Abstract
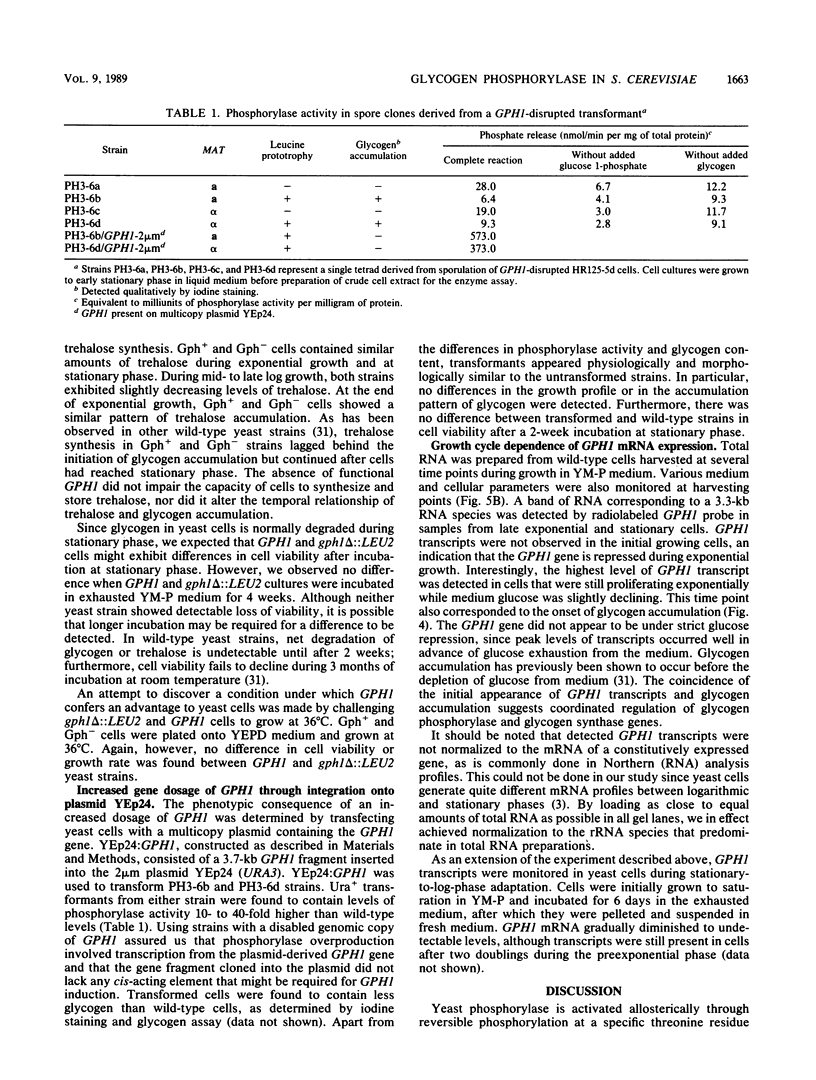
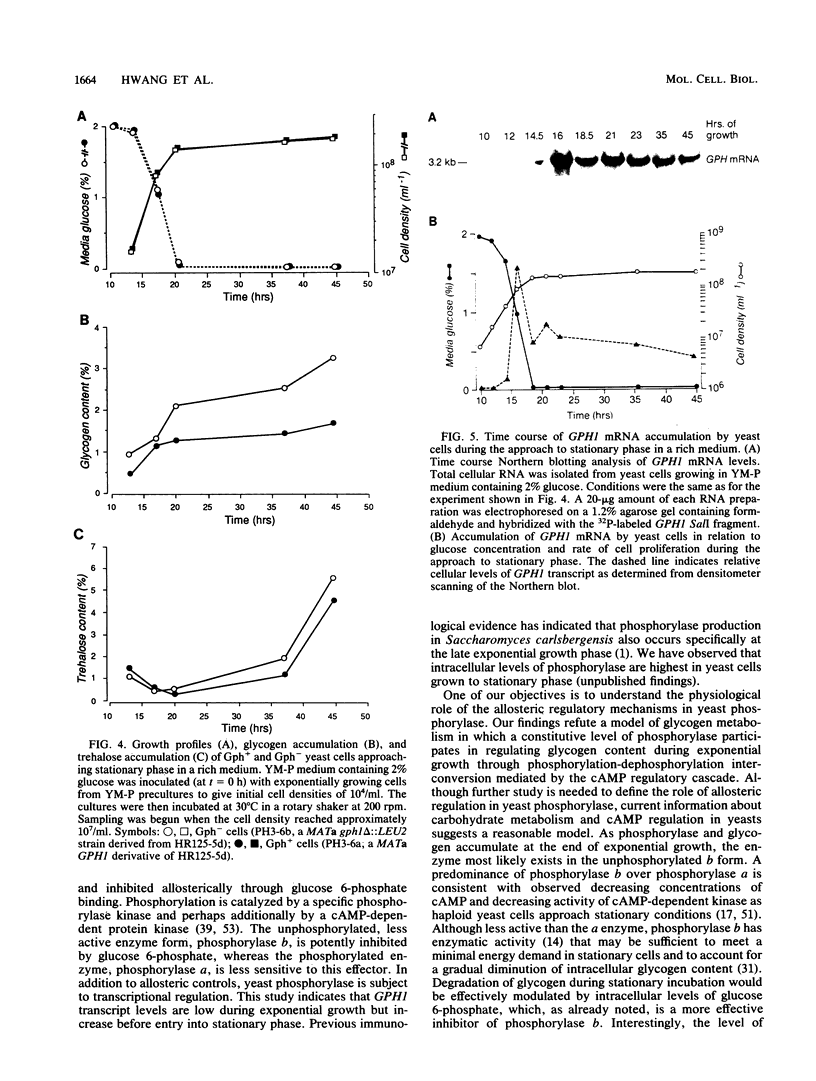
In yeast cells, the activity of glycogen phosphorylase is regulated by cyclic AMP-mediated phosphorylation of the enzyme. We have previously cloned the gene for glycogen phosphorylase (GPH1) in Saccharomyces cerevisiae. To assess the role of glycogen and phosphorylase-catalyzed glycogenolysis in the yeast life cycle, yeast strains lacking a functional GPH1 gene or containing multiple copies of the gene were constructed. GPH1 was found not to be an essential gene in yeast cells. Haploid cells disrupted in GPH1 lacked phosphorylase activity and attained higher levels of intracellular glycogen but otherwise were similar to wild-type cells. Diploid cells homozygous for the disruption were able to sporulate and give rise to viable ascospores. Absence of functional GPH1 did not impair cells from synthesizing and storing trehalose. Increases in phosphorylase activity of 10- to 40-fold were detected in cells carrying multiple copies of GPH1-containing 2 microns plasmid. Northern (RNA) analysis indicated that GPH1 transcription was induced at the late exponential growth phase, almost simultaneous with the onset of intracellular glycogen accumulation. Thus, the low level of glycogen in exponential cells was not primarily maintained through regulating the phosphorylation state of a constitutive amount of phosphorylase. GPH1 did not appear to be under formal glucose repression, since transcriptional induction occurred well in advance of glucose depletion from the medium.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Becker J. U. Mechanisms of regulation of glycogen phosphorylase activity in Saccharomyces carlsbergensis. J Gen Microbiol. 1982 Mar;128(3):447–454. doi: 10.1099/00221287-128-3-447. [DOI] [PubMed] [Google Scholar]
- Boucherie H. Protein synthesis during transition and stationary phases under glucose limitation in Saccharomyces cerevisiae. J Bacteriol. 1985 Jan;161(1):385–392. doi: 10.1128/jb.161.1.385-392.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Browner M. F., Nakano K., Bang A. G., Fletterick R. J. Human muscle glycogen synthase cDNA sequence: a negatively charged protein with an asymmetric charge distribution. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1443–1447. doi: 10.1073/pnas.86.5.1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carle G. F., Olson M. V. An electrophoretic karyotype for yeast. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3756–3760. doi: 10.1073/pnas.82.11.3756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carney I. T., Beynon R. J., Kay J., Birket N. A semicontinuous assay for glycogen phosphorylase. Anal Biochem. 1978 Mar;85(1):321–324. doi: 10.1016/0003-2697(78)90309-3. [DOI] [PubMed] [Google Scholar]
