Abstract
Cyclooxygenases and their metabolites are important regulators of inflammatory responses and play critical roles in regulating the differentiation of T helper cell subsets in inflammatory diseases. In this review, we highlight new information on regulation of T helper cell subsets by cyclooxygenases and their metabolites. Prostanoids influence cytokine production on both antigen presenting cells and T cells to regulate the differentiation of naïve CD4+ T cells to Th1, Th2 and Th17 cell phenotypes. Cyclooxygenases and PGE2 generally exacerbate Th2 and Th17 phenotypes, while suppressing Th1 differentiation. Thus, cycloxygenases may play a critical role in diseases that involve immune cell dysfunction. Targeting of cyclooxygenases and their eicosanoid products may represent a new approach for treatment of inflammatory diseases, tumors and autoimmune disorders.
Keywords: Cyclooxygenases, T helper cells, Prostanoids, PGE2
1. Introduction
Cyclooxygenases (COXs) metabolize arachidonic acid (AA) to prostaglandin (PG) H2, which is further metabolized by specific synthases to biologically active lipids including PGD2, PGE2, PGF2α, prostacyclin (PGI2) and thromboxane (TXA2). These AA-derived signaling molecules are collectively known as prostanoids, and elicit various effects through their respective cognate receptors, named DP, EP, FP, IP and TP. These receptors are widely expressed in the immune system, including on T cells, B cells, dendritic cells (DCs) and macrophages (Figure 1). Prostanoids also play an important role in modulating the inflammatory response, especially in conditions such as allergic airway inflammation, chronic infections and cancer [1].
Figure 1.
Arachidonic acid metabolism by COX-1 and COX-2. COX1 and COX-2 convert arachidonic acid to PGH2. PGH2 is a substrate for terminal synthases which generate PGE2, PGD2, PGF2α, PGI2 and TXA2, which activate cell signaling through their cognate receptors, EP1-4, DP1-2, FP, IP and TP.
There are two COX isoforms: COX-1 and COX-2. COX-1 is the constitutive isoform, with relatively stable expression, and plays important roles in many physiologic “housekeeping” functions (Figure 1). COX-2 is generally expressed a low levels under basal conditions but becomes induced by various stimuli such as bacterial endotoxins, pro-inflammatory cytokines and growth factors. COX-2-dependent prostanoid production plays an essential role in inflammation and cell proliferation [2]. COX-2 and downstream synthases represent important targets for the treatment of a wide range of diseases from autoimmunity to cancer [3].
Both COX isoforms play critical, but distinct roles in T cell maturation and lymphocyte development [4]. In this review, we highlight recent developments in the regulation of T-cell subsets (e.g., Th1, Th2 and Th17 cells) by COXs and PGs. We also summarize the current understanding of the role of COXs and PGs in regulating the function of T cells in immune diseases, which may provide guidance for future basic, translational and clinical research.
2. T helper cell differentiation and function
During inflammatory and immune responses to pathogens and tumors, naïve, CD4+ T cells differentiate into different effector/helper T cells after activation by antigen-presenting cells (APCs). Two lineages of effector/helper CD4+ T cells, named Th1 and Th2, were first described by Coffman and Mosman over two decades ago [5]. Subsequent studies showed that different stimuli could direct T cells preferentially toward Th1 or Th2 lineages. These lineages are involved in immunity against intracellular and extracellular pathogens, as well as immunopathologies such as autoimmunity and allergy. In general, Th1 cells mediate a more aggressive response by promoting the inflammatory/cytotoxic form of immunity, while Th2 cells mediate less tissue-destructive forms of immunity [6, 7]. Th1 cells are implicated in the pathogenesis of many diseases such as autoimmune disorders, Crohn's disease, Helicobacter pylori-induced peptic ulcer and acute kidney allograft rejection [8]. Allergic atopic disorders, such as allergic rhinitis, asthma and atopic dermatitis, are the result of systemic inflammatory reactions triggered by enhanced Th2-type responses to allergens [9].
The field of immune regulation was dominated by the Th1/Th2 dichotomy until 2005, when IL-17-expressing T helper cells (Th17) were identified as a third lineage of CD4+ effector/helper T cells distinct from the Th1 and Th2 lineages. In addition to Th17 cells, other T-cell subsets were recently discovered, such as Foxp3+ regulatory T (Treg) cells, T follicular helper (Tfh) cells and IL-9-expressing Th9 cells [10, 11].
Differentiation of Th cells is regulated by the inflammatory cytokines found within the local microenvironment. Naïve CD4+ T cell differentiation to Th1 or Th2 subsets often occurs after migration to secondary lymphoid organs. There, APCs, such as DCs, play an important role in the maturation of these naïve cells to distinct T cell lineages. DC-derived IL-12 is a potent inducer of Th1 differentiation. IL-12 induces Th1 differentiation via signal transducer and activator of transcription (Stat) 4 and T-bet signaling [12]. Thus, factors that lower IL-12 production will lead to reduced Th1-promoting capacity. Th1 cells secrete IL-2, IFN-γ, and TNF-α that activate tissue cells and macrophages which are responsible for cell-mediated immunity and phagocyte-dependent protective responses [13]. In contrast, IL-2 and IL-4 activate Stat6 and GATA3 signaling to induce Th2 cell differentiation [12]. Th2 cells secrete IL-4, -5, -6, -10, and -13, which control macrophage function as well as antibody production by B cells [2]. Mature DCs also secrete chemokines that induce the migration of Th1 and Th2 cells. The IFN-γ inducible protein-10 (IP-10) is a Th1-recruiting chemokine, while macrophage-derived chemokine is a Th2 chemoattractant [14–16].
Differentiation of Th17 cells in the mouse is promoted by TGF-β and IL-6, which induce the STAT3-dependent expression of IL-21, IL-23R and the transcription factor RORγt. DC-derived IL-21 and IL-23 then regulate the establishment and clonal expansion of Th17 cells, while RORγt induces secretion of IL-17A, IL-17F and IL-22. IL-17, IL-21, and IL-22 are all potent pro-inflammatory mediators, although IL-22 may also be involved in promoting tissue protection and regeneration [13, 17]. In addition, these Th17-related cytokines can stimulate chemokine secretion by resident tissue cells, leading to the recruitment of neutrophils and macrophages to sites of inflammation. Recruited cells, in turn, produce additional cytokines and proteases that further exacerbate the immune response [13, 17].
In contrast to mouse Th17 differentiation, human Th17 polarization is dependent upon IL-1β, IL-6, IL-21, and IL-23, but does not require TGF-β. Thus, the mechanisms of Th17 differentiation may vary by species. Another notable difference is that human Th17 cells can secrete IL-26, a member of the IL-10 family of cytokines, for which there is no known murine homologue [18, 19].
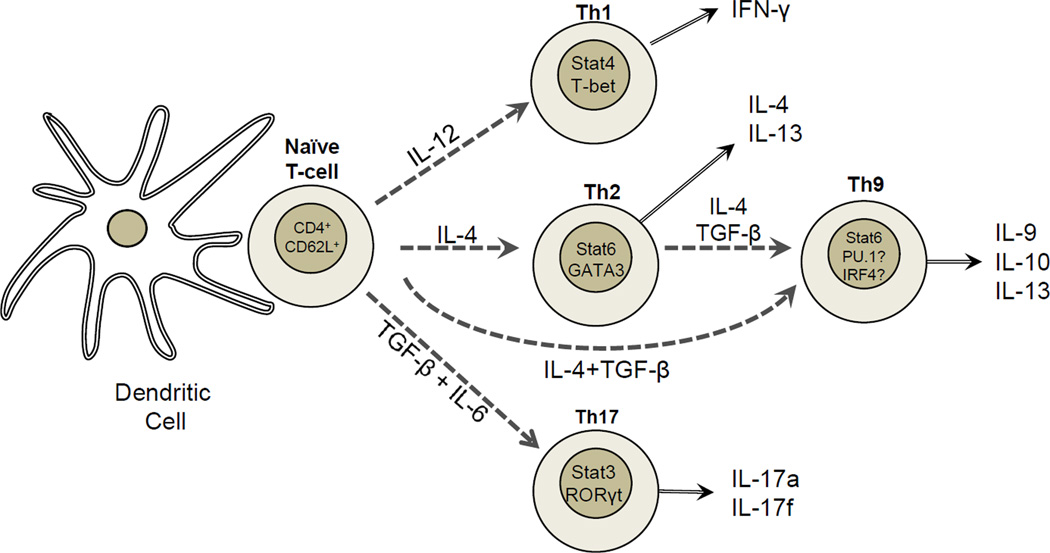
The cytokines produced by Th17 cells can have both beneficial and pathogenic effects. They play a central role in eliminating harmful microbes, but persistent secretion promotes characteristics of autoimmune diseases such as chronic inflammation and tissue destruction. For these reasons, factors involved in Th17 differentiation may serve as potential targets for preventing a variety of autoimmune conditions, including rheumatoid arthritis, multiple sclerosis, and inflammatory bowel disorders [11, 17, 18, 20–26]. The cytokines, signaling molecules and transcription factors involved in Th cell differentiation are summarized in Figure 2. Following activation by antigen-presenting cells such as dendritic cells (DCs), naive CD4+ T cells can be polarized into different effector T cell subsets, including Th1, Th2, Th17 and Th9 cells, depending on the local cytokine environment (IL-12-Th1; IL-4-Th2; IL-6+TGF-β-Th17 and IL-4+TGF-β-Th9). The differentiation of each of these effector T cell subsets is controlled by distinct sets of transcription factors (Stat4/T-bet-Th1; Stat6/GATA3-Th2; Stat3/RORγt; and Stat6/PU.1/IRF4-Th9). Th1, Th2, Th17 and Th9 cell subsets produce their specific cytokines and have different function (Figure 2).
Figure 2.
