Abstract
OBJECTIVE
Impaired insulin sensitivity increases the risk of cardiovascular disease. Although calorie restriction and weight loss increase insulin sensitivity, the effects of modifying macronutrient composition on insulin sensitivity are uncertain. The purpose of this study is to determine the effects on insulin sensitivity of a carbohydrate-rich diet (CARB; similar to the Dietary Approaches to Stop Hypertension [DASH] diet), a protein-rich diet (PROT; protein predominantly from plant sources), and an unsaturated fat–rich diet (UNSAT; predominantly monounsaturated).
RESEARCH DESIGN AND METHODS
This study was a randomized, controlled, three-period, crossover feeding study. The study participants were 164 individuals with prehypertension or stage 1 hypertension without diabetes. Diets were administered for 6 weeks each, with a washout period between diets of 2–4 weeks. Weight was held constant throughout the study. For our primary outcome, we calculated the quantitative insulin sensitivity check index (QUICKI) using the end-of-period fasting serum glucose and insulin. QUICKI is a validated measure of insulin sensitivity. The primary analyses used generalized estimating equations.
RESULTS
At baseline, mean (SD) BMI was 30.2 (6.1) kg/m2, and mean (SD) QUICKI was 0.35 (0.04). The UNSAT diet increased QUICKI by 0.005, more than the CARB diet (P = 0.04). PROT had no significant effect compared with CARB.
CONCLUSIONS
A diet that partially replaces carbohydrate with unsaturated fat may improve insulin sensitivity in a population at risk for cardiovascular disease. Given the well-recognized challenges of sustaining weight loss, our results suggest an alternative approach for improving insulin sensitivity.
Insulin resistance is a physiological decrease in sensitivity to the actions of insulin (1). The prevalence of insulin resistance is dramatically increasing as a result of the global obesity epidemic (2). Aside from leading to type 2 diabetes, insulin resistance increases the risk of cardiovascular disease (3–5). It is well established that daily caloric restriction and weight loss improve insulin sensitivity (6). Whether modifying the macronutrient composition of diet has beneficial effects on insulin sensitivity is unclear but may be an effective approach for increasing insulin sensitivity, given the challenges of sustaining weight loss.
We evaluated the effects of three diets of differing macronutrient intake on insulin sensitivity: a diet high in carbohydrates (CARB), compared with diets in which carbohydrate is partially replaced with protein (PROT) and unsaturated fat (UNSAT) (7). We assessed changes in fasting insulin, glucose, and the quantitative insulin sensitivity check index (QUICKI), a fasting steady-state index of insulin sensitivity (8). We hypothesized that diets with increased proportions of proteins and unsaturated fats, in comparison with a high-carbohydrate diet, would increase insulin sensitivity.
RESEARCH DESIGN AND METHODS
The Optimal Macronutrient Intake Trial to Prevent Heart Disease (OmniHeart) was an investigator-initiated, National Heart, Lung, and Blood Institute–funded trial (7). The study methods and primary results have been published elsewhere (7,9). In brief, it was a randomized, three-period, crossover feeding study. This protocol was reviewed and approved by institutional review boards at the two participating clinical centers (The Johns Hopkins University medical institutions and Brigham and Women’s Hospital).
Participants
Trial participants were adults in overall good health, 30 years of age and older, with either prehypertension or stage 1 hypertension (systolic blood pressure of 120–159 mmHg or a diastolic blood pressure of 80–99 mmHg). The exclusion criteria were preexisting diabetes; active or prior cardiovascular disease; LDL cholesterol >220 mg/dL; fasting triglycerides >750 mg/dL; weight >350 lbs; use of insulin, hypoglycemic agents, medications that affect blood pressure or blood lipid levels, or vitamin and mineral supplements; and self-reported alcohol intake of >14 drinks per week. By design, the trial enrolled ∼50% African Americans.
Participant flow
Eligibility was ascertained and baseline data were collected during three screening visits. Participants subsequently underwent a 6-day run-in period in which participants ate meals from each study diet for 2 days to ensure tolerance to study foods. They were then randomly assigned to one of six sequences of the three diets through computer-generated assignments. Each feeding period lasted 6 weeks. A washout period of 2–4 weeks separated the feeding periods. During the washout, participants ate their own foods.
