the live vaccine model is crucial for the rational design of human vaccine candidates [33••]. Generation of a T-cell epitope-based recombinant protein vaccine was initiated as a strategy for the design of a clinically acceptable reagent. Three assumptions have emerged from evaluations of recombinant protein vaccines against coccidioidomycosis: CD4+ T-cell immunity is essential for protection, activation of Th1, Th2 and Th17 signal pathways contribute to early protective response to infection, and APC processing and presentation of immunodominant T-cell epitopes representing different stages of the parasitic cycle are necessary for induction of optimal vaccine immunity to coccidioidomycosis [72–75].
With these suppositions in mind, a T-cell epitope-based, recombinant protein vaccine was constructed and subsequently shown to protect mice against intranasal challenge with Coccidioides spores [73, 76•]. The T-cell epitope peptides incorporated into the vaccine were derived from three T-cell-reactive proteins expressed during the parasitic cycle of C. posadasii. These full-length recombinant proteins (Pep1, Amn1, and Plb) [75] were selected from studies which showed that when they were combined and used to immunize C57BL/6 mice they induced a robust protective immune response to coccidioidal infection [74, 75]. T-cell epitopes of these three proteins were first identified by computational prediction of their ability to bind promiscuously to human MHC class II molecules. An algorithm was employed (ProPred) which contains matrix-based motifs of 51 human leukocyte antigen (HLA) subregion DR alleles derived from a MHC class II pocket profile database [77]. Promiscuous epitopes were defined as peptides that were predicted to bind to at least 80% of the MHC class II molecules expressed by the 51 HLA-DR alleles.
T-cell-reactivity of synthetic peptides (20–32 mer) spanning all predicted promiscuous epitopes of each of the three proteins was evaluated by interferon-gamma (IFN-γ) enzyme-linked immunospot (ELISPOT) assays. Immune lymphocyte response was evaluated with CD4+ T cells obtained from human MHC class II-expressing transgenic mice. Each epitope peptide was also tested for its ability to bind to human MHC II molecules by an in vitro competitive inhibition assay [78]. On the basis of results of these immunoassays a DNA construct was synthesized for bacterial expression of a single recombinant epitope-based protein vaccine (rEBV) which contained five epitopes with highest immune T cell reactivity and affinity for human MHC II [76•]. Each epitope in the construct was fused with leader and spacer peptides to optimize epitope processing and presentation to T cell receptors [79, 80]. Recall assays of immune T lymphocytes confirmed that the epitopes were processed. Transgenic mice immunized with the epitope-based vaccine admixed with a synthetic oligodeoxynucleotide adjuvant (CpG ODN) and challenged by the intranasal route with Coccidioides spores showed early lung infiltration of activated Th1, Th2 and Th17 cells, elevated IFN-γ and IL-17A production, and significant reduction of the fungal burden compared to nonvaccinated mice [73, 76•]. CD4+ T cell epitope-driven vaccine constructs against microbial diseases have been revealed to be immunologically potent, relatively easy to produce, and predicted to be safe for human use [81].
Experimental Adjuvants
The synthetic oligodeoxynucleotide adjuvant employed in evaluation of the T-cell epitope-based vaccine [82–84] has been suggested to function by direct activation of dendritic cells and macrophages and stimulation of IL-12, IFN-γ, and TNF-α production resulting in creation of a Th1-like milieu in lymphoid tissue [85]. On the other hand, CpG ODN has proved to be a suboptimal adjuvant for promotion of the Th17 signal pathway [76•]. Two novel adjuvant/delivery systems are currently under investigation in combination with candidate vaccines against coccidioidomycosis and both have improved the outcome of the protective host response to infection. EP67 is a peptide agonist of the biologically active C-terminal region of human complement component C5a [44]. Its N-terminus has been chemically modified to enable conjugation of the adjuvant to lysine residues of candidate protein vaccines. Binding of EP67 to the complement receptor of APCs is proposed to enable efficient receptor-mediated endocytosis and processing of the conjugate vaccines [86–89]. The EP67 decapeptide (YSFKDMP[Mel]aR) is the product of specific residue substitutions of the C5a65–74 fragment. The purpose of the substitutions is to restrict backbone flexibility in order to bias certain topographical features that allow a conformational distinction between beneficial C5a-like immune stimulatory responses versus undesirable inflammatory activity [90]. The unique conformational features of the adjuvant are accommodated by the C5a receptor (C5aR/CD88) expressed on APCs, but not by C5a receptors on neutrophils [91, 92]. In support of this concept, BALB/c mice immunized with an EP67-conjugate vaccine against coccidioidomycosis reveal significantly dampened neutrophil response to lung infection and associated reduction of inflammatory pathology at 11 days after challenge, but polarized activation of Th1 and Th17 effector cells during early stages of the disease compared to mice immunized with a nonconjugated vaccine [44•].
