Abstract
The new superbug Neisseria gonorrhoeae has retained resistance to antimicrobials previously recommended for first-line treatment and has now demonstrated its capacity to develop resistance to the extended-spectrum cephalosporin, ceftriaxone, the last remaining option for first-line empiric treatment of gonorrhea. An era of untreatable gonorrhea may be approaching, which represents an exceedingly serious public health problem. Herein, we review the evolution, origin and spread of antimicrobial resistance and resistance determinants (with a focus on extended-spectrum cephalosporins) in N. gonorrhoeae, detail the current situation regarding verified treatment failures with extended-spectrum cephalosporins and future treatment options, and highlight essential actions to meet the large public health challenge that arises with the possible emergence of untreatable gonorrhea. Essential actions include: implementing action/response plans globally and nationally; enhancing surveillance of gonococcal antimicrobial resistance, treatment failures and antimicrobial use/misuse; and improving prevention, early diagnosis and treatment of gonorrhea. Novel treatment strategies, antimicrobials (or other compounds) and, ideally, a vaccine must be developed.
Keywords: action plan, antimicrobial resistance, cefixime, ceftriaxone, cephalosporins, Neisseria gonorrhoeae, penA, surveillance, treatment failure
Public health concerns regarding gonorrhea & antimicrobial-resistant Neisseria gonorrhoeae
The sexually transmitted infection gonorrhea (etiological agent Neisseria gonorrhoeae) is associated with high morbidity and socioeconomic consequences, remaining a major public health concern worldwide. In 2008, the WHO estimated 106 million new gonorrhea cases among adults (a similar global incidence to genital chlamydial infections), which represented a 21% increase since 2005 [201]. If gonorrhea remains undetected and/or untreated, the additional morbidity may be severe and includes pelvic inflammatory disease, infertility, ectopic pregnancy, first trimester abortion, neonatal conjunctivitis leading to blindness and, less commonly, male infertility and disseminated gonococcal infections. Gonorrhea also enhances the transmission of HIV [1,2,202]. Control of gonorrhea relies on prevention, appropriate diagnostics and effective antimicrobial treatment [1]. Antimicrobial therapy should cure individual cases to reduce the risk of complications as well as end further transmission of the infection, which is an important public health strategy to decrease the gonorrhea burden in a population.
The first truly effective antigonococcal agent, penicillin G, was introduced in 1943 and was effective for about 40 years (albeit by repeated increases in the dose) until widespread resistance necessitated a change to other antimicrobials. Unfortunately, antimicrobial resistance (AMR) has emerged for essentially all antimicrobials following their introduction into clinical practice. Over the last 70–80 years, treatment options have diminished rapidly due to the emergence and spread of AMR to all drugs previously used or considered for first-line treatment (penicillins, tetracyclines, spectinomycin, narrow-spectrum cephalosporins, amphenicols, sulfonamide and trimethoprim combinations, macrolides and fluoroquinolones). Currently, the extended-spectrum cephalosporins (ESCs), ceftriaxone (injectable) and cefixime (oral), are the only first-line options for the antimicrobial monotherapy of gonorrhea in most settings globally; however, during the most recent decade, global susceptibility to these antibiotics has markedly decreased [1,3–20] and clinical treatment failures with these ESCs have been verified [21–30]. In particular, it is of concern that during the last 2 years, the first three extensively drug-resistant (XDR; defined in [1]) N. gonorrhoeae strains with high-level resistance to ceftriaxone, the last remaining option for empiric single antimicrobial treatment, were reported from Japan [23], France [25] and Spain [31]. The threat of widespread ceftriaxone resistance and untreatable gonorrhea, particularly in settings where dual antimicrobial therapy is not feasible and/or affordable, is real [1,4,17,23,25,32]. In response to this developing situation, the WHO has published the ‘Global Action Plan to Control the Spread and Impact of Antimicrobial Resistance in Neisseria gonorrhoeae’ [33,202]. The European CDC and the US CDC have published region-specific response plans for the EU/European Economic Area (EU/EEA) countries [203] and the USA, respectively [204].
Evolution, origin & spread of AMR in N. gonorrhoeae
Since the beginning of the antimicrobial era (1930s), N. gonorrhoeae has repeatedly demonstrated its extraordinary capacity to develop resistance to all antimicrobials introduced for treatment of gonorrhea. After introduction of a new antimicrobial, resistance has emerged and been spread internationally after only 1–2 decades [17]. Gonococci utilize most of the known mechanisms of AMR: drug inactivation, alteration of antimicrobial targets, increased export and decreased uptake. Of particular concern are the mechanisms that alter the permeability of the gonococcal cell that can affect a wide range of antimicrobials with different modes of action, including penicillins, cephalosporins, tetracyclines and macrolides. AMR mechanisms arise from specific chromosomal mutations, conjugation of resistance plasmids and external gene transfer (transformation and subsequent recombination of whole or parts of resistance genes), particularly from other Neisseria species. Since N. gonorrhoeae is naturally competent for uptake and recombination of external DNA during its entire life cycle, the transfer of chromosomally encoded resistance genes is both rapid and extensive [1,11,17].
Exposure of Neisseria spp. to antimicrobials (for treatment of gonorrhea or other infections) can result in the selection of resistant strains, due to spontaneous gene mutations and/or acquisition of whole or parts of resistance genes. The commensal Neisseria spp. frequently inhabit the human body and, consequently, are exposed more frequently to antimicrobials than the transiently acquired gonococcus. Thus, resistance may initially emerge in commensal Neisseria spp., which act as a reservoir of AMR genes that can be readily transferred to gonococci through transformation, particularly in pharyngeal gonorrhea [1,17,34–36]. Pharyngeal gonorrhea is mostly asymptomatic, and gonococci and commensal Neisseria spp. can coexist for extended time periods in the pharynx and share genetic material. Once AMR determinants are acquired by N. gonorrhoeae, efficient intraspecies transfer between gonococcal strains, via transformation, can spread these determinants rapidly throughout the population. Horizontal gene transfer has likely played a pivotal role in the spread of penA mosaic alleles, resulting in decreased susceptibility/resistance to ESCs [23,25,37].
Acquired AMR determinants are stably maintained in the gonococcal population many decades after withdrawal of the specific antimicrobials from treatment regimens [1,17,38]. This persistence and selection of AMR-resistant clones may be as a result of maintained selective pressure in the community due to antimicrobials being used/misused to treat other bacterial infections. However, the persistence may also be because the AMR determinants: do not significantly affect biological fitness; lower the fitness, although this is compensated by having a stabilizing second-site mutation(s); or even enhance the fitness and make the AMR clones more successful in transmission and virulence [17,38]. For example, gonococci lacking a functional MtrC–MtrD–MtrE multidrug-resistance efflux pump (a pump that expels multiple antimicrobials and host-derived compounds from the cell) or MtrA (an activator of mtrCDE transcription) had decreased fitness in murine competition experiments, whereas gonococci lacking MtrR (a repressor of mtrCDE transcription) had increased fitness compared with wild-type gonococci [39–41]. Interestingly, a spontaneous compensatory in-host mutation in the mtrR locus of an mtrA mutant restored fitness to wild-type levels [40]. MtrR can regulate the expression of nearly 70 gonococcal genes and, accordingly, this repressor may also be important for processes involved in basic metabolism, adaptation, biological fitness and pathogenicity [42]. Similarly, gyrA91/95 mutations (encoding an altered GyrA subunit of DNA gyrase), which cause decreased susceptibility/resistance to ciprofloxacin, increased fitness compared with wild-type gonococci in the same murine model. Inclusion of an additional parC86 mutation (encoding an altered ParC subunit of topoisomerase IV), which further increases ciprofloxacin resistance, abrogated this fitness advantage, but compensatory mutations could be isolated that restored in vivo fitness [43]. These examples demonstrate how AMR mechanisms can increase bacterial survival and fitness, and suggest why AMR mechanisms are maintained despite discontinuation of specific antimicrobials in treatment regimens. Thus, highly successful gonococcal clones have acquired additional virulence determinants/enhanced biological fitness that allow them to evade or render ineffective mucosal host defenses and spread widely to predominate in local populations, with subsequent emergence in national and even international populations. Attributes that determine success may or may not be affected by AMR determinants [1,44], but when one of these successful clones acquires AMR determinants for the antimicrobials used for treatment and is subsequently introduced to a high-frequency transmitting population, resistance spreads quickly and widely, and these clones can represent a large fraction of the gonococcal population [1,45,46].
Historically, most gonococcal AMR appears to have originated in the WHO Western Pacific Region (WPR). For example, β-lactamase-mediated penicillin resistance, spectinomycin resistance and fluoroquinolone resistance (with the latter being a well-documented example), all appear to have originated and demonstrated high resistance prevalence in the WHO WPR and subsequently spread worldwide, clearly illustrating that gonococcal AMR is a global concern [1,17,23,25,47]. The first cases and/or outbreaks were identified in this region and, particularly, resistance prevalence was high prior to mainly any resistance being detected in other settings. These observations suggest that resistance emerged in the WHO WPR and, subsequently, spread via sex tourists, long-distance truck drivers and forced migration, to the Pacific Rim countries within the WHO WPR, the USA, southeast and central Asia, and Europe. To provide an evidence-basis for this hypothesis, it would be valuable to model the emergence and spread of gonococcal AMR with an international perspective; however, sufficient quality-assured geographical and temporal metadata (regarding gonorrhoea patients, their sexual networks and the gonococcal population, including its AMR and additional phenotypic and genetic characteristics) are lacking in most settings worldwide. The reasons for emergence of AMR in the WHO WPR can only be hypothesized, but likely factors include extensive use and particularly misuse of antimicrobials in general, which results in AMR emergence in many bacterial species, including N. gonorrhoeae, and in the treatment of gonorrhea itself. This misuse is catalyzed by the unrestricted access to a huge variety of antimicrobials with consequent inappropriate selection, overuse and, in some settings, the use of generic antimicrobials of suboptimal quality and/or dose. This lack of optimal antimicrobial selection and use (in general and for gonorrhea) is presumably due to deficient monitoring of AMR in vitro, clinical failures, antimicrobial use and clinical efficacy of antimicrobials. Of particular concern, is that in regions with a high incidence of gonorrhea and ineffective disease control measures to decrease the gonorrhea burden, resistance can spread rapidly, nationally and internationally [1].
Emergence and spread of in vitro & in vivo decreased susceptibility/resistance to ESCs
Over the past decade, strains of N. gonorrhoeae with decreased susceptibility/resistance to ESCs appear to have emerged in Japan and spread internationally. In the 1990s through to the early 2000s, ceftriaxone was not permitted for treatment of gonorrhea in Japan. Therefore, a variety of oral ESCs, many comprising suboptimal efficacy, and dose regimens were used in monotherapy (if resistance was indicated, cefodizime or spectinomycin was used) [48,49]. Longer, multiple low-dose regimens of oral cephalosporins were commonly utilized. Some of these regimens resulted in ESC concentrations that were subinhibitory, which may thereby have selected for resistant clones [21,49–51]. In many settings worldwide, only the most potent oral ESC, cefixime (in optimized 400 mg × 1 dose), has been used for treatment. However, when applied, single dose cefixime therapy in Japan included only 300 mg as a standard dose [52]. Thus, between 1995 and 2000, in Fukuoka, Japan, the MIC peak of cefixime and ceftriaxone in gonococcal isolates increased from 0.008 μg/ml to 0.25 μg/ml, and from 0.015 μg/ml to 0.064 μg/ml, respectively [49]. Furthermore, in six hospitals in central Japan between 1999 and 2002, the proportion of gonococcal isolates with decreased susceptibility/resistance to cefixime (MIC ≥0.5 μg/ml) and ceftriaxone (MIC ≥0.5 μg/ml) increased, from 0 to 30.2% and from 0 to 0.9%, respectively [50]. From 1999 to 2001, eight treatment failures with cefixime (200 mg orally × 2; 6 h apart) were reported and isolates cultured from these patients had cefixime MICs of 0.125–0.25 μg/ml [21]. In 2002–2003, four treatment failures with an extended cefixime regimen (200 mg orally twice a day for 3 days) were documented [30]. Isolates obtained from these treatment failures exhibited substantially elevated MICs of cefixime (0.5–1 μg/ml) and ceftriaxone (0.125–0.5 μg/ml). In 2006, all oral ESCs were excluded from treatment guidelines in Japan.
The theory that gonococcal strains with decreased susceptibility/resistance to ESCs, particularly oral ESCs, have spread internationally from Japan is in concordance with decreased in vitro susceptibility to cefixime and ceftriaxone in WHO WPR countries, including Australia, China, Hong Kong, Taiwan, USA, Canada, South America, southeast Asia, the EEA and Russia, identified in subsequent years [1,3–20,23,25,32,33,45,47,51,53–58,205]. For example, from 2009 to 2010, decreased susceptibility/resistance to cefixime in EU/EEA gonococcal isolates increased from 4 to 9%, and this resistance phenotype was detected in 17 countries [203,205]. Worryingly, gonococcal AMR surveillance remains sporadic, limited or even lacking in many regions worldwide, including Eastern Europe, Central Asia, Africa, and large parts of South America and the Caribbean [1,12,20,59–61]; these regions also suffer from a high burden of gonorrhea [59,60,201]. Accordingly, the global burden of decreased susceptibility/resistance to ESCs is unknown.
It is of concern that the first treatment failures with cefixime outside Japan [22,25–27] and with ceftriaxone [23,24,28,29] have been verified in recent years. Furthermore, the first three XDR [1] gonococcal isolates with high-level ceftriaxone resistance were recently identified in Japan [23], France [25] and Spain [31].
Recently verified treatment failures with cefixime & ceftriaxone, international spread of successful gonococcal strains & first strains with high-level ceftriaxone resistance
Cefixime treatment failures
In 2010, the first verified cefixime treatment failures beyond Japan were reported in Norway [27] and, subsequently, verified failures were documented in the UK [22], Austria [26] and France [25]. The main characteristics of all microbiologically verified failures are described in Table 1. Briefly, the cefixime MICs of the gonococcal isolates resulting in treatment failure were 0.25–4 μg/ml and the country of infection varied widely. Nevertheless, all genetically typed isolates belonged to either the multilocus sequence typing (MLST) ST1901 and N. gonorrhoeae multiantigen sequence typing (NG-MAST) ST1407 or genetically closely related NG-MAST STs, such as ST3779 and ST3431 (Table 1) [22,25–27]. Disquietingly, the strain (referred to as F89) resulting in cefixime failure in a man who has sex with men (MSM) in Quimper, France [25], appears to represent a subclone of the MLST ST1901/NG-MAST ST1407 clone that has developed high-level resistance to cefixime (MIC = 4 μg/ml) and ceftriaxone (MIC = 1–2 μg/ml). F89 was the second identified XDR [1] gonococcal strain [25]. A genetically and phenotypically similar ceftriaxone-resistant strain has subsequently been described in Spain, causing gonorrhea in two MSMs with sexual contact [31]. This report illustrates that this ceftriaxone-resistant strain can spread and, accordingly, does not have a deleteriously low biological fitness.
