Abstract
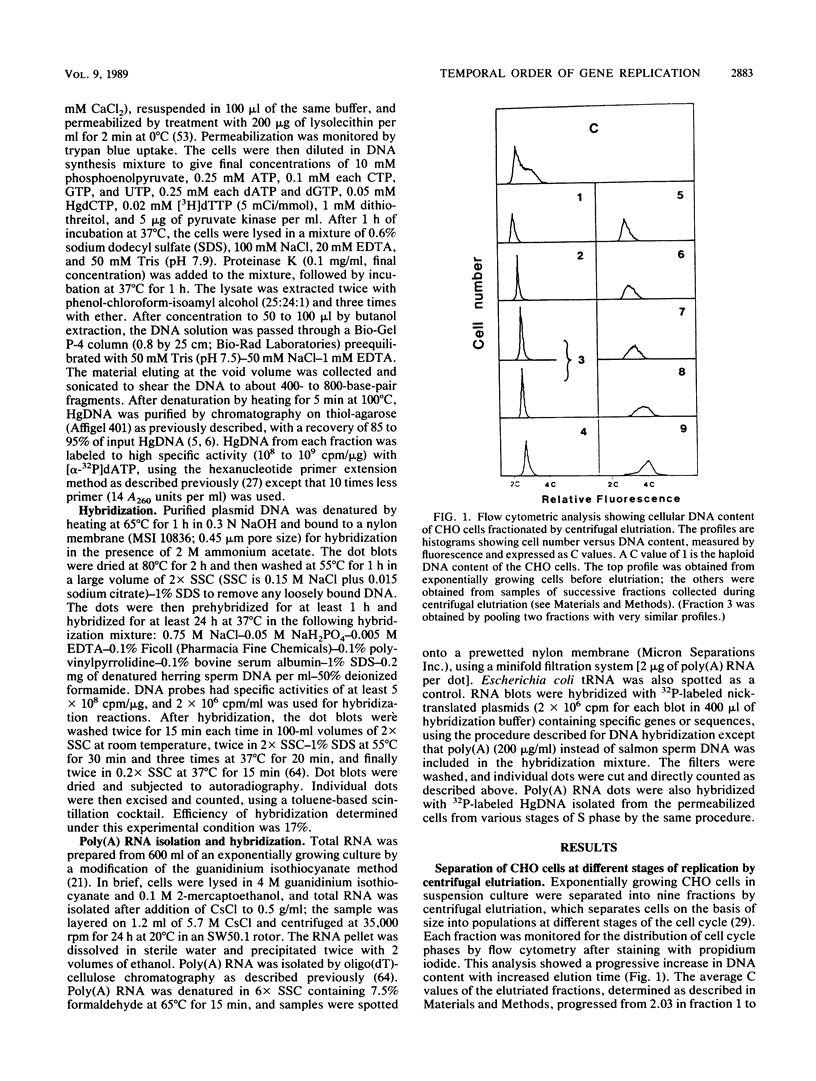
To investigate the molecular basis of the regulatory mechanisms responsible for the orderly replication of the mammalian genome, we have developed an experimental system by which the replication order of various genes can be defined with relative ease and precision. Exponentially growing CHO-K1 cells were separated into populations representing various stages of the cell cycle by centrifugal elutriation and analyzed for cell cycle status flow cytometry. The replication of specific genes in each elutriated fraction was measured by labeling with 5-mercuri-dCTP and [3H]dTPP under conditions of optimal DNA synthesis after cell permeabilization with lysolecithin. Newly synthesized mercurated DNA from each elutriated fraction was purified by affinity chromatography on thiol-agarose and replicated with the large fragment of Escherichia coli DNA polymerase I by using [alpha-32P]dATP and random primers. The 32P-labeled DNA representative of various stages of the cell cycle was then hybridized with dot blots of plasmid DNA containing specific cloned genes. From these results, it was possible to deduce the nuclear DNA content at the time each specific gene replicated during S phase (C value). The C values of 29 genes, which included single-copy genes, multifamily genes, oncogenes, and repetitive sequences, were determined and found to be distributed over the entire S phase. Of the 28 genes studied, 19 had been examined by others using in vivo labeling techniques, with results which agreed with the replication pattern observed in this study. The replication times of nine other genes are described here for the first time. Our method of analysis is sensitive enough to determine the replication time of single-copy genes. The replication times of various genes and their levels of expression in exponentially growing CHO cells were compared. Although there was a general correlation between transcriptional activity and replication in the first half of S phase, examination of specific genes revealed a number of exceptions. Approximately 25% of total poly(A) RNA was transcribed from the late-replicating DNA.
