Abstract
Sox2 is expressed in developing foregut endoderm, with highest levels in the future esophagus and anterior stomach. By contrast, Nkx2.1 (Titf1) is expressed ventrally, in the future trachea. In humans, heterozygosity for SOX2 is associated with anopthalmiaesophageal-genital syndrome (OMIM 600992), a condition including esophageal atresia (EA) and tracheoesophageal fistula (TEF), in which the trachea and esophagus fail to separate. Mouse embryos heterozygous for the null allele, Sox2EGFP, appear normal. However, further reductions in Sox2, using Sox2LP and Sox2COND hypomorphic alleles, result in multiple abnormalities. Approximately 60% of Sox2EGFP/COND embryos have EA with distal TEF in which Sox2 is undetectable by immunohistochemistry or western blot. The mutant esophagus morphologically resembles the trachea, with ectopic expression of Nkx2.1, a columnar, ciliated epithelium, and very few p63+ basal cells. By contrast, the abnormal foregut of Nkx2.1-null embryos expresses elevated Sox2 and p63, suggesting reciprocal regulation of Sox2 and Nkx2.1 during early dorsal/ventral foregut patterning. Organ culture experiments further suggest that FGF signaling from the ventral mesenchyme regulates Sox2 expression in the endoderm. In the 40% Sox2EGFP/COND embryos in which Sox2 levels are ~18% of wild type there is no TEF. However, the esophagus is still abnormal, with luminal mucus-producing cells, fewer p63+ cells, and ectopic expression of genes normally expressed in glandular stomach and intestine. In all hypomorphic embryos the forestomach has an abnormal phenotype, with reduced keratinization, ectopic mucus cells and columnar epithelium. These findings suggest that Sox2 plays a second role in establishing the boundary between the keratinized, squamous esophagus/forestomach and glandular hindstomach.
Keywords: Sox2, Nkx2.1, p63, Mouse embryo, Mutant, Foregut development, Tracheoesophageal fistula, Metaplasia
INTRODUCTION
The foregut of the vertebrate embryo initially consists of a simple epithelial tube of endoderm surrounded by mesoderm. As development proceeds, the foregut gives rise to distinct organ primordia, including those of the trachea, esophagus, stomach and lungs. The endoderm within each organ generates differentiated cells with characteristic specialized functions. In addition, it becomes organized into different classes of epithelia. For example, in the mouse trachea ciliated and secretory cells reside within a pseudostratified columnar monolayer, with only a discontinuous population of p63+ basal cells. By contrast, in the esophagus and forestomach the endoderm develops into a stratified squamous keratinized epithelium with a continuous basal layer of p63+ progenitor cells. Moreover, there is a sharp boundary between this stratified epithelium and the columnar glandular epithelium of the posterior stomach. Understanding how the patterning of the foregut is established and maintained may throw light on human metaplasias and congenital disorders in which morphogenesis and differentiation are disrupted. For example, in the premalignant condition of Barrett’s esophagus the stratified squamous epithelium of the esophagus is replaced by glandular epithelium expressing products characteristic of the posterior stomach or intestine (for a review, see Wild and Hardie, 2003). Another example is the congenital defect known as tracheoesophageal fistula (TEF), in which the separation of the foregut into trachea and esophagus is abnormal (for reviews, see Brunner and van Bokhoven, 2005; Que et al., 2006).
Insights into the mechanisms controlling the patterning, differentiation and morphogenesis of foregut organs have come from a variety of experimental and genetic manipulations (for reviews, see Fukuda and Yasugi, 2005; Grapin-Botton and Melton, 2000; Lemaigre and Zaret, 2004; Roberts, 2000). These studies highlight the importance of reciprocal signaling between the endoderm and mesoderm in foregut patterning. For example, tissue recombination studies in the chick embryo have shown that during a window of early development culturing the epithelium of the small intestine with stomach (gizzard) mesenchyme results in the epithelium acquiring a stomach phenotype. By contrast, combining gizzard epithelium with intestinal mesenchyme has the opposite effect (Fukuda and Yasugi, 2005; Ishii et al., 1998). Typically, such epithelial-mesenchymal interactions involve conserved intercellular signaling pathways and transcription factors, including those of the hedgehog, fibroblast growth factor (FGF), activin, bone morphogenetic protein (BMP), Wnt and retinoic acid pathways (Calmont et al., 2006; Kim, B. et al., 2005; Kim, J. et al., 2005; Kim et al., 2000; Listyorini and Yasugi, 2006; Matsuda et al., 2005; Narita et al., 1998; Narita et al., 2000; Ramalho-Santos et al., 2000; Shin et al., 2006; Theodosiou and Tabin, 2003). However, relatively little is known about the identity and function of the downstream transcription factors (Aubin et al., 2002; Jacobsen et al., 2002; Kawazoe et al., 2002; Kim, J. et al., 2005).
Here, we address the role in anterior foregut development of Sox2, a member of a family of evolutionarily conserved transcription factors containing an Sry-related HMG box. Sox2 is already known to be critically important for early vertebrate development. For example, in mouse and human it is essential for maintaining the pluripotential phenotype of epiblast and embryonic stem (ES) cells, in part by regulating the expression of nanog and repressing genes that promote differentiation (Avilion et al., 2003; Boyer et al., 2005; Rodda et al., 2005). In addition, Sox2 regulates the differentiation of neural stem cells in the CNS and sensory cells in the neuroepithelium of the retina and inner ear (Taranova et al., 2006; Kiernan et al., 2005). Sox2 is expressed at varying levels in the endoderm of foregut-derived organs, including the tongue, esophagus, trachea and proximal lung and stomach (Ishii et al., 1998; Okubo et al., 2005; Williamson et al., 2006). Several lines of evidence indicate that this expression is functionally significant. First, heterozygosity for SOX2 in humans is associated with anophthalmia-esophageal-genital (AEG) syndrome (OMIM 600992). In some affected infants the esophagus and trachea fail to separate normally and the trachea is connected to the stomach by an abnormal distal esophagus (Fantes et al., 2003; Hagstrom et al., 2005; Williamson et al., 2006). Second, we recently established that Sox2 is required for the differentiation of endoderm progenitor cells of the embryonic tongue into taste bud cells versus keratinocytes (Okubo et al., 2006). Finally, crucial processes in both the morphogenesis and differentiation of the embryonic chick lung and proventriculus are associated with dramatic changes in the level of Sox2. Significantly, chick Sox2 levels are downregulated in the epithelium as it responds to morphogenetic signals from the surrounding mesenchyme and gives rise to either distal buds (lung) or secretory glands (proventriculus) (Ishii et al., 1998). In the developing gizzard, chick Sox2 levels are downregulated by combination with intestinal mesenchyme whereas CDXA, a key regulator of intestinal development, is upregulated (Fukuda and Yasugi, 2005; Ishii et al., 1998).
Addressing the in vivo function of Sox2 in the mouse is hampered by the fact that Sox2-null embryos die before gastrulation because of a requirement in the inner cell mass of the blastocyst (Avilion et al., 2003). Recently, two hypomorphic alleles of Sox2, Sox2LP and Sox2COND have been generated. Combining these alleles with the Sox2EGFP-null allele gives compound mutant embryos in which the level of Sox2 varies below 50% (Taranova et al., 2006). Here, we use these mutants to explore the dose-dependent role of Sox2 in the differentiation and morphogenesis of several foregut-derived organs, including the posterior pharynx, trachea, esophagus and stomach.
MATERIALS AND METHODS
Mice
Sox2EGFP/+, Sox2LP/+ and Sox2COND/+ mouse lines were as described (Taranova et al., 2006). Sox2EGFP/+ has been backcrossed for multiple generations to C57Bl/6 whereas the other two lines are maintained on the (129/SvxC57Bl/6) background. A total of 42 Sox2EGFP/LP mutants were generated and analyzed either postnatally or at E18.5. The number of Sox2EGFP/COND mutants examined is shown in Table 1. Embryos of the Fgf10Tg(Myl3-lacZ)24Buck (Fgf10lacZ) line (Kelly et al., 2001) were generously provided by Saverio Bellusci (Children’s Hospital Los Angeles). Titf1tm1Shk heterozygotes are maintained on the Black Swiss background. Homozygous null embryos were examined at E15.5 (n=2) and E16.5 (n=1), together with wild-type littermates.
Table 1.