Beta-glucan particles (GPs) engender both an adjuvant effect as a result of their uptake by dendritic cells mediated by the Dectin-1 receptor, and an efficient delivery system for GP-incorporated vaccine candidates [93, 94•]. GPs are purified S. cerevisiae cell walls treated so that they are primarily β-1,3-D-glucans free of mannans and proteins. The hollow, porous nature of the GP structure allows loading high concentrations of antigen. The GP-based vaccine system appears to combine adjuvanicity with efficient antigen delivery to induce strong antibody-mediated and Th1/Th17-biased CD4+ T-cell immunity. Preliminary studies have demonstrated that when GPs are used for loading and delivery of the rEBV protein described above a significant improvement is observed in the protective immune response of transgenic mice to Coccidioides infection [76•, 95].
Conclusions
Substantial progress has been made toward the generation of a human vaccine against San Joaquin Valley fever since the last major reviews of this area of research were published in 2004 [72, 96]. Evaluation of both heat-killed and live vaccines in murine models of coccidioidomycosis have contributed to our current understanding of the required elements of the innate and adaptive immune repertoires for protection against this respiratory disease. A critical balance must be maintained between early host granulocyte infiltration and inflammatory cytokine production on the one hand, and prevention of excessive host tissue damage due to inflammatory pathology on the other. CD4+ and CD8+ T-cell immunity contribute synergistically to the protective response to fungal infection. CD8+ T cells are associated with long-term protective antifungal memory, which can be maintained even in the absence of CD4+ T cell help or persistent antigen [65••]. Recent evidence indicates that DCs are particularly adept at activating CD4+ or CD8+ T cells in response to antifungal vaccines through different antigen presentation pathways [97, 98]. Experiments in mice immunized with a live vaccine have revealed that activation of Th1 and Th17 cells and their production of inflammatory cytokines (e.g., IFN-γ, TNF, IL-17, and IL-22) are essential for protection [33••]. In spite of the clinical evidence that a rise in titer of Coccidioides-specific antibody in infected patients signals a poor prognosis, activation of the Th2 signal pathway is nevertheless a consistent feature of vaccine immunity to coccidioidomycosis in mice [42, 43•]. If protective antibodies against Coccidioides exist, however, they have not yet been identified.
At least in animal models of other mycoses antibodies have been shown to contribute to Th17 differentiation, collaborate with phagocytic cells and T lymphocytes to kill the fungal pathogen, and modulate inflammation to prevent host tissue damage [99, 100, 101••]. The roles of antibodies, Th17 cells and their interaction with other CD4+ T cell subsets in fungal vaccines are currently subjects of intense research. The introduction of live vaccines against coccidioidomycosis has revealed promising results in animal models but has the drawback of limited use in immunosuppressed or otherwise debilitated subjects since even attenuated variants can sometimes cause disease in individuals with impaired immunity [102•]. Construction and evaluation of a recombinant T-cell epitope-driven vaccine is an attempt to generate a defined, single protein construct which provides adequate protection in a humanized mouse model, achieves the necessary standards for large-scale production, and can be subjected to clinical studies at reasonable cost [76•]. To a significant degree, its success as a vaccine depends on the choice of the adjuvant which will be employed in clinical evaluations. A few adjuvants have been licensed for use in human vaccines (e.g., Alum, MF59, AS03, Virosomes, AS04) but numerous products are in the research pipeline, including EP67 and glucan/chitin particles [103]. Adjuvants are recognized to play a pivotal role in vaccine immunity by more than just enhancement of the magnitude of the adaptive immune response; they also contribute to the specificity and clonotypic diversity of the responding CD4+ T-cell repertoire [44•, 104]. In many cases vaccines which are potentiated by adjuvants are specifically designed to stimulate the aging immune system to respond better to vaccination [103]. This is a particularly relevant area of Coccidioides research since the elderly (≥65 years of age) are the cohort with the highest incidence of symptomatic coccidioidal infection, at least in endemic regions of the US [16].
Acknowledgments
Support for preparation of this review was provided by Public Health Service grants AI071118 awarded to GTC and an Institutional Research Training Fellowship awarded to BJH (AI007271) from the National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health. Additional support was provided by the Margaret Batts Tobin Foundation of San Antonio.
Footnotes
Disclosure
No potential conflicts of interest relevant to this article were reported.