Study diets
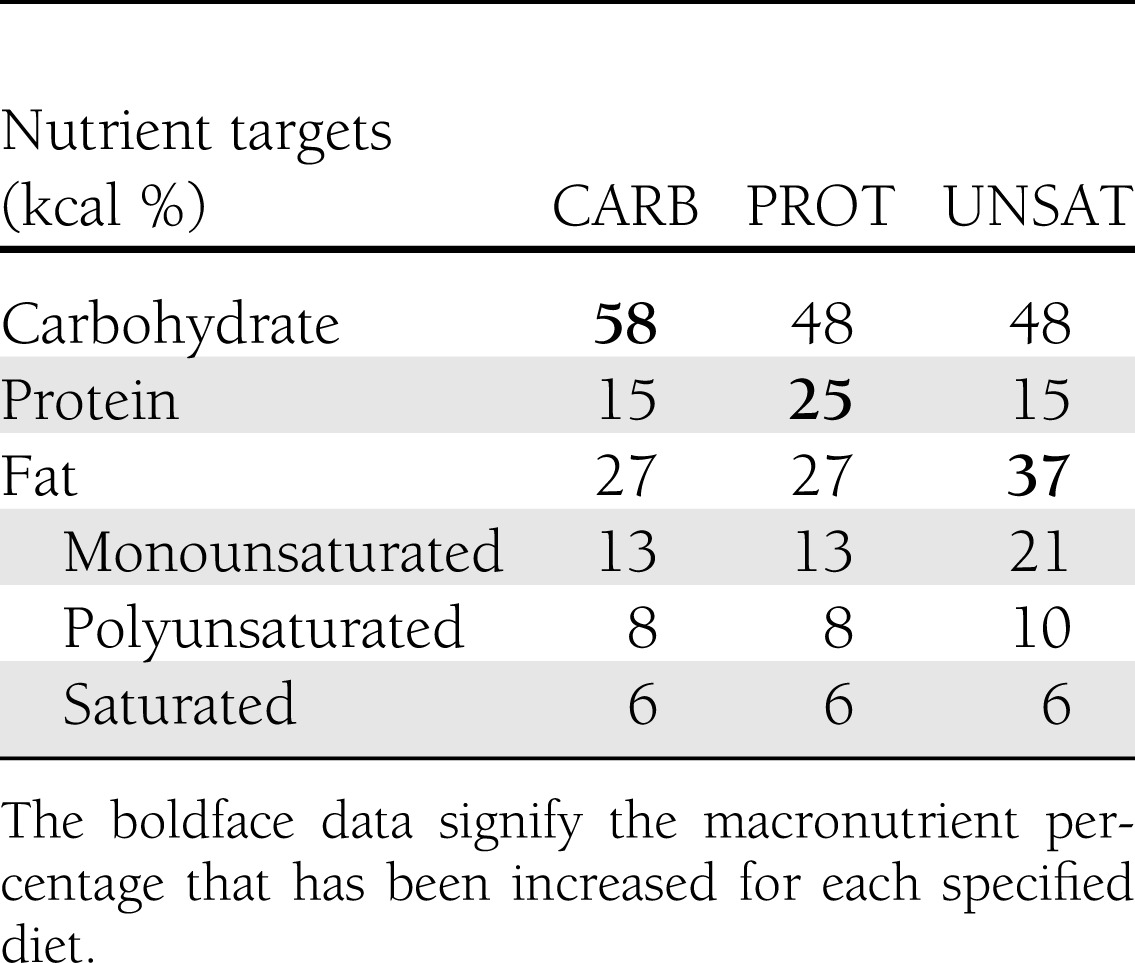
The macronutrient composition for study diets is presented in Table 1. The CARB diet provided 58% of kilocalories from carbohydrate, 15% from protein, and 27% from fat, and was similar to the Dietary Approaches to Stop Hypertension (DASH) diet (10). The PROT diet replaced 10% of carbohydrate calories with protein and provided 25% of calories from protein, 48% from carbohydrates, and 27% from fat. The UNSAT diet replaced 10% of carbohydrate calories with unsaturated fats and provided 48% of calories from carbohydrates, 15% from protein, and 37% from fat. The increase in fat in the UNSAT diet primarily resulted from an increase in monounsaturated fat from olive, canola, and safflower oils and nuts and seeds. Each diet provided 6% of calories from saturated fat. Protein sources included meat, poultry, egg product substitutes, and dairy; however, most of the increase in the PROT diet came from plant-based sources. The type of carbohydrate in each diet was similar, as indicated by the total dietary glycemic index (68 in CARB diet, 71 in the PROT diet, and 75 in UNSAT diet, relative to the white bread index).
Table 1.
Macronutrient composition of study diets
Controlled feeding
For each diet, there was a 7-day menu cycle. Calorie intake was adjusted so that each participant maintained a constant body weight (to within 2% of individual baseline) during the entire study. Participants were weighed daily. All meals and snacks were prepared in research kitchens and provided to the participants. Participants were asked to maintain exercise frequency and intensity and to limit alcohol consumption to usual patterns.
Fasting blood samples were collected after an 8–12-h fast at baseline and at 4 and 6 weeks of each feeding period, for assessment of insulin and glucose levels. They were stored at −70°C. Glucose was measured using the enzymatic hexokinase kit from Roche on the Hitachi 917. Insulin was measured using microparticle enzyme immunoassay technology on the Abbott IMx analyzer. To enhance precision, we averaged the values taken at 4 and 6 weeks to produce one intervention value for each diet in each participant.
Indices of insulin sensitivity
We assessed insulin sensitivity by calculating the QUICKI (quantitative insulin check index) measure of insulin sensitivity (8). The equation for QUICKI, 1/{[log(fasting insulin µU/mL) + log(fasting glucose mg/dL)]}, uses log transformations of fasting glucose and insulin measurements and is a steady-state measure with a high correlation with the gold standard hyperinsulinemic-euglycemic glucose clamp (8). In a sensitivity analysis, homeostasis model assessment of insulin resistance (HOMA-IR), an alternate assessment of insulin sensitivity, was also calculated and reported as 1/HOMA, calculated as 1/[glucose (mmol/L) × insulin (μIU/L)/22.5] (11). As the linear slopes of HOMA change in relation to varying insulin sensitivities, the reciprocal and log transformation of HOMA inherent in QUICKI make it a more flexible measure, usable in normal weight, overweight, and the insulin-resistant state of obese individuals, and more appropriate for the range of BMI in our population (8,12–14). A higher numerical value for 1/HOMA or QUICKI reflects greater insulin sensitivity.
