Abstract
OBJECTIVE
Distal sensorimotor polyneuropathy (DSPN) is a severe complication of type 2 diabetes. This study aimed to assess the prevalence of unawareness of DSPN in prediabetes and diabetes in a sample of the older population of Augsburg, Germany.
RESEARCH DESIGN AND METHODS
Glucose tolerance status was determined in 61- to 82-year-old participants of the population-based KORA F4 Study (2006–2008) (n = 1,100). Clinical DSPN was defined as the presence of bilaterally impaired foot-vibration perception and/or bilaterally impaired foot-pressure sensation. DSPN case subjects were considered unaware of their condition when answering “no” to the question, “Has a physician ever told you that you are suffering from nerve damage, neuropathy, polyneuropathy, or diabetic foot?”
RESULTS
Clinical DSPN was prevalent in 154 (14%) participants, 140 of whom were unaware of their disorder. At a prevalence of 23.9% (95% CI 12.6–38.8), participants with combined impaired fasting glucose and impaired glucose tolerance had the highest prevalence of DSPN. Of these, 10 of 11 (91%) were unaware of having clinical DSPN. Participants with known diabetes had an equally high prevalence of DSPN [22.0% (16.2–28.9)], with 30 of the 39 (77%) DSPN case subjects unaware of having the disorder. Among subjects with known diabetes who reported to have had their feet examined by a physician, 18 of 25 (72%) clinical DSPN case subjects emerged unaware of having DSPN.
CONCLUSIONS
Our findings showed a high prevalence of unawareness of having clinical DSPN among the prediabetic and diabetic groups and an insufficient frequency of professional foot examinations, suggesting inadequate attention to diabetic foot prevention practice.
Diabetic peripheral neuropathy is a severe complication of type 2 diabetes related to chronic hyperglycemia and the presence of cardiovascular risk factors (1). Symmetrical distal sensorimotor polyneuropathy (DSPN), the most common form of peripheral neuropathies in patients with diabetes, is a heterogeneous disorder covering a wide range of abnormalities that affect peripheral sensory and motor nerves as well as the autonomic nervous system (2). Of all diabetes complications, DSPN is responsible for the highest number of hospital admissions and, being the foremost cause of foot ulcers, for 50–75% of all nontraumatic amputations after ulceration (3). Next to substantial morbidity, DSPN leads to reduced quality of life and an increased risk of mortality (1,4).
In recognition of the importance of early detection and prevention of DSPN, American (5), British (6), and German (7) national guidelines for diabetes care state that all patients with type 2 diabetes should be screened for clinical DSPN at the time of their diabetes diagnosis and yearly thereafter. Screening is to be performed using simple clinical tests, such as vibration perception, pressure sensation, assessment of ankle reflexes, and pinprick sensation. Still, several reports have indicated that in primary care practice, where most of the diabetic patients are being treated, screening for polyneuropathy was underused (8–11). Neurologic tests and physical examination of the feet are being carried out rarely in asymptomatic diabetic patients, and neuropathic pain often remains unrecognized and untreated (12). To date, there are only sparse data on the prevalence of undiagnosed DSPN (13,14). Although the two studies differed in methodology, both observed that over one-half of their study sample of diabetic patients had undiagnosed DSPN. The aim of the current study was to examine the prevalence of unawareness of having clinical DSPN among older prediabetic and diabetic individuals from a population-based sample in Germany.
RESEARCH DESIGN AND METHODS
The Cooperative Health Research in the Region of Augsburg (KORA) was initiated to study the prevalence and incidence of various chronic diseases in the general population, including diabetes, and to identify novel risk factors of these diseases. The current study is based on the follow-up examination of the KORA S4 Survey that was conducted in 1999–2001. The study design and subject enrollment have previously been described in detail (15). Briefly, 2,656 men and women aged 55–74 years were randomly selected from the region of Augsburg in the south of Germany to participate in the KORA S4 Survey. From the 2,564 eligible subjects, 1,653 (64%) completed the survey and a subsequent 1,353 subjects without known diabetes successfully completed an oral glucose tolerance test (OGTT). In 2006–2008, the 7-year follow-up examination (F4 Survey) of this cohort took place, including a second OGTT. Of the initial 1,353 subjects, a total of 1,209 (89%) participated in the follow-up measurements. For 177 participants, a previous diagnosis of diabetes could be validated, and a further 923 participants successfully completed the OGTT, resulting in a total sample size of 1,100 (81%) subjects. All participants gave written informed consent, and the study was approved by the ethics committee of the Bavarian Medical Association.
