Abstract
Benzodiazepines are prescribed widely but their utility is limited by unwanted side effects, including abuse potential. The mechanisms underlying the abuse-related effects of benzodiazepines are not well understood, although α1 subunit-containing GABAA receptors have been proposed to have a critical role. Here, we examine the reinforcing effects of several compounds that vary with respect to intrinsic efficacy at α2, α3, and α5 subunit-containing GABAA receptors but lack efficacy at α1 subunit-containing GABAA receptors (‘α1-sparing compounds'): MRK-623 (functional selectivity for α2/α3 subunit-containing receptors), TPA023B (functional selectivity for α2/α3/α5 subunit-containing receptors), and TP003 (functional selectivity for α3 subunit-containing receptors). The reinforcing effects of the α1-sparing compounds were compared with those of the non-selective benzodiazepine receptor partial agonist MRK-696, and non-selective benzodiazepine receptor full agonists, midazolam and lorazepam, in rhesus monkeys trained to self-administer midazolam or cocaine, under a progressive-ratio schedule of intravenous (i.v.) drug injection. The α1-sparing compounds were self-administered significantly above vehicle levels in monkeys maintained under a midazolam baseline, but not under a cocaine baseline over the dose ranges tested. Importantly, TP003 had significant reinforcing effects, albeit at lower levels of self-administration than non-selective benzodiazepine receptor agonists. Together, these results suggest that α1 subunit-containing GABAA receptors may have a role in the reinforcing effects of benzodiazepine-type compounds in monkeys with a history of stimulant self-administration, whereas α3 subunit-containing GABAA receptors may be important mediators of the reinforcing effects of benzodiazepine-type compounds in animals with a history of sedative-anxiolytic/benzodiazepine self-administration.
Keywords: benzodiazepine, GABAA receptor, alpha subunit, self-administration, drug history, rhesus monkey (Macaca mulatta)
INTRODUCTION
Although significant new information has accrued in recent years on mechanisms of action underlying the therapeutic effects of benzodiazepines, the precise mechanism(s) of action underlying the addictive effects of these widely-abused drugs remains elusive. Benzodiazepines potentiate the effects of the neurotransmitter γ-aminobutyric acid (GABA) via positive allosteric modulation at the γ-aminobutyric acid type A (GABAA) receptor. GABAA receptors are heteropentameric chloride ion channels assembled in a typical stoichiometry of 2 α, 2 β, and 1 γ subunits; conventional benzodiazepines bind to GABAA receptors containing α1, α2, α3, or α5 subunits (α1GABAA, α2GABAA, α3GABAA, or α5GABAA receptors, respectively; Rudolph and Knoflach, 2011; Tan et al, 2011). Previous studies have shown localization of α1GABAA receptors on inhibitory interneurons that synapse with DA neurons in the ventral tegmental area (VTA; Heikkinen et al, 2009; Tan et al, 2010) while α3GABAA receptors are expressed at lower levels on the DA neurons themselves (Fritschy and Mohler, 1995; Ciccarelli et al, 2012). Benzodiazepines decrease firing of these interneurons via α1GABAA receptors, resulting in a net effect of decreased activity, ie, ‘disinhibition', of DA neurons. The ultimate outcome would be an increase in DA release in the nucleus accumbens (NAc; Tan et al, 2010, 2011), although this effect has yet to be established for benzodiazepines (eg Finlay et al, 1992; Murai et al, 1994). If this hypothesis is correct, then compounds lacking activity at α1GABAA receptors (ie, ‘α1-sparing compounds') should lack abuse potential (Tan et al, 2010, 2011).
Currently, there is mixed evidence for the idea that α1-sparing compounds lack abuse potential. Supporting this, the α1-sparing compound, TPA023, was not self-administered by baboons up to doses that completely occupied benzodiazepine binding sites as measured via positron emission tomography (Ator et al, 2010). Moreover, mice with point mutations rendering the α1GABAA receptor insensitive to benzodiazepines did not show a preference for oral midazolam vs sucrose solutions, in contrast to wild-type mice (Tan et al, 2010). We have shown, however, that the α1-sparing compound, L-838,417, was reliably self-administered by rhesus monkeys (Rowlett et al, 2005), a finding clearly inconsistent with the hypothesis that α1GABAA receptors mediate the reinforcing effects of benzodiazepines.
The reasons for the discrepancies in the results with TPA023 and L-838,417 are unclear. A possibility is the differences in the pharmacological profiles of the two compounds: TPA023 has notably lower intrinsic efficacy in vitro (measured via Cl- conductance in cloned human receptor subtypes) than L-838,417, with the highest efficacy for TPA023 at α3GABAA receptors whereas L-838 417 is equi-effective at α2GABAA, α3 GABAA, and α5GABAA receptors, raising the possibility that differences in levels of intrinsic efficacy could be key factors mediating self-administration (for reviews, see Licata and Rowlett, 2008; Atack, 2011). Moreover, TPA023 likely has a relatively long duration of action (Atack et al, 2006, 2010; Ator et al, 2010) while that of L-838 417 is shorter (Rowlett et al, 2005; Licata et al, 2010; J.R. Atack, unpublished data). Benzodiazepines with relatively long durations of action typically are less robustly self-administered compared with short-acting drugs (Griffiths and Weerts, 1997; Platt and Rowlett, 2012).
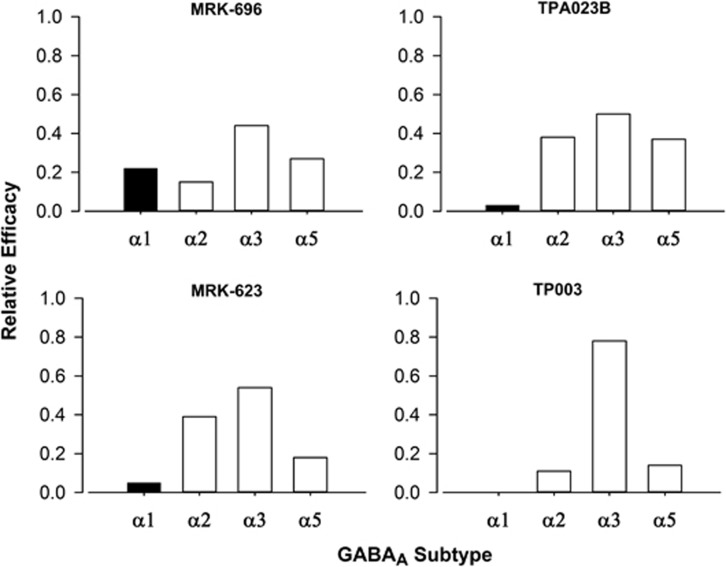
To address the extent to which our findings with L-838,417 generalize to compounds with different pharmacodynamic and pharmacokinetic characteristics, the present study evaluated the reinforcing effects of novel α1-sparing compounds from different chemical classes with different relative durations of action and functional selectivity profiles. These compounds included a long-acting imidazotriazine referred to as TPA023B, the relatively short-acting imidazopyrimidine MRK-623, and the relatively long-acting fluoroimidazopyridine TP003. Each of the novel compounds has a non-selective binding affinity profile (see Table 1), but demonstrates higher intrinsic efficacy at one or more receptor subtypes compared with the α1GABAA receptor (ie, they are functionally selective in vitro; see Figure 1).