Genotype/phenotype of Sox2 mutant embryos
| Genotype | Sox2COND/+ | Sox2EGFP/COND | Sox2EGFP/COND | |
|---|---|---|---|---|
| Number of embryos | 64 | 29 | 34 | 5 |
| EA/TEF | – | – | EA with distal TEF | Isolated EA |
| Percentage of total embryos | 48.5 | 21.9 | 25.8 | 3.8 |
| Percentage of total Sox2EGFP/COND embryos | 42.7 | 50.0 | 7.3 |
EA, esophageal atresia; TEF, tracheoesophageal fistula.
Immunohistochemistry
Tissues were fixed in 4% paraformaldehyde (PFA) in phosphate buffered saline (PBS) pH 7.4 for 3-4 hours at 4°C and embedded in paraffin for sectioning and immunohistochemistry using the following antibodies: Anti-p63 (clone 4A4; SantaCruz), anti-BrdU (clone BU-33; Sigma), anti-K14 (clone LL002; NeoMarkers, Fremont, CA), anti-CC10/Scgb1a1 (rabbit polyclonal; kindly provided by Barry Stripp, University of Pittsburgh, PA), anti-β-tubulin (BioGenex), anti-involucrin (kindly provided by Fiona M. Watt, CR-UK Cambridge Research Institute, Cambridge, UK), anti-Muc5AC monoclonal mouse antibody (Clone 45M1; NeoMarkers), anti-Tff2 (mouse monoclonal; kindly provided by Nick Wright, Cancer Research UK), anti-smooth muscle actin (mouse monoclonal clone1A4; Sigma), anti-Sox2 [rabbit polyclonal (Taranova et al., 2006) and Chemicon AB5603 affinity-purified rabbit polyclonal] and anti-Nkx2.1 (mouse monoclonal clone 8G7G3/1; Lab Vision, Fremont, CA). For wholemount immunostaining with phosphorylated ERK1/2 antibody (rabbit polyclonal; Cell Signaling Technology, Danvers, MA) and X-gal staining followed by paraffin embedding and sectioning, see Que et al. (Que et al., 2006). Anterior stomach samples were taken proximally, whereas posterior samples were from the corpus and/or antrum, well away from the junction with the forestomach.
PAS and Alcian blue staining
Sections were treated with or without amylase (alpha-amylase from Aspergillus oryzae, Fluka #10065: 0.5% in distilled water at 37°C for 15 minutes), then stained in Periodic Acid Schiff (PAS) solution. For Alcian blue staining, sections were treated with 3% acetic acid solution for 3 minutes, stained in Alcian blue solution for 30 minutes, and counterstained with eosin.
Organ culture
Explants of E10.0 (32 somite) foregut tissues (from posterior pharynx to the stomach) or E11.0 isolated esophagi from Sox2EGFP/+ embryos were cultured in suspension in groups of 2-3 in a 500-700 μl drop of Dulbecco’s modified Eagle’s medium (DMEM)/F-12(1:1) medium (Gibco) on a glass coverslip in a humidified chamber. The medium contained 0.5% bovine serum albumin (BSA) Fraction V (Roche), 5 μg/ml each of transferrin and insulin and 5 ng/ml selenous acid (ITS, Mediatech), 100 U/ml penicillin G, 100 μg/ml streptomycin, 0.1 mM non-essential amino acid (Gibco) and 1:100 chemically defined lipid concentrate (Gibco, 11905). They were cultured for 36 hours at 37°C in 5% CO2, with or without 50 ng/ml Fgf10 (R&D Systems), and the optimal dose was found to inhibit the separation of foregut into trachea and esophagus. The cultures were repeated three times. After culture, foregut explants were either photographed, fixed in 4% PFA, dehydrated, embedded in paraffin and sectioned at 7 μm, or harvested for RNA extraction.
BrdU labeling
BrdU (Amersham Bioscience, Little Chalfont, UK) was injected intraperitoneally into pregnant females at a dose of 10 μl per gram bodyweight. After 1 hour, embryonic tissues were collected and fixed in 4% PFA in PBS (pH 7.4). For immunohistochemistry, BrdU monoclonal antibody (clone BU33; Sigma) was used, in combination with MOM kit (Vector Laboratories, Burlingame, CA). For quantifying proliferation indexat least 2000 cells were counted from at least seven sections of three different embryos of the following phenotype: wild type, Sox2GFP/COND with TEF, and Sox2GFP/COND without TEF.
Western blot
For each experiment, two E15.5 esophagi (endoderm and mesoderm) of each phenotype were lysed on ice in 30 μl lysis reagent (C2978; Sigma) supplemented with 1:100 protease inhibitor cocktail (P8340; Sigma). Handplates from distal limbs were used as control. Samples containing 20 μg protein were denatured and proteins separated on 12% SDS-polyacrylamide gel under reducing conditions followed by immunoblotting. Primary antibodies were Sox2 (1:2000; Chemicon) and beta actin (1:2000; Abcam). IRDye680-conjugated secondary antibodies (Li-Cor) were used at 1:5000. Image densities were quantified with a LI-COR Odyssey Imaging System and Sox2 levels normalized relative to actin loading control. The experiment was repeated three times and the values averaged.
RT-PCR
Total RNA was extracted by RNeasy (Qiagen), and cDNA was synthesized from 500 ng total RNA by SuperScript (Invitrogen). PCR was performed using the following primer sets: Tff1: 5′-GCACAAGGTGATCTGTGTCCTC-3′, 5′-CCCGGACACT-GTCATCAAAAC-3′; Tff2: 5′-GTGAGCAGTGCTTTGATCTTGG-3′, 5′-GGCACTTCAAAGATCAGGTTGG-3′; Muc5B: 5′-CGGTACAG-CCCAATGAAACC-3′, 5′-GGAAGCAATCATGGAAGTCC-3′; Agr2: 5′-TCCAAGACAAGCAACAGACC-3′, 5′-GTCTTTAGCAGCTTGAG-AGC-3′; Clca3: 5′-TCACTGACCTGAAGGCCAGC-3′, 5′-CAGTGC-AAACCTAGTGTCAC-3′; Reg3g: 5′-AGATGCTTCCCCGTATAACC-3′, 5′-ACTCTAGGCCTTGAATTTGC-3′; Cdx1: 5′-GGACGCCCTACGA-ATGGATG-3′, 5′-AACTCCTCCTTGACGGGCAC-3′; Sox2: 5′-AAC-GGCTCGCCCACCTACAGC-3′, 5′-CAGGGGCAGTGTGCCGTATT-TGG-3′; Nkx2.1: 5′-CGCCATGTCTTGTTCTACCTT-3′, 5′-AGCGTCT-GTCCTCCCTTCT-3′; TAp63: 5′-GCATATCTGGGATTTTCTGGAAC-3′, 5′-GGAGCCCCAGGTTCGTGTA-3′; deltaNp63: 5′-GCAGAAA-AGAGGAGAGCAGC-3′, 5′-GGAGCCCCAGGTTCGTGTA-3′; β-actin: see Okubo et al. (Okubo et al., 2005).
RESULTS
Expression of Sox2 during anterior foregut development
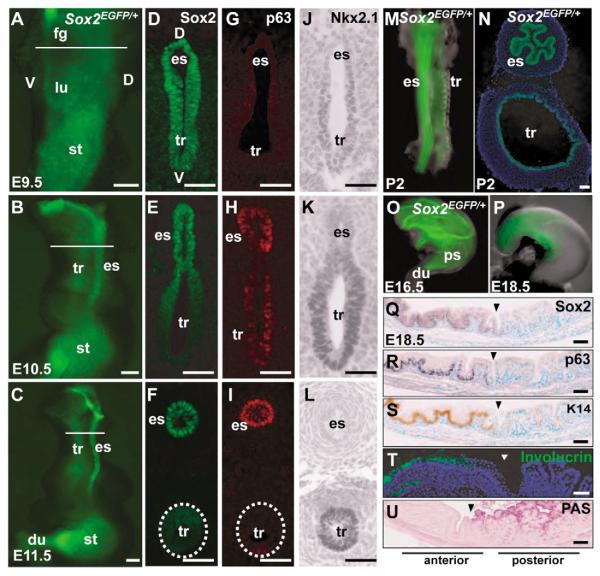
We previously reported expression of Sox2 in the epithelium of the embryonic tongue, with highest levels in fungiform papillae and taste buds (Okubo et al., 2006). Sox2EGFP is also expressed in developing lingual and palatine glands (see Fig. S1 in the supplementary material). We detail here the temporal and spatial pattern of Sox2 expression in the foregut giving rise to the esophagus, trachea and stomach (Fig. 1A-F,M-Q). At E9.5, Sox2 is localized in all endoderm cells of the undivided foregut. Protein levels are somewhat higher dorsally, in the future esophagus, and lower ventrally, in the future trachea (Fig. 1D). This dorsal-ventral difference is very clear one day later (Fig. 1E), and is even more marked after separation of the two tubes (Fig. 1F,N). Subsequently, the esophageal endoderm becomes multilayered and develops into a stratified squamous epithelium. Sox2 levels are always highest in the cells adjacent to the basal lamina (Fig. 1F, Fig. 2H), with lower levels suprabasally. Significantly, the Sox2hi cells also express high levels of p63 (Fig. 1H,I, Fig. 2I), a transcription factor required for the development of many stratified epithelia, including the esophagus, and the basal cells of the trachea (Daniely et al., 2004). RT-PCR shows that the E11.5-18.5 esophagus expresses a higher level of the TAp63 isoform than of the ΔNp63 isoform (data not shown).
