Abstract
The mechanisms by which migratory birds achieve their often spectacular navigational performance are still largely unclear, but perception of cues from the Earth's magnetic field is thought to play a role. Birds that possess migratory experience can use map-based navigation, which may involve a receptor that uses ferrimagnetic material for detecting gradients in the magnetic field. Such a mechanism can be experimentally disrupted by applying a strong magnetic pulse that re-magnetizes ferrimagnetic materials. In captivity, this treatment indeed affected bearings of adult but not of naive juvenile birds. However, field studies, which expose birds to various navigational cues, yielded mixed results. Supportive studies were difficult to interpret because they were conducted in spring when all age groups navigate back to breeding areas. The present study, therefore, applied a magnetic pulse treatment in autumn to naturally migrating, radio-tagged European robins. We found that, although overall bearings were seasonally correct, orientation of adult but not juvenile robins was compromised by a pulse. Pulsed adults that departed within 10 days of treatment failed to show significant orientation and deviated more from mean migration direction than adult controls and juveniles. Thus, our data give field-based support for a possible ferrimagnetic map-sense during bird migration.
Keywords: navigation, ferrimagnetic, map, migration, robin
1. Introduction
The cues and mechanisms that migratory passerine birds use to navigate to their breeding and wintering grounds remain hotly debated [1,2]. The most straight-forward approach to this question is the study of migrants in autumn, when juvenile birds are still naive while adults can use information based on previous journeys. Evidence suggests that, in the majority of cases, juveniles making their first autumn journey migrate in a population-specific compass direction that is genetically controlled and seasonally appropriate. By contrast, adults additionally call upon a mechanism that allows them to correct for displacements from the normal path [3,4]. This mechanism has been described as a ‘map’ [5,6] and is thought to be based on environmental gradients that are learned during the first migratory journey [7], possibly forming a bi-coordinate grid [8]. A number of environmental cues have been proposed as candidates for such a map, but, as yet, conclusive evidence for a bi-coordinate navigational map remains elusive [2]. One cue that has been proposed as a potential gradient for navigation is the intensity of the Earth's magnetic field, which varies latitudinally from poles to equator, and in some places longitudinally [7]. Birds have been demonstrated to respond to changes in the magnetic field [9] and possibly possess two sensory pathways by which they detect the magnetic field. The ‘radical pair’ mechanism proposes that the Earth's magnetic field alters the unpaired electron spin state of photoreceptive chemicals in the eye, most likely cryptochromes [10–14]. It has been demonstrated that this sensory pathway is involved in compass orientation in passerine birds, but there is no evidence for a role in the map step [15].
A second proposed mechanism of magnetoreception is based on a receptor that uses ferrimagnetic particles [16] and that is innervated by the ophthalmic branch of the trigeminal nerve [17–19]. One such putative sensory receptor was located in the upper beak of homing pigeons [20,21] and other birds, including European robins, Erithacus rubecula [22]. This sensory pathway is apparently not involved in seasonally appropriate compass orientation [15], and recent evidence has questioned whether the particles identified are indeed magnetosensory cells [23]. An alternative site for the ferrimagnetic receptor has been proposed to be the inner ear lagena, which is part of the vestibular system in the ear. The proposition is based on neurological evidence that lesions of this area eliminated responses in regions of the brain that are activated by magnetic activity [24]. Recent evidence suggests that vestibular brainstem neurons encode direction, intensity and polarity of the magnetic field [25], but these studies do not test the role of the sense in navigation behaviour. However, an intriguing line of evidence suggests a possible role of the ferrimagnetic sense in the navigational map of migratory songbirds. Previous experiments on magnetotactic bacteria had indicated that a brief, strong magnetic pulse (greater than 0.1 T) re-magnetizes the magnetic material and leads to incorrect orientation [26]. Based on these findings, it was proposed that, if birds used a ferrimagnetic sensory pathway to detect the magnetic field, they should likewise orient incorrectly after treatment with a magnetic pulse. This idea was partly supported by tests on passerine birds in Emlen funnels [27–30]. Interestingly, the effect is only seen in adult birds, not juveniles, whose autumnal, seasonally appropriate compass orientation remained unaffected [31]. Because adult but not juvenile birds can rely on a map, it has been argued that the pulse is affecting a receptor that is involved in detecting the Earth's magnetic field to be used specifically in the navigational map mechanism.
For technical reasons, the role of magnetic cues in the navigation mechanisms of naturally migrating birds had been poorly studied, but, recently, tests of magnetic pulses on naturally migrating robins and reed warblers (Acrocephalus scirpaceus) indicated an effect on departure direction [32]. This suggested a role for the ferrimagnetic sense, but, in spring, both adults and juveniles are expected to be navigating back to familiar breeding grounds based on an experience derived map [33]. Therefore, the experiment could not distinguish between an effect of the magnetic pulse on a map or a compass mechanism. To clarify this question, the present study was carried out at the same European stopover site during autumn. The study examines the effect of magnetic pulses on the departure directions of naturally migrating adult and juvenile European robins (i) to test whether the ferrimagnetic sense plays a role in autumn migration and (ii) to compare orientation of juvenile and adult birds to interpret possible effects of magnetic pulses. We predict that if the ferrimagnetic sense plays a role in the navigational map then adult but not juvenile birds will either be deflected or show increased scatter in their departure directions.
2. Material and methods
2.1. Subjects
European robins (Erithacus rubecula) are common passage migrants at our study site, the Mettnau Peninsula at Lake Constance in southern Germany (47.7° N, 9.0° E, near Radolfzell) [34]. Robins in central Europe are partial migrants [35] that are known to use genetically inherited migration programmes [36] and are, therefore, commonly used in navigation studies [37]. The previous study of magnetic pulses which is hereby extended has successfully used robins under similar experimental conditions [32]. In the current study, robins were caught by mist netting at the Mettnau monitoring station from 8 September until 21 October 2010. Birds were ringed, weighed, checked for moult, and scored for migratory fattening and state of breast muscle [38]. We also collected back feathers (a side product of trimming for transmitter attachment) for subsequent isotope analysis in related projects and measured tarsus length, wing length and length of the 3rd primary [39]. In autumn distinction between adult and juvenile robins is generally possible based on characteristics summarized, for example, by Svensson [39] and Jenni & Winkler [40]. In cases in which characters were too intermediate to allow unambiguous ageing, or in which physical condition was poor (as based on visible breast muscle), the bird was released after ringing. In total, 87 birds (46 adults, 41 juveniles) were retained in the experiment. Birds were alternately assigned to experimental and control ‘sham’ pulse treatments.
