Abstract
The Cdh23erl/erl mice are a novel mouse model for DFNB12 and characterized by progressive hearing loss. In this study, erythropoietin (EPO) was given to the Cdh23erl/erl mice by intraperitoneally injection every other day from P7 for seven weeks. PBS treated or untreated Cdh23erl/erl mice were used as controls. Auditory-evoked brainstem response (ABR) thresholds and distortion product oto-acoustic emission (DPOAE) were measured in the mouse groups at age of 4, 6 and 8 weeks. The results show that EPO can significantly decrease the ABR thresholds in the Cdh23erl/erl mice as compared with those of the untreated mice at stimulus frequencies of click, 8-, 16- and 32- kHz at three time points. Meanwhile, DPOAE amplitudes in the EPO treated Cdh23erl/erl mouse group were significantly higher than those of the untreated groups at f2 frequency of 15,383 Hz at the three time points. Furthermore, the mean percentage of OHC loss at middle through basal turns of cochleae was significantly lower in EPO-treated Cdh23erl/erl mice than in the untreated mice (P < 0.05). This is the first report that EPO acts as an otoprotectant in a DFNB12 mouse model with progressive hearing loss.
Keywords: ABR, DPOAE, erythropoietin, genetics, hearing loss, mouse model
INTRODUCTION
Erythropoietin (EPO) is a cytokine hormone with multiple functions. It is well-known that exogenous administration of the recombinant EPO stimulates erythropoiesis, but EPO also acts on non-haemopoietic cells in a cytoprotective manner in damaged tissues and organs (Ghezzi and Brines, 2004). This is possible because many of the cells in these tissues and organs have EPO receptors (EPORs). Similar to the way that EPO stimulates erythropoiesis by protecting the erythroid progenitor cells from apoptosis, its main cytoprotective mechanism appears to be anti-apoptotic (Vesey et al., 2004, Johnson et al., 2006). EPO can cross the blood-brain barrier and exerts neuroprotective effects (Brines et al., 2000, Maiese et al., 2008). Systemic administration of EPO prevented neuronal apoptosis after a cerebral ischemia in rats (Siren et al., 2001). EPO reduced the apoptosis rate in hippocampal neurons and enhanced the regenerative effect of brain-derived neurotrophic factor (BDNF) on spiral ganglion neurons (Berkingali et al., 2008). Study on guinea pigs revealed that the EPO receptor was widely expressed by several cell types within the cochlea (Caye-Thomasen et al., 2005). In vitro, EPO attenuated hair cell loss in rat cochlear explants that were treated with gentamycin (Monge et al., 2006, Monge Naldi et al., 2009). Experiments also showed recombinant human erythropoietin (rhEPO) prevented ischemia-induced apoptosis and necrosis in explant cultures of the rat organ of Corti (Andreeva et al., 2006). However, there are no reports of EPO on otoprotection in genetic mouse models.
We recently reported that mutation in the Cdh23erl/erl mice lead to hearing loss through apoptosis and that hearing was preserved in pan-caspase inhibitor Z-VAD-FMK-treated mutants compared with untreated mutants (Han et al., 2012). In this study, we further show that hearing in Cdh23erl/erl mice was protected by EPO as evaluated by the measurement of ABR thresholds and DPOAE amplitudes.
EXPERIMENTAL PROCEDURES
Cdh23erl/erl mouse preparation
Cdh23erl/erl mice were used as a model to test anti-apoptotic drug therapy (Han et al., 2012). The studies were conducted in accordance with the principles set for the Guide for the Care and Use of Laboratory Animals, Institute Of Laboratory Animal Resources, and were approved by The Case Western Reserve University of the Health Sciences Institutional Animal Use and Care Committee. Twenty-two Cdh23erl/erl mice at the age of 7 days were randomly divided into 3 groups: an EPO (in 1×PBS, Phosphate-Balanced Saline, pH7.4) treated group (5 mice, n = 10 ears), PBS treated group (5 mice, n = 10 ears) and untreated group (6 mice, n = 12 ears).