Table 1.
Verified treatment failures with cefixime beyond the early reports from Japan.
| Country | Country of exposure | Cefixime treatment | Final successful treatment | MIC (μg/ml) | MLST/NG-MAST | Site of failure | Ref. |
|---|---|---|---|---|---|---|---|
| Norway (n = 2) | Philippines/Spain/Norway | 400 mg × 1 | Ceftriaxone 500 mg × 1 | 0.25–0.5† | ST1901/ST1407 | Urethra | [27] |
| UK (n = 2) | UK | 400 mg × 1/400 mg × 1 plus doxycycline 100 mg twice daily for 7 days | Ceftriaxone 250 mg × 1 | 0.25 | ND/ST3779 and ST3431 | Urethra | [22] |
| Austria (n = 1) | Germany | 400 mg × 1 for 7 days | Azithromycin 2 g × 1 | 1.0† | ST1901/ST1407 | Urethra | [26] |
| France (n = 1) | France | 200 mg × 2 (6 h apart) | Gentamicin 160 mg × 1 | 4.0† (XDR) | ST1901/ST1407 | Urethra | [25] |
Genetic resistance determinants (penA, mtrR and penB) elucidated.
MLST: Multilocus sequence typing; ND: Not determined; NG-MAST: Neisseria gonorrhoeae multiantigen sequence typing; ST: Sequence type; XDR: Extensively drug-resistant.
Ceftriaxone treatment failures
Five failures in treating pharyngeal gonorrhea with ceftriaxone, the last remaining treatment option, have been verified in Australia (n = 2) [29], Sweden (n = 1) [24], Slovenia (n = 1) [28], and Japan (n = 1) [23]. The Swedish [24] and Slovenian [28] treatment failures were caused by the MLST ST1901/NG-MAST ST1407 clone. The main characteristics of these failures are described in Table 2. The relatively low MICs of the gonococcal strains (with exception of the one in Japan) causing these failures, especially those from Australia that had near wild-type MICs (Table 2), likely reflecting the difficulty in treating pharyngeal gonorrhea compared with urogenital infections [1,3,14,19,20,62–64]. Thus, it appears likely that treatment failures with ceftriaxone will initially arise from pharyngeal gonorrhea. Unfortunately, the failure to treat pharyngeal gonorrhea in a female sex worker with ceftriaxone 1 g × 1 in Kyoto, Japan, was caused by the first ever XDR [1] N. gonorrhoeae strain (‘H041’; the first gonococcal ‘superbug’), which displayed high-level resistance to cefixime (MIC = 8 μg/ml) and ceftriaxone (MIC = 2–4 μg/ml), and to most other antimicrobials tested (n = 28) [23]. Ceftriaxone treatment (1 g × 1) results in a median time of free ceftriaxone above the MIC of only 6.0 h and 0 h for the detected MICs of 2 μg/ml (agar dilution method) and 4 μg/ml (Etest®), respectively [51]. Consequently, even with this treatment, H041 will escape eradication in almost any patient. H041 was assigned to MLST ST7363 and the novel NG-MAST ST4220 (Table 2). H041 may of accordingly represent a subclone of the successful cefixime-resistant MLST ST7363, which has been prevalent in Japan, that has also developed high-level ceftriaxone resistance [23,37,65].
Table 2.
Verified treatment failures with ceftriaxone globally.
| Country | Country of exposure | Ceftriaxone treatment | Final successful treatment | MIC (μg/ml) | MLST/NG-MAST | Site of failure | Ref. |
|---|---|---|---|---|---|---|---|
| Australia (n = 2) | Australia | 250 mg × 1 | Ceftriaxone 500 mg × 1†/ceftriaxone 1 g × 1 | 0.032/0.016 | ND/ST5 and ST2740 | Pharynx | [29] |
| Sweden n = 1) | Japan | 250 mg × 1 and 500 mg × 1 | Ceftriaxone 1 g × 1 | 0.125–0.25‡ | ST1901/ST2958 | Pharynx | [24] |
| Slovenia (n = 1) | Serbia | 250 mg × 1 | Ceftriaxone 250 mg × 1 plus azithromycin 1 g × 1 | 0.125‡ | ST1901/ST1407 | Pharynx | [28] |
| Japan (n = 1) | Japan | 1 g × 1 | None§ | 2.0–4.0‡ (XDR) | ST7363/ST4220 | Pharynx | [23] |
No test-of-cure to confirm clearance of infection performed.
Genetic resistance determinants (penA, mtrR and penB) elucidated.
The pharyngeal infection was considered to have resolved spontaneously within 3 months.
MLST: Multilocus sequence typing; ND: Not determined; NG-MAST: Neisseria gonorrhoeae multiantigen sequence typing; ST: Sequence type; XDR: Extensively drug-resistant.
International spread of successful gonococcal clones causing treatment failures with ESCs
The gonococcal clones MLST ST1901 (commonly NG-MAST ST1407) and MLST ST7363, both of which likely originated in Japan [23,25], were responsible for all of the verified cefixime treatment failures and at least three of the five ceftriaxone treatment failures (Tables 1 & 2); both of these clones are spreading worldwide [6,8,22,23,25–28,37,55,65–73]. During the early 2000s, MLST ST1901 (frequently NG-MAST ST1407 in many countries) started to replace MLST ST7363 as the most predominant MLST ST in Japan, and it is currently responsible for most of the decreased susceptibility/resistance to ESCs in Europe [6,8,23,25,65]. Nevertheless, although NG-MAST ST1407 is the most prevalent ST of MLST ST1901 in Europe, many NG-MAST STs of this MLST clone have been identified globally, particularly in Japan, where the MLST ST1901 clone has been spreading for many years [6,8,22,25–28,65–72; 206].
Thus, two successful gonococcal clones (MLST ST7363 and MLST ST1901) are spreading worldwide and both have demonstrated the capacity to develop high-level resistance to cefixime and ceftriaxone, resulting in ESC treatment failures. Worryingly, the first three strains with high-level ceftriaxone resistance were identified in high-risk, frequently transmitting populations, such as H041 from a female sex worker in Japan [23], F89 in France [25] and the Spanish strain from MSMs [31]. To date, further spread of H041 has not been reported. However, F89 [25] and the genetically similar Spanish strain, transmitted between two MSMs in Spain [31], may represent the first international transmission of N. gonorrhoeae with high-level ceftriaxone resistance. If strains with high-level ceftriaxone resistance spread globally, gonorrhea may become untreatable.
Verification of treatment failures
It is crucial to survey for and appropriately verify as well as follow-up treatment failures with ESCs [1,202–204,207]. Recent WHO publications [1,202,207] have described in detail important laboratory parameters to verify a treatment failure, which ideally requires assessing pre- and post-treatment isolates for MICs, genotype, and genetic resistance determinants. Additionally, a detailed clinical history that excludes reinfection and records the treatment regimen used is mandatory. The correlates between genetic resistance determinants, ESC MICs, and treatment outcome remain weak. It is crucial to examine all gonococcal strains causing treatment failure as well as isolates showing high-level ESC resistance in vitro, including determination of resistance determinants. This is essential to establish appropriate MIC breakpoints for ESC resistance (together with pharmacokinetic/pharmacodynamic simulations), to develop genetic methods for screening to identify specific ESC-resistant clones and, ideally, to carry out genetic ESC resistance (and AMR in general) testing.
Decreased susceptibility/resistance to ESCs: genetic resistance determinants
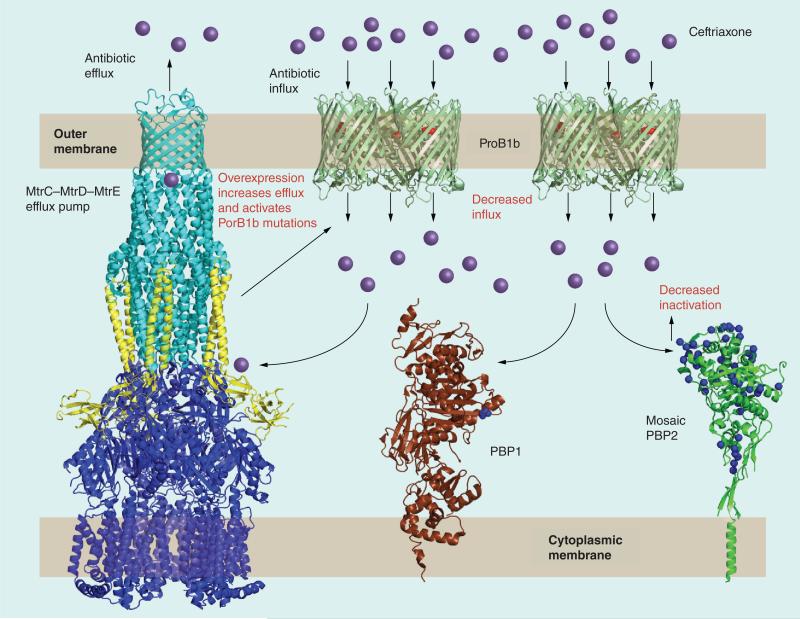
Acquisition of resistance determinants from a resistant strain to a susceptible strain can be achieved in the laboratory by step-wise transformation. Many studies have investigated the steps of transformation for penicillin and ESC resistance, and these reveal a complex and multifaceted interaction of at least four known resistance determinants (Figure 1): penA, encoding altered forms of PBP2, the lethal target for β-lactam antibiotics; mtrR, resulting in overexpression of the MtrC–MtrD–MtrE efflux pump; penB, encoding mutations in the major outer membrane porin; and ponA, encoding an altered form of PBP1. These and other determinants are discussed in more detail below.
Figure 1. An illustration of the known resistance determinants responsible for decreased susceptibility and resistance to extended-spectrum cephalosporins in Neisseria gonorrhoeae.
penA encodes altered forms of PBP2, the lethal target for β-lactam antimicrobials. The purple spheres indicate amino acid alterations in a mosaic PBP2 compared with wild-type PBP2. Expression of mtrR results in overexpression of the MtrC–MtrD–MtrE efflux pump and activation of penB. penB encodes mutations in the major outer membrane porin (PorB1b) that decrease influx of antimicrobials. ponA, encoding an altered form of PBP1, has been demonstrated to be involved in penicillin resistance, but not yet in resistance to extended-spectrum cephalosporins. The structure of PBP2 is from Neisseria gonorrhoeae. The other structures are from closely related structures in other bacteria, that is, the MtrCDE are of AcrAB-TolC from Escherichia coli; the structure of PorB1b is of Neisseria meningitidis PorB and the structure of PBP1 is of E. coli PBP1b. The PDB codes are: 2f1M (AcrA), 1IWG (AcrB), 1EK9 (TolC), 3A2R (PorB), 3FWM (PBP1) and 3EQU (PBP2).
Alteration of PBP2, the lethal target for ESCs
The first step in decreased susceptibility/resistance to ESCs is alteration of the penA gene encoding the transpeptidase PBP2 (Figure 2), the lethal target for ESCs and other β-lactam antibiotics. Acquisition of penA mosaic alleles, and non-mosaic penA alleles with mutations at A501, are associated with enhanced ESC MICs [8,23–28,31,36,37,52,68,69,71–87]. The mosaic penA alleles appear to have evolved by DNA uptake and subsequent recombination of partial penA genes from commensal Neisseria spp. commonly residing in the oropharynx (i.e., Neisseria sicca, Neisseria perflava, Neisseria cinerea, Neisseria polysaccharea and/or Neisseria flavescens) into the gonococcal penA gene [36,75,76,79]. Pharyngeal gonorrhea may have provided the means for this in vivo intrageneric horizontal transfer [1,34–36]. The relative increases in MICs due to acquisition of a penA mosaic allele appear to be higher for cefixime than for ceftriaxone [23,25,86], which may be due to structure/activity relationships associated with the longer side chain at the C-3 position of the cephem skeleton of ceftriaxone [1,78–80,87].
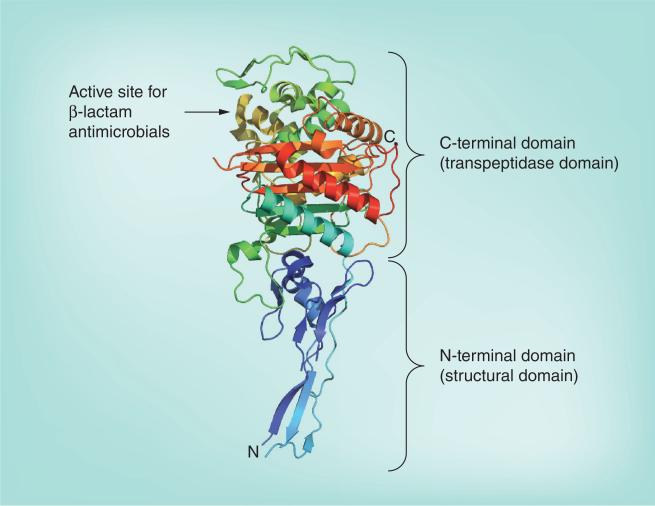
Figure 2. The structure of PBP2 from Neisseria gonorrhoeae.
The C-terminal domain (transpeptidase domain) with the active site for binding of β-lactam antimicrobials is at the top and the N-terminal domain (structural domain) that anchors PBP2 to the cytoplasmic membrane at the bottom. The structure is color-ramped blue-to-red in the N-terminal to C-terminal direction.