Statistical analysis
Statistical analysis was performed using linear regression analysis with generalized estimating equations with exchangeable correlations to account for repeated measures with robust estimation of the variance. The primary outcome measure is the change in QUICKI index, representing the difference between the index calculated at baseline and after each diet, respectively. We also modeled fasting glucose (mg/dL), fasting insulin (μIU/mL), and 1/HOMA in a similar fashion. The continuous variables for change in QUICKI were approximately normally distributed. There was a high correlation between QUICKI and 1/HOMA measures, with a Pearson correlation coefficient of 0.92. At baseline, we assessed correlations between QUICKI and traditional cardiovascular disease risk factors using Pearson correlation coefficients.
There were few data missing; only 4 out of 164 participants did not complete all three intervention diets. Only participants with complete data were included in the regression analysis. In the initial OmniHeart Trial analysis, there was a change in weight of 1 kg across the first period, although mean end-of-period weights were similar regardless of diet intervention. To account for any impact on insulin sensitivity from this change in weight and by period, we adjusted for feeding period. Between-diet comparisons were modeled as multiple linear regression analyses of the difference in change in QUICKI and 1/HOMA. To assess for carryover, we lagged exposure by one feeding period and found no evidence of a statistically significant carryover effect. We tested for interaction between diet and BMI, sex, race, age, and prediabetes status through interaction terms, which were insignificant for all stated variables. We then performed stratified analyses based on BMI category (normal, overweight, or obese) and clinical status of prediabetes or normoglycemia. As a sensitivity analysis, we modeled the first period in isolation to examine the effects without preceding intervention. Further sensitivity analyses demonstrated that outliers did not have a marked effect on our regression model.
Based on a fixed sample size (n = 160), we estimated minimal detectable differences in QUICKI of 0.001, assuming a significance level of 0.05 and a power of 80%. No adjustments were made for multiple testing. Primary analyses were performed using STATA version 11.1 (StataCorp, College Station, TX). Statistical significance was defined as P < 0.05.
RESULTS
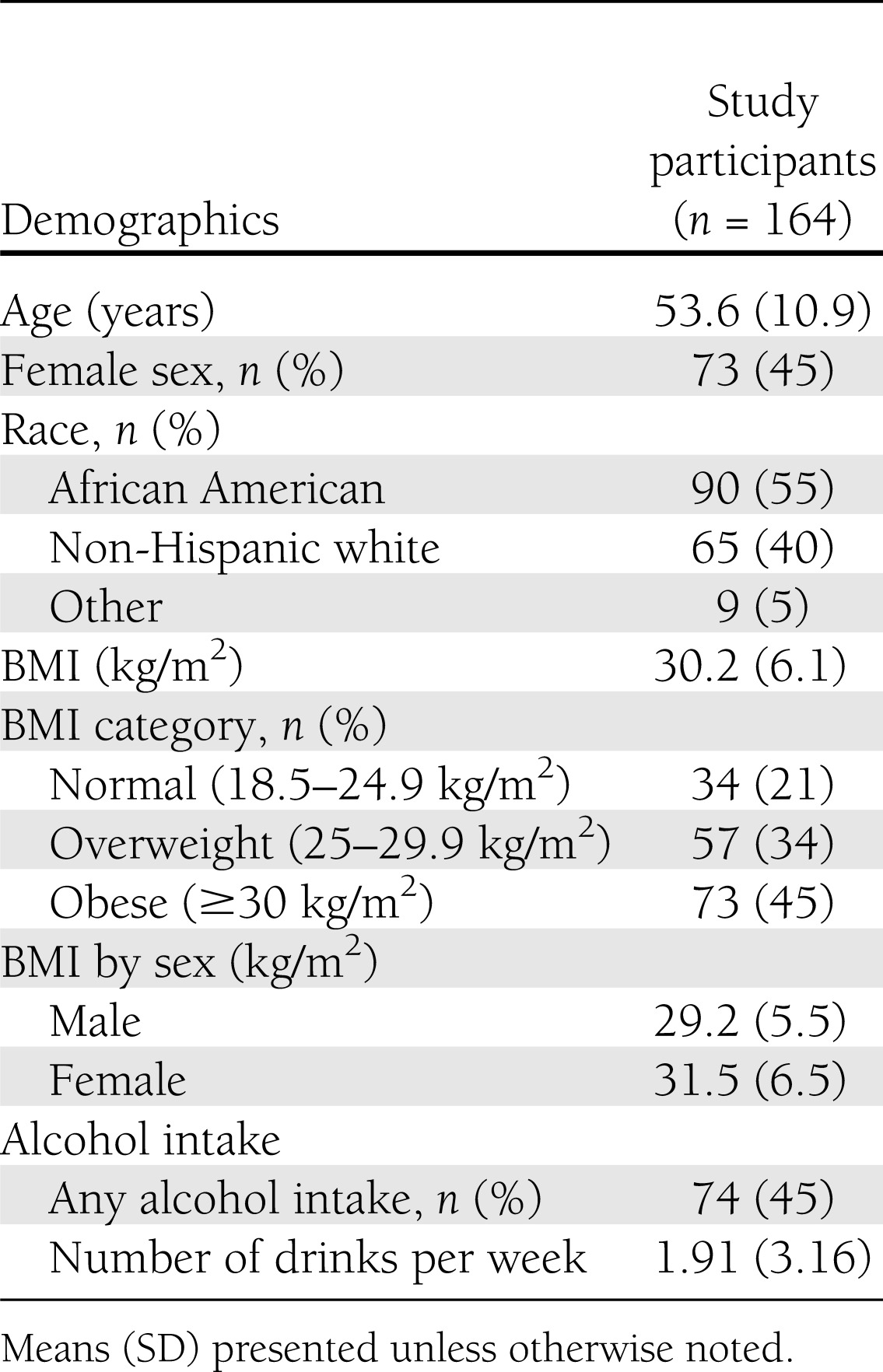
At baseline, the participants had a mean age of 54 years; 45% were women, 55% were African American, and 79% were overweight or obese. Mean BMI was 30.2 kg/m2, and mean QUICKI was 0.35. Other baseline characteristics are reported in Table 2.