References
Papers recently published and of particular interest have been highlighted as:
• Of importance
•• Of major importance
- 1.Centers for Disease Control and Prevention (CDC) Increase in coccidioidomycosis - California, 2000–2007. MMWR Morb Mortal Wkly Rep. 2009;58:105–109. [PubMed] [Google Scholar]
- 2.Hage CA, Knox KS, Wheat LJ. Endemic mycoses: overlooked causes of community acquired pneumonia. Resp Med. 2012;106:769–776. doi: 10.1016/j.rmed.2012.02.004. [DOI] [PubMed] [Google Scholar]
- 3•.Blodget E, Jan Geiseler P, Larsen RA, Stapfer M, Qazi Y, Petrovic LM. Donor-derived Coccidioides immitis fungemia in solid organ transplant recipients. Transpl Infect Dis. 2012;14:305–310. doi: 10.1111/j.1399-3062.2011.00698.x. This paper reports the risk of coccidioidomycosis in SOT among patients who receive an organ from a donor in the endemic regions and illustrates the need for diagnostic evaluation. [DOI] [PubMed] [Google Scholar]
- 4.Dierberg KL, Marr KA, Subramanian A, Nace H, Desai N, Locke JE, et al. Donor-derived organ transplant transmission of coccidioidomycosis. Transpl Infect Dis. 2012;14:300–304. doi: 10.1111/j.1399-3062.2011.00696.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Singh N, Huprikar S, Burdette SD, Morris MI, Blair JE, Wheat LJ, et al. Donor-derived fungal infections in organ transplant recipients: guidelines of the American Society of Transplantation, infectious diseases community of practice. Am J Transplant. 2012;12:2414–2428. doi: 10.1111/j.1600-6143.2012.04100.x. [DOI] [PubMed] [Google Scholar]
- 6.Kwon-Chung KJ, Bennett JE. Medical mycology. Malvern, Pa: Lea & Febiger; 1992. pp. 356–396. [Google Scholar]
- 7.Pappagianis D. Epidemiology of coccidioidomycosis. Curr Topics Med Mycol. 1988;2:199–238. doi: 10.1007/978-1-4612-3730-3_6. [DOI] [PubMed] [Google Scholar]
- 8•.Gobbi F, Angheben A, Farina C, Buonfrate D, Postiglione C, Rossanese A, et al. Coccidioidomycosis: first imported case in Italy. J Travel Med. 2012;19:192–194. doi: 10.1111/j.1708-8305.2012.00599.x. This paper illustrates the need for physicians to consider coccidioidomycosis in their differential diagnosis of febrile patients who have resided even for a short period in a Coccidioides endemic region, especially if they present with hypereosinophilia. [DOI] [PubMed] [Google Scholar]
- 9.Sunenshine RH, Anderson S, Erhart L, Vossbrink A, Kelly PC, Engelthaler D, et al. Public health surveillance for coccidioidomycosis in Arizona. Ann N Y Acad Sci. 2007;1111:96–102. doi: 10.1196/annals.1406.045. [DOI] [PubMed] [Google Scholar]
- 10.Kim MM, Blair JE, Carey EJ, Wu Q, Smilack JD. Coccidioidal pneumonia, Phoenix, Arizona, USA, 2000–2004. Emerg Infect Dis. 2009;15:397–401. doi: 10.3201/eid1503.081007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ampel NM. New perspectives on coccidioidomycosis. Proc Am Thorac Soc. 2010;7:181–185. doi: 10.1513/pats.200907-080AL. [DOI] [PubMed] [Google Scholar]
- 12.Laniado-Laborin R. Expanding understanding of epidemiology of coccidioidomycosis in the Western Hemisphere. Ann N Y Acad Sci. 2007;1111:19–34. doi: 10.1196/annals.1406.004. [DOI] [PubMed] [Google Scholar]
- 13.Fraser D, Ward JI, Ajello L, Plikaytis BD. Aspergillosis and other systemic mycoses: the growing problem. JAMA. 1979;242:1631–1635. [PubMed] [Google Scholar]
- 14.Blair JE, Mayer AP, Currier J, Files JA, Wu Q. Coccidioidomycosis in elderly persons. Clin Infect Dis. 2008;47:1513–1518. doi: 10.1086/593192. [DOI] [PubMed] [Google Scholar]
- 15.Crum-Cianflone NF. Coccidioidomycosis in the U.S. military. Ann N Y Acad Sci. 2007;1111:112–121. doi: 10.1196/annals.1406.001. [DOI] [PubMed] [Google Scholar]
- 16.Baddley JW, Winthrop KL, Patkar NM, Delzell E, Beukelman T, Xie F, et al. Geographic distribution of endemic fungal infections among older persons, United States. Emerg Infect Dis. 2011;17:1664–1669. doi: 10.3201/eid1709.101987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baptista Rosas RC, Riquelme M. The epidemiology of coccidioidomycosis in Mexico. Rev Iberoam Micol. 2007;24:100–105. doi: 10.1016/s1130-1406(07)70022-0. [DOI] [PubMed] [Google Scholar]
- 18•.Sifuentes-Osornio J, Corzo-Leon DE, Ponce-de-Leon LA. Epidemiology of invasive fungal infections in Latin America. Curr Fungal Infect Rep. 2012;6:23–34. doi: 10.1007/s12281-011-0081-7. This paper provides an update on the geographic location of endemic regions of Coccidioides as well as other dimorphic fungal pathogens in Central and South America. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pappagianis D. Coccidioides immitis antigen. J Infect Dis. 1999;180:243–244. doi: 10.1086/314835. [DOI] [PubMed] [Google Scholar]