2.2. Experimental treatments
Immediately after ringing and measurements birds were treated with one of two treatments using a magnetic pulse device. The pulse device was a silicon-controlled rectifier-fired capacitive discharge unit (a SOTA magnetic pulser) modified by the addition of a double-wrapped, 10 cm diameter Lee Whittling coil [41]. The coil system produced a unidirectional magnetic pulse of approximately 0.1 ms duration, with a peak amplitude slightly over 0.1 T, and a rise time of approximately 100 ns. A pair of fine wire Helmholtz coils produced a 320 μT biasing field that could be aligned parallel or anti-parallel to the pulse direction. The pulse device was aligned on the magnetic east–west axis. Experimental birds (adults, n = 24; juveniles, n = 16) were treated south anterior with a pulse anti-parallel to the artificial biasing field (figure 1). Control birds (adults, n = 22; juveniles, n = 25) were put into the coil in the same way as experimental birds with the biasing field on, but received a ‘sham’ treatment in which, although the pulse device fired, no current was directed into the pulse coil and so the bird did not receive a magnetic treatment [41].
Figure 1.
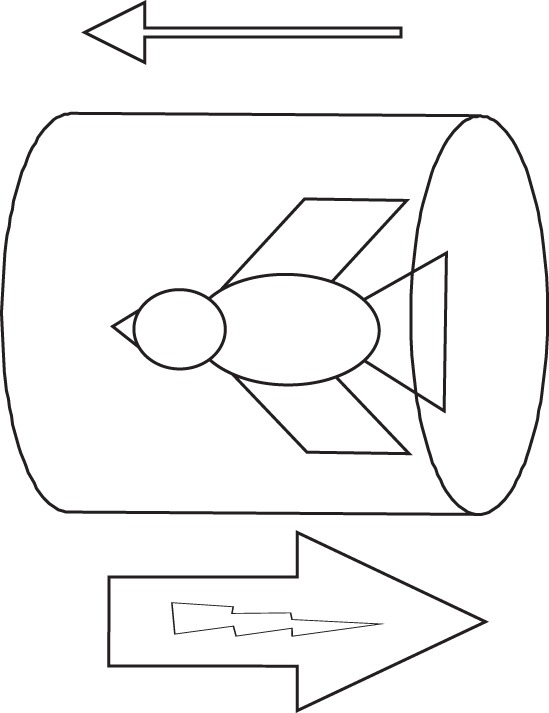
Treatment of experimental birds. Application of a magnetic pulse to illustrate the alignment of the bird to the pulse (large arrow with lightning symbol below coil) and biasing field (small arrow above coil) under the experimental condition. Controls were treated in the same way but no current was directed into the pulse coil.
2.3. Data collection
After treatment, birds were fitted with a radio transmitter (66 × 0.4 g Holohil Systems Ltd LB2N and 21 × 0.36 g Biotrack NTQB-2). The transmitter was sewn to a square of cotton cloth and the cloth was attached to the birds' back using latex-based eyelash glue. This method has been demonstrated to allow the transmitter to stay attached for approximately 24 days, which is longer than the 14–21 day battery life of the transmitter [42]. Transmitters produce an electromagnetic field that has the potential to disrupt magnetic information if not considered, particularly if negative results are obtained [43]. However, previous measurements of these transmitters indicate no measurable electromagnetic field 2 cm from its centre ([4]; the approximate distance of the transmitter to the bird's head), and magnetic treatments have previously been shown to be effective on birds and bats using similar transmitters [32,44,45]. Once the glue had dried the bird was immediately released back onto the Mettnau Peninsula. The signals were monitored from two automated radio receiver units (ARU; Sparrow Systems) placed on a 30 m tower at the Mettnau Peninsula. These devices scanned through the frequencies programmed into the receiver sequentially and logged the signal strength received. Each ARU had four 4-element yagi antennae attached to it, and the antennae pointed approximately 90° to each other, thus covering the cardinal compass directions. The antennae of the two units were fixed one directly above the other such that each of the four antennae for both ARUs pointed in the same direction as each other. No more than 10 birds were monitored by a single ARU at any time. Data were collected on flash cards that were transferred out each time the ARU program was changed and were saved as a comma separated value file.
For determining migratory departure direction (i.e. vanishing bearing), we first identified the time of departure from constant data recording. A migratory departure could be detected by observing a graphical trace of the signal strength of the antennae and determining the point at which the signal dropped to a level which indicated that only noise was being received on all four antennae. The signal for each bird was observed over the course of the time it was present at the Mettnau, and was also checked for 3 days after the determined departure date to make sure that it had not returned. In most cases, departure occurred during night time, in keeping with predominantly nocturnal migration of robins [46]. The point immediately prior to the drop to noise level was then used to calculate the bearing of the departure by comparing the relative signal strength (ratio of signal strength to noise) of the two strongest antennae. This was done by calculating a mean vector from the bearings of these two antennae by vector addition [47] weighted by the relative signal strength of each of these antennae. Bearings were calculated with only the transmitter number (a six-digit number) available as an identifier and as a consequence the data were analysed blind to the treatment condition of the bird. This procedure was established by Holland [32] and was demonstrated to provide results comparable to bearings taken with a handheld antenna and receiver. Because the transmitter life was predicted to be 21 days, any bearing that was obtained after this period was not included in the analysis, as it would be more likely that the reduction in signal was the result of battery failure. Using this procedure 65 bearings were obtained, four bearings were rejected owing to the bird taking longer than 21 days to leave and 18 bearings were lost through ARU battery failure.
2.4. Statistical analysis
2.4.1. Orientation performance
Circular statistics were calculated in Oriana v. 4.0 (Kovach Computing). Mean departure bearing and vector length (r, a measure of concentration of the angles in the sample) were calculated for the age-specific treatment groups: adult control (AC), adult anti-parallel pulsed (AAP), juvenile control (JC) and juvenile anti-parallel pulsed (JAP). We tested for orientation within each group by Rayleigh test and compared the mean bearings of groups using the Watson–Williams test (WW). We then examined differences in variance between groups as a proxy for disruption of navigational accuracy. For this, we transformed the original bearings to deviations from group-specific, circular means (calculated as the absolute difference of a given bearing from the pertinent, age-specific treatment group mean). These deviations were then compared using Levene's test, a test for the equality of variances between groups [48], which does not assume underlying normality of the data. Previous data from orientation cage tests have shown that the pulse effect is temporary, with orientation returning to the seasonally appropriate direction 10 days after treatment [29,49]. We thus performed circular–linear correlations between bearings and time after treatment to test for any effect of time between treatment and departure on the orientation of the groups, using Oriana v. 4.0. We also compared orientation and scatter of birds within the control and experimental groups which departed within 10 days from treatment and after 10 days from treatment, respectively.