Differentiation of Th cell subsets. Naïve CD4+ T cells are activated by antigen-presenting cells (APCs) in the presence of co-stimulation through CD28 and/or inducible T-cell co-stimulator (ICOS). The cytokine microenvironment determines the terminal lineage commitment and cytokine expression profiles. IL-12, through STAT4, potentiates the production of IFN-γ and the expression of the transcription factor T-bet. T-bet increases IFN-γ which maintains Th1-cell-lineage commitment. IL-4, activates STAT6 to increase expression of GATA3. GATA3 potentiates IL-4 and IL-13 expression, which determines Th2-cell-lineage differentiation. TGF-β and IL-6 function through STAT6 to increases the expression of RORγt and promote CD4+ T cells to Th17 effector cells that produce IL-17A and IL-17F. TGF-β can convert Th2 cells to Th9 cells that produce IL-9, IL-10 and IL-13 by increasing expression of PU.1 and IRF4 transcription factors. T-bet- T-box expressed in T cells; GATA3, GATA-binding protein 3; IFNγ, interferon-γ; RORγt-retinoic acid receptor-related orphan receptor-γt.
3. COX-derived PGs inhibit the differentiation and activation of Th1 cells
COX-1, via PGE2 production and EP2 receptor signaling, is required for early T cell development (i.e. CD4/CD8 double negative (DN) to double positive (DP) transition). COX-2-derived PGs also stimulate T cell maturation [2]. After maturation to naïve CD4+ cells, differentiation of T helper cell lineages is driven by chemokine signaling and interaction with surface molecules on APCs [5, 27]. Recent studies indicate that several chemical mediators, hormones and neuropeptides are also important in modulating the Th1/Th2 balance and immune responses [28–31]. PGE2 produced by APCs, such as macrophages and DCs, is one of the well-known factors that influence Th1 activation.
Numerous examples indicate that COX-2-derived prostaglandins, such as PGE2, suppress Th1 cell differentiation, particularly in anti-tumor and anti-infection immune responses. Macrophages from BALB/c mice produce greater amounts of PGE2 after LPS stimulation than those from any other strain of mice. This is associated with decreased macrophage-derived IL-12 synthesis, Th1 differentiation, and T cell-derived IFN-γ production [32]. COX-2 activation suppresses Th1 polarization in response to Helicobacter pylori [33]. Conversely, short-term therapy with the specific COX-2 inhibitor celecoxib increases the Th1 immune response in human gastric mucosa [34, 35]. COX-2 inhibition promotes IFN-γ-dependent enhancement of anti-tumor responses [36]. Deletion of COX-2 in mouse mammary epithelial cells delays breast cancer onset through augmentation of type 1 immune responses in tumors [37].
PGE2 downregulates signaling primarily through the Th1-inducing IL-12 pathway. PGE2 has been shown to suppresses LPS-induced IL-12 production by macrophages and DCs [38, 39], and down-regulates IL-12 receptor expression [40–42] to inhibit Th1 differentiation. APC-derived PGE2 down-regulates IFN-γ production by T cells [43–45]. By suppressing Th1 differentiation, PGE2 mediates a shift in the immune response from Th1 to Th2. For example, Han et al. demonstrated that infection of Balb/c mice with low-level doses of Mycobacterium bovix led to Th1 immunity; however, high-dose infection increased levels of COX-2 and PGE2-releasing macrophages, total serum immunoglobulin E (IgE), and serum IgG1 antibodies against heat shock protein 65 (Th2 responses). Cultures of spleen cells isolated from these mice expressed Th2 cytokines, IL-4 and IL-10, in recall responses. Treatment of these mice with the selective COX-2 inhibitor NS-398 reversed these effects. NS-398 increased IFN-γ levels, reduced IL-4 and IL-10 production in recall responses and decreased total serum IgE levels [46]. Thus, low doses of mycobacteria induced low levels of COX-2/PGE2 and Th1 immunity, while high mycobacteria doses induced strong COX-2/PGE2 and Th2 responses.
In vitro evidence indicates that PGs reduce the maturation and function of monocyte-derived dendritic cells (MDC). MDCs express COX-1 and COX-2 and produce prostaglandins that reduce MDC maturation and IL-12 production in an autocrine fashion. Whittaker et al. found that while monocytes express COX-1 constitutively and only express COX-2 when activated, immature MDCs (I-MDC) and mature MDCs constitutively express both COX-1 and COX-2. In I-MDCs, whether COX-1 or COX-2 catalyzes increases in PG production is dependent on several factors, such as IFN-γ and CD40 [47]. PGs produced through COX-2 are autocoids that inhibit IL-12 production and subsequent Th1 differentiation. Treatment of maturing MDCs with NS-398 yields MDCs that stimulate significantly more IFN-γ production (Th1 response). These studies demonstrate that MDCs express both COX isoforms constitutively and produce PGs that regulate Th1 maturation, differentiation and activation.
Another important mediator of Th1 cell differentiation is prostacyclin (PGI2). In vivo, DCs are the primary producers of IL-12, which induces Th1 differentiation. PGI2 analogs not only inhibit IL-12 production by BMDCs, but also directly decrease CD4+ Th1 cytokine production in a partially IP receptor-specific manner [48, 49]. Thus, like PGE2, PGI2 signaling also limits Th1 immune responses.
In summary, COXs metabolites, such as PGE2 and PGI2, inhibit Th1 cell activation in anti-tumor and anti-intracellular infection immune responses via down-regulation of IL-12 and IFN-γ production from APCs and Th2 cells. At high doses, PGE2 can induce a switch from Th1- to Th2- dominated immune responses through downregulated activation of Th1-inducing IL-12 pathway (Figure 3). Thus, modulating PGE2 and PGI2 levels may be a viable therapeutic target for altering Th1 immune responses.
Figure 3.
Regulation of Th1 differentiation and activation by PGE2. Differentiation to Th1 vs. Th2 cells is controled by the balance of IL-12 and IL-4 cytokines. PGE2 downregulates IL-12 production and Th1-inducing capacity of APCs. In the absence of IL-12 production, Th2 differentiation is enhanced. Th2 cells secrete IL-4 which augments Th2 cell development. Th2 cell-derived IL-4 also provides feedback inhibition on APCs to diminish COX-2 expression to restore Th1/Th2 balance.
4. COX-derived PGE2 promotes Th2 type responses
Prostaglandins play important roles in modulating Th2 immunity [45]. Regulation of Th2 cell responses by PGE2 is well documented. In support of its involvement in Th2-mediated human pathology, overproduction of PGE2 is observed in multiple Th2-associated diseases, most notably atopic dermatitis and asthma [50]. The variability in suppressive effects of PGE2 in different mouse strains is determined largely by strain differences in the number of PGE2 receptors [45]. An in vivo Th2-dominant immune response in BALB/c mice has been shown to be dependent on PGE2, further confirming that PGE2 can shift the balance of CD4+ helper T cells toward a Th2 type immune response [45]. In many cases, PGE2 does not specifically upregulate cytokines or transcription factors that promote Th2 differentiation. Instead, PGE2 strongly suppresses Th1-inducing factors generated by T lymphocytes, DCs, APCs and monocytes, and this can shift the balance toward more Th2 cell differentiation.
PGE2 regulates CD4+ T cell differentiation by altering the local cytokine microenvironment through actions on APC. The balance of Th1 vs. Th2 differentiation is determined in large part by relative levels of IL-12 and IL-4. High IL-12 levels direct naïve CD4+ T cells toward Th1 differentiation, while IL-4 directs them toward Th2 differentiation. The Th1-suppressive effect of PGE2 also relies largely on its ability to lower the production of IL-12 in monocytes and DCs. In the absence of IL-12 signaling, IL-4 stimulates Th2 differentiation. Since Th2 cells secrete IL-4, these T cells participate in a positive feedback mechanism that drives Th2 differentiation. Thus, PGE2 suppresses cytokine secretion which, in turn, leads to Th1 differentiation, rather than promotes Th2-inducing cytokines (Figure 3). While PGE2 has been found to enhance IL-12 production from immature DCs stimulated by TNF-α or other signals, PGE2 suppresses the capacity of mature DCs to secrete IL-12 during DC maturation and antigen presentation. Induction of CTL-, Th1-, and NK cell-mediated type 1 immune response is also diminished by PGE2, lowering their Th1-promoting capacity, which results in Th2 responses [3]. In addition, PGE2 also stimulates the production of Th2 chemoattractants from CD40-activated DCs. Similar to its effects on T cells, PGE2 regulates the inflammatory phenotype and function of DCs through EP2 and EP4 receptor subtypes [51].
PGE2 can enhance Th2-type responses, in part through direct actions on T lymphocytes [52]. When added during the priming of naive CD4+ T cells, PGE2 selectively suppresses the production of Th1 cytokines, IL-2 and IFN-γ, which indirectly facilitates the development of a Th2-type immune response. IL-12 receptor expression and IL-12 responsiveness are also reduced by PGE2 in peripheral blood lymphocytes stimulated by anti-CD3, which limits Th1 differentiation [41, 53]. PGE2 leads to enhanced Th2 differentiation, but does so without promoting Th2-inducing cytokines. Bao et al. found that PGE2 inhibits both Th1 and Th2-inducing cytokines in differentiating CD4+ T cells; however, Th1-inducing cytokines were inhibited to a much higher degree [54]. PGE2 increases IL-5 secretion in Th2 clones, but has no effect on production of the key Th2-inducing cytokine IL-4 [2]. Thus, PGE2 promotes the Th2 phenotype by strongly suppressing Th1 differentiation.
The PGE2-dependent effects on naïve CD4+ T cells, Th1 cells and Th2 cells appear to be associated with increased intracellular cyclic AMP (cAMP), suggesting that the Gαs-linked EP2 and/or EP4 receptors may mediate these responses. The EP4 receptor is expressed in Jurkat T cells and responds to PGE2 by raising cAMP levels [55]. COX-2 is upregulated upon T cell activation and may also promote T cell activation through as yet unidentified PGs [56].