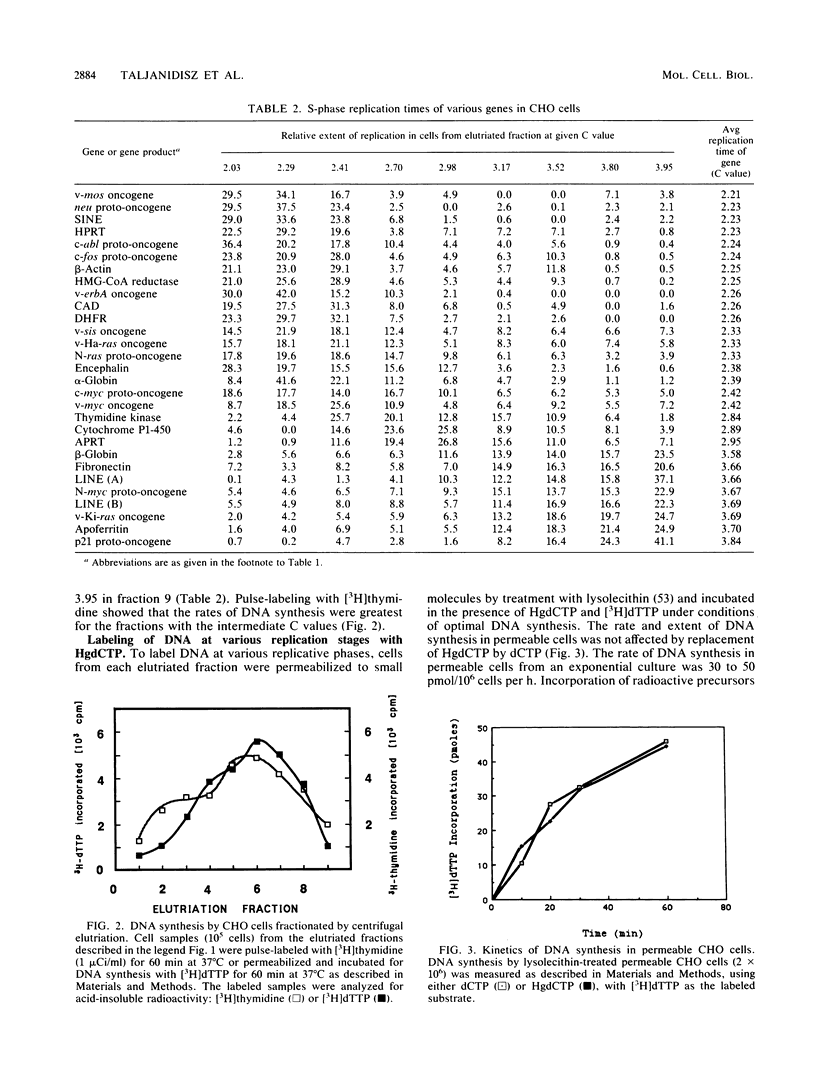
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alitalo K., Bishop J. M., Smith D. H., Chen E. Y., Colby W. W., Levinson A. D. Nucleotide sequence to the v-myc oncogene of avian retrovirus MC29. Proc Natl Acad Sci U S A. 1983 Jan;80(1):100–104. doi: 10.1073/pnas.80.1.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amaldi F., Giacomoni D., Zito-Bignami R. On the duplication of ribosomal RNA cistrons in Chinese hamster cells. Eur J Biochem. 1969 Dec;11(3):419–423. doi: 10.1111/j.1432-1033.1969.tb00790.x. [DOI] [PubMed] [Google Scholar]
- Ariga H., Itani T., Iguchi-Ariga S. M. Autonomous replicating sequences from mouse cells which can replicate in mouse cells in vivo and in vitro. Mol Cell Biol. 1987 Jan;7(1):1–6. doi: 10.1128/mcb.7.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balazs L., Schildkraut C. L. DNA replication in synchronized cultured mammalian cells. II. Replication of ribosomal cistrons in thymidine-synchronized HeLa cells. J Mol Biol. 1971 Apr 14;57(1):153–158. doi: 10.1016/0022-2836(71)90125-2. [DOI] [PubMed] [Google Scholar]
- Banfalvi G., Bhattacharya S., Sarkar N. Selective isolation of mercurated DNA by affinity chromatography on thiol matrices. Anal Biochem. 1985 Apr;146(1):64–70. doi: 10.1016/0003-2697(85)90396-3. [DOI] [PubMed] [Google Scholar]
- Banfalvi G., Sarkar N. Analysis of the 5'-termini of nascent DNA chains synthesized in permeable cells of Bacillus subtilis. J Mol Biol. 1983 Jan 15;163(2):147–169. doi: 10.1016/0022-2836(83)90001-3. [DOI] [PubMed] [Google Scholar]
- Banfalvi G., Sarkar N. Origin and degradation of the RNA primers at the 5' termini of nascent DNA chains in Bacillus subtilis. J Mol Biol. 1985 Nov 20;186(2):275–282. doi: 10.1016/0022-2836(85)90104-4. [DOI] [PubMed] [Google Scholar]
- Banfalvi G., Sooki-Toth A., Sarkar N., Csuzi S., Antoni F. Nascent DNA chains synthesized in reversibly permeable cells of mouse thymocytes. Eur J Biochem. 1984 Mar 15;139(3):553–559. doi: 10.1111/j.1432-1033.1984.tb08041.x. [DOI] [PubMed] [Google Scholar]
- Bargmann C. I., Hung M. C., Weinberg R. A. Multiple independent activations of the neu oncogene by a point mutation altering the transmembrane domain of p185. Cell. 1986 Jun 6;45(5):649–657. doi: 10.1016/0092-8674(86)90779-8. [DOI] [PubMed] [Google Scholar]
- Bhattacharya S., Sarkar N. Study of deoxyribonucleic acid replication in permeable cells of Bacillus subtilis using mercurated nucleotide substrates. Biochemistry. 1981 May 26;20(11):3029–3034. doi: 10.1021/bi00514a007. [DOI] [PubMed] [Google Scholar]
- Bhattacharya S., Sarkar N. Transforming activity of mercury-substituted DNA synthesized in vitro by permeable cells of Bacillus subtilis. J Biol Chem. 1982 Feb 25;257(4):1610–1612. [PubMed] [Google Scholar]