- Chen G. S., Segel I. H. Purification and properties of glycogen phosphorylase from Escherichia coli. Arch Biochem Biophys. 1968 Sep 20;127(1):175–186. doi: 10.1016/0003-9861(68)90214-2. [DOI] [PubMed] [Google Scholar]
- Church G. M., Gilbert W. Genomic sequencing. Proc Natl Acad Sci U S A. 1984 Apr;81(7):1991–1995. doi: 10.1073/pnas.81.7.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fletterick R. J., Madsen N. B. The structures and related functions of phosphorylase a. Annu Rev Biochem. 1980;49:31–61. doi: 10.1146/annurev.bi.49.070180.000335. [DOI] [PubMed] [Google Scholar]
- Fosset M., Muir L. W., Nielsen L. D., Fischer E. H. Purification and properties of yeast glycogen phosphorylase a and b. Biochemistry. 1971 Oct 26;10(22):4105–4113. doi: 10.1021/bi00798a015. [DOI] [PubMed] [Google Scholar]
- Fraenkel D. G. On ras gene function in yeast. Proc Natl Acad Sci U S A. 1985 Jul;82(14):4740–4744. doi: 10.1073/pnas.82.14.4740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- François J., Eraso P., Gancedo C. Changes in the concentration of cAMP, fructose 2,6-bisphosphate and related metabolites and enzymes in Saccharomyces cerevisiae during growth on glucose. Eur J Biochem. 1987 Apr 15;164(2):369–373. doi: 10.1111/j.1432-1033.1987.tb11067.x. [DOI] [PubMed] [Google Scholar]
- HENION W. F., SUTHERLAND E. W. Immunological differences of phosphorylases. J Biol Chem. 1957 Jan;224(1):477–488. [PubMed] [Google Scholar]
- Hall M. N., Hereford L., Herskowitz I. Targeting of E. coli beta-galactosidase to the nucleus in yeast. Cell. 1984 Apr;36(4):1057–1065. doi: 10.1016/0092-8674(84)90055-2. [DOI] [PubMed] [Google Scholar]
- Hers H. G. The control of glycogen metabolism in the liver. Annu Rev Biochem. 1976;45:167–189. doi: 10.1146/annurev.bi.45.070176.001123. [DOI] [PubMed] [Google Scholar]
- Hodgson C. P., Fisk R. Z. Hybridization probe size control: optimized 'oligolabelling'. Nucleic Acids Res. 1987 Aug 11;15(15):6295–6295. doi: 10.1093/nar/15.15.6295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang K. P., Cabib E. Yeast glycogen synthetase in the glucose 6-phosphate-dependent form. I. Purification and properties. J Biol Chem. 1974 Jun 25;249(12):3851–3857. [PubMed] [Google Scholar]
- Hwang P. K., Fletterick R. J. Convergent and divergent evolution of regulatory sites in eukaryotic phosphorylases. Nature. 1986 Nov 6;324(6092):80–84. doi: 10.1038/324080a0. [DOI] [PubMed] [Google Scholar]