Fig. 1. Dynamic expression of Sox2EGFP in foregut-derived organs during mouse development.
(A-C,M-P) Fluorescence microscopy of foregut organs from Sox2EGFP/+ embryos. N is a cryosection of M. (D-F) Immunohistochemical localization of Sox2 protein in sections of foregut before and after separation into trachea and esophagus. Nuclei are counterstained with DAPI. (G-I) Immunohistochemical localization of p63. (J-L) Immunohistochemical localization of Nkx2.1. (Q-U) Sections through boundary (arrowhead) between stratified keratinized epithelium of the E18.5 forestomach and glandular epithelium of posterior stomach. Immunohistochemistry localizes Sox2 (Q), p63 (R), Keratin 14 (S) and involucrin (T) in keratinized epithelium whereas Periodic Acid Schifff (PAS) staining marks mucus-producing luminal cells of glandular stomach (U). Scale bars: 500 μm in A-C; 50 μm in all others. du, duodenum; es, esophagus; fg, foregut; lu, lung; st, stomach; tr, trachea.
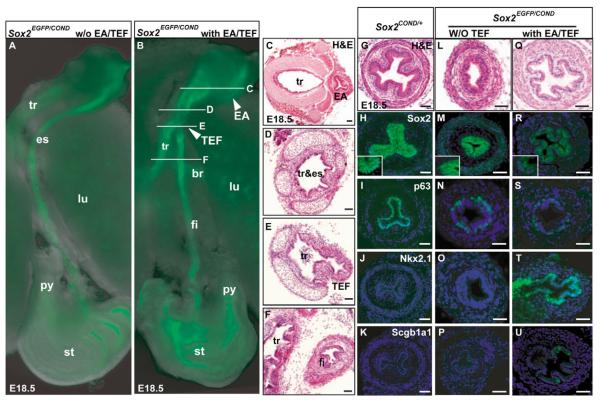
Fig. 2. Tracheoesophageal fistula and esophageal phenotype of Sox2EGFP/COND hypomorphic mutants.
(A,B) Fluorescence microscopy of dissected foregut of Sox2EGFP/COND mutant without (A) and with (B) distal tracheoesophageal fistula (TEF). In B the arrowhead marked EA shows the blind-ended proximal esophagus known in this condition as esophageal atresia (Brunner and van Bokhoven, 2005). White lines indicate level of sections shown in C-F. (C-F) Histological sections through trachea and esophagus of mutant similar to that shown in B. Note the EA in C, and in E the presence of a continuation (TEF) between the dorsal common tube and the distal esophagus (here called the fistula). (G-K) Sections of Sox2COND/+ esophagus demonstrating cellular organization (G), immunohistochemical localization of Sox2, with high-power inset showing strong nuclear staining of basal cells and background cytoplasmic staining of suprabasal cells (H), high p63 in nuclei of basal cells (I) and absence of expression for Nkx2.1 (J) and Scgb1a1 (K). (L-P) Phenotype of esophagus of typical compound mutant without TEF showing cellular organization (note that scale bar is different from G) (L), low Sox2 nuclear staining that is just above background cytoplasmic staining (inset) (M), reduced number of p63-positive cells compared with control (N) and absence of Nkx2.1 and Scgb1a1 (O,P). (Q-U) Phenotype of esophagus of typical compound mutant with TEF, showing cellular organization with a clear monolayered epithelium (Q), absence of Sox2 nuclear staining (R), very few p63-positive cells (S), strong nuclear staining for Nkx2.1 (T) and scattered cells positive for Scgb1a1 (U). Nuclei in H-U are counterstained with DAPI. Scale bars: 100 μm. br, primary bronchus of lung; fi, fistula or distal esophagus; py, pyloric sphincter; tr&es, undivided foregut tube that represents both trachea and esophagus.
The high expression of Sox2 in the dorsal foregut is clearly reciprocal to that of Nkx2.1 (also known as thyroid transcription factor 1, Titf1 – Mouse Genome Informatics). This homeodomain protein plays a key role in regulating trachea and lung development, and promotes the transcription of genes expressed in mature airway epithelial cells, including Scgb1a1 (also known as CC10, CCSP) (Ray et al., 1996). As shown in Fig. 1J-L, levels of Nkx2.1 in the undivided foregut are highest ventrally, in the future trachea and primary lung buds.
In the developing stomach, Sox2EGFP expression initially extends to the junction with the duodenum (Fig. 1O). However, levels are gradually downregulated in the posterior, glandular stomach (corpus and antrum) (Fig. 1P). As the epithelium of the forestomach becomes stratified, high Sox2 protein levels are restricted to the basal layer, where the protein again colocalizes with p63 (Fig. 1Q,R). At E18.5 there is a sharp boundary between the Sox2+, Keratin 14+ (K14+) and p63hi basal epithelial cells of the stratified, keratinized forestomach and the Sox2-, K14- and p63-negative columnar secretory epithelium of the glandular corpus and antrum (Fig. 1S-U).
Hypomorphic mutants die postnatally and have defects in the posterior pharynx
No Sox2EGFP/+, Sox2LP/+ or Sox2COND/+ heterozygous embryos examined at any stage had obvious defects in foregut development. However, at P0 all Sox2EGFP/COND compound mutants showed labored breathing and died with air in the stomach. In approximately 60% of these mutants this phenotype might have resulted from the presence of TEF (see below). However, all the Sox2EGFP/LP mutants also died soon after birth, some with labored breathing and air in the stomach, and none of these mutants had even a small TEF, after serial sectioning of the trachea and esophagus. This raises the possibility that defects in the dynamic interaction between the epiglottis and the soft palate are responsible for the misrouting of air into the esophagus. Histological analysis did reveal abnormalities in the posterior pharyngeal region of P0 Sox2EGFP/LP mutants, in addition to the absence of taste buds previously described (Okubo et al., 2006). Although the circumvallate papilla forms, there are only a few, undeveloped Ebner’s glands arising from it. In addition, there is a complete absence of serous- and mucous-secreting glands in the posterior tongue and soft palate (see Fig. S1D-G in the supplementary material). It is possible that the abnormalities in the soft palate disturb its interactions with the epiglottis, but whether this is the only cause of death is not presently known.
Hypomorphic Sox2EGFP/COND mutants have isolated EA or EA/TEF, with variable penetrance
Approximately 60% of Sox2EGFP/COND mutants have distal TEF, with proximal esophageal atresia (EA) (Fig. 2A-F). In the remaining Sox2EGFP/COND mutants the esophagus has clearly separated from the trachea but is of variable diameter. In a few cases parts of the esophagus are very thin or absent (Table 1 and see Fig. S2A in the supplementary material), a condition classified in humans as isolated EA (Brunner and van Bokhoven, 2005). As discussed below, analysis of Sox2EGFP/COND mutants revealed a clear correlation between levels of Sox2 in the esophagus and the TEF phenotype.