- Bishop J. M. Viral oncogenes. Cell. 1985 Aug;42(1):23–38. doi: 10.1016/s0092-8674(85)80098-2. [DOI] [PubMed] [Google Scholar]
- Blair D. G., McClements W. L., Oskarsson M. K., Fischinger P. J., Vande Woude G. F. Biological activity of cloned Moloney sarcoma virus DNA: Terminally redundant sequences may enhance transformation efficiency. Proc Natl Acad Sci U S A. 1980 Jun;77(6):3504–3508. doi: 10.1073/pnas.77.6.3504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braunstein J. D., Schulze D., DelGiudice T., Furst A., Schildkraut C. L. The temporal order of replication of murine immunoglobulin heavy chain constant region sequences corresponds to their linear order in the genome. Nucleic Acids Res. 1982 Nov 11;10(21):6887–6902. doi: 10.1093/nar/10.21.6887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brennand J., Konecki D. S., Caskey C. T. Expression of human and Chinese hamster hypoxanthine-guanine phosphoribosyltransferase cDNA recombinants in cultured Lesch-Nyhan and Chinese hamster fibroblasts. J Biol Chem. 1983 Aug 25;258(16):9593–9596. [PubMed] [Google Scholar]
- Brown E. H., Iqbal M. A., Stuart S., Hatton K. S., Valinsky J., Schildkraut C. L. Rate of replication of the murine immunoglobulin heavy-chain locus: evidence that the region is part of a single replicon. Mol Cell Biol. 1987 Jan;7(1):450–457. doi: 10.1128/mcb.7.1.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calza R. E., Eckhardt L. A., DelGiudice T., Schildkraut C. L. Changes in gene position are accompanied by a change in time of replication. Cell. 1984 Mar;36(3):689–696. doi: 10.1016/0092-8674(84)90349-0. [DOI] [PubMed] [Google Scholar]
- Carothers A. M., Urlaub G., Ellis N., Chasin L. A. Structure of the dihydrofolate reductase gene in Chinese hamster ovary cells. Nucleic Acids Res. 1983 Apr 11;11(7):1997–2012. doi: 10.1093/nar/11.7.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin D. J., Gil G., Russell D. W., Liscum L., Luskey K. L., Basu S. K., Okayama H., Berg P., Goldstein J. L., Brown M. S. Nucleotide sequence of 3-hydroxy-3-methyl-glutaryl coenzyme A reductase, a glycoprotein of endoplasmic reticulum. Nature. 1984 Apr 12;308(5960):613–617. doi: 10.1038/308613a0. [DOI] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Comb M., Seeburg P. H., Adelman J., Eiden L., Herbert E. Primary structure of the human Met- and Leu-enkephalin precursor and its mRNA. Nature. 1982 Feb 25;295(5851):663–666. doi: 10.1038/295663a0. [DOI] [PubMed] [Google Scholar]