PGE2 may also interfere with feedback regulation of the Th1/Th2 balance. As the immune system skews toward the Th2 phenotype, production of the major Th2 cytokine, IL-4 will increase. However, IL-4 can suppress COX-2 expression and enhance Th1-promoting IL-12 production from DCs, which could return the immune system toward a more balanced Th1/Th2 response [52]. Since PGE2 inhibits IL-12 production, it may interfere with this feedback mechanism and continue to drive the immune system toward a Th2 response. Similar to mature DCs, human monocyte production of IL-12 following LPS stimulation is also inhibited by PGE2 in a dose-dependent manner. Macrophages are able to secrete both PGE2 and IL-12 during antigen presentation, and the balance of these two factors may determine whether the T cells differentiate into Th1 or Th2 cells [39, 57].
By shifting the balance of the cellular immune response toward Th2-type cytokine production, PGE2 ultimately potentiates the B cell-mediated humoral response. PGE2 also attenuates Th2-mediated responses indirectly through actions on the effector B lymphocytes. PGE2 inhibits several early events of LPS and IL-4 activation in normal mouse B cells such as lowering class II MHC molecule expression [58, 59]. This may indirectly blunt some of the T cell responses stimulated by B cells. In addition, B cells have an APC capacity and participate in the development of the Th2 subset. Stimulation of CD40 ligand on B cells is necessary for the induction of Th2 responses. Th2 chemoattractant production is potentiated by PGE2 in CD40 or LPS-stimulated B cells [58, 59].
Several studies also indicate a role of COX-1 in Th2 regulation. COX-1−/− mice have exacerbated allergen-induced bronchoconstriction associated with increased numbers of CD4+ and CD8+ T cells. These effects are associated with ex aggerated levels of the Th2 cytokines, IL-4, -5, and -13, and increased levels of eotaxin and thymus- and activation-regulated chemokines (TARC) in their airways. This data suggests a potential role of COX-1 in regulating T cell recruitment, Th2 cytokine secretion, and lung function in the allergic airway [4].
4.1 Regulation of Th2-type response by other PGs
PGD2 also promotes the shift toward a Th2 response. PGD2 is the major mast cell product released during the allergic response. Two G-protein-coupled receptors for PGD2 are chemoattractant receptor homologous molecule expressed on Th2 cells (CRTH2) and D-type prostanoid receptor (DP) [60]. PGD2 stimulates the chemotaxis of eosinophils, basophils and Th2 lymphocytes [60]. In addition, most of the PGD synthase-expressing CD4+ lymphocytes show a typical Th2-type cytokine pattern [61]. Part of the Th2 cell population can also produce PGD2 at sites of antigen presentation, and this may contribute to Th2-related immune responses [61]. PGD2-DP signaling activates p38 MAPK, p44/42 MAPK, and protein kinase C pathways that lead to NF-κB activation and COX-2 gene expression. This positive feedback loop amplifies PGD2 production and perpetuates the Th2 phenotype [62].
PGI2 analogs also stimulate T cell proliferation and Th2 cytokine production in an antigen-specific fashion [48] through the IP receptor [49]. In vivo studies from several groups suggest that PGI2 restrains allergen-induced lung inflammation [63–66]. Taken together, these results suggest that PGI2 signaling modulates Th2 immune responses.
4.2 Discrepancies in reported COX-2/PGE2 effects on Th2-type responses
Even though the majority of published studies suggest that PGE2 enhances Th2 type responses [52], several lines of evidence also support an inhibitory effect of COX-2 on Th2 immune responses. Thus, Th2 regulation by COX-2 is much more complicated than previously thought. Jaffar et al. showed that selective COX-2 inhibition in vivo reduces PGE2 biosynthesis and results in a marked increase in Th2-mediated, but not Th1-mediated, lung inflammation [67]. Carey et al. reported that acute pharmacologic inhibition of COX-1 or COX-2 during allergen sensitization and exposure leads to enhanced Th2 airway responses [4]. Laouini et al. reported a similar effect of COX-2 inhibition, in which it enhanced the Th2 immune response to percutaneous sensitization in a mouse model of atopic dermatitis [68]. Furthermore, using COX-2−/− mice, Hamada et al. demonstrated that COX-2 deficiency enhances Th2 type responses and impairs neutrophil recruitment upon hepatic ischemia/reperfusion injury [69].
The observed differences in COX-2/PGE2 effects may be due, at least in part, to alterations in various developmental phases of the immune response and may depend on the timing of COX-2 inhibitor application. For example, COX-2 inhibition during the immune response (late) phase, but not during the antigen priming (early) phase, can attenuate the development of experimental autoimmune myocarditis [70]. In contrast, in persistent post-infective enteric muscle hypercontractility, it is during the acute infection but not thereafter when increased expression of Th2 cytokines, IL -4, -5, and -13, is observed [71]. Moreover, levels of Th2 cytokines are significantly different between the acute infection phase and post-infection phase in a model of post-infective gut dysfunction [72]. A Th1 to Th2 cytokine transition was observed in the progression of thyroid-associated ophthalmopathy from the early to late stages [46]. Furthermore, it was found that COX inhibition during immune sensitization, but not during the allergic challenge phase, is necessary and sufficient for increasing the Th2 response to OVA challenge after sensitization [73]. Other factors that may affect the direction of actual Th responses include the doses of infective agents and differences in the COX inhibitors used in the studies.
4.3 Regulation of COXs in immune cells
COX-2 expression and activity is regulated in T cells, DCs and monocytes. For instance, CD40 engagement on murine bone marrow-derived DCs upregulates COX-2, PGE2 biosynthesis and EP2 receptor levels via activation of p38 and ERK1/2 MAPK signaling. These events contribute to the induction of Th2 immune responses [51]. T helper cell-derived chemokines can also induce COX-2 expression. Co-culture with monocytes exposed to Th2-inducing cytokines activates COX-2 function in human basal cell carcinoma cells and subsequently increases cellular invasion and angiogenesis [74]. Th2-cell-associated cytokines, particularly IL-4, have also been shown to suppress stimulation of PGE2 biosynthesis and COX activity mainly through the downregulation of COX-2 in various cell lines, including circulating monocytes [75], alveolar macrophages [75], mature DCs [52] and fibroblasts [46]. In contrast, Th1-associated cytokines, such as IFN-γ, have varying effects on COX-2 expression depending on the cell type [7, 46, 76]. T cell-derived cytokines can activate both p38 MAPK and STAT signaling pathways, which are known to reduce COX-2 levels [46]. IL-13, which is secreted by Th2 cells, also decreases fibroblast PGE2 release, and this is accompanied by a decrease in the expression of both COX-1 and COX-2 [77, 78].
4.4 Role of COXs in Th2-related pathological immunologic responses
Alterations in Th1 and Th2 responses exist in many immune disorders, such as immune tolerance, allograft rejection, atopy, cancer and HIV infection [79]. In many cases, both COXs and PGs are involved in this process [33]. In the gut mucosa, LPS induces COX-2-dependent PGE2 production by mononuclear and/or stromal cells, creating a Th2-permissive local microenvironment. PGD2 derived from the gastric mucosa and fibroblasts is protective against inflammatory changes in H. pylori-induced gastritis [80]. In addition, increased PGE2 protects against allograft liver transplant rejection in rats [81].
Atopic disorders, such as allergic rhinitis, asthma and atopic dermatitis, are the result of a systemic inflammatory reaction triggered by a Th2-type immune response [9]. High levels of IL-4, IL-5 and IL-13, and diminished IL-12 and IFN-γ production from APCs appear to be central to the pathophysiology of these disorders [50, 82]. Inhibition of PGE2 biosynthesis would increase IL-12 levels and promote a shift toward Th1 cytokine production.
PGE2 also modulates the actions of bacteria and viruses in evading and subverting the host immune response [83]. For example, Francisella tularensis alter host T cell responses by causing macrophages to release PGE2 that blocks T cell proliferation and promotes a Th2-like responses [84]. A similar scenario is observed for lepromatous leprosy and H. pylori gastritis [85]. HIV infection induces a Th2-type immune response and defects in Th1 differentiation. High levels of PGE2 released from monocytes/macrophages and reduced IL-12 production have also been observed in HIV [86]. Similar to bacteria or viruses, tumor cell growth increases when host immune responses are compromised. PGE2-mediated mechanisms may contribute to an attenuated anti-tumor immune response in humans. Reduced IL-12 production by host MFGs and DCs could result in a lowered Th1 response that is known to contribute to the anti-tumor, cell-mediated immune response [87, 88].
An increase in COX-2 and PGE2 release is associated with the susceptibility of smokers to cancer, infection, and allergy. Cigarette smoke extract (CSE) induces COX-2 protein levels and augments endogenous PGE2 release [89]. This suppresses key DC functions and favors the development of Th2 immunity [89]. CSE inhibits the secretion of the Th1 cytokine IFN-γ and enhances production of the Th2 cytokine IL-4 in the mixed lymphocyte reaction (MLR).
4.5 Chemical regulation of COX-2 and Th2 immunity
Because of their essential roles in modulating immune responses, COX-2 and Th2 immune cells are promising targets for the treatment of many diseases. Several natural plant ingredients are active in various inflammatory diseases based on their effects on COX-2 and Th1–Th2 immunity. In mouse models of allergic diseases, suppression of Th2 differentiation leads to reduced inflammatory outcomes. Examples include curcumin for treatment of inflammatory bowel disease (IBD) [90], thymoquinone for allergic lung inflammation [1], anthocyanins for asthma [91], and 2-amino-3H-phenoxazin-3-one for treatment of T cell-mediated inflammatory autoimmune diseases and bacteria-induced chronic-inflammatory diseases [79].