Table 1. Potencies for Novel Compounds at GABAA Receptors (Affinities) and in Self-Administration (Dose Engendering Half-Maximal Effects, ED50) Under Midazolam Baseline Conditions.
|
GABAA
subtype (αxβ3γ2) |
|||||
|---|---|---|---|---|---|
|
α1 |
α2 |
α3 |
α5 |
||
| Compound | Ki (nM)a | Self-administrationED50(mean mg/kg/injection± SEM)b | |||
| MRK-696 | 0.14 | 0.22 | 0.11 | 0.14 | 0.0011±0.0009 |
| TP003 | 0.32 | 0.54 | 0.50 | 0.26 | 0.49±0.21 |
| TPA023B | 0.73 | 2.00 | 1.80 | 1.10 | 0.0032±0.001 |
| MRK-623 | 0.85 | 3.70 | 4.00 | 0.53 | 0.09±0.01 |
Ki values determined from radioligand binding assays with [3H]flumazenil in human recombinant GABAA receptors containing β3 and γ2 subunits, combined with α1, α2, α3, or α5 subunits (adapted from Atack, 2011).
ED50 values determined for monkeys responding under a progressive-ratio schedule of i.v. midazolam injection (‘midazolam baseline' group). Data are from N=4 rhesus monkeys.
Figure 1.
Intrinsic efficacy profiles of novel compounds with functional selectivity for GABAA receptor subtypes. Intrinsic efficacy values were determined in human recombinant GABAA receptors containing β3 and γ2 subunits, combined with α1, α2, α3, or α5 subunits, using whole-cell patch-clamp electrophysiology. Values are expressed relative to the efficacy measured at each subtype using the nonselective high efficacy benzodiazepine agonist chlordiazepoxide. Graphs are adapted from (Atack 2010, 2011).
Although there are several differences between the studies of Ator et al (2010) and Rowlett et al (2005), one potentially important difference is training/maintenance drug (cocaine vs methohexital). Drug history, including the type of drug used for self-administration training, has been shown to be a major determinant of the reinforcing effects of benzodiazepines (Nelson et al, 1983; Bergman and Johanson, 1985; Falk and Tang, 1989). Therefore, we examined self-administration of the different α1-sparing compounds described above in monkeys trained to self-administer either a benzodiazepine agonist (midazolam), or the psychomotor stimulant cocaine, in order to match the training conditions of Ator et al (2010). For comparisons across the two baseline conditions, we also included tests with non-selective benzodiazepine full agonists (midazolam, lorazepam) and a non-selective benzodiazepine partial agonist (MRK-696, see Table 1; Figure 1).
MATERIALS AND METHODS
Subjects, Surgery, and Design
Eight adult rhesus monkeys (Macaca mulatta; four males, four females) were housed in a colony room with a 12 h light/dark cycle, had unrestricted access to water, and were fed Harlan Teklad monkey diet, supplemented by fresh fruit. Animals were maintained in accordance with the guidelines of the Committee on Animals of the Harvard Medical School and the Guide for Care and Use of Laboratory Animals (eighth edition, 2011). Research protocols were approved by the Harvard Medical School Institutional Animal Care and Use Committee.
Monkeys were prepared with chronic indwelling venous catheters (polyvinyl chloride, i.d.: 0.64 mm; o.d.: 1.35 mm) following the general surgical procedures described by (Platt et al, 2011). Monkeys were anesthetized with 10–20 mg/kg i.m. of ketamine for preparation for the procedure. Throughout surgery, anesthesia was maintained by an isofluorane/oxygen mixture. Under aseptic conditions, a catheter was implanted in the femoral, brachial, or jugular vein and passed to the level of the right atrium. The distal end of the catheter was passed subcutaneously and exited in the mid-scapular region. The external end of the catheter was fed through a fitted jacket and tether system (Lomir Biomedical, Toronto, Canada) and attached to a fluid swivel mounted to the animal's cage. The catheters were flushed daily with heparinized saline (150–200 U/ml).
Two groups of monkeys (each consisting of two males and two females) were trained to self-administer either the benzodiazepine agonist midazolam (0.03 mg/kg/injection, referred to as ‘midazolam baseline' group) or cocaine (0.03 mg/kg/injection, referred to as ‘cocaine baseline' group) under a progressive-ratio (PR) schedule of i.v. drug injection (Rowlett et al, 2005; Rowlett and Lelas, 2007). Previously, the monkeys in the midazolam baseline group had received single-day tests with other benzodiazepines (clonazepam, diazepam, alprazolam, triazolam) over an approximately 6-month period (unpublished). The monkeys in the cocaine baseline group had received single-day tests with cocaine and/or opioid agonists (alfentanil and nalbuphine) over the course of approximately a year (Rowlett et al, 2002). Both groups of monkeys had received numerous injections of ketamine as part of routine clinical care.
Self-Administration Training and Testing
Each monkey's cage was fitted with a custom-made panel (MetalSmiths) containing stimulus lights and levers (Med Associates). At the beginning of a session, a set of two white stimulus lights above a response lever was illuminated. Upon completion of a response requirement, the white lights were extinguished and a set of two red stimulus lights were illuminated for 1 s, coinciding with a 1-s infusion. Each trial ended with either an injection or the expiration of a 30-min limited hold. Trials were separated by a 30-min timeout period, during which all lights were extinguished and responding had no programmed consequences.
Experimental sessions consisted of five components made up of four trials each. The response requirement remained constant for each of the four trials within a component, and doubled during each successive component. The session ended when a monkey self-administered a maximum of 20 injections or when the response requirement was not completed for two consecutive trials. The PR schedule consisted of the sequence of response requirements 40, 80, 160, 320, and 640 responses per injection. Once performance was stable under these conditions (no increasing or decreasing trend in the number of injections/session for three consecutive sessions), drug (midazolam or cocaine, depending on the baseline condition) or saline was available on alternating days. Test sessions (T) with experimental compounds were added to the alternating sequence of drug (D) and saline (S) sessions according to the following sequence: DTSDTSTDST, etc.