Measurements and interviews
Height, weight, waist circumference, and systolic and diastolic blood pressure were measured according to standard protocols as previously described (15). Trained medical interviewers collected information on medical history, physical activity, smoking behavior, and alcohol consumption. Furthermore, participants with known diabetes were asked the question, “When has a physician examined your feet lately?” which could be answered with 1) within the past 12 months, 2) >12 months ago, 3) not ever, and 4) I don’t know. As an indication for ever having had one’s feet examined, answers 1 and 2 were considered confirmative of a foot examination and answer 3 was considered nonconfirmative.
Patients with known diabetes completed an additional self-administered questionnaire on diabetes care, which inquired about the presence of complications, the course of treatment, and whether the subject had been enrolled in a type 2 diabetes disease-management program (DDMP). Since DDMP names vary considerably between the supplying social health insurance companies and might not reveal the disease-management aspect, the family physician of each participant with known diabetes was contacted to validate DDMP participation.
Assessment of glucose metabolism
Cases of self-reported diabetes, as well as the date of diagnosis, were validated through contacting the participants’ general practitioners. All other participants underwent an OGTT (World Health Organization criteria). After an overnight fasting period of at least 10 h, fasting blood samples were taken and participants were given an oral dose of 75 g anhydrous glucose (Dextro OGT; Boehringer Mannheim, Mannheim, Germany). Another blood sample was collected 2 h after the glucose load. Blood samples were collected without stasis. After withdrawal, the samples were centrifuged and refrigerated at 4°C until analysis in the central laboratory of the Augsburg Central Hospital at maximum 4 h after withdrawal. Blood glucose levels were assessed using the hexokinase method (Glu-Flex; Dade Behring, Marburg, Germany). Glucose tolerance categories were defined according to the 1999 World Health Organization diagnostic criteria (16). We considered participants with isolated impaired fasting glucose (IFG), isolated impaired glucose tolerance (IGT), or combined IFG-IGT as subjects with prediabetes. Furthermore, overall diabetes was defined as the combined group of subjects with known and newly diagnosed diabetes. It can be assumed that the great majority of case subjects with newly diagnosed diabetes in this age-group had type 2 diabetes.
Neurologic assessment
The neurologic examination of the F4 survey consisted of two parts. The first involved a detailed interview addressing the presence of (current) pain in the feet and other parts of the body, the presence of neurologic diseases, and the participant’s history of foot ulcers and amputations. The second part comprised a foot inspection and a series of neurologic tests involving sensation to touch, vibration, and temperature and testing of ankle reflexes and sudomotor function.
We defined the presence of clinical DSPN as bilateral impairment of foot-vibration perception and/or bilateral impairment of foot-pressure sensation. Vibration perception was assessed at the dorsal side of the left and right big toe, using a calibrated 64-Hz Rydel Seiffer tuning fork. Increased thresholds were calculated according to the study of Martina et al. (17). Pressure sensation was measured at the dorsal side of the left and right big toe in between the nail fold and the metatarsophalangeal joint, using a 10-g monofilament (Twin-Tip, Heinsberg, Germany). Participants were asked to close their eyes during the test and to respond with “yes” each time the monofilament was sensed. No negative stimuli were tested. At least 8 of 10 correct responses were considered to indicate normal sensibility (18). Less than eight perceived applications indicated reduced sensibility, and when none of the applications were perceived, sensibility to touch was considered absent. Measurements of vibration perception and pressure sensation were performed by trained investigators under supervision of an experienced diabetologist (19) and according to the practical guidelines for the diabetic foot from the American Diabetes Association and the International Diabetic Foot Working Group (5,20). Our choice for these two specific tests lies in their quantitative nature to detect the insensate foot and the fact that both tests are predictors of future foot ulceration (21). Also, the two tests have previously been studied as being the most accurate tools for diagnosing large-fiber polyneuropathy in patients with diabetes. We have validated our clinical DSPN definition against nerve conduction studies as previously described (19).
During the interview on possible neurologic complaints, before undergoing the foot inspection and the neurologic testing, participants were asked whether a physician had ever told them that they were suffering from nerve damage, neuropathy, polyneuropathy, or diabetic foot. We defined subjects with clinical DSPN as being unaware of their disorder if they had answered this question with “no.”
