Abstract
Individual differences in maternal behavior are affected by both early life experiences and oxytocin, but little is known about genetic variation in oxytocin genes and its effects on mothering. We examined two polymorphisms in the oxytocin peptide gene OXT (rs2740210 and rs4813627) and one polymorphism in the oxytocin receptor gene OXTR (rs237885) in 187 Caucasian mothers at six months postpartum. For OXT, both rs2740210 and rs4813627 significantly associated with maternal vocalizing to the infant. These polymorphisms also interacted with the quality of care mothers experienced in early life, to predict variation in maternal instrumental care and postpartum depression. However, postpartum depression did not mediate the gene-environment effects of the OXT SNPs on instrumental care. In contrast, the OXTR SNP rs237885 did not associate with maternal behavior, but it did associate with pre-natal (but not post-natal) depression score. The findings illustrate the importance of variation in oxytocin genes, both alone and in interaction with early environment, as predictors of individual differences in human mothering. Furthermore, depression does not appear to have a causal role on the variation we report in instrumental care. This suggests that variation in instrumental care varies in association with a gene-early environment effect regardless of current depressive symptomatology. Finally, our findings highlight the importance of examining multiple dimensions of human maternal behavior in studies of genetic associations.
Introduction
Early life experiences including adversity and stress influence development in many mammals. Early social isolation in rats results in a high proportion of mothers who are erratic, less attentive, and show reduced licking and grooming [1], [2]. Maternally deprived monkeys are more likely to reject nursing attempts by their own infants, they are less likely to ‘retrieve’ a crying infant, and tend to engage in more physical aggression towards their infants [3]. Human mothers who have experienced early adversity also show problematic mothering. They are more likely to be abusive and neglectful or to be less sensitive and responsive to their babies [4]. In all of these species, including humans, negative parenting experiences are transmitted across generations [5]–[7]. However, not all mothers are affected to the same extent. Differences in vulnerability to early experiences are related to protective factors throughout life, including nonparent support in early development, social support networks, and marrying a supportive spouse [8], [9]. Differences in vulnerability may also be influenced by individual differences in genetic profile [10]. Yet the role of genetic variation is largely unexplored in the study of individual differences in mothering.
In the present study, we first explored associations between genetic variation on both the oxytocin peptide-coding gene (OXT) and the oxytocin receptor gene (OXTR), and variations in human maternal care. The oxytocin system is an excellent candidate gene system in the maternal context. Extensive pharmacological, physiological, and behavioral evidence points to a role of oxytocin in mammalian mothering [11]–[15]. Oxytocin promotes maternal behavior in rats and sheep [16], [17] and oxytocin receptor antagonists inhibit the onset of rat maternal behavior [18]. Oxytocin receptor levels correlate with natural variations in maternal licking and grooming in rats [19] and with maternal behavior toward foreign lambs in sheep [20]. In humans, oxytocin is clearly associated with aspects of mother-infant relationships [21]. Plasma oxytocin levels in pregnancy predict attachment to the foetus [22], whereas postpartum serum and salivary oxytocin levels predict maternal behaviors toward infants [23], [24]. To date, two studies show associations between OXTR genotype and observed maternal sensitivity [25] and touching of the infant [26]. In contrast, genetic variants in the OXT gene are unexplored in association with human mothering.
Next, we examined the moderating effects of OXT and OXTR genotypes on the relationships between mothers’ early life experiences and present maternal care. Early experiences correlate with subsequent functioning of the oxytocin system in many mammalian species. Greater maternal licking/grooming in female rat pups predicts greater oxytocin receptor binding in adulthood [19], [27]. Early handling in female prairie voles predicts decreased oxytocin receptor binding in the nucleus accumbens and bed nucleus of the stria terminalis, two regions implicated in the regulation of maternal behavior [28]. Early maternal separation in both rhesus macaques and humans predicts lower adult levels of OXT in the cerebrospinal fluid [29], [30]. Moreover, the influences of early experience on oxytocin function may affect later social [28] or maternal behavior [31]. Therefore, genetic variation in the OXT and/or OXTR genes may moderate the extent to which early life experiences influences oxytocinergic function and associated behavior.
Finally, we explored the possibility that the interactive effect of OXT genotype and early experiences on behavior will be mediated by postpartum maternal mood state. Mothers with postpartum depression are more intrusive and irritable, and respond with less attentiveness and sensitivity to infants [32]–[38]. Depression itself is associated with early adversity and genetic variation [39]. A history of childhood adversity, including parental factors such as abuse, insecure attachment, rejection and lack of warmth [40], is a major risk factor for depression. Among the many factors that moderate and/or mediate the relationship between early adversity and adult depression are the timing of the early adversity [41], the presence of social support during development [42], oxytocin function [43] and OXTR genotypes [44]. Moreover, OXTR genotype is directly related to depression [39].
We explored these three questions in a sample of N = 187 Caucasian mothers during interactions with their six-month old infants. Early life experiences were assessed through questionnaires about abuse, neglect, childhood relationships with a mother’s own parents, and consistency of caregivers across childhood. There were four outcome measures of observed maternal care: orienting away from the infant, infant-directed vocalizing, instrumental care, and overall sensitivity of maternal responses. Firstly, the extent of maternal orienting away from the infant may be a measure of maternal distractibility. Orienting away is associated with both serotonin transporter and dopamine D1 receptor genotypes and is negatively correlated with maternal sensitivity [45], [46]. Secondly, maternal sensitivity, or the overall rating of maternal quality of interactions with the infant, associates with genetic variation in the serotonin transporter, OXTR [25], [26], and the vasopressin receptor gene [47], but not in the dopamine D1 or D2 receptor genes [45], [46]. Thirdly, maternal vocalizations to the infant, relate both to genetic variation in the dopamine D2 receptor and to maternal depression [33], [46]. The previous findings indicate that the maternal phenotypic outcomes we use in the present analysis have shown sensitivity in prior genetic association studies, and also highlight the potential specificity of polymorphisms relating to different dimensions of mothering phenotypes. Finally, instrumental care includes adjusting the infant’s clothing and grooming her face, nose, ears, etc. Maternal grooming is present across many mammalian species and has been related to oxytocin signalling in rats [19], [48] and monkeys [49]. However, in humans, younger and less experienced mothers exhibit more instrumental care (and fewer affectionate behaviors like kissing and caressing) [50]. Furthermore, employed mothers spend more time socially engaging with their infants than performing instrumental care [51]. This suggests that the appropriateness of instrumental care in humans may context specific. The previous studies highlight the need to investigate multiple dimensions of human maternal behavior.
Methods
Participants
Subjects were part of the longitudinal study on Maternal Adversity, Vulnerability, and Neurodevelopment (MAVAN). The study follows two cohorts of mothers and their infants: one in Montreal, QC, and another in Hamilton, ON, Canada. Behavioral data were available for the Hamilton cohort of mothers; the present study examines this cohort. Ethnic descent in this sample was mostly Caucasian (90%), with 3% mixed ethnicity, 2% African, 1.5% Hispanic, and 1% East Indian; the remainder were unspecified. This ethnic distribution is typical of the greater Hamilton region. Allele frequency distributions can differ across ethnic groups [52] and heterogeneous ancestry samples can reduce power [53]. Thus we examined the 187 Caucasian mothers in the Hamilton cohort. Subjects were ages 18–45 and recruited in their second trimester of pregnancy (weeks 12 to 24) from referrals to the St. Joseph’s Health Center (SJHC) Women’s Health Concerns Clinic and SJHC Ultrasound Department, Hamilton, Ontario, Canada. Two hundred and fifty-five mothers were originally enrolled. Fifty-one subjects were excluded due to: attrition (n = 28), premature delivery (n = 18), stillbirth or termination (n = 3), or involvement of the Children’s Aid Society (n = 2). A Chi-square test with Yates’ continuity correction revealed that the mothers who were lost to attrition did not differ significantly from the mothers who participated in the study on the level of intake adversity (including past or present psychiatric disturbances, early life abuse or neglect, or significant stressful life events) (χ2 (1, N = 249) = 0.57, p = 0.45). Most subjects reported having a partner (94%). Mean (±SD) maternal age was 31.2 (±4.9); mean maternal education was 4.81 (±2.3) on a scale of zero to ten, where zero is ‘not completed high-school’, and ten is ‘post-graduate degree’.
Procedure
Subjects signed written consent to participate in the MAVAN project. Ethics approval for this study was obtained from the ethics review boards at the University of Toronto, Toronto, ON; and St Joseph’s Healthcare, Hamilton, ON. We assessed mothers and their children through questionnaires, diagnostic tools, and behavioral tasks, during 20 home and lab visits starting at weeks 12 to 24 of pregnancy and continuing until 72 months postpartum. Participants received $25 compensation after each visit.