Functional Selectivity Profiles and Drug/Compound Information
Three novel α1-sparing compounds were evaluated: TPA023B, MRK-623, and TP003 (see Table 1 and Figure 1 for details). TPA023B is functionally selective for α2GABAA, α3GABAA, and α5GABAA receptors, similar to the previously-tested L-838,417, whereas MRK-623 exhibits functional selectivity at α2GABAA and α3GABAA receptors compared with α1GABAA and α5GABAA receptors. In contrast, TP003 has appreciable intrinsic efficacy for α3GABAA receptors, but essentially no measurable intrinsic efficacy at α1GABAA, α2GABAA, and α5GABAA receptors. Therefore, in addition to providing information about the necessity of action at α1GABAA receptors for self-administration, these studies provided key information on the role of α2GABAA, α3GABAA, and α5GABAA receptors in the reinforcing effects of benzodiazepine-type drugs. Comparisons were made with conventional benzodiazepines (midazolam, lorazepam) as well as with a non-selective partial agonist, MRK-696.
All drugs were administered intravenously. The base forms of midazolam (0.003—0.1 mg/kg) and lorazepam (0.001—0.03 mg/kg) (Sigma-Aldrich, St Louis, MO) were dissolved in small amounts of 95% ethanol or 100% propylene glycol, and diluted to desired concentration using propylene glycol (50%) and water. Cocaine HCl (Sigma-Aldrich, St Louis, MO) was dissolved in 0.9% saline solution. MRK-696, 7-Cyclobutyl-6-(2-methyl-2H-1,2,4-triazol-3-ylmethoxy)-3-(2-fluorophenyl)-1,2,4-triazolo(4,3-b)pyridazine (0.001—0.03 mg/kg) was prepared in 100% propylene glycol and diluted using 50–80% propylene glycol and water solutions. MRK-623, 2-(3-(4-fluoro-3-pyridin-3-yl-phenyl)-imidazo(1,2-a)pyrimidin-7-yl)-propan-2-ol (0.03—3.0 mg/kg); TPA023B, 6,2′-difluoro-5′-(3-(1-hydroxy-1-methylethyl)imidazo(1,2-b((1,2,4)triazin-7-yl) (1,1′-biphenyl)-2-carbonitrile (0.003—0.3 mg/kg); and TP003, 4,2′-difluoro-5′-(8-fluoro-7-(1-hydroxy-1-methylethyl)imidazo(1,2-a)pyridine-3-yl)biphenyl-2-carbonitrile (0.1—1.8 mg/kg) were synthesized at the Merck, Sharp & Dohme Neuroscience Research Centre (Terlings Park, Harlow, UK) as summarized by (Atack 2011). MRK-623, TPA023B, and TP003 were prepared in solutions of 10% benzoyl ethanol, 50% propylene glycol, and 40% water.
Data Analysis
The number of injections/session and the last response requirement completed in a session (break point, BP) were determined for individual monkeys for each test drug/dose. The injections/session data were analyzed by repeated measures analysis of variance (ANOVA) with dose as the factor. A dose of drug was determined to be self-administered significantly above vehicle levels by comparing mean injections/session for each dose to the corresponding vehicle control value (Bonferroni t-test, alpha level equal to P<0.05).
The maximum BP (BPmax) was calculated as the highest BP, irrespective of dose, for each test drug. The BPmax measure provides an index of reinforcing strength that takes into account individual differences in peak BP values. Medians rather than means were used for this analysis because BP values characteristically violate the assumption of homogeneity of variance for parametric tests (Rowlett et al, 1996, 2002). The BPmax data were analyzed with the nonparametric Friedman's repeated measures ANOVA on ranks. Multiple comparisons of BPmax medians to those for midazolam were conducted using Dunnett's tests for ranks (P<0.05).
The doses of compound needed to engender 50% of the maximum effect (ED50) were calculated based on the number of injections/session data, only after significant self-administration was obtained (ie, at least one dose of compound engendered an average number of injections/session that was significantly different from vehicle). The ED50 values were determined by non-linear regression analysis. An iterative curve-fitting procedure for sigmoidal dose-response functions with variable slopes was used (Rowlett, 2000). The equation used was the four-parameter logistic equation: Y = min + max−(max/(1+eslope/dose—ED50)), where min equals the number of injections/session obtained with vehicle availability and max equals the highest values obtained with compound. All parameters in the nonlinear regression analysis were free to vary.