Statistical analysis
Follow-up characteristics are presented as means ± SD for normally distributed variables and as median (interquartile range) for variables without a normal distribution. Age- and sex-adjusted differences in characteristics were evaluated for participants with clinical DSPN who were either aware or unaware of their disorder using ANOVA. For log-normal variables, ANOVA was performed on a log scale. P values <0.05 were considered to indicate statistical significance. Analyses were performed with the STATA statistical software package (version 11; Stata).
RESULTS
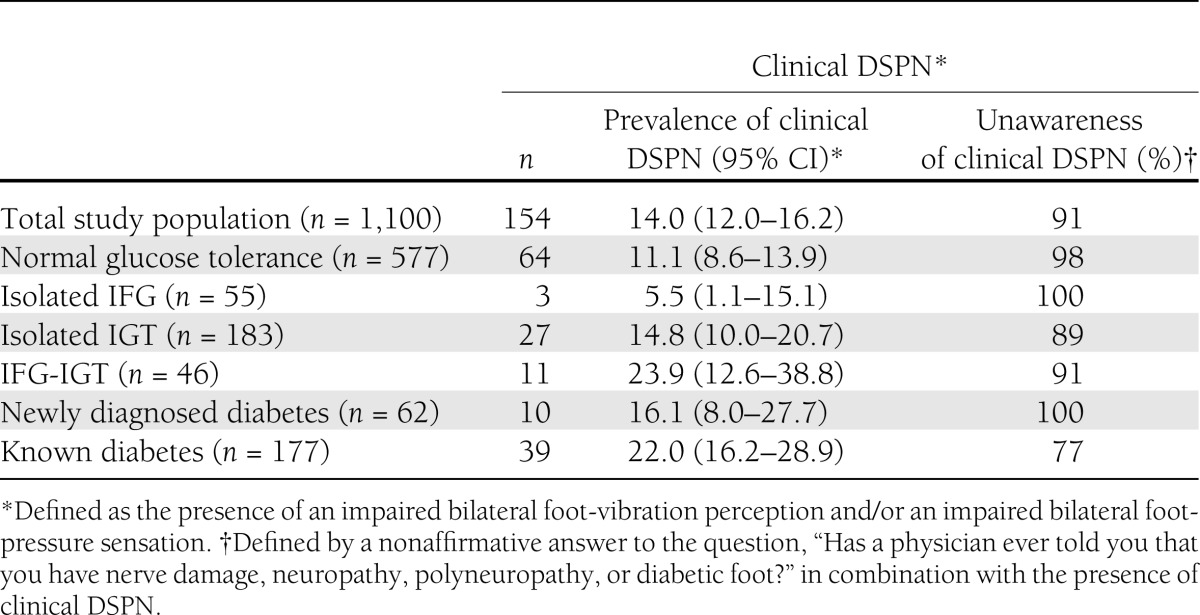
The KORA F4 Survey comprised a total of 1,100 participants with complete information on the presence of clinical DSPN, glucose tolerance status, and other covariables. According to our definition, clinical DSPN was present in 154 (14%) subjects, only 14 (9%) of whom were classified as being aware of their disorder and as many as 140 (91%) as being unaware. The prevalence of clinical DSPN and subsequent unawareness of having the disorder is presented in Table 1 according to glucose tolerance status. Participants with IFG-IGT and with known diabetes had the highest prevalence of clinical DSPN [23.9% (95% CI 12.6–38.8) and 22.0% (16.2–28.9), respectively]. Among those with IFG-IGT, 10 of 11 (91%) case subjects with clinical DSPN were unaware of having the disorder. This proportion was only slightly lower among those with known diabetes, among whom 30 of 39 (77%) case subjects with clinical DSPN were unaware of having the disorder.
Table 1.
Prevalence of clinical DSPN according to glucose tolerance status: KORA F4 (2006–2008)
In Table 2, differences in characteristics are presented between participants who were aware of having clinical DSPN and those who were unaware. Compared with the latter, subjects who were aware of their disorder had on average a higher systolic blood pressure and were more likely to have known diabetes. No differences between the two groups were observed for the prevalence of neurologic diseases, the prevalence of prediabetes, foot examinations, and DDMP participation (the latter two variables were only available for participants with known diabetes). With regard to neurologic characteristics, participants aware of having clinical DSPN more often had complaints of pain, paresthesias, and numbness in the feet over the last 24 h; foot ulcers; and absent ankle reflexes. The continuous monofilament test scores showed that clinical DSPN was more severe in subjects aware of having clinical DSPN as indicated by lower test scores compared with those who were unaware of having the disorder. The continuous tuning fork test scores indicated this as well; yet, only borderline statistical significance was reached.