Video-recorded mother-infant interaction
We assessed maternal behavior and maternal sensitivity by video-coding 30-minute in-home mother-infant recordings at six months postpartum. During the first 20 minutes mothers were free to interact with their infants as normal, but without feeding or changing the infants. In the last ten minutes mothers completed self-report questionnaires in the presence of their infants, to assess responsiveness during maternal divided attention.
One hundred and fifty-eight of the 187 Caucasian-sample mothers agreed to be recorded. These mothers did not differ significantly from mothers who did not participate in the video recording with respect to having prior children (parity), prenatal household income, maternal age, or maternal education.
Maternal and infant behavior
The first 20-minutes of the mother-infant interactions were coded by two raters using the Behavioral Evaluation Strategies and Taxonomies (BEST) coding system (Educational Consulting, Inc. Florida, US) [50]. This analysis generated duration and frequency data for multiple maternal behaviors by use of a computer keyboard with keys indexed for each behavior. We limited the analyses to behaviors that were present in 15% or more of the mothers. Inter-rater reliability was high (r = 0.80, n = 10). We quantified the following maternal behaviors as major outcomes: orienting away from infant (frequency), infant-directed vocalizing (duration), and instrumental care (duration). Orienting away frequency was the number of times a mother’s gaze was directed away from the infant’s head or body. This measure was transformed with the natural log (Ln) transformation due to positive skew. Infant-directed vocalizing was defined as any infant directed speech including motherese, nonsense words, and onomatopoeic sounds. All mothers vocalized during the 20-minute recordings and the measure had a normal distribution. Instrumental care included all maternal grooming and cleaning of the infant’s body and face, as well as adjusting clothes, bibs, soothers, and hats. We corrected the skew in this measure by using the square root transformation. We quantified the following three infant behavioral durations: reaching toward mother, smiling at mother, and crying. These three durations were aggregated to obtain a total duration of infant activity. We later used this measure as a covariate in the regression analyses.
Maternal sensitivity
A single rater coded the full-length 30-minute videos for maternal sensitivity using the Ainsworth Maternal Sensitivity Scales [54] (n = 158), which contain four subscales: Cooperation, Accessibility, Acceptance, and Sensitivity. Mothers receive a rating (1–9) on each of the four subscales, and these are later aggregated to obtain a total Ainsworth Sensitivity Score. Inter-rater reliability was high across the individual subscales (r = 0.83 across all four subscales; n = 10) in comparison with another experienced rater. Raters were blind to subjects’ genotype and early experience status.
Maternal mood
We used the Center for Epidemiological Studies Depression Scale (CES-D; [55]), a 20-item self-assessment tool, to assess maternal mood at 12–24 weeks of pregnancy and again at 6 months postpartum. Each item contains response options scored between 0 and 3, for an overall score ranging from 0 (no depression) to 60 (highest level of depression). The CES-D scale is one of the most widely used depression scales [56] with excellent psychometric properties across clinical and community samples, English speaking and non-English speaking [57], [58].
Early care quality
Early experience was assessed from the following three questionnaires. The Life History Calendar (LHC; [59]) assessed the number of experienced changes in primary caregiver during the subjects' first 18 years of life. The Childhood Trauma Questionnaire (CTQ; [60]), assessed five types of childhood trauma: physical, emotional and sexual abuse; emotional and physical neglect. The Parental Bonding Instrument (PBI; [61]), assessed quality of parenting experienced during the subjects' first 16 years of life. The PBI and CTQ have good psychometric properties [62], [63]. The LHC and CTQ were delivered at 12–24 weeks of pregnancy, whereas the PBI was delivered at 6 months postpartum. The PBI has been shown to exhibit stability over time [63], [64]. We used factor analysis to reduce the multiple dimensions within these three questionnaires to obtain an early care quality factor [45]. The early care quality values range from negative (high early life adversity) to positive (low early life adversity). This dimension was the first principal component (of two) derived from principal component analysis (quartimax rotation) with nine input variables from CTQ, PBI, and LHC, and had an eigenvalue of 4.0. Early experience dimensions that loaded highly on care quality were the following: CTQ physical abuse, CTQ emotional abuse, CTQ physical neglect, CTQ emotional neglect, PBI maternal care and PBI maternal overprotection (intercorrelations between these six dimensions were significant and the absolute value Kendall’s tau coefficients ranged between 0.25 and 0.56, p<0.01 across all cases). Missing values in the derived early care quality dimension were imputed with stochastic regression single imputation in SPSS, where the original nine CTQ, PBI and LHC variables were used as predictors (methods described in [45]).
Buccal cell swabs and genotyping
We collected subjects’ DNA through buccal swabs. DNA extraction and genotyping were carried out at the Center for Addiction and Mental Health (CAMH), Toronto, Ontario, Canada. Because of the relative lack of known functional polymorphisms in the two candidate genes, particularly the OXT peptide gene, we chose the SNPs based on available evidence of associations with mood disorders [65] and antipsychotic treatment response [66]. Furthermore, there is evidence that the OXTR SNP in this study (rs237885) is part of a haplotype block for OXTR, which is significantly associated with pro-social behaviors [67]. Rs2740210 is in the 3′ flanking region of the OXT gene, rs4813627 is 56 kb downstream of the OXT gene, and rs237885 is in the intron region of the OXTR gene. The genotypes of the OXT polymorphisms were determined by the Taqman assay method using the ABI PRISM 7000 (Applied Biosystems, Foster City, California, USA). The IDs for on-demand assays available from ABI were as follows: C__16061225_10 (rs2740210), C___2712196_10 (rs4813627), and C___3290319_1_ (rs237885).
Analysis
Hardy-Weinberg Equilibrium tests and linkage disequilibrium tests were carried out with the genetics package in the open-source statistical framework R (http://cran.r-project.org/). We first tested for main effects of maternal OXT genotype on maternal behavioral outcomes using analysis of variance (ANOVA). OXT SNPs (rs2740210 and rs4813627) were entered in the models both separately and together. The direction of effects was the same, but because of high LD (see above) results are presented for the analyses in which these SNPs are entered together.
Next, we used multivariate regression models to test for genotype×early experience (G×E) effects on 1) maternal behavior and 2) maternal mood (postnatal depression, CES-D score). We first ran full models with genotype, early experience, and their cross-product as predictor variables; maternal age, education, parity (whether this was the mothers’ first child), and postnatal depression score (in the model predicting maternal behavior) as maternal covariates; and infant gender and activity as infant covariates. Model reduction with the F-test enabled the removal of non-significant covariates for the “reduced models”. We present output from both the full and the reduced models in the regression tables.
For the SNP main effect models and the G×E regression models, we used multiple imputation to impute values for missing data in the behavioral outcomes (orienting away from infant, infant-directed vocalizing, instrumental care, maternal sensitivity, and infant activity), with the Hmisc package in R. Imputations were performed with predictive mean matching and Bayesian approximation methods over ten iterations, and results were combined with the fit.mult.impute function, which returns likelihood ratio test chi-squared values as a test of overall fit of the test model compared to the null model (intercept only). This function also returns average beta coefficients. Imputations were performed for the following percentages of missing data in the sample (N = 187): 14% for behavioral outcomes, including maternal and infant behaviors; 9% for parity; 14% for maternal education; and 12% for CES-D Depression scores.
For mediation analyses, we followed steps outlined in Edwards and Lambert [68] to test a full mediation moderation model. We tested a full moderated mediation model [68] by entering our predictors, moderators, and mediators into the following regression equations:
| (1) |
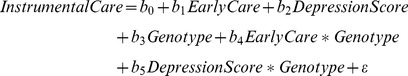 |
(2) |
Where Genotype is OXT rs2740210 (coded as C/C = 0; and A/C or A/A = 1) and Depression Score is maternal CES-D score at 6 months, centered at the standard cut-off score of 16. We used this model to derive path coefficients for a direct effect of early experience (early care quality) on instrumental care, Y; and indirect paths (A) from early care quality to depression (CES-D) and (B) from CES-D to instrumental care. Each of the three paths was modeled to be potentially moderated by genotype. Regression coefficients and bias-corrected boot-strapped confidence intervals (n = 1000) were derived using the boot and boot.ci functions in the boot package in R. For mediation analyses we used the original rather than the imputed data.
Finally, path models using prenatal depression as a mediator were also examined, to strengthen any arguments based on the previous model where the mediator (depression) and parental outcome (instrumental care) occur at the same time-point (6 months postpartum). We present the model with prenatal depression in the supplementary materials.