RESULTS
Self-Administration of Midazolam and Lorazepam
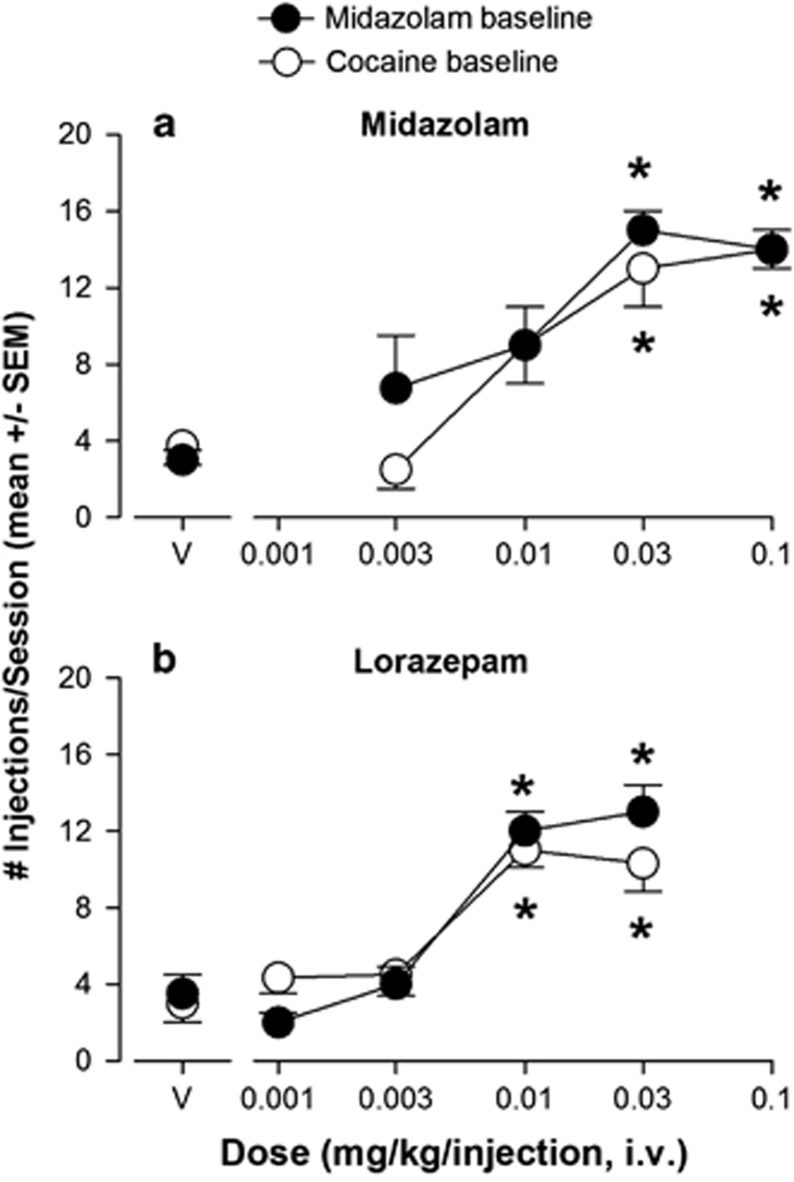
Under midazolam baseline conditions, the 0.03 mg/kg/injection dose of midazolam maintained an average of 15 injections/session (SEM=1.0) whereas saline availability resulted in an average of 3.5 injections/session (SEM=0.57; data not shown). Under cocaine baseline conditions, the 0.03 mg/kg/injection dose of cocaine maintained an average of 17 injections/session (SEM=1.7) whereas saline availability resulted in an average of 3.0 injections/session (SEM=1.0; data not shown). Note there were no differences between the two baseline conditions when comparing the mean numbers of injections/session after baseline drug (midazolam vs cocaine), or between the groups in terms of mean numbers of injections/session following vehicle availability (t-tests, P>0.05). No substantial changes in baseline responding occurred for either drug during the ∼1.5 year duration of this study.
As anticipated, both midazolam and lorazepam were self-administered reliably above vehicle levels when the number of injections/session was analyzed (Figure 2). Doses of midazolam at 0.03 and 0.1 mg/kg/injection were self-administered significantly above vehicle levels under both midazolam- and cocaine-baseline conditions (Figure 2a., Bonferroni t-tests, P<0.05 vs vehicle). When the conventional benzodiazepine agonist lorazepam was made available to these monkeys, significant self-administration was observed under both baseline conditions (Figure 2b). When 0.001−0.03 mg/kg/injection of lorazepam was made available to monkeys under midazolam baseline conditions, the mean number of injections/session of the two highest doses (0.01 and 0.03 mg/kg/injection) but not the two lower doses were significantly above vehicle levels (Figure 2b, Bonferroni t-tests, P<0.05 vs vehicle). Similarly, under cocaine baseline conditions, lorazepam maintained mean number of injections/session above vehicle levels for 0.01 and 0.03 mg/kg/injection (Figure 2b, Bonferroni t-tests, P<0.05 vs vehicle).
Figure 2.
Self-administration of conventional benzodiazepine agonists under a progressive-ratio schedule of i.v. midazolam or cocaine injection. (a) Intravenous self-administration of midazolam by rhesus monkeys trained to press a lever to obtain injections of 0.03 mg/kg/injection of midazolam (N=4; filled symbols, ‘midazolam baseline') or 0.03 mg/kg/injection of cocaine (N=4; open symbols, ‘cocaine baseline'). Data are mean (± SEM) injections per session, out of a total of 20 injections available in a daily session. Points above ‘V': vehicle tests. Note that *P<0.05 vs vehicle (Bonferroni t-tests). (b) Self-administration of lorazepam under the same conditions as described for panel a.
MRK-696 and Functionally-Selective Compounds
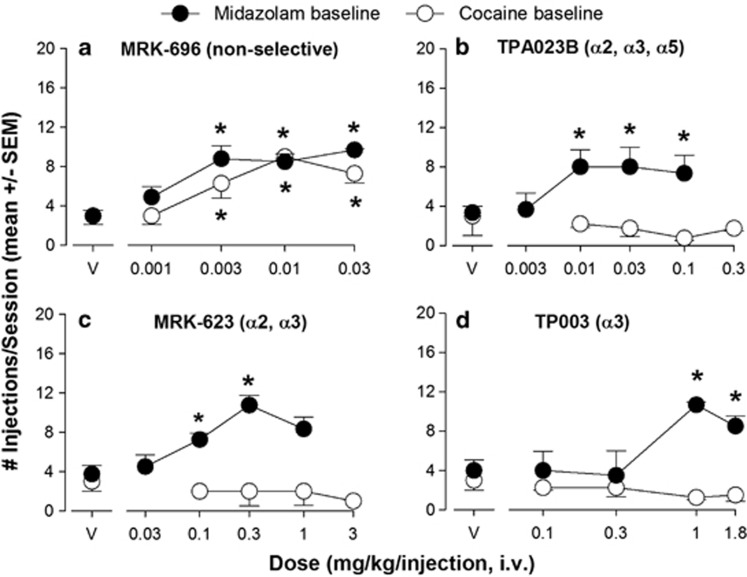
After establishment of self-administration of the conventional benzodiazepines midazolam and lorazepam under both baseline conditions, we evaluated the four novel compounds for self-administration. As with the non-selective conventional benzodiazepines, the non-selective partial agonist MRK-696 maintained a mean number of injections/session above vehicle levels at doses of 0.003–0.03 mg/kg/injection under both midazolam and cocaine conditions (Figure 3a, Bonferroni t-tests, P<0.5 vs vehicle).
Figure 3.
Differential self-administration of compounds with varying degrees of efficacy GABAA receptor subtypes. (a) Intravenous self-administration of MRK-696 (no differences in efficacy across subtypes) in rhesus monkeys trained under a progressive-ratio schedule of midazolam (0.03 mg/kg/injection; N=4, ‘midazolam baseline') or cocaine (0.03 mg/kg/injection, N=4; ‘cocaine baseline'). Data are mean (± SEM) injections per session, out of a total of 20 injections available in a daily session. Points above ‘V': vehicle tests. Note that *P<0.05 vs vehicle (Bonferroni t-tests). (b) Self-administration of TPA023B (near zero efficacy at α1 subunit-containing receptors, ie, ‘α1-sparing' partial agonist at α2, α3, and α5 subunit-containing receptors) under the same conditions as described for panel a. (c) Self-administration of MRK-623 (near zero efficacy at α1 subunit-containing receptors, ie, ‘α1-sparing' highest efficacy at α2, α3 subunit-containing receptors) under the same conditions as described for panel a. (d) Self-administration of TP003 (zero efficacy at α1 subunit-containing receptors, ie, ‘α1-sparing' highest efficacy at α3 subunit-containing receptors; near zero efficacy at α2 and α5 subunit-containing receptors) under the same conditions as described for panel a. See Figure 1 for receptor subtype selectivity profiles.