- 20.Spinello IM, Munoz A, Johnson RH. Pulmonary coccidioidomycosis. Semin Respir Crit Care Med. 2008;29:166–173. doi: 10.1055/s-2008-1063855. [DOI] [PubMed] [Google Scholar]
- 21.Cole GT, Hung C-Y, Delgado N. Parasitic phase-specific gene expression in Coccidioides. ASM News. 2002;68:603–611. [Google Scholar]
- 22.Converse JL. Effect of physico-chemical environment of spherulation of Coccidioides immitis in a chemically defined medium. J Bacteriol. 1956;72:784–792. doi: 10.1128/jb.72.6.784-792.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Levine HB, Pappagianis D, Cobb JM. Development of vaccines for coccidioidomycosis. Mycopath Mycol Appl. 1970;41:177–185. doi: 10.1007/BF02051493. [DOI] [PubMed] [Google Scholar]
- 24.Pappagianis D. Evaluation of the protective efficacy of the killed Coccidioides immitis spherule vaccine in humans. The Valley Fever Vaccine Study Group. Am Rev Respir Dis. 1993;148:656–660. doi: 10.1164/ajrccm/148.3.656. [DOI] [PubMed] [Google Scholar]
- 25.Liu M, Clemons KV, Johansen ME, Martinez M, Chen V, Stevens DA. Saccharomyces as a vaccine against systemic candidiasis. Immunol Invest. 2012 doi: 10.3109/08820139.2012.692418. [DOI] [PubMed] [Google Scholar]
- 26.Stevens DA, Clemons KV, Liu M. Developing a vaccine against aspergillosis. Med Mycol. 2011;49(Suppl 1):S170–S176. doi: 10.3109/13693786.2010.497775. [DOI] [PubMed] [Google Scholar]
- 27••.Capilla J, Clemons KV, Liu M, Levine HB, Stevens DA. Saccharomyces cerevisiae as a vaccine against coccidioidomycosis. Vaccine. 2009;27:3662–3668. doi: 10.1016/j.vaccine.2009.03.030. The authors present an argument for the protective efficacy of a heat-killed, heterologous yeast vaccine in outbred mice. [DOI] [PubMed] [Google Scholar]
- 28.Liu M, Capilla J, Johansen ME, Alvarado D, Martinez M, Chen V, et al. Saccharomyces as a vaccine against systemic aspergillosis: ‘the friend of man’ a friend again? J Med Microbiol. 2011;60:1423–1432. doi: 10.1099/jmm.0.033290-0. [DOI] [PubMed] [Google Scholar]
- 29.Zimmermann CR, Johnson SM, Martens GW, White AG, Zimmer BL, Pappagianis D. Protection against lethal murine coccidioidomycosis by a soluble vaccine from spherules. Infect Immun. 1998;66:2342–2345. doi: 10.1128/iai.66.5.2342-2345.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Johnson SM, Kerekes KM, Lunetta JM, Pappagianis D. Characteristics of the protective subcellular coccidioidal T27K vaccine. Ann N Y Acad Sci. 2007;1111:275–289. doi: 10.1196/annals.1406.016. [DOI] [PubMed] [Google Scholar]
- 31.van Kooyk Y. C-type lectins on dendritic cells: key modulators for the induction of immune responses. Biochem Soc Trans. 2008;36:1478–1481. doi: 10.1042/BST0361478. [DOI] [PubMed] [Google Scholar]
- 32.Rabinovich GA, van Kooyk Y, Cobb BA. Glycobiology of immune responses. Ann N Y Acad Sci. 2012;1253:1–15. doi: 10.1111/j.1749-6632.2012.06492.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33••.Leibundgut-Landmann S, Wüthrich M, Hohl TM. Immunity to fungi. Cur Opin Immunol. 2012;24:449–458. doi: 10.1016/j.coi.2012.04.007. This paper examines pathways of fungal innate immune activation and fungicidal mechanisms, signaling pathways that lead to induction of adaptive immunity, and strategies for development of fungal vaccines. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Torosantucci A, Bromuro C, Chiani P, De Bernardis F, Berti F, Galli C, et al. A novel glyco-conjugate vaccine against fungal pathogens. J Exp Med. 2005;202:597–606. doi: 10.1084/jem.20050749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pietrella D, Rachini A, Torosantucci A, Chiani P, Brown AJ, Bistoni F, et al. A beta-glucan-conjugate vaccine and anti-b-glucan antibodies are effective against murine vaginal candidiasis as assessed by a novel in vivo imaging technique. Vaccine. 2010;28:1717–1725. doi: 10.1016/j.vaccine.2009.12.021. [DOI] [PubMed] [Google Scholar]
- 36•.Vautier S, MacCallum DM, Brown GD. C-type lectin receptors and cytokines in fungal immunity. Cytokine. 2012;58:89–99. doi: 10.1016/j.cyto.2011.08.031. An excellent review of the C-type lectins, their influence on cytokine production and subsequent immune responses in antifungal immunity. [DOI] [PubMed] [Google Scholar]