EPO administration
Ten micrograms of recombinant mouse EPO (R&D systems, Inc., catalog number, 959-ME-010/CF; Lot Number: EUP0409061) were dissolved in 100 μl sterile PBS (equivalent to 50 U/μl or 100 ng/ μl) and aliquots of 10 μl were stored in −20 C°. The stock solution was diluted 10 times in PBS (5U/μl or 10 ng/ μl) before using. The Cdh23erl/erl mice at age of 7 days in EPO group were injected intraperitoneally under sterile conditions with 5 U of EPO per gram body weight (5 U/g or 10 ng/g) every other day. Administration was stopped when the mice were at 8 weeks of age. The EPO dosage was chosen with the reference to a previous report (Wiese et al., 2008). The starting point was based on the aim of preventing caspase increases, detected as early as P14 in untreated Cdh23erl/erl mice (Han et al., 2012). The Cdh23erl/erl mice in the PBS group received equal amounts of PBS (1 μl/g) at the same time points. ABR and DPOAE were tested at 4, 6, and 8 weeks for all the mice.
Auditory-evoked brainstem response
ABR was measured at various intervals at 4 weeks, 6 weeks and 8 weeks of age for the Cdh23erl/erl mice. A computer-aided evoked potential system (Intelligent Hearing Systems, Miami, FL, USA) was used to test mice for ABR thresholds as previously described (Johnson et al., 1997, Zheng et al., 1999). Briefly, mice were anesthetized and body temperature maintained at 37–38 C° by placing them on a heating pad in a sound-attenuating chamber. Sub-dermal needle electrodes were inserted at the vertex (active) and ventrolaterally to (reference) the right ear and to the left ear (ground). Clicks and 8- 16- and 32-kHz tone-bursts were channeled through plastic tubes into the animal’s ear canals. The amplified brainstem responses were averaged by a computer and displayed on a computer screen. Auditory thresholds were obtained for each stimulus by reducing the sound pressure level (SPL) at 10-dB steps and finally at 5-dB steps up and down to identify the lowest level at which an ABR pattern could be recognized. ABR threshold values above 55 (for click stimulus), 40 (for 8 kHz), 35 (for16 kHz) or 60 (for 32 kHz) dB SPL were considered to be hearing impaired.
Distortion product oto-acoustic emission (DPOAE)
To test the function of outer hair cells (OHCs) of different mice at different time points, we used the IHSS Smart EP 3.30 and USB ez Software (Intelligent Hearing Systems) for DPOAE measurement, which was conducted for pure tones from 2 to 36 kHz (Polak et al., 2004). An Etymotic10Bt (Etymotic Research, Inc., Elk Grove Village, IL, USA) probe was inserted into the external ear canal and used with two different types of transducers depending on the range of the stimulation frequency. For frequencies ranging from 2 to16 kHz, an Etyfrequencies frommotic ER2 Stimulator was used and for frequencies ranging from 16 to 30 kHz, an HIS high-frequency transducer was used. Stimulus response signals were sampled at a rate of 128 kHz using a 16-bit D/A converter; L1 and L2 amplitudes were set to the same level. Frequencies were acquired with an F2:F1 ratio of 1.22. The stimuli were presented starting from the lowest frequencies tested and increasing to the highest frequencies tested. Five stimulation levels ranging from 65 to 25 dB SPL in 10-dB steps were used.
Observation of hair cells in the cochleae
The organ of Corti was carefully microdissected out and mounted in glycerin on glass slides. The surface preparations were stained for F-actin with Alexa Fluor 568 conjugated to phalloidin to show hair bundles and examined with a fluorescence microscope (Leica DM4000 B, Leica Microsystems, Wetzlar, Germany). OHC counts were made in each turn of the organs of Corti in the EPO-treated and untreated mouse groups and showed as percentage of OHC missing (Han et al., 2012), i.e. the number of missing OHC per view at 63X objective divided by a total of 90–100 OHC in the same view. Counts of two continuous views for each turn were computed for statistical analysis.
Statistical analysis
ANOVA test was used for analyzing data from testing ABR and DPOAE. Hair cell loss data were analyzed by X2-test. P < 0.05 was considered to be significant.