Table 2.
Baseline characteristics of adults participating in the OmniHeart Trial
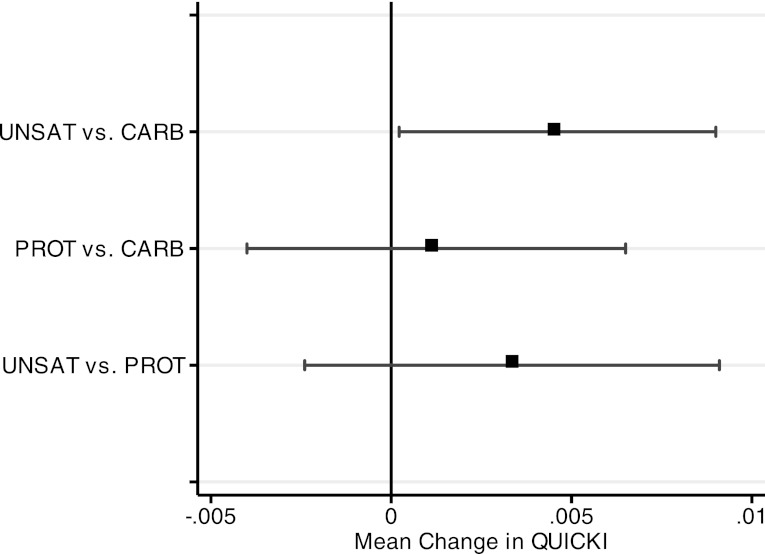
In between-diet comparisons, adjusted for period effects, the QUICKI index increased by a mean of 0.005 (95% CI 0.000–0.009) on the UNSAT diet, compared with the CARB diet (Fig. 1). The PROT diet showed no significant difference, compared with the CARB diet (−0.004 to 0.007) or the UNSAT diet (−0.009 to 0.002) (Table 3). 1/HOMA index also improved 0.11 (0.03–0.20) on the UNSAT diet as compared with the CARB diet. 1/HOMA did not significantly differ between the PROT and CARB (−0.07 to 0.14) or UNSAT and PROT diets (−0.05 to 0.20). There were no between-diet differences for glucose or insulin (Table 3).
Figure 1.
Between-diet comparisons of change in QUICKI measure. Change in QUICKI determined by generalized estimating equations. Error bars indicate 95% CIs.
Table 3.
Changes from baseline and between-diet changes in indices of insulin sensitivity for each diet*
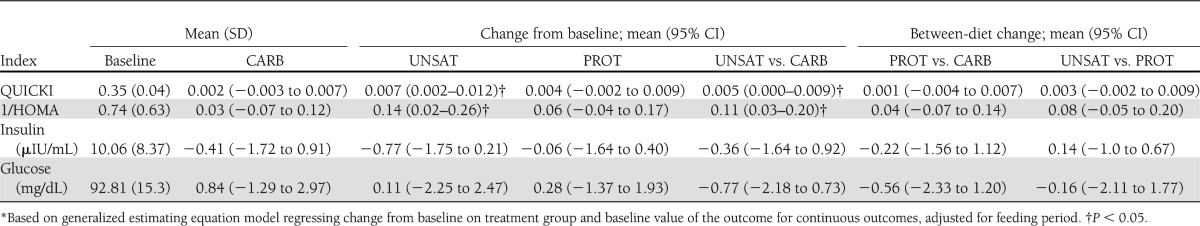
In models adjusting for feeding period (Table 3), the UNSAT diet increased QUICKI by 0.007 (95% CI 0.002–0.012) from baseline, but there was no change with the PROT diet (0.004 [−0.002 to 0.009]) or CARB diet (0.002 [−0.003 to 0.007]). 1/HOMA showed similar diet-specific responses with an increase of 0.14 (0.02–0.26) after consumption of the UNSAT diet, 0.06 (−0.002 to 0.009) after the PROT diet, and 0.04 (−0.003 to 0.007) after the CARB diet compared with baseline. Similar findings were seen in analyses not adjusted for feeding period.