2.4.2. Environmental conditions and physical characteristics
Unlike in an orientation cage, or in a displacement experiment in which the animals attempt to home immediately, we released birds directly at the capture site. Hence, there was no experimental control over their decision as to when to migrate, and, consequently, over the consistency of physiological state and of environmental cues that birds faced. Many passerines, and European robins in particular, are sensitive to environmental conditions and state during migratory departure [46,50,51]. To account for this, we investigated the effects of known environmental and physical factors on the bearings of the birds that could have been independent of or additional to the experimental treatments [46,50,51]. See the electronic supplementary material for details of the method of analysis.
2.4.3. Wind
It has been demonstrated that take-off bearings are influenced by winds of speeds as low as 4 m s−1, or 3 on the Beaufort scale [52]. Only two of our birds took off in winds as strong as 3.4 m s−1 (bottom of 3 on the Beaufort scale). A total of 53 per cent departed in winds of 0.3–1.5 m s−1 (1 on the Beaufort scale) and 43 per cent took off in winds of 1.6–3.4 m s−1 (2 on the Beaufort scale). These wind speeds were lower than those known to influence bearings, but we nonetheless scrutinized our data for possible effects of wind on departures. In order to analyse the effects of wind on departure, headings (intended direction without wind drift) were calculated (see the electronic supplementary material). As the analysis gave no indication that different groups took off in different wind directions, or that there was any difference between the headings and the bearings, all subsequent analyses were performed on bearings.
3. Results
The departure directions of adult and juvenile birds are shown in figure 2. All groups were significantly oriented (table 1). There was no difference between the mean bearings of adult experimental and control birds or juvenile experimental and control birds (WW; table 1). In contrast, after treatment with an anti-parallel pulse, precision of orientation was significantly reduced in adult but not in juvenile robins in the experimental groups relative to respective control birds (Levene's test; table 1).
Figure 2.
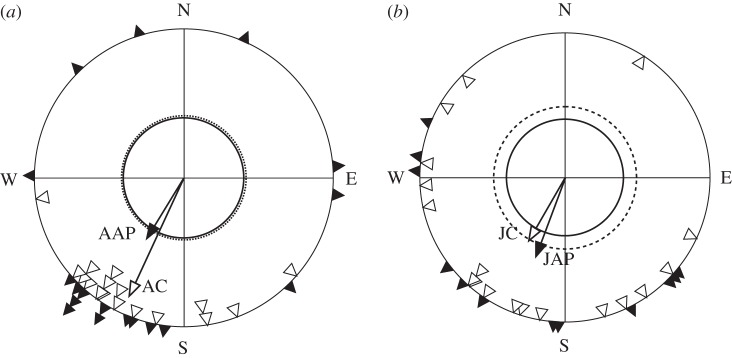
(a) Departure directions of adult and (b) juvenile robins in relation to type of treatment. Open triangles, control; filled triangles, anti-parallel pulsed birds. Arrows represent mean bearing and vector length. Inner circles (solid for anti-parallel pulsed birds, dotted for controls) represent the vector length required for significant orientation by the Rayleigh test at p = 0.05.
Table 1.
Summary of statistical tests of orientation behaviour of adult and juvenile birds. Results for all birds (rows 2–5) and divided by time between treatment and departure, i.e. less than or greater than 10 days (rows 7–11), are shown. AC, adult control; AAP, adult anti-parallel pulsed birds; JC, juvenile control; JAP, juvenile anti-parallel pulsed birds. The length of the mean vector (r) and direction (α) are reported. Differences in mean angular direction (Watson–Williams test) and variance around the mean direction (Levene's test) are reported.
| treatment | n | r | α (°) | Watson–Williams test | Levene's test |
|---|---|---|---|---|---|
| AC | 17 | 0.872*** | 204 | F1,33 = 0.119n.s. | F1,33 = 23.84** |
| AAP | 18 | 0.475* | 211 | ||
| JC | 18 | 0.501* | 210 | F1,28 = 0.129n.s. | F1,28 = 1.187n.s. |
| JAP | 12 | 0.578* | 201 | ||
| AC < 10 | 11 | 0.899*** | 209 | F1,21 = 0.248n.s. | F1,21 = 28.917** |
| AAP < 10 | 12 | 0.287n.s. | 223 | ||
| AC > 10 | 6 | 0.830** | 198 | F1,10 = 0.201n.s. | F1,10 = 0.034n.s. |
| AAP > 10 | 6 | 0.871** | 204 | ||
| JC < 10 | 14 | 0.508* | 213 | F1,23 = 0.456n.s. | F1,23 = 0.116n.s. |
| JAP < 10 | 11 | 0.617* | 192 |
*p < 0.05.
**p < 0.01.
***p < 0.001.
n.s. = p > 0.05.
Neither type of treatment nor age nor their interaction affected the length of time a bird remained at the staging site after treatment (restricted maximum likelihood (REML), Wald on d.f. = 1; for all factors, p > 0.15). However, there was a significant correlation between mean vector length and days between treatment and departure in the AAP group (circular–linear correlation: r = 0.578, p = 0.008). The longer after treatment the bird departed, the less it deviated from the overall group mean. In contrast, mean vector length in adult and juvenile controls was unaffected by time after treatment (circular–linear correlation: adults, r = 0.298, p = 0.263; juveniles, r = 0.256, p = 0.572) and in JAP birds (r = 0.422, p = 0.198).
Figure 3 shows a more detailed analysis of the directionality of birds depending on time after treatment, separated by whether they departed before or after the reported end of treatment effects after 10 days. Only one juvenile pulsed bird departed more than 10 days after treatment and so no analysis was performed for this group. All tested groups were significantly oriented except experimental adults which departed within 10 days after treatment (table 1). There was no significant difference between the mean bearings of the groups; however, there was a significant difference between the residual values of adult experimental compared with adult control birds departing within 10 days after treatment (Levene's test; figure 3a), but not between any other groups (table 1). Thus, adult robins departing within 10 days after receiving an anti-parallel pulse were thus distinct from other groups by lack of significant orientation and by heightened variation compared with controls (figure 3a).