- D'Andrea A. D., Tantravahi U., Lalande M., Perle M. A., Latt S. A. High resolution analysis of the timing of replication of specific DNA sequences during S phase of mammalian cells. Nucleic Acids Res. 1983 Jul 25;11(14):4753–4774. doi: 10.1093/nar/11.14.4753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhar V., Mager D., Iqbal A., Schildkraut C. L. The coordinate replication of the human beta-globin gene domain reflects its transcriptional activity and nuclease hypersensitivity. Mol Cell Biol. 1988 Nov;8(11):4958–4965. doi: 10.1128/mcb.8.11.4958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis R. W., Defeo D., Shih T. Y., Gonda M. A., Young H. A., Tsuchida N., Lowy D. R., Scolnick E. M. The p21 src genes of Harvey and Kirsten sarcoma viruses originate from divergent members of a family of normal vertebrate genes. Nature. 1981 Aug 6;292(5823):506–511. doi: 10.1038/292506a0. [DOI] [PubMed] [Google Scholar]
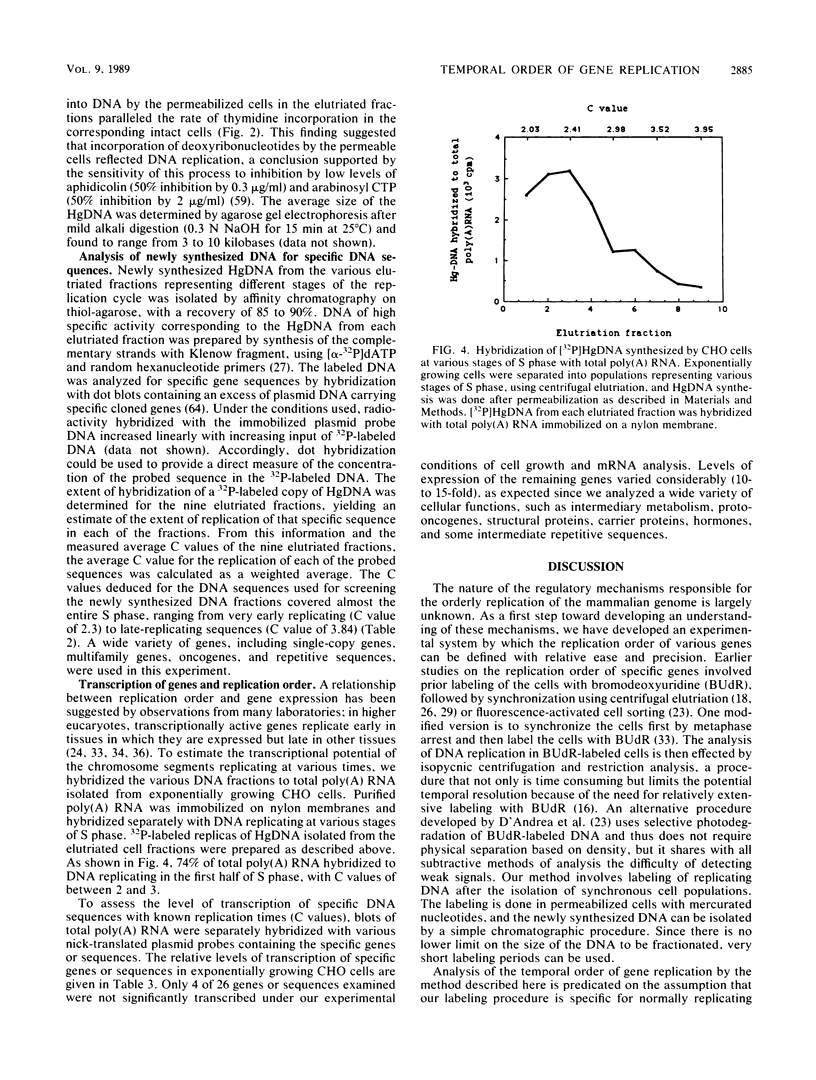
- Epner E., Rifkind R. A., Marks P. A. Replication of alpha and beta globin DNA sequences occurs during early S phase in murine erythroleukemia cells. Proc Natl Acad Sci U S A. 1981 May;78(5):3058–3062. doi: 10.1073/pnas.78.5.3058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Frykberg L., Palmieri S., Beug H., Graf T., Hayman M. J., Vennström B. Transforming capacities of avian erythroblastosis virus mutants deleted in the erbA or erbB oncogenes. Cell. 1983 Jan;32(1):227–238. doi: 10.1016/0092-8674(83)90513-5. [DOI] [PubMed] [Google Scholar]
- Furst A., Brown E. H., Braunstein J. D., Schildkraut C. L. alpha-Globulin sequences are located in a region of early-replicating DNA in murine erythroleukemia cells. Proc Natl Acad Sci U S A. 1981 Feb;78(2):1023–1027. doi: 10.1073/pnas.78.2.1023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gannon J. V., Lane D. P. p53 and DNA polymerase alpha compete for binding to SV40 T antigen. Nature. 1987 Oct 1;329(6138):456–458. doi: 10.1038/329456a0. [DOI] [PubMed] [Google Scholar]
- Giacomoni D., Finkel D. Time of duplication of ribosomal RNA cistrons in a cell line of Potorous tridactylis (rat kangaroo). J Mol Biol. 1972 Oct 14;70(3):725–728. doi: 10.1016/0022-2836(72)90570-0. [DOI] [PubMed] [Google Scholar]
- Gimmler G. M., Schweizer E. rDNA replication in a synchronized culture of Saccharomyces cerevisiae. Biochem Biophys Res Commun. 1972 Jan 14;46(1):143–149. doi: 10.1016/0006-291x(72)90642-0. [DOI] [PubMed] [Google Scholar]
- Goldman M. A., Holmquist G. P., Gray M. C., Caston L. A., Nag A. Replication timing of genes and middle repetitive sequences. Science. 1984 May 18;224(4650):686–692. doi: 10.1126/science.6719109. [DOI] [PubMed] [Google Scholar]