RESULTS
EPO decreases the ABR thresholds in Cdh23erl/erl mice
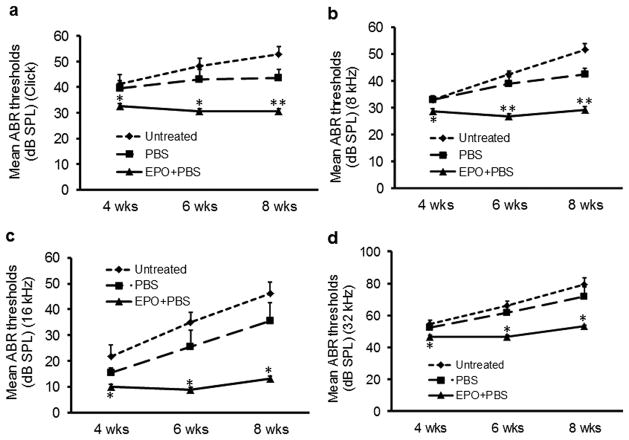
For the three groups of Cdh23erl/erl mice, ABR thresholds were measured at stimulus frequencies of click, 8 kHz, 16 kHz and 32 kHz at the age of 4 weeks, 6 weeks and 8 weeks. ABR thresholds in the EPO group were significantly lower than those of the PBS group or untreated group (P < 0.05) for all the stimulus frequencies at the three time points. There was no significant difference for the ABR thresholds between the PBS group and the untreated group at any stimulus frequencies at any time points. The detailed results were shown in Fig. 1.
Fig. 1. Time course observation of ABR thresholds in the three Cdh23erl/erl mouse groups.
ABR thresholds were measured and a time course comparison of the ABR thresholds between the three mouse groups were shown at stimulus frequencies of click (a), 8 kHz (b), 16 kHz (c) and 32 kHz (d). ABR thresholds (Mean + SE) in the EPO group (solid line, n = 10 ears) were significantly lower than those of the PBS (dash line, n = 10 ears) or untreated group ( dot line, n = 12 ears) (P < 0.05 or 0.01) at 4 week (4 wks), 6 wks and 8 wks of age for all the stimulus frequencies. There were no significant differences for the ABR thresholds between the PBS group and the untreated group at any stimulus frequencies at any time points. Error bar represents the s.e. from the mean. *P <0.05, ** P < 0.01
EPO increases DPOAE amplitudes in Cdh23erl/erl mice
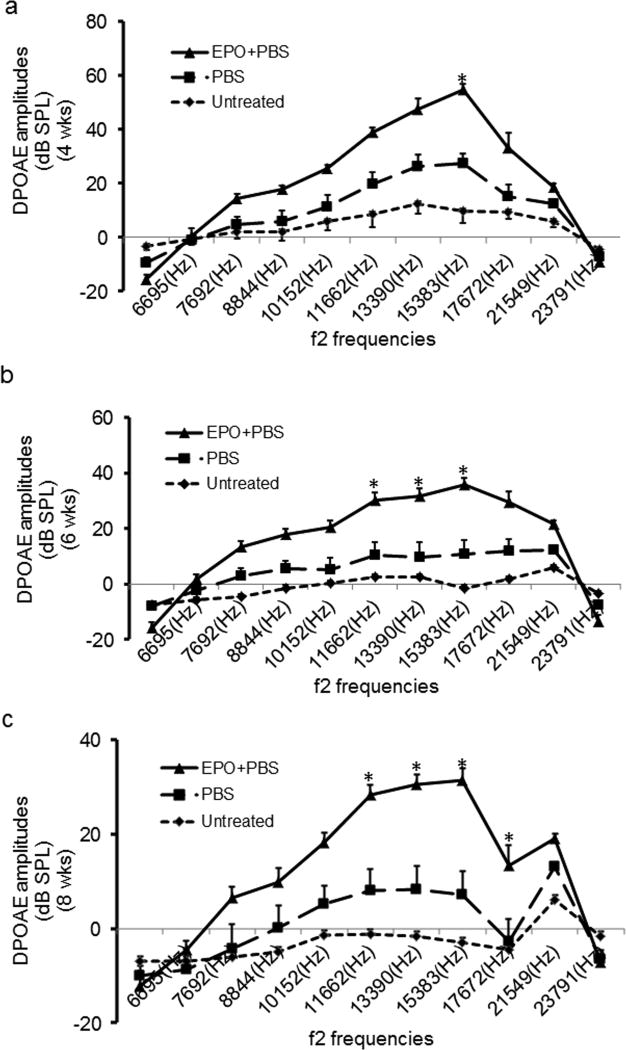
DPOAE amplitudes were measured in the 3 Cdh23erl/erl mouse groups at the age of 4 weeks, 6 weeks and 8 weeks at f2 frequencies from 6,995 Hz to 21,391 Hz. DPOAE amplitudes in the EPO group were generally higher than those of the PBS group and untreated group at any f2 frequencies at any time points; DPOAE amplitudes in the EPO group at f2 frequency of 15,383 Hz were significantly higher than those of the PBS group and untreated group at 4 weeks, 6 weeks and 8 weeks of age. There were no significant differences in DPOAE amplitudes between the PBS group and the untreated group at any f2 frequencies at any time points (Fig. 2).