Table 2.
Characteristics of KORA F4 participants according to awareness and unawareness of having clinical DSPN: KORA F4 (2006–2008)

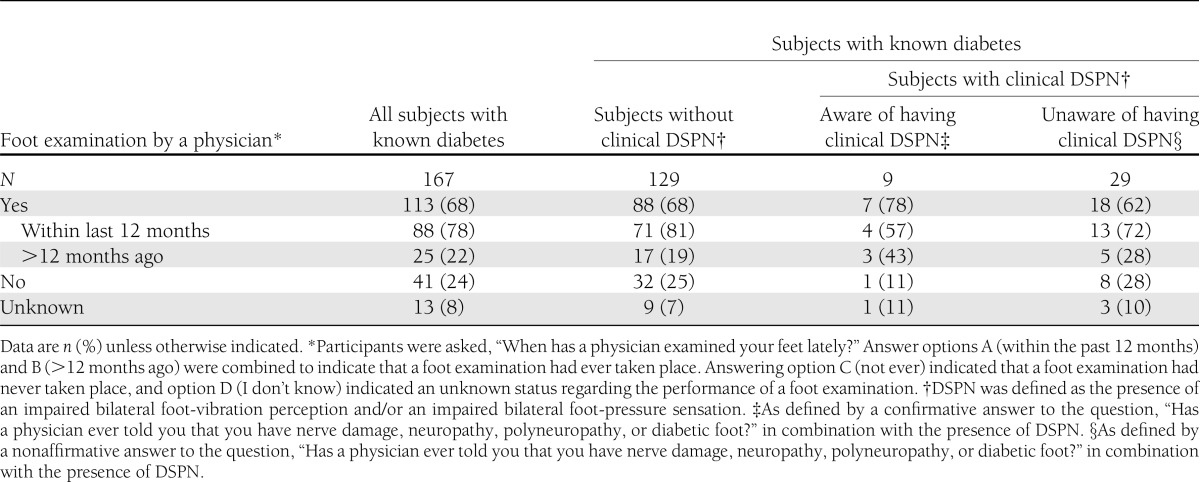
Information on the performance of a foot examination by a physician was assessed for participants with known diabetes only (n = 177). Excluding those with missing data (n = 10), 113 of 167 (68%) subjects with known diabetes reported to have ever had their feet examined by a physician (Table 3). Of these foot examinations, 88 of 113 (78%) had taken place within the last 12 months and 25 of 113 (22%) had been performed >12 months ago. Approximately one-quarter of the subjects with known diabetes had never undergone a foot examination, and 13 (8%) could not remember. In total, 38 patients with known diabetes had clinical DSNP according to our definition, 29 (76%) of whom were unaware of having the disorder. Eighteen of these 29 (62%) subjects indicated having ever undergone a foot examination by a physician, whereas 8 stated to have never had their feet examined. Thirteen of the 18 (72%) foot examinations had taken place within the last 12 months and 5 (18%) >12 months ago. Of the nine case subjects aware of having clinical DSPN, a foot examination had been performed in seven.
Table 3.
Performance of a foot examination by a physician on participants with known diabetes: KORA F4 (2006–2008)
According to the German National Disease Management Guidelines for neuropathy in adults with diabetes, individuals with diabetes should be screened for DSPN at diagnosis and yearly thereafter (7). Of the participants with available information on diabetes duration (n = 154), only 11 of 24 (46%) with a duration ≤1 year reported having ever undergone a foot examination by a physician. Of the 49 participants with a diabetes duration of ≤5 years, 30 (61%) indicated having ever had their feet examined, and this was also true for 75 of 105 (71%) subjects diagnosed with diabetes >5 years ago.
CONCLUSIONS
The results of this cross-sectional population-based study demonstrated that a large proportion of subjects with prediabetes and diabetes were unaware of having clinical DSPN. While 113 of 167 (68%) participants with known diabetes had ever undergone a foot examination by a physician, only 7 of the 25 (28%) with clinical DSPN were aware of having clinical DSPN, whereas 18 of these 25 (72%) were unaware of having the disorder. Overall, the majority of the reported foot examinations had been carried out within the preceding 12 months.