- 37.Liu M, Clemons KV, Bigos M, Medovarska I, Brummer E, Stevens DA. Immune responses induced by heat killed Saccharomyces cerevisiae: a vaccine against fungal infection. Vaccine. 2011;29:1745–1753. doi: 10.1016/j.vaccine.2010.12.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38••.Wüthrich M, Gern B, Hung C-Y, Ersland K, Rocco N, Pick-Jacobs J, et al. Vaccine-induced protection against 3 systemic mycoses endemic to North America requires Th17 cells in mice. J Clin Invest. 2011;121:554–568. doi: 10.1172/JCI43984. The authors present a cogent argument for the critical need of activated Th17 cells for a protective immune response to Coccidioides and other dimorphic fungal infections of the respiratory system. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wüthrich M, Hung C-Y, Gern BH, Pick-Jacobs JC, Galles KJ, Filutowicz HI, et al. A TCR transgenic mouse reactive with multiple systemic dimorphic fungi. J Immunol. 2011;187:1421–1431. doi: 10.4049/jimmunol.1100921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wuthrich M, Deepe GS, Jr, Klein B. Adaptive immunity to fungi. Annu Rev Immunol. 2012;30:115–148. doi: 10.1146/annurev-immunol-020711-074958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kamei A, Coutinho-Sledge YS, Goldberg JB, Priebe GP, Pier GB. Mucosal vaccination with a multivalent, live-attenuated vaccine induces multi-factorial immunity against Pseudomonas aeruginosa acute lung infection. Infect Immun. 2011;79:1289–1299. doi: 10.1128/IAI.01139-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xue J, Chen X, Selby D, Hung C-Y, Yu J-J, Cole GT. A genetically engineered live attenuated vaccine of Coccidioides posadasii protects BALB/c mice against coccidioidomycosis. Infect Immun. 2009;77:3196–3208. doi: 10.1128/IAI.00459-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43•.Hung C-Y, Gonzalez A, Wüthrich M, Klein BS, Cole GT. Vaccine immunity to coccidioidomycosis occurs by early activation of three signal pathways of T helper cell response (Th1, Th2, and Th17) Infect Immun. 2011;79:4511–4522. doi: 10.1128/IAI.05726-11. This paper summarizes features of vaccine immunity to coccidioidomycosis in mice vaccinated with a live, genetically-engineered mutant strain of Coccidioides. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44•.Hung C-Y, Hurtgen BJ, Bellecourt M, Sanderson SD, Morgan EL, Cole GT. An agonist of human complement fragment C5a enhances vaccine immunity against Coccidioides infection. Vaccine. 2012;30:4681–4690. doi: 10.1016/j.vaccine.2012.04.084. The authors present evidence that the EP67 adjuvant enhances protection of a live Coccidioides vaccine by augmentation of T-cell immunity, especially through Th1- and Th17-mediated host responses to infection. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hung C-Y, Castro-Lopez N, Cole GT. Diverse pulmonary T cell subsets of vaccinated and non-vaccinated C57BL/6 mice produce immunomodulatory IL-10 during Coccidioides infection [abstract]. 112th General Meeting, American Society for Microbiology; San Francisco, CA. 16–19 June 2012. [Google Scholar]
- 46.Balamayooran G, Batra S, Fessler MB, Happel KI, Jeyaseelan S. Mechanisms of neutrophil accumulation in the lungs against bacteria. Am J Resp Cell Mol Biol. 2010;43:5–16. doi: 10.1165/rcmb.2009-0047TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Frey CL, Drutz DJ. Influence of fungal surface components on the interaction of Coccidioides immitis with polymorphonuclear neutrophils. J Infect Dis. 1986;153:933–943. doi: 10.1093/infdis/153.5.933. [DOI] [PubMed] [Google Scholar]
- 48.Echols RM, Palmer DL, Long GW. Tissue eosinophilia in human coccidioidomycosis. Rev Infect Dis. 1982;4:656–664. doi: 10.1093/clinids/4.3.656. [DOI] [PubMed] [Google Scholar]
- 49.Gonzalez A, Hung C-Y, Cole GT. Coccidioides releases a soluble factor that suppresses nitric oxide production by murine primary macrophages. Microb Pathogen. 2011;50:100–108. doi: 10.1016/j.micpath.2010.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gonzalez A, Hung C-Y, Cole GT. Nitric oxide synthase activity has limited influence on the control of Coccidioides infection in mice. Microb Pathogen. 2011;51:161–168. doi: 10.1016/j.micpath.2011.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kawakami K, Tohyama M, Qifeng X, Saito A. Expression of cytokines and inducible nitric oxide synthase mRNA in the lungs of mice infected with Cryptococcus neoformans: effects of interleukin-12. Infect Immun. 1997;65:1307–1312. doi: 10.1128/iai.65.4.1307-1312.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chinen T, Qureshi MH, Koguchi Y, Kawakami K. Candida albicans suppresses nitric oxide (NO) production by interferon-gamma (IFN-γ) and lipopolysaccharide (LPS)-stimulated murine peritoneal macrophages. Clin Exp Immunol. 1999;115:491–497. doi: 10.1046/j.1365-2249.1999.00822.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schroppel K, Kryk M, Herrmann M, Leberer E, Rollinghoff M, Bogdan C. Suppression of type 2 NO-synthase activity in macrophages by Candida albicans. Int J Med Microbiol. 2001;290:659–668. doi: 10.1016/S1438-4221(01)80003-5. [DOI] [PubMed] [Google Scholar]
- 54.Flavia-Popi A, Daniel Lopes J, Mariano M. GP43 from Paracoccidioides brasiliensis inhibits macrophage functions. An evasion mechanism of the fungus. Cell Immunol. 2002;218:87–94. doi: 10.1016/s0008-8749(02)00576-2. [DOI] [PubMed] [Google Scholar]