In summary, Th2 regulation by COXs and PGs is complex and involves both positive/negative feedback loops and direct/indirect pathways (Figure 4). PGE2 promotes IL-4 production in Th2 cells and inhibits IFN-γ and IL-2 secretion in Th1 cells. PGE2 inhibits IL-12 and increases IL-4 production by DCs and macrophages, which in turn decreases Th1 cell and increases Th2 cell immune responses. PGE2 triggers EP2 and EP4 receptor signaling to promote COX-2 transcription in macrophages and DCs, thus forming the COX-2 positive regulation loop. It also increases IL-4 production in Th2 cells, resulting in inhibition of COX-2 expression in dendritic cell and macrophages and forming a negative feedback loop in the Th2 cell immune response. Other PGs such as PGD2 and PGI2 directly promote Th2 cell immune responses. These results indicate that COXs and prostaglandins are critical regulatory mediators of the Th2 cell responses and are involved in the pathogenesis of a variety of inflammatory disorders.
Figure 4.
Regulation of Th2 cell immune response by COXs and PGs. Th2 cell differentiation is regulated by COXs and PGs in the inflammatory cytokine microenvironment. (A) PGE2 inhibits IL-12 production by DCs and macrophages, which reduces Th1 differentiation. PGE2 inhibits IL-12 receptor expression and IFN-γ and IL-2 secretion by Th1 cells. (B) PGE2 indirectly promotes Th2 differentiation and autocrine signaling through increased IL-4 in Th2 cells. (C) PGE2, via EP2 and EP4 receptors, promotes COX-2 transcription in macrophages and DCs, resulting in a COX-2 positive feedback loop. (D) Increased IL-4 production in Th2 cells inhibits COX-2 expression in DCs and macrophages, which forms a negative feedback loop in Th2 cell immune response. (E) PGD2 and PGI2 directly promote Th2 cell immune response.
5. COX-2 and PGs enhance Th17 responses
COX-2 and PGs promote Th17 cell development and function through both direct and indirect pathways. There are several examples whereby PGE2 indirectly promotes Th17 differentiation by altering cytokine production of APCs. IL-23 and IL-12/IL-27 have opposite effects, supporting Th17 and Th1 phenotypes, respectively. PGE2 actively shifts the IL-12/IL-23 balance in DCs in favor of IL-23. PGE2 promotes IL-23 expression, and inhibits IL-12 and IL-27 release from stimulated DCs to enhance the pathogenic inflammatory Th17 phenotype [92, 93]. PGE2 enhances the production of IL-23 and IL-1β in macrophages and DCs while down-regulating IL-12 production [94]. PGE2 promotes Th17 development by enhancing IL-6 production from macrophages and increasing IL-23 and IL-6 secretion by DCs. B. bronchiseptica-infected macrophages can induce IL-17 production from naive CD4+ splenocytes and lung tissues from B. bronchiseptica-infected mice exhibit a strong Th17 immune response. PGE2 enhances DC-derived IL-6 production and shifts the IL-23/IL-12 balance in favor of IL-23, resulting in increased IL-17 production [92]. In humans, peripheral blood mononuclear cells (PBMCs) produce PGE2, which, together with IL-23, favors the expansion of human Th17 cells from and further enhances IL-17 production induced by IL-23 from memory CD4+ T cells [95].
COXs and PGs also directly regulate Th17 cells. PGE2 can directly promote the differentiation and pro-inflammatory functions of human and murine Th17 cells. In human purified naïve T cells, PGE2 acts via EP2- and EP4-mediated signaling and cyclic AMP pathways to upregulate IL-23 and IL-1 receptor expression [96, 97]. In the presence of IL-1β and IL-23, PGE2 may favor the development and/or expansion of Th17 cells, while downregulating proliferation of non-Th17 cells. In addition, it may convert T cell lineages into IL-17 producers, resulting in an “apparent” increase in the number of Th17 cells. Moreover, PGE2 promotes the selective enrichment of Th17 cells by differentially modulating the proliferation of memory T cell subsets in vitro [98].
In 2003, our lab reported that COX-1 and COX-2 regulate the Th1–Th2 balance in allergic and non-allergic lung diseases [4]. Recently, we found that COX-2-derived PGF2α and PGI2 regulate Th17 cell differentiation during allergic lung inflammation through FP and IP receptors on T cells. Together, these data indicate that PGI2, PGF2α and PGE2 can promote Th17 cell differentiation via binding to FP, IP, EP2 and EP4 receptors (Figure 5) [99, 100].
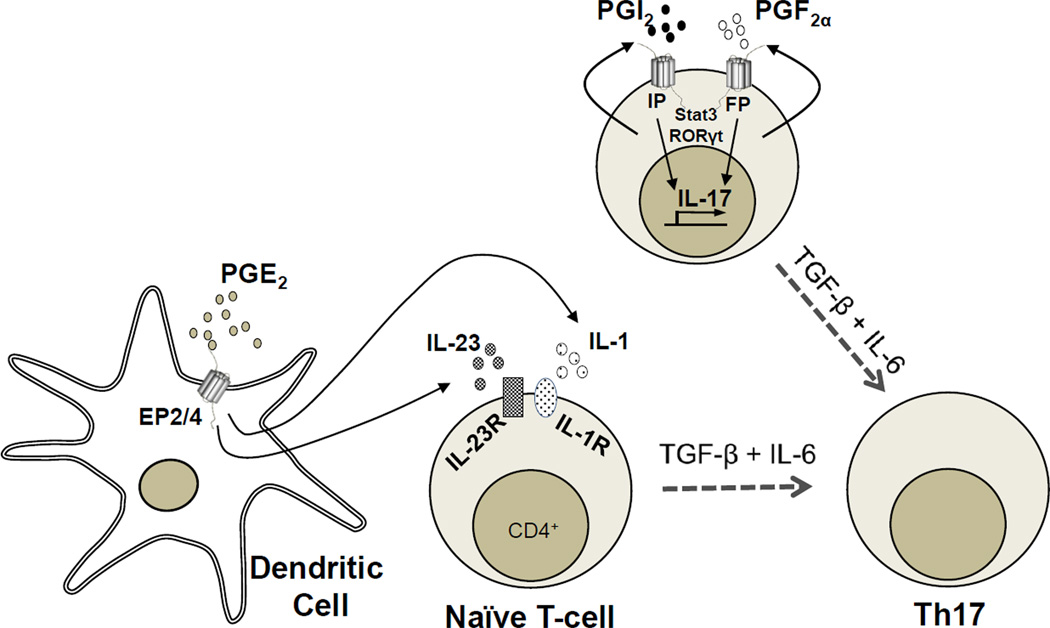
Figure 5.
Regulation of Th17 cell differentiation by COX-2 and PGs. (A) Binding of PGE2 to EP2 and EP4 on DCs leads to increased IL-23 and IL-1β expression. In addition, PGE2 induces IL-23R and IL-1R expression on CD4+ T cells. The IL-23 and IL-1β signals activate Stat3 and upregulate RORγt, which in conjunction with TGF-β and IL-6, leads to IL-17 expression. (B) Autocrine effect of PGI2 and PGF2α on Th17 cell differentiation. CD4+ T cells generate PGI2 and PGF2α. Activation of IP and FP receptors on CD4+ T cells induces Stat3 signaling and upregulation of RORγt, which enhances TGF-β and IL-6 induction of IL-17 expression.
In contrast, PGE2 downregulates some functions of Th17 cells. Napolitani et al. demonstrated that PGE2 inhibits IFN-γ and IL-10 production by Th17 cells, which attenuates their pathogenic potential. These opposing effects of PGE2 are mediated by differential signaling through EP2 and EP4 receptors, leading to a rapid increase in levels of RORγt and a decrease in T cell-specific T-box transcription factor 21 (T-bet) expression [98].
Overall, COXs and PGs promote Th17 cell development via paracrine and autocrine regulation. In a paracrine manner, PGE2 triggers EP2 and EP4 receptor signaling to increase IL-23 and IL-6 secretion, thus changing the IL-23/IL-12 balance of DCs and macrophages. IL-6 and TGF-β then drive naïve CD4+ T cells to Th17 cells, which proliferate under the influence of IL-23. In the autocrine pathway, PGI2 and PGF2α promote Th17 differentiation via their respective FP and IP receptors, independent of DC interactions (Figure 5). Findings from these studies suggest possible therapies to treat Th17 cell-related inflammatory diseases.
6. Clinical consequences of COX inhibition in asthma and other T cell-mediated allergic diseases
Prostaglandins are critical mediators of inflammation and affect both humoral and T cell-mediated immune responses in humans. In experimental allergic diseases, COX-2 regulates the development of T helper cell subsets [101–103]. Examination of tissue sections from patients with chronic tonsillitis show that activated B and T cells express both COX-1 and COX-2 [101]. Further studies will be necessary to elucidate the role of COX expression and/or function in immunologic responses in chronic tonsillitis [23].
Pharmacologic manipulation of COX enzyme activity also affects allergic and inflammatory responses in humans. The COX-1 inhibitor aspirin induces a form of asthma termed aspirin-induced asthma (AIA), which is part of a syndrome that includes rhinitis and nasal polyposis. Aspirin suppresses PGE2 levels in sensitive individuals, which leads to upregulation of lipid biosynthesis and respiratory distress. AIA can be at least partly reversed by treatment with PGE2, which provides direct evidence that prostanoids can have direct effects on asthma pathogenesis [104].
Other non-steroidal anti-inflammatory drugs (NSAIDs) inhibit both COX-1 and COX-2, and offer relief from many inflammatory conditions; however, NSAIDs can induce serious side effects, including upper gastrointestinal injury. Selective COX-2 inhibitors (e.g. celecoxib, rofecoxib, valdecoxib, parecoxib, etoricoxib and lumiracoxib) avoid COX-1-dependent gastrointestinal effects. COX-2 produces prostaglandins almost exclusively responsible for pain, inflammation and fever. COX-2 inhibitors are often prescribed for the treatment of osteoarthritis, rheumatoid arthritis and acute pain. Some studies suggest that selective COX-2 inhibitors reduce the incidence of gastrointestinal side effects compared with conventional NSAIDs. COX-2 selective inhibitors are used as alternative anti-inflammatory drugs in patients with ASA/NSAID intolerance [105, 106], asthma [107–120] and other allergic diseases [105, 106, 121, 122]. COX-2-selective inhibitors also have efficacy similar to NSAIDs in other inflammatory arthropathies including ankylosing spondylitis and gout [123]; however, recent concerns about prothrombotic cardiovascular effects in humans have limited widespread use of COX-2 inhibitors [124]. COX-2 regulates the development T helper cell subsets in experimental models of allergic diseases [101–103]. Thus, COX-2 inhibitors may suppress inflammation through attenuation of T cell differentiation. It is unclear to what degree suppression of immune responses contributes to the efficacy of COX-2 inhibitors in T cell-mediated allergic diseases in humans.