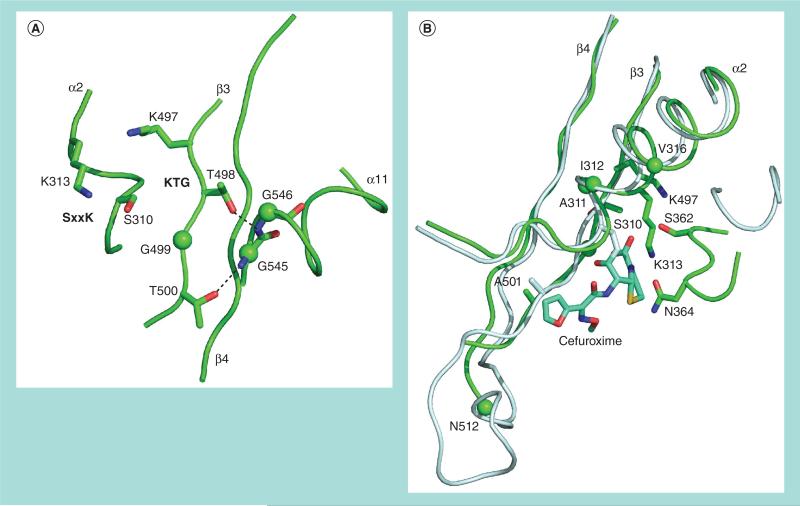
Mosaic PBP2 variants contain up to 70 amino acid alterations compared with the wild-type PBP2 [23]. Takahata et al. initially suggested that three of these alterations, G545S, I312M and V316T, were responsible for the ESC resistance (by altering the β-lactam binding pocket) [79], especially to cefixime [80]. However, it was subsequently shown that while reversion of these three amino acids to wild-type dramatically decreased ESC resistance, introduction of these same amino acids into the wild-type PBP2 sequence had little to no effect on resistance. These data suggest that the three mutations only increase resistance in the presence of other mutations in the penA mosaic alleles that have little or no apparent effect on their own [81], a phenomenon known as epistasis. Inspection of the crystal structure of PBP2 reveals that the G545S mutation is present at the beginning of the a11 helix, one of the two helices on top of the five anti-parallel β-strands typical of β-lactam-interacting proteins. The main chain amides of G545 and G546 are within hydrogen-bonding distance of the side chain hydroxyls of Thr498 and Thr500, respectively, located within the KTG(T) active site motif (Figure 3a). The main chain amide of Thr500 is predicted to form the oxyanion hole for stabilization of the transition state and, thus, one effect of the G545S mutation might be to lower acylation by compromising the geometry of the transition state/tetrahedral intermediate. Alternatively, the hydroxyl side chains of Thr498 and Thr500 may interact with the β-lactam carboxylate in the covalent complex, and alteration of these contacts may also lower the acylation [81]. Ile312 and Val316 are located on the opposite side of Ser310 and Lys313 of the SxxK active site motif on the α2 helix and pack into a hydrophobic pocket (Figure 3b). Mutation to larger (I312M) or more hydrophilic (V316T) side chains might disrupt these interactions and alter the position of the SxxK active site motif, which would also result in decreased acylation [81]. Tomberg et al. also verified that an additional mutation, N512Y, contributes to decreased susceptibility/resistance to ESCs (without affecting penicillin susceptibility), although it plays a lesser role than the three alterations discussed above. N512 is relatively distant from the active site (Figure 3b) on the β3–β4 loop, and it is possible that the N512Y alteration perturbs the architecture of the KTG active site motif of the β3 strand [81].
Figure 3. Crystal structure of PBP2 displaying all amino acids currently verified to be involved in enhancing the MICs of extended-spectrum cephalosporins.
(A) Location of the G545S mutation and the β-lactam active site sequence motifs at the crystal structure of PBP2. The interactions of Thr498 and Thr500 within the KTG(T) active site motif with the main chain amides of Gly545 and Gly546 are illustrated. Mutation of Gly545 to Ser incorporates a hydroxylated side chain that potentially could perturb the interactions between the two Thr residues on β3 and the main chain of the α11 helix [81]. The SxxK active site motif is also shown. (B) The crystal structure of PBP2 showing the location of all amino acid alterations (with the exception of G545S shown in A) currently verified to increase the MICs of extended-spectrum cephalosporins. Ile312 and Val316 are located on the opposite side of Ser310 and Lys313 of the SxxK active site motif on the α2 helix and pack into a hydrophobic pocket. Mutation to larger (I312M) or more hydrophilic (V316T) side chains might disrupt these interactions and alter the position of the SxxK active site motif. N512 is relatively distant from the active site on the β3–β4 loop and it is possible that the N512Y alteration perturbs the architecture of the KTG active site motif of the β3 strand [81]. Replacement of the methyl side chain of A501, which is located on the β3–β4 loop close to the KTG active site motif, with the more bulky side chains of valine (A501V), threonine (A501T) or proline (A501P) might inhibit the binding of extended-spectrum cephalosporins by clashing with their R1 substituents [25,81]. The novel A311V and T316S alterations, which are in close proximity to the β-lactam active site, appear to contribute to high-level resistance to extended-spectrum cephalosporins [23]. Elucidation of their effects in detail is in progress. Images were created in PyMol and were adapted from [81].
Characterization of the novel penA mosaic allele in the first described XDR [1] strain, H041, with high-level resistance to cefixime (MIC = 8 μg/ml) and ceftriaxone (MIC = 2–4 μg/ml), revealed 12 additional amino acid alterations compared with penA mosaic allele X, which has been associated with cefixime treatment failures in Japan [23]. Transformation experiments using full-length and partial penAH041 mosaic alleles indicated that the novel A311V and T316S alterations may contribute to high-level ESC resistance, due to the proximity to the β-lactam active site in PBP2 (Figure 3b) [23]. However, the full cause of ESC resistance in H041 needs to be appropriately elucidated, which is in progress.
A single amino acid alteration at position A501 (mostly A501V, but sometimes A501T) in PBP2, which until recently was found mainly in non-mosaic penA alleles, is associated with decreased susceptibility/resistance to ESCs, particularly ceftriaxone [8,25,31,52,68,69,74,77,80,81,83–85]. The rarity of strains containing a penA mosaic allele with an A501 mutation may suggest that these strains have a lower biological fitness [25,81], although this has not been formally investigated. A501 alterations could be gonococcal-specific, as they have not yet been reported in any other Neisseria species, suggesting that they may have evolved and been selected in gonococci through spontaneous mutation and selective pressure with ESCs instead of by transformation from another species [8,25,81,85]. In support of this hypothesis, the isolation of a spontaneous A501V mutation during transformation experiments with mosaic penA alleles has been reported [80]. Given the concern that A501 mutations might emerge in mosaic penA alleles, Tomberg et al. examined the effects of introducing an A501V mutation into a mosaic penA allele and transforming it into both a penicillin-susceptible and a penicillin-resistant gonococcal strain. Introduction of the A501V mutation increased the MIC of both cefixime and ceftriaxone by two- to three-fold, while the penicillin MIC decreased [81]. As originally feared, a strain containing a traditional penA mosaic allele with an A501 mutation was reported only a year later [25]. This strain (F89), which contained a penA mosaic allele XXXIV with an additional A501P alteration, was the second XDR [1] strain identified and displayed high-level resistance to cefixime (MIC = 4 μg/ml) and ceftriaxone (MIC = 1–2 μg/ml). Transformation experiments confirmed that this novel penA mosaic allele was responsible for the high-level ESC resistance [25]. Isolates harboring the penA mosaic allele XXXIV, which commonly belong to the successful MLST ST1901/NG-MAST ST1407 clone, have been reported in many countries worldwide [25,28,55,67–69,71,73,74], and it is of major concern that an A501P (or similar) alteration will emerge in additional strains containing the penA mosaic allele XXXIV or other penA mosaic allele. It has been suggested that replacement of the methyl side chain of Ala501, which is located on the β3–β4 loop close to the KTG active site motif of PBP2, with the more bulky side chains of valine (A501V) or threonine (A501T), inhibits the binding of cefixime and ceftriaxone to PBP2 by clashing with their R1 substituents [25,81]. The change of Ala501 to a proline (A501P) introduces secondary structure alterations that may result in even more dramatic changes, which is consistent with the much higher MICs conferred by the A501P alteration than the A501V and A501T alterations [25]. The crystal structure of PBP2 showing all amino acids currently verified to be involved in enhancing the MICs of ESCs is illustrated in Figure 3.
Enhanced efflux & decreased influx of ESCs
Specific mutations in the promoter and/or coding sequence of mtrR, a transcriptional repressor of the mtrCDE operon, markedly increase the expression of the MtrC– MtrD–MtrE efflux pump (‘mtrR resistance determinant’) that enhances the efflux of ESCs, particularly ceftriaxone, from the periplasmic space of the gonococcus and increases their MICs. The mtrR determinant is most often a single-base-pair (A) deletion in the 13-base pair (bp) inverted repeat of the promoter region, with G45D mutations (and other mutations) within the coding sequence of MtrR being less common [8,23–28,31,36,39–41,58,68,74,78,82,86,88–91]. Other rare alterations that increase expression of the MtrC–MtrD–MtrE efflux pump have been described: a 153-bp insertion sequence located between the mtrR/mtrC promoter and the mtrC gene [92], and a C-to-T transition 120 bp upstream of the mtrC start codon (termed mtr120) that results in a consensus -10 element and a novel promoter for mtrCDE transcription [93].
mtrR mutations affect resistance in two ways: by directly increasing efflux of the ESC and by activating the penB resistance determinant, which has a resistant phenotype only when a mtrR mutation is present [86]. The penB resistance determinant encodes an altered porB1b allele with mutations that alter amino acid G101 (e.g., G101K) or both G101 and A102 (e.g., G101D/A102D) in the putative loop 3 of the PorB1b outer membrane porin. Acquisition of penB results in decreased influx of ESCs, particularly ceftriaxone, into the cell and increases their MICs [8,23–28,31,36,58,68,74,78,82,86,89,94,95]. Synergy between the penA mosaic allele, mtrR, and penB has been reported in isolates with decreased susceptibility/resistance to ESCs, particularly ceftriaxone [23,25,86]. Interestingly, the great majority of cefixime resistance appears to be conferred by the penA mosaic allele, with mtrR and penB having only a minor effect on its MIC, whereas ceftriaxone resistance is nearly equally dependent upon penA and mtrR/penB. This result suggests that either cefixime does not readily diffuse into the periplasm through PorB or that such diffusion is not altered by the penB determinant. These data also suggest that cefixime is not an effective substrate for the MtrC–MtrD–MtrE efflux pump. The net charge of cefixime (-2) differs from the net charge of penicillin (-1) and ceftriaxone (-1), which may affect the permeation properties of cefixime [86].
Other current or possible future resistance determinants
Alterations in ponA (the ponA1 allele encoding PBP1 with an L421P mutation) and pilQ (encoding loss-of-function mutations in the pore-forming secretin PilQ protein of the type IV pili), both of which are involved in high-level penicillin resistance in strains containing penA alterations, mtrR and penB, do not appear to alter the ESC MICs in clinical isolates [8,86,96–98]. The ponA1 allele is present in most isolates with decreased susceptibility/resistance to ESCs. However, this likely only reflects that these strains arose by transfer of penA mosaic alleles into pre-existing chromosomally mediated penicillin-resistant strains, which persist in high prevalence in the gonococcal population, despite the fact that penicillin was withdrawn from gonorrhea treatment regimens decades ago [86]. An unusual aspect of the ponA1 allele in penicillin resistance is that the introduction of ponA1 into a strain with penA, mtrR and penB resistance determinants has no effect on resistance, while reversion of ponA1 to wild-type in a penicillin-resistant strain decreases the MIC by twofold [96]. This may indicate epistasis or the presence of ‘factor X’, which is described in more detail below. pilQ2 (or any other pilQ loss-of-function mutation) has never been reported in clinical isolates, probably because it compromises type IV pilus formation and, thus, pathogenic potential [17,97,99]. Nevertheless, the effect(s) of ponA and/or pilQ polymorphisms in future ESC-resistant strains cannot be totally excluded.
While penA, mtrR and penB determinants markedly increase resistance, they do not confer donor levels of resistance to penicillin or ESCs (MICs ~three- to six-fold lower than donor strains). Attempts by many labs to transform strains already containing penA, mtrR and penB to donor MIC levels have been unsuccessful and, thus, the identity of this non-transformable penicillin and ESC resistance determinant (termed ‘factor X’) remains unknown [8,23,25,78,81,86].
Fortunately, acquisition or development of an extended-spectrum β-lactamase (ESBL), which could degrade all ESCs, has not yet been reported in any gonococcal strain. However, in many strains the blaTEM-1 gene (encoding TEM-1 β-lactamase) appears to have evolved by one single nucleotide polymorphism (SNP) to blaTEM-135 and only one additional specific SNP could result in an ESBL [100].
Public health actions to meet the large challenge of emergence of high-level ceftriaxone-resistant & untreatable gonorrhea
In the age of easy international travel, the global trend of AMR in gonococci and resistance to ESCs, including treatment failures, demands a global approach to respond to the potential public health disaster that may occur with the emergence of untreatable gonorrhea. This is imperative, since gonococcal AMR data are lacking in many settings and the true global problem is, therefore, unknown. Accordingly, the WHO Global Gonococcal Antimicrobial Surveillance Programme (WHO Global GASP), initially established in the 1990s, was revisited and revamped in 2009, in close liaison with other existing AMR surveillance programs, in order to enable a coordinated global response. The WHO Global GASP network aims to recruit laboratories worldwide to: monitor quality-assured gonococcal AMR data (with particular attention to ESCs); provide support to establish gonococcal culture and AMR testing; inform public health authorities and treatment guidelines on trends in gonococcal AMR; optimize early detection of emerging resistance; and identify and verify treatment failures with ESCs [33].
To facilitate the functioning of this WHO GASP network and engage the support of governments, the international community and appropriate donors, the WHO recently published the ‘Global action plan to control the spread and impact of AMR in Neisseria gonorrhoeae’ [33,202]. This plan aims to enhance AMR surveillance worldwide through the WHO Global GASP, facilitate early detection and verification of resistance to recommended treatment (particularly ESCs) and combine these with a public health response to mitigate the impact of ESC-resistant gonococci on sexual and reproductive health morbidity. The WHO global action plan calls for a public health approach to the control of gonorrhea and the emergence of untreatable gonorrhea. The main components of the WHO global action plan are summarized in Box 1. Several region-specific and national action/response plans to mitigate the spread of multidrug-resistant gonorrhea, which are also crucial for the global response, are under preparation or have been published. For example, the European CDC and the US CDC have launched public health response plans for EU/EEA countries [203] and the USA, respectively [204].
Future options for treatment & prevention of gonorrhea
Even if the aforementioned action/response plans will be fully implemented, they would likely only delay, as opposed to halt, the inevitable international emergence and spread of ceftriaxone-resistant gonorrhea. Ultimately, new treatment strategies and novel antimicrobials are essential for the effective treatment of gonorrhea.
Increased doses of ESCs
Use of an increased dose of ceftriaxone has been implemented in most settings [1,21,26,51,59,101,102]. In some countries, such as Japan and China, ceftriaxone 1 g × 1 is recommended [59,101,103,104], and likely up to 2 g × 1 would be safe, based on the dosage used for treatment of community-acquired pneumonia. Cefixime is not licensed for doses higher than the currently used 400 mg × 1, due to frequent adverse gastrointestinal effects observed with a higher dose (800 mg × 1) [105]. Even so, increased doses of ceftriaxone will only provide a short-term solution. No currently available alternative cephalosporins, either injectable or oral, offer any advantage over ceftriaxone or cefixime in terms of efficacy and pharmokinetics/pharmacodynamics, and efficacy for pharyngeal infection is even less certain [14,19,62,64].