Figure 3.
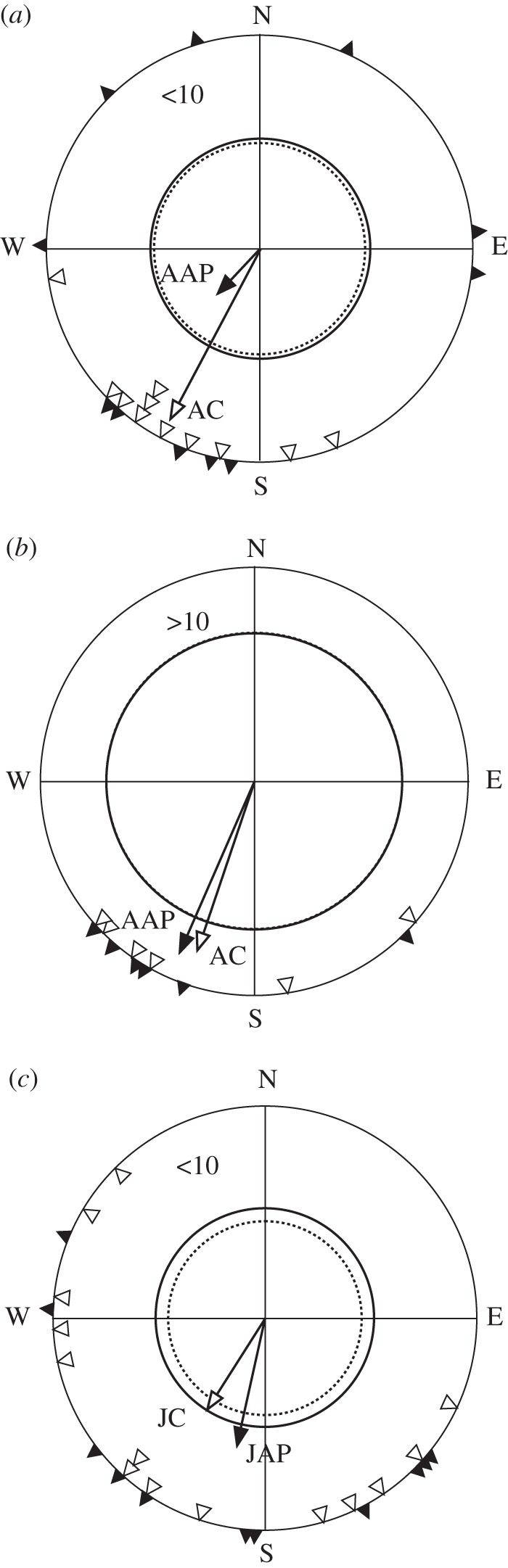
Bearings during and after the time of effective treatments. (a) Adults which departed within 10 days after treatment, (b) adults which departed more than 10 days after treatment and (c) juveniles which departed within 10 days after treatment. Key as for figure 2 (experimental (anti-parallel pulse), filled triangle; control, open triangle).
We also analysed the effects of additional factors on precision of migratory directions (i.e. on absolute deviation from age-specific mean group departure direction). While taking account of age, treatment type and days from treatment, the only factor that significantly influenced precision was the date of capture. The later in a year a bird was examined, the more precise was its choice of direction upon migratory departure (REML, Wald = 5.79 on d.f. = 1; p = 0.016; for all other factors, p > 0.15). All other investigated factors were not significant (see the electronic supplementary material).
4. Discussion
4.1. Main findings
The findings of our experimental study on naturally migrating robins support a putative ferrimagnetic map-sense during bird migration. Application of a strong, magnetic pulse affected departure direction of experienced adult, but not of naive juvenile birds during effective treatment time. These results are in overall agreement with findings in captivity. When Australian silvereyes (Zosterops lateralis) were tested in an orientation cage, the only evidence for an effect of the treatment was on experienced adult birds [31]. In that study, pulse-treated adults reoriented by approximately 90°. However, this reoriented behaviour was only present in the first 2–3 days, following which pulsed birds tested in orientation cages become disoriented. A previous captive study of pulsed robins in spring, which also produced disorientation in the treated group, speculated that this was due to different individuals having different final goals and thus interpreting the altered magnetic information differently [53]. Given the high precision of the control group in our study, this seems an unlikely explanation for our results, and we hypothesize that, for the robins in our study, available natural, additional navigational cues may have resulted in cue-conflict, possibly resulting in a bearing that was a compromise between two conflicting cues for some birds. Surprisingly, we found little direct evidence for effects of environment and physical state on the birds' navigational performance. The only factors that affected bearings were age, with greater precision in adult than in juvenile robins, and date, with increasing precision as autumn progressed. None of the weather variables or body characteristics that we considered had any effect on the birds' directional choice.
4.2. Effect of time between treatment and departure
Detailed analysis of magnetic pulse treatment effects yielded further insights. Again agreeing with data from silvereyes, the pulse effect on robins seems to be dependent on the number of days between treatment and departure. As in orientation cage studies [29,49], pulsed birds were only disoriented if their bearing was obtained within 10 days after treatment. After this time period, the pulsed group was not different from controls. Time dependence was not seen in the spring migration of robins and reed warblers at the same site [32], most likely because the mean departure time was much shorter in that study, with very few birds departing more than 10 days after treatment. Temporary effects of pulse treatment may also explain the lack of an effect of a pulse on migrating catbirds (Dumetella carolinensis) in autumn in a previous study, because most of these birds departed more than 14 days after treatment [54]. However, direct comparison of this experiment with the current study is difficult because it had used different orientation measures (final bearing obtained at greater distance and time after initial departure by tracking from air). Conversely, the temporary nature of the pulse does not fit with hypotheses on the nature of the ferrimagnetic material proposed to play a role in the sensory receptor [30]. If the material is single domain magnetite with a permanent magnetic moment, the strong magnetic pulse should re-magnetize it permanently in the opposite direction. The unexpectedly temporary effects of the treatment could result from the eventual downgrading of faulty magnetic information in the hierarchy of cues, from recalibration, or from repair of the sensory system. It is possible that the disorientation seen in our experiment was a result of conflict with other navigational cues (e.g. visual landmarks, olfactory cues), and that the lack of effect greater than 10 days after treatment was because of a reversion to those alternative cues. However, in Wiltschko et al. [49] a second pulse treatment was applied after observable effects of the first pulse had disappeared, resulting in an increase in scatter in the treated group. If the magnetic information was downgraded in the hierarchy, then an effect of the subsequent pulse would not be expected, which argues against this explanation for the temporary nature of the pulse. Another possible explanation is that if the magnetoreceptor is based on superparamagnetic material [20], which does not have a permanent magnetic moment and thus cannot be re-magnetized, then this might cause temporary disruption of the sensory system [30], although it remains unclear whether such a receptor structure exists [23].