Not all COX-2 inhibitors alleviate inflammatory diseases. The ability of selective COX-2 inhibitors to exacerbate colonic injury in different models of experimental colitis suggests that PGs derived from COX-2 are anti-inflammatory and mucosal protective in this condition. Indeed, PGE2 inhibits inflammatory cytokines and stimulates mucus secretion in the gastrointestinal mucosa through activation of (EP4) receptors. Pretreatment with intraluminal PGE2 analogs (e.g. 16,16’-dimethyl PGE2) reduces the severity of injury induced by trinitrobenzenesulfonic acid and acetic acid [125–127]. Together, these studies argue against the use of COX-2-selective inhibitors for the treatment of human or experimental colitis.
7. Conclusions
In summary, COXs and their metabolites are important regulators of inflammatory responses and play critical roles in regulating the differentiation of T helper cell subsets in inflammatory diseases, tumor immunity and autoimmune diseases. PGs influence cytokine production on both APCs and T cells to regulate the differentiation of naïve CD4+ T cells to Th1, Th2 and Th17 phenotypes. COXs and PGE2 generally exacerbate Th2 and Th17 phenotypes, while suppressing Th1 differentiation. Thus, they may play a critical role in diseases that involve immune cell dysfunction. COX-2 inhibitors are used in clinical treatment of asthma and T cell-mediated allergic diseases. These findings may provide new avenues of treating inflammatory diseases, tumors and autoimmune disorders.
Highlights.
Prostanoids influence cytokine production on both antigen presenting cells and T cells to regulate the differentiation of naïve CD4+ T cells to Th1, Th2 and Th17 cell phenotypes
COXs and PGE2 generally suppressing Th1 differentiation and exacerbate Th2 and Th17 phenotypes
Targeting of cyclooxygenases and their eicosanoid products may represent a new approach for treatment of inflammatory diseases, tumors and autoimmune disorders.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.El Mezayen R, El Gazzar M, Nicolls MR, et al. Effect of thymoquinone on cyclooxygenase expression and prostaglandin production in a mouse model of allergic airway inflammation. Immunology Letters. 2006;106:72–81. doi: 10.1016/j.imlet.2006.04.012. [DOI] [PubMed] [Google Scholar]
- 2.Rocca B, FitzGerald GA. Cyclooxygenases and prostaglandins: shaping up the immune response. Int Immunopharmacol. 2002;2:603–630. doi: 10.1016/s1567-5769(01)00204-1. [DOI] [PubMed] [Google Scholar]
- 3.Kalinski P. Regulation of immune responses by prostaglandin E2. Journal of Immunology. 2012;188:21–28. doi: 10.4049/jimmunol.1101029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carey MA, Germolec DR, Bradbury JA, et al. Accentuated T helper type 2 airway response after allergen challenge in cyclooxygenase-1−/− but not cyclooxygenase-2−/− mice. Am J Respir Crit Care Med. 2003;167:1509–1515. doi: 10.1164/rccm.200211-1383OC. [DOI] [PubMed] [Google Scholar]
- 5.Mosmann TR, Coffman RL. TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties. Annual Review of Immunology. 1989;7:145–173. doi: 10.1146/annurev.iy.07.040189.001045. [DOI] [PubMed] [Google Scholar]
- 6.Morel PA, Oriss TB. Crossregulation between Th1 and Th2 cells. Crit Rev Immunol. 1998;18:275–303. doi: 10.1615/critrevimmunol.v18.i4.10. [DOI] [PubMed] [Google Scholar]
- 7.Endo T, Ogushi F, Sone S. LPS-dependent cyclooxygenase-2 induction in human monocytes is down-regulated by IL-13, but not by IFN-gamma. Journal of Immunology. 1996;156:2240–2246. [PubMed] [Google Scholar]
- 8.Romagnani S. Th1/Th2 cells. Inflammatory Bowel Diseases. 1999;5:285–294. doi: 10.1097/00054725-199911000-00009. [DOI] [PubMed] [Google Scholar]
- 9.Romagnani S. The increased prevalence of allergy and the hygiene hypothesis: missing immune deviation, reduced immune suppression, or both? Immunology. 2004;112:352–363. doi: 10.1111/j.1365-2567.2004.01925.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Diaconu CC, Neagu AI, Lungu R, et al. Plasticity of regulatory T cells under cytokine pressure. Roumanian Archives of Microbiology and Immunology. 2010;69:190–196. [PubMed] [Google Scholar]
- 11.Zhu J, Paul WE. Heterogeneity and plasticity of T helper cells. Cell Research. 2010;20:4–12. doi: 10.1038/cr.2009.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hsieh CS, Macatonia SE, Tripp CS, et al. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science. 1993;260:547–549. doi: 10.1126/science.8097338. [DOI] [PubMed] [Google Scholar]
- 13.Peck A, Mellins ED. Plasticity of T-cell phenotype and function: the T helper type 17 example. Immunology. 2010;129:147–153. doi: 10.1111/j.1365-2567.2009.03189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Honda T, Imaizumi K, Yokoi T, et al. Differential Th1/Th2 chemokine expression in interstitial pneumonia. Am J Med Sci. 2010;339:41–48. doi: 10.1097/MAJ.0b013e3181c14aaa. [DOI] [PubMed] [Google Scholar]
- 15.Jordan WJ, Eskdale J, Srinivas S, et al. Human interferon lambda-1 (IFN-lambda1/IL-29) modulates the Th1/Th2 response. Genes Immun. 2007;8:254–261. doi: 10.1038/sj.gene.6364382. [DOI] [PubMed] [Google Scholar]
- 16.Imai T, Nagira M, Takagi S, et al. Selective recruitment of CCR4-bearing Th2 cells toward antigen-presenting cells by the CC chemokines thymus and activation-regulated chemokine and macrophage-derived chemokine. International immunology. 1999;11:81–88. doi: 10.1093/intimm/11.1.81. [DOI] [PubMed] [Google Scholar]
- 17.Peck A, Mellins ED. Breaking old paradigms: Th17 cells in autoimmune arthritis. Clinical Immunology. 2009;132:295–304. doi: 10.1016/j.clim.2009.03.522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miossec P. IL-17 and Th17 cells in human inflammatory diseases. Microbes and Infection / Institut Pasteur. 2009;11:625–630. doi: 10.1016/j.micinf.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 19.Annunziato F, Cosmi L, Liotta F, et al. The phenotype of human Th17 cells and their precursors, the cytokines that mediate their differentiation and the role of Th17 cells in inflammation. International Immunology. 2008;20:1361–1368. doi: 10.1093/intimm/dxn106. [DOI] [PubMed] [Google Scholar]
- 20.Korn T, Bettelli E, Oukka M, Kuchroo VK. IL-17 and Th17 Cells. Annual Review of Immunology. 2009;27:485–517. doi: 10.1146/annurev.immunol.021908.132710. [DOI] [PubMed] [Google Scholar]
- 21.Mommert S, Gschwandtner M, Koether B, et al. Human memory Th17 cells express a functional histamine H4 receptor. The American Journal of Pathology. 2012;180:177–185. doi: 10.1016/j.ajpath.2011.09.028. [DOI] [PubMed] [Google Scholar]
- 22.Kryczek I, Zhao E, Liu Y, et al. Human TH17 cells are long-lived effector memory cells. Science Translational Medicine. 2011;3 doi: 10.1126/scitranslmed.3002949. 104ra100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Leipe J, Grunke M, Dechant C, et al. Role of Th17 cells in human autoimmune arthritis. Arthritis and Rheumatism. 2010;62:2876–2885. doi: 10.1002/art.27622. [DOI] [PubMed] [Google Scholar]
- 24.Lee WW, Kang SW, Choi J, et al. Regulating human Th17 cells via differential expression of IL-1 receptor. Blood. 2010;115:530–540. doi: 10.1182/blood-2009-08-236521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Annunziato F, Cosmi L, Liotta F, et al. Human Th17 cells: are they different from murine Th17 cells? European Journal of Immunology. 2009;39:637–640. doi: 10.1002/eji.200839050. [DOI] [PubMed] [Google Scholar]
- 26.Romagnani S. Human Th17 cells. Arthritis Research & Therapy. 2008;10:206. doi: 10.1186/ar2392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Constant SL, Bottomly K. Induction of Th1 and Th2 CD4+ T cell responses: the alternative approaches. Annual Review of Immunology. 1997;15:297–322. doi: 10.1146/annurev.immunol.15.1.297. [DOI] [PubMed] [Google Scholar]
- 28.Blotta MH, DeKruyff RH, Umetsu DT. Corticosteroids inhibit IL-12 production in human monocytes and enhance their capacity to induce IL-4 synthesis in CD4+ lymphocytes. Journal of Immunology. 1997;158:5589–5595. [PubMed] [Google Scholar]
- 29.Delgado M, Munoz-Elias EJ, Gomariz RP, Ganea D. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide prevent inducible nitric oxide synthase transcription in macrophages by inhibiting NF-kappa B and IFN regulatory factor 1 activation. Journal of Immunology. 1999;162:4685–4696. [PubMed] [Google Scholar]