Fig. 2. DPOAE amplitudes in the three Cdh23erl/erl mouse groups at the three time points.
DPOAE amplitudes were measured and comparison of the DPOAE amplitudat the three time pointses between the three mouse groups were shown at 4 week (4 wks) (a), 6 wks (b) and 8 wks (c) of age. (a) DPOAE amplitudes in the EPO group (solid line, n = 10 ears) at f2 frequency of 15,383 Hz were significantly higher than those of the PBS group(dash line, n = 10 ears) and untreated group (dot line, n = 12 ears) at 4 wks of age; (b) DPOAE amplitudes in the EPO group at f2 frequencies of 11,662 Hz, 13,390 Hz and 15,383 Hz were significantly higher than those of the PBS group and untreated group at 6 wks of age; (c) DPOAE amplitudes in the EPO group at f2 frequencies of 11,662 Hz, 13,390 Hz, 15,383 Hz and 17,672 Hz were significantly higher than those of the PBS group and untreated group at 8 wks of age. There were no significant differences for the DPOAE amplitudes between the PBS group and the untreated group at any f2 frequencies at any time point. Error bar represents the s.e. from the mean. * P < 0.05.
EPO protects hair cells from loss in the cochleae
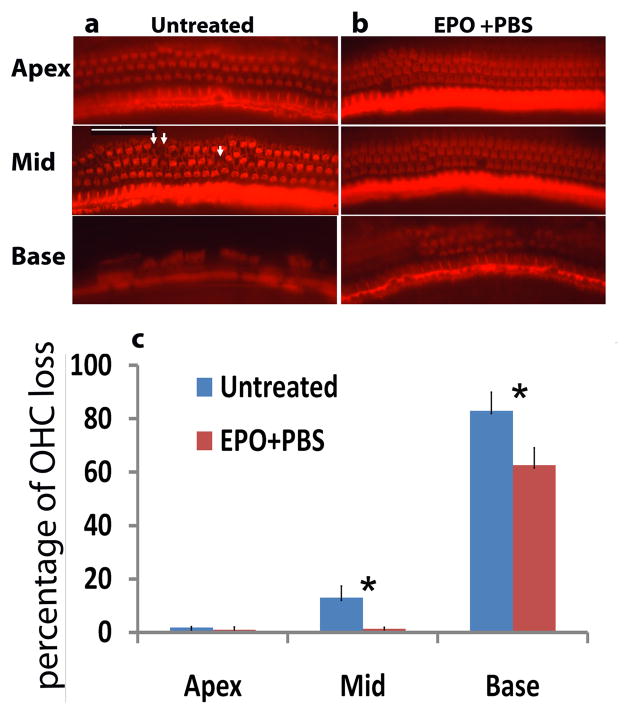
Loss of OHC was evident in the middle-basal turns of cochleae of the untreated mouse group at 8 weeks of age; whereas, only a few OHC losses were seen in the corresponding areas of the EPO-treated mice (Fig. 3a, 3b). OHC loss was counted and averaged in each turn of cochlear surface preparations in untreated and EPO-treated mouse groups at 8 weeks of age (Fig.3c). The mean percentage of OHC loss at middle-basal turns of cochleae was significantly lower in EPO-treated Cdh23erl/erl mice than in the untreated mice (P < 0.05). Loss of OHC was 78–91% in the basal turn of cochleae of the untreated mouse group at 8 weeks of age; whereas, the OHC loss rate was 58–70% in the corresponding areas of the EPO-treated mice. Loss of OHC in the epical turn is 1–2% in both treated and untreated mice. No inner hair cell loss was observed in either EPO-treated or untreated ears from epical to basal turns.