- Guinta D. R., Tso J. Y., Narayanswami S., Hamkalo B. A., Korn L. J. Early replication and expression of oocyte-type 5S RNA genes in a Xenopus somatic cell line carrying a translocation. Proc Natl Acad Sci U S A. 1986 Jul;83(14):5150–5154. doi: 10.1073/pnas.83.14.5150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hand R. Eucaryotic DNA: organization of the genome for replication. Cell. 1978 Oct;15(2):317–325. doi: 10.1016/0092-8674(78)90001-6. [DOI] [PubMed] [Google Scholar]
- Hatton K. S., Dhar V., Brown E. H., Iqbal M. A., Stuart S., Didamo V. T., Schildkraut C. L. Replication program of active and inactive multigene families in mammalian cells. Mol Cell Biol. 1988 May;8(5):2149–2158. doi: 10.1128/mcb.8.5.2149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hentze M. W., Keim S., Papadopoulos P., O'Brien S., Modi W., Drysdale J., Leonard W. J., Harford J. B., Klausner R. D. Cloning, characterization, expression, and chromosomal localization of a human ferritin heavy-chain gene. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7226–7230. doi: 10.1073/pnas.83.19.7226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmquist G. P., Caston L. A. Replication time of interspersed repetitive DNA sequences in hamsters. Biochim Biophys Acta. 1986 Nov 13;868(2-3):164–177. doi: 10.1016/0167-4781(86)90019-9. [DOI] [PubMed] [Google Scholar]
- Holmquist G. P. Role of replication time in the control of tissue-specific gene expression. Am J Hum Genet. 1987 Feb;40(2):151–173. [PMC free article] [PubMed] [Google Scholar]
- Holmquist G., Gray M., Porter T., Jordan J. Characterization of Giemsa dark- and light-band DNA. Cell. 1982 Nov;31(1):121–129. doi: 10.1016/0092-8674(82)90411-1. [DOI] [PubMed] [Google Scholar]
- Housman D., Huberman J. A. Changes in the rate of DNA replication fork movement during S phase in mammalian cells. J Mol Biol. 1975 May 15;94(2):173–181. doi: 10.1016/0022-2836(75)90076-5. [DOI] [PubMed] [Google Scholar]
- Huberman J. A., Riggs A. D. On the mechanism of DNA replication in mammalian chromosomes. J Mol Biol. 1968 Mar 14;32(2):327–341. doi: 10.1016/0022-2836(68)90013-2. [DOI] [PubMed] [Google Scholar]
- Iguchi-Ariga S. M., Itani T., Yamaguchi M., Ariga H. c-myc protein can be substituted for SV40 T antigen in SV40 DNA replication. Nucleic Acids Res. 1987 Jun 25;15(12):4889–4899. doi: 10.1093/nar/15.12.4889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iqbal M. A., Chinsky J., Didamo V., Schildkraut C. L. Replication of proto-oncogenes early during the S phase in mammalian cell lines. Nucleic Acids Res. 1987 Jan 12;15(1):87–103. doi: 10.1093/nar/15.1.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelinek W. R., Schmid C. W. Repetitive sequences in eukaryotic DNA and their expression. Annu Rev Biochem. 1982;51:813–844. doi: 10.1146/annurev.bi.51.070182.004121. [DOI] [PubMed] [Google Scholar]