- 30.van der Pouw Kraan TC, Snijders A, Boeije LC, et al. Histamine inhibits the production of interleukin-12 through interaction with H2 receptors. J Clin Invest. 1998;102:1866–1873. doi: 10.1172/JCI3692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Elenkov IJ, Webster E, Papanicolaou DA, et al. Histamine potently suppresses human IL-12 and stimulates IL-10 production via H2 receptors. Journal of Immunology. 1998;161:2586–2593. [PubMed] [Google Scholar]
- 32.Kuroda E, Yamashita U. Mechanisms of enhanced macrophage-mediated prostaglandin E2 production and its suppressive role in Th1 activation in Th2-dominant BALB/c mice. Journal of Immunology. 2003;170:757–764. doi: 10.4049/jimmunol.170.2.757. [DOI] [PubMed] [Google Scholar]
- 33.Meyer F, Ramanujam KS, Gobert AP, et al. Cutting edge: cyclooxygenase-2 activation suppresses Th1 polarization in response to Helicobacter pylori. Journal of Immunology. 2003;171:3913–3917. doi: 10.4049/jimmunol.171.8.3913. [DOI] [PubMed] [Google Scholar]
- 34.Larussa T, Suraci E, Leone I, et al. Short-term therapy with celecoxib and lansoprazole modulates Th1/ Th2 immune response in human gastric mucosa. Helicobacter. 2010;15:449–459. doi: 10.1111/j.1523-5378.2010.00796.x. [DOI] [PubMed] [Google Scholar]
- 35.Miyamoto K, Miyake S, Mizuno M, et al. Selective COX-2 inhibitor celecoxib prevents experimental autoimmune encephalomyelitis through COX-2-independent pathway. Brain : a Journal of Neurology. 2006;129:1984–1992. doi: 10.1093/brain/awl170. [DOI] [PubMed] [Google Scholar]
- 36.Sharma S, Zhu L, Yang SC, et al. Cyclooxygenase 2 inhibition promotes IFN-gamma-dependent enhancement of antitumor responses. Journal of Immunology. 2005;175:813–819. doi: 10.4049/jimmunol.175.2.813. [DOI] [PubMed] [Google Scholar]
- 37.Markosyan N, Chen EP, Ndong VN, et al. Deletion of cyclooxygenase 2 in mouse mammary epithelial cells delays breast cancer onset through augmentation of type 1 immune responses in tumors. Carcinogenesis. 2011;32:1441–1449. doi: 10.1093/carcin/bgr134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van der Pouw Kraan TC, Boeije LC, Smeenk RJ, et al. Prostaglandin-E2 is a potent inhibitor of human interleukin 12 production. The Journal of Experimental Medicine. 1995;181:775–779. doi: 10.1084/jem.181.2.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harizi H, Juzan M, Pitard V, et al. Cyclooxygenase-2-issued prostaglandin e(2) enhances the production of endogenous IL-10, which down-regulates dendritic cell functions. Journal of Immunology. 2002;168:2255–2263. doi: 10.4049/jimmunol.168.5.2255. [DOI] [PubMed] [Google Scholar]
- 40.Katamura K, Shintaku N, Yamauchi Y, et al. Prostaglandin E2 at priming of naive CD4+ T cells inhibits acquisition of ability to produce IFN-gamma and IL-2, but not IL-4 and IL-5. Journal of Immunology. 1995;155:4604–4612. [PubMed] [Google Scholar]
- 41.Demeure CE, Yang LP, Desjardins C, et al. Prostaglandin E2 primes naive T cells for the production of anti-inflammatory cytokines. European Journal of Immunology. 1997;27:3526–3531. doi: 10.1002/eji.1830271254. [DOI] [PubMed] [Google Scholar]
- 42.Wu C, Ferrante J, Gately MK, Magram J. Characterization of IL-12 receptor beta1 chain (IL-12Rbeta1)-deficient mice: IL-12Rbeta1 is an essential component of the functional mouse IL-12 receptor. Journal of Immunology. 1997;159:1658–1665. [PubMed] [Google Scholar]
- 43.Hilkens CM, Messer G, Tesselaar K, et al. Lack of IL-12 signaling in human allergen-specific Th2 cells. Journal of Immunology. 1996;157:4316–4321. [PubMed] [Google Scholar]
- 44.Hilkens CM, Snijders A, Vermeulen H, et al. Accessory cell-derived IL-12 and prostaglandin E2 determine the IFN-gamma level of activated human CD4+ T cells. Journal of Immunology. 1996;156:1722–1727. [PubMed] [Google Scholar]
- 45.Kuroda E, Sugiura T, Zeki K, et al. Sensitivity difference to the suppressive effect of prostaglandin E2 among mouse strains: a possible mechanism to polarize Th2 type response in BALB/c mice. Journal of Immunology. 2000;164:2386–2395. doi: 10.4049/jimmunol.164.5.2386. [DOI] [PubMed] [Google Scholar]
- 46.Han R, Smith TJ. T helper type 1 and type 2 cytokines exert divergent influence on the induction of prostaglandin E2 and hyaluronan synthesis by interleukin-1beta in orbital fibroblasts: implications for the pathogenesis of thyroid-associated ophthalmopathy. Endocrinology. 2006;147:13–19. doi: 10.1210/en.2005-1018. [DOI] [PubMed] [Google Scholar]
- 47.Whittaker DS, Bahjat KS, Moldawer LL, Clare-Salzler MJ. Autoregulation of human monocyte-derived dendritic cell maturation and IL-12 production by cyclooxygenase-2-mediated prostanoid production. Journal of Immunology. 2000;165:4298–4304. doi: 10.4049/jimmunol.165.8.4298. [DOI] [PubMed] [Google Scholar]
- 48.Zhou W, Hashimoto K, Goleniewska K, et al. Prostaglandin I2 analogs inhibit proinflammatory cytokine production and T cell stimulatory function of dendritic cells. Journal of Immunology. 2007;178:702–710. doi: 10.4049/jimmunol.178.2.702. [DOI] [PubMed] [Google Scholar]
- 49.Zhou W, Blackwell TS, Goleniewska K, et al. Prostaglandin I2 analogs inhibit Th1 and Th2 effector cytokine production by CD4 T cells. Journal of Leukocyte Biology. 2007;81:809–817. doi: 10.1189/jlb.0606375. [DOI] [PubMed] [Google Scholar]
- 50.Kapsenberg ML, Hilkens CM, Wierenga EA, Kalinski P. The paradigm of type 1 and type 2 antigen-presenting cells. Implications for atopic allergy. Clin Exp Allergy. 1999;29(Suppl 2):33–36. [PubMed] [Google Scholar]
- 51.Harizi H, Limem I, Gualde N. CD40 engagement on dendritic cells induces cyclooxygenase-2 and EP2 receptor via p38 and ERK MAPKs. Immunology and Cell Biology. 2011;89:275–282. doi: 10.1038/icb.2010.94. [DOI] [PubMed] [Google Scholar]
- 52.Teloni R, Giannoni F, Rossi P, et al. Interleukin-4 inhibits cyclo-oxygenase-2 expression and prostaglandin E production by human mature dendritic cells. Immunology. 2007;120:83–89. doi: 10.1111/j.1365-2567.2006.02482.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wu CY, Wang K, McDyer JF, Seder RA. Prostaglandin E2 and dexamethasone inhibit IL-12 receptor expression and IL-12 responsiveness. Journal of Immunology. 1998;161:2723–2730. [PubMed] [Google Scholar]
- 54.Bao YS, Zhang P, Xie RJ, et al. The regulation of CD4+ T cell immune responses toward Th2 cell development by prostaglandin E2. Int Immunopharmacol. 2011;11:1599–1605. doi: 10.1016/j.intimp.2011.05.021. [DOI] [PubMed] [Google Scholar]
- 55.Gerlo S, Verdood P, Gellersen B, et al. Mechanism of prostaglandin (PG)E2-induced prolactin expression in human T cells: cooperation of two PGE2 receptor subtypes, E-prostanoid (EP) 3 and EP4, via calcium- and cyclic adenosine 5'-monophosphate-mediated signaling pathways. J Immunol. 2004;173:5952–5962. doi: 10.4049/jimmunol.173.10.5952. [DOI] [PubMed] [Google Scholar]
- 56.Rodriguez C, Lopez P, Pozo M, et al. COX2 expression and Erk1/Erk2 activity mediate Cot-induced cell migration. Cellular Signalling. 2008;20:1625–1631. doi: 10.1016/j.cellsig.2008.05.008. [DOI] [PubMed] [Google Scholar]
- 57.Harizi H, Gualde N. Dendritic cells produce eicosanoids, which modulate generation and functions of antigen-presenting cells. Prostaglandins Leukot Essent Fatty Acids. 2002;66:459–466. doi: 10.1054/plef.2002.0383. [DOI] [PubMed] [Google Scholar]
- 58.Conboy IM, DeKruyff RH, Tate KM, et al. Novel genetic regulation of T helper 1 (Th1)/Th2 cytokine production and encephalitogenicity in inbred mouse strains. J Exp Med. 1997;185:439–451. doi: 10.1084/jem.185.3.439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Roper RL, Phipps RP. Prostaglandin E2 and cAMP inhibit B lymphocyte activation and simultaneously promote IgE and IgG1 synthesis. J Immunol. 1992;149:2984–2991. [PubMed] [Google Scholar]
- 60.Schuligoi R, Sedej M, Waldhoer M, et al. Prostaglandin H2 induces the migration of human eosinophils through the chemoattractant receptor homologous molecule of Th2 cells, CRTH2. Journal of Leukocyte Biology. 2009;85:136–145. doi: 10.1189/jlb.0608387. [DOI] [PubMed] [Google Scholar]
- 61.Tanaka K, Ogawa K, Sugamura K, et al. Cutting edge: differential production of prostaglandin D2 by human helper T cell subsets. Journal of Immunology. 2000;164:2277–2280. doi: 10.4049/jimmunol.164.5.2277. [DOI] [PubMed] [Google Scholar]