Results
Sample characteristics and study variables are described in Table S1. The three SNPs were in Hardy-Weinberg equilibrium (Table 1). There was significant linkage disequilibrium (LD) between the OXT SNPs rs2740210 and rs4813627 (D’ = 0.79, χ2 = 86.1, p<0.001). When SNPs are in high LD, the individual effects of SNPs in regression models should be interpreted cautiously. Results from haplotype analyses are presented in Table S2.
Table 1. Minor allele and genotype frequencies in the three OXT/OXTR SNPs.
| % Genotyped | Minor allele frequency | Genotype frequency | |||
| OXT rs4813627 | 87 | A (.50) | G/G (44) | A/G (73) | A/A (45) |
| OXT rs2740210 | 84 | A (.31) | C/C (77) | A/C (63) | A/A (18) |
| OXTR rs237885 | 84 | G (.38) | T/T (43) | G/T (72) | G/G (42) |
There was a significant negative correlation between infant-directed vocalizing and frequency of orienting away from the infant (Spearman’s rho (158) = −0.3, p<0.01), and between frequency of orienting away and sensitivity (Spearman’s rho (158) = −0.19, p = 0.02) (Table 2). Instrumental care correlated negatively with maternal education (Spearman’s rho (139) = −0.17, p = 0.04), and positively with infant activity (Spearman’s rho (158) = 0.22, p<0.01). There was a trend for a positive correlation between instrumental care and CES-D postnatal depression score (Spearman’s rho (145) = 0.16, p = 0.05). Depression score negatively correlated with maternal education (Spearman’s rho (143) = −0.25, p<0.01) (Table 2). ANOVAs revealed no association between genotype and early maternal care quality (p<0.05 for all three SNPs tested).
Table 2. Correlations between maternal socioeconomic, behavioral, and mood variables.
| Orienting Away | Vocalizing | Instrumental Care | Ainsworth Sensitivity | Depression Score (CES-D postnatal) | |
| Orienting away | – | ||||
| Infant-directed Vocalizing | −.30 *** | – | |||
| Instrumental Care | .03 | .06 | – | ||
| Ainsworth Sensitivity | −.19 * | .13 | −.11 | – | |
| Maternal Age | .06 | −.10 | −.10 | .09 | −.15∼ |
| Education | .05 | −.15 | −.17 * | .14 | −.25** |
| Parity | .14 | .00 | −.05 | −.12 | .12 |
| Depression Score (CES-D prenatal) | −.07 | .01 | .11 | −.10 | .60 *** |
| Depression Score (CES-D postnatal) | −.01 | .02 | .16∼ | −.14 | – |
| Infant Gender | −.07 | −.01 | −.02 | −.06 | .06 |
| Infant Activity | −.19 * | .13 | .22** | −.15∼ | .09 |
NOTE: Values are Spearman’s rho coefficients, 2-tailed; ∼ p<0.1;
p<0.05;
p<0.001; N = 128–187.
Main Effects of Genotype
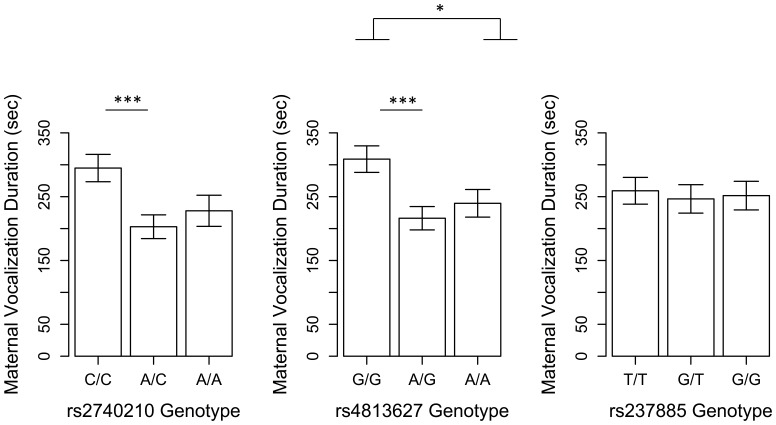
There were significant main effects of both rs2740210 A/C and rs4813627 A/G genotype (reference group was rs2740210 C/C and rs4813627 G/G respectively) on infant directed vocalizing (β = −52.8, t(147) = −2.09, p = 0.04 and β = −56.3, t(147) = −2.06, p = 0.04, respectively). Together rs2740210 and rs4813627 genotypes explained a significant proportion of variance in infant-directed vocalizing (R2 = 0.10, LR χ2 (4, N = 152) = 15.7, p = 0.004) (Fig. 1). Based on the pattern of effects in Figure 1, we grouped genotypes A/C and A/A for rs2740210 and genotypes A/G and A/A for rs4813627. For rs2740210 the A/A genotype group had quite a small sample size (N = 18), providing further justification for this grouping. Haplotype analyses (Table S4) revealed a consistent pattern of results.
Figure 1. Main effects of OXT genotype on infant-directed vocalizing.
Mothers with C/C and G/G genotypes for rs2740210 and rs4813627, respectively, vocalize significantly longer to their infants; values plotted represent non-imputed values of the original sample; regression coefficients and statistics presented here and in the text are based on multiply imputed values for maternal outcomes (see Methods); ***p<0.001; †p<0.1; values are means ± SEM.
Genotype, Experience, and Mothering
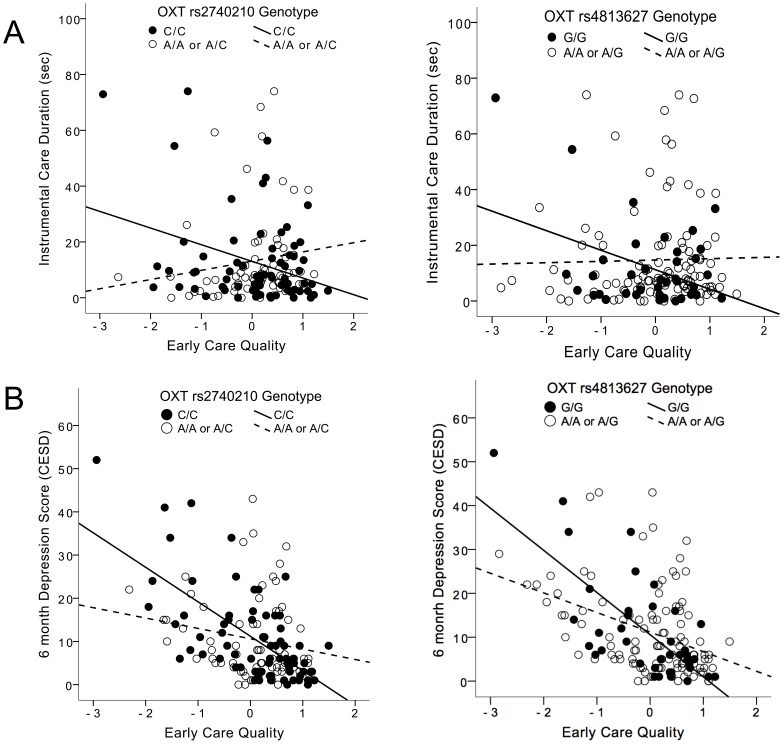
In both the full and reduced models for rs2740210 and rs4813627 there were significant main effects of early care quality on the total duration of maternal instrumental care (Table 3). Moreover, there was a significant genotype×early care quality on the overall duration of maternal instrumental care (Fig. 2, Table 3). For rs2740210 genotype C/C and rs4813627 genotype G/G, respectively, mothers engaged in less instrumental care as reported early care quality increased. For the other genotype groups, this relationship was reversed: mothers had longer total duration of instrumental care with increasing early care quality. Finally, there was a main effect in these models of infant activity, but no other infant or maternal covariate tested.