Dual antimicrobial therapy
An alternative approach to increasing the dose in ESC monotherapy is to use dual antimicrobial therapy, which also covers concurrent chlamydial infection, and recently this has been introduced in the UK [64], the whole of Europe [Bignell C, Unemo M. European guideline on the diagnosis and treatment of gonorrhoea in adults (2012), Submitted] and the USA [19,106]. In the UK, ceftriaxone 500 mg × 1 (intramuscularly) plus azithromycin 1 g × 1 (orally) have been recommended since 2011 for uncomplicated anogenital gonorrhea [64]. The recommendation in the USA is ceftriaxone 250 mg × 1 (intramuscularly) plus azithromycin 1 g × 1 or doxycycline 100 mg (orally) twice-daily for 7 days [19,106]. Synergy has been reported for ESCs and azithromycin in treatment, including for pharyngeal gonorrhea [107,108], and synergistic and/or additive effects can also be observed in vitro [Unemo M, Unpublished Data]. Dual antimicrobial therapy might also inhibit future development of gonococcal AMR. Accordingly, dual antimicrobial therapy appears effective, and, in fact, has been practiced in several years, primarily due to the frequent co-administration of azithromycin to gonorrhea patients for possible concomitant chlamydial infection. Unfortunately, susceptibility to ceftriaxone is decreasing, resistance to doxycycline is common and resistance to azithromycin has been selected rapidly in settings where it has been frequently used. In recent years, isolates with high-level resistance to azithromycin (MIC ≥256 μg/ml), due to an A2059G alteration (Escherichia coli numbering) in domain V in three or four of the four alleles of the 23S rRNA gene, have also been identified in several countries [109–113]. In the USA, a randomized clinical multicenter trial to evaluate the efficacy (and safety) of gentamicin (240 mg × 1 intramuscularly) plus azithromycin (2 g × 1 orally) and gemifloxacin (320 mg × 1 orally) plus azithromycin (2 g × 1 orally) for treatment of uncomplicated urogenital gonorrhea is also ongoing. In order to minimize the gastrointestinal side effects when administering 2 g × 1 of azithromycin, it would be valuable to have the new extended-release microsphere formulation of azithromycin available worldwide. Nevertheless, dual antimicrobial regimens, especially those with injections, might not be feasible and/or affordable in less-resourced settings, many of which suffer from the highest gonorrhea burden.
Antimicrobial monotherapy: any future options?
Effective antimicrobial monotherapy, or at least new antimicrobials for inclusion in a dual antimicrobial therapy regimens, appear essential for a long-term solution and from a global perspective. The aminocyclitol spectinomycin (2 g × 1 intramuscularly) is a theoretical option, and resistance to spectinomycin is also rare worldwide [1,3,7,14,18,23]. Unfortunately, high-level resistance to spectinomycin rapidly emerged when it was widely used as a first-line option among US military personnel in Korea in the 1980s [114]. This resistance was due to only one SNP, C1192T (E. coli numbering), in the 16S rRNA gene. High-level resistance can also result from alterations in ribosomal proteins [Unemo M, Unpublished Data]. Furthermore, spectinomycin is not effective for treatment of pharyngeal gonorrhea and it is currently not available in many countries [1,3,11,14,23,115], making it a poor candidate for first-line treatment. There are few promising candidates in sight for new (or rediscovery of old) antimicrobials or other compounds for effective treatment of gonorrhea [1,3,11,14,19,23,25].
Nevertheless, the aminoglycoside gentamicin is now being considered for treatment of gonorrhea in several countries. Gentamicin (240 mg × 1 intramuscularly) has also been used as first-line treatment in Malawi since 1993 without any reported emergence of in vitro resistance [116,117], and the in vitro susceptibility in Europe appears high [118]. However, global susceptibility data remain sparse, appropriate correlates between MICs of gentamicin and treatment outcome are lacking (e.g., no microbiological resistance breakpoints exist), no data for treatment of pharyngeal or anorectal gonorrhea exist and it is not clear whether appropriate pharmacokinetic/pharmacodynamic targets for treatment of all cases of anogenital and pharyngeal gonorrhea can be reached. A recent meta-analysis also found that treatment with a single dose of gentamicin resulted in a pooled cure rate of only 91.5% (95% CI: 88.1–94.0%) [119], which is substantially lower than the current criteria of ≥95% effectiveness for recommended treatment of gonorrhea. Furthermore, gentamicin has mainly been used in Malawi in syndromic management administered together with doxycycline and, thus, in a dual antimicrobial therapy regimen and not as a single antimicrobial therapy. Valuable information regarding the efficacy and safety of gentamicin in dual antimicrobial therapy of gonorrhea will soon be available from the ongoing clinical trial mentioned above.
The new fluoroketolide (antimicrobial class: macrolides), solithromycin (CEM-101), has recently been demonstrated to have high in vitro activity against gonococci, including ESC- and multidrug-resistant isolates [120]. Solithromycin had an activity that was fourfold (MIC50) to 32-fold (MIC90) better than that of azithromycin and also appeared superior to most other antimicrobials tested [120]. The three binding sites for solithromycin (compared with two for other macrolides) on the bacterial ribosome probably result in this higher activity and are also expected to delay resistance development [120,121]. Solithromycin is well absorbed orally, with high plasma levels, intracellular concentrations and tissue distribution, has a long post-antibiotic effect, and a 1.6 g × 1 oral dose appears well-tolerated [122]. Accordingly, solithromycin might be appropriate for single antimicrobial therapy, especially of infections by ESC-resistant strains, and particularly in dual antimicrobial therapy of gonorrhea, which likely would further delay resistance development. Furthermore, the susceptibility of Chlamydia trachomatis [123] and Mycoplasma genitalium [124] to solithromycin indicates that it may be an option for treatment of several sexually transmitted infections, including management of urethral and vaginal discharge syndromes. Nevertheless, gonococcal isolates with ribosomal mutations resulting in high-level resistance to azithromycin (MIC ≥256 μg/ml) have MICs of 4–32 μg/ml for solithromycin, which presumably reflects resistance [120]. In the USA, a minor open-label, single-center study to evaluate the efficacy and safety of an oral 1.2 g × 1 dose of solithromycin for treatment of uncomplicated urogenital gonorrhea is ongoing.
The in vitro activity of ertapenem (parenteral 1-β-methyl-carbapenem) against N. gonorrhoeae also appears to be high [125]. Ertapenem did not have any in vitro advantage over ceftriaxone for isolates that were susceptible or intermediate-susceptible to ceftriaxone. Nevertheless, for isolates with resistance to ceftriaxone (MIC = 0.5–4 μg/ml), the corresponding MICs of ertapenem remained low (0.016–0.064 μg/ml) [125]. Ertapenem was also highly active against multidrug-resistant isolates [125], as well as being safe, well-tolerated and effective against urinary tract infections [126,127]. Thus, ertapenem might be a treatment option for gonorrhea, particularly for the currently identified ESC-resistant cases and especially in dual antimicrobial therapy. However, despite the observation that ceftriaxone-resistant isolates (n = 4) had relatively low ertapenem MICs, ESC resistance determinants, such as mosaic penA alleles, mtrR and penB, increase the MIC of ertapenem [125]. The decrease in susceptibility to ertapenem indicates that gonococci have already acquired initial determinants for ertapenem resistance, and the eventual remodeling of PBP2 to specifically exclude ertapenem, which would work synergistically with the existing resistance determinants, seems likely. Due to these facts and the pharmacokinetic/pharmacodynamic considerations of ertapenem, it may not be suitable for future gonorrhea treatment. Finally, the future possibility of acquisition of a carbapenemase or a TEM-1 β-lactamase that evolves into an ESBL to degrade ertapenem cannot be excluded, especially when the blaTEM-1 gene appears to be evolving [100].
Tigecycline, a broad-spectrum parenteral glycylcycline, is a tetracycline derivative that has shown high in vitro activity against gonococci, including tetracycline-resistant isolates [3,14,128]. However, tigecycline has not been examined against multidrug-resistant gonococci (including ESC-resistant isolates) or evaluated in vivo for treatment of gonorrhea. Furthermore, because tigecycline is mainly eliminated into the bile and a relatively small proportion of the administered drug is excreted unchanged in the urine, the use of tigecycline for treatment of urinary tract infections has been debated [129–131].
Evaluation of novel treatment options
All novel single or dual antimicrobial treatment regimens for gonorrhea ideally require up-to-date and comprehensive in vitro and in vivo evaluation, including appropriately designed, randomized and controlled treatment studies evaluating parameters such as efficacy, safety, toxicity, cost, optimal dose and pharmacokinetic/pharmacodynamics data for genital and extragenital (especially pharyngeal) gonorrhea. Furthermore, knowledge regarding current and future genetic resistance determinants (in vitro selected and in vivo emerged) for these antimicrobials (both in gonococci and bystander organisms when treating gonorrhea) clear correlates between genetic and phenotypic laboratory parameters, and clinical treatment outcomes, would be exceedingly valuable.
Pipeline for future therapeutic options
There are no novel antimicrobial options for the effective treatment of gonorrhea in late development [1,11,14,17,19,23]. N. gonorrhoeae has rapidly developed resistance to all antimicrobials introduced for first-line therapy during the last 70–80 years [1,11,17]. Given the fear of emergence of untreatable gonorrhea, it is essential to think ‘outside the box’ and investigate new targets, compounds and strategies for treatment, and, ideally, to develop an effective gonococcal vaccine.
Pleuromutilin antimicrobials are potent inhibitors of bacterial protein synthesis that bind to novel sites on bacterial ribosome, which are active against gonococci and suitable for systemic use [132,133]. Both the pleuromutilins and novel inhibitors of bacterial topoisomerases targeting regions different from the fluoroquinolone-binding site [134] may deserve attention for potential antimicrobial treatment of gonorrhea.
Other novel compounds that are under development or could be developed include: species-specific FabI inhibitors with activity against gonococci, such as MUT056399 [135]; inhibitors of efflux pumps that increase the susceptibility to certain antimicrobials, innate host defense and toxic metabolites [136]; LpxC inhibitors having potent antigonococcal activity [137]; molecules mimicking host defensins; and host defense peptides such as LL-37 (multifunctional cathelicidin peptide) [138].
Prospects for a gonococcal vaccine
The search for a gonococcal vaccine is complicated, since even a natural uncomplicated gonococcal infection results only in a mostly local, relatively low and non-persistent immune response that provides little to no protection to reinfection even with an identical strain. To date, no vaccine has been developed, despite the fact that whole organisms, numerous purified gonococcal components and recombinant proteins have been tested. The major obstacles for developing a vaccine have been: gonococcal production of non-bactericidal antibodies (e.g., against Rmp) that block bactericidal antibodies (e.g., against PorB and LOS); sialylation of gonococci and subsequent binding of alternative complement regulator factor H; potential elicitation of gonococcal IL-17 responses that suppress Th1/Th2 adaptive immune responses to natural infection; high antigenic heterogeneity in gonococci (including antigenic variability and phase variation of surface structures, e.g., pili, Opa proteins and LOS); general lack of correlates for immune protection; and lack of an ideal animal model for this obligate human pathogen [139–141]. Nevertheless, several recent advances may enable future gonococcal vaccine research: development of transgenic mouse models expressing human genes required for gonococcal infection; better definition of the course of natural infection through the use of experimental infection in male volunteers [142]; enhanced knowledge regarding development of polyvalent vaccines (which might be required for gonorrhea) gained from the reverse vaccinology technology used for meningococcal vaccines [143]; modern molecular biology, including genome sequencing for the identification of novel single or multiple vaccine candidate antigens; recombinant protein vaccines; discoveries in genital mucosal immunology; and intranasal immunization for increased local mucosal immune responses [140,141,144]. A vaccine that generates a partial immunity, which might not inhibit uncomplicated gonorrhea but eradicates the severe complications of the disease, would still be valuable.
Future perspective
It appears inevitable that ceftriaxone-resistant gonococcal strains with retained resistance determinants to previously recommended antimicrobials will spread internationally, possibly heralding an era of untreatable gonorrhea. This would be an exceedingly serious public health problem that would result in substantial morbidity from the severe sequelae caused by untreated gonorrhea worldwide.
Accordingly, timely, decisive, multidisciplinary and multicomponent public health actions are essential worldwide. Strict implementation of the WHO global action plan [33,202] and region-specific action/response plans [203,204] is crucial. The WHO Global GASP, in close liaison with other regional/national GASPs, needs to: substantially enhance gonococcal AMR surveillance worldwide, with a focus on ESCs; inform public health authorities about national and regional treatment guidelines; optimize early detection of emerging AMR; and support the early identification, verification and follow-up (including tracing of sexual contacts) of treatment failures [33,202,207].
Currently, quality-assured surveillance of gonococcal AMR and treatment failures remains sporadic, limited or lacking in many regions globally [1,12,20,59–61]. The limited use of gonococcal culture and AMR testing worldwide is mostly the result of using syndromic management and microscopy for diagnosis in less-resourced settings, and nucleic acid amplification tests in more resourced settings. It is crucial to culture more isolates, examine AMR, and identify specific ESC resistance determinants, to facilitate AMR surveillance and develop sensitive and specific genetic ESC resistance testing assays. The recently revised WHO standards [207] describe appropriate sampling, transportation and culture of gonococcal isolates, quality-assured AMR testing using WHO gonococcal reference strains [82] and methods to increase the sampling for AMR testing, such as using targeted culture of N. gonorrhoeae microscopy and/or nucleic acid amplification test-positive samples. Furthermore, well-established GASPs should increase the number and representativeness of gonococcal isolates tested (most GASPs include mainly isolates from symptomatic male urethritis from relatively few clinics per country) and epidemiological data collected to prevent emergence of AMR in overlooked populations. Finally, it is also crucial to optimize and, as much as feasible, harmonize the methods, interpretative criteria (breakpoints) and internal and external quality assurance used globally. This is essential to be able to compare validated and quality assured AMR data internationally.
The WHO global action plan [33,202] additionally stresses the importance of combining enhanced surveillance of AMR and treatment failures with a public health response to mitigate the emergence, spread and impact of gonorrhea, particularly ESC-resistant gonococci. Accordingly, crucial actions also include: encouraging increased awareness of gonococcal AMR; correct use of antimicrobials; effective early prevention (e.g., information, counseling and safe sex), diagnosis and management of gonorrhea; effective drug regulation and prescription policies (using only ideal antimicrobials of appropriate quality and optimized doses); development of molecular methods for detecting and monitoring ESC resistance; and, most importantly, research (basic science, in vitro and in vivo studies) to identify new effective treatment strategies, novel antimicrobials and ideally a vaccine (Box 1) [1,33,202]. An increased focus on preventing, diagnosing and treating pharyngeal gonorrhea, with special attention to pharmacokinetic/pharmacodynamic considerations, is also crucial. This includes increasing the sampling of pharyngeal specimens and promoting condom use when practicing oral sex. Pharyngeal infections are harder to treat than urogenital infection, they are relatively common and represent an asymptomatic reservoir for infection and emergence of resistance [1,3,14,17,19,20,34–36,62–64]. Finally, widespread implementation of test-of-cure is important to identify treatment failures and reinfection – particularly, but not only, for pharyngeal infections.