Fig. 3. OHC loss in each turns of cochleae in Cdh23erl/erl mice at age of 8 weeks.
(a) & (b): Representative whole-mount phalloidin-stained preparations from each turn of cochleae. Abnormal appearance of OHC morphology and arrangement in untreated mouse (a) was apparent compared to that of the EPO-treated mouse (b). White arrows indicate areas of OHC loss. Scale bar = 50 μm (applies to all panels). (c) Percentage of OHC loss at each turn of cochleae in Cdh23erl/erl mice. Mean percentages of OHC loss for middle and basal turn were significantly lower in EPO-treated mice than in untreated mice (n = 3–5 for each group). Error bar represents the s.e. from the mean. *P < 0.05
DISCUSSION
Hearing loss (HL) affects an estimated 28 million Americans and 22.5 million Europeans (Dror and Avraham, 2009). Genetics is an important aspect of HL, especially in children(Matsunaga, 2009). Animal models are very important for advancing medical treatment of human disease. Though the mechanisms leading to genetic hearing loss are not completely understood, caspase-family proteases function as important signals in the inner ear pathology. Age-related loss of hair cells and spiral ganglion neurons through apoptosis has come from an association of aging with the expression of apoptosis-related proteins in the cochlea (Alam et al., 2001, Nevado et al., 2006). Several mutations in apoptosis genes, such as TJP2, DFNA5 and MSRB3, were identified as the cause of monogenic hearing impairment (Op de Beeck et al., 2011). Apoptosis was also involved in mouse models of inherited deafness (Han et al., 2012, Schwander et al., 2009, Zheng et al., 2012). The unique genetic mouse model, Cdh23erl/erl, is much more suitable for screening and evaluating otoprotective drugs (Han et al., 2012).
EPO has been shown to be a neuroprotective agent for both central and peripheral nervous system via activation of anti-apoptotic pathways. Its systemic administration prevents apoptosis and inflammatory responses (Genc et al., 2004). Our results in this study show that EPO can preserve the hearing of Cdh23erl/erl mice as tested by ABR thresholds (Fig. 1). It can also maintain the function of OHCs as tested by DPOAE (Fig. 2). These results were further supported by less OHC loss in EPO-treated than in the untreated cochleae of Cdh23erl/erl mice (Fig. 3). Since apoptosis has been identified as the final common pathway in degradation of the organ of Corti in several types of genetic hearing loss (Op de Beeck et al., 2011, Laine et al., 2007, Niu et al., 2007, Ji et al., 2011, Yang et al., 2011, Liu et al., 2012, Zheng et al., 2012), especially in aging-related hearing loss, EPO might show otoprotective effects on these animal models as well, and thus further studies are warranted.
It is reported that systemic administration of EPO may have higher bioavailability in the inner ear than the cerebrospinal fluid (CSF). The intraperitoneal injection of EPO could improve the hearing threshold shift induced by implantation (Quesnel et al., 2011). The concentrations of EPO were much higher in the perilymph (194 mU/mL) than in the CSF (4 mU/mL) in the guinea pigs. Due to the small size of the mice, we could not directly measure EPO concentration in the perilymph or endolymph. Presumably EPO can cross the blood-perilymph barrier in Cdh23erl/erl mice. Thus we chose to use intraperitoneal injection of EPO instead of local surgical drug delivery.
In vitro experiments on inner ear hair cells showed a protective effect of EPO in hair cell damage(Andreeva et al., 2006, Monge et al., 2006). In contrast, an in vivo study using an animal model of noise-induced hearing loss showed a negative effect of EPO (Frederiksen et al., 2007). It is possible that their negative effect of EPO could be due to cochlear stress caused by local EPO delivery. Even though their “trial 3” included intraperitoneal injection of EPO instead of local surgical drug delivery, the potential setback was that their one-time EPO administration might not maintain effective treatment. Thus we delivered EPO every other day for seven weeks and demonstrated significant therapeutic effect.