- Johnson E. M., Jelinek W. R. Replication of a plasmid bearing a human Alu-family repeat in monkey COS-7 cells. Proc Natl Acad Sci U S A. 1986 Jul;83(13):4660–4664. doi: 10.1073/pnas.83.13.4660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P. B., Galeazzi D. R., Fisher J. M., Whitlock J. P., Jr Control of cytochrome P1-450 gene expression by dioxin. Science. 1985 Mar 22;227(4693):1499–1502. doi: 10.1126/science.3856321. [DOI] [PubMed] [Google Scholar]
- Krainer A. R., Maniatis T., Ruskin B., Green M. R. Normal and mutant human beta-globin pre-mRNAs are faithfully and efficiently spliced in vitro. Cell. 1984 Apr;36(4):993–1005. doi: 10.1016/0092-8674(84)90049-7. [DOI] [PubMed] [Google Scholar]
- Land H., Chen A. C., Morgenstern J. P., Parada L. F., Weinberg R. A. Behavior of myc and ras oncogenes in transformation of rat embryo fibroblasts. Mol Cell Biol. 1986 Jun;6(6):1917–1925. doi: 10.1128/mcb.6.6.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis J. A. Structure and expression of the Chinese hamster thymidine kinase gene. Mol Cell Biol. 1986 Jun;6(6):1998–2010. doi: 10.1128/mcb.6.6.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchionni M. A., Roufa D. J. Replication of viral DNA sequences integrated within the chromatin of SV40-transformed Chinese hamster lung cells. Cell. 1981 Oct;26(2 Pt 2):245–258. doi: 10.1016/0092-8674(81)90307-x. [DOI] [PubMed] [Google Scholar]
- Miller A. D., Curran T., Verma I. M. c-fos protein can induce cellular transformation: a novel mechanism of activation of a cellular oncogene. Cell. 1984 Jan;36(1):51–60. doi: 10.1016/0092-8674(84)90073-4. [DOI] [PubMed] [Google Scholar]
- Miller M. R., Castellot J. J., Jr, Pardee A. B. A permeable animal cell preparation for studying macromolecular synthesis. DNA synthesis and the role of deoxyribonucleotides in S phase initiation. Biochemistry. 1978 Mar 21;17(6):1073–1080. doi: 10.1021/bi00599a021. [DOI] [PubMed] [Google Scholar]
- Miller M. R., Ulrich R. G., Wang T. S., Korn D. Monoclonal antibodies against human DNA polymerase-alpha inhibit DNA replication in permeabilized human cells. J Biol Chem. 1985 Jan 10;260(1):134–138. [PubMed] [Google Scholar]
- Murray M. J., Cunningham J. M., Parada L. F., Dautry F., Lebowitz P., Weinberg R. A. The HL-60 transforming sequence: a ras oncogene coexisting with altered myc genes in hematopoietic tumors. Cell. 1983 Jul;33(3):749–757. doi: 10.1016/0092-8674(83)90017-x. [DOI] [PubMed] [Google Scholar]
- Nishioka Y., Leder P. The complete sequence of a chromosomal mouse alpha--globin gene reveals elements conserved throughout vertebrate evolution. Cell. 1979 Nov;18(3):875–882. doi: 10.1016/0092-8674(79)90139-9. [DOI] [PubMed] [Google Scholar]