- 62.Mandal AK, Zhang Z, Ray R, et al. Uteroglobin represses allergen-induced inflammatory response by blocking PGD2 receptor-mediated functions. The Journal of Experimental Medicine. 2004;199:1317–1330. doi: 10.1084/jem.20031666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Takahashi Y, Tokuoka S, Masuda T, et al. Augmentation of allergic inflammation in prostanoid IP receptor deficient mice. British Journal of Pharmacology. 2002;137:315–322. doi: 10.1038/sj.bjp.0704872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Nagao K, Tanaka H, Komai M, et al. Role of prostaglandin I2 in airway remodeling induced by repeated allergen challenge in mice. American Journal of Respiratory Cell and Molecular Biology. 2003;29:314–320. doi: 10.1165/rcmb.2003-0035OC. [DOI] [PubMed] [Google Scholar]
- 65.Jaffar Z, Ferrini ME, Buford MC, et al. Prostaglandin I2- IP signaling blocks allergic pulmonary inflammation by preventing recruitment of CD4+ Th2 cells into the airways in a mouse model of asthma. Journal of Immunology. 2007;179:6193–6203. doi: 10.4049/jimmunol.179.9.6193. [DOI] [PubMed] [Google Scholar]
- 66.Idzko M, Hammad H, van Nimwegen M, et al. Inhaled iloprost suppresses the cardinal features of asthma via inhibition of airway dendritic cell function. J Clin Invest. 2007;117:464–472. doi: 10.1172/JCI28949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jaffar Z, Wan KS, Roberts K. A key role for prostaglandin I2 in limiting lung mucosal Th2, but not Th1, responses to inhaled allergen. Journal of Immunology. 2002;169:5997–6004. doi: 10.4049/jimmunol.169.10.5997. [DOI] [PubMed] [Google Scholar]
- 68.Laouini D, Elkhal A, Yalcindag A, et al. COX-2 inhibition enhances the TH2 immune response to epicutaneous sensitization. The Journal of Allergy and Clinical Immunology. 2005;116:390–396. doi: 10.1016/j.jaci.2005.03.042. [DOI] [PubMed] [Google Scholar]
- 69.Hamada T, Tsuchihashi S, Avanesyan A, et al. Cyclooxygenase-2 deficiency enhances Th2 immune responses and impairs neutrophil recruitment in hepatic ischemia/reperfusion injury. Journal of Immunology. 2008;180:1843–1853. doi: 10.4049/jimmunol.180.3.1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Suzuki J, Ogawa M, Futamatsu H, et al. A cyclooxygenase-2 inhibitor alters Th1/Th2 cytokine balance and suppresses autoimmune myocarditis in rats. Journal of Molecular and Cellular Cardiology. 2006;40:688–695. doi: 10.1016/j.yjmcc.2006.01.006. [DOI] [PubMed] [Google Scholar]
- 71.Song GY, Chung CS, Chaudry IH, Ayala A. MAPK p38 antagonism as a novel method of inhibiting lymphoid immune suppression in polymicrobial sepsis. American Journal of Physiology. Cell Physiology. 2001;281:C662–C669. doi: 10.1152/ajpcell.2001.281.2.C662. [DOI] [PubMed] [Google Scholar]
- 72.Akiho H, Deng Y, Blennerhassett P, et al. Mechanisms underlying the maintenance of muscle hypercontractility in a model of postinfective gut dysfunction. Gastroenterology. 2005;129:131–141. doi: 10.1053/j.gastro.2005.03.049. [DOI] [PubMed] [Google Scholar]
- 73.Zhou W, Newcomb DC, Moore ML, et al. Cyclooxygenase inhibition during allergic sensitization increases STAT6-independent primary and memory Th2 responses. Journal of Immunology. 2008;181:5360–5367. doi: 10.4049/jimmunol.181.8.5360. [DOI] [PubMed] [Google Scholar]
- 74.Tjiu JW, Chen JS, Shun CT, et al. Tumor-associated macrophage-induced invasion and angiogenesis of human basal cell carcinoma cells by cyclooxygenase-2 induction. The Journal of Investigative Dermatology. 2009;129:1016–1025. doi: 10.1038/jid.2008.310. [DOI] [PubMed] [Google Scholar]
- 75.Endo T, Ogushi F, Kawano T, Sone S. Comparison of the regulations by Th2-type cytokines of the arachidonic-acid metabolic pathway in human alveolar macrophages and monocytes. American Journal of Respiratory Cell and Molecular Biology. 1998;19:300–307. doi: 10.1165/ajrcmb.19.2.2915. [DOI] [PubMed] [Google Scholar]
- 76.Noguchi K, Shitashige M, Watanabe H, et al. Interleukin-4 and interferon-gamma inhibit prostaglandin production by interleukin-1beta-stimulated human periodontal ligament fibroblasts. Inflammation. 1999;23:1–13. doi: 10.1023/a:1020231331932. [DOI] [PubMed] [Google Scholar]
- 77.Liu X, Kohyama T, Wang H, et al. Th2 cytokine regulation of type I collagen gel contraction mediated by human lung mesenchymal cells. American Journal of Physiology. Lung Cellular and Molecular Physiology. 2002;282:L1049–L1056. doi: 10.1152/ajplung.00321.2001. [DOI] [PubMed] [Google Scholar]
- 78.Saito A, Okazaki H, Sugawara I, et al. Potential action of IL-4 and IL-13 as fibrogenic factors on lung fibroblasts in vitro. International Archives of Allergy and Immunology. 2003;132:168–176. doi: 10.1159/000073718. [DOI] [PubMed] [Google Scholar]
- 79.Kohno K, Miyake M, Sano O, et al. Anti-inflammatory and immunomodulatory properties of 2-amino-3H-phenoxazin-3-one. Biological & Pharmaceutical Bulletin. 2008;31:1938–1945. doi: 10.1248/bpb.31.1938. [DOI] [PubMed] [Google Scholar]
- 80.Hokari R, Nagata N, Kurihara C, et al. Increased expression and cellular localization of lipocalin-type prostaglandin D synthase in Helicobacter pylori-induced gastritis. The Journal of Pathology. 2009;219:417–426. doi: 10.1002/path.2615. [DOI] [PubMed] [Google Scholar]
- 81.Nassiri M, Viciana A, Padmanabhan J, et al. Lymphoid organ production of immunomodulatory eicosanoids in mice resistant to neonatal tolerance induction. Transplantation. 1994;57:1643–1652. [PubMed] [Google Scholar]
- 82.Heinzmann A, Mao XQ, Akaiwa M, et al. Genetic variants of IL-13 signalling and human asthma and atopy. Hum Mol Genet. 2000;9:549–559. doi: 10.1093/hmg/9.4.549. [DOI] [PubMed] [Google Scholar]
- 83.Bansal K, Sinha AY, Ghorpade DS, et al. Src homology 3-interacting domain of Rv1917c of Mycobacterium tuberculosis induces selective maturation of human dendritic cells by regulating PI3K-MAPK-NF-kappaB signaling and drives Th2 immune responses. The Journal of Biological Chemistry. 2010;285:36511–36522. doi: 10.1074/jbc.M110.158055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Woolard MD, Wilson JE, Hensley LL, et al. Francisella tularensis-infected macrophages release prostaglandin E2 that blocks T cell proliferation and promotes a Th2-like response. Journal of Immunology. 2007;178:2065–2074. doi: 10.4049/jimmunol.178.4.2065. [DOI] [PubMed] [Google Scholar]
- 85.Kiszewski AE, Becerril E, Baquera J, et al. Expression of cyclooxygenase type 2 in lepromatous and tuberculoid leprosy lesions. The British Journal of Dermatology. 2003;148:795–798. doi: 10.1046/j.1365-2133.2003.05179.x. [DOI] [PubMed] [Google Scholar]
- 86.Meyaard L, Hovenkamp E, Pakker N, et al. Interleukin-12 (IL-12) production in whole blood cultures from human immunodeficiency virus-infected individuals studied in relation to IL-10 and prostaglandin E2 production. Blood. 1997;89:570–576. [PubMed] [Google Scholar]
- 87.Nastala CL, Edington HD, McKinney TG, et al. Recombinant IL-12 administration induces tumor regression in association with IFN-gamma production. J Immunol. 1994;153:1697–1706. [PubMed] [Google Scholar]
- 88.Baxevanis CN, Reclos GJ, Gritzapis AD, et al. Elevated prostaglandin E2 production by monocytes is responsible for the depressed levels of natural killer and lymphokine-activated killer cell function in patients with breast cancer. Cancer. 1993;72:491–501. doi: 10.1002/1097-0142(19930715)72:2<491::aid-cncr2820720227>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 89.Vassallo R, Tamada K, Lau JS, et al. Cigarette smoke extract suppresses human dendritic cell function leading to preferential induction of Th-2 priming. Journal of Immunology. 2005;175:2684–2691. doi: 10.4049/jimmunol.175.4.2684. [DOI] [PubMed] [Google Scholar]
- 90.Hanai H, Sugimoto K. Curcumin has bright prospects for the treatment of inflammatory bowel disease. Current Pharmaceutical Design. 2009;15:2087–2094. doi: 10.2174/138161209788489177. [DOI] [PubMed] [Google Scholar]
- 91.Park SJ, Shin WH, Seo JW, Kim EJ. Anthocyanins inhibit airway inflammation and hyperresponsiveness in a murine asthma model. Food and Chemical Toxicology : an international journal published for the British Industrial Biological Research Association. 2007;45:1459–1467. doi: 10.1016/j.fct.2007.02.013. [DOI] [PubMed] [Google Scholar]
- 92.Sheibanie AF, Khayrullina T, Safadi FF, Ganea D. Prostaglandin E2 exacerbates collagen-induced arthritis in mice through the inflammatory interleukin-23/interleukin-17 axis. Arthritis and Rheumatism. 2007;56:2608–2619. doi: 10.1002/art.22794. [DOI] [PubMed] [Google Scholar]