As reported by Bohne’s laboratory, counts of total hair cells could not be made in the base and round window (RW) hook because many specimens often have moderate to severe hair cell loss in this region (Henry et al., 2001). Similarly in the Cdh23erl/erl mice, the far basal turn and hook region of the basilar membrane are usually lost during the sample preparation.
CONCLUSION
In this study, we show that EPO has otoprotective effects on the Cdh23erl/erl mice as evaluated by cochleograms, a time course measurement of ABR thresholds and DPOAE amplitudes. This is the first report that EPO shows otoprotection on a DFNB12 mouse model with progressive hearing loss. Future studies will focus on the effects of EPO on the apoptosis-related pathways in the Cdh23erl/erl mice.
Highlights.
The Cdh23erl/erl mice are a novel mouse model for progressive hearing loss.
We evaluated EPO as an effective otoprotectant on the Cdh23erl/erl mice.
EPO can protect hearing by 20–30 dB in the Cdh23erl/erl mice.
Acknowledgments
This project was supported partially by NIH grants (R01DC009246, R01DC007392), Foundation of Taishan Scholar (tshw20110515), Natural Science Foundation (ZR2012HZ004) of Shandong Province, National Natural Science Foundation of China (No. 81271085 and 81271092) and Research Initiation Grant of Binzhou Medical University (BY2012KYQD01).
Footnotes
Author contribution statement
Conceived and designed the experiments: Fengchan Han and Qing Yin Zheng. Performed the experiments and analyzed the data: Fengchan Han, Heping Yu, Tihua Zheng, Xiufang Ma, Xin Zhao, Ping Li, Linda Le, Yipeng Su and Qing Yin Zheng. Wrote the paper: Fengchan Han and Qing Yin Zheng.
Conflict of interest
Conflict of interest statement: QYZ, F.H. and H. Y. have filed a patent covering the general approach of EPO and its derivatives as a therapeutic approach for genetic hearing loss and using the erl mouse model for evaluating otoprotective drugs. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Alam SA, Oshima T, Suzuki M, Kawase T, Takasaka T, Ikeda K. The expression of apoptosis-related proteins in the aged cochlea of Mongolian gerbils. Laryngoscope. 2001;111:528–534. doi: 10.1097/00005537-200103000-00026. [DOI] [PubMed] [Google Scholar]
- Andreeva N, Nyamaa A, Haupt H, Gross J, Mazurek B. Recombinant human erythropoietin prevents ischemia-induced apoptosis and necrosis in explant cultures of the rat organ of Corti. Neurosci Lett. 2006;396:86–90. doi: 10.1016/j.neulet.2005.11.013. [DOI] [PubMed] [Google Scholar]
- Berkingali N, Warnecke A, Gomes P, Paasche G, Tack J, Lenarz T, Stover T. Neurite outgrowth on cultured spiral ganglion neurons induced by erythropoietin. Hear Res. 2008;243:121–126. doi: 10.1016/j.heares.2008.07.003. [DOI] [PubMed] [Google Scholar]
- Brines ML, Ghezzi P, Keenan S, Agnello D, de Lanerolle NC, Cerami C, Itri LM, Cerami A. Erythropoietin crosses the blood-brain barrier to protect against experimental brain injury. Proc Natl Acad Sci U S A. 2000;97:10526–10531. doi: 10.1073/pnas.97.19.10526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caye-Thomasen P, Wagner N, Lidegaard Frederiksen B, Asal K, Thomsen J. Erythropoietin and erythropoietin receptor expression in the guinea pig inner ear. Hear Res. 2005;203:21–27. doi: 10.1016/j.heares.2004.11.017. [DOI] [PubMed] [Google Scholar]
- Dror AA, Avraham KB. Hearing loss: mechanisms revealed by genetics and cell biology. Annu Rev Genet. 2009;43:411–437. doi: 10.1146/annurev-genet-102108-134135. [DOI] [PubMed] [Google Scholar]