Sox2EGFP/COND compound mutants with EA/TEF have a dorsally located fistula connecting the undivided foregut/trachea with a tube leading to the stomach (Fig. 2B,E,F). This tube is known as the distal esophagus (Brunner and van Bokhoven, 2005) but for simplicity will be called hereafter the fistula. In wild-type littermates at E18.5 the epithelium of the esophagus is stratified (but not yet keratinized) and most of the cells adjacent to the basal lamina are Sox2hi and p63hi, and negative for Nkx2.1 (Fig. 2G-J). In striking contrast, the Sox2EGFP/COND fistula is mostly composed of a simple columnar epithelium, although multilayered regions are present in some sections (Fig. 2Q and see Fig. S2B,C in the supplementary material). Significantly, nuclear staining for Sox2 is absent or barely above background by immunohistochemistry and only a few p63+ cells are present (Fig. 2R,S). However, the nuclei of most of the columnar cells are strongly positive for Nkx2.1 (Fig. 2T). Scattered cells score positively for Scgb1a1, a secreted glycoprotein characteristic of Clara cells of the trachea and proximal airways of the lung (Fig. 2U). Analysis also shows the presence of ciliated cells, and mucus-producing cells staining positively with Alcian blue (see Fig. S2B,C in the supplementary material). The epithelium therefore resembles that of the normal E18.5 trachea, with Clara, ciliated and mucus-secreting cells (the mucus cells later disappear).
It is possible that the ectopic expression of Nkx2.1 in the fistula of Sox2EGFP/COND mutants is a late event, occurring well after the failure of the foregut to separate. We therefore examined Sox2EGFP/COND mutants at E12.5, shortly after the separation normally takes place. This revealed ectopic Nkx2.1 expression and reduced Sox2 in the dorsal undivided foregut even at this time (data not shown).
In the Sox2EGFP/COND mutants without TEF, and in all Sox2EGFP/LP mutants, the esophageal phenotype is variable. There are columnar regions where the epithelium is mostly multilayered, although not as thick as wild type and not squamous (Fig. 2L, note scale difference). There are fewer p63+ and K14+ basal cells than normal (Fig. 2N and Fig. 3F). In addition, many cells in the uppermost (luminal) layer are positive for markers of mucus production, namely Alcian blue, amylase-resistant PAS and Muc5AC (Fig. 3G,H and data not shown). These markers are never seen in wild-type esophagus at any stage of development. Significantly, none of the esophageal cells in these hypomorphic mutants ectopically express Nkx2.1 (Fig. 2O).
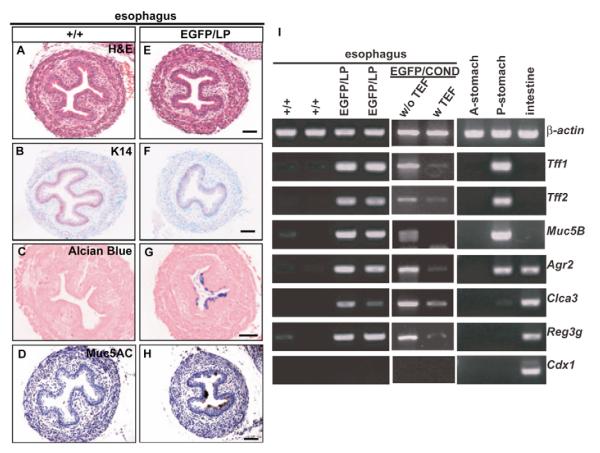
Fig. 3. Abnormal esophagus phenotype of hypomorphic mutants at P0.
Sections of esophagi of wild type (A-D) and Sox2EGFP/LP compound mutants (E-H) stained with haematoxylin and eosin (A,E), anti-Keratin 14 (B,F), Alcian blue (C,G), and anti-Muc5AC (D,H). (I) Semi-quantitative RT-PCR for expression in mutant esophagi of several glandular stomach- and intestine-specific genes. Tff1, Tff2 and Muc5B are normally specifically expressed in posterior (antral/fundic) stomach (P-stomach), not in forestomach (A-stomach). Agr2 is normally expressed in both posterior stomach and intestine. Clca3, Reg3g and Cdx1 are normally expressed in intestine. β-actin is internal control for RT-PCR. Scale bars: 50 μm in A-H.
The immunohistochemical studies described above suggest that there is a correlation between the levels of Sox2 expression in Sox2EGFP/COND mutants and the presence/absence of TEF. To quantify this finding we performed western blot analysis of protein extracted from either the esophagus or the fistula of E15.5 embryos of various genotypes and phenotypes (Fig. 4). As expected, the level of Sox2 protein in the Sox2EGFP/+ esophagus is approximately 50% of that in wild-type (+/+) embryos. The level of Sox2 protein is further reduced, to approximately 18% of wild type, in the esophagus of Sox2EGFP/COND embryos without TEF. In agreement with the immunohistochemical findings, Sox2 protein cannot be detected above background in the fistula of compound mutants with TEF.
Fig. 4. Western blot analysis to compare the levels of Sox2 protein in the E15.5 esophagus of mutants with different genotypes and phenotypes.
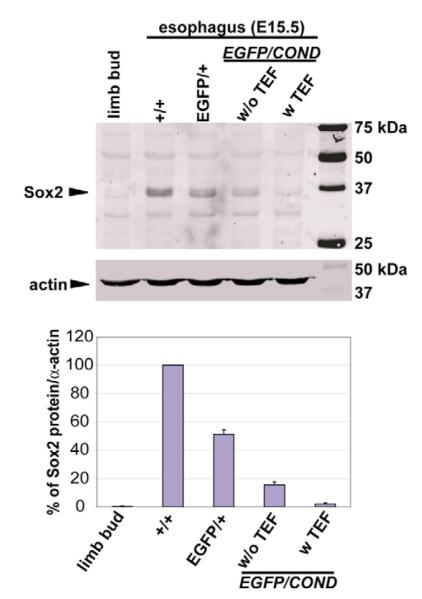
The example shown is typical of three independent experiments. Control sample is from limb bud mesenchyme that does not express Sox2. Quantification showed that the level of Sox2 in Sox2EGFP/COND mutants without fistula was approximately 18% of wild type (mean of three experiments).
To analyze any effect of Sox2 levels on endodermal cell proliferation, E18.5 embryos were exposed to BrdU for 1 hour in utero. Subsequent analysis showed that in Sox2COND/+ control embryos (n=3) approximately 12% of the epithelial cells in the esophagus are labeled, mostly in the basal position (see Fig. S3 in the supplementary material). There is no significant difference in proliferation in the esophagus of Sox2EGFP/COND mutants without TEF (n=3) (P>0.1), but the value was reduced to ~7% in the fistula of mutants with TEF (n=3) (P<0.05). However, this value is not significantly different from that seen in the wild-type trachea (~5%) (P>0.05). It therefore appears that the reduced epithelial proliferation of the mutant fistula is secondary to its acquisition of a tracheal-like phenotype.
In Nkx2.1-null mutants the undivided foregut resembles the esophagus
The ectopic expression of Nkx2.1 in the fistula of Sox2EGFP/COND mutants with TEF raised the possibility that Sox2 and Nkx2.1 normally negatively regulate each other’s expression during the dorsal/ventral (D/V) patterning of the foregut. We therefore examined the undivided foregut of embryos homozygous for a null mutation of Nkx2.1 at E15.5-E16.5 (n=3). In Nkx2.1-null mutants the foregut does not separate into trachea and esophagus and the primary lung buds fail to branch but form sacs that do not express genes specific to mature pulmonary phenotypes (Minoo et al., 1999). As previously reported, the undivided foregut anterior to the lung sacs has an abnormal morphology and the epithelium is multilayered. Immunohistochemistry clearly shows upregulation of Sox2 in the mutant foregut, so that expression in the cells adjacent to the basal lamina resembles that in the esophagus of wild-type littermate controls (Fig. 5A,D and insets). In addition, the basal epithelial cells stain strongly for p63, again as in the normal esophagus and unlike the trachea (Fig. 5B,E). Finally, staining with antibody to smooth muscle actin shows a continuous ring of smooth muscle around the mutant foregut anterior to the lung sacs. This abnormal muscle pattern (and the absence of tracheal cartilage) has been described before (Minoo et al., 1999) and is characteristic of that seen in the esophagus (Fig. 5C,F). Upregulation of Sox2 and p63 was less dramatic in the most anterior undivided foregut (data not shown), although cartilage development is much reduced. Taken together, these results support a model in which absence of Nkx2.1 can result in the dorsalization of the ventral foregut endoderm and its acquisition of an esophagus-like phenotype.