At baseline, there was a strong direct correlation between QUICKI and HDL cholesterol (P < 0.001) and QUICKI and BMI (P < 0.0001), and an inverse correlation between QUICKI and triglyceride level (P < 0.001). The correlation between baseline QUICKI and systolic and diastolic blood pressure was present but less strong (P < 0.04). We found no significant correlation between change in QUICKI and change in weight, change in systolic or diastolic blood pressure, total, HDL, or LDL cholesterol, or triglycerides (not presented). The average weight decrease from baseline was similar across all three diets: mean (SD) 0.93 kg (2.05 kg) after intervention with the CARB diet, 0.90 kg (2.14 kg) after the UNSAT diet, and 1.12 kg (2.09 kg) after the PROT diet.
We adhered to conservative guidelines for analysis of subgroups (15), and as our interaction terms between diet and BMI, diabetes status, age, race, or sex were not significant, we did not highlight subgroup results in Table 3. When stratified by BMI, normal-weight participants experienced an increase in QUICKI with the UNSAT as compared with the CARB diet that trended toward significance (0.012, P = 0.06). There was no change in those who were overweight (0.007, P = 0.09) or obese (0.001, P = 0.83). In participants with prediabetes, defined as a fasting glucose between 100 and 125 mg/dL, choice of diet did not alter QUICKI (0.003, P = 0.48); however, in those who were normoglycemic, QUICKI trended toward significance with an increase of 0.005 (P = 0.06). There were no differences in the PROT versus CARB or UNSAT versus PROT comparisons.
CONCLUSIONS
In this trial that enrolled predominantly overweight and obese individuals, the UNSAT diet increased QUICKI and 1/HOMA to a greater degree than the CARB diet. The UNSAT diet increased the QUICKI index from baseline, whereas the CARB and PROT diets did not. The improvements in insulin sensitivity were both statistically significant and clinically relevant, and this comparative difference between three healthy diets has not previously been demonstrated.
The UNSAT diet is composed of 37% kilocalories from unsaturated fat, which is similar to Mediterranean-style diets, which typically include 28–40% of kilocalories from fat, mostly from unsaturated sources (16,17). These data suggest that an isocaloric diet similar in macronutrient proportions to Mediterranean-style diets may improve insulin sensitivity in patients without type 2 diabetes.
A key feature of this study is the examination of changes in insulin sensitivity by differing macronutrient intake while weight was held constant. The association of weight loss among overweight or obese individuals with increases in insulin sensitivity has been extensively studied (18–20). For example, in an analysis of participants in the Women’s Health Initiative, a similar increase in QUICKI to what was observed on the UNSAT diet in our results was seen in women who lost an average of 1 kg of body weight (21). In the OmniHeart Trial, weight change during the three feeding periods was small and not significantly different between intervention diets. Thus, the results suggest that reducing carbohydrate while increasing unsaturated fat in a healthy diet improves insulin sensitivity in the setting of stable weight. Interestingly, there was no corresponding improvement when participants consumed the reduced-carbohydrate, increased-protein diet. Hence, the improvement in insulin sensitivity on UNSAT appears to result from an increase in unsaturated fat rather than a decrease in carbohydrate.
We chose QUICKI as our primary measure of insulin sensitivity. Both HOMA and QUICKI indices have been validated as methods to assess insulin sensitivity in comparison with the gold standard hyperinsulinemic-euglycemic glucose clamp (8,11). Although both are widely used, the log transformations of fasting insulin and glucose inherent in the construction of QUICKI allow for a better correlation with the gold standard over a wider distribution of fasting insulin values, in both obese and hypertensive patients (8,12,22). There is evidence to suggest that β-cell dysfunction may occur prior to changes in fasting insulin or glucose levels (23). Changes in insulin sensitivity revealed by the 1/HOMA and QUICKI indices may precede clinical diagnosis and allow for earlier detection, and subsequent intervention, of altered glucose-insulin homeostasis.
Investigations into the effects of unsaturated fat– or carbohydrate-rich diets on glycemic control have yielded conflicting results. An isocaloric feeding trial of 720 participants demonstrated no differences in insulin sensitivity after replacing saturated fatty acids with monounsaturated fats or carbohydrates, regardless of the glycemic index of the diet (24). In this trial, weight was not held constant throughout the feeding periods. A second randomized trial, comparing a standard carbohydrate-rich diet with one high in fat and reduced carbohydrates, measured both insulin sensitivity and β-cell function. It suggested that a higher-fat, lower-carbohydrate diet may actually lower β-cell responsiveness, whereas dynamic insulin sensitivity measurements remained unaffected (25). Possible reasons for the difference in results are fewer total participants who had a higher average BMI than in our analysis and who had a significant increase in their fasting glucose measurements.