- Frederiksen BL, Caye-Thomasen P, Lund SP, Wagner N, Asal K, Olsen NV, Thomsen J. Does erythropoietin augment noise induced hearing loss? Hear Res. 2007;223:129–137. doi: 10.1016/j.heares.2006.11.002. [DOI] [PubMed] [Google Scholar]
- Genc S, Koroglu TF, Genc K. Erythropoietin and the nervous system. Brain Res. 2004;1000:19–31. doi: 10.1016/j.brainres.2003.12.037. [DOI] [PubMed] [Google Scholar]
- Ghezzi P, Brines M. Erythropoietin as an antiapoptotic, tissue-protective cytokine. Cell Death Differ. 2004;11(Suppl 1):S37–44. doi: 10.1038/sj.cdd.4401450. [DOI] [PubMed] [Google Scholar]
- Han F, Yu H, Tian C, Chen HE, Benedict-Alderfer C, Zheng Y, Wang Q, Han X, Zheng QY. A new mouse mutant of the Cdh23 gene with early-onset hearing loss facilitates evaluation of otoprotection drugs. Pharmacogenomics J. 2012;12:30–44. doi: 10.1038/tpj.2010.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry O, Barbara B, Gary H. Handbook of Mouse Auditory Research. CRC Press; 2001. Preparation and Evaluation of the Mouse Temporal Bone; pp. 171–187. [Google Scholar]
- Ji Y-B, Han D-Y, Lan L, Wang D-Y, Zong L, Zhao F-F, Liu Q, Benedict-Alderfer C, Zheng Q-Y, et al. Molecular epidemiological analysis of mitochondrial DNA12SrRNA A1555G, GJB2, and SLC26A4 mutations in sporadic outpatients with nonsyndromic sensorineural hearing loss in China. Acta Oto-Laryngologica. 2011;131:124–129. doi: 10.3109/00016489.2010.483479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson DW, Pat B, Vesey DA, Guan Z, Endre Z, Gobe GC. Delayed administration of darbepoetin or erythropoietin protects against ischemic acute renal injury and failure. Kidney Int. 2006;69:1806–1813. doi: 10.1038/sj.ki.5000356. [DOI] [PubMed] [Google Scholar]
- Johnson KR, Erway LC, Cook SA, Willott JF, Zheng QY. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear Res. 1997;114:83–92. doi: 10.1016/s0378-5955(97)00155-x. [DOI] [PubMed] [Google Scholar]
- Laine H, Doetzlhofer A, Mantela J, Ylikoski J, Laiho M, Roussel MF, Segil N, Pirvola U. p19(Ink4d) and p21(Cip1) collaborate to maintain the postmitotic state of auditory hair cells, their codeletion leading to DNA damage and p53-mediated apoptosis. J Neurosci. 2007;27:1434–1444. doi: 10.1523/JNEUROSCI.4956-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S, Li S, Zhu H, Cheng S, Zheng QY. A mutation in the cdh23 gene causes age-related hearing loss in Cdh23 nmf308/nmf308 mice. Gene. 2012;499:309–317. doi: 10.1016/j.gene.2012.01.084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maiese K, Chong ZZ, Li F, Shang YC. Erythropoietin: elucidating new cellular targets that broaden therapeutic strategies. Prog Neurobiol. 2008;85:194–213. doi: 10.1016/j.pneurobio.2008.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsunaga T. Value of genetic testing in the otological approach for sensorineural hearing loss. Keio J Med. 2009;58:216–222. doi: 10.2302/kjm.58.216. [DOI] [PubMed] [Google Scholar]
- Monge A, Nagy I, Bonabi S, Schmid S, Gassmann M, Bodmer D. The effect of erythropoietin on gentamicin-induced auditory hair cell loss. Laryngoscope. 2006;116:312–316. doi: 10.1097/01.mlg.0000199400.08550.3f. [DOI] [PubMed] [Google Scholar]
- Monge Naldi A, Gassmann M, Bodmer D. Erythropoietin but not VEGF has a protective effect on auditory hair cells in the inner ear. Cell Mol Life Sci. 2009;66:3595–3599. doi: 10.1007/s00018-009-0144-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nevado J, Sanz R, Casqueiro JC, Ayala A, Garcia-Berrocal JR, Ramirez-Camacho R. Ageing evokes an intrinsic pro-apoptotic signalling pathway in rat cochlea. Acta Otolaryngol. 2006;126:1134–1139. doi: 10.1080/00016480600672592. [DOI] [PubMed] [Google Scholar]