For the compounds lacking efficacy at α1GABAA receptors but with functional selectivity for different GABAA receptor subtypes, a different pattern of effects was observed. In this regard, TPA023B, a compound lacking significant efficacy at α1GABAA receptors but with functional selectivity for α2GABAA, α3GABAA and α5GABAA receptors, maintained a mean number of injections above vehicle levels for doses of 0.01, 0.03, and 0.1 mg/kg/injection under the midazolam baseline (Figure 3b, Bonferroni t-tests, P<0.05 vs vehicle). However, this compound did not maintain self-administration under the cocaine baseline, even when a higher dose (0.3 mg/kg/injection) was tested (Figure 3b, Bonferroni t-tests, P>0.05 vs vehicle).
A similar profile of effects was observed for MRK-623, which has functional selectivity for α2GABAA and α3GABAA receptors. MRK-623 maintained a mean number of injections/session above vehicle levels at doses of 0.1 and 0.3 mg/kg/injection in the midazolam baseline group (Figure 3c, Bonferroni t-tests, P<0.05 vs vehicle). Under cocaine baseline conditions, however, MRK-623 did not maintain average injections/session above vehicle levels at any dose, even when the dose was increased above the maximum tested under midazolam conditions (Figure 3c, Bonferroni t-tests, P>0.05 vs vehicle).
Finally, the most selective of the compounds tested, TP003 (functional selectivity for α3GABAA receptors), maintained a mean number of injections/session above vehicle levels at doses of 1.0 and 1.8 mg/kg/injection under the midazolam baseline (Figure 3d, Bonferroni t-tests, P<0.05 vs vehicle). As with TPA023B and MRK-623, TP003 did not maintain a mean number of injections/session above vehicle levels at any dose tested under cocaine baseline conditions (Figure 3d, Bonferroni t-tests, P>0.05 vs vehicle). Note that doses higher than 1.8 mg/kg/injection of TP003 were not evaluated due to solubility limits.
Comparison of Potencies
Potencies based on the number of injections/session could be calculated for MRK-696, TPA023B, MRK-623, and TP003 under midazolam baseline conditions, although note that the extent to which the dose-response functions for both MRK-623 and TP003 are ‘asymptotic', ie, reach a maximum level for several doses, is unclear. Nevertheless, all non-linear regression analyses resulted in relatively high correlation coefficients (R2=0.71—0.93). The ED50 values (±SEM) are shown in Table 1, and reveal a rank order of potency of MRK-696>TPA023B>MRK-623>TP003.
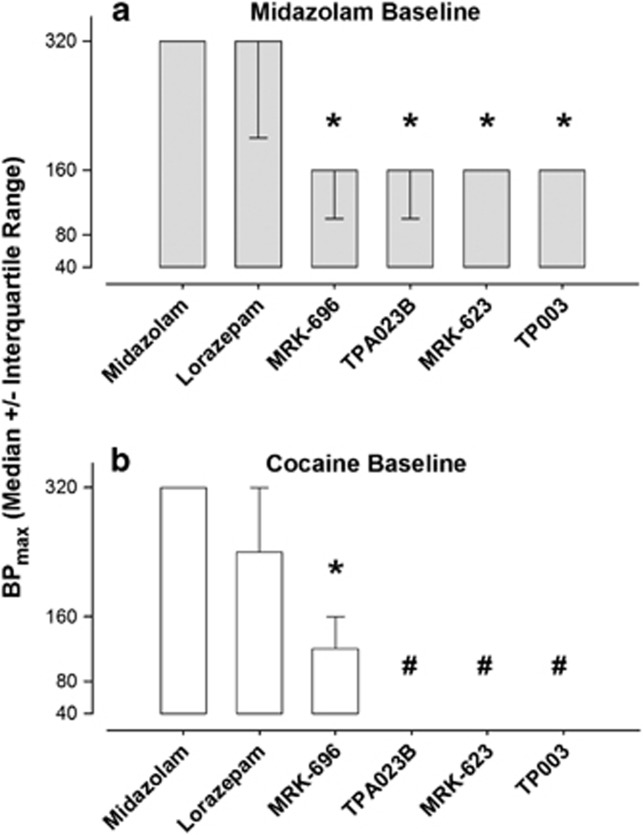
Comparison of Relative Reinforcing Strength
Evaluation of BPmax values (ie, maximum break point, BP, irrespective of dose) showed median values for midazolam and lorazepam of 240−320 responses/injection under both midazolam and cocaine baseline conditions (Figure 4). Under the midazolam baseline condition, comparisons with lorazepam and the four novel compounds revealed significant effects (repeated measures Friedman's ANOVA) that were due to BPmax values for MRK-696, TPA023B, MRK-623, and TP003; but not lorazepam, being lower than the BPmax values obtained with midazolam (Figure 4a, Dunnett's tests, P<0.05).
Figure 4.
Relative reinforcing effectiveness of benzodiazepine agonists and novel compounds under progressive-ratio schedules of midazolam (a) or cocaine (b) injection. Data are derived from break points, ie, the highest response requirement completed in a session, and expressed in terms of BPmax, which is the highest break point obtained irrespective of dose. Data are median (± interquartile range) due to lack of homogeneity of variance associated with break point data sets (see text for details). Note that *P<0.05 vs median BPmax for midazolam, Dunnett's test for ranks. Also note that # indicates that self-administration was not significantly different from vehicle for any dose which did not allow determination of break point values.
Under the cocaine baseline, median BPmax values for TPA023B, MRK-623, and TP003 were not calculated due to the lack of statistical significance between vehicle and any dose of the three compounds (see above). Thus, median BPmax values were calculated for midazolam, lorazepam, and MRK-696 only, and when the values for lorazepam and MRK-696 were compared with those of midazolam, only MRK-696 maintained a lower median BPmax value (Figure 4b, Dunnett's tests, P<0.05).