Table 3. Beta regression coefficients (t-statistics in brackets) for analyses predicting maternal instrumental care duration.
| OXT rs2740210 (N = 158) | OXT rs4813627 (N = 162) | |||||
| Full Model | Reduced Model | Full Model | Reduced Model | |||
| Intercept | 11.57 (0.98) | 2.54 (0.50) | 7.04 (0.60) | 0.13 (0.02) | ||
| Genotype (G) | 0.99 (0.35) | 0.97 (0.34) | 2.72 (0.82) | 3.16 (0.96) | ||
| Early Care Quality (E) | −4.69 (−1.68)∼ | −5.67 (−2.50)* | −5.67 (−1.60) | −6.61 (−2.13)* | ||
| Maternal Age | −0.13 (−0.39) | − | −0.07 (−0.22) | − | ||
| Parity | −3.17 (−1.04) | − | −2.64 (−0.84) | − | ||
| Education | −0.83 (−1.08) | − | −0.91 (−1.14) | − | ||
| Postnatal depression score (CES-D) | 0.14 (0.71) | − | 0.10 (0.54) | − | ||
| Infant Gender | −0.62 (−0.20) | − | 1.10 (0.36) | − | ||
| Infant Activity | 2.68 (2.23)* | 2.84 (2.41)* | 3.09 (2.56)* | 3.11 (2.62)** | ||
| G×E | 8.37 (2.15)* | 8.60 (2.64)** | 7.49 (1.96)* | 7.91 (2.23)* | ||
| R2 (adj.) | 0.10 | 0.08 | 0.08 | 0.06 | ||
| Likelihood Ratio χ2 | 26.79** | 18.02** | 22.01** | 14.70** | ||
| df | (9, 147) | (4, 153) | (9, 151) | (4, 157) | ||
NOTE: For Tables 3 and 4, coefficients represent averages from ten imputations; the ‘full model’ contains several maternal and infant covariates whereas reduced model (obtained after F-test reduction in number of predictor variables) contains only variables that contribute significantly to the model. ∼p<0.10,
p<0.05,
p<0.01.
Figure 2. Interaction between OXT genotypes (rs2740210 and rs4813627) and maternal self-reported early care quality (factor derived from multiple early experience measures, see Methods) on (A) duration of instrumental care during a 20 minute maternal-infant recorded interaction and (B) depression score assessed with the CES-D depressioni scale at 6 months postpartum.
Figures do not take into account covariates used in analyses (Tables 3 and 4).
There were no main effects of early care quality or interactions of care quality with OXT genotypes in relation to maternal sensitivity or orienting away. Because of high linkage disequilibrium between the two OXT SNPs, the results of the regressions using both SNPs in the model should be interpreted cautiously. There were no effects of OXTR genotype (rs237885) on any maternal measure.
Genotype, Experience, and Depression
There were significant genotype×early care quality interactive effects (for both rs2740210 and rs4813627) on maternal depression score (CES-D) at 6 months (Fig. 2, Table 4). Mothers scored lower on depression as the early care quality increased, but the slopes for genotypes C/C (rs2740210) and G/G (rs4813627) were more negative, indicating that early life experience had a stronger moderating effect in this group of mothers than in mothers with other genotypes (A/A or A/C for rs2740210; and A/A or A/G for rs4813627) (Table 4). For rs4813627, there was also a significant main effect of maternal education, so that depression score decreased with increasing education. There were no effects of OXTR genotype (rs237885) on depression levels at 6 months postpartum, but when we used a prenatal depression measure, there were significant associations between rs237885 genotype G/G, which explained a significant proportion of the variance (R2 = 0.07, LR χ2 (2, N = 154) = 11.72, p = 0.003).
Table 4. Regression analyses predicting maternal depression score (CES-D) at 6 months postpartum.
| OXT rs2740210 (N = 158) | OXT rs4813627 (N = 162) | ||||
| Full Model | Reduced Model | Full Model | Reduced Model | ||
| Intercept | 20.85 (2.81)** | 11.23 (9.57)*** | 16.45 (2.19)* | 12.99 (3.52)*** | |
| Genotype (G) | −0.34 (−0.19) | −0.34 (−0.18) | 0.66 (0.37) | 0.63 (0.36) | |
| Early Care Quality (E) | −7.45 (−5.92) | −7.77 (−6.28)*** | −9.35 (−5.58)*** | −9.36 (−5.55)*** | |
| Maternal Age | −0.29 (−1.31) | − | −0.20 (−0.89) | − | |
| Parity | 0.98 (0.52) | − | 1.65 (0.92) | − | |
| Education | −0.37 (−0.97) | − | −0.70 (−1.82)∼ | −0.86 (−2.54)* | |
| Infant Gender | 1.30 (0.80) | − | 1.77 (1.09) | − | |
| Infant Activity | 0.09 (0.12) | – | 0.55 (0.76) | – | |
| G×E | 5.33 (2.56)* | 5.89 (2.91)** | 5.83 (2.97)** | 5.95 (2.98)** | |
| R2 (adj.) | 0.23 | 0.21 | 0.26 | 0.25 | |
| F | 48.98*** | 39.52*** | 56.82*** | 51.05*** | |
| Df | (8, 148) | (3, 154) | (8, 152) | (5, 156) | |
NOTE: Table coefficients represent averages from ten imputations. ∼ p<0.10,
p<0.05,
p<0.01,
p<0.001.
Mediation/Moderation Model
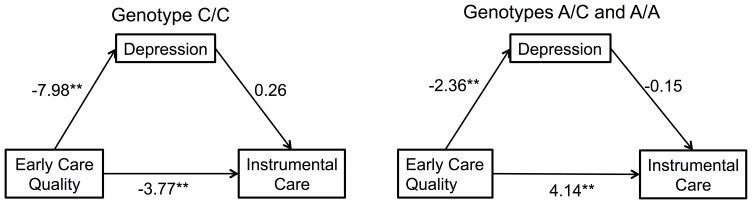
In mediated moderation analysis, we compared rs2740210 genotypes C/C with genotypes A/C and A/A, and found direct effects and indirect effects in the model from early experience to instrumental care. For both rs2740210 genotypes, there was a significant negative association between early care quality and postnatal depression (path coefficients are −7.98 (C/C) and −2.40 (A/C; A/A)) (Fig. 3; Table 5 and Table 6). The direct path from early care quality to instrumental care was significant and negative for C/C mothers (−3.77); and significant and positive for A/C or A/A mothers (4.30). However, the path from depression to instrumental care was not significant for either C/C or A/C and A/A genotypes of rs2740210.
Figure 3. Path models depicting relationships between early experience, postnatal depression, and maternal instrumental care in two genotypes of rs2740210: C/C and A/C+A/A using coefficients from Table 6 .
Significance values are based on bias-corrected bootstrap adjusted confidence intervals. Postnatal depression is not a significant mediator of the early care quality associations with instrumental care, for either genotype.
Table 5. Coefficient estimates from path models.
| Equation 1a | Equation 2 b | |||||||||
| Early Care | Genotypec | Early Care*Genotype | R2 | Early Care | Depressiond | Genotype | Early Care*Genotype | Depression*Genotype | R2 | |
| Coefficients | −7.98** | −0.72 | 5.62** | 0.24*** | −3.77* | 0.26 | −0.26 | 7.91** | −0.11 | 0.04∼ |
| CI 2.5% | −10.10 | −3.91 | 6.40 | −11.22 | −0.20 | −10.05 | 6.65 | −1.11 | ||
| CI 97.5% | −7.81 | 1.59 | 6.45 | −0.59 | 1.14 | 7.00 | 13.56 | 0.54 | ||
NOTE: acoefficients were derived using Equation 1 (Methods);
Coefficients were derived using Equation 2 (Methods);
OXTrs2740210 genotype (grouped as genotype C/C versus genotype A/C and A/A);
Depression measured postnatally, with the CES-D scale. CI: bias-corrected confidence intervals which are Bootstrap adjusted values;∼p<0.1,
p<0.05,
p<0.01.
Table 6. Simple effects of path models (using postnatal depression score as a mediator).
| Stage | Effect | ||||
| rs2740210 Genotype | First | Second | Direct | Indirect | Total |
| C/C | −7.98** | 0.26 | −3.77** | −2.06* | −5.85** |
| A/C and A/A | −2.36** | −0.15 | 4.14** | −0.35 | 3.79** |
| Differences | 5.62** | −0.11 | 7.91** | 1.72 | 9.63** |
Note: N = 129–140. Simple effects computed using equation 25 in Edwards and Lambert (2007) using coefficients estimates from Table 5. Zs = 0 for C/C and 1 for A/C and A/A genotypes (rs2740210). Differences in simple effects were calculated by subtracting the A/C+A/A effects from the C/C simple effects. Significance values are based on bias-corrected boot-strap adjusted confidence intervals.
p<0.05,
p<0.01.
Models using prenatal depression revealed a similar pattern of effects for the C/C genotype mothers (significant first stage effect (early care depression) and direct effect (early care instrumental care)), and a similar direction (though not significant) of effects for the alternate genotype (Tables S3, S4, and Fig. S1). The differences between the effects in the two genotypes were significant. Thus the overall prenatal depression model had the same pattern of effects as the postnatal model.