- Niu X, Trifunovic A, Larsson NG, Canlon B. Somatic mtDNA mutations cause progressive hearing loss in the mouse. Exp Cell Res. 2007;313:3924–3934. doi: 10.1016/j.yexcr.2007.05.029. [DOI] [PubMed] [Google Scholar]
- Op de Beeck K, Schacht J, Van Camp G. Apoptosis in acquired and genetic hearing impairment: the programmed death of the hair cell. Hear Res. 2011;281:18–27. doi: 10.1016/j.heares.2011.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polak M, Eshraghi AA, Nehme O, Ahsan S, Guzman J, Delgado RE, He J, Telischi FF, Balkany TJ, Van De Water TR. Evaluation of hearing and auditory nerve function by combining ABR, DPOAE and eABR tests into a single recording session. J Neurosci Methods. 2004;134:141–149. doi: 10.1016/j.jneumeth.2003.11.003. [DOI] [PubMed] [Google Scholar]
- Quesnel S, Nguyen Y, Campo P, Hermine O, Ribeil JA, Elmaleh M, Grayeli AB, Ferrary E, Sterkers O, Couloigner V. Protective effect of systemic administration of erythropoietin on auditory brain stem response and compound action potential thresholds in an animal model of cochlear implantation. Ann Otol Rhinol Laryngol. 2011;120:737–747. doi: 10.1177/000348941112001108. [DOI] [PubMed] [Google Scholar]
- Schwander M, Xiong W, Tokita J, Lelli A, Elledge HM, Kazmierczak P, Sczaniecka A, Kolatkar A, Wiltshire T, Kuhn P, Holt JR, Kachar B, Tarantino L, Muller U. A mouse model for nonsyndromic deafness (DFNB12) links hearing loss to defects in tip links of mechanosensory hair cells. Proc Natl Acad Sci U S A. 2009;106:5252–5257. doi: 10.1073/pnas.0900691106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siren AL, Fratelli M, Brines M, Goemans C, Casagrande S, Lewczuk P, Keenan S, Gleiter C, Pasquali C, Capobianco A, Mennini T, Heumann R, Cerami A, Ehrenreich H, Ghezzi P. Erythropoietin prevents neuronal apoptosis after cerebral ischemia and metabolic stress. Proc Natl Acad Sci U S A. 2001;98:4044–4049. doi: 10.1073/pnas.051606598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesey DA, Cheung C, Pat B, Endre Z, Gobe G, Johnson DW. Erythropoietin protects against ischaemic acute renal injury. Nephrol Dial Transplant. 2004;19:348–355. doi: 10.1093/ndt/gfg547. [DOI] [PubMed] [Google Scholar]
- Wiese L, Hempel C, Penkowa M, Kirkby N, Kurtzhals JA. Recombinant human erythropoietin increases survival and reduces neuronal apoptosis in a murine model of cerebral malaria. Malar J. 2008;7:3. doi: 10.1186/1475-2875-7-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang B, Tian C, Zhang Z-g, Han F-c, Azem R, Yu H, Zheng Y, Jin G, Arnold JE, et al. Sh3pxd2b mice are a model for craniofacial dysmorphology and otitis media. PLoS One. 2011;6(7):e22622. doi: 10.1371/journal.pone.0022622. doi: 10.1371/journal.pone.0022622. Epub 2011 Jul 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng QY, Johnson KR, Erway LC. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear Res. 1999;130:94–107. doi: 10.1016/s0378-5955(99)00003-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng QY, Scarborough JD, Zheng Y, Yu H, Choi D, Gillespie PG. Digenic inheritance of deafness caused by 8J allele of myosin-VIIA and mutations in other Usher I genes. Human Molecular Genetics. 2012;21:2588–2598. doi: 10.1093/hmg/dds084. [DOI] [PMC free article] [PubMed] [Google Scholar]