Fig. 5. Dorsalization of the abnormal foregut of Nkx2.1−/− mutants.
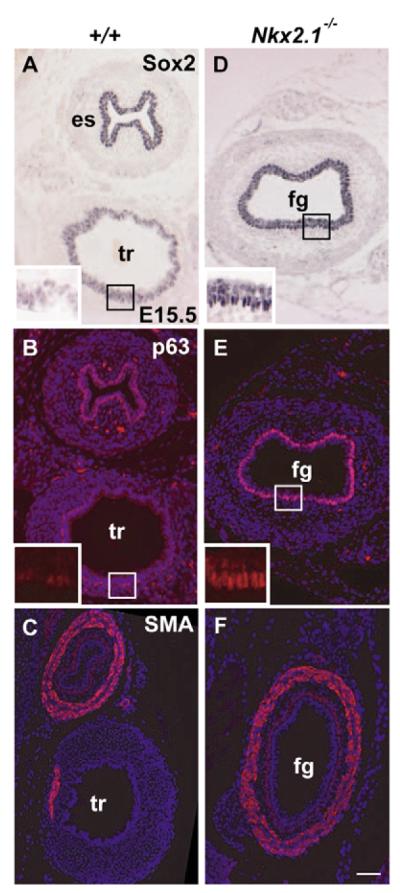
Histological sections were taken through the undivided foregut of Nkx2.1 mutant embryos just anterior to the abnormal lung sacs, and through the trachea and esophagus of wild-type embryos at about the same level. (A,B) Immunohistochemical analysis of wild-type sections showing high levels of Sox2 (A) and p63 (B) in basal cells of the esophagus, and lower levels in the trachea (higher magnification of trachea shown in the insets). By contrast, the foregut of Nkx2.1-null mutants shows high levels of Sox2 (D) and p63 (E) throughout the basal layer of the undivided foregut (high magnification in the insets). The ectopic expression of Sox2hi and p63hi was less pronounced in more rostral sections of the undivided foregut (data not shown). Immunohistochemistry for smooth muscle actin shows only a localized region of smooth muscle in the wild-type trachea (C) but a continuous layer around the mutant foregut (F). Scale bars: 100 μm. fg, undivided foregut; SMA, smooth muscle actin.
Patterning of the undivided foregut and early esophagus is regulated by Fgf10
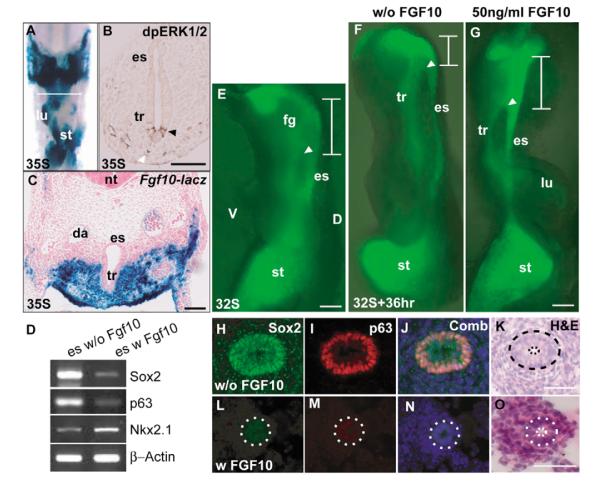
We next addressed the question of how the differential D/V expression of Sox2 and Nkx2.1 in the foregut is established and maintained. Numerous studies have shown that transcription factors in the endoderm of the gastrointestinal tract are regulated by mesenchymal signals, including FGFs. More specifically, FGFs upregulate Nkx2.1 in the early anterior foregut endoderm (Serls et al., 2005), whereas overexpression of Fgf10 in the stomach downregulates Sox2 (Nyeng et al., 2007). In the chick embryo, chick Sox2 levels decrease in distal lung primordia undergoing budding morphogenesis in response to the distal mesenchyme (Ishii et al., 1998) that specifically expresses Fgf10 (Cardoso and Lu, 2006). We therefore hypothesized that FGFs, including Fgf10, regulate Sox2 expression in the foregut endoderm. In support of this idea, analysis of Fgf10lacZ embryos at E10.5 shows that Fgf10 is expressed in the mesoderm surrounding the ventral undivided foregut (Fig. 6A,C). Moreover, immunohistochemistry shows phosphoERK1/2-positive cells in the adjacent ventral endoderm (Fig. 6B). To test the functional significance of these findings we exploited the fact that the foregut at this stage continues to separate into trachea and esophagus when cultured in vitro (Fig. 6E,F). Addition of 50 ng/ml Fgf10 to the medium inhibits this continued separation (Fig. 6G). Moreover, analysis of the samples showed that Fgf10 induces a significant decrease in both Sox2 and p63 in the esophagus, which does not become multilayered (Fig. 6H-O). We also tested the effect of exogenous Fgf10 on Sox2 expression in the isolated esophagus. As shown in Fig. 6D, Fgf10 downregulates Sox2 and p63 and promotes Nkx2.1 expression in the E11.0 esophagus in culture. Taken together, these results support a model in which FGF signaling from the ventral mesenchyme regulates the reciprocal expression patterns of Sox2 and Nkx2.1 in the early foregut endoderm.
Fig. 6. Evidence that Fgf10 plays a role in foregut patterning.
(A,C) Wholemount and section of foregut of E10.5 FGF10lacZ embryo after staining for β-galactosidase. (A) Ventral view after dissection of the heart. Section in C shows strong Fgf10 expression in ventral mesenchyme. (B) Ventral epithelium (black arrowhead) and mesenchyme (white arrowhead) are positive for phosphorylated ERK1/2 staining. (E-G) Fluorescence microscopy of foreguts from 32-somite stage Sox2EGFP/+ embryo before (E) and after culture for 36 hours without (F) or with (G) 50 ng/ml Fgf10 in the culture medium. White arrowhead marks the junction of the esophagus with the foregut and the white line marks the undivided proximal foregut. The anterior limit was determined from phase microscopy. Note that in samples incubated with Fgf10 the region of undivided foregut is longer than in control incubated without Fgf. (H-O) Section through the esophagus of samples similar to those shown in F,G, stained with antibodies to Sox2 (H,L), p63 (I,M), merged (J,N) and haematoxlyin and eosin (K,O). (H-K) Cultured without Fgf10, (L-O) cultured with Fgf10. The dotted line in K-O outlines the esophageal epithelial layer. Note the difference in length of the scale bars. (D) RT-PCR of transcript levels in isolated esophagi from E11 embryos cultured with and without Fgf10. Primers for p63 were for the transactivating isoform. Scale bars: 100 μm in E-G; 50 μm in H-O.
The esophagus of Sox2EGFP/LP and Sox2EGFP/COND mutants without TEF ectopically expresses genes characteristic of glandular stomach and intestine
Based on the abnormal histology of the esophagus of compound mutants without TEF described above, we performed microarray analysis to compare global gene expression in the E18.5 esophagus of Sox2EGFP/LP and Sox2EGP/+ embryos (data not shown). This revealed the misexpression in the mutant esophagus of genes such as Tff1 (trefoil factor 1), Tff2 (trefoil factor 2), Muc5B (mucin 5 subtype B), Agr2 (anterior gradient 2), Clca3 (chloride channel calcium activated 3) and Reg3g (regenerating islet-derived 3 gamma). All these genes are normally expressed in the posterior glandular stomach or intestine. However, there was no significant change in the level of transcripts for the homeodomain genes, Cdx1 and Cdx2, crucial regulators of the intestinal lineage (Roberts, 2000). RT-PCR analysis of cDNA derived from the esophagus of Sox2EGFP/LP mutants confirmed these results (Fig. 3I). They also showed ectopic expression of all of the genes, again except Cdx1, in the esophagus of Sox2EGFP/COND mutants without TEF.
By western blot analysis, our preliminary data showed that only 30% of wild-type Sox2 protein remains in the esophagus of Sox2EGFP/LP mutants (see Fig. S2D in the supplementary material). These findings suggest that a reduction in the level of Sox2 in the esophagus below 50% releases repression of genes that are normally expressed in secretory cells of the wild type and Sox2 heterozygous stomach and intestinal tract, but does not cause a lineage switch from esophagus to intestine.