DISCUSSION
Expanding on our previous findings with the α1-sparing compound L-838,417 (Rowlett et al, 2005), we show that α1-sparing compounds with varying pharmacodynamic and pharmacokinetic profiles can have reinforcing effects in rhesus monkeys. However, self-administration of α1-sparing compounds appears to depend critically on baseline conditions, as all compounds tested had reinforcing effects in monkeys experienced primarily with midazolam, but not in monkeys experienced primarily with cocaine. Under cocaine baseline conditions, our findings are consistent with the prediction advanced by Tan et al (2010) that α1-sparing compounds should be without reinforcing effects, possibly due to a lack of disinhibition of VTA dopaminergic neurons that innervate the NAc. In contrast, our findings that α1-sparing compounds have reinforcing effects under the midazolam baseline condition is more consistent with recent results reported by Reynolds et al (2012), in which the α1GABAA receptor appeared to have little role in benzodiazepine-induced enhancement of reward thresholds in an intra-cranial self-stimulation procedure.
In our studies, the α1-sparing compounds had lower relative reinforcing strength (as determined by the BPmax) than conventional benzodiazepines such as midazolam and lorazepam. These findings collectively suggest that α1-sparing compounds may have reduced abuse liability compared with non-selective benzodiazepines. Conversely, drugs that bind preferentially to α1GABAA receptors have a higher degree of relative reinforcing strength than conventional benzodiazepines and α1-sparing compounds (eg, Ator, 2002; Rowlett et al, 2005). On the basis of these findings, we speculated previously that the α1GABAA subtype might have a facilitative role in the reinforcing effects of benzodiazepines (Rowlett et al, 2005; Licata and Rowlett, 2011). One prediction from this idea is that a non-selective partial agonist would have a higher degree of relative reinforcing strength relative to α1-sparing compounds that are partial agonists at the other GABAA receptors. This prediction is based, in part, on the finding by Licata and Rowlett (2011) that the non-selective partial agonist, bretazenil, had reinforcing strength similar to that of midazolam. However, in the present study, a direct comparison of the non-selective partial agonist MRK-696 with the α1-sparing compounds revealed no differences in relative reinforcing strength; thus providing little support for the idea of a facilitative role for the α1GABAA receptor subtype.
As described above, recent work has implicated α1 subunit-containing GABAA receptors on GABAergic interneurons in the VTA as key mediators of the reinforcing effects of benzodiazepines (Heikkinen et al, 2009; Tan et al, 2010, 2011). In this regard, benzodiazepines would increase inhibition of these neurons, thus relieving their tonic inhibition of DA neurons (Tan et al, 2010, 2011), and resulting in the hallmark DA increase observed with other drugs of abuse (Lüscher and Ungless, 2006). Although these findings assume that increased DA in the NAc is the mechanism for benzodiazepine self-administration, there is no direct evidence to date documenting such an increase. In fact, there is evidence suggesting that benzodiazepines may actually decrease DA release (eg, Lavielle et al, 1979; Finlay et al, 1992; Harada et al, 1992; Murai et al, 1994). Moreover, in healthy drug-inexperienced people, benzodiazepine-type drugs and drugs that bind preferentially to α1 subunit-containing receptors (eg, zolpidem) are characteristically found to be unpleasant and/or aversive (Licata and Rowlett, 2008; Licata et al, 2008).
Although no unifying hypothesis of the reinforcing/rewarding effects of benzodiazepine-type drugs is evident at this time, our findings with different training baselines may help to reconcile some aspects of the differing results across species and procedures. Notably, the suggestion by (Tan et al, 2010, 2011) that α1-sparing compounds should lack or have reduced reinforcing effects is supported by our findings, but only with cocaine-experienced monkeys. Moreover, this observation is generally consistent with an earlier report that the conventional benzodiazepine agonist diazepam was not self-administered by a majority of monkeys trained to self-administer cocaine, but was self-administered by all monkeys trained with the barbiturate GABAergic modulator, pentobarbital (Bergman and Johanson, 1985). One potential implication of these findings is that exposure to GABAergic positive modulators vs cocaine exposure results in different neuroplastic outcomes, such that increases in DA neurotransmission in the mesolimbic system are critical for reinforcing effects of benzodiazepines after cocaine exposure, but not midazolam (or pentobarbital) exposure. That is, the hypothesis of (Tan et al, 2010, 2011) that α1GABAA receptors mediate the addictive effects of benzodiazepines applies to cocaine-exposed, but not midazolam-exposed subjects.
In addition to differences in neuroplasticity, there are other possible explanations for the divergence in self-administration of α1-sparing compounds for midazolam- vs cocaine-experienced monkeys to consider. For example, self-administration of α1-sparing compounds might depend on the extent to which compounds share discriminative stimulus effects with the two training drugs. Cocaine characteristically does not share discriminative stimulus effects with benzodiazepines (eg, Negus et al, 2000). However, in the present study, midazolam and lorazepam, as well as the non-selective partial agonist MRK-696, were self-administered under the cocaine baseline condition in a manner similar to the midazolam baseline condition, both in terms of potency and relative reinforcing strength. Conversely, we have shown previously that the α1-sparing compound L-838,417 was self-administered under a methohexital baseline, yet did not share discriminative stimulus effects with the non-selective BZ, triazolam (Rowlett et al, 2005). These findings suggest that the differences in self-administration between midazolam vs cocaine baseline conditions were not simply a matter of benzodiazepine-like vs cocaine-like discriminative stimulus effects. These results are in line with previous studies, which have demonstrated that animals will self-administer drugs that do not share discriminative stimuli with the training drug (Ator, 2002). Another possibility is that α1-sparing compounds possess effects that suppress, or impair, lever pressing; and these effects are attenuated in midazolam- but not cocaine-experienced monkeys. However, α1-sparing compounds generally lack sedative-motor effects in monkeys and do not impair performance in tasks requiring lever pressing (Rowlett et al, 2005; Fischer et al, 2010, 2011).
Although our findings are not consistent with the idea that α1-sparing compounds lack abuse potential entirely, we have evidence for a GABAA receptor subtype that is perhaps critical for mediating the reinforcing effects of benzodiazepines, at least under midazolam baseline conditions. In this regard, TP003, a compound with functional selectivity for the α3GABAA receptor (Dias et al, 2005), had reinforcing effects in the benzodiazepine-experienced monkeys. Interestingly, selective activation of the α3GABAA receptor would be predicted to decrease DA release in the NAc (Tan et al, 2011). If borne out, the combination of TP003 self-administration with decreased DA release in the NAc would provide a clear exception to current hypotheses regarding the neurobiological mechanisms of reinforcement.