Discussion
There is accumulating evidence for the role of oxytocin in the regulation of human mothering [21]. We examined polymorphic variation on two oxytocin genes – one which codes for the oxytocin peptide (OXT), and one which codes for the oxytocin receptor (OXTR) – and found that polymorphisms in OXT (but not OXTR) associate with both infant-directed vocalizing and maternal instrumental care.
There was a main effect of genotype on vocalizing for both OXT SNPs (rs2740210 and rs4813627). Vocalizing to infants is a universal behavior of mothers that includes infant-directed speech, or “motherese”, characterised by exaggerated prosodic cues and is present in all studied languages [69]. The distinctive vocal characteristics of infant-directed speech are believed to act as cues for the infant, and to aid communication and language learning and promote closeness [70], [71]. Maternal speech can be further subdivided, for instance into affect-salient and information-salient content [70]. In the present research, we opted for quantifying the entire duration of the vocalization. In this sense, though a large component of these vocalizations are “motherese”, our measure of infant-directed vocalization is a measure of maternal engagement with the infant.
We previously showed that genetic variation in the dopamine D1 and D2 receptor genes (DRD1 and DRD2) associates with individual variation in human maternal care, including infant-directed vocalizing [46]. Specifically, there was a notable association between DRD2 genotypes and haplotypes and maternal infant-directed vocalizing. The link between genetic variation in both the OXT and DRD2 genes and individual differences in the duration of maternal vocalizing to infants is plausible. Oxytocin neurons express DRD2-like receptors in the rat brain [72]. There is also evidence for a functional link between dopamine and oxytocin in the rat mesolimbic dopamine pathway, which is important not only for reward-processing but also for the expression of maternal behavior [73]. Oxytocin infusion into the ventral tegmental area (VTA) enhances the dopamine signal in the nucleus accumbens [74]. Moreover, antagonist oxytocin infusion into the VTA disrupts rat maternal care [18]. Therefore genetic variants on these genes may have similar or even interactive effects on behavioral outcomes. Our sample size precluded us from examining gene-gene interactions between oxytocin and dopamine pathway genes, but this should be an important future step for studies in mothering. The many complex neural pathways that regulate mothering do so by acting in concert; therefore multiple genes and genetic polymorphisms underlying these pathways are likely to influence mothering. Indeed, the prior evidence relating genotypic variation in the serotonin transporter gene [25], [45], the vasopressin peptide gene [47], the dopamine D1 and D2 receptor genes [46], [75], and the oxytocin receptor gene [25], to differences in mothering suggest opportunities for studying gene-gene interactions in individual variation in maternal care, given large enough samples.
In the present report we found that maternal OXT genotype also associates with maternal instrumental care, in interaction with early maternal life experience. Mothers with genotypes C/C or G/G (for rs2740210 and rs4813627, respectively) displayed shorter overall instrumental care duration with increasing early care quality; for mothers with the other two genotypes for these SNPs, instrumental care duration increased with increasing early care quality (Fig. 2A).
In non-human animals, grooming is an essential caregiving behavior. Maternal rat dams spend considerable amounts of time in the early postpartum licking/grooming their pups and variations in the time spent licking/grooming predicts a number of developmental and behavioral outcomes in the growing and adult offspring [27], [76], [77]. In humans, mothers groom as part of instrumental or caretaking behaviors. Younger mothers engage in more instrumental caretaking behaviors than older mothers [50]. Instrumental behavior did not correlate with maternal age in our sample, but negatively correlated with maternal education. The more educated the mother, the less time she spent performing instrumental tasks like grooming, adjusting, and cleaning the infant. Indeed, research supports the notion that maternal education and employment status influence how mothers allocate their time while interacting with their infants. For instance, employed women engage in significantly less instrumental care, and significantly more socialization with their infants than non-employed women [51]. However, instrumental care is positively correlated with maternal age and education in this study [51], whereas education was negatively related to instrumental care in our sample. These differences may arise from the inherent differences in instrumental care between the two studies. In our study, free-play interactions were designed to minimize instrumental care – mothers were instructed to refrain from changing diapers, breast- or bottle-feeding, bathing, or other time-consuming instrumental care behaviors. In contrast, in the Huston & Rosenkrantz Aronson study [51], mothers filled out questionnaire reports about the total duration of caretaking activities over a full two days.
There was also a notable main effect of infant activity on instrumental care: the longer the infants cried, smiled, or reached toward their mothers, the longer the duration of maternal instrumental care, though this relationship may be bi-directional (Table 3). In other words, infants may cry, reach, or smile more in response to maternal caretaking behaviors. Conversely, mothers may engage in instrumental behavior in response to infant fussiness or signals of attention-seeking. There is a potential role for infant genotype on this dyadic interaction, which should be studied in future. Observed instrumental behaviors during a free-play interaction therefore likely represent an important aspect of maternal strategizing; for some mothers instrumental care may be more prominent than for others, and we have shown that the levels of maternal instrumental care is predicted by the interactive effects of genotype and early life experiences.
Instrumental caregiving behaviors may also be part of a more generally intrusive (over-stimulating, controlling) style of mothering sometimes associated with depressed mothers [32]. Indeed, we found a trend for a negative correlation between depression score and instrumental care in our cohort of mothers (Table 2). We subsequently explored the possibility that depression mediates the genetic associations with mothering. Moreover, early stress influences oxytocin signalling in adulthood. We thus tested the interaction between OXT genotype and early care quality, and found a highly significant association on CES-D scores at 6 months postpartum (Table 4). However, when we tested these in a mediation/moderation model, depression (both prenatal and postnatal) showed no significant association with maternal instrumental care, regardless of maternal genotype. Although OXT genotype×early care quality predicted both maternal instrumental care and CES-D score, these appear to be separate effects rather than causally linked outcomes. There is support for this in the regression models, which show a significant main effect of infant activity on maternal instrumental care, but not on maternal postnatal depression.
In addition to being intrusive, depressed mothers are sometimes withdrawn - vocalizing less with their infants and looking away more often [78]. Yet there was no correlation between depression score and maternal infant-directed vocalization in our sample (Table 2). However, ours was not a clinically depressed sample and the majority of mothers scored below the clinical cut-off, 16. Moreover, mean CES-D score in the prenatal period was higher than in the postnatal period, possibly because mothers who presented with depression or depressive symptoms prenatally were seen by a psychiatrist and received behavioral or pharmacological therapy. It would be interesting to replicate this study in a clinically depressed sample of mothers. In such a sample, greater depressive symptoms may more directly influence mothering behavior, and may be a significant mediator of OXT genotype×early life experience influences on mothering.
Interestingly, the genotype groups that showed the longest duration of overall infant-directed vocalizing (Figure 1) also showed greater moderation by early environment on depression score. For rs2740210 and rs4813627, genotypes C/C and G/G, respectively, showed steeper negative slopes when CES-D scores were regressed onto early care quality (Figure 2). Variations in the quality of early care among these mothers had a greater effect on postpartum depression scores. Very similar findings are reported for the OXTR polymorphism rs2254298, which associates with depression and anxiety in adults [79] and, in the face of early adversity, associates with anxiety and depression in adolescent girls [44]. It is unknown if the two non-coding OXT SNPs in our study exhibit gene-gene interactions with the OXTR rs2254298 SNP in the above studies, or if they have similar or overlapping functions. There is an indication that rs2254298 influences amygdala volume, but this effect may be ethnicity-dependent as it is found in Asian, but not Caucasian populations [80], [81].
Another OXTR polymorphism, rs53576, has previously been shown to associate with maternal sensitivity [25] and depression [39]. We did not have available genotypes for this SNP, but did examine the rs237885 polymorphism, which also lies on the OXTR gene. We found a notable absence of associations between this SNP and any maternal outcome tested. The previously studied rs53576 is a silent (AG) mutation and along with the SNP we assessed, rs237885, lies in the intronic region of the OXTR gene, with no known effects on expression or protein function. Yet because rs53576 has effects in other association studies, future research should examine this SNP in association with early adversity for effects on maternal behavior.
None of the three SNPs we explored showed associations with maternal sensitivity, a global measure of maternal quality of care; or with orienting away, a measure of maternal responsiveness potentially related to attention. We have previously reported that orienting away associates with dopamine D1 receptor genotype and with genotype at the serotonin transporter-linked polymorphic region (5HTTLPR) [45], [46], and have argued that this measure reflects maternal inattention or distractibility. The lack of association between OXT SNPs and orienting away is not surprising. Oxytocin seems to have functions centered on sociality and affect, and therefore attention-related dimensions of mothering are unlikely to be influenced by genetic variation in the OXT gene.