Anterior stomach of all hypomorphic Sox2 mutants has characteristics of posterior stomach
Sox2 is initially expressed throughout the embryonic stomach but becomes restricted to the anterior before birth (Fig. 1O,P). In the mouse this region is stratified and keratinized and Sox2 expression is highest in the basal cells that are also K14+ and p63hi (Fig. 1Q-S). In all classes of compound mutant, the anterior stomach is highly dysmorphic and variable in histology (Fig. 7F-J and see Fig. S4 in the supplementary material). Some regions resemble the simple secretory epithelium of the posterior glandular stomach in which the cells are negative for K14 and p63 (Fig. 7F,G and see Fig. S4 in the supplementary material). Other regions are multilayered but do not stain for involucrin, a marker for keratinization (Fig. 7H). Like the esophagus of Sox2EGFP/LP mutants, and Sox2EGFP/COND mutants without TEF, the forestomach also contains numerous ectopic luminal cells that stain positively with Alcian blue, PAS (amylase resistant) and antibodies to Muc5AC and Tff2 (Fig. 7I,J and data not shown). These mucin-producing cells are present both in the columnar regions and in the upper layers of the multilayered epithelium.
Fig. 7. Abnormal phenotype of forestomach of Sox2EGFP/LP mutants.
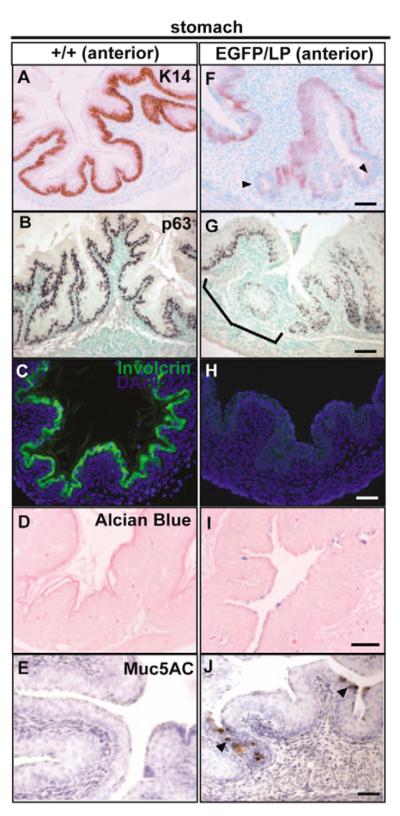
Sections of the wild type (A-E) and mutant (F-J) P0 anterior stomach were stained with anti-Keratin14 (A,F), anti-p63 (B,G), anti-involucrin (C,H), Alcian blue (D,I) and Muc5AC (E,J). Note that Keratin14 expression is reduced or absent (arrowheads) in the mutant forestomach (F). Likewise, anti-p63 immunostaining shows regions of reduced or absent staining in the mutant forestomach (G). Ectopic Alcian blue-positive (I) and Muc5AC-positive (J, arrowheads) cells are present in the mutant forestomach. Scale bars: 50 μm.
DISCUSSION
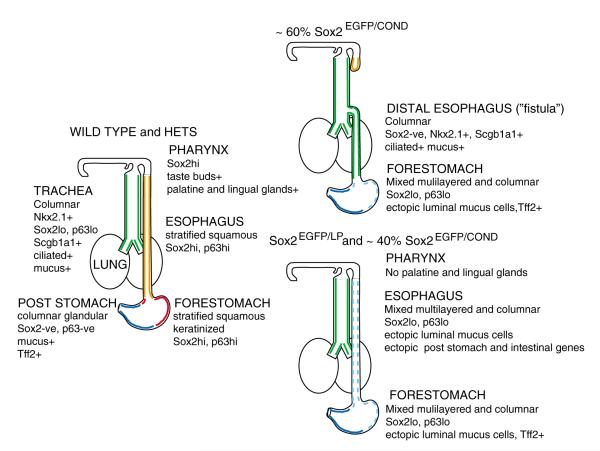
The allelic series of Sox2EGFP, Sox2LP and Sox2COND compound mutants has uncovered multiple roles for Sox2 in the development of the posterior pharynx, trachea, esophagus and forestomach. Compound mutants display a range of phenotypes, summarized in Fig. 8. Several factors are probably responsible for the variation in the severity and penetrance of the phenotypes observed. For example, different foregut regions, and different developmental processes, may require different threshold levels of Sox2 protein. Whether these thresholds are reached will depend on the transcriptional efficiency of the Sox2LP and Sox2COND alleles in different cell types, and/or the activity of modifier genes in the mixed genetic background on which the alleles are maintained. In spite of this phenotypic variability, we can confidently conclude that Sox2 regulates at least two independent processes. The first is the early D/V patterning of the anterior foregut that is coupled to the specification and morphogenesis of the trachea and esophagus. The second is the patterning and differentiation of the foregut into keratinized esophagus and forestomach versus posterior glandular stomach. Our discussion will focus on these two aspects of Sox2 function.
Fig. 8. Summary of the various phenotypes seen in Sox2 hypomorphic mutants.
For details, see text.
D/V patterning of the foregut and the etiology of tracheoesophageal fistula
One model to explain our findings is that Sox2 and Nkx2.1 play reciprocal roles in the D/V patterning of the undivided foregut and the specification of the future dorsal esophagus and ventral trachea, respectively. In particular, we suggest that Sox2 normally directly or indirectly represses Nkx2.1, and vice versa. According to this model, if the level of Sox2 in the undivided foregut falls below a certain threshold early in development Nkx2.1 is ectopically expressed dorsally and there is a cell-autonomous ventralization of the foregut. This, in turn, disrupts the normal separation of the tube, which is mediated by cellular mechanisms as yet largely unknown (Que et al., 2006). In addition, the endodermal tube connected to the stomach expresses Nkx2.1 and is specified as a trachea and develops a tracheal rather than esophageal phenotype. By contrast, when Nkx2.1 is absent, Sox2 is strongly upregulated ventrally, at least in the region of the foregut anterior to the abnormal lung sacs, and this region now acquires characteristics of the esophagus rather than trachea. The fact that this dorsalization was less evident in the most anterior foregut suggests that here other genes, for example Nkx2.5, may compensate for the absence of Nkx2.1. Future studies must determine whether the reciprocal regulation of Sox2 and Nkx2.1 is direct or indirect. Significantly, it has been reported that Sox2 represses Nkx2.1 in human ES cells (Boyer et al., 2005), but the precise mechanism by which this is achieved is not known. In addition, it will be important to develop genetic tools to conditionally inactivate or overexpress Sox2, Nkx2.1 and other potential key regulatory genes in different regions of the early foregut. We also need to identify more genes that function as specific markers for early esophagus and trachea in order to follow changes in cell fate.
Another important goal for the future is to identify all of the factors regulating the levels of Sox2 and Nkx2.1 in the early foregut and to understand how they are functionally coordinated. Candidates include Shh, NMyc, Bmp4, noggin and retinoic acid, all factors associated with TEF in humans and mice (Brunner and van Bokhoven, 2005; Que et al., 2006). Here, we provide strong evidence that Fgf10 made in the ventral mesenchyme is one such factor. We show that exogenous Fgf10 upregulates Nkx2.1 and downregulates Sox2 and interferes with normal separation of the trachea and esophagus. However, Fgf10 cannot be the only FGF mediating normal patterning in vivo because Fgf10-null mutants have a separate trachea and esophagus (Min et al., 1998). Other FGFs expressed in the ventral foregut mesenchyme (Serls et al., 2005) may compensate for the absence of Fgf10 in these mutants. Our findings raise the possibility that mutations in genes acting in the FGF signaling pathway function as modifiers of TEF with variable penetrance, as seen in Sox2EGFP/COND hypomorphic mutants. This could be tested in the future by introducing specific mutations into the genetic background of Sox2EGFP/COND embryos.
Role of Sox2 in regulating epithelial morphogenesis
Our data suggest that in addition to regulating the D/V patterning of the anterior foregut Sox2 also plays a crucial role in the two other processes, which may be tightly interconnected: the epithelial organization of the endoderm and the patterning and differentiation of the esophagus and stomach.