- 55.Xiao G, Miyazato A, Inden K, Nakamura K, Shiratori K, Nakagawa K, et al. Cryptococcus neoformans inhibits nitric oxide synthesis caused by CpG-oligodeoxynucleotide-stimulated macrophages in a fashion independent of capsular polysaccharides. Microbiol Immunol. 2008;52:171–179. doi: 10.1111/j.1348-0421.2008.00019.x. [DOI] [PubMed] [Google Scholar]
- 56.Gonzalez A, Hung C-Y, Cole GT. Absence of phagocyte NADPH oxidase 2 leads to severe inflammatory response in lungs of mice infected with Coccidioides. Microb Pathogen. 2011;51:432–441. doi: 10.1016/j.micpath.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Margolis DA, Viriyakosol S, Fierer J, Kirkland TN. The role of reactive oxygen intermediates in experimental coccidioidomycosis in mice. BMC Microbiol. 2011;11:71e. doi: 10.1186/1471-2180-11-71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Metzler KD, Fuchs TA, Nauseef WM, Reumaux D, Roesler J, Schulze I, et al. Myeloperoxidase is required for neutrophil extracellular trap formation: implications for innate immunity. Blood. 2011;117:953–959. doi: 10.1182/blood-2010-06-290171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59•.Kirchner T, Moller S, Klinger M, Solbach W, Laskay T, Behnen M. The impact of various reactive oxygen species on the formation of neutrophil extracellular traps. Mediat Inflamm. 2012;2012:849136. doi: 10.1155/2012/849136. The authors discuss the requirement of NADPH oxidase and myeloperoxidase but not superoxide dismutase activity for the release of NETs. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Urban CF, Reichard U, Brinkmann V, Zvchlinsky A. Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell Microbiol. 2006;8:668–676. doi: 10.1111/j.1462-5822.2005.00659.x. [DOI] [PubMed] [Google Scholar]
- 61.Bianchi M, Niemiec MJ, Siler U, Urban CF, Reichenbach J. Restoration of anti-Aspergillus defense by neutrophil extracellular traps in human chronic granulomatous disease after gene therapy is calprotectin-dependent. J Allergy Clin Immunol. 2011;127:1243–1252. doi: 10.1016/j.jaci.2011.01.021. [DOI] [PubMed] [Google Scholar]
- 62.Neeli I, Radic M. Knotting the NETs: analyzing histone modifications in neutrophil extracellular traps. Arthritis Res Ther. 2012;14:115. doi: 10.1186/ar3773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wüthrich M, Filutowicz HI, Warner T, Deepe GS, Klein BS. Vaccine immunity to pathogenic fungi overcomes the requirement for CD4 help in exogenous antigen presentation to CD8+ T cells. J Exp Med. 2003;197:1405–1416. doi: 10.1084/jem.20030109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fierer J, Waters C, Walls L. Both CD4+ and CD8+ T cells can mediate vaccine-induced protection against Coccidioides immitis infection in mice. J Infect Dis. 2006;193:1323–1331. doi: 10.1086/502972. [DOI] [PubMed] [Google Scholar]
- 65••.Nanjappa SG, Heninger E, Wüthrich M, Sullivan T, Klein B. Protective antifungal memory CD8+ T cells are maintained in the absence of CD4+ T cell help and cognate antigen in mice. J Clin Invest. 2012;122:987–999. doi: 10.1172/JCI58762. The authors demonstrate in a mouse model that immunization with a live vaccine against a dimorphic fungal pathogen results in production of antifungal CD8+ T cells which are maintained in the absence of CD4+ T cells without loss of function or number for at least 6 months and protect against infection. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Duplessis CA, Tilley D, Bavaro M, Hale B, Holland SM. Two cases illustrating successful adjunctive interferon-γ immunotherapy in refractory disseminated coccidioidomycosis. J Infect. 2011;63:223–228. doi: 10.1016/j.jinf.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nishikaku AS, Molina RFS, Albe BP, Cunha CDS, Scavone R, Pizzo CRP, et al. Immunolocalization of IFN-gamma in the lesions of resistant and susceptible mice to Paracoccidioides brasiliensis infection. FEMS Immunol Med Microbiol. 2011;63:281–288. doi: 10.1111/j.1574-695X.2011.00851.x. [DOI] [PubMed] [Google Scholar]
- 68.Allendörfer R, Brunner GD, Deepe GS. Complex requirements for nascent and memory immunity in pulmonary histoplasmosis. J Immunol. 1999;162:7389–7396. [PubMed] [Google Scholar]