One limitation to our study is the use of a retrospective measure of early care quality. However, the subcomponents of our measure were the Childhood Trauma Questionnaire (CTQ) and the Parental Bonding Instrument (PBI), both of which have long-term stability. The PBI has been examined in clinical samples where depressed individuals improved over time; in mothers who were followed from the early postpartum for 30 months [64]; and in a sample where scores were taken 20 years apart [63]. These studies indicate stability of scores across time, regardless of depression or postpartum status. Furthermore, there is a high correlation between CTQ (assessed prenatally) and the PBI (assessed at 6 months postpartum). Another limitation is the relatively small sample size for a genetics study and the lack of a replication sample. After exclusion of the non-Caucasian mothers, we had 187 remaining mothers. Quantification of complex phenotypes such as maternal behavior is time-intensive and this size of sample is quite large for psychological studies of this kind.
The present report is the first, to our knowledge, to examine genetic variation in the oxytocin peptide gene OXT in relation to maternal behavior. We examined two polymorphisms in the OXT gene (rs2740210 and rs4813627), and both showed a main effect on maternal vocalizing to the infant; as well as a significant interactive effect with early life experience on maternal instrumental care of the infant. The significant associations for type (i.e. vocalizing, instrumental care) but not quality (i.e. maternal sensitivity) of maternal care, highlights the importance of exploring multiple levels of behavioral phenotypes in genetic association analyses. Finally, it is important to keep in mind that mother-child interactions may change over time. Thus, they represent a dynamic relationship, which at different times may exhibit different influences from hereditary and non-hereditary factors. Genetic studies should begin to delve into the temporal stability of association between genetic variation and multiple outcomes over time.
Supporting Information
Path models depicting relationships between early experience, prenatal depression, and maternal instrumental care in two genotypes of rs2740210: C/C and A/C using coefficients from Table 6. Significance values are based on bias-corrected bootstrap adjusted confidence intervals. Prenatal depression is not a significant mediator of the early care quality associations with instrumental care, for either genotype.
(TIFF)
Sample characteristics and study variables.
(DOCX)
OXT haplotypes and regression models with these haplotypes.
(DOCX)
Coefficient estimates of path models (using prenatal CES-D).
(DOCX)
Simple effects of path models (using prenatal prenatal depression score as a mediator).
(DOCX)
Acknowledgments
We would like to thank Amber Rieder, Carmen MacPherson, and their team for their lead in the Hamilton project; and Natalie Freeman, Subi Shehaan, Maria Tampakeras, Sajid Shaikh, Clement Zai, and their team at the Centre for Addiction and Mental Health for their assistance with genotyping and genetic analyses.
Funding Statement
This study was funded by a Canadian Institutes of Health Research (CIHR, http://www.cihr-irsc.gc.ca/e/193.html) trajectory grant for the MAVAN, principal investigators (PIs) M.J.M. and S.G.M., by CIHR grants to A.S.F. and J.L.K., by a University of Toronto Connaught Global Challenge Grant to A.S.F. and M.B.S., by a Canadian Institutes for Advanced Research Grant to M.J.M. and M.B.S., and by a Natural Sciences and Engineering Research Council Canada (NSERC; http://www.nserc-crsng.gc.ca/index_eng.asp) PhD scholarship to V.M.-S. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Rees SL, Akbari E, Steiner M, Fleming AS (2008) Effects of early deprivation and maternal separation on pup-directed behavior and HPA axis measures in the juvenile female rat. Dev Psychobiol 50: 315–321. [DOI] [PubMed] [Google Scholar]
- 2. Barrett J, Fleming AS (2011) All mothers are not created equal: neural and psychobiological perspectives on mothering and the importance of individual differences. J Child Psychol Psychiatry 52: 368–397 doi:10.1111/j.1469-7610.2010.02306.x. [DOI] [PubMed] [Google Scholar]
- 3. Maestripieri D (2005) Early experience affects the intergenerational transmission of infant abuse in rhesus monkeys. Proc Natl Acad Sci U S A 102: 9726–9729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Moehler E, Biringen Z, Poustka L (2007) Emotional availability in a sample of mothers with a history of abuse. Am J Orthopsychiatry 77: 624–628 doi:10.1037/0002-9432.77.4.624. [DOI] [PubMed] [Google Scholar]
- 5. Champagne F, Meaney MJ (2001) Like mother, like daughter: evidence for non-genomic transmission of parental behavior and stress responsivity. Prog Brain Res 133: 287–302. [DOI] [PubMed] [Google Scholar]
- 6. Gonzalez A, Lovic V, Ward GR, Wainwright PE, Fleming AS (2001) Intergenerational effects of complete maternal deprivation and replacement stimulation on maternal behavior and emotionality in female rats. Dev Psychobiol 38: 11–32. [DOI] [PubMed] [Google Scholar]
- 7. Belsky J, Conger RD, Capaldi DM (2009) The intergenerational transmission of parenting: introduction to the special section. Dev Psychol 45: 1201–1204 doi:10.1037/a0016245. [DOI] [PubMed] [Google Scholar]
- 8.Egeland B, Bosquet M, Levy A (2002) Continuities and discontinuities in the intergenerational transmission of child maltreatment: Implications for breaking the cycle of abuse. In: K Browne, H Hanks, P. Stratton CH, editor. The prediction and prevention of child abuse: A handbook. New York, NY: John Wiley & Sons. 217–232. [Google Scholar]
- 9. Langeland W, Dijkstra S (1995) Breaking the intergenerational transmission of child abuse: Beyond the mother-child relationship. Child abuse review 4: 4–13 doi:10.1002/car.2380040104. [Google Scholar]
- 10. Belsky J, Bakermans-Kranenburg MJ, Van IJzendoorn MH (2007) For Better and For Worse: Differential Susceptibility to Environmental Influences. Curr Dir Psychol Sci 16: 300–304 doi:10.1111/j.1467-8721.2007.00525.x. [Google Scholar]
- 11.Numan M, Fleming AS, Levy F (2006) Maternal behavior. In: Neill JD, editor. Knobil & Neill’s Physiology of Reproduction. New York: Elsevier Inc. 1921–1993. [Google Scholar]
- 12. Insel TR, Shapiro LE (1992) Oxytocin receptors and maternal behavior. Annals Of The New York Academy Of Sciences 652: 122–141. [DOI] [PubMed] [Google Scholar]
- 13. Febo M, Numan M, Ferris CF (2005) Functional magnetic resonance imaging shows oxytocin activates brain regions associated with mother-pup bonding during suckling. J Neurosci 25: 11637–11644 doi:10.1523/JNEUROSCI.3604-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Pedersen CA, Vadlamudi S V, Boccia ML, Amico JA (2006) Maternal behavior deficits in nulliparous oxytocin knockout mice. Genes Brain Behav 5: 274–281 doi:10.1111/j.1601-183X.2005.00162.x. [DOI] [PubMed] [Google Scholar]
- 15. Dwyer CM (2008) Individual variation in the expression of maternal behaviour: a review of the neuroendocrine mechanisms in the sheep. J Neuroendocrinol 20: 526–534 doi:10.1111/j.1365-2826.2008.01657.x. [DOI] [PubMed] [Google Scholar]
- 16. Keverne EB (1996) Psychopharmacology of maternal behaviour. J Psychopharmacol 10: 16–22 doi:10.1177/026988119601000104. [DOI] [PubMed] [Google Scholar]
- 17. Kendrick KM, Keverne EB, Baldwin BA (1986) Intracerebroventricular oxytocin stimulates maternal-behavior in the sheep. Neuroendocrinology 46: 56–61. [DOI] [PubMed] [Google Scholar]
- 18. Pedersen CA, Caldwell JD, Walker C, Ayers G, Mason GA (1994) Oxytocin activates the postpartum onset of rat maternal behavior in the ventral tegmental and medial preoptic areas. Behav Neurosci 108: 1163. [DOI] [PubMed] [Google Scholar]