Although provocative, the results with TP003 must be approached with some caution. In particular, the rank order of potencies of the α1-sparing compounds did not match the rank order of potencies for binding affinities for any of the GABAA receptor subtypes. This mismatch was due primarily to TP003 having relatively high potency in vitro, but the lowest potency of all the compounds in self-administration. This raises the possibility of TP003 acting at a non-GABAA site, although there is no evidence to date for this compound having activity at other receptor systems (Dias et al, 2005). One possible explanation for this relative lack of in vivo potency for TP003 is that this compound consistently requires a high degree of binding site occupancy to engender behavioral effects, in contrast to most benzodiazepine-type compounds (Dias et al, 2005). Nevertheless, it is unknown at present what degree of binding site occupancy is required for behavioral effects in rhesus monkeys and, in fact, the pharmacokinetic profile of TP003 in this species has not yet been established.
Given that drugs that bind preferentially to α1GABAA receptors (eg, zolpidem) have reinforcing effects, we propose that intrinsic efficacy at α3GABAA receptors alone is sufficient for benzodiazepine self-administration under midazolam baseline conditions. In contrast, intrinsic efficacy at α1GABAA receptors is necessary for benzodiazepine self-administration under cocaine baseline conditions. The neurobiological basis for these differential mechanisms of action remains to be discovered.
Acknowledgments
This project was funded by an unrestricted research grant from Merck & Co., USPHS grants DA011792, DA033795 (JKR), AA016179 (DMP); and RR000168 (currently Office of Research Infrastructure Programs grant OD011103). The authors would like to thank Donna Reed, Annemarie Duggan, and Laura Teixeira for technical assistance.
During the course of these studies, the research was supported, in part, by a research agreement from Merck Research Laboratories with Dr Rowlett as the Principal Investigator. During the studies, Drs Atack, Reynolds, and Dawson were employees of Merck Research Laboratories (Merck, Sharp and Dohme, Ltd.). Ms. Shinday, Drs Sawyer, Fischer, Platt, and Licata had no financial relationship with Merck Research Laboratories. The agreement between Harvard Medical School and Merck Research Laboratories was a scientific collaboration, and did not involve studies that by the publication thereof would engender financial gain or loss from public disclosure. Therefore, the authors declare no conflict of interest.
References
- Atack JR. GABAA receptor alpha2/alpha3 subtype-selective modulators as potential nonsedating anxiolytics. Curr Top Behav Neurosci. 2010;2:331–360. doi: 10.1007/7854_2009_30. [DOI] [PubMed] [Google Scholar]
- Atack JR. GABAA receptor subtype-selective modulators. I. α2/α3-selective agonists as non-sedating anxiolytics. Curr Top Med Chem. 2011;11:1176–1202. doi: 10.2174/156802611795371350. [DOI] [PubMed] [Google Scholar]
- Atack JR, Wafford KA, Tye SJ, Cook SM, Sohal B, Pike A, et al. TPA023 (7-(1,1-dimethylethyl)-6-(2-ethyl-2H-1,2,4-triazol-3-ylmethoxy)-3-(2-fluorophenyl)-1,2,4-triazolo(4,3-b)pyridazine), an agonist selective for alpha2- and alpha3-containing GABAA receptors, is a nonsedating anxiolytic in rodents and primates. J Pharmacol Exp Ther. 2006;316:410–422. doi: 10.1124/jpet.105.089920. [DOI] [PubMed] [Google Scholar]
- Ator NA. Relation between discriminative and reinforcing effects of midazolam, pentobarbital, chlordiazepoxide, zolpidem, and imidazenil in baboons. Psychopharmacology (Berl) 2002;163:477–487. doi: 10.1007/s00213-002-1076-4. [DOI] [PubMed] [Google Scholar]
- Ator NA, Atack JR, Hargreaves RJ, Burns HD, Dawson GR. Reducing abuse liability of GABAA/benzodiazepine ligands via selective partial agonist efficacy at alpha1 and alpha2/3 subtypes. J Pharmacol Exp Ther. 2010;332:4–16. doi: 10.1124/jpet.109.158303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergman J, Johanson C-E. The reinforcing properties of diazepam under several conditions in the rhesus monkey. Psychopharmacology (Berl) 1985;86:108–113. doi: 10.1007/BF00431693. [DOI] [PubMed] [Google Scholar]
- Ciccarelli A, Calza A, Panzanelli P, Concas A, Giustetto M, Sassoè-Pognetto M. Organization of GABAergic synaptic circuits in the rat ventral tegmental area. PLoS One. 2012;7:e46250. doi: 10.1371/journal.pone.0046250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dias R, Sheppard WF, Fradley RL, Garrett EM, Stanley JL, Tye SJ, et al. Evidence for a significant role of alpha 3-containing GABAA receptors in mediating the anxiolytic effects of benzodiazepines. J Neurosci. 2005;46:10682–10688. doi: 10.1523/JNEUROSCI.1166-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falk JL, Tang M. Schedule-induced chlordiazepoxide intake: differential effect of cocaine and ethanol histories. Pharmacol Biochem Behav. 1989;33:393–396. doi: 10.1016/0091-3057(89)90520-0. [DOI] [PubMed] [Google Scholar]
- Finlay J, Damsma G, Fibiger H. Benzodiazepine-induced decreases in extracellular concentrations of dopamine in the nucleus accumbens after acute and repeated administration. Psychopharmacology (Berl) 1992;106:202–208. doi: 10.1007/BF02801973. [DOI] [PubMed] [Google Scholar]
- Fischer BD, Atack JR, Platt DM, Reynolds DS, Dawson GR, Rowlett JK. Contribution of GABAA receptors containing alpha 3 subunits to the therapeutic-related and side effects of benzodiazepine-type drugs in monkeys. Psychophamacology (Berl) 2011;2:311–319. doi: 10.1007/s00213-010-2142-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer BD, Licata SC, Edwankar RV, Wang ZJ, Huang S, He X, et al. Anxiolytic-like effects of 8-acetylene imidazobenzodiazepines in a rhesus monkey conflict procedure. Neuropharmacology. 2010;59:612–618. doi: 10.1016/j.neuropharm.2010.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritschy JM, Mohler H. GABAA-receptor heterogeneity in the adult rat brain: differential regional and cellular distribution of seven major subunits. J Comp Neurol. 1995;359:154–194. doi: 10.1002/cne.903590111. [DOI] [PubMed] [Google Scholar]