In terms of epithelial organization we argue that high levels of Sox2 promote the morphogenesis of the endoderm into a stratified, squamous epithelium versus a pseudostratified, columnar epithelium. Thus, in the normal esophagus and forestomach of the mouse, where Sox2 levels are high in the basal cells, the epithelium develops into a thick stratified and keratinized layer to protect the organs from damage by food. Such stratified epithelia, including those in the esophagus, are characteristically renewed from a population of p63+, K14+ basal cells (Seery and Watt, 2000). Both loss- and gain-of-function studies suggest that p63 isoforms play crucial roles in regulating the development of stratified epithelia, including that of the esophagus (for a review, see McKeon, 2004). Instead of being stratified and keratinized the epithelium of the p63-null mutant esophagus consists of a simple columnar ciliated epithelium, and both the esophagus and the trachea lack basal cells (Daniely et al., 2004). By contrast, misexpression of p63 in the epithelium of the distal lung leads to the development of a multilayered epithelium that is Nkx2.1+ (Koster et al., 2004). Recent evidence suggests that p63 isoforms regulate the expression of multiple genes associated with epithelial cell adhesion and shape, as well as proliferation and survival (Carroll et al., 2006; Vigano et al., 2006). Here, we find that Sox2 and p63 are coexpressed in basal cells of the esophagus and forestomach and that a reduction in the level of Sox2 in hypomorphic mutants is associated with a reduction in the number of p63+ and K14+ cells. This raises the possibility that Sox2 directly regulates the transcription of p63 and/or K14 in epithelial cells of developing foregut organs.
Role of Sox2 in patterning of the esophagus and stomach
A striking feature of the phenotype of all Sox2 hypomorphic embryos is that the esophagus and forestomach ectopically express genes normally restricted to the posterior glandular stomach, including mucin genes and genes encoding trefoil factor peptides. Moreover, mucin-producing cells are present on the luminal surface of the epithelium, even when it is multilayered. This finding has several implications. First, it suggests that Sox2 normally plays a role in establishing the boundary between the forestomach and glandular stomach by directly or indirectly repressing genes expressed in the posterior epithelium. Second, regions of multilayered epithelium with luminal goblet-like cells have been seen in the dysmorphic lesions of patients with Barrett’s esophagus. It has been suggested that this phenotype is a transition phase in the complex pathogenic process by which the epithelium of the esophagus is replaced by epithelium resembling stomach or intestine (Glickman et al., 2001; Shields et al., 2001). Our results therefore raise the possibility that downregulation of Sox2 plays a role in the etiology of columnar mucous cell metaplasia in the esophagus.
Supplementary Material
Acknowledgments
We thank Saverio Bellusci for kindly providing stained Fgf10lacZ embryos, Jan Jensen (University of Colorado) for helpful discussion and unpublished information and Emma Rawlins and Terry Lechler for stimulating discussion and critical reading. This work was supported by NIMH 641798-01 (L.H.P.), a VA Merit Review Award and a Discovery Award from the Vanderbilt-Ingram Cancer Center (J.R.G.), the Intramural Research Program of the National Cancer Institute, Center for Cancer Research (to Shioko Kimura) and HL071303 (B.L.M.H.).
Footnotes
Supplementary material Supplementary material for this article is available at http://dev.biologists.org/cgi/content/full/134/13/2521/DC1
References
- Aubin J, Dery U, Lemieux M, Chailler P, Jeannotte L. Stomach regional specification requires Hoxa5-driven mesenchymal-epithelial signaling. Development. 2002;129:4075–4087. doi: 10.1242/dev.129.17.4075. [DOI] [PubMed] [Google Scholar]
- Avilion AA, Nicolis SK, Pevny LH, Perez L, Vivian N, Lovell-Badge R. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev. 2003;17:126–140. doi: 10.1101/gad.224503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyer LA, Lee TI, Cole MF, Johnstone SE, Levine SS, Zucker JP, Guenther MG, Kumar RM, Murray HL, Jenner RG, et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell. 2005;122:947–956. doi: 10.1016/j.cell.2005.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunner HG, van Bokhoven H. Genetic players in esophageal atresia and tracheoesophageal fistula. Curr. Opin. Genet. Dev. 2005;15:341–347. doi: 10.1016/j.gde.2005.04.010. [DOI] [PubMed] [Google Scholar]
- Calmont A, Wandzioch E, Tremblay KD, Minowada G, Kaestner KH, Martin GR, Zaret KS. An FGF response pathway that mediates hepatic gene induction in embryonic endoderm cells. Dev. Cell. 2006;11:339–348. doi: 10.1016/j.devcel.2006.06.015. [DOI] [PubMed] [Google Scholar]
- Cardoso WV, Lu J. Regulation of early lung morphogenesis: questions, facts and controversies. Development. 2006;133:1611–1624. doi: 10.1242/dev.02310. [DOI] [PubMed] [Google Scholar]
- Carroll DK, Carroll JS, Leong CO, Cheng F, Brown M, Mills AA, Brugge JS, Ellisen LW. p63 regulates an adhesion programme and cell survival in epithelial cells. Nat. Cell Biol. 2006;8:551–561. doi: 10.1038/ncb1420. [DOI] [PubMed] [Google Scholar]
- Daniely Y, Liao G, Dixon D, Linnoila RI, Lori A, Randell SH, Oren M, Jetten AM. Critical role of p63 in the development of a normal esophageal and tracheobronchial epithelium. Am. J. Physiol. Cell Physiol. 2004;287:C171–C181. doi: 10.1152/ajpcell.00226.2003. [DOI] [PubMed] [Google Scholar]
- Fantes J, Ragge NK, Lynch SA, McGill NI, Collin JR, Howard-Peebles PN, Hayward C, Vivian AJ, Williamson K, van Heyningen V, et al. Mutations in SOX2 cause anophthalmia. Nat. Genet. 2003;33:461–463. doi: 10.1038/ng1120. [DOI] [PubMed] [Google Scholar]
- Fukuda K, Yasugi S. The molecular mechanisms of stomach development in vertebrates. Dev. Growth Differ. 2005;47:375–382. doi: 10.1111/j.1440-169X.2005.00816.x. [DOI] [PubMed] [Google Scholar]
- Glickman JN, Yang A, Shahsafaei A, McKeon F, Odze RD. Expression of p53-related protein p63 in the gastrointestinal tract and in esophageal metaplastic and neoplastic disorders. Hum. Pathol. 2001;32:1157–1165. doi: 10.1053/hupa.2001.28951. [DOI] [PubMed] [Google Scholar]
- Grapin-Botton A, Melton DA. Endoderm development: from patterning to organogenesis. Trends Genet. 2000;16:124–130. doi: 10.1016/s0168-9525(99)01957-5. [DOI] [PubMed] [Google Scholar]
- Hagstrom SA, Pauer GJ, Reid J, Simpson E, Crowe S, Maumenee IH, Traboulsi EI. SOX2 mutation causes anophthalmia, hearing loss, and brain anomalies. Am. J. Med. Genet. A. 2005;138:95–98. doi: 10.1002/ajmg.a.30803. [DOI] [PubMed] [Google Scholar]
- Ishii Y, Rex M, Scotting PJ, Yasugi S. Region-specific expression of chicken Sox2 in the developing gut and lung epithelium: regulation by epithelial-mesenchymal interactions. Dev. Dyn. 1998;213:464–475. doi: 10.1002/(SICI)1097-0177(199812)213:4<464::AID-AJA11>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Jacobsen CM, Narita N, Bielinska M, Syder AJ, Gordon JI, Wilson DB. Genetic mosaic analysis reveals that GATA-4 is required for proper differentiation of mouse gastric epithelium. Dev. Biol. 2002;241:34–46. doi: 10.1006/dbio.2001.0424. [DOI] [PubMed] [Google Scholar]
- Kawazoe Y, Sekimoto T, Araki M, Takagi K, Araki K, Yamamura K. Region-specific gastrointestinal Hox code during murine embryonal gut development. Dev. Growth Differ. 2002;44:77–84. doi: 10.1046/j.1440-169x.2002.00623.x. [DOI] [PubMed] [Google Scholar]