- 19. Francis DD, Champagne F, Meaney MJ (2000) Variations in maternal behaviour are associated with differences in oxytocin receptor levels in the rat. J Neuroendocrinol 12: 1145–1148. [DOI] [PubMed] [Google Scholar]
- 20. Keverne EB, Kendrick KM, Poindron P (1988) Oxytocin and maternal-behavior. Psychopharmacology (Berl) 96: S30. [Google Scholar]
- 21. Galbally M, Lewis AJ, Ijzendoorn M Van, Permezel M, Van Ijzendoorn MH (2011) The role of oxytocin in mother-infant relations: a systematic review of human studies. Harv Rev Psychiatry 19: 1–14 doi:10.3109/10673229.2011.549771. [DOI] [PubMed] [Google Scholar]
- 22. Levine A, Zagoory-Sharon O, Feldman R, Weller A (2007) Oxytocin during pregnancy and early postpartum: individual patterns and maternal-fetal attachment. Peptides 28: 1162–1169 doi:10.1016/j.peptides.2007.04.016. [DOI] [PubMed] [Google Scholar]
- 23. Gordon I, Zagoory-Sharon O, Leckman JF, Feldman R (2010) Oxytocin, cortisol, and triadic family interactions. Physiol Behav 101: 679–684 doi:10.1016/j.physbeh.2010.08.008. [DOI] [PubMed] [Google Scholar]
- 24. Feldman R, Gordon I, Schneiderman I, Weisman O, Zagoory-Sharon O (2010) Natural variations in maternal and paternal care are associated with systematic changes in oxytocin following parent-infant contact. Psychoneuroendocrinology 35: 1133–1141 doi:10.1016/j.psyneuen.2010.01.013. [DOI] [PubMed] [Google Scholar]
- 25. Bakermans-Kranenburg MJ, Van Ijzendoorn MH (2008) Oxytocin receptor (OXTR) and serotonin transporter (5-HTT) genes associated with observed parenting. Soc Cogn Affect Neurosci 3: 128–134 doi:10.1093/scan/nsn004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Feldman R, Zagoory-Sharon O, Weisman O, Schneiderman I, Gordon I, et al. (2012) Sensitive parenting is associated with plasma oxytocin and polymorphisms in the OXTR and CD38 genes. Biol Psychiatry 72: 175–181 doi:10.1016/j.biopsych.2011.12.025. [DOI] [PubMed] [Google Scholar]
- 27. Champagne F, Diorio J, Sharma S, Meaney MJ (2001) Naturally occurring variations in maternal behavior in the rat are associated with differences in estrogen-inducible central oxytocin receptors. Proc Natl Acad Sci U S A 98: 12736–12741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Bales KL, Boone E, Epperson P, Hoffman G, Carter CS (2011) Are behavioral effects of early experience mediated by oxytocin? Frontiers in psychiatry 2: 24 doi:10.3389/fpsyt.2011.00024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Winslow JT, Noble PL, Lyons CK, Sterk SM, Insel TR (2003) Rearing effects on cerebrospinal fluid oxytocin concentration and social buffering in rhesus monkeys. Neuropsychopharmacology 28: 910–918 doi:10.1038/sj.npp.1300128. [DOI] [PubMed] [Google Scholar]
- 30. Heim C, Young LJ, Newport DJ, Mletzko T, Miller a H, et al. (2009) Lower CSF oxytocin concentrations in women with a history of childhood abuse. Mol Psychiatry 14: 954–958 doi:10.1038/mp.2008.112. [DOI] [PubMed] [Google Scholar]
- 31.Bakermans-Kranenburg MJ, Van IJzendoorn MH, Riem MME, Tops M, Alink LRA (2011) Oxytocin decreases handgrip force in reaction to infant crying in females without harsh parenting experiences. Soc Cogn Affect Neurosci. doi:10.1093/scan/nsr067. [DOI] [PMC free article] [PubMed]
- 32. Field T, Hernandezreif M, Diego M (2006) Intrusive and withdrawn depressed mothers and their infants. Dev Rev 26: 15–30 doi:10.1016/j.dr.2005.04.001. [Google Scholar]
- 33. Fleming AS, Ruble DN, Flett GL, Shaul DL (1988) Postpartum adjustment in first-time mothers: Relations between mood, maternal attitudes, and mother-infant interactions. Dev Psychol 24: 71. [Google Scholar]
- 34. Chung EK, McCollum KF, Elo IT, Lee HJ, Culhane JF (2004) Maternal Depressive Symptoms and Infant Health Practices Among Low-Income Women. Pediatrics 113: e523–e529 doi:10.1542/peds.113.6.e523. [DOI] [PubMed] [Google Scholar]
- 35. Milgrom J, Westley D, Gemmill A (2004) The mediating role of maternal responsiveness in some longer term effects of postnatal depression on infant development. Infant Behav Dev 27: 443–454 doi:10.1016/j.infbeh.2004.03.003. [Google Scholar]
- 36. Herrera E, Reissland N, Shepherd J (2004) Maternal touch and maternal child-directed speech: effects of depressed mood in the postnatal period. J Affect Disord 81: 29–39 doi:10.1016/j.jad.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 37. Feldman R, Eidelman AI (2007) Maternal postpartum behavior and the emergence of infant–mother and infant–father synchrony in preterm and full-term infants: The role of neonatal vagal tone. Dev Psychobiol 49: 290–302 doi:10.1002/dev. [DOI] [PubMed] [Google Scholar]
- 38. Righetti-Veltema M, Conne-Perréard E, Bousquet A, Manzano J (2002) Postpartum depression and mother-infant relationship at 3 months old. J Affect Disord 70: 291–306. [DOI] [PubMed] [Google Scholar]
- 39.Saphire-Bernstein S, Way BM, Kim HS, Sherman DK, Taylor SE (2011) Oxytocin receptor gene (OXTR) is related to psychological resources. Proc Natl Acad Sci U S A: 1–5. doi:10.1073/pnas.1113137108. [DOI] [PMC free article] [PubMed]
- 40. Heim C, Plotsky PM, Nemeroff CB (2004) Importance of studying the contributions of early adverse experience to neurobiological findings in depression. Neuropsychopharmacology 29: 641–648 doi:10.1038/sj.npp.1300397. [DOI] [PubMed] [Google Scholar]
- 41. Karevold E, Røysamb E, Ystrom E, Mathiesen KS (2009) Predictors and pathways from infancy to symptoms of anxiety and depression in early adolescence. Dev Psychol 45: 1051–1060 doi:10.1037/a0016123. [DOI] [PubMed] [Google Scholar]
- 42. Thoits PA (2011) Mechanisms linking social ties and support to physical and mental health. J Health Soc Behav 52: 145–161 doi:10.1177/0022146510395592. [DOI] [PubMed] [Google Scholar]
- 43.Skrundz M, Bolten M, Nast I, Hellhammer DH, Meinlschmidt G (2011) Plasma Oxytocin Concentration during Pregnancy is associated with Development of Postpartum Depression. Neuropsychopharmacology: 1–8. doi:10.1038/npp.2011.74. [DOI] [PMC free article] [PubMed]
- 44. Thompson RJ, Parker KJ, Hallmayer JF, Waugh CE, Gotlib IH (2011) Oxytocin receptor gene polymorphism (rs2254298) interacts with familial risk for psychopathology to predict symptoms of depression and anxiety in adolescent girls. Psychoneuroendocrinology 36: 144–147 doi:10.1016/j.psyneuen.2010.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Mileva-Seitz V, Kennedy J, Atkinson L, Steiner M, Levitan R, et al. (2011) Serotonin transporter allelic variation in mothers predicts maternal sensitivity, behavior and attitudes toward 6-month-old infants. Genes Brain Behav 10: 325–333 doi:10.1111/j.1601-183X.2010.00671.x. [DOI] [PubMed] [Google Scholar]
- 46. Mileva-Seitz V, Fleming AS, Meaney MJ, Mastroianni A, Sinnwell JP, et al. (2012) Dopamine receptors D1 and D2 are related to observed maternal behavior. Genes Brain Behav 11: 684–694 doi:10.1111/j.1601-183X.2012.00804.x. [DOI] [PubMed] [Google Scholar]
- 47. Bisceglia R, Jenkins JM, Wigg KG, O’Connor TG, Moran G, et al. (2012) Arginine vasopressin 1a receptor gene and maternal behavior: evidence of association and moderation. Genes Brain Behav 11: 262–268 doi:10.1111/j.1601-183X.2012.00769.x. [DOI] [PubMed] [Google Scholar]
- 48. Stivers JA, Kaltwasser MT, Hill PS, Hruby VJ, Crawley JN (1988) Ventral tegmental oxytocin induces grooming. Peptides 9 Suppl 1223–231. [DOI] [PubMed] [Google Scholar]
- 49. Maestripieri D, Hoffman CL, Anderson GM, Carter CS, Higley JD (2009) Mother-infant interactions in free-ranging rhesus macaques: relationships between physiological and behavioral variables. Physiol Behav 96: 613–619 doi:10.1016/j.physbeh.2008.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Krpan KM, Coombs R, Zinga D, Steiner M, Fleming AS (2005) Experiential and hormonal correlates of maternal behavior in teen and adult mothers. Horm Behav 47: 112–122 doi:10.1016/j.yhbeh.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 51. Huston AC, Rosenkrantz Aronson S (2005) Mothers’ time with infant and time in employment as predictors of mother–child relationships and children’s early development. Child Dev 76: 467–482. [DOI] [PubMed] [Google Scholar]
- 52. Kidd KK, Morar B, Castiglione CM, Zhao H, Pakstis a J, et al. (1998) A global survey of haplotype frequencies and linkage disequilibrium at the DRD2 locus. Hum Genet 103: 211–227. [DOI] [PubMed] [Google Scholar]
- 53. Tian C, Gregersen PK, Seldin MF (2008) Accounting for ancestry: population substructure and genome-wide association studies. Hum Mol Genet 17: R143–50 doi:10.1093/hmg/ddn268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ainsworth M, Bell S, Stayton D (1974) Infant-mother attachment and social development: “socialization” as a product of reciprocal responsiveness to signals. In: MPM Richards, editor. The Integration of a Child into a Social World. London: Cambridge University Press. 99–135. [Google Scholar]
- 55. Radloff LS (1977) The CES-D Scale: A Self-Report Depression Scale for Research in the General Population. Appl Psychol Meas 1: 385–401 doi:10.1177/014662167700100306. [Google Scholar]
- 56. Shafer AB (2006) Meta-analysis of the factor structures of four depression questionnaires: Beck, CES-D, Hamilton, and Zung. J Clin Psychol 62: 123–146 doi:10.1002/jclp.20213. [DOI] [PubMed] [Google Scholar]
- 57. Knight RG, Williams S, McGee R, Olaman S (1997) Psychometric properties of the Centre for Epidemiologic Studies Depression Scale (CES-D) in a sample of women in middle life. Behav Res Ther 35: 373–380. [DOI] [PubMed] [Google Scholar]
- 58. Morin AJS, Moullec G, Maïano C, Layet L, Just J-L, et al. (2011) Psychometric properties of the Center for Epidemiologic Studies Depression Scale (CES-D) in French clinical and nonclinical adults. Revue d’épidémiologie et de santé publique 59: 327–340 doi:10.1016/j.respe.2011.03.061. [DOI] [PubMed] [Google Scholar]
- 59.Caspi A, Moffitt TE, Thornton A, Freedman D, Amwell JW, et al.. (1996) AID-MPR156>3.3.CO;2-E. [Google Scholar]
- 60. Bernstein DP (2003) Development and validation of a brief screening version of the Childhood Trauma Questionnaire. Child Abuse Negl 27: 169–190 doi:10.1016/S0145-2134(02)00541–0. [DOI] [PubMed] [Google Scholar]
- 61. Parker G (1990) The parental bonding instrument. Soc Psychiatry Psychiatr Epidemiol 25: 281–282. [DOI] [PubMed] [Google Scholar]
- 62. Bernstein DP, Fink L, Handelsman L, Foote J, Lovejoy M, et al. (1994) Initial reliability and validity of a new retrospective measure of child abuse and neglect. Am J Psychiatry 151: 1132–1136. [DOI] [PubMed] [Google Scholar]
- 63. Wilhelm K, Niven H, Parker G, Hadzi-Pavlovic D (2005) The stability of the Parental Bonding Instrument over a 20-year period. Psychol Med 35: 387–393. [DOI] [PubMed] [Google Scholar]
- 64. Gotlib IH, Mount JH, Cordy NI, Whiffen VE (1988) Depression and perceptions of early parenting: a longitudinal investigation. Br J Psychiatry 152: 24–27 doi:10.1192/bjp.152.1.24. [DOI] [PubMed] [Google Scholar]
- 65.Strauss JS, Freeman NL, Shaikh S a., Vetró Á, Kiss E, et al.. (2010) No association between oxytocin or prolactin gene variants and childhood-onset mood disorders. Psychoneuroendocrinology: 1422–1428. doi:10.1016/j.psyneuen.2010.04.008. [DOI] [PMC free article] [PubMed]
- 66.Souza RP, De Luca V, Meltzer HY, Lieberman JA, Kennedy JL (2010) Schizophrenia severity and clozapine treatment outcome association with oxytocinergic genes. Int J Neuropsychopharmacol: 1–6. doi:10.1017/S1461145710000167. [DOI] [PubMed]
- 67. Israel S, Lerer E, Shalev I, Uzefovsky F, Riebold M, et al. (2009) The oxytocin receptor (OXTR) contributes to prosocial fund allocations in the dictator game and the social value orientations task. PLoS One 4: e5535 doi:10.1371/journal.pone.0005535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Edwards JR, Lambert LS (2007) Methods for integrating moderation and mediation: a general analytical framework using moderated path analysis. Psychol Methods 12: 1–22 doi:10.1037/1082-989X.12.1.1. [DOI] [PubMed] [Google Scholar]
- 69. Bryant GA, Barrett HC (2007) Recognizing intentions in infant-directed speech: evidence for universals. Psychol Sci 18: 746–751 doi:10.1111/j.1467-9280.2007.01970.x. [DOI] [PubMed] [Google Scholar]
- 70. Bornstein MH, Tal J, Rahn C, Galperin CZ, Pêcheux MG, et al. (1992) Functional analysis of the contents of maternal speech to infants of 5 and 13 months in four cultures: Argentina, France, Japan, and the United States. Dev Psychol 28: 593. [Google Scholar]
- 71. Bloom L, Margulis C, Tinker E, Fujita N (1996) Early Conversations and Word Learning: Contributions from Child and Adult. Child Dev 67: 3154–3175 doi:10.1111/j.1467-8624.1996.tb01907.x. [PubMed] [Google Scholar]
- 72. Baskerville TA, Douglas AJ (2010) Dopamine and oxytocin interactions underlying behaviors: potential contributions to behavioral disorders. CNS Neurosci Ther 16: e92–123 doi:10.1111/j.1755-5949.2010.00154.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Numan M (2007) Motivational systems and the neural circuitry of maternal behavior in the rat. Dev Psychobiol 49: 12–21 doi:10.1002/dev. [DOI] [PubMed] [Google Scholar]
- 74. Shahrokh DK, Zhang T-Y, Diorio J, Gratton A, Meaney MJ (2010) Oxytocin-dopamine interactions mediate variations in maternal behavior in the rat. Endocrinology 151: 2276–2286 doi:10.1210/en.2009-1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Van Ijzendoorn MH, Bakermans-Kranenburg MJ, Mesman J (2008) Dopamine system genes associated with parenting in the context of daily hassles. Genes Brain Behav 7: 403–410 doi:10.1111/j.1601-183X.2007.00362.x. [DOI] [PubMed] [Google Scholar]
- 76. Francis DD, Kuhar MJ (2008) Frequency of maternal licking and grooming correlates negatively with vulnerability to cocaine and alcohol use in rats. Pharmacol Biochem Behav 90: 497–500 doi:10.1016/j.pbb.2008.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Caldji C, Tannenbaum B, Sharma S, Francis DD, Plotsky PM, et al. (1998) Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proc Natl Acad Sci U S A 95: 5335–5340 doi:10.1073/pnas.95.9.5335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Field T (1992) Infants of depressed mothers. Dev Psychopathol 4: 49–66 doi:10.1017/S0954579400005551. [DOI] [PubMed] [Google Scholar]
- 79. Costa B, Pini S, Gabelloni P, Abelli M, Lari L, et al. (2009) Oxytocin receptor polymorphisms and adult attachment style in patients with depression. Psychoneuroendocrinology 34: 1506–1514 doi:10.1016/j.psyneuen.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 80. Inoue H, Yamasue H, Tochigi M, Abe O, Liu X, et al. (2010) Association Between the Oxytocin Receptor Gene and Amygdalar Volume in Healthy Adults. Biol Psychiatry 68: 1066–1072 doi:10.1016/j.biopsych.2010.07.019. [DOI] [PubMed] [Google Scholar]
- 81.Tost H, Kolachana B, Verchinski BA, Bilek E, Goldman AL, et al.. (2011) Neurogenetic effects of OXTR rs2254298 in the extended limbic system of healthy Caucasian adults. Biol Psychiatry 70: e37–9; author reply e41-2. doi:10.1016/j.biopsych.2011.06.034. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Path models depicting relationships between early experience, prenatal depression, and maternal instrumental care in two genotypes of rs2740210: C/C and A/C using coefficients from Table 6. Significance values are based on bias-corrected bootstrap adjusted confidence intervals. Prenatal depression is not a significant mediator of the early care quality associations with instrumental care, for either genotype.
(TIFF)
Sample characteristics and study variables.
(DOCX)
OXT haplotypes and regression models with these haplotypes.
(DOCX)
Coefficient estimates of path models (using prenatal CES-D).
(DOCX)
Simple effects of path models (using prenatal prenatal depression score as a mediator).
(DOCX)