Genetic methods for detecting and monitoring ESC resistance would be extremely valuable, and some methods identifying different or specific penA mosaic alleles or other chromosomal AMR determinants have been developed [71,145–149]. Unfortunately, the currently available penA mosaic PCR detection methods have suboptimal sensitivity and specificity in their predictive power of ESC resistance, in general, and are suboptimal for clinical specimens. The reasons for this are that the MICs of ESCs for strains with different mosaic and Ala501-altered non-mosaic penA alleles vary markedly, making it crucial to sequence the penA gene (rather than simply using some penA mosaic PCR that detects all the different types of penA mosaic alleles or only a specific penA mosaic allele) and identify other AMR determinants in these isolates. Additionally, most penA mosaic sequences originate in commensal Neisseria species, making it hard to determine ESC resistance particularly in extragenital specimen such as those taken from the pharynx [1,8,23,25,72,77,80,83,84]. Initially, statistical associations between specific amino acid alterations and elevated ESC MICs are exceedingly important to identify. However, this approach also requires subsequent verification of the amino acid alterations (including their possible epistasis) that enhance the MICs of ESCs by transformation of site-directed penA mutants into isogenic strain backgrounds. It is also important to agree on the nomenclature of gonococcal penA alleles (or PBP2 alleles), which includes generating an internationally accessible database containing the full-length sequence, the name of the allele, and, ideally, appropriate additional microbiological, genetic and epidemiologic data.
Nevertheless, accumulating knowledge regarding ESC resistance determinants combined with recent advances in genome sequencing, nanotechnology and other molecular approaches might enable sensitive and specific genetic ESC resistance assays (ideally at point-of-care [POC]), as well as genetic POC diagnostic tests, which might substantially improve early detection, particularly in less resourced settings worldwide. These POC tests should be able to detect not only ESC resistance/susceptibility, but also resistance/susceptibility to many of the antimicrobials previously recommended for treatment of gonorrhea, and the results should be available in a timely manner and used not only for gonococcal AMR surveillance, but also to direct the treatment of the individual patient. Nevertheless, it is essential to retain culture and phenotypic AMR testing methods, that is, to be able to detect novel resistance and inform the design and redesign of the genetic AMR assays. New sequencing technologies can also substantially improve the molecular epidemiological typing of gonococci [150], elucidate the dynamics of the national and international spread of AMR gonococcal clones (microepidemiology and microepidemiology), and detail the evolution of successful AMR gonococcal clones and their AMR determinants, including prediction of emergence.
Unfortunately, the aforementioned action/response plans will only delay and limit, but not halt, the inevitable spread of ceftriaxone resistance globally. Therefore, it is essential to develop novel treatment strategies and antimicrobials in a timely manner. Using an increased dose of ceftriaxone will provide a short-term solution, but introduction of dual antimicrobial therapy (currently ceftriaxone and azithromycin) covering concurrent chlamydial infection [19,64,106], is recommended for most settings and, hopefully, will also inhibit future development of gonococcal AMR. Additional research is needed regarding which antimicrobials (and at what dosages) to combine, and to document efficacy, side effects, pharmacokinetic/pharmacodynamic parameters, resistance development (in gonococci and bystander organisms), in vitro methods for synergy testing and microbiological resistance breakpoints. Unfortunately, gonococcal strains with decreased susceptibility/resistance to ceftriaxone with concomitant resistance to both azithromycin and doxycycline are already circulating globally, making it crucial to develop new alternatives for dual antimicrobial therapy. Furthermore, dual antimicrobial therapy, especially with injectable antimicrobials, might not be feasible and/or affordable in less-resourced settings (with a substantial gonorrhea burden) and, thus, may not significantly mitigate AMR emergence and global spread.
Accordingly, from a global perspective, an antimicrobial monotherapy appears essential [1,17,23,25], however, currently there are no promising options [1,3,11,14,19,23,25]. Spectinomycin is a theoretical option, but it is not available in many countries and resistance can be rapidly selected [1,3,14,114,115]. Nevertheless, it would be valuable to have spectinomycin available to treat emergent ESC-resistant cases and possibly for inclusion in a dual antimicrobial treatment regimen, which could inhibit the development of resistance and effectively treat gonorrhoea, including pharyngeal infection. Although new antimicrobials (i.e., gentamicin [116–119], solithromycin [120] and ertapenem [125]) have been suggested for treatment, comprehensive and quality-assured in vitro and/or in vivo evaluation of these antimicrobials remains lacking, and none appear ideal for single antimicrobial therapy. N. gonorrhoeae has rapidly developed resistance to all antimicrobials introduced for first-line therapy during the last 70–80 years [1,11,17] and, thus, it is essential to develop new antimicrobials, investigate novel non-antimicrobial targets and define the mechanisms of action for more sustainable treatment. A gonococcal vaccine would be especially useful, however progress in this area remains minimal.
In conclusion, an era of untreatable gonorrhea may be approaching, which represents a serious public health problem. It is crucial to implement action/response plans at global and national levels, focus research and funding on enhanced surveillance of gonococcal AMR (phenotypic and genetic) and treatment failures, and to improve the prevention, early diagnosis and effective treatment of gonorrhea. Novel effective treatment strategies, new antimicrobials (or other compounds) and, ideally, a gonococcal vaccine should be developed.
Box 1. Some of the main components and actions of the WHO global action plan to control the spread and impact of antimicrobial resistance in Neisseria gonorrhoeae.
-
■
Advocacy for increased awareness on correct use of antibiotics among healthcare providers and the consumers, particularly in key populations, including men who have sex with men and sex workers
-
■
Effective prevention, diagnosis and control of gonorrhea, using prevention messages, interventions, appropriate and recommended diagnosis and treat ment regimens
-
■
Systematic monitoring, early detection and follow-up of treatment failures with recommended treatment (cefixime and ceftriaxone) by developing a standard case definition of treatment failure and protocols for verification, reporting and management of failure
-
■
Effective drug regulations and prescription policies
-
■
Strengthened antimicrobial resistance surveillance, especially in countries with a high burden of gonorrhea (and/or gonococcal antimicrobial resistance), other sexually transmitted infections and HIV
-
■
Capacity building to establish regional networks of laboratories to perform quality-assured gonococcal culture and antimicrobial susceptibility testing
-
■
Research to identify new molecular methods for detecting and monitoring antimicrobial resistance
-
■
Research (basic science, in vitro and clinical studies) to identify alternative effective strategies and/or novel antimicrobials (or other effective compounds) for treatment of gonorrhea (and, ideally, a vaccine)
Executive summary.
Public health concerns regarding gonorrhea & antimicrobial-resistant Neisseria gonorrhoeae
-
■
Gonorrhea burden and multidrug-resistance in N. gonorrhoeae remain major public health concerns that are poorly surveyed and controlled in global terms.
Evolution, origin & spread of antimicrobial resistance in N. gonorrhoeae
-
■
Since the beginning of the antimicrobial era, N. gonorrhoeae has repeatedly demonstrated an extraordinary capacity to develop resistance to all antimicrobials introduced for treatment, mostly within only 1–2 decades.
-
■
The antimicrobial resistance determinants do not appear to significantly decrease the biological fitness of the gonococci, with some even increasing fitness, and are stably maintained in the gonococcal population decades after withdrawal of the specific antimicrobials from treatment regimens.
Emergence & spread of in vitro & in vivo decreased susceptibility/resistance to extended-spectrum cephalosporins
-
■
N. gonorrhoeae has now demonstrated its capacity to develop resistance to the extended-spectrum cephalosporin ceftriaxone, the last remaining option for first-line empiric monotherapy of gonorrhea, which has resulted in treatment failures. An era of untreatable gonorrhea may be approaching, which represents an exceedingly serious global public health problem.
Recent verified treatment failures with cefixime & ceftriaxone, international spread of successful gonococcal strains & the first isolation of strains with high-level ceftriaxone resistance
-
■
During recent years, cefixime treatment failures have been verified in many countries, and the first confirmed clinical failures with ceftriaxone were recently reported.
-
■
The gonococcal clones MLST ST1901 (commonly NG-MAST ST1407) and MLST ST7363, both of which likely originated in Japan but have now spread worldwide, have been responsible for most of these verified treatment failures with cefixime and ceftriaxone. Both clones have also shown the capacity to develop high-level resistance to ceftriaxone.
-
■
It is crucial to survey for and appropriately verify treatment failures with cefixime and ceftriaxone.
Decreased susceptibility/resistance to extended-spectrum cephalosporins: genetic resistance determinants
-
■
The mechanisms of resistance to extended-spectrum cephalosporins in N. gonorrhoeae are complex and include diverse alterations in many genes such as penA, mtrR and porB1b. It is crucial to elucidate the resistance determinants in gonococcal isolates responsible for treatment failures with extended-spectrum cephalosporins, as well as in isolates showing high in vitro resistance. Rapid assays for genetic determination of resistance to extended-spectrum cephalosporins would be exceedingly valuable for resistance surveillance as well as clinical management of gonorrhea patients.
Public health actions to meet the large challenge of emergence of high-level ceftriaxone-resistant & untreatable gonorrhea
-
■
Essential actions to meet the large public health challenge of the possible emergence of untreatable gonorrhea include implementing the WHO global action plan and supporting regional/national response plans globally and nationally; enhancing surveillance of gonococcal antimicrobial resistance, treatment failures (including appropriate verification and follow-up of failure) and antimicrobial use/misuse worldwide; and improving prevention, early diagnosis and treatment to reduce the global burden of gonorrhea.
Future options for treatment & prevention of gonorrhea
-
■
There are few promising candidates in sight for new (or rediscovery of old) antimicrobials or other compounds for effective treatment of gonorrhea.
-
■
Dual antimicrobial therapy (presently, ceftriaxone plus azithromycin) can be recommended, particularly for settings suffering from decreased susceptibility/resistance to extended-spectrum cephalosporins. However, currently available antimicrobials (in current doses or in increased and/or multiple doses) used alone or in combination may offer little prospect as long-term solutions for effective gonorrhea treatment.
-
■
Novel effective antimicrobials (or other compounds) for treatment of gonorrhea, and ideally a gonococcal vaccine must be developed.
Acknowledgments
Financial & competing interests disclosure
Research at the WHO Collaborating Centre for Gonorrhoea and other STIs is supported by grants from the WHO, the Örebro County Council Research Committee and the Foundation for Medical Research at Örebro University Hospital, Sweden (M Unemo). Research by RA Nicholas is supported by grants from the National Institutes of Health.
Footnotes
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
Papers of special note have been highlighted as:
■ of interest
■■ of considerable interest
- 1■.Tapsall JW, Ndowa F, Lewis DA, Unemo M. Meeting the public health challenge of multidrug- and extensively drug-resistant Neisseria gonorrhoeae. Expert Rev. Anti. Infect. Ther. 2009;7:821–834. doi: 10.1586/eri.09.63. [Comprehensive review describing initiatives by the WHO to enhance global antimicrobial resistance surveillance and meet the public health challenges posed by international spread of multidrug-resistant and extensively drug-resistant Neisseria gonorrhoeae] [DOI] [PubMed] [Google Scholar]
- 2.Cohen MS, Hoffman IF, Royce RA, et al. Reduction of concentration of HIV-1 in semen after treatment of urethritis: implications for prevention of sexual transmission of HIV-1. Lancet. 1997;349:1868–1873. doi: 10.1016/s0140-6736(97)02190-9. [DOI] [PubMed] [Google Scholar]
- 3.Barry PM, Klausner JD. The use of cephalosporins for gonorrhea: the impending problem of resistance. Expert Opin. Pharmacother. 2009;10:555–577. doi: 10.1517/14656560902731993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bolan GA, Sparling PF, Wasserheit JN. The emerging threat of untreatable gonococcal infection. N. Engl. J. Med. 2012;366:485–487. doi: 10.1056/NEJMp1112456. [DOI] [PubMed] [Google Scholar]
- 5.Centers for Disease Control and Prevention (CDC) Cephalosporin susceptibility among Neisseria gonorrhoeae isolates – United States, 2000–2010. MMWR Morb. Mortal. Wkly Rep. 2011;60:873–877. [PubMed] [Google Scholar]
- 6.Chisholm SA, Alexander S, Desouza-Thomas L, et al. Emergence of a Neisseria gonorrhoeae clone showing decreased susceptibility to cefixime in England and Wales. J. Antimicrob. Chemother. 2011;66:2509–2512. doi: 10.1093/jac/dkr332. [DOI] [PubMed] [Google Scholar]
- 7.Cole MJ, Unemo M, Hoffmann S, Chisholm SA, Ison CA, van de Laar MJ. The European gonococcal antimicrobial surveillance programme, 2009. Euro. Surveill. 2011;16(42):19995. [PubMed] [Google Scholar]
- 8.Golparian D, Hellmark B, Fredlund H, Unemo M. Emergence, spread and characteristics of Neisseria gonorrhoeae isolates with in vitro decreased susceptibility and resistance to extended-spectrum cephalosporins in Sweden. Sex. Transm. Infect. 2010;86:454–460. doi: 10.1136/sti.2010.045377. [DOI] [PubMed] [Google Scholar]
- 9.Kirkcaldy RD, Ballard RC, Dowell D. Gonococcal resistance: are cephalosporins next? Curr. Infect. Dis. Rep. 2011;13:196–204. doi: 10.1007/s11908-011-0169-9. [DOI] [PubMed] [Google Scholar]
- 10.Kubanova A, Frigo N, Kubanov A, et al. The Russian gonococcal antimicrobial susceptibility programme (RU-GASP) – national resistance prevalence in 2007 and 2008, and trends during 2005–2008. Euro. Surveill. 2010;15(14):19533. [PubMed] [Google Scholar]