The prevalence of unawareness of having clinical DSPN in participants with newly diagnosed diabetes was high. Yet, since these subjects were not receiving regular professional diabetes care, a high proportion of unaware DSPN cases was to be expected. Unexpected, though, was the high prevalence of unawareness of having DSPN among participants with known diabetes. Since these subjects are receiving regular diabetes care, the large proportion of unaware case subjects suggests that the current diabetes care practice may have serious shortcomings concerning appropriate attention to foot care. Next to subjects with diabetes, participants with IFG-IGT showed a strikingly high prevalence of unawareness of having clinical DSPN. In a previous report, we showed that IFG-IGT represents a high-risk group for developing DSPN (19). And since clinical DSPN is a strong risk predictor for the subsequent development of diabetic foot ulcers (21), these prediabetic individuals may also benefit from receiving preventive foot care.
To date, there are only two publications on the prevalence of undiagnosed DSPN in patients with known type 2 diabetes (13,14). Although a direct comparison of results is hampered by differences in study design, sample size, and assessment of undiagnosed DSPN, the main finding of the two is concordant: DSPN was underdiagnosed in over one-half of the diabetic patients. Wang et al. (14) have speculated that the underdiagnosis in their population sample might be the result of the low number of foot examinations performed by a health professional, since only 16.2% of their study sample had received preventive foot care. Herman and Kennedy did not have data on foot examinations (13). It is known that a large proportion of diabetic foot complications are preventable and that regular foot examinations by a general practitioner, a physician, or other health care providers play an important role in prevention (5). As such, a number of studies have reported on (frequency of) preventive foot care in individuals with diabetes (8,11,14,22–25). An Australian population-based cohort study on diabetes care practice reported that only 50% of the 396 participants with known type 2 diabetes had received a foot examination by a health professional within the last 12 months (11). Of those who were classified as being at risk for a future foot ulcer, only 46 of 81 (57%) reported to have had a foot examination. Another large cohort study of 3,564 patients with type 2 diabetes randomly selected from outpatient clinics and general practitioners found similar results (8). As many as 50% of these patients reported not to have had their feet examined in the last 12 months. And among the patients with symptomatic neuropathy or peripheral vascular disease—both risk factors of foot complications—over one-third had not undergone a foot examination. In summary, the general picture sketched by the previous literature on diabetes care is that attention to foot complications was poor and that a large proportion of patients were not offered regular foot examinations—not even those at high risk for developing foot complications. Whereas our data on preventive foot care are less elaborate, our findings fit this picture of insufficient and inadequate practice of preventive foot care. In addition, case subjects aware of having clinical DSPN showed a significantly higher proportion of absent ankle reflexes and foot ulcers compared with case subjects who were unaware of having DSPN (Table 2). Whereas there were no differences between the two groups concerning abnormal test scores on monofilament and tuning fork tests (data not shown), we can only speculate that overall, ankle reflex testing and the presence of ulcers have been the only criteria used to diagnose DSPN and that foot examinations were thus not performed according to clinical guidelines (7).
Some limitations and strengths of our study need to be discussed. First, the data on previous foot examinations being performed are self-reported and may suffer from recall bias. Also, the question of whether a physician has ever told the participant that he/she was suffering from nerve damage, neuropathy, polyneuropathy, or diabetic foot may also be subject to recall bias. Yet, the latter data were collected by a trained interviewer (aware of the importance of this specific information) before the actual neurologic examination of the participant took place. It is unlikely that false memory may have had a substantial effect on our results and conclusion. Second, there is no uniform consensus on a definition of diagnosing DSPN for use in epidemiological studies. Also, we cannot rule out that our definition of clinical DSPN allowed for the inclusion of some case subjects that had developed DSPN due to a different cause rather than to chronic hyperglycemia. Subsequently, we performed a validation study to strengthen the validity of the present findings, and we observed that our clinical DSPN definition had an excellent diagnostic performance (19). A further strength of the current study includes the use of different bedside tests of peripheral sensory function, facilitating the construction of a relatively accurate definition of clinical DSPN.
In conclusion, our findings show a high prevalence of unawareness of having clinical DSPN among subjects with prediabetes and with diabetes. The high frequency of unawareness among the latter group is of particular concern, given that subjects with diabetes receive regular diabetes care. Despite the performance of foot examinations by a physician, the proportion of subjects with known diabetes unaware of having DSPN is high. Overall, these results suggest inadequate attention to diabetic foot prevention practice and insufficient adherence to the clinical guidelines for diabetes care necessary to prevent further development of severe diabetic foot complications.