- Griffiths RR, Weerts EM. Benzodiazepine self-administration in humans and laboratory animals-implications for problems of long-term use and abuse. Psychopharmacology (Ber) 1997;134:1–37. doi: 10.1007/s002130050422. [DOI] [PubMed] [Google Scholar]
- Harada K, Noguchi K, Wakusawa R. Effects of immobilization stress and of a benzodiazepine derivative on rat central dopamine system. J Anesth. 1992;6:167–171. doi: 10.1007/s0054020060167. [DOI] [PubMed] [Google Scholar]
- Heikkinen AE, Möykkynen TP, Korpi ER. Long-lasting modulation of glutamatergic transmission in VTA dopamine neurons after a single dose of benzodiazepine agonists. Neuropsychopharmacology. 2009;34:290–298. doi: 10.1038/npp.2008.89. [DOI] [PubMed] [Google Scholar]
- Lavielle S, Tassin JP, Thierry AM, Blanc G, Herve D, Barthelemy C, et al. Blockade by benzodiazepines of the selective high increase in dopamine turnover induced by stress in mesocortical dopaminergic neurons of the rat. Brain Res. 1979;168:585–594. doi: 10.1016/0006-8993(79)90311-1. [DOI] [PubMed] [Google Scholar]
- Licata SC, Platt DM, Rüedi-Bettschen D, Atack JR, Dawson GR, Van Linn ML, et al. Discriminative stimulus effects of L-838,417 (7-tert-butyl-3-(2,5-difluoro-phenyl)-6-(2-methyl-2H-(1,2,4)triazol-3-ylmethoxy)-(1,2,4)triazolo(4,3-b)pyridazine): role of GABAA receptor subtypes. Neuropharmacology. 2010;58:357–364. doi: 10.1016/j.neuropharm.2009.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Licata SC, Rowlett JK. Abuse and dependence liability of benzodiazepine-type drugs: GABAA receptor modulation and beyond. Pharmacol Biochem Behav. 2008;90:74–89. doi: 10.1016/j.pbb.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Licata SC, Rowlett JK. Self-administration of bretazenil under progressive-ratio schedules: behavioral economic analysis of the role intrinsic efficacy plays in the reinforcing effects of benzodiazepines. Drug Alcohol Depend. 2011;113:157–164. doi: 10.1016/j.drugalcdep.2010.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lüscher C, Ungless MA. The mechanistic classification of addictive drugs. PLoS Med. 2006;3:e437. doi: 10.1371/journal.pmed.0030437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murai T, Koshikawa N, Kanayama T, Takada K, Tomiyama K, Kobayashi M. Opposite effects of midazolam and beta-carboline-3-carboxylate ethyl ester on the release of dopamine from rat nucleus accumbens measured by in vivo microdialysis. Eur J Pharmacol. 1994;261:65–71. doi: 10.1016/0014-2999(94)90301-8. [DOI] [PubMed] [Google Scholar]
- Negus SS, Mello NK, Fival PA. Effects of GABA agonists and GABAA receptor modulators on cocaine discrimination in rhesus monkeys. Psychopharmacology (Berl) 2000;152:398–407. doi: 10.1007/s002130000543. [DOI] [PubMed] [Google Scholar]
- Nelson LR, Potthoff AD, Ellison GD. Increased consumption of diazepam during continuous amphetamine administration. Pharmacol Biochem Behav. 1983;18:863–865. doi: 10.1016/s0091-3057(83)80006-9. [DOI] [PubMed] [Google Scholar]
- Platt DM, Carey G, Spealman RD. Models of neurological disease (substance abuse): self-administration in monkeys. John Wiley & Sons, Inc.: Hoboken, NJ; Current Protocols in Pharmacology. 2011;56:10.5.1–10.5.17. doi: 10.1002/0471141755.ph1005s56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platt DM, Rowlett JK.2012Nonhuman primates in drug and alcohol addiction researchIn: Abee C, Mansfield K, Morris T, Tardif S, (eds).Nonhuman Primates in Biomedical Research 2Elsevier Press: Waltham; 817–839. [Google Scholar]
- Reynolds LM, Engin E, Tantillo G, Lau HM, Muschamp JW, Carlezon WA, et al. Differential Roles of GABAA receptor subtypes in benzodiazepine-induced enhancement of brain-stimulation reward. Neuropsychopharmacology. 2012;37:2531–2540. doi: 10.1038/npp.2012.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowlett JK. A labor-supply analysis of cocaine self-administration under progressive-ratio schedules: antecedents, methodologies, and perspectives. Psychopharmacology (Berl) 2000;153:1–16. doi: 10.1007/s002130000610. [DOI] [PubMed] [Google Scholar]
- Rowlett JK, Lelas S. Comparison of zolpidem and midazolam self-administration under progressive-ratio schedules: consumer demand and labor supply analyses. Exp Clin Psychopharmacology. 2007;15:328–337. doi: 10.1037/1064-1297.15.4.328. [DOI] [PubMed] [Google Scholar]
- Rowlett JK, Massey BW, Kleven MS, Woolverton WL. Parametric analysis of cocaine self-administration under a progressive-ratio schedule in rhesus monkeys. Psychopharmacology (Berl) 1996;125:361–370. doi: 10.1007/BF02246019. [DOI] [PubMed] [Google Scholar]
- Rowlett JK, Platt DM, Lelas S, Atack JR, Dawson GR. Different GABAA receptor subtypes mediate the anxiolytic, abuse-related, and motor effects of benzodiazepine-type drugs in primates. Proc. Natl. Acad. Sci. USA. 2005;102:915–920. doi: 10.1073/pnas.0405621102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowlett JK, Rodefer JS, Spealman RD. Self-administration of cocaine, alfentanil, and nalbuphine under progressive-ratio schedules: Consumer demand and labor supply analyses of relative reinforcing effectiveness. Exp Clin Psychopharmacol. 2002;10:367–375. doi: 10.1037//1064-1297.10.4.367. [DOI] [PubMed] [Google Scholar]
- Rudolph U, Knoflach F. Beyond classical benzodiazepines: novel therapeutic potential of GABAA receptor subtypes. Nat Rev Drug Disc. 2011;10:685–697. doi: 10.1038/nrd3502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan KR, Brown M, Labouèbe G, Yvon C, Creton C, Fritschy JM, et al. Neural bases for addictive properties of benzodiazepines. Nature. 2010;463:769–775. doi: 10.1038/nature08758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan KR, Rudolph U, Luscher C. Hooked on benzodiazepines: GABAA receptor subtypes and addiction. Trends Neurosci. 2011;4:188–197. doi: 10.1016/j.tins.2011.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]