4.3. Does a magnetic pulse affect a magnetoreceptor?
It has been argued that the pulse treatment affects a sensory receptor based on ferrimagnetic material [26]. Existence and location of such a receptor is still disputed and mostly rests on two lines of evidence, one being histological and the other neurological. Previous studies suggested that the magnetoreceptors were located in the beak area [20,22,55]. These studies had identified iron clusters in the beak that were thought to be functionally related to magnetoreception. However, recent evidence arguing that the iron clusters identified by Fleissner et al. [20] may be macrophages [23] brings this interpretation into question. In contrast, support for this hypothesis comes from the observation that, after local anaesthetic was applied to the beak area, pulse treatments had no effect [56]. The role of the trigeminal nerve in magnetoreception [17,18], which innervates the beak, does still argue for magnetoreceptors located in this area. Treiber et al. [23] propose that, similarly to the rainbow trout [57], the olfactory mucosa is a potential location. Single domain magnetic particles have previously been identified in the ethmoid region and the nasal cavity of birds [58–60].
The second line of evidence that the pulse affects magnetoreceptors is based on neurological studies. A conditioning study demonstrates that the trigeminal nerve is responsible for conveying magnetic intensity information [18]. Bobolinks (Dolichonyx oryzivorus) which were deflected by a pulse treatment reverted to the species-specific migratory direction when this nerve was blocked directly with a local anaesthetic [19], suggesting that the pulse effects trigeminally mediated magnetoreception (although see [61] for criticisms of local anaesthetic treatments in this context). This study remains the only direct evidence that the trigeminal nerve plays a role in navigation behaviour, with all other attempts to disrupt navigation in birds by trigeminal nerve section failing to affect the initial orientation or homing performance of pigeons [62–64]. An alternative location for ferrimagnetic magnetoreception has been proposed to be the inner ear lagena [65]. In accordance with this idea, recent neuroanatomical evidence indicates that brain areas that are innervated by vestibular neurons in pigeons are activated by magnetic cues [25]. Ablation of the lagena eliminates activation of these regions [24]. However, as with the trigeminal nerve, evidence for a role of this mechanism in navigation is equivocal. While one study suggests disruption of navigation performance in pigeons with the inner ear ablated [65], an earlier comprehensive study with the same treatment found no effect on initial orientation or homing performance ([66]; see [67] for discussion of the limitations of the Harada et al. [65] study).
In addition to these ideas, the magnetic pulse treatment may also affect other systems that are influenced by electromagnetic fields. Given that transcranial magnetic stimulation in humans, using such pulses, results in non-specific behavioural cognitive deficits [68], this possibility should also be considered as an alternative to a putative ferrimagnetic mechanism. It is noteworthy that the pulse affects adult but not juvenile birds [31], and that it also depends on the orientation of the pulse. Previous research shows different effects depending on the treatment, with reorientation depending on the alignment of the pulse [27,32]. This would argue against a non-specific effect and does argue in favour of a magnetic receptor being disrupted.
4.4. What aspect of navigation is affected?
If the magnetic pulse is affecting a magnetic receptor, what aspect of the navigation mechanism is it affecting? None of the previous work indicates that a pulse disrupts normal compass orientation in juvenile or adult migrating birds [18,19,31,69]. Instead, the specificity of effects on experienced adult, and not naive juvenile, migrants supports the interpretation of disruption of a possible map-sense. A recent study on migratory Australian silvereyes (Zosterops l. lateralis) that simulated displacement by changing magnetic intensity signatures indicated that only adult, not juvenile, migrants were affected by the displacement [70]. However, the existence of a magnetic map continues to be strongly debated [1,2,7]. Given that neither the current experiment nor those carried out in orientation cages use displacement outside the normal migratory route as part of the design, it cannot be entirely certain that the adults are indeed accessing a true navigational map based on cues experienced outside the familiar migration route [71]. Although arguably less parsimonious given the evidence in [29,30,32,68], an alternative navigational explanation could include different roles of the ferrimagnetic sense in the adult and juvenile magnetic compass systems, as some have suggested that the ferrimagnetic sense may play an as yet unidentified role in the avian magnetic compass [61,72]. Magnetic cues have also been shown to play a role in ‘signposting’ latitudes at which birds should refuel to prepare for energetically demanding crossings of geographical barriers [73]. It is, therefore, possible that the pulse affects the adults' ability to recognize the latitude at which refuelling is necessary, perhaps resulting in a lack of oriented behaviour (owing to a desire to refuel at the stopover site rather than make a directed migration flight).
4.5. Conclusion
With this paper there is now independent evidence that a strong magnetic pulse affects the orientation of migrating birds in both the laboratory and the field and affects an aspect of migratory behaviour that is expressed by adult but not juvenile birds. This suggests that a magnetoreceptor with ferrimagnetic properties plays some role in experience-based migratory orientation in songbirds. The challenge now is for clear links to be made between the behavioural aspects of magnetoreception and the neurobiology, molecular biology and physics of magnetoreception, to describe how the putative receptors are altered to produce the behavioural effects seen in nature.
Acknowledgements
We are indebted to Heidi Schmid, Wolfgang Fiedler, Kim Mortega, Marina Lehmann and the many volunteers who helped out at the ringing station. Martin Wikelski supported the project by additional monitoring of birds from an aeroplane. The authors thank the Max Planck Society, the NSF, the Young Scholar Fund of Konstanz University and the European Social Fund in Baden-Wuerttemberg for financial support.