Our trial tested moderate, achievable differences in macronutrients, rather than more extreme changes, such as carbohydrate elimination or ketogenic diets. A high-fat, low-protein, very-low-carbohydrate diet has attracted considerable attention, particularly in the lay press. Although often leading to substantial weight loss, the ketogenic diet, as an extreme of fat-predominant macronutrient composition, may actually be detrimental to overall insulin sensitivity. In animal studies, dynamic measures of insulin sensitivity have shown an increase in hepatic insulin resistance after consumption of ketogenic diets (26,27).
Nutrients, other than carbohydrates and unsaturated fat, and behavioral factors might influence insulin sensitivity. Saturated fatty acids decrease responsiveness of the cell membrane to insulin-mediated actions through a decrease in binding affinity, which contributes to increased insulin resistance (28). High protein intake, especially from animal-based sources, has been associated with an increase in type 2 diabetes risk (29–31). The European Prospective Investigation into Cancer and Nutrition (EPIC-NL) study, a large, prospective investigation of 38,094 participants, suggests a 30% increase in type 2 diabetes risk with consumption of five energy percent of protein in the place of fat or carbohydrate (32). The same association was not seen for vegetable protein. Although the PROT diet in our study did not exceed 25% kilocalories of total calories from protein, the increase in both vegetable and animal sources of protein may be responsible for the null effect observed in our study. All diets in the OmniHeart Trial were high in fiber, with a nutrient target of >30 g per day in each diet. Physical activity and alcohol consumption were constant for each participant throughout the trial.
Impaired insulin sensitivity in the obese may attenuate substantial changes in insulin sensitivity brought about by changes in macronutrient intake (33). In our subgroup analyses stratified by weight category, we found that change in QUICKI after consumption of the UNSAT diet was most pronounced in participants of normal weight, and the effect decreases as weight increases. Similarly, participants adhering to the Mediterranean diet in the European Prospective Investigation into Cancer and Nutrition (EPIC-NL) cohort demonstrated slightly decreased type 2 diabetes risk as compared with those with low adherence; however, this association was less robust for those who were obese and <50 years of age (34). It is likely that the choice of macronutrient to prevent insulin resistance and type 2 diabetes is less important than overall weight loss in the obese, whereas partial replacement of carbohydrates by unsaturated fat intake can mitigate risk in those of normal weight. In light of recent evidence suggesting that diabetes in those of normal weight leads to higher mortality (35), altering macronutrient intake may be another tool in a limited arsenal to combat risk of cardiovascular disease and diabetes in those of normal weight.
The main OmniHeart Trial evaluated these three healthy diets differing in macronutrient composition and their comparative effects on blood pressure and lipids (7). Compared with the CARB diet, which lowered blood pressure and cholesterol from baseline, consumption of the PROT and UNSAT diets further reduced systolic and diastolic blood pressure, and the PROT diet resulted in the greatest reduction in LDL cholesterol. Additionally, compared with the CARB diet, there was a greater reduction in triglycerides with consumption of both the PROT and UNSAT diets, and the UNSAT diet comparatively increased HDL. In a secondary analysis of this trial, the PROT diet reduced harmful apolipoprotein C-III more than the CARB diet and was found to be more effective in white participants than black participants (36,37). Another analysis reported associations between the UNSAT diet and improvement of high-molecular-weight and total adiponectin when compared with the CARB or PROT diets (38). Taken together, these findings suggest that partial replacement of carbohydrates beneficially affects three major components of the metabolic syndrome: systolic blood pressure, triglycerides, and HDL cholesterol. In this context, the increases in insulin sensitivity with the UNSAT diet demonstrated with our investigation may serve as further evidence for its place in the prevention of type 2 diabetes and the metabolic syndrome.
These results should be viewed in the context of nutrition recommendations and usual dietary intake. Although the American Diabetes Association does not recommend a low-carbohydrate diet, other institutes prominent in diabetes care do advocate for carbohydrate composition of 40%, with replacement of a portion of carbohydrate with unsaturated fat or protein (39). The average carbohydrate consumption in the U.S., as collected by the 2005–2006 National Health and Nutrition Examination Survey, was ∼48% for males and 50% for females (40). In comparison, the OmniHeart CARB diet provided 58% of kilocalories from carbohydrate, and the UNSAT diet provided 48% of kilocalories from carbohydrate.
Our study does have limitations. First, the period on each diet was only 6 weeks. However, prior feeding studies by our group have shown that blood pressure and lipid responses occur quickly, within 2–4 weeks of a dietary change. Second, we assessed insulin sensitivity through the use of fasting indices, rather than the gold standard physiologic clamps. In the absence of dynamic measurements, we have several measures of insulin sensitivity, and QUICKI appears the best suited for our population (8,14,22). Third, insulin sensitivity is only a surrogate outcome; correlations with clinical outcomes such as incident type 2 diabetes or clinical complications related to insulin sensitivity were not assessed. The strengths of our trial remain that it is a large, randomized, tightly controlled feeding study that enrolled a diverse population. Our subgroup analyses, although provocative, rely on numbers of participants that are too small to effectively draw bold conclusions, and the assessments for interaction do not meet the criteria for statistical significance.