- 11.Lewis DA. The gonococcus fights back: is this time a knock out? Sex. Transm. Infect. 2010;86:415–421. doi: 10.1136/sti.2010.042648. [DOI] [PubMed] [Google Scholar]
- 12.Lewis DA, Lukehart SA. Antimicrobial resistance in Neisseria gonorrhoeae and Treponema pallidum: evolution, therapeutic challenges and the need to strengthen global surveillance. Sex. Transm. Infect. 2011;87(Suppl. 2):ii39–ii43. doi: 10.1136/sti.2010.047712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Martin I, Jayaraman G, Wong T, Liu G, Gilmour M. Canadian Public Health Laboratory Network. Trends in antimicrobial resistance in Neisseria gonorrhoeae isolated in Canada: 2000–2009. Sex. Transm. Dis. 2011;38:892–898. doi: 10.1097/OLQ.0b013e31822c664f. [DOI] [PubMed] [Google Scholar]
- 14.Newman LM, Moran JS, Workowski KA. Update on the management of gonorrhea in adults in the United States. Clin. Infect. Dis. 2007;44(Suppl.3):S84–S101. doi: 10.1086/511422. [DOI] [PubMed] [Google Scholar]
- 15.Tanaka M, Koga Y, Nakayama H, et al. Antibiotic-resistant phenotypes and genotypes of Neisseria gonorrhoeae isolates in Japan: identification of strain clusters with multidrug-resistant phenotypes. Sex. Transm. Dis. 2011;38:871–875. doi: 10.1097/OLQ.0b013e31821d0f98. [DOI] [PubMed] [Google Scholar]
- 16.Tanaka M, Shimojima M, Saika T, et al. Nationwide antimicrobial susceptibility survey of Neisseria gonorrhoeae isolates in Japan. Kansenshogaku Zasshi. 2011;85:360–365. doi: 10.11150/kansenshogakuzasshi.85.360. [DOI] [PubMed] [Google Scholar]
- 17.Unemo M, Shafer WM. Antibiotic resistance in Neisseria gonorrhoeae: origin, evolution, and lessons learned for the future. Ann. NY Acad. Sci. 2011;1230:E19–E28. doi: 10.1111/j.1749-6632.2011.06215.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.WHO Western Pacific and South East Asian Gonococcal Antimicrobial Surveillance Programmes Surveillance of antibiotic resistance in Neisseria gonorrhoeae in the WHO Western Pacific and South East Asian Regions, 2010. Commun. Dis. Intell. 2012;36:95–100. [PubMed] [Google Scholar]
- 19.Workowski KA, Berman S. Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm. Rep. 2010;59(RR-12):1–110. [PubMed] [Google Scholar]
- 20■.Stoltey JE, Barry PM. The use of cephalosporins for gonorrhea: an update on the rising problem of resistance. Expert Opin. Pharmacother. 2012;13:1411–1420. doi: 10.1517/14656566.2012.690396. [Comprehensive updated review emphasizing many important actions needed to meet the challenge of multidrug-resistant gonorrhea] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Deguchi T, Yasuda M, Yokoi S, et al. Treatment of uncomplicated gonococcal urethritis by double-dosing of 200 mg cefixime at a 6-h interval. J. Infect. Chemother. 2003;9:35–39. doi: 10.1007/s10156-002-0204-8. [DOI] [PubMed] [Google Scholar]
- 22.Ison CA, Hussey J, Sankar KN, Evans J, Alexander S. Gonorrhoea treatment failures to cefixime and azithromycin in England, 2010. Euro. Surveill. 2011;16(14):19833. [PubMed] [Google Scholar]
- 23■■.Ohnishi M, Golparian D, Shimuta K, et al. Is Neisseria gonorrhoeae initiating a future era of untreatable gonorrhea? Detailed characterization of the first strain with high-level resistance to ceftriaxone. Antimicrob. Agents Chemother. 2011;55:3538–3545. doi: 10.1128/AAC.00325-11. [Detailed phenotypic and genetic characterization of the first N. gonorrhoeae strain with high-level ceftriaxone resistance (identified in Japan)] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Unemo M, Golparian D, Hestner A. Ceftriaxone treatment failure of pharyngeal gonorrhoea verified by international recommendations, Sweden, July 2010. Euro. Surveill. 2011;16(6):19792. [PubMed] [Google Scholar]
- 25■.Unemo M, Golparian D, Nicholas R, Ohnishi M, Gallay A, Sednaoui P. High-level cefixime- and ceftriaxone-resistant N. gonorrhoeae in France: novel penA mosaic allele in a successful international clone causes treatment failure. Antimicrob. Agents Chemother. 2012;56:1273–1280. doi: 10.1128/AAC.05760-11. [Detailed phenotypic and genetic characterization of the second N. gonorrhoeae strain with high-level ceftriaxone resistance (identified in France)] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Unemo M, Golparian D, Stary A, Eigentler A. First Neisseria gonorrhoeae strain with resistance to cefixime causing gonorrhoea treatment failure in Austria, 2011. Euro. Surveill. 2011;16(43):19998. [PubMed] [Google Scholar]
- 27.Unemo M, Golparian D, Syversen G, Vestrheim DF, Moi H. Two cases of verified clinical failures using internationally recommended first-line cefixime for gonorrhoea treatment, Norway, 2010. Euro. Surveill. 2010;15(47):19721. doi: 10.2807/ese.15.47.19721-en. [DOI] [PubMed] [Google Scholar]
- 28.Unemo M, Golparian D, Potočnik M, Jeverica S. Treatment failure of pharyngeal gonorrhoea with internationally recommended first-line ceftriaxone verified in Slovenia, September 2011. Euro. Surveill. 2012;17(25):20200. [PubMed] [Google Scholar]
- 29.Tapsall J, Read P, Carmody C, et al. Two cases of failed ceftriaxone treatment in pharyngeal gonorrhoea verified by molecular microbiological methods. J. Med. Microbiol. 2009;58(Pt 5):683–687. doi: 10.1099/jmm.0.007641-0. [DOI] [PubMed] [Google Scholar]
- 30.Yokoi S, Deguchi T, Ozawa T, et al. Threat to cefixime treatment of gonorrhea. Emerg. Infect. Dis. 2007;13:1275–1277. doi: 10.3201/eid1308.060948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cámara J, Serra J, Ayats J, et al. Molecular characterization of two high-level ceftriaxone-resistant Neisseria gonorrhoeae isolates detected in Catalonia, Spain. J. Antimicrob. Chemother. 2012;67:1858–1860. doi: 10.1093/jac/dks162. [DOI] [PubMed] [Google Scholar]
- 32.Whiley DM, Goire N, Lahra MM, et al. The ticking time bomb: escalating antibiotic resistance in Neisseria gonorrhoeae is a public health disaster in waiting. J. Antimicrob. Chemother. 2012;67:2059–2061. doi: 10.1093/jac/dks188. [DOI] [PubMed] [Google Scholar]
- 33.Ndowa F, Lusti-Narasimhan M, Unemo M. The serious threat of multidrug-resistant and untreatable gonorrhoea: the pressing need for global action to control the spread of antimicrobial resistance, and mitigate the impact on sexual and reproductive health. Sex. Transm. Infect. 2012;88:317–318. doi: 10.1136/sextrans-2012-050674. [DOI] [PubMed] [Google Scholar]
- 34.Furuya R, Onoye Y, Kanayama A, et al. Antimicrobial resistance in clinical isolates of Neisseria subflava from the oral cavities of a Japanese population. J. Infect. Chemother. 2007;13:302–304. doi: 10.1007/s10156-007-0541-8. [DOI] [PubMed] [Google Scholar]
- 35.Saika T, Nishiyama T, Kanayama A, et al. Comparison of Neisseria gonorrhoeae isolates from the genital tract and pharynx of two gonorrhea patients. J. Infect. Chemother. 2001;7:175–179. doi: 10.1007/s101560100031. [DOI] [PubMed] [Google Scholar]
- 36.Tanaka M, Nakayama H, Huruya K, et al. Analysis of mutations within multiple genes associated with resistance in a clinical isolate of Neisseria gonorrhoeae with reduced ceftriaxone susceptibility that shows a multidrug-resistant phenotype. Int. J. Antimicrob. Agents. 2006;27:20–26. doi: 10.1016/j.ijantimicag.2005.08.021. [DOI] [PubMed] [Google Scholar]
- 37.Ohnishi M, Watanabe Y, Ono E, et al. Spreading of a chromosomal cefixime-resistant penA gene among different Neisseria gonorrhoeae lineages. Antimicrob. Agents Chemother. 2010;54:1060–1067. doi: 10.1128/AAC.01010-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dillon JA, Parti RP. Fluoroquinolone resistance in Neisseria gonorrhoeae: fitness cost or benefit? J. Infect. Dis. 2012;205:1775–1777. doi: 10.1093/infdis/jis281. [DOI] [PubMed] [Google Scholar]
- 39.Jerse AE, Sharma ND, Simms AN, Crow ET, Snyder L, Shafer WM. A gonococcal efflux pump system enhances bacterial survival in a female mouse model of genital tract infection. Infect. Immun. 2003;71:5576–5582. doi: 10.1128/IAI.71.10.5576-5582.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Warner DM, Folster JP, Shafer WM, Jerse AE. Regulation of the MtrC-MtrD-MtrE efflux-pump system modulates the in vivo fitness of Neisseria gonorrhoeae. J. Infect. Dis. 2007;196:1804–1812. doi: 10.1086/522964. [DOI] [PubMed] [Google Scholar]
- 41.Warner DM, Shafer WM, Jerse AE. Clinically relevant mutations that cause derepression of the Neisseria gonorrhoeae MtrC-MtrD-MtrE Efflux pump system confer different levels of antimicrobial resistance and in vivo fitness. Mol. Microbiol. 2008;70:462–478. doi: 10.1111/j.1365-2958.2008.06424.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Johnson PJ, Stringer VA, Shafer WM. Off-target gene regulation mediated by transcriptional repressors of antimicrobial efflux pump genes in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2011;55:2559–2565. doi: 10.1128/AAC.00010-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kunz AN, Begum AA, Wu H. Impact of fluoroquinolone resistance mutations on gonococcal fitness and in vivo selection for compensatory mutations. J. Infect. Dis. 2012;205:1821–1829. doi: 10.1093/infdis/jis277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tapsall J, Whiley D, Sloots T. Applications of molecular testing in clinical laboratories for the diagnosis and control of gonorrhea. Future Microbiol. 2006;1:317–324. doi: 10.2217/17460913.1.3.317. [DOI] [PubMed] [Google Scholar]
- 45.Tapsall JW, Limnios EA, Murphy DM, Australian Gonococcal Surveillance Programme Analysis of trends in antimicrobial resistance in Neisseria gonorrhoeae isolated in Australia 1997–2006. J. Antimicrob. Chemother. 2008;61:150–155. doi: 10.1093/jac/dkm434. [DOI] [PubMed] [Google Scholar]
- 46.Morris SR, Knapp JS, Moore DF, et al. Using strain typing to characterise a fluoroquinolone-resistant Neisseria gonorrhoeae transmission network in southern California. Sex. Transm. Infect. 2008;84:290–291. doi: 10.1136/sti.2008.030163. [DOI] [PubMed] [Google Scholar]
- 47.Tapsall J. Antibiotic resistance in Neisseria gonorrhoeae is diminishing available treatment for gonorrhea: some possible remedies. Exp. Rev. Anti Infect. Ther. 2006;4:619–628. doi: 10.1586/14787210.4.4.619. [DOI] [PubMed] [Google Scholar]
- 48.Akasaka S, Muratani T, Yamada Y, Inatomi H, Takahashi K, Matsumoto T. Emergence of cephem- and aztreonam-high-resistant Neisseria gonorrhoeae that does not produce beta-lactamase. J. Infect. Chemother. 2001;7:49–50. doi: 10.1007/s101560170034. [DOI] [PubMed] [Google Scholar]
- 49.Tanaka M, Nakayama H, Tunoe H, et al. A remarkable reduction in the susceptibility of Neisseria gonorrhoeae isolates to cephems and the selection of antibiotic regimens for the single-dose treatment of gonococcal infection in Japan. J. Infect. Chemother. 2002;8:81–86. doi: 10.1007/s101560200011. [DOI] [PubMed] [Google Scholar]
- 50.Ito M, Yasuda M, Yokoi S, et al. Remarkable increase in central Japan in 2001–2002 of Neisseria gonorrhoeae isolates with decreased susceptibility to penicillin, tetracycline, oral cephalosporins, and fluoroquinolones. Antimicrob. Agents Chemother. 2004;48:3185–3187. doi: 10.1128/AAC.48.8.3185-3187.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chisholm SA, Mouton JW, Lewis DA, Nichols T, Ison CA, Livermore DM. Cephalosporin MIC creep among gonococci: time for a pharmacodynamic rethink? J. Antimicrob. Chemother. 2010;65:2141–2148. doi: 10.1093/jac/dkq289. [DOI] [PubMed] [Google Scholar]
- 52.Takahashi S, Kurimura Y, Hashimoto J, et al. Antimicrobial susceptibility and penicillin-binding protein 1 and 2 mutations in Neisseria gonorrhoeae isolated from male urethritis in Sapporo, Japan. J. Infect. Chemother. 2012 doi: 10.1007/s10156-012-0450-3. doi:10.1007/s10156-012-0450-3. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 53.Lo JY, Ho KM, Leung AO, et al. Ceftibuten resistance and treatment failure of Neisseria gonorrhoeae infection. Antimicrob. Agents Chemother. 2008;52:3564–3567. doi: 10.1128/AAC.00198-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Martin I, Sawatzky P, Allen V, et al. Emergence and characterization of Neisseria gonorrhoeae isolates with decreased susceptibilities to ceftriaxone and cefixime in Canada: 2001–2010. Sex. Transm. Dis. 2012;39:316–323. doi: 10.1097/OLQ.0b013e3182401b69. [DOI] [PubMed] [Google Scholar]
- 55.Li SY. Global transmission of multiple-drug resistant Neisseria gonorrhoeae strains refractive to cephalosporin treatment. J. Formos. Med. Assoc. 2012;111:463–464. doi: 10.1016/j.jfma.2012.03.004. [DOI] [PubMed] [Google Scholar]
- 56.Starnino S, Galarza P, Carvallo ME, et al. Retrospective analysis of antimicrobial susceptibility trends (2000–2009) in Neisseria gonorrhoeae isolates from countries in Latin America and the Caribbean shows evolving resistance to ciprofloxacin, azithromycin and decreased susceptibility to ceftriaxone. Sex. Transm. Dis. 2012;39:813–821. doi: 10.1097/OLQ.0b013e3182631c9f. [DOI] [PubMed] [Google Scholar]
- 57.Su X, Jiang F, Qimuge, Dai X, Sun H, Ye S. Surveillance of antimicrobial susceptibilities in Neisseria gonorrhoeae in Nanjing, China, 1999–2006. Sex. Transm. Dis. 2007;34:995–999. doi: 10.1097/OLQ.0b013e3180ca8f24. [DOI] [PubMed] [Google Scholar]
- 58.Liao M, Gu WM, Yang Y, Dillon JA. Analysis of mutations in multiple loci of Neisseria gonorrhoeae isolates reveals effects of PIB, PBP2 and MtrR on reduced susceptibility to ceftriaxone. J. Antimicrob. Chemother. 2011;66:1016–1023. doi: 10.1093/jac/dkr021. [DOI] [PubMed] [Google Scholar]
- 59.Unemo M, Shipitsyna E, Domeika M, on behalf of the Eastern European Sexual and Reproductive Health (EE SRH) Network Antimicrobial Resistance Group Recommended antimicrobial treatment of uncomplicated gonorrhoea in 2009 in 11 East European countries: implementation of a Neisseria gonorrhoeae antimicrobial susceptibility programme in this region is crucial. Sex. Transm. Infect. 2010;86:442–444. doi: 10.1136/sti.2010.042317. [DOI] [PubMed] [Google Scholar]
- 60.Unemo M, Shipitsyna E, Domeika M, the Eastern European Sexual and Reproductive Health (EE SRH) Network Antimicrobial Resistance Group Gonorrhoea surveillance, laboratory diagnosis and antimicrobial susceptibility testing of Neisseria gonorrhoeae in 11 countries of the eastern part of the WHO European region. APMIS. 2011;119:643–649. doi: 10.1111/j.1600-0463.2011.02780.x. [DOI] [PubMed] [Google Scholar]
- 61.Dillon JA. Sustainable antimicrobial surveillance programs essential for controlling Neisseria gonorrhoeae superbug. Sex. Transm. Dis. 2011;38:899–901. doi: 10.1097/OLQ.0b013e318232459b. [DOI] [PubMed] [Google Scholar]
- 62.Moran JS, Levine WC. Drugs of choice in the treatment of uncomplicated gonococcal infection. Clin. Infect. Dis. 1995;20(Suppl. 1):S47–S65. doi: 10.1093/clinids/20.supplement_1.s47. [DOI] [PubMed] [Google Scholar]
- 63.Moran JS. Treating uncomplicated Neisseria gonorrhoeae infections: is the anatomic site of infection important? Sex. Transm. Dis. 1995;22:39–47. doi: 10.1097/00007435-199501000-00007. [DOI] [PubMed] [Google Scholar]
- 64■■.Bignell C, Fitzgerald M, Guideline Development Group UK national guideline for the management of gonorrhoea in adults, 2011. Int. J. STD AIDS. 2011;22:541–547. doi: 10.1258/ijsa.2011.011267. [Updated guidelines for the management of gonorrhea in adults in the UK] [DOI] [PubMed] [Google Scholar]