- 69.Vinh DC, Schwartz B, Hsu AP, Miranda DJ, Valdez PA, Fink D, et al. Interleukin-12 receptor-β1 deficiency predisposing to disseminated coccidioidomycosis. Clin Infect Dis. 2011;52:99–102. doi: 10.1093/cid/ciq215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.MacCallum DM, Castillo L, Brown AJP, Gow NAR, Odds FC. Early-expressed chemokines predict kidney immunopathology in experimental disseminated Candida albicans infections. PLoS One. 2009;4:e6420. doi: 10.1371/journal.pone.0006420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71•.Ouyang W, Rutz S, Crellin NK, Valdez PA, Hymowitz SG. Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annu Rev Immunol. 2011;29:71–109. doi: 10.1146/annurev-immunol-031210-101312. The authors discuss the importance of the family of IL-10 cytokines in facilitating the tissue-healing process caused by infection and inflammation. [DOI] [PubMed] [Google Scholar]
- 72.Cole GT, Xue JM, Okeke CN, Tarcha EJ, Basrur V, Schaller RA, et al. A vaccine against coccidioidomycosis is justified and attainable. Med Mycol. 2004;42:189–216. doi: 10.1080/13693780410001687349. [DOI] [PubMed] [Google Scholar]
- 73.Hurtgen BJ, Hung C-Y, Cole GT. A human T-cell epitope-driven vaccine against San Joaquin Valley Fever [abstract]. 111th General Meeting, American Society for Microbiology; New Orleans, LA. 21–24 May 2011. [Google Scholar]
- 74.Tarcha EJ, Basrur V, Hung C-Y, Gardner MJ, Cole GT. A recombinant aspartyl protease of Coccidioides posadasii induces protection against pulmonary coccidioidomycosis in mice. Infect Immun. 2006;74:516–527. doi: 10.1128/IAI.74.1.516-527.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tarcha EJ, Basrur V, Hung C-Y, Gardner MJ, Cole GT. Multivalent recombinant protein vaccine against coccidioidomycosis. Infect Immun. 2006;74:5802–5813. doi: 10.1128/IAI.00961-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76•.Hurtgen BJ, Hung C-Y, Ostroff GR, Levitz SM, Cole GT. Construction and evaluation of a novel recombinant T cell epitope-based vaccine against coccidioidomycosis. Infect Immun. 2012 doi: 10.1128/IAI.00566-12. Forthcoming. The authors describe a protein vaccine consisting of computationally selected and validated T-cell epitopes fused with leader and spacer peptides proposed to optimize epitope processing and presentation to T cell receptors. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Singh H, Raghava GP. ProPred: prediction of HLA-DR binding sites. Bioinformatics. 2001;17:1236–1237. doi: 10.1093/bioinformatics/17.12.1236. [DOI] [PubMed] [Google Scholar]
- 78.Forsthuber TG, Shive CL, Wienhold W, De Graaf K, Spack EG, Sublett R, et al. T cell epitopes of human myelin oligodendrocyte glycoprotein identified in HLA-DR4 (DRB1*0401) transgenic mice are encephalitogenic and are presented by human B cells. J Immunol. 2001;167:7119–7125. doi: 10.4049/jimmunol.167.12.7119. [DOI] [PubMed] [Google Scholar]
- 79.Germain RN. Uncovering the role of invariant chain in controlling MHC class II antigen capture. J Immunol. 2011;187:1073–1075. doi: 10.4049/jimmunol.1101663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Livingston B, Crimi C, Newman M, Higashimoto Y, Appella E, Sidney J, et al. A rational strategy to design multiepitope immunogens based on multiple Th lymphocyte epitopes. J Immunol. 2002;168:5499–5506. doi: 10.4049/jimmunol.168.11.5499. [DOI] [PubMed] [Google Scholar]
- 81.Rosa DS, Ribeiro SP, Cunha-Neto E. CD4 T cell epitope discovery and rational vaccine design. Arch Immunol Ther Exp. 2010;58:121–130. doi: 10.1007/s00005-010-0067-0. [DOI] [PubMed] [Google Scholar]
- 82.Li K, Yu J-J, Hung C-Y, Lehmann PF, Cole GT. Recombinant urease and urease DNA of Coccidioides immitis elicit an immunoprotective response against coccidioidomycosis in mice. Infect Immun. 2001;69:2878–2887. doi: 10.1128/IAI.69.5.2878-2887.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Delgado N, Xue J, Yu J-J, Hung C-Y, Cole GT. A recombinant beta-1,3-glucanosyltransferase homolog of Coccidioides posadasii protects mice against coccidioidomycosis. Infect Immun. 2003;71:3010–3019. doi: 10.1128/IAI.71.6.3010-3019.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hanagata N. Structure-dependent immunostimulatory effect of CpG oligodeoxynucleotides and their delivery system. Int J Nanomed. 2012;7:2181–2195. doi: 10.2147/IJN.S30197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Krieg AM. CpG still rocks! Update on an accidental drug. Nucleic Acid Ther. 2012;22:77–89. doi: 10.1089/nat.2012.0340. [DOI] [PubMed] [Google Scholar]
- 86.Morgan EL, Thoman ML, Sanderson SD, Phillips JA. A novel adjuvant for vaccine development in the aged. Vaccine. 2010;28:8275–8279. doi: 10.1016/j.vaccine.2010.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hegde GV, Meyers-Clark E, Joshi SS, Sanderson SD. A conformationally-biased, response-selective agonist of C5a acts as a molecular adjuvant by modulating antigen processing and presentation activities of human dendritic cells. Int Immunopharmacol. 2008;8:819–827. doi: 10.1016/j.intimp.2008.01.031. [DOI] [PubMed] [Google Scholar]