In conclusion, our analysis suggests that a diet rich in unsaturated fats, which is commonplace in Mediterranean-style diets, improves insulin sensitivity in a population at risk for cardiovascular disease. Given the well-recognized challenges of sustaining weight loss, our results suggest an alternative approach to improving insulin sensitivity.
Acknowledgments
Funding for this study was provided by grants from the National Heart, Lung, and Blood Institute (T32-HL-007180) and the Optimal Macronutrient Intake to Prevent Heart Disease (HL-67098, DK-63214, HL-68712, and RR-02635) from the National Institutes of Health. E.H.Y. was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health.
No potential conflicts of interest relevant to this article were reported.
M.D.G. performed statistical analysis and wrote the manuscript. L.J.A., F.M.S., and E.R.M. were involved in the study concept and design of the original trial, contributed to the discussion, and reviewed and edited the manuscript. E.Y. and C.A.M.A. contributed to the discussion and reviewed and edited the manuscript. E.R.M. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
This paper was presented as an oral abstract at the 2011 Scientific Sessions of the American Heart Association, Orlando, Florida, 12–16 November 2011.
The authors thank Mark Van Natta (Johns Hopkins Bloomberg School of Public Health) for his advice on statistical analysis.
Footnotes
Clinical trial reg. no. NCT00051350, clinicaltrials.gov.
References
- 1.Nolan CJ, Damm P, Prentki M. Type 2 diabetes across generations: from pathophysiology to prevention and management. Lancet 2011;378:169–181 [DOI] [PubMed] [Google Scholar]
- 2.Mokdad AH, Ford ES, Bowman BA, et al. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA 2003;289:76–79 [DOI] [PubMed] [Google Scholar]
- 3.Milman S, Crandall JP. Mechanisms of vascular complications in prediabetes. Med Clin North Am. 2011;95:309–325, vii [DOI] [PubMed]
- 4.Reaven GM. The role of insulin resistance and hyperinsulinemia in coronary heart disease. Metabolism 1992;41(Suppl. 1):16–19 [DOI] [PubMed] [Google Scholar]
- 5.Zavaroni I, Bonora E, Pagliara M, et al. Risk factors for coronary artery disease in healthy persons with hyperinsulinemia and normal glucose tolerance. N Engl J Med 1989;320:702–706 [DOI] [PubMed] [Google Scholar]
- 6.Wing RR, Look AHEAD Research Group Long-term effects of a lifestyle intervention on weight and cardiovascular risk factors in individuals with type 2 diabetes mellitus: four-year results of the Look AHEAD trial. Arch Intern Med 2010;170:1566–1575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Appel LJ, Sacks FM, Carey VJ, et al. OmniHeart Collaborative Research Group Effects of protein, monounsaturated fat, and carbohydrate intake on blood pressure and serum lipids: results of the OmniHeart randomized trial. JAMA 2005;294:2455–2464 [DOI] [PubMed] [Google Scholar]
- 8.Katz A, Nambi SS, Mather K, et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 2000;85:2402–2410 [DOI] [PubMed] [Google Scholar]
- 9.Carey VJ, Bishop L, Charleston J, et al. Rationale and design of the optimal macro-nutrient intake heart trial to prevent heart disease (OMNI-heart). Clin Trials 2005;2:529–537 [DOI] [PubMed] [Google Scholar]
- 10.Appel LJ, Moore TJ, Obarzanek E, et al. DASH Collaborative Research Group A clinical trial of the effects of dietary patterns on blood pressure. N Engl J Med 1997;336:1117–1124 [DOI] [PubMed] [Google Scholar]
- 11.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–419 [DOI] [PubMed] [Google Scholar]
- 12.Muniyappa R, Lee S, Chen H, Quon MJ. Current approaches for assessing insulin sensitivity and resistance in vivo: advantages, limitations, and appropriate usage. Am J Physiol Endocrinol Metab 2008;294:E15–E26 [DOI] [PubMed] [Google Scholar]
- 13.Chen H, Sullivan G, Yue LQ, Katz A, Quon MJ. QUICKI is a useful index of insulin sensitivity in subjects with hypertension. Am J Physiol Endocrinol Metab 2003;284:E804–E812 [DOI] [PubMed] [Google Scholar]
- 14.Cheng C, Campbell KL, Kushner H, Falkner BE. Correlation of oral glucose tolerance test-derived estimates of insulin sensitivity with insulin clamp measurements in an African-American cohort. Metabolism 2004;53:1107–1112 [DOI] [PubMed] [Google Scholar]
- 15.Wang R, Lagakos SW, Ware JH, Hunter DJ, Drazen JM. Statistics in medicine—reporting of subgroup analyses in clinical trials. N Engl J Med 2007;357:2189–2194 [DOI] [PubMed] [Google Scholar]
- 16.Trichopoulou A, Costacou T, Bamia C, Trichopoulos D. Adherence to a Mediterranean diet and survival in a Greek population. N Engl J Med 2003;348:2599–2608 [DOI] [PubMed] [Google Scholar]
- 17.Willett WC, Sacks F, Trichopoulou A, et al. Mediterranean diet pyramid: a cultural model for healthy eating. Am J Clin Nutr 1995;61(Suppl.):1402S–1406S [DOI] [PubMed] [Google Scholar]
- 18.Tuomilehto J, Lindström J, Eriksson JG, et al. Finnish Diabetes Prevention Study Group Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001;344:1343–1350 [DOI] [PubMed] [Google Scholar]