Acknowledgments
The current study was funded by a grant of the German Research Foundation (RA-45913/3-1). The German Diabetes Center is funded by the German Federal Ministry of Health and the Ministry of Innovation, Science, Research and Technology of the State of North Rhine-Westphalia. The KORA research platform was initiated and financed by the Helmholtz Zentrum München, German Research Centre for Environmental Health (GmbH), which is funded by the German Federal Ministry of Education, Science, Research and Technology and by the State of Bavaria.
No potential conflicts of interest relevant to this article were reported.
B.W.C.B. performed data analyses and wrote the manuscript. W.R. planned the study, contributed to data analyses, and wrote the manuscript. M.H., B.K., C.H., D.S., and C.M. reviewed and edited the manuscript. D.Z. planned the study, contributed to data analyses and discussion, and reviewed and edited the manuscript. W.R. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
References
- 1.Vinik AI, Park TS, Stansberry KB, Pittenger GL. Diabetic neuropathies. Diabetologia 2000;43:957–973 [DOI] [PubMed] [Google Scholar]
- 2.Tesfaye S, Boulton AJ, Dyck PJ, et al. Toronto Diabetic Neuropathy Expert Group Diabetic neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care 2010;33:2285–2293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Caputo GM, Cavanagh PR, Ulbrecht JS, Gibbons GW, Karchmer AW. Assessment and management of foot disease in patients with diabetes. N Engl J Med 1994;331:854–860 [DOI] [PubMed] [Google Scholar]
- 4.Seshasai SR, Kaptoge S, Thompson A, et al. Emerging Risk Factors Collaboration Diabetes mellitus, fasting glucose, and risk of cause-specific death. N Engl J Med 2011;364:829–841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.American Diabetes Association Standards of medical care in diabetes—2011. Diabetes Care 2011;34(Suppl. 1):S11–S61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McIntosh A, Peters J, Young R, et al. Prevention and Management of Foot Problems in Type 2 Diabetes: Clinical Guidelines and Evidence. Sheffield, U.K., University of Sheffield, 2003 [PubMed] [Google Scholar]
- 7.Bundesärztekammer, Kassenärztliche Bundesvereinigung, Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften. Nationale VersorgungsLeitlinie Neuropathie bei Diabetes im Erwachsenenalter-Langfassung: version 1.1 [article online]. Available from http://www.diabetes.versorgungsleitlinien.de Accessed 23 July 2012
- 8.De Berardis G, Pellegrini F, Franciosi M, et al. QuED Study Group-Quality of Care and Outcomes in Type 2 Diabetes Are Type 2 diabetic patients offered adequate foot care? The role of physician and patient characteristics. J Diabetes Complications 2005;19:319–327 [DOI] [PubMed] [Google Scholar]
- 9.Kirkman MS, Williams SR, Caffrey HH, Marrero DG. Impact of a program to improve adherence to diabetes guidelines by primary care physicians. Diabetes Care 2002;25:1946–1951 [DOI] [PubMed] [Google Scholar]
- 10.O’Brien KE, Chandramohan V, Nelson DA, Fischer JR, Jr, Stevens G, Poremba JA. Effect of a physician-directed educational campaign on performance of proper diabetic foot exams in an outpatient setting. J Gen Intern Med 2003;18:258–265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tapp RJ, Zimmet PZ, Harper CA, et al. Australian Diabetes Obesity and Lifestyle Study Group Diabetes care in an Australian population: frequency of screening examinations for eye and foot complications of diabetes. Diabetes Care 2004;27:688–693 [DOI] [PubMed] [Google Scholar]
- 12.Daousi C, Benbow SJ, Woodward A, MacFarlane IA. The natural history of chronic painful peripheral neuropathy in a community diabetes population. Diabet Med 2006;23:1021–1024 [DOI] [PubMed] [Google Scholar]
- 13.Herman WH, Kennedy L. Underdiagnosis of peripheral neuropathy in type 2 diabetes. Diabetes Care 2005;28:1480–1481 [DOI] [PubMed] [Google Scholar]
- 14.Wang W, Balamurugan A, Biddle J, Rollins KM. Diabetic neuropathy status and the concerns in underserved rural communities: challenges and opportunities for diabetes educators. Diabetes Educ 2011;37:536–548 [DOI] [PubMed] [Google Scholar]
- 15.Rathmann W, Haastert B, Icks A, et al. High prevalence of undiagnosed diabetes mellitus in Southern Germany: target populations for efficient screening. The KORA survey 2000. