Abstract
Background
Healthy young individuals benefit from sleep to promote offline enhancement of a variety of explicitly learned discrete motor tasks. It remains unknown if sleep will promote learning of other types of explicit tasks. The purpose of this study is to verify the role of sleep in learning an explicitly instructed discrete motor task and to determine if participants who practice an explicitly instructed continuous tracking task demonstrate sleep-dependent offline learning of this task.
Methods
In experiment 1, 28 healthy young adults (mean age 25.6 ± 3.8 years) practiced a serial reaction time (SRT) task at either 8 am (SRT no-sleep group) or 8 pm (SRT sleep group) and underwent retention testing 12 ± 1 hours later. In experiment 2, 20 healthy young individuals (mean age 25.6 ± 3.3 years) practiced a continuous tracking task and were similarly divided into a no-sleep (continuous tracking no-sleep group) or sleep group (continuous tracking sleep group). Individuals in both experiments were provided with explicit instruction on the presence of a sequence in their respective task prior to practice.
Results
Individuals in the SRT sleep group demonstrated a significant offline reduction in reaction time whereas the SRT no-sleep group did not. Results for experiment 1 provide concurrent evidence that explicitly learned discrete tasks undergo sleep-dependent offline enhancement. Individuals in the continuous tracking sleep group failed to demonstrate a significant offline reduction in tracking error. However, the continuous tracking no-sleep group did demonstrate a significant offline improvement in performance. Results for experiment 2 indicate that sleep is not critical for offline enhancement of an explicit learned continuous task.
Conclusion
The findings that individuals who practiced an explicitly instructed discrete task experienced sleep-dependent offline learning while those individuals who practiced an explicitly instructed continuous task did not may be due to the difference in motor control or level of complexity between discrete and continuous tasks.
Keywords: sleep, motor learning, discrete task, continuous task
Background
Evidence has accumulated supporting the notion that processes during sleep contribute significantly to motor learning and memory consolidation.1–7 Memory consolidation refers to either the stabilization or the enhancement (offline learning) of a memory through the passage of time without additional practice.8,9 Individuals who sleep following practice of a motor task demonstrate an improvement in performance of that task compared with participants who do not sleep.10–14 While sleep has been shown to promote offline motor skill learning, there are factors to consider, including the type of instruction provided and the type of task utilized, when examining the role of sleep on offline motor skill learning and memory consolidation.
The type of instruction provided to participants before or during practice influences whether offline motor learning is related to sleep or simply the passage of time.15,16 Studies have confirmed that sleep enhances offline learning if the task is explicitly learned, that is, the individuals practicing the task are aware of the pattern to be learned.15,17,18 In a study by Robertson et al,15 participants who were provided explicit information about the sequence to be learned and then practiced the motor task demonstrated an improvement in performance only if the training was followed by a period of sleep. In contrast, participants who were unaware of the sequence (learned the sequence implicitly) demonstrated an improvement in performance following sleep as well as a similar period of being awake. Supporting the findings from Robertson et al,15 Nemeth et al19 found that general skill learning of an implicit probabilistic task improved offline regardless of whether the offline period contained sleep or did not. Findings from a series of studies by Yordanova et al20–22 suggest that performance on an implicit task (the number reduction task) is enhanced overnight due to activation of the hippocampal system, which is associated with explicit learning and memory consolidation.23,24 In examining prior studies11–14 that also demonstrated offline enhancement of skill ability following a period of sleep, the similarity between these studies, although not stated outright, is that the participants in these studies all had explicit awareness of the skill being learning and, thus, lends support to the notion that offline motor memory consolidation of explicitly learned tasks is sleep-dependent. Taken together, these findings indicate that while memory consolidation of implicitly learned tasks is typically time-dependent, memory consolidation using explicit instruction is sleep-dependent.
While concurrent evidence supports the theory that explicit memory is preferentially enhanced offline during sleep,15,17,18 it remains unclear if sleep enhances all types of explicitly learned motor skills. Two important classifications for motor skills are discrete skills and continuous skills. Continuous motor skills are those in which the movement is cyclical and repetitive, with an arbitrary beginning and end.25,26 Examples of continuous skills include swimming, running, or performing a tracking task. In contrast, discrete motor skills represent a distinct, manipulation-type skill that has a specific beginning and end.26,27 Examples of discrete skills include kicking a soccer ball or pressing a key on the keyboard.
The two different classifications of motor skills are thought to rely on different mechanisms of motor control. Due to the often rapid nature of discrete tasks, these types of skills are thought to rely on a motor program to produce the rapid movement.26,28,29 There is often not enough time to use online feedback to correct the discrete movement while the movement is being performed. On the other hand, continuous tasks are thought to rely on the ability to use feedback to correct movements while the movement is being produced.30 Thus, sleep may preferentially enhance one type of skill due to the differences in motor control.
In addition to different mechanisms of motor control, studies demonstrate that discrete movements result in more extensive neuronal activation than continuous movements.25,26,31 Habas et al25 examined the cerebral and the cerebellar networks involved in performing a unimanual continuous movement and a unimanual discrete movement. They found overlap in the brain areas activated during the performance of the discrete and continuous motor tasks, but found that the discrete movements were associated with a stronger and bilateral activation of some of the brain areas. In addition, performance of the discrete movement specifically resulted in the recruitment of additional brain areas not observed during performance of the continuous movement. The authors suggest that the discrete movement produced more extensive neural activation than the continuous movement because the discrete movement requires more attentional and computational load to coordinate the sequential movements of a discrete task.25
In line with the findings of Habas et al25, Spencer et al32 found that a region within the cerebellum was more active when participants produce flexion-extension of the index finger with a brief pause prior to the next movement (discrete movement) compared with when the movements were produced without this discontinuity (continuous movement). The authors proposed that the discrete movements are represented as a sequence of successively timed events, and the cerebellum has a central role in the representation of the timing of these events. In contrast, continuous movements lack an event structure. Therefore, their timing can be achieved through the control of kinematic variables. Due to the various degrees of neuronal activation and differences in brain areas involved in producing a discrete task compared with a continuous task, sleep may impact the offline enhancement of these two types of tasks differently.
Most of the studies examining the beneficial role of sleep in motor performance enhancement have utilized discrete tasks such as a finger-to-thumb opposition task,12,18 a sequential finger-tapping task,11,13,14 and the serial reaction time (SRT) task.15 However, a few sleep studies have utilized continuous tasks to assess the role of sleep in motor learning or performance. Elmenhorst et al33 demonstrated that partial sleep deprivation over four nights significantly impaired performance on an unstable tracking task. The pursuit rotor task, another type of continuous task, has been utilized to elucidate changes in the brain representation of motor memory after sleep34 and to compare the changes that occur in sleep architecture following the acquisition of this task in young and older subjects.35 However, the pursuit rotor does not incorporate a novel sequence of movement, and it is difficult to assess the immediate effect of sleep on motor skill learning and memory consolidation because the retest sessions took place three days34 or one week36 following initial practice. One study used a continuous tracking task to assess sleep-dependent motor skill learning in individuals with stroke and demonstrated a benefit from sleep to promote learning of an explicitly learned continuous tracking task.37 Individuals with stroke performed with less error on the continuous tracking task following a night of sleep but not following a period of being awake. However, the interaction of sleep and explicit instruction on offline motor learning of a novel tracking task in young healthy individuals has never been considered.
Therefore, the purpose of this study is to verify performance on an explicitly instructed discrete task undergoes sleep-dependent enhancement (experiment 1) and to determine if performance on an explicitly instructed continuous task will benefit from sleep to promote overnight skill enhancement (experiment 2) in young healthy adults.
Experiment 1
Materials and methods
Subjects
In the first experiment, 28 healthy young adults (25.6 ± 3.8 years, 19 females, nine males) were recruited from the University of Kansas Medical Center and the community to practice the SRT task, a discrete task, either at 8 pm or 8 am and then return for retention testing after a 12 ± 1 hour period either including sleep (SRT sleep group) or not including sleep (SRT no-sleep group). Participants in the sleep group slept the night between practice and retention in their home, and participants in the no-sleep group conducted their normal daily activities. The study was conducted according to the regulations of and with approval from the Human Subjects Committee at the University of Kansas Medical Center. Written informed consent was received from all participants. All participants who consented completed the study. Participants did not receive payment for participation. Participants were excluded if they presented with acute medical problems, uncorrected vision loss, previous history of psychiatric admission or neurological disease, or scored below 26 on the Mini-Mental State Exam (MMSE). The Stanford Sleepiness Scale38 was used to assess level of sleepiness prior to practice and retention testing. The Stanford Sleepiness Scale is a seven-point ordinal scale, whereby 1 = wide awake and 7 = nearly asleep. Participants maintained a sleep log to assess the amount of sleep achieved by participants for the week prior to practice. Participants were instructed to refrain from alcohol and caffeine for 24 hours prior to and during testing. There were no differences between the sleep and no-sleep group for age (F1,26 = 0.406, P = 0.530), MMSE score (F1,26 = 0.000, P = 1.00), amount of sleep the week prior to practice (F1,26 = 1.349, P = 0.256), or the level of sleepiness at practice (F1,26 = 1.650, P = 0.210) or retention testing (F1,26 = 1.000, P = 0.327, Table 1).
Table 1.
Descriptive information
| Group | Age (years) | MMSE | Average sleep (hours) | SSS1 | SSS2 |
|---|---|---|---|---|---|
| SRT Task | |||||
| Sleep | 26.1 (5.0) | 29.9 (0.4) | 7.6 (0.6) | 2.4 (0.7) | 2.4 (1.0) |
| No-sleep | 25.1 (2.1) | 29.9 (0.4) | 7.4 (0.6) | 2.7 (0.7) | 2.0 (0.9) |
| CT Task | |||||
| Sleep | 26.5 (4.0) | 30.0 (0.0) | 7.4 (0.8) | 2.9 (1.7) | 2.3 (1.1) |
| No-Sleep | 24.6 (2.1) | 30.0 (0.0) | 7.3 (0.9) | 2.5 (0.8) | 1.5 (0.7) |
Note: Data are mean (standard deviation).
Abbreviations: MMSE, Mini-mental Status Exam; Average sleep, average amount of sleep the week prior to testing determined by sleep log; SSS1, Stanford Sleepiness Scale at practice session; SSS2, Stanford Sleepiness Scale at retention testing.
Serial reaction time task
During SRT task practice, participants sat in front of a computer with the most centered letters on the centrally placed keyboard (v, b, n, and m) capped with the colors red, yellow, blue, and green, respectively. Only one colored circle was displayed on the computer screen at a time. Participants responded using the first four fingers of their dominant hand, pressing one of the four keys corresponding to the appropriately colored circle. Participants were instructed to respond as quickly and accurately as possible.
Fifteen blocks of the SRT task were performed during the practice session. Each block included 100 responses. The first block and second to last block (block 14) of responses contained randomly ordered stimuli. The middle 12 blocks and last block (block 15) consisted of a repeating 10-element sequence (blue-yellow-red-blue-green-red-blue-red-green-yellow) with an ambiguous or minimal probabilistic relationship between the elements. The sequence was designed to contain no more than one trill (eg, red, blue, red) and have no repeating positions or colors. The transition between the end of one sequence and the beginning of the next within a block was not marked. In total, during practice, the 10-element sequence was performed 130 times. Participants returned for delayed retention testing 12 hours (± 1 hour) following practice either including sleep (sleep group) or not including sleep (no-sleep group). Retention testing consisted of two blocks, ie, one random block followed by a repeating sequence block.
Explicit instruction for serial reaction time task
Participants received instruction prior to practice regarding the presence of the repeating sequence. First, they studied a pictorial representation of, but were not allowed to physically practice, the sequence. A recognition test prior to practice verified that participants had acquired explicit knowledge regarding the repeating sequence.39 Ten iterations of a pictorial representation of either the sequence they were instructed to learn (n = 3) or a foil sequence (n = 7) were shown; participants had to decide (forced choice) if the sequence was one they recognized as the sequence they explicitly learned. Participants were required to score at least 80% correct on the recognition test or would undergo the instruction process until 80% was achieved. No participants required reinstruction.
Statistical analysis
The median response time for each 10-element sequence was calculated, and the mean median response time for each block was calculated as the summary score. Acquisition practice performance was examined using a two factor (group [sleep, no-sleep] X block [2–13,15]) repeated-measures analysis of variance, with response time score as the dependent variable. Offline learning was calculated by subtracting the retention block response time from the response time of the last practice block. Parameter estimates were then generated by a repeated-measures analysis of variance model to assess the significance of change in motor behavior associated with offline motor skill learning attributable to sleep for the sleep group or the passage of time for the no-sleep group.
Results
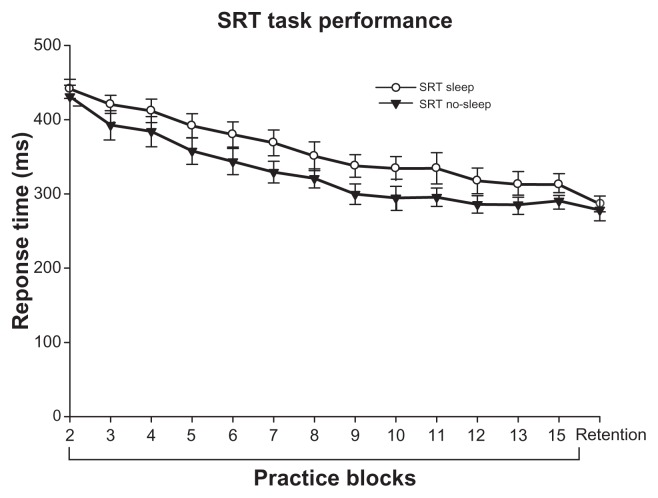
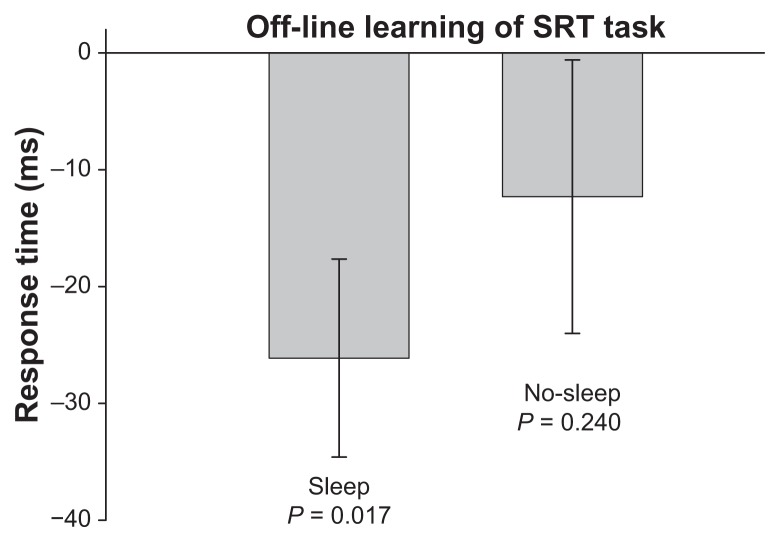
As Figure 1 shows, performance for both the sleep group and no-sleep group benefited from practice, as shown by a main effect of block (F12,312 = 62.849, P < 0.001), indicating that both the SRT sleep and SRT no-sleep groups became faster with training. The time of day of practice did not impact acquisition performance of the SRT task, as evidenced by the insignificant group effect (main effect of group, F1,26 = 2.493, P = 0.126). The group by block interaction was not significant. Only the SRT sleep group demonstrated significant offline motor learning of the SRT task, achieving a faster response time at retention compared with the last block of practice (P = 0.017, Figure 2); the SRT no-sleep group did not (P = 0.240, Figure 2). This first experiment supports previous literature demonstrating that sleep promotes offline motor learning of an explicitly learned discrete motor task in healthy young adults.
Figure 1.
RT performance (in milliseconds) on the SRT task across the practice blocks and at retention. Error bars are SEM.
Figure 2.
The off-line learning score or change in RT performance (in milliseconds) between the last practice block and the retention test for the SRT sleep and SRT no-sleep group. A negative score indicates a faster RT at retention compared to the last block of practice. Error bars are SEM.
Experiment 2
Materials and methods
Subjects
Twenty-two healthy young individuals were recruited to participate in the second experiment. Two of the participants did not complete testing and their data was not included in data analysis. Twenty individuals (25.6 ± 3.3 years, 13 females, seven males) practiced a continuous tracking task consisting of a repeating sequence embedded within two random segments. A continuous tracking task was originally developed by Pew40 and has since been used by others41–45 to examine continuous motor sequence learning. Similar to the first experiment, participants practiced the continuous tracking task at either 8 pm or 8 am with a retention test approximately 12 hours later. Individuals in the continuous tracking sleep group slept between practice and retention testing while individuals in the continuous tracking no-sleep group stayed awake. As in Experiment 1, participants were recruited from the University of Kansas Medical Center and the community, written informed consent was obtained, and participants were not paid for participation. Similar exclusion criteria and sleep assessments were used as in Experiment 1. The continuous tracking sleep and continuous tracking no-sleep group did not differ in age (F1,18 = 1.738, P = 0.204), MMSE score (F1,18 = 0.000, P = 1.000), amount of sleep the week prior to practice (F1,18 = 0.026, P = 0.873), or level of sleepiness at practice (F1,18 = 0.459, P = 0.507) or at retention testing (F1,18 = 3.470, P = 0.079, Table 1).
Continuous tracking task
To practice the continuous tracking task, participants were instructed to control a joystick to track a target on the computer screen that moved in a sinusoidal wave pattern.42,45 Only the target (white box) and the participant’s cursor position (red circle) were visible to the participant; there was no residual trace of the wave on the screen. Each participant practiced the continuous tracking task for eight blocks of 10 trials, each for a total of 80 iterations of the repeating wave pattern. Each trial consisted of three segments, ie, one repeating segment imbedded between two random segments. Each segment was 12 seconds long, for a total trial length of 36 seconds, with a three-second stable baseline trial divider. To assess offline motor learning, participants completed one block (10 trials) of the continuous tracking task at a retention test 12 ± 1 hours after practice.
Explicit instruction for continuous tracking task
All participants received explicit instruction prior to practice that a wave sequence would be repeated throughout practice of the continuous tracking task. Participants were first verbally instructed that a sequence would occur, but that the sequence might be difficult to identify during practice because it would be embedded between random waves. Participants then watched the sequence for as many times as they requested but did not perform the repeating sequence on the computer. Participants then underwent a recognition test prior to practice to ensure acquisition of explicit knowledge of the repeating sequence. Participants watched a target move on the computer screen and were asked to indicate whether or not the sequence displayed was the repeating sequence. Ten iterations were shown, ie, three of the repeating sequence and seven foils. Similar to experiment 1, participants were required to score at least 80% correct on the recognition test. If an 80% correct was not achieved, participants would again receive the explicit instruction until they were able to score 80% correct on the recognition test. Two participants required reinstruction to achieve a score of at least 80%.
Statistical analysis
The root mean square error (RMSE) for the sequence was calculated for each trial, and median RMSE was calculated for each block as a summary score for tracking accuracy of the tracking task.42,44,45 Similar to the first experiment, a two factor (group [sleep, no-sleep] X block [1–8]) repeated-measures analysis of variance with RMSE as the dependent variable assessed practice performance, and an offline learning score was generated by subtracting the retention block RMSE from the last practice block RMSE. Parameter estimates generated by a repeated-measures analysis of variance model assessed sleep-dependent or time-dependent offline motor learning.
Results
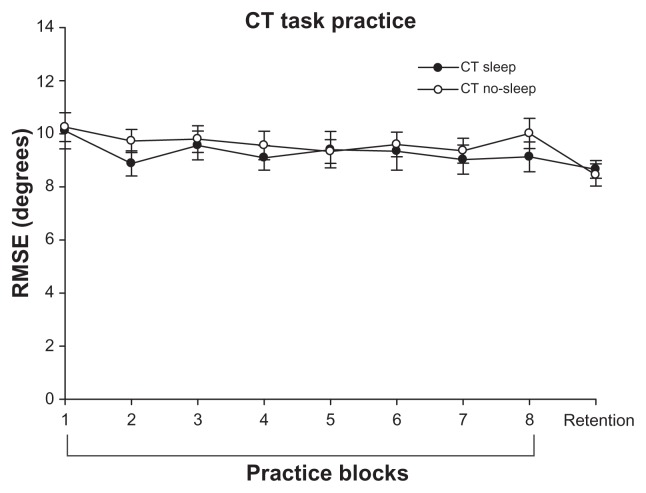
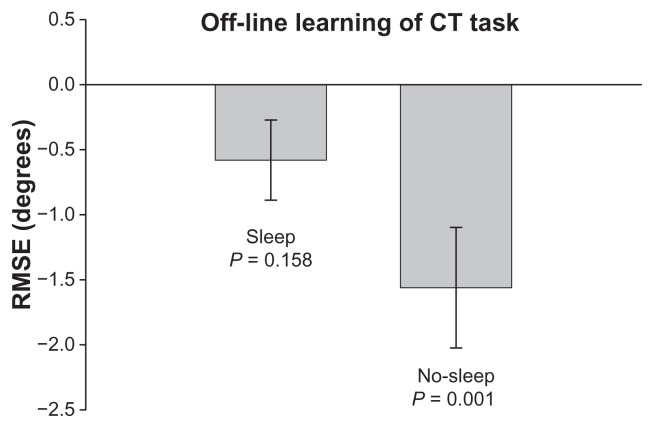
As Figure 3 shows, both the continuous tracking sleep group and continuous tracking no-sleep group demonstrated a significant reduction in error during practice (main effect of block, F7,126 = 3.130, P = 0.004) despite practicing at different times of day (main effect of group, F1,18 = 0.306, P = 0.587). The interaction was not significant. The continuous tracking no-sleep group demonstrated significant offline motor learning of the continuous tracking task (P = 0.001, Figure 4) but the continuous tracking sleep group failed to demonstrate offline motor learning (P = 0.158, Figure 4). This suggests that while sleep may promote offline learning of explicit discrete tasks, these findings do not generalize to a continuous task learned explicitly.
Figure 3.
RMSE performance (in degrees) on the CT task across the practice blocks and at retention. Error bars are SEM.
Figure 4.
The off-line learning score or change in tracking error (in RMSE) between the last practice block and the retention test for the CT sleep and CT no-sleep group. A negative score indicates less tracking error at retention compared to the last block of practice. Error bars are SEM.
Discussion
The results of this study expand on previous work that has examined the role of sleep in explicitly learned motor tasks. The results support prior studies11–15 that demonstrate explicitly learned discrete tasks benefit from sleep to promote offline learning of the task for healthy young individuals. This study is the first to demonstrate that explicitly learning a continuous task is not enhanced overnight, but is enhanced through the passage of time not involving sleep.
One reason we propose that the explicitly learned discrete task benefited from sleep while the explicitly learned continuous task did not is the difference in motor control between these tasks. Discrete tasks are thought to rely on motor programs, while continuous tasks are thought to rely on feedback to produce movement.26,28,29 These differences in motor control may result in a differential effect of sleep on learning these skills. Perhaps the benefits of sleep are related to the development of the motor program, and would therefore assist the performance of discrete tasks but not continuous tasks. Future studies are needed to support this contention.
The findings of this study appear to support the findings of Kuriyama et al46 that more complex tasks experience a larger improvement in performance following sleep compared with simpler tasks. In the study by Kuriyama et al, participants who practiced a bimanual nine-element sequence showed a larger percentage improvement following a night of sleep compared with those participants who practiced a unimanual five-element sequence. Kuriyama et al used a discrete sequential finger-tapping task to assess offline learning, and discrete tasks have been found to be more complex than continuous tasks. By studying the neural networks involved in controlling simple discrete and continuous movements, neuroimaging studies19,20,23 found that discrete movements requires more extensive neuronal activation than continuous movements. Habas et al25 found that a discrete task is accompanied by more widespread and stronger brain activations than a continuous task, although the muscles and number of joints involved in producing both tasks are the same. They suggest that these differences are attributed to a greater complexity of the discrete task.25 In particular, increased activation in the sensorimotor, premotor cortex, the basal ganglia, and in the cerebellum have been found with the performance of a discrete task,19,20,23,32 and these areas have been positively correlated with task difficulty.47 Our findings, along with these previous studies, would suggest that sleep is more likely to enhance learning of complex tasks.
While we cannot completely rule out the influence of circadian rhythm or time-of-day of testing on the results, we feel that a time-of-day effect is an unlikely explanation for our findings. Both the sleep and no-sleep SRT groups demonstrated improvements in performance across practice (demonstrated by a main effect of block) but did not perform significantly different from each other across practice (demonstrated by no significant main effect of group) regardless of the time of day the practice session occurred. A similar improvement across practice but no group difference at practice was also demonstrated by the sleep and no-sleep continuous tracking groups. This suggests that the sleep and no-sleep groups for both the SRT and continuous tracking task performed similarly to each other despite practicing at different times of day. Furthermore, the Stanford Sleepiness Scale did not reveal group differences at practice or retention in either the SRT or continuous tracking condition at practice or retention regardless of time of day the testing occurred. In addition, if time-of-day of testing influenced our results, we would have anticipated the sleep groups and the no-sleep groups to experience similar offline learning changes regardless of task, which was not the case.
A limitation of this study is that objective sleep parameters were not acquired to verify sleep or quality of sleep during the night between the practice and retention session for the sleep groups. We did include the intervening night of sleep on the subject-maintained sleep log to ensure participants in the sleep groups did sleep during the night between practice and retention testing. Another limitation is we did not reassess explicit awareness at retention testing. We tested to ensure participants had explicit awareness prior to the initiation of practice, but did not reassess explicit awareness to verify explicit awareness had been retained. Another limitation is we cannot verify that the participants in the SRT groups had the same degree of explicit awareness as the participants in the continuous tracking groups. We ensured that all participants were able to recognize the sequence with 80% accuracy, but we did not assess for more salient explicit awareness with a recall task or production task.
The participants in the first experiment practiced the SRT sequence 130 times while the participants in the second experiment practiced the continuous tracking task sequence 80 times. We think it is unlikely that more practice of the sequence led to sleep-dependent offline learning of the SRT task and less practice inhibited sleep-dependent learning of the continuous tracking task. This contention is supported by Walker et al14 who determined that doubling the amount of practice did not result in additional offline improvement of the task. One would suspect that a critical amount of practice would be required to encode the memory to allow the memory to be consolidated, but this critical amount remains to be determined and would likely be task-specific. Eighty iterations of practice of the continuing tracking task sequence was sufficient to produce time-dependent offline learning of the sequence for the no-sleep group. Because it is likely that over-the-day memory consolidation is a different process than overnight memory consolidation,48,49 it cannot be ruled out that a continuous task requires a critical amount of practice to produce sleep-dependent offline learning and this critical amount was not achieved for this current study. Future studies are needed to determine the critical amount of practice required to produce sleep-dependent offline learning and if this critical amount of practice is task dependent.
Conclusion
Supporting previous studies,11–15 participants who practiced an explicit discrete task and slept following practice demonstrated a significant offline improvement in performance, while those participants who stayed awake did not demonstrate offline learning. However, those individuals who practiced an explicit continuous task did not demonstrate sleep-dependent offline learning, but did demonstrate time-dependent offline learning. It is proposed that the differences in motor control and differences in task complexity support sleep-dependent offline enhancement of discrete tasks but not continuous ones. These results have important implications for learning different types of tasks for healthy young adults.
Acknowledgment
The authors would like to acknowledge the contribution of E Lezi, PhD student at the University of Kansas Medical Center, for data collection.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Walker MP, Stickgold R. Sleep, memory, and plasticity. Annu Rev Psychol. 2006;57:139–166. doi: 10.1146/annurev.psych.56.091103.070307. [DOI] [PubMed] [Google Scholar]
- 2.Walker MP, Stickgold R. Sleep-dependent learning and memory consolidation. Neuron. 2004;44(1):121–133. doi: 10.1016/j.neuron.2004.08.031. [DOI] [PubMed] [Google Scholar]
- 3.Rauchs G, Desgranges B, Foret J, Eustache F. The relationships between memory systems and sleep stages. J Sleep Res. 2005;14(2):123–140. doi: 10.1111/j.1365-2869.2005.00450.x. [DOI] [PubMed] [Google Scholar]
- 4.Peigneux P, Laureys S, Delbeuck X, Maquet P. Sleeping brain, learning brain. The role of sleep for memory systems. Neuroreport. 2001;12(18):A111–A124. doi: 10.1097/00001756-200112210-00001. [DOI] [PubMed] [Google Scholar]
- 5.Stickgold R, Walker MP. Sleep and memory: The ongoing debate. Sleep. 2005;28(10):1225–1227. doi: 10.1093/sleep/28.10.1225. [DOI] [PubMed] [Google Scholar]
- 6.Stickgold R. Sleep-dependent memory consolidation. Nature. 2005;437( 7063):1272–1278. doi: 10.1038/nature04286. [DOI] [PubMed] [Google Scholar]
- 7.Stickgold R, Walker MP. Memory consolidation and reconsolidation: What is the role of sleep? Trends Neurosci. 2005;28(8):408–415. doi: 10.1016/j.tins.2005.06.004. [DOI] [PubMed] [Google Scholar]
- 8.McGaugh JL. Memory – a century of consolidation. Science. 2000;287( 5451):248–251. doi: 10.1126/science.287.5451.248. [DOI] [PubMed] [Google Scholar]
- 9.Robertson EM, Pascual-Leone A, Miall RC. Current concepts in procedural consolidation. Nat Rev Neurosci. 2004;5(7):576–582. doi: 10.1038/nrn1426. [DOI] [PubMed] [Google Scholar]
- 10.Korman M, Raz N, Flash T, Karni A. Multiple shifts in the representation of a motor sequence during the acquisition of skilled performance. Proc Natl Acad Sci U S A. 2003;100(21):12492–12497. doi: 10.1073/pnas.2035019100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Walker MP, Brakefield T, Morgan A, Hobson JA, Stickgold R. Practice with sleep makes perfect: Sleep-dependent motor skill learning. Neuron. 2002;35(1):205–211. doi: 10.1016/s0896-6273(02)00746-8. [DOI] [PubMed] [Google Scholar]
- 12.Fischer S, Hallschmid M, Elsner AL, Born J. Sleep forms memory for finger skills. Proc Natl Acad Sci U S A. 2002;99(18):11987–11991. doi: 10.1073/pnas.182178199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Walker MP, Brakefield T, Hobson JA, Stickgold R. Dissociable stages of human memory consolidation and reconsolidation. Nature. 2003;425(6958):616–620. doi: 10.1038/nature01930. [DOI] [PubMed] [Google Scholar]
- 14.Walker MP, Brakefield T, Seidman J, Morgan A, Hobson JA, Stickgold R. Sleep and the time course of motor skill learning. Learn Mem. 2003;10(4):275–284. doi: 10.1101/lm.58503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Robertson EM, Pascual-Leone A, Press DZ. Awareness modifies the skill-learning benefits of sleep. Curr Biol. 2004;14(3):208–212. doi: 10.1016/j.cub.2004.01.027. [DOI] [PubMed] [Google Scholar]
- 16.Diekelmann S, Born J. The memory function of sleep. Nat Rev Neurosci. 2010;11(2):114–126. doi: 10.1038/nrn2762. [DOI] [PubMed] [Google Scholar]
- 17.Drosopoulos S, Wagner U, Born J. Sleep enhances explicit recollection in recognition memory. Learn Mem. 2005;12(1):44–51. doi: 10.1101/lm.83805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fischer S, Drosopoulos S, Tsen J, Born J. Implicit learning – explicit knowing: A role for sleep in memory system interaction. J Cogn Neurosci. 2006;18(3):311–319. [PubMed] [Google Scholar]
- 19.Nemeth D, Janacsek K, Londe Z, Ullman MT, Howard DV, Howard JH., Jr Sleep has no critical role in implicit motor sequence learning in young and old adults. Exp Brain Res. 2010;201(2):351–358. doi: 10.1007/s00221-009-2024-x. [DOI] [PubMed] [Google Scholar]
- 20.Yordanova J, Kolev V, Verleger R, Bataghva Z, Born J, Wagner U. Shifting from implicit to explicit knowledge: Different roles of early-and late-night sleep. Learn Mem. 2008;15(7):508–515. doi: 10.1101/lm.897908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yordanova J, Kolev V, Wagner U, Verleger R. Covert reorganization of implicit task representations by slow wave sleep. PLos One. 2009;4(5):e5675. doi: 10.1371/journal.pone.0005675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yordanova J, Kolev V, Verleger R. Awareness of knowledge or awareness of processing? Implications for sleep-related memory consolidation. Front Hum Neurosci. 2009;3:40. doi: 10.3389/neuro.09.040.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Squire LR, Zola SM. Structure and function of declarative and nondeclarative memory systems. Proc Natl Acad Sci U S A. 1996;93(24):13515–13522. doi: 10.1073/pnas.93.24.13515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Squire LR, Stark CE, Clark RE. The medial temporal lobe. Annu Rev Neurosci. 2004;27:279–306. doi: 10.1146/annurev.neuro.27.070203.144130. [DOI] [PubMed] [Google Scholar]
- 25.Habas C, Cabanis EA. Neural correlates of simple unimanual discrete and continuous movements: A functional imaging study at 3 T. Neuroradiology. 2008;50(4):367–375. doi: 10.1007/s00234-007-0354-6. [DOI] [PubMed] [Google Scholar]
- 26.Schaal S, Sternad D, Osu R, Kawato M. Rhythmic arm movement is not discrete. Nat Neurosci. 2004;7(10):1136–1143. doi: 10.1038/nn1322. [DOI] [PubMed] [Google Scholar]
- 27.Sternad D, Dean WJ. Rhythmic and discrete elements in multi-joint coordination. Brain Res. 2003;989(2):152–171. doi: 10.1016/s0006-8993(03)03292-x. [DOI] [PubMed] [Google Scholar]
- 28.Howard IS, Ingram JN, Wolpert DM. Separate representations of dynamics in rhythmic and discrete movements: Evidence from motor learning. J Neurophysiol. 2011;105(4):1722–1731. doi: 10.1152/jn.00780.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ikegami T, Hirashima M, Taga G, Nozaki D. Asymmetric transfer of visuomotor learning between discrete and rhythmic movements. J Neurosci. 2010;30(12):4515–4521. doi: 10.1523/JNEUROSCI.3066-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schmidt RA, Lee TD. Motor Control and Learning: A Behavioral Emphasis. 3rd ed. Champaign, IL: Human Kinetics; 1999. [Google Scholar]
- 31.Gowen E, Miall RC. Differentiation between external and internal cuing: An fMRI study comparing tracing with drawing. Neuroimage. 2007;36(2):396–410. doi: 10.1016/j.neuroimage.2007.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Spencer RM, Verstynen T, Brett M, Ivry R. Cerebellar activation during discrete and not continuous timed movements: An fMRI study. Neuroimage. 2007;36(2):378–387. doi: 10.1016/j.neuroimage.2007.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Elmenhorst D, Elmenhorst EM, Luks N, et al. Performance impairment during four days partial sleep deprivation compared with the acute effects of alcohol and hypoxia. Sleep Med. 2009;10(2):189–197. doi: 10.1016/j.sleep.2007.12.003. [DOI] [PubMed] [Google Scholar]
- 34.Maquet P, Schwartz S, Passingham R, Frith C. Sleep-related consolidation of a visuomotor skill: Brain mechanisms as assessed by functional magnetic resonance imaging. J Neurosci. 2003;23(4):1432–1440. doi: 10.1523/JNEUROSCI.23-04-01432.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Peters KR, Ray L, Smith V, Smith C. Changes in the density of stage 2 sleep spindles following motor learning in young and older adults. J Sleep Res. 2008;17(1):23–33. doi: 10.1111/j.1365-2869.2008.00634.x. [DOI] [PubMed] [Google Scholar]
- 36.Peters KR, Smith V, Smith CT. Changes in sleep architecture following motor learning depend on initial skill level. J Cogn Neurosci. 2007;19(5):817–829. doi: 10.1162/jocn.2007.19.5.817. [DOI] [PubMed] [Google Scholar]
- 37.Siengsukon CF, Boyd LA. Sleep to learn after stroke: Implicit and explicit offline motor learning. Neurosci Lett. 2009;451(1):1–5. doi: 10.1016/j.neulet.2008.12.040. [DOI] [PubMed] [Google Scholar]
- 38.Hoddes E, Zarcone V, Smythe H, Phillips R, Dement WC. Quantification of sleepiness: A new approach. Psychophysiology. 1973;10(4):431–436. doi: 10.1111/j.1469-8986.1973.tb00801.x. [DOI] [PubMed] [Google Scholar]
- 39.Boyd LA, Winstein CJ. Impact of explicit information on implicit motor-sequence learning following middle cerebral artery stroke. Phys Ther. 2003;83(11):976–989. [PubMed] [Google Scholar]
- 40.Pew RW. Levels of analysis in motor control. Brain Res. 1974;71(2–3):393–400. doi: 10.1016/0006-8993(74)90983-4. [DOI] [PubMed] [Google Scholar]
- 41.Shea CH, Wulf G, Whitacre CA, Park JH. Surfing the implicit wave. Q J Exp Psychol A. 2001;54(3):841–862. doi: 10.1080/713755993. [DOI] [PubMed] [Google Scholar]
- 42.Boyd LA, Winstein CJ. Providing explicit information disrupts implicit motor learning after basal ganglia stroke. Learn Mem. 2004;11(4):388–396. doi: 10.1101/lm.80104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Boyd LA, Winstein CJ. Cerebellar stroke impairs temporal but not spatial accuracy during implicit motor learning. Neurorehabil Neural Repair. 2004;18(3):134–143. doi: 10.1177/0888439004269072. [DOI] [PubMed] [Google Scholar]
- 44.Boyd LA, Winstein CJ. Explicit information interferes with implicit motor learning of both continuous and discrete movement tasks after stroke. J Neurol Phys Ther. 2006;30(2):46–57. doi: 10.1097/01.npt.0000282566.48050.9b. [DOI] [PubMed] [Google Scholar]
- 45.Wulf G, Schmidt RA. Variability of practice and implicit motor learning. J Exp Psychol Learn Mem Cogn. 1997;23(4):987–1006. [Google Scholar]
- 46.Kuriyama K, Stickgold R, Walker MP. Sleep-dependent learning and motor-skill complexity. Learn Mem. 2004;11(6):705–713. doi: 10.1101/lm.76304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Seidler RD, Noll DC, Thiers G. Feedforward and feedback processes in motor control. Neuroimage. 2004;22(4):1775–1783. doi: 10.1016/j.neuroimage.2004.05.003. [DOI] [PubMed] [Google Scholar]
- 48.Robertson EM, Press DZ, Pascual-Leone A. Offline learning and the primary motor cortex. J Neurosci. 2005;25(27):6372–6378. doi: 10.1523/JNEUROSCI.1851-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Diekelmann S, Born J. One memory, two ways to consolidate? Nat Neurosci. 2007;10(9):1085–1086. doi: 10.1038/nn0907-1085. [DOI] [PubMed] [Google Scholar]