References
- 1.Alerstam T. 2006. Conflicting evidence about long-distance animal navigation. Science 313, 791–794 10.1126/science.1129048 (doi:10.1126/science.1129048) [DOI] [PubMed] [Google Scholar]
- 2.Thorup K, Holland R. 2009. The bird GPS. J. Exp. Biol. 212, 3597–3604 10.1242/jeb.021238 (doi:10.1242/jeb.021238) [DOI] [PubMed] [Google Scholar]
- 3.Perdeck AC. 1958. Two types of orientation in migrating Sturnus vulgaris and Fringilla coelebs. Ardea 46, 1–37 [Google Scholar]
- 4.Thorup K, Bisson I, Bowlin M, Holland RA, Ramenofsky M, Wingfield J, Wikelski M. 2007. Migration routes of adult and juvenile white-crowned sparrows differ after continent-wide displacement during migration. Proc. Natl Acad. Sci. USA 104, 18 115–18 119 10.1073/pnas.0704734104 (doi:10.1073/pnas.0704734104) [DOI] [Google Scholar]
- 5.Kramer G. 1953. Wird die Sonnehöhe bei der Heimfindeorientierung verwertet? J. Orn. 94, 201–219 10.1007/BF01922508 (doi:10.1007/BF01922508) [DOI] [Google Scholar]
- 6.Wiltschko R, Wiltschko W. 2003. Avian navigation: from historical to modern concepts. Anim. Behav. 65, 257–272 10.1006/anbe.2003.2054 (doi:10.1006/anbe.2003.2054) [DOI] [Google Scholar]
- 7.Phillips JB, Schmidt-Koenig K, Muheim R. 2006. True navigation: sensory bases of gradient maps. In Animal spatial cognition: comparative, neural, and computational approaches (eds Brown MF, Cook RG.). Comparative Cognition Press. See http://www.pigeon.psy.tufts.edu/asc/phillips/ [Google Scholar]
- 8.Wallraff HG. 1974. Das Navigationssystem der Vögel. Munich, Germany: R. Oldenbourg [Google Scholar]
- 9.Wiltschko R, Wiltschko W. 1995. Magnetic orientation in animals. Berlin, Germany: Springer [Google Scholar]
- 10.Ritz T, Adem S, Schulten K. 2000. A model for vision based magnetoreception in birds. Biophys. J. 78, 707–718 10.1016/S0006-3495(00)76629-X (doi:10.1016/S0006-3495(00)76629-X) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rogers CT, Hore PJ. 2009. Chemical magnetoreception in birds: the radical pair mechanism. Proc. Natl Acad. Sci. USA 106, 353–360 10.1073/pnas.0711968106 (doi:10.1073/pnas.0711968106) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schulten K, Swenberg CE, Weller A. 1978. A biomagnetic sensory mechanism based on magnetic field modulated coherent electron spin motion. Z. Phys. Chem. NF111, 1–5 10.1524/zpch.1978.111.1.001 (doi:10.1524/zpch.1978.111.1.001) [DOI] [Google Scholar]
- 13.Schulten K, Windemuth A. 1986. Model for a physiological magnetic compass. In Biophysical effects of steady magnetic fields (eds Maret G, Boccara N, Kiepenheuer J.), pp. 99–106 Berlin, Germany: Springer [Google Scholar]
- 14.Liedvogel M, Maeda K, Henbest K, Schleicher E, Simon T, Timmel CR, Hore PJ, Mouritsen H. 2007. Chemical magnetoreception: bird cryptochrome 1a is excited by blue light and forms long-lived radical-pairs. PLoS ONE 2, e1106. 10.1371/journal.pone.0001106 (doi:10.1371/journal.pone.0001106) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zapka M, et al. 2009. Visual but not trigeminal mediation of magnetic compass information in a migratory bird. Nature 461, 1274–1277 10.1038/nature08528 (doi:10.1038/nature08528) [DOI] [PubMed] [Google Scholar]
- 16.Kirschvink JL, Gould JL. 1981. Biogenetic magnetite as a basis for magnetic field detection in animals. BioSystems 13, 181–201 10.1016/0303-2647(81)90060-5 (doi:10.1016/0303-2647(81)90060-5) [DOI] [PubMed] [Google Scholar]
- 17.Heyers D, Zapka M, Hoffmeister M, Wild JM, Mouritsen H. 2010. Magnetic field changes activate the trigeminal brainstem complex in a migratory bird. Proc. Natl Acad. Sci. USA 107, 9394–9399 10.1073/pnas.0907068107 (doi:10.1073/pnas.0907068107) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mora CV, Davison M, Wild JM, Walker MM. 2004. Magnetoreception and its trigeminal mediation in the homing pigeon. Nature 432, 508–511 10.1038/nature03077 (doi:10.1038/nature03077) [DOI] [PubMed] [Google Scholar]
- 19.Beason RC, Semm P. 1996. Does the avian ophthalmic nerve carry magnetic navigational information? J. Exp. Biol. 199, 1241–1244 [DOI] [PubMed] [Google Scholar]
- 20.Fleissner G, Holtkamp-Rotzler E, Hanzlik M, Winklhofer M, Petersen N, Wiltschko W. 2003. Ultrastructural analysis of a putative magnetoreceptor in the beak of homing pigeons. J. Comp. Neurol. 458, 350–360 10.1002/cne.10579 (doi:10.1002/cne.10579) [DOI] [PubMed] [Google Scholar]
- 21.Fleissner G, Stahl B, Thalau P, Falkenberg G, Fleissner G. 2007. A novel concept of Fe-mineral-based magnetoreception: histological and physicochemical data from the upper beak of homing pigeons. Naturwiss 91, 632–641 [DOI] [PubMed] [Google Scholar]
- 22.Falkenberg G, Fleissner G, Schuchardt K, Kuehbacher M, Thalau P, Mouritsen H, Heyers D, Wellenreuther G, Fleissner G. 2010. Avian magnetoreception: elaborate iron mineral containing dendrites in the upper beak seem to be a common feature of birds. PLoS ONE 5, e9231. 10.1371/journal.pone.0009231 (doi:10.1371/journal.pone.0009231) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Treiber CD, et al. 2012. Clusters of iron-rich cells in the upper beak of pigeons are macrophages not magnetosensitive neurons. Nature 484, 367–370 10.1038/nature11046 (doi:10.1038/nature11046). [DOI] [PubMed] [Google Scholar]