- 65.Ohnishi M, Saika T, Hoshina S, et al. Ceftriaxone-resistant Neisseria gonorrhoeae, Japan. Emerg. Infect. Dis. 2010;17(1):148–149. doi: 10.3201/eid1701.100397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chisholm S, Unemo M, Quaye N-N, et al. Molecular epidemiological typing within the European gonococcal antimicrobial resistance surveillance programme (EURO-GASP) reveals predominance of a multi-drug resistant clone.. Presented at: 18th International Pathogenic Neisseria Conference; Wurzburg, Germany. 9–14 September 2012. [Google Scholar]
- 67.Buono S, Wu A, Hess DC, et al. Using the Neisseria gonorrhoeae multiantigen sequence-typing method to assess strain diversity and antibiotic resistance in San Francisco, California. Microb. Drug Resist. 2012;18:510–517. doi: 10.1089/mdr.2011.0229. [DOI] [PubMed] [Google Scholar]
- 68.Heymans R, Bruisten SM, Golparian D, Unemo M, de Vries HJ, van Dam AP. Clonally related Neisseria gonorrhoeae isolates with decreased susceptibility to the extended-spectrum cephalosporin cefotaxime in Amsterdam, The Netherlands. Antimicrob. Agents Chemother. 2012;56:1516–1522. doi: 10.1128/AAC.05481-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Huang CT, Yen MY, Wong WW, et al. Characteristics and dissemination of mosaic penicillin-binding protein 2-harboring multidrug-resistant Neisseria gonorrhoeae isolates with reduced cephalosporin susceptibility in northern Taiwan. Antimicrob. Agents Chemother. 2010;54:4893–4895. doi: 10.1128/AAC.00482-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mavroidi A, Tzelepi E, Siatravani E, Godoy D, Miriagou V, Spratt BG. Analysis of emergence of quinolone-resistant gonococci in Greece by combined use of Neisseria gonorrhoeae multiantigen sequence typing and multilocus sequence typing. J. Clin. Microbiol. 2011;49:1196–1201. doi: 10.1128/JCM.02233-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Pandori M, Barry PM, Wu A, et al. Mosaic penicillin-binding protein 2 in Neisseria gonorrhoeae isolates collected in 2008 in San Francisco, California. Antimicrob. Agents Chemother. 2009;53:4032–4034. doi: 10.1128/AAC.00406-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tapsall JW, Ray S, Limnios A. Characteristics and population dynamics of mosaic penA allele-containing Neisseria gonorrhoeae isolates collected in Sydney, Australia, in 2007–2008. Antimicrob. Agents Chemother. 2010;54:554–556. doi: 10.1128/AAC.01108-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hess D, Wu A, Golparian D, et al. Genome sequencing of a Neisseria gonorrhoeae isolate of a successful international clone with decreased susceptibility/resistance to extended-spectrum cephalosporins. Antimicrob. Agents Chemother. 2012;56(11):5633–5641. doi: 10.1128/AAC.00636-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Allen VG, Farrell DJ, Rebbapragada A, et al. Molecular analysis of antimicrobial resistance mechanisms in Neisseria gonorrhoeae isolates from Ontario, Canada. Antimicrob. Agents Chemother. 2011;55:703–712. doi: 10.1128/AAC.00788-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ameyama S, Onodera S, Takahata M, et al. Mosaic-like structure of penicillin-binding protein 2 gene (penA) in clinical isolates of Neisseria gonorrhoeae with reduced susceptibility to cefixime. Antimicrob. Agents Chemother. 2002;46:3744–3749. doi: 10.1128/AAC.46.12.3744-3749.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ito M, Deguchi T, Mizutani KS, et al. Emergence and spread of Neisseria gonorrhoeae clinical isolates harboring mosaic-like structure of penicillin-binding protein 2 in central Japan. Antimicrob. Agents Chemother. 2005;49:137–143. doi: 10.1128/AAC.49.1.137-143.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lee SG, Lee SG, Lee H, et al. Various penA mutations together with mtrR, porB and ponA mutations in Neisseria gonorrhoeae isolates with reduced susceptibility to cefixime or ceftriaxone. J. Antimicrob. Chemother. 2010;65:669–675. doi: 10.1093/jac/dkp505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lindberg R, Fredlund H, Nicholas R, Unemo M. Neisseria gonorrhoeae isolates with reduced susceptibility to cefixime and ceftriaxone: association with genetic polymorphisms in penA, mtrR, porB1b, and ponA. Antimicrob. Agents Chemother. 2007;51:2117–2122. doi: 10.1128/AAC.01604-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Osaka K, Takakura T, Narukawa K, et al. Analysis of amino acid sequences of penicillin-binding protein 2 in clinical isolates of Neisseria gonorrhoeae with reduced susceptibility to cefixime and ceftriaxone. J. Infect. Chemother. 2008;14:195–203. doi: 10.1007/s10156-008-0610-7. [DOI] [PubMed] [Google Scholar]
- 80.Takahata S, Senju N, Osaki Y, Yoshida T, Ida T. Amino acid substitutions in mosaic penicillin-binding protein 2 associated with reduced susceptibility to cefixime in clinical isolates of Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2006;50:3638–3645. doi: 10.1128/AAC.00626-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81■■.Tomberg J, Unemo M, Davies C, Nicholas RA. Molecular and structural analysis of mosaic variants of penicillin-binding protein 2 conferring decreased susceptibility to expanded-spectrum cephalosporins in Neisseria gonorrhoeae: role of epistatic mutations. Biochemistry. 2010;49:8062–8070. doi: 10.1021/bi101167x. [Detailed analysis and verification of amino acid alterations in PBP2 that are important for decreased susceptibility to expanded-spectrum cephalosporins in N. gonorrhoeae] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Unemo M, Fasth O, Fredlund H, Limnios A, Tapsall JW. Phenotypic and genetic characterization of the 2008 WHO Neisseria gonorrhoeae reference strain panel intended for global quality assurance and quality control of gonococcal antimicrobial resistance surveillance for public health purposes. J. Antimicrob. Chemother. 2009;63:1142–1151. doi: 10.1093/jac/dkp098. [DOI] [PubMed] [Google Scholar]
- 83.Whiley DM, Limnios A, Ray S, Sloots TP, Tapsall JW. Diversity of penA alterations and subtypes in Neisseria gonorrhoeae strains from Sydney, Australia, that are less susceptible to ceftriaxone. Antimicrob. Agents Chemother. 2007;51:3111–3116. doi: 10.1128/AAC.00306-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Whiley DM, Goire N, Lambert SB, et al. Reduced susceptibility to ceftriaxone in Neisseria gonorrhoeae is associated with mutations G542S, P551S and P551L in the gonococcal penicillin-binding protein 2. J. Antimicrob. Chemother. 2010;65:1615–1618. doi: 10.1093/jac/dkq187. [DOI] [PubMed] [Google Scholar]
- 85.Whiley DM, Goire N, Lambert SB, Nissen MD, Sloots TP, Tapsall JW. Reduced susceptibility to ceftriaxone in Neisseria gonorrhoeae is spread internationally by genetically distinct gonococcal populations. J. Antimicrob.Chemother. 2011;66:1186–1187. doi: 10.1093/jac/dkr052. [DOI] [PubMed] [Google Scholar]
- 86■.Zhao S, Duncan M, Tomberg J, Davies C, Unemo M, Nicholas RA. Genetics of chromosomally mediated intermediate resistance to ceftriaxone and cefixime in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2009;53:3744–3751. doi: 10.1128/AAC.00304-09. [Detailed analysis of the effects of penA mosaic allele, and synergy with mtrR and penB, on the susceptibility to extended-spectrum cephalosporins in N. gonorrhoeae] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ochiai S, Sekiguchi S, Hayashi A, et al. Decreased affinity of mosaic-structure recombinant penicillin-binding 2 for oral cephalosporins in Neisseria gonorrhoeae. J. Antimicrob. Chemother. 2007;60:54–60. doi: 10.1093/jac/dkm166. [DOI] [PubMed] [Google Scholar]
- 88.Hagman KE, Pan W, Spratt BG, Balthazar JT, Judd RC, Shafer WM. Resistance of Neisseria gonorrhoeae to antimicrobial hydrophobic agents is modulated by the mtrR CDE efflux system. Microbiology. 1995;141:611–622. doi: 10.1099/13500872-141-3-611. [DOI] [PubMed] [Google Scholar]
- 89.Shafer WM, Folster JP. Towards an understanding of chromosomally mediated penicillin resistance in Neisseria gonorrhoeae: evidence for a porin-efflux pump collaboration. J. Bacteriol. 2006;188:2297–2299. doi: 10.1128/JB.188.7.2297-2299.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Veal WL, Nicholas RA, Shafer WM. Overexpression of the MtrC-MtrD-MtrE efflux pump due to an mtrR mutation is required for chromosomally mediated penicillin resistance in Neisseria gonorrhoeae. J. Bacteriol. 2002;184:5619–5624. doi: 10.1128/JB.184.20.5619-5624.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Zarantonelli L, Borthagaray G, Lee EH, Shafer WM. Decreased azithromycin susceptibility of Neisseria gonorrhoeae due to mtrR mutations. Antimicrob. Agents Chemother. 1999;43:2468–2472. doi: 10.1128/aac.43.10.2468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Johnson SR, Sandul AL, Parekh M, et al. Mutations causing in vitro resistance to azithromycin in Neisseria gonorrhoeae. Int. J. Antimicrob. Agents. 2003;21:414–419. doi: 10.1016/s0924-8579(03)00039-6. [DOI] [PubMed] [Google Scholar]
- 93.Ohneck EA, Zalucki YM, Johnson PJ, et al. A novel mechanism of high-level, broad-spectrum antibiotic resistance caused by a single base pair change in Neisseria gonorrhoeae. MBio. 2011;202(5):e00187–e00111. doi: 10.1128/mBio.00187-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Olesky M, Hobbs M, Nicholas RA. Identification and analysis of amino acid mutations in porin IB that mediate intermediate-level resistance to penicillin and tetracycline in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2002;46:2811–2820. doi: 10.1128/AAC.46.9.2811-2820.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Olesky M, Zhao S, Rosenberg RL, Nicholas RA. Porin-mediated antibiotic resistance in Neisseria gonorrhoeae: ion, solute, and antibiotic permeation through PIB proteins with penB mutations. J. Bacteriol. 2006;188:2300–2308. doi: 10.1128/JB.188.7.2300-2308.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ropp PA, Hu M, Olesky M, Nicholas RA. Mutations in ponA, the gene encoding penicillin-binding protein 1, and a novel locus, penC, are required for high-level chromosomally mediated penicillin resistance in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2002;46:769–777. doi: 10.1128/AAC.46.3.769-777.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Whiley DM, Jacobsson S, Tapsall JW, et al. Alterations of the pilQ gene in Neisseria gonorrhoeae are unlikely contributors to decreased susceptibility to ceftriaxone and cefixime in clinical gonococcal strains. J. Antimicrob. Chemother. 2010;65:2543–2547. doi: 10.1093/jac/dkq377. [DOI] [PubMed] [Google Scholar]
- 98.Zhao S, Tobiason DM, Hu M, Seifert HS, Nicholas RA. The penC mutation conferring antibiotic resistance in Neisseria gonorrhoeae arises from a mutation in the PilQ secretin that interferes with multimer stability. Mol. Microbiol. 2005;57:1238–1251. doi: 10.1111/j.1365-2958.2005.04752.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Helm RA, Barnhart MM, Seifert HS. pilQ missense mutations have diverse effects on PilQ multimer formation, piliation, and pilus function in Neisseria gonorrhoeae. J. Bacteriol. 2007;189:3198–3207. doi: 10.1128/JB.01833-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Nakayama SI, Tribuddharat C, Prombhul S, et al. Molecular analyses of TEM genes and their corresponding penicillinase-producing Neisseria gonorrhoeae isolates in Bangkok, Thailand. Antimicrob. Agents Chemother. 2012;56:916–920. doi: 10.1128/AAC.05665-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Tapsall JW. Implications of current recommendations for third-generation cephalosporin use in the WHO Western Pacific Region following the emergence of multiresistant gonococci. Sex. Transm. Infect. 2009;85:256–258. doi: 10.1136/sti.2008.035337. [DOI] [PubMed] [Google Scholar]
- 102.Bignell C. 2009 European (IUSTI/WHO) guideline on the diagnosis and treatment of gonorrhoea in adults. Int. J. STD AIDS. 2009;20:453–457. doi: 10.1258/ijsa.2009.009160. [DOI] [PubMed] [Google Scholar]
- 103.Japanese Society of Sexually Transmitted Infection Gonococcal infection. Sexually transmitted infections, diagnosis and treatment guidelines 2011. Jpn J. Sex. Transm. Dis. 2011;22(Suppl. 1):52–59. [Google Scholar]
- 104.Muratani T, Inatomi H, Ando Y, Kawai S, Akasaka S, Matsumoto T. Single dose 1 g ceftriaxone for urogenital and pharyngeal infection caused by Neisseria gonorrhoeae. Int. J. Urol. 2008;15:837–842. doi: 10.1111/j.1442-2042.2008.02100.x. [DOI] [PubMed] [Google Scholar]
- 105.Handsfield HH, McCormack WM, Hook EW, III, et al. A comparison of single-dose cefixime with ceftriaxone as treatment for uncomplicated gonorrhea. The Gonorrhea Treatment Study Group. N. Engl. J. Med. 1991;325:1337–1341. doi: 10.1056/NEJM199111073251903. [DOI] [PubMed] [Google Scholar]
- 106.Centers for Disease Control and Prevention (CDC) Update to CDC's Sexually Transmitted Diseases Treatment Guidelines, 2010: oral cephalosporins no longer a recommended treatment for gonococcal infections. MMWR Morb. Mortal. Wkly Rep. 2012;61:590–594. [PubMed] [Google Scholar]
- 107.Furuya R, Nakayama H, Kanayama A, et al. In vitro synergistic effects of double combinations of beta-lactams and azithromycin against clinical isolates of Neisseria gonorrhoeae. J. Infect. Chemother. 2006;12:172–176. doi: 10.1007/s10156-006-0445-z. [DOI] [PubMed] [Google Scholar]
- 108.Sathia L, Ellis B, Phillip S, et al. Pharyngeal gonorrhoea – is dual therapy the way forward? Int. J. STD AIDS. 2007;18:647–648. doi: 10.1258/095646207781568556. [DOI] [PubMed] [Google Scholar]
- 109.Palmer HM, Young H, Winter A, Dave J. Emergence and spread of azithromycin-resistant Neisseria gonorrhoeae in Scotland. J. Antimicrob. Chemother. 2008;62:490–494. doi: 10.1093/jac/dkn235. [DOI] [PubMed] [Google Scholar]
- 110.Chisholm SA, Dave J, Ison CA. High-level azithromycin resistance occurs in Neisseria gonorrhoeae as a result of a single point mutation in the 23S rRNA genes. Antimicrob. Agents Chemother. 2010;54:3812–3816. doi: 10.1128/AAC.00309-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Starnino S, Stefanelli P, Neisseria gonorrhoeae Italian Study Group I Azithromycin-resistant Neisseria gonorrhoeae strains recently isolated in Italy. J. Antimicrob. Chemother. 2009;63:1200–1204. doi: 10.1093/jac/dkp118. [DOI] [PubMed] [Google Scholar]
- 112.Galarza PG, Abad R, Canigia LF, et al. New mutation in 23S rRNA gene associated with high level of azithromycin resistance in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2010;54:1652–1653. doi: 10.1128/AAC.01506-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Katz AR, Komeya AY, Soge OO, et al. Neisseria gonorrhoeae with high-level resistance to azithromycin: case report of the first isolate identified in the United States. Clin. Infect. Dis. 2012;54:841–843. doi: 10.1093/cid/cir929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Boslego JW, Tramont EC, Takafuji ET, et al. Effect of spectinomycin use on the prevalence of spectinomycin-resistant and penicillinase-producing Neisseria gonorrhoeae. N. Engl. J. Med. 1987;317:272–278. doi: 10.1056/NEJM198707303170504. [DOI] [PubMed] [Google Scholar]
- 115.Discontinuation of spectinomycin Morbidity and Mortality Weekly Report. Apr 7, 2006.