- Ito H., Fukuda Y., Murata K., Kimura A. Transformation of intact yeast cells treated with alkali cations. J Bacteriol. 1983 Jan;153(1):163–168. doi: 10.1128/jb.153.1.163-168.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen R., Sprague G. F., Jr, Herskowitz I. Regulation of yeast mating-type interconversion: feedback control of HO gene expression by the mating-type locus. Proc Natl Acad Sci U S A. 1983 May;80(10):3035–3039. doi: 10.1073/pnas.80.10.3035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kane S. M., Roth R. Carbohydrate metabolism during ascospore development in yeast. J Bacteriol. 1974 Apr;118(1):8–14. doi: 10.1128/jb.118.1.8-14.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krebs E. G., Beavo J. A. Phosphorylation-dephosphorylation of enzymes. Annu Rev Biochem. 1979;48:923–959. doi: 10.1146/annurev.bi.48.070179.004423. [DOI] [PubMed] [Google Scholar]
- Küenzi M. T., Fiechter A. Regulation of carbohydrate composition of Saccharomyces cerevisiae under growth limitation. Arch Mikrobiol. 1972;84(3):254–265. doi: 10.1007/BF00425203. [DOI] [PubMed] [Google Scholar]
- Lerch K., Fischer E. H. Amino acid sequence of two functional sites in yeast glycogen phosphorylase. Biochemistry. 1975 May 6;14(9):2009–2014. doi: 10.1021/bi00680a031. [DOI] [PubMed] [Google Scholar]
- Lillie S. H., Pringle J. R. Reserve carbohydrate metabolism in Saccharomyces cerevisiae: responses to nutrient limitation. J Bacteriol. 1980 Sep;143(3):1384–1394. doi: 10.1128/jb.143.3.1384-1394.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto K., Uno I., Ishikawa T. Control of cell division in Saccharomyces cerevisiae mutants defective in adenylate cyclase and cAMP-dependent protein kinase. Exp Cell Res. 1983 Jun;146(1):151–161. doi: 10.1016/0014-4827(83)90333-6. [DOI] [PubMed] [Google Scholar]
- Nakano K., Fukui T. The complete amino acid sequence of potato alpha-glucan phosphorylase. J Biol Chem. 1986 Jun 25;261(18):8230–8236. [PubMed] [Google Scholar]
- Newgard C. B., Hwang P. K., Fletterick R. J. The family of glycogen phosphorylases: structure and function. Crit Rev Biochem Mol Biol. 1989;24(1):69–99. doi: 10.3109/10409238909082552. [DOI] [PubMed] [Google Scholar]
- Palm D., Goerl R., Burger K. J. Evolution of catalytic and regulatory sites in phosphorylases. Nature. 1985 Feb 7;313(6002):500–502. doi: 10.1038/313500a0. [DOI] [PubMed] [Google Scholar]
- Panek A. D., Sampaio A. L., Braz G. C., Baker S. J., Mattoon J. R. Genetic and metabolic control of trehalose and glycogen synthesis. New relationships between energy reserves, catabolite repression and maltose utilization. Cell Mol Biol Incl Cyto Enzymol. 1979;25(5):345–354. [PubMed] [Google Scholar]
- Pohlig G., Wingender-Drissen R., Becker J. U. Characterization of phosphorylase kinase activities in yeast. Biochem Biophys Res Commun. 1983 Jul 18;114(1):331–338. doi: 10.1016/0006-291x(83)91632-7. [DOI] [PubMed] [Google Scholar]
- Purwin C., Leidig F., Holzer H. Cyclic AMP-dependent phosphorylation of fructose-1,6-bisphosphatase in yeast. Biochem Biophys Res Commun. 1982 Aug 31;107(4):1482–1489. doi: 10.1016/s0006-291x(82)80166-6. [DOI] [PubMed] [Google Scholar]