- 93.Khayrullina T, Yen JH, Jing H, Ganea D. In vitro differentiation of dendritic cells in the presence of prostaglandin E2 alters the IL-12/IL-23 balance and promotes differentiation of Th17 cells. Journal of Immunology. 2008;181:721–735. doi: 10.4049/jimmunol.181.1.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sheibanie AF, Tadmori I, Jing H, et al. Prostaglandin E2 induces IL-23 production in bone marrow-derived dendritic cells. FASEB journal : official publication of the Federation of American Societies for Experimental Biology. 2004;18:1318–1320. doi: 10.1096/fj.03-1367fje. [DOI] [PubMed] [Google Scholar]
- 95.Chizzolini C, Chicheportiche R, Alvarez M, et al. Prostaglandin E2 synergistically with interleukin-23 favors human Th17 expansion. Blood. 2008;112:3696–3703. doi: 10.1182/blood-2008-05-155408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Boniface K, Bak-Jensen KS, Li Y, et al. Prostaglandin E2 regulates Th17 cell differentiation and function through cyclic AMP and EP2/EP4 receptor signaling. The Journal of Experimental Medicine. 2009;206:535–548. doi: 10.1084/jem.20082293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kleinschek MA, Boniface K, Sadekova S, et al. Circulating and gut-resident human Th17 cells express CD161 and promote intestinal inflammation. The Journal of Experimental Medicine. 2009;206:525–534. doi: 10.1084/jem.20081712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Napolitani G, Acosta-Rodriguez EV, Lanzavecchia A, Sallusto F. Prostaglandin E2 enhances Th17 responses via modulation of IL-17 and IFN-gamma production by memory CD4+ T cells. European Journal of Immunology. 2009;39:1301–1312. doi: 10.1002/eji.200838969. [DOI] [PubMed] [Google Scholar]
- 99.Li H, Bradbury JA, Dackor RT, et al. Cyclooxygenase-2 regulates Th17 cell differentiation during allergic lung inflammation. Am J Respir Crit Care Med. 2011;184:37–49. doi: 10.1164/rccm.201010-1637OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Boswell MG, Zhou W, Newcomb DC, Peebles RS., Jr PGI2 as a regulator of CD4+ subset differentiation and function. Prostaglandins & Other Lipid Mediators. 2011;96:21–26. doi: 10.1016/j.prostaglandins.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Dilek FH, Sahin O, Tokyol C, et al. Expression of cyclooxygenase-1 and 2 in chronic tonsillitis. Indian J Pathol Microbiol. 2010;53:451–454. doi: 10.4103/0377-4929.68272. [DOI] [PubMed] [Google Scholar]
- 102.Muthian G, Raikwar HP, Johnson C, et al. COX-2 inhibitors modulate IL-12 signaling through JAK-STAT pathway leading to Th1 response in experimental allergic encephalomyelitis. J Clin Immunol. 2006;26:73–85. doi: 10.1007/s10875-006-8787-y. [DOI] [PubMed] [Google Scholar]
- 103.Rivero M, Santiago B, Galindo M, et al. Cyclooxygenase-2 inhibition lacks immunomodulatory effects on T cells. Clin Exp Rheumatol. 2002;20:379–385. [PubMed] [Google Scholar]
- 104.Farooque SP, Lee TH. Aspirin-sensitive respiratory disease. Annu Rev Physiol. 2009;71:465–487. doi: 10.1146/annurev.physiol.010908.163114. [DOI] [PubMed] [Google Scholar]
- 105.Bavbek S, Celik G, Ediger D, et al. The use of nimesulide in patients with acetylsalicylic acid and nonsteroidal anti-inflammatory drug intolerance. J Asthma. 1999;36:657–663. doi: 10.3109/02770909909055417. [DOI] [PubMed] [Google Scholar]
- 106.Bavbek S, Celik G, Ozer F, et al. Safety of selective COX-2 inhibitors in aspirin/nonsteroidal anti-inflammatory drug-intolerant patients: comparison of nimesulide, meloxicam, and rofecoxib. J Asthma. 2004;41:67–75. doi: 10.1081/jas-120026063. [DOI] [PubMed] [Google Scholar]
- 107.Fraj J, Valero A, Vives R, et al. Safety of triflusal (antiplatelet drug) in patients with aspirin-exacerbated respiratory diseases. Allergy. 2008;63:112–115. doi: 10.1111/j.1398-9995.2007.01473.x. [DOI] [PubMed] [Google Scholar]
- 108.Goksel O, Aydin O, Misirligil Z, et al. Safety of meloxicam in patients with aspirin/non-steroidal anti-inflammatory drug-induced urticaria and angioedema. J Dermatol. 2010;37:973–979. doi: 10.1111/j.1346-8138.2010.00948.x. [DOI] [PubMed] [Google Scholar]
- 109.Hamad AM, Sutcliffe AM, Knox AJ. Aspirin-induced asthma: clinical aspects, pathogenesis and management. Drugs. 2004;64:2417–2432. doi: 10.2165/00003495-200464210-00004. [DOI] [PubMed] [Google Scholar]
- 110.Hinz B, Brune K. Paracetamol and cyclooxygenase inhibition: is there a cause for concern? Ann Rheum Dis. 2012;71:20–25. doi: 10.1136/ard.2011.200087. [DOI] [PubMed] [Google Scholar]
- 111.Hinz B, Renner B, Brune K. Drug insight: cyclo-oxygenase-2 inhibitors--a critical appraisal. Nat Clin Pract Rheumatol. 2007;3:552–560. doi: 10.1038/ncprheum0619. quiz 551 p following 589. [DOI] [PubMed] [Google Scholar]
- 112.Kowalski ML, Makowska J. Use of nonsteroidal anti-inflammatory drugs in patients with aspirin hypersensitivity : safety of cyclo-oxygenase-2 inhibitors. Treat Respir Med. 2006;5:399–406. doi: 10.2165/00151829-200605060-00005. [DOI] [PubMed] [Google Scholar]
- 113.Loewen PS. Review of the selective COX-2 inhibitors celecoxib and rofecoxib: focus on clinical aspects. Cjem. 2002;4:268–275. doi: 10.1017/s1481803500007508. [DOI] [PubMed] [Google Scholar]
- 114.Marks F, Harrell K, Fischer R. Successful use of cyclooxygenase-2 inhibitor in a patient with aspirin-induced asthma. South Med J. 2001;94:256–257. [PubMed] [Google Scholar]
- 115.Martin-Garcia C, Hinojosa M, Berges P, et al. Celecoxib, a highly selective COX-2 inhibitor, is safe in aspirin-induced asthma patients. J Investig Allergol Clin Immunol. 2003;13:20–25. [PubMed] [Google Scholar]
- 116.Umemoto J, Tsurikisawa N, Nogi S, et al. Selective cyclooxygenase-2 inhibitor cross-reactivity in aspirin-exacerbated respiratory disease. Allergy Asthma Proc. 2011;32:259–261. doi: 10.2500/aap.2011.32.3413. [DOI] [PubMed] [Google Scholar]
- 117.West PM, Fernandez C. Safety of COX-2 inhibitors in asthma patients with aspirin hypersensitivity. Ann Pharmacother. 2003;37:1497–1501. doi: 10.1345/aph.1C497. [DOI] [PubMed] [Google Scholar]
- 118.Woessner KM, Simon RA, Stevenson DD. The safety of celecoxib in patients with aspirin-sensitive asthma. Arthritis Rheum. 2002;46:2201–2206. doi: 10.1002/art.10426. [DOI] [PubMed] [Google Scholar]
- 119.Woessner KM, Simon RA, Stevenson DD. Safety of high-dose rofecoxib in patients with aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2004;93:339–344. doi: 10.1016/S1081-1206(10)61392-0. [DOI] [PubMed] [Google Scholar]
- 120.Yadav UC, Ramana KV, Aguilera-Aguirre L, et al. Inhibition of aldose reductase prevents experimental allergic airway inflammation in mice. PLoS One. 2009;4:e6535. doi: 10.1371/journal.pone.0006535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Bavbek S, Dursun AB, Dursun E, et al. Safety of meloxicam in aspirin-hypersensitive patients with asthma and/or nasal polyps. A challenge-proven study. Int Arch Allergy Immunol. 2007;142:64–69. doi: 10.1159/000096000. [DOI] [PubMed] [Google Scholar]
- 122.Bennett A. The importance of COX-2 inhibition for aspirin induced asthma. Thorax. 2000;55(Suppl 2):S54–S56. doi: 10.1136/thorax.55.suppl_2.S54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Bingham CO., 3rd Development and clinical application of COX-2-selective inhibitors for the treatment of osteoarthritis and rheumatoid arthritis. Cleve Clin J Med. 2002;69(Suppl 1):SI5–S12. doi: 10.3949/ccjm.69.suppl_1.si5. [DOI] [PubMed] [Google Scholar]
- 124.Mukherjee D, Nissen SE, Topol EJ. Risk of cardiovascular events associated with selective COX-2 inhibitors. Jama. 2001;286:954–959. doi: 10.1001/jama.286.8.954. [DOI] [PubMed] [Google Scholar]
- 125.Feng L, Sun W, Xia Y, et al. Cloning two isoforms of rat cyclooxygenase: differential regulation of their expression. Arch Biochem Biophys. 1993;307:361–368. doi: 10.1006/abbi.1993.1601. [DOI] [PubMed] [Google Scholar]
- 126.Sasaki S, Hirata I, Maemura K, et al. Prostaglandin E2 inhibits lesion formation in dextran sodium sulphate-induced colitis in rats and reduces the levels of mucosal inflammatory cytokines. Scand J Immunol. 2000;51:23–28. doi: 10.1046/j.1365-3083.2000.00623.x. [DOI] [PubMed] [Google Scholar]
- 127.Tso JY, Sun XH, Kao TH, et al. Isolation and characterization of rat and human glyceraldehyde-3-phosphate dehydrogenase cDNAs: genomic complexity and molecular evolution of the gene. Nucleic Acids Res. 1985;13:2485–2502. doi: 10.1093/nar/13.7.2485. [DOI] [PMC free article] [PubMed] [Google Scholar]