- 24.Wu L-Q, Dickman JD. 2011. Magnetoreception in an avian brain in part mediated by inner ear lagena. Curr. Biol. 21, 418–423 10.1016/j.cub.2011.01.058 (doi:10.1016/j.cub.2011.01.058) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wu L-Q, Dickman JD. 2012. Neural correlates of a magnetic sense. Science 336, 1054–1057 10.1126/science.1216567 (doi:10.1126/science.1216567) [DOI] [PubMed] [Google Scholar]
- 26.Kirschvink JL. 1982. Birds, bees and magnetism: a new look at the old problem of magnetoreception. Trends Neurosci. 5, 160–167 10.1016/0166-2236(82)90090-X (doi:10.1016/0166-2236(82)90090-X) [DOI] [Google Scholar]
- 27.Beason RC, Dussourd N, Deutschlander ME. 1995. Behavioral evidence for the use of magnetic material in magnetoreception by a migratory bird. J. Exp. Biol. 198, 141–146 [DOI] [PubMed] [Google Scholar]
- 28.Wiltschko W, Munro U, Beason RC, Ford H, Wiltschko R. 1994. A magnetic pulse leads to a temporary deflection in the orientation of migratory birds. Experientia 50, 697–700 10.1007/BF01952877 (doi:10.1007/BF01952877) [DOI] [Google Scholar]
- 29.Wiltschko W, Munro U, Ford H, Wiltschko R. 1998. Effect of a magnetic pulse on the orientation of silvereyes Zosterops l. lateralis, during spring migration. J. Exp. Biol. 201, 3257–3261 [DOI] [PubMed] [Google Scholar]
- 30.Wiltschko W, Munro U, Wiltschko R, Kirschvink JL. 2002. Magnetite-based magnetoreception in birds: the effect of a biasing field and a pulse on migratory behavior. J. Exp. Biol. 205, 3031–3037 [DOI] [PubMed] [Google Scholar]
- 31.Munro U, Munro JA, Phillips JB, Wiltschko R, Wiltschko W. 1997. Evidence for a magnetite-based navigational ‘map’ in birds. Naturwiss 84, 26–28 10.1007/s001140050343 (doi:10.1007/s001140050343) [DOI] [Google Scholar]
- 32.Holland RA. 2010. Differential effects of magnetic pulses on the departure directions of naturally migrating birds. J. R. Soc. Interface 7, 1617–1625 10.1098/rsif.2010.0159 (doi:10.1098/rsif.2010.0159) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chernetsov N, Kishkinev D, Mouritsen H. 2008. A long distance avian migrant compensates for longitudinal displacement during spring migration. Curr. Biol. 18, 188–190 10.1016/j.cub.2008.01.018 (doi:10.1016/j.cub.2008.01.018) [DOI] [PubMed] [Google Scholar]
- 34.Berthold P, Fliege G, Heine G, Querner U, Schlenker R. 1991. Wegzug, Rastverhalten, Biometrie und Mauser von Kleinvögeln in Mitteleuropa. Vogelwarte 36, 72–76 [Google Scholar]
- 35.Adriaensen F, Dhondt A, Matthysen E. 1990. Bird migration. Nature 347, 23. 10.1038/347023b0 (doi:10.1038/347023b0) [DOI] [Google Scholar]
- 36.Biebach H. 1983. Genetic determination of partial migration in the European Robin, Erithacus rubecola. Auk 100, 602–606 [Google Scholar]
- 37.Wiltschko W, Wiltschko R. 1972. Magnetic compass of European robins. Science 176, 62–64 10.1126/science.176.4030.62 (doi:10.1126/science.176.4030.62) [DOI] [PubMed] [Google Scholar]
- 38.Deutsche-Ornithologen-Gesellschaft 2011. Measuring birds: Vögel vermessen. Minden, Germany: Christ Media Natur [Google Scholar]
- 39.Svensson L. 1992. Identification guide to European passerines, 4th edn. Stockholm, Sweden: Rosersberg [Google Scholar]
- 40.Jenni L, Winkler R. 1994. Molt and ageing of European passerines. London, UK: Academic Press [Google Scholar]
- 41.Kirschvink JL. 1992. Uniform magnetic-fields and double-wrapped coil systems—improved techniques for the design of bioelectromagnetic experiments. Bioelectromagnetics 13, 401–411 10.1002/bem.2250130507 (doi:10.1002/bem.2250130507) [DOI] [PubMed] [Google Scholar]
- 42.Raim A. 1978. A radio transmitter attachment for small passerine birds. Bird Banding 49, 326–332 10.2307/4512391 (doi:10.2307/4512391) [DOI] [Google Scholar]
- 43.Freake MJ, Muheim R, Phillips JB. 2006. Magnetic maps in animals: a theory comes of age? Q. Rev. Biol. 81, 327–347 10.1086/511528 (doi:10.1086/511528) [DOI] [PubMed] [Google Scholar]
- 44.Holland RA, Borissov I, Siemers B. 2010. A nocturnal mammal calibrates a magnetic compass by the sun. Proc. Natl Acad. Sci. USA 107, 6941–6945 10.1073/pnas.0912477107 (doi:10.1073/pnas.0912477107) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Holland RA, Kirschvink JL, Doak TG, Wikelski M. 2008. Bats use magnetite to detect the Earth's magnetic field. PLoS ONE 3, e1676. 10.1371/journal.pone.0001676 (doi:10.1371/journal.pone.0001676) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bolshakov CV, Chernetsov N, Mukhin A, Bulyuk VN, Kosarev V, Ktitorov-Leoke D, Tsvey A. 2007. Time of nocturnal departures in European robins, Erithacus rubecula, in relation to celestial cues, season, stopover duration and fat stores. Anim. Behav. 74, 855–865 10.1016/j.anbehav.2006.10.024 (doi:10.1016/j.anbehav.2006.10.024) [DOI] [Google Scholar]
- 47.Batschelet E. 1981. Circular statistics in biology. New York, NY: Academic Press [Google Scholar]
- 48.Levene H. 1960. Robust tests for equality of variances. In Contributions to probability and statistics: essays in honor of Harold Hotelling (ed. Olkin I, Ghurye SG, Hoeffding W, Madow WG, Mann HB.), pp. 278–292 San Fransisco, CA: Stanford University Press [Google Scholar]
- 49.Wiltschko W, Ford H, Munro U, Winklhofer M, Wiltschko R. 2007. Magnetite-based magnetoreception: the effect of repeated pulsing on the orientation of migratory birds. J. Comp. Physiol. 193, 515–522 10.1007/s00359-006-0207-5 (doi:10.1007/s00359-006-0207-5) [DOI] [PubMed] [Google Scholar]
- 50.Schaub M, Liechti F, Jenni L. 2004. Departure of migrating European robins, Erithacus rubecula, from a stopover site in relation to wind and rain. Anim. Behav. 67, 229–237 10.1016/j.anbehav.2003.03.011 (doi:10.1016/j.anbehav.2003.03.011) [DOI] [Google Scholar]