- Rothstein R. J. One-step gene disruption in yeast. Methods Enzymol. 1983;101:202–211. doi: 10.1016/0076-6879(83)01015-0. [DOI] [PubMed] [Google Scholar]
- Schane H. P. Molecular weight estimation of rat uterine phosphorylase. Anal Biochem. 1965 May;11(2):371–374. doi: 10.1016/0003-2697(65)90025-4. [DOI] [PubMed] [Google Scholar]
- Struhl K. The new yeast genetics. 1983 Sep 29-Oct 5Nature. 305(5933):391–397. doi: 10.1038/305391a0. [DOI] [PubMed] [Google Scholar]
- TREVELYAN W. E., HARRISON J. S. Studies on yeast metabolism. 7. Yeast carbohydrate fractions. Separation from nucleic acid, analysis, and behaviour during anaerobic fermentation. Biochem J. 1956 May;63(1):23–33. doi: 10.1042/bj0630023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatchell K., Robinson L. C., Breitenbach M. RAS2 of Saccharomyces cerevisiae is required for gluconeogenic growth and proper response to nutrient limitation. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3785–3789. doi: 10.1073/pnas.82.11.3785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toda T., Uno I., Ishikawa T., Powers S., Kataoka T., Broek D., Cameron S., Broach J., Matsumoto K., Wigler M. In yeast, RAS proteins are controlling elements of adenylate cyclase. Cell. 1985 Jan;40(1):27–36. doi: 10.1016/0092-8674(85)90305-8. [DOI] [PubMed] [Google Scholar]
- Uno I., Matsumoto K., Adachi K., Ishikawa T. Genetic and biochemical evidence that trehalase is a substrate of cAMP-dependent protein kinase in yeast. J Biol Chem. 1983 Sep 25;258(18):10867–10872. [PubMed] [Google Scholar]
- WOSILAIT W. D., SUTHERLAND E. W. The relationship of epinephrine and glucagon to liver phosphorylase. II. Enzymatic inactivation of liver phosphorylase. J Biol Chem. 1956 Jan;218(1):469–481. [PubMed] [Google Scholar]
- Wingender-Drissen R., Becker J. U. Regulation of yeast phosphorylase by phosphorylase kinase and cAMP-dependent protein kinase. FEBS Lett. 1983 Oct 31;163(1):33–36. doi: 10.1016/0014-5793(83)81156-9. [DOI] [PubMed] [Google Scholar]
- Wingender-Drissen R. Yeast cyclic AMP-dependent protein kinase. FEBS Lett. 1983 Oct 31;163(1):28–32. doi: 10.1016/0014-5793(83)81155-7. [DOI] [PubMed] [Google Scholar]
- Yamashita I., Fukui S. Transcriptional control of the sporulation-specific glucoamylase gene in the yeast Saccharomyces cerevisiae. Mol Cell Biol. 1985 Nov;5(11):3069–3073. doi: 10.1128/mcb.5.11.3069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Plaat J. B. Cyclic 3',5'-adenosine monophosphate stimulates trehalose degradation in baker's yeast. Biochem Biophys Res Commun. 1974 Feb 4;56(3):580–587. doi: 10.1016/0006-291x(74)90643-3. [DOI] [PubMed] [Google Scholar]