- 19.Knowler WC, Barrett-Connor E, Fowler SE, et al. Diabetes Prevention Program Research Group Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393–403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.LeRoith D. Dyslipidemia and glucose dysregulation in overweight and obese patients. Clin Cornerstone 2007;8:38–52 [DOI] [PubMed] [Google Scholar]
- 21.Shikany JM, Margolis KL, Pettinger M, et al. Effects of a low-fat dietary intervention on glucose, insulin, and insulin resistance in the Women’s Health Initiative (WHI) Dietary Modification trial. Am J Clin Nutr 2011;94:75–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen H, Sullivan G, Quon MJ. Assessing the predictive accuracy of QUICKI as a surrogate index for insulin sensitivity using a calibration model. Diabetes 2005;54:1914–1925 [DOI] [PubMed] [Google Scholar]
- 23.Matthews DR. Insulin resistance and beta-cell function—a clinical perspective. Diabetes Obes Metab 2001;3(Suppl. 1):S28–S33 [PubMed] [Google Scholar]
- 24.Jebb SA, Lovegrove JA, Griffin BA, et al. RISCK Study Group Effect of changing the amount and type of fat and carbohydrate on insulin sensitivity and cardiovascular risk: the RISCK (Reading, Imperial, Surrey, Cambridge, and Kings) trial. Am J Clin Nutr 2010;92:748–758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Goree LL, Chandler-Laney P, Ellis AC, Casazza K, Granger WM, Gower BA. Dietary macronutrient composition affects β cell responsiveness but not insulin sensitivity. Am J Clin Nutr 2011;94:120–127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jornayvaz FR, Jurczak MJ, Lee HY, et al. A high-fat, ketogenic diet causes hepatic insulin resistance in mice, despite increasing energy expenditure and preventing weight gain. Am J Physiol Endocrinol Metab 2010;299:E808–E815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lan-Pidhainy X, Wolever TM. The hypoglycemic effect of fat and protein is not attenuated by insulin resistance. Am J Clin Nutr 2010;91:98–105 [DOI] [PubMed] [Google Scholar]
- 28.Manco M, Calvani M, Mingrone G. Effects of dietary fatty acids on insulin sensitivity and secretion. Diabetes Obes Metab 2004;6:402–413 [DOI] [PubMed] [Google Scholar]
- 29.Micha R, Wallace SK, Mozaffarian D. Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: a systematic review and meta-analysis. Circulation 2010;121:2271–2283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Risérus U. Fatty acids and insulin sensitivity. Curr Opin Clin Nutr Metab Care 2008;11:100–105 [DOI] [PubMed] [Google Scholar]
- 31.Aune D, Ursin G, Veierød MB. Meat consumption and the risk of type 2 diabetes: a systematic review and meta-analysis of cohort studies. Diabetologia 2009;52:2277–2287 [DOI] [PubMed] [Google Scholar]
- 32.Sluijs I, Beulens JW, van der A DL, Spijkerman AM, Grobbee DE, van der Schouw YT. Dietary intake of total, animal, and vegetable protein and risk of type 2 diabetes in the European Prospective Investigation into Cancer and Nutrition (EPIC)-NL study. Diabetes Care 2010;33:43–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seino S, Shibasaki T, Minami K. Dynamics of insulin secretion and the clinical implications for obesity and diabetes. J Clin Invest 2011;121:2118–2125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Romaguera D, Guevara M, Norat T, et al. InterAct Consortium Mediterranean diet and type 2 diabetes risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study: the InterAct project. Diabetes Care 2011;34:1913–1918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Carnethon MR, De Chavez PJ, Biggs ML, et al. Association of weight status with mortality in adults with incident diabetes. JAMA 2012;308:581–590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Furtado JD, Campos H, Appel LJ, et al. Effect of protein, unsaturated fat, and carbohydrate intakes on plasma apolipoprotein B and VLDL and LDL containing apolipoprotein C-III: results from the OmniHeart Trial. Am J Clin Nutr 2008;87:1623–1630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Furtado JD, Campos H, Sumner AE, Appel LJ, Carey VJ, Sacks FM. Dietary interventions that lower lipoproteins containing apolipoprotein C-III are more effective in whites than in blacks: results of the OmniHeart trial. Am J Clin Nutr 2010;92:714–722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yeung EH, Appel LJ, Miller ER, 3rd, Kao WH. The effects of macronutrient intake on total and high-molecular weight adiponectin: results from the OMNI-Heart trial. Obesity (Silver Spring) 2010;18:1632–1637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Acheson KJ. Carbohydrate for weight and metabolic control: where do we stand? Nutrition 2010;26:141–145 [DOI] [PubMed] [Google Scholar]
- 40.Austin GL, Ogden LG, Hill JO. Trends in carbohydrate, fat, and protein intakes and association with energy intake in normal-weight, overweight, and obese individuals: 1971-2006. Am J Clin Nutr 2011;93:836–843 [DOI] [PubMed] [Google Scholar]