- 51.Ramenofsky M, Cornelius JM, Helm B. 2012. Physiological and behavioural responses of migrants to environmental cues. J. Orn. 153, 181–191 10.1007/s10336-012-0817-3 (doi:10.1007/s10336-012-0817-3) [DOI] [Google Scholar]
- 52.Mouritsen H. 1998. Redstarts, Phoenicurus phoenicurus, can orient in a true-zero magnetic field. Anim. Behav. 55, 1311–1324 10.1006/anbe.1997.0696 (doi:10.1006/anbe.1997.0696) [DOI] [PubMed] [Google Scholar]
- 53.Wiltschko W, Wiltschko R. 1995. Migratory orientation of European robins is affected by the wavelength of light as well as by a magnetic pulse. J. Comp. Physiol. A 177, 363–369 10.1007/BF00192425 (doi:10.1007/BF00192425) [DOI] [Google Scholar]
- 54.Holland RA, Thorup K, Gagliardo A, Bisson I, Knecht E, Mizrahi D, Wikelski M. 2009. Testing the role of sensory systems in the migratory heading of a songbird. J. Exp. Biol. 212, 4065–4071 10.1242/jeb.034504 (doi:10.1242/jeb.034504) [DOI] [PubMed] [Google Scholar]
- 55.Wiltschko R, Wiltschko W. 2013. The magnetite-based receptors in the beak of birds and their role in avian navigation. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 199, 89–98 10.1007/s00359-012-0769-3 (doi:10.1007/s00359-012-0769-3) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wiltschko W, Munro U, Ford H, Wiltschko R. 2009. Avian orientation: the pulse effect is mediated by the magnetite receptors in the upper beak. Proc. R. Soc. B 276, 2227–2232 10.1098/rspb.2009.0050 (doi:10.1098/rspb.2009.0050). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Diebel CE, Proksch R, Green CR, Neilson P, Walker MM. 2000. Magnetite defines a vertebrate magnetoreceptor. Nature 406, 299–302 10.1038/35018561 (doi:10.1038/35018561) [DOI] [PubMed] [Google Scholar]
- 58.Beason RC, Nichols JE. 1984. Magnetic orientation and magnetically sensitive material in a transequatorial migratory bird. Nature 309, 151–153 10.1038/309151a0 (doi:10.1038/309151a0) [DOI] [Google Scholar]
- 59.Williams NM, Wild JM. 2001. Trigeminally innervated iron-containing structures in the beak of homing pigeons, and other birds. Brain Res. 889, 243–246 10.1016/S0006-8993(00)03114-0 (doi:10.1016/S0006-8993(00)03114-0) [DOI] [PubMed] [Google Scholar]
- 60.Beason RC, Brennan WJ. 1986. Natural and induced magnetization in the Bobolink Dolichonyx oryzivorus (Aves, Icteridae). J. Exp. Biol. 125, 49–56 [Google Scholar]
- 61.Mouritsen H, Hore PJ. 2012. The magnetic retina: light-dependent and trigeminal magnetoreception in migratory birds. Curr. Opin. Neurobiol. 22, 343–352 10.1016/j.conb.2012.01.005 (doi:10.1016/j.conb.2012.01.005) [DOI] [PubMed] [Google Scholar]
- 62.Gagliardo A, Ioale P, Savini M, Wild JM. 2006. Having the nerve to home: trigeminal magnetoreceptor versus olfactory mediation of homing in pigeons. J. Exp. Biol. 209, 2888–2892 10.1242/jeb.02313 (doi:10.1242/jeb.02313) [DOI] [PubMed] [Google Scholar]
- 63.Gagliardo A, Ioale P, Savini M, Wild JM. 2008. Navigational abilities of homing pigeons deprived of olfactory or trigeminally mediated magnetic information when young. J. Exp. Biol. 211, 2046–2051 10.1242/jeb.017608 (doi:10.1242/jeb.017608) [DOI] [PubMed] [Google Scholar]
- 64.Gagliardo A, Ioale P, Savini M, Wild JM. 2009. Navigational abilities of adult and experienced homing pigeons deprived of olfactory or trigeminally mediated magnetic information. J. Exp. Biol. 212, 3119–3124 10.1242/jeb.031864 (doi:10.1242/jeb.031864) [DOI] [PubMed] [Google Scholar]
- 65.Harada Y, Taniguchi M, Namatame H, Iida A. 2001. Magnetic materials in otoliths of bird and fish lagena and their function. Acta Otolaryngol. 121, 590. 10.1080/000164801316878872 (doi:10.1080/000164801316878872) [DOI] [PubMed] [Google Scholar]
- 66.Wallraff HG. 1972. Homing of pigeons after extirpation of their cochleae and lagenae. Nat. New Biol. 236, 223–224 10.1038/236223a0 (doi:10.1038/236223a0) [DOI] [PubMed] [Google Scholar]
- 67.Wallraff HG. 2005. Avian navigation: pigeon homing as a paradigm. Berlin, Germany: Springer [Google Scholar]
- 68.Pascual-Leone A, Davey N, Rothwell J, Wassermann EM, Puri BK. 2002. Handbook of transcranial magnetic stimulation. New York, NY: Hodder Arnold [Google Scholar]
- 69.Wiltschko W, Munro U, Ford H, Wiltschko R. 2006. Bird navigation: what type of information does the magnetite-based receptor provide. Proc. R. Soc. B 273, 2815–2820 10.1098/rspb.2006.3651 (doi:10.1098/rspb.2006.3651) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Deutchlander ME, Phillips JB, Munro U. 2012. Age-dependent orientation to magnetically-simulated geographic displacements in migratory Australian silvereyes (Zosterops l. lateralis). Wilson J. Ornithol. 124, 467–477 10.1676/11-043.1 (doi:10.1676/11-043.1) [DOI] [Google Scholar]
- 71.Griffin DR. 1952. Bird navigation. Biol. Rev. 27, 359–400 10.1111/j.1469-185X.1952.tb01509.x (doi:10.1111/j.1469-185X.1952.tb01509.x) [DOI] [Google Scholar]
- 72.Kirschvink JL, Winklhofer M, Walker MM. 2010. Biophysics of magnetic orientation: strengthening the interface between theory and experimental design. J. R. Soc. Interface 7, S179–S191 10.1098/rsif.2009.0491.focus (doi:10.1098/rsif.2009.0491.focus) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fransson T, Jakobsson S, Johansson P, Kullberg C, Lind J, Vallin A. 2001. Bird migration: magnetic cues trigger extensive refuelling. Nature 414, 35–36 10.1038/35102115 (doi:10.1038/35102115) [DOI] [PubMed] [Google Scholar]