- Oliver N., Newby R. F., Furcht L. T., Bourgeois S. Regulation of fibronectin biosynthesis by glucocorticoids in human fibrosarcoma cells and normal fibroblasts. Cell. 1983 May;33(1):287–296. doi: 10.1016/0092-8674(83)90357-4. [DOI] [PubMed] [Google Scholar]
- Robbins K. C., Devare S. G., Aaronson S. A. Molecular cloning of integrated simian sarcoma virus: genome organization of infectious DNA clones. Proc Natl Acad Sci U S A. 1981 May;78(5):2918–2922. doi: 10.1073/pnas.78.5.2918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar N., List J. F., Banfalvi G. Replication of the origin region of simian virus 40 DNA in permeabilized monkey cells. Eur J Biochem. 1987 Oct 15;168(2):263–268. doi: 10.1111/j.1432-1033.1987.tb13415.x. [DOI] [PubMed] [Google Scholar]
- Schwab M., Ellison J., Busch M., Rosenau W., Varmus H. E., Bishop J. M. Enhanced expression of the human gene N-myc consequent to amplification of DNA may contribute to malignant progression of neuroblastoma. Proc Natl Acad Sci U S A. 1984 Aug;81(15):4940–4944. doi: 10.1073/pnas.81.15.4940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stambrook P. J. The temporal replication of ribosomal genes in synchronized Chinese hamster cells. J Mol Biol. 1974 Jan 25;82(3):303–313. doi: 10.1016/0022-2836(74)90592-0. [DOI] [PubMed] [Google Scholar]
- Studzinski G. P., Brelvi Z. S., Feldman S. C., Watt R. A. Participation of c-myc protein in DNA synthesis of human cells. Science. 1986 Oct 24;234(4775):467–470. doi: 10.1126/science.3532322. [DOI] [PubMed] [Google Scholar]
- TAYLOR J. H. Asynchronous duplication of chromosomes in cultured cells of Chinese hamster. J Biophys Biochem Cytol. 1960 Jun;7:455–464. doi: 10.1083/jcb.7.3.455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- TAYLOR J. H. DNA SYNTHESIS IN RELATION TO CHROMOSOME REPRODUCTION AND THE REUNION OF BREAKS. J Cell Physiol. 1963 Oct;62:SUPPL1–SUPPL1:86. doi: 10.1002/jcp.1030620408. [DOI] [PubMed] [Google Scholar]
- Taljanidisz J., Decker R. S., Guo Z. S., DePamphilis M. L., Sarkar N. Initiation of simian virus 40 DNA replication in vitro: identification of RNA-primed nascent DNA chains. Nucleic Acids Res. 1987 Oct 12;15(19):7877–7888. doi: 10.1093/nar/15.19.7877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taljanidisz J., Karnik P., Sarkar N. Messenger ribonucleic acid for the lipoprotein of the Escherichia coli outer membrane is polyadenylated. J Mol Biol. 1987 Feb 5;193(3):507–515. doi: 10.1016/0022-2836(87)90263-4. [DOI] [PubMed] [Google Scholar]
- Wahl G. M., Padgett R. A., Stark G. R. Gene amplification causes overproduction of the first three enzymes of UMP synthesis in N-(phosphonacetyl)-L-aspartate-resistant hamster cells. J Biol Chem. 1979 Sep 10;254(17):8679–8689. [PubMed] [Google Scholar]