- Kelly RG, Brown NA, Buckingham ME. The arterial pole of the mouse heart forms from Fgf10-expressing cells in pharyngeal mesoderm. Dev. Cell. 2001;1:435–440. doi: 10.1016/s1534-5807(01)00040-5. [DOI] [PubMed] [Google Scholar]
- Kiernan AE, Pelling AL, Leung KK, Tang AS, Bell DM, Tease C, Lovell-Badge R, Steel KP, Cheah KS. Sox2 is required for sensory organ development in the mammalian inner ear. Nature. 2005;434:1031–1035. doi: 10.1038/nature03487. [DOI] [PubMed] [Google Scholar]
- Kim BM, Buchner G, Miletich I, Sharpe PT, Shivdasani RA. The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient Wnt signaling. Dev. Cell. 2005;8:611–622. doi: 10.1016/j.devcel.2005.01.015. [DOI] [PubMed] [Google Scholar]
- Kim JH, Huang Z, Mo R. Gli3 null mice display glandular overgrowth of the developing stomach. Dev. Dyn. 2005;234:984–991. doi: 10.1002/dvdy.20542. [DOI] [PubMed] [Google Scholar]
- Kim SK, Hebrok M, Li E, Oh SP, Schrewe H, Harmon EB, Lee JS, Melton DA. Activin receptor patterning of foregut organogenesis. Genes Dev. 2000;14:1866–1871. [PMC free article] [PubMed] [Google Scholar]
- Koster MI, Kim S, Mills AA, DeMayo FJ, Roop DR. p63 is the molecular switch for initiation of an epithelial stratification program. Genes Dev. 2004;18:126–131. doi: 10.1101/gad.1165104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lemaigre F, Zaret KS. Liver development update: new embryo models, cell lineage control, and morphogenesis. Curr. Opin. Genet. Dev. 2004;14:582–590. doi: 10.1016/j.gde.2004.08.004. [DOI] [PubMed] [Google Scholar]
- Listyorini D, Yasugi S. Expression and function of Wnt5a in the development of the glandular stomach in the chicken embryo. Dev. Growth Differ. 2006;48:243–252. doi: 10.1111/j.1440-169X.2006.00861.x. [DOI] [PubMed] [Google Scholar]
- Matsuda Y, Wakamatsu Y, Kohyama J, Okano H, Fukuda K, Yasugi S. Notch signaling functions as a binary switch for the determination of glandular and luminal fates of endodermal epithelium during chicken stomach development. Development. 2005;132:2783–2793. doi: 10.1242/dev.01853. [DOI] [PubMed] [Google Scholar]
- McKeon F. p63 and the epithelial stem cell: more than status quo? Genes Dev. 2004;18:465–469. doi: 10.1101/gad.1190504. [DOI] [PubMed] [Google Scholar]
- Min H, Danilenko DM, Scully SA, Bolon B, Ring BD, Tarpley JE, DeRose M, Simonet WS. Fgf-10 is required for both limb and lung development and exhibits striking functional similarity to Drosophila branchless. Genes Dev. 1998;12:3156–3161. doi: 10.1101/gad.12.20.3156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minoo P, Su G, Drum H, Bringas P, Kimura S. Defects in tracheoesophageal and lung morphogenesis in Nkx2.1 (−/−) mouse embryos. Dev. Biol. 1999;209:60–71. doi: 10.1006/dbio.1999.9234. [DOI] [PubMed] [Google Scholar]
- Narita T, Ishii Y, Nohno T, Noji S, Yasugi S. Sonic hedgehog expression in developing chicken digestive organs is regulated by epithelial-mesenchymal interactions. Dev. Growth Differ. 1998;40:67–74. doi: 10.1046/j.1440-169x.1998.t01-5-00008.x. [DOI] [PubMed] [Google Scholar]
- Narita T, Saitoh K, Kameda T, Kuroiwa A, Mizutani M, Koike C, Iba H, Yasugi S. BMPs are necessary for stomach gland formation in the chicken embryo: a study using virally induced BMP-2 and Noggin expression. Development. 2000;127:981–988. doi: 10.1242/dev.127.5.981. [DOI] [PubMed] [Google Scholar]
- Nyeng P, Norgaard GA, Kobberup S, Jensen J. Fgf10 signaling controls stomach morphogenesis. Dev. Biol. 2007;303:295–310. doi: 10.1016/j.ydbio.2006.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okubo T, Knoepfler PS, Eisenman RN, Hogan BL. Nmyc plays an essential role during lung development as a dosage-sensitive regulator of progenitor cell proliferation and differentiation. Development. 2005;132:1363–1374. doi: 10.1242/dev.01678. [DOI] [PubMed] [Google Scholar]
- Okubo T, Pevny LH, Hogan BL. Sox2 is required for development of taste bud sensory cells. Genes Dev. 2006;20:2654–2659. doi: 10.1101/gad.1457106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Que J, Choi M, Ziel JW, Klingensmith J, Hogan BL. Morphogenesis of the trachea and esophagus: current players and new roles for noggin and Bmps. Differentiation. 2006;74:422–437. doi: 10.1111/j.1432-0436.2006.00096.x. [DOI] [PubMed] [Google Scholar]
- Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development. 2000;127:2763–2772. doi: 10.1242/dev.127.12.2763. [DOI] [PubMed] [Google Scholar]
- Ray MK, Chen CY, Schwartz RJ, DeMayo FJ. Transcriptional regulation of a mouse Clara cell-specific protein (mCC10) gene by the NKx transcription factor family members thyroid transcription factor 1 and cardiac muscle-specific homeobox protein (CSX) Mol. Cell. Biol. 1996;16:2056–2064. doi: 10.1128/mcb.16.5.2056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts DJ. Molecular mechanisms of development of the gastrointestinal tract. Dev. Dyn. 2000;219:109–120. doi: 10.1002/1097-0177(2000)9999:9999<::aid-dvdy1047>3.3.co;2-y. [DOI] [PubMed] [Google Scholar]
- Rodda DJ, Chew JL, Lim LH, Loh YH, Wang B, Ng HH, Robson P. Transcriptional regulation of nanog by OCT4 and SOX2. J. Biol. Chem. 2005;280:24731–24737. doi: 10.1074/jbc.M502573200. [DOI] [PubMed] [Google Scholar]
- Seery JP, Watt FM. Asymmetric stem-cell divisions define the architecture of human oesophageal epithelium. Curr. Biol. 2000;10:1447–1450. doi: 10.1016/s0960-9822(00)00803-4. [DOI] [PubMed] [Google Scholar]
- Serls AE, Doherty S, Parvatiyar P, Wells JM, Deutsch GH. Different thresholds of fibroblast growth factors pattern the ventral foregut into liver and lung. Development. 2005;132:35–47. doi: 10.1242/dev.01570. [DOI] [PubMed] [Google Scholar]
- Shields HM, Rosenberg SJ, Zwas FR, Ransil BJ, Lembo AJ, Odze R. Prospective evaluation of multilayered epithelium in Barrett’s esophagus. Am. J. Gastroenterol. 2001;96:3268–3273. doi: 10.1111/j.1572-0241.2001.05324.x. [DOI] [PubMed] [Google Scholar]
- Shin M, Noji S, Neubuser A, Yasugi S. FGF10 is required for cell proliferation and gland formation in the stomach epithelium of the chicken embryo. Dev. Biol. 2006;294:11–23. doi: 10.1016/j.ydbio.2005.12.019. [DOI] [PubMed] [Google Scholar]
- Taranova OV, Magness ST, Fagan BM, Wu Y, Surzenko N, Hutton SR, Pevny LH. SOX2 is a dose-dependent regulator of retinal neural progenitor competence. Genes Dev. 2006;20:1187–1202. doi: 10.1101/gad.1407906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theodosiou NA, Tabin CJ. Wnt signaling during development of the gastrointestinal tract. Dev. Biol. 2003;259:258–271. doi: 10.1016/s0012-1606(03)00185-4. [DOI] [PubMed] [Google Scholar]
- Vigano MA, Lamartine J, Testoni B, Merico D, Alotto D, Castagnoli C, Robert A, Candi E, Melino G, Gidrol X, et al. New p63 targets in keratinocytes identified by a genome-wide approach. EMBO J. 2006;25:5105–5116. doi: 10.1038/sj.emboj.7601375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wild CP, Hardie LJ. Reflux, Barrett’s oesophagus and adenocarcinoma: burning questions. Nat. Rev. Cancer. 2003;3:676–684. doi: 10.1038/nrc1166. [DOI] [PubMed] [Google Scholar]
- Williamson KA, Hever AM, Rainger J, Rogers RC, Magee A, Fiedler Z, Keng WT, Sharkey FH, McGill N, Hill CJ, et al. Mutations in SOX2 cause anophthalmia-esophageal-genital (AEG) syndrome. Hum. Mol. Genet. 2006;15:1413–1422. doi: 10.1093/hmg/ddl064. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.