- 88.Duryee MJ, Bevins RA, Reichel CM, Murray JE, Dong Y, Thiele GM, et al. Immune responses to methamphetamine by active immunization with peptide-based, molecular adjuvant-containing vaccines. Vaccine. 2009;27:2981–2988. doi: 10.1016/j.vaccine.2009.02.105. [DOI] [PubMed] [Google Scholar]
- 89.Sanderson SD, Cheruku SR, Padmanilayam MP, Vennerstrom JL, Thiele GM, Palmatier MI, et al. Immunization to nicotine with a peptide-based vaccine composed of a conformationally biased agonist of C5a as a molecular adjuvant. Int Immunopharmacol. 2003;3:137–146. doi: 10.1016/s1567-5769(02)00260-6. [DOI] [PubMed] [Google Scholar]
- 90.Morgan EL, Morgan BN, Stein EA, Vitrs EL, Thoman ML, Sanderson SD, et al. Enhancement of in vivo and in vitro immune functions by a conformationally biased, response-selective agonist of human C5a: implications for a novel adjuvant in vaccine design. Vaccine. 2009;28:463–469. doi: 10.1016/j.vaccine.2009.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Taylor SM, Sherman SA, Kirnarsky L, Sanderson SD. Development of response-selective agonists of human C5a anaphylatoxin: conformational, biological, and therapeutic considerations. Curr Med Chem. 2001;8:675–684. doi: 10.2174/0929867013373156. [DOI] [PubMed] [Google Scholar]
- 92.Vogen SM, Paczkowski NJ, Kirnarsky L, Short A, Whitmore JB, Sherman SA, et al. Differential activities of decapeptide agonists of human C5a: the conformational effects of backbone N-methylation. Int Immunopharmacol. 2001;1:2151–2162. doi: 10.1016/s1567-5769(01)00141-2. [DOI] [PubMed] [Google Scholar]
- 93.Huang H, Ostroff GR, Lee CK, Specht CA, Levitz SM. Robust stimulation of humoral and cellular immune responses following vaccination with antigen-loaded b-glucan particles. MBio. 2010;1:e00164–10. doi: 10.1128/mBio.00164-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94•.Huang H, Ostroff GR, Lee CK, Agarwal S, Ram S, Rice PA, et al. Relative contributions of Dectin-1 and complement to immune responses to particulate beta-glucans. J Immunol. 2012;189:312–317. doi: 10.4049/jimmunol.1200603. The authors suggest a dominant role for complement and a redundant role for Dectin-1 in both phagocytosis of GPs and CD4+ Th17-biased immune response after GP-based vaccination. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hurtgen BJ, Castro-Lopez N, Hung C-Y, Cole GT. Heterogeneity of immune response of vaccinated HLA-DR4 (DRB1*0401) transgenic mice to coccidioidomycosis [abstr]. 112th General Meeting, American Society for Microbiology; San Francisco, CA. 16–19 June 2012. [Google Scholar]
- 96.Cox RA, Magee DM. Coccidioidomycosis: host response and vaccine development. Clin Microbiol Rev. 2004;17:804–839. doi: 10.1128/CMR.17.4.804-839.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cutler JE, Deepe GS, Klein BS. Advances in combating fungal diseases: vaccines on the threshold. Nat Rev Microbiol. 2007;5:13–28. doi: 10.1038/nrmicro1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Iannitti RG, Carvalho A, Romani L. From memory to antifungal vaccine design. Trends Microbiol. 2012;33:467–474. doi: 10.1016/j.it.2012.04.008. [DOI] [PubMed] [Google Scholar]
- 99.Magliani W, Conti S, Arseni S, Salati A, Ravanetti L, Maffei DL, et al. Antibody-mediated protective immunity in fungal infections. New Microbiol. 2005;28:299–309. [PubMed] [Google Scholar]
- 100.Rapaka RR, Ricks DM, Alcorn JF, Chen K, Khader SA, Zheng M, et al. Conserved natural IgM antibodies mediate innate and adaptive immunity against the opportunistic fungus Pneumocystis murina. J Exp Med. 2010;207:2907–2919. doi: 10.1084/jem.20100034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101••.Casadevall A, Pirofski LA. Immunoglobulins in defense, pathogenesis, and therapy of fungal diseases. Cell Host Microbe. 2012;11:447–456. doi: 10.1016/j.chom.2012.04.004. This paper reviews evidence that fungal-specific antibodies can alter the course of mycoses to the benefit or detriment of the host. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102•.Cassone A, Casadevall A. Recent progress in vaccines against fungal diseases. Curr Opin Microbiol. 2012;15:1–7. doi: 10.1016/j.mib.2012.04.004. Discussion of progress in the generation of vaccines against certain opportunistic fungal infections, with a review of those vaccines currently in clinical trial. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Rappuoli R, Mandl CW, Black S, De Gregorio E. Vaccines for the twenty-first century society. Nat Rev Immunol. 2011;11:865–872. doi: 10.1038/nri3085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Baumgartner CK, Malherbe LP. Antigen-driven T-cell repertoire selection during adaptive immune responses. Immunol Cell Biol. 2011;89:54–59. doi: 10.1038/icb.2010.117. [DOI] [PubMed] [Google Scholar]