- 116.Brown LB, Krysiak R, Kamanga G, et al. Neisseria gonorrhoeae antimicrobial susceptibility in Lilongwe, Malawi, 2007. Sex. Transm. Dis. 2010;37:169–172. doi: 10.1097/OLQ.0b013e3181bf575c. [DOI] [PubMed] [Google Scholar]
- 117.Ross JD, Lewis DA. Cephalosporin resistant Neisseria gonorrhoeae: time to consider gentamicin? Sex. Transm. Infect. 2012;88:6–8. doi: 10.1136/sextrans-2011-050362. [DOI] [PubMed] [Google Scholar]
- 118.Chisholm SA, Quaye N, Cole MJ, et al. An evaluation of gentamicin susceptibility of Neisseria gonorrhoeae isolates in Europe. J. Antimicrob. Chemother. 2011;66:592–595. doi: 10.1093/jac/dkq476. [DOI] [PubMed] [Google Scholar]
- 119.Dowell D, Kirkcaldy RD. Effectiveness of gentamicin for gonorrhoea treatment: systematic review and meta-analysis. Sex. Transm. Infect. 2012 doi: 10.1136/sextrans-2012-050604. doi:10.1136/sextrans-2012-050604. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 120.Golparian D, Fernandes P, Ohnishi M, Jensen JS, Unemo M. In vitro activity of the new fluoroketolide solithromycin (CEM-101) against a large collection of clinical Neisseria gonorrhoeae isolates and international reference strains including those with various high-level antimicrobial resistance - potential treatment option for gonorrhea? Antimicrob. Agents Chemother. 2012;56:2739–2742. doi: 10.1128/AAC.00036-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Llano-Sotelo B, Dunkle J, Klepacki D, et al. Binding and action of CEM-101, a new fluoroketolide antibiotic that inhibits protein synthesis. Antimicrob. Agents Chemother. 2010;54:4961–4970. doi: 10.1128/AAC.00860-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Still JG, Schranz J, Degenhardt TP, et al. Pharmacokinetics of solithromycin (CEM-101) after single or multiple oral doses and effects of food on single-dose bioavailability in healthy adult subjects. Antimicrob. Agents Chemother. 2011;55:1997–2003. doi: 10.1128/AAC.01429-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Roblin PM, Kohlhoff SA, Parker C, Hammerschlag MR. In vitro activity of CEM-101, a new fluoroketolide antibiotic, against Chlamydia trachomatis and Chlamydia (Chlamydophila) pneumoniae. Antimicrob. Agents Chemother. 2010;54:1358–1359. doi: 10.1128/AAC.01343-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Waites KB, Crabb DM, Duffy LB. Comparative in vitro susceptibilities of human mycoplasmas and ureaplasmas to a new investigational ketolide, CEM-101. Antimicrob. Agents Chemother. 2009;53:2139–2141. doi: 10.1128/AAC.00090-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Unemo M, Golparian D, Limnios A, et al. In vitro activity of ertapenem vs. ceftriaxone against Neisseria gonorrhoeae isolates with highly diverse ceftriaxone MIC values and effects of ceftriaxone resistance determinants – ertapenem for treatment of gonorrhea? Antimicrob. Agents Chemother. 2012;56:3603–3609. doi: 10.1128/AAC.00326-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Burkhardt O, Derendorf H, Welte T. Ertapenem: the new carbapenem 5 years after first FDA licensing for clinical practice. Expert Opin. Pharmacother. 2007;8:237–256. doi: 10.1517/14656566.8.2.237. [DOI] [PubMed] [Google Scholar]
- 127.Congeni BL. Ertapenem. Expert Opin. Pharmacother. 2010;11:669–672. doi: 10.1517/14656561003631397. [DOI] [PubMed] [Google Scholar]
- 128.Deshpande LM, Gales AC, Jones RN. GAR-936 (9-t-butylglycylamidominocycline) susceptibility test development for streptococci, Haemophilus influenzae and Neisseria gonorrhoeae: preliminary guidelines and interpretive criteria. Int. J. Antimicrob. Agents. 2001;18:29–35. doi: 10.1016/s0924-8579(01)00355-7. [DOI] [PubMed] [Google Scholar]
- 129.Falagas ME, Karageorgopoulos DE, Dimopoulos G. Clinical significance of the pharmacokinetic and pharmacodynamic characteristics of tigecycline. Curr. Drug Metab. 2009;10:13–21. doi: 10.2174/138920009787048356. [DOI] [PubMed] [Google Scholar]
- 130.Nix DE, Matthias KR. Should tigecycline be considered for urinary tract infections? A pharmacokinetic re-evaluation. J. Antimicrob. Chemother. 2010;65:1311–1312. doi: 10.1093/jac/dkq116. [DOI] [PubMed] [Google Scholar]
- 131.Alexander BT, Marschall J, Tibbetts RJ, Neuner EA, Dunne WM, Jr, Ritchie DJ. Treatment and clinical outcomes of urinary tract infections caused by KPC-producing Enterobacteriaceae in a retrospective cohort. Clin. Ther. 2012;34:1314–1323. doi: 10.1016/j.clinthera.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Novak R. Are pleuromutilin antibiotics finally fit for human use? Ann. NY Acad. Sci. 2011;1241:71–81. doi: 10.1111/j.1749-6632.2011.06219.x. [DOI] [PubMed] [Google Scholar]
- 133.Novak R, Shlaes DM. The pleuromutilin antibiotics: a new class for human use. Curr. Opin. Investig. Drugs. 2010;11:182–191. [PubMed] [Google Scholar]
- 134.Bradbury BJ, Pucci MJ. Recent advances in bacterial topoisomerase inhibitors. Curr. Opin. Pharmacol. 2008;8:574–581. doi: 10.1016/j.coph.2008.04.009. [DOI] [PubMed] [Google Scholar]
- 135.Then RL, Sahl HG. Anti-infective strategies of the future: is there room for species-specific antibacterial agents? Curr. Pharm. Des. 2010;16:555–566. doi: 10.2174/138161210790361407. [DOI] [PubMed] [Google Scholar]
- 136.Golparian D, Shafer M, Ohnishi M, Unemo M. Inactivation of the MtrCDE, MacAB, and NorM efflux pumps in Neisseria gonorrhoeae strains with clinical resistance to extended-spectrum cephalosporins make them susceptible to several antimicrobials.. Presented at: 18th International Pathogenic Neisseria Conference; Wurzburg, Germany. 9–14 September 2012. [Google Scholar]
- 137.Swanson S, Lee C-J, Liang X, Toone E, Zhou P, Nicholas R. LpxC inhibitors as novel therapeutics for treatment of antibiotic-resistant Neisseria gonorrhoeae.. Presented at: 18th International Pathogenic Neisseria Conference; Wurzburg, Germany. 9–14 September 2012. [Google Scholar]
- 138.Bucki R, Leszczyńska K, Namiot A, Sokołowski W. Cathelicidin LL-37: a multitask antimicrobial peptide. Arch. Immunol. Ther. Exp. (Warsz.) 2010;58:15–25. doi: 10.1007/s00005-009-0057-2. [DOI] [PubMed] [Google Scholar]
- 139.Wetzler LM. The development of a gonococcal vaccine: fact or fiction? Int. J. STD AIDS. 2001;12(Suppl. 2):12. [Google Scholar]
- 140.Zhu W, Chen CJ, Thomas CE, Anderson JE, Jerse AE, Sparling PF. Vaccines for gonorrhea: can we rise to the challenge. Front. Microbiol. 2011;2:124. doi: 10.3389/fmicb.2011.00124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Hadad R, Jacobsson S, Pizza M, et al. Novel meningococcal 4CMenB vaccine antigens – prevalence and polymorphisms of the encoding genes in Neisseria gonorrhoeae. APMIS. 2012;120:750–760. doi: 10.1111/j.1600-0463.2012.02903.x. [DOI] [PubMed] [Google Scholar]
- 142.Hobbs MM, Sparling PF, Cohen MS, Shafer WM, Deal CD, Jerse AE. Experimental gonococcal infection in male volunteers: cumulative experience with Neisseria gonorrhoeae strains FA1090 and MS11mkC. Front. Microbiol. 2011;2:123. doi: 10.3389/fmicb.2011.00123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Guiliani MM, Adu-Bobie J, Comanducci M, et al. A universal vaccine for serogroup B meningococcus. Proc. Natl Acad. Sci. USA. 2006;103:10834–10839. doi: 10.1073/pnas.0603940103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Borges O, Lebre F, Bento D, Borchard G, Junginger HE. Mucosal vaccines: recent progress in understanding the natural barriers. Pharm. Res. 2010;27:211–223. doi: 10.1007/s11095-009-0011-3. [DOI] [PubMed] [Google Scholar]
- 145.Goire N, Sloots TP, Nissen MD, Whiley DM. Protocol for the molecular detection of antibiotic resistance mechanisms in Neisseria gonorrhoeae. Methods Mol. Biol. 2012;903:319–328. doi: 10.1007/978-1-61779-937-2_22. [DOI] [PubMed] [Google Scholar]
- 146.Unemo M, Olcén P, Fredlund H, Thulin S. Real-time PCR and subsequent pyrosequencing for screening of penA mosaic alleles and prediction of reduced susceptibility to expanded-spectrum cephalosporins in Neisseria gonorrhoeae. APMIS. 2008;116:1004–1008. doi: 10.1111/j.1600-0463.2008.01062.x. [DOI] [PubMed] [Google Scholar]
- 147.Whiley SM, Bates J, Limnios A, Nissen MD, Tapsall J, Sloots TP. Use of a novel screening PCR indicates presence of Neisseria gonorrhoeae with a mosaic penA gene sequence in Australia. Pathology. 2007;39:445–446. doi: 10.1080/00313020701444515. [DOI] [PubMed] [Google Scholar]
- 148.Ochiai S, Ishiko H, Yasuda M, Deguchi T. Rapid detection of the mosaic structure of the Neisseria gonorrhoeae penA gene, which is associated with decreased susceptibilities to oral cephalosporins. J. Clin. Microbiol. 2008;46:1804–1810. doi: 10.1128/JCM.01800-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Goire N, Ohnishi M, Limnios AE, et al. Enhanced gonococcal antimicrobial surveillance in the era of ceftriaxone resistance: a real-time PCR assay for direct detection of the Neisseria gonorrhoeae H041 strain. J. Antimicrob. Chemother. 2012;67:902–905. doi: 10.1093/jac/dkr549. [DOI] [PubMed] [Google Scholar]
- 150.Unemo M, Dillon JA. Review and international recommendation of methods for typing Neisseria gonorrhoeae isolates and their implications for improved knowledge of gonococcal epidemiology, treatment, and biology. Clin. Microbiol. Rev. 2011;24:447–458. doi: 10.1128/CMR.00040-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Websites
- 201.WHO Global incidence and prevalence of selected curable sexually transmitted infections – 2008. www.who.int/reproductivehealth/ publications/rtis/2008_STI_estimates.pdf (Accessed 6 October 2012) [PubMed]
- 202■■.WHO, Department of Reproductive Health and Research Global action plan to control the spread and impact of antimicrobial resistance in Neisseria gonorrhoeae. www.who.int/reproductivehealth/publications/rtis/9789241503501 (Accessed 6 October 2012) [WHO global action plan describing essential actions to control the spread and impact of gonococcal antimicrobial resistance and possible emergence of untreatable gonorrhea]
- 203.European Centre for Disease Prevention and Control Response plan to control and manage the threat of multidrug-resistant gonorrhoea in Europe. www.ecdc.europa.eu/en/publications/Publications/1206-ECDC-MDR-gonorrhoea-response-plan.pdf (Accessed 6 October 2012)
- 204.Centers for Disease Control and Prevention Cephalosporin-resistant Neisseria gonorrhoeae public health response plan. 2012 www.cdc.gov/std/gonorrhea/default.htm (Accessed 6 October 2012)
- 205.European Centre for Disease Prevention and Control Gonococcal antimicrobial susceptibility surveillance in Europe – 2010. www.ecdc.europa.eu/en/publications/Publications/1206-Gonococcal-AMR.pdf (Accessed 6 October 2012)
- 206.NG-MAST Query global sequence and ST database. www.ng-mast.net (Accessed 26 October 2012)
- 207.WHO Strategies and laboratory methods for strengthening surveillance of sexually transmitted infections. www.who.int/iris/bitstream/10665/75729/1/9789241504478_eng.pdf (Accessed 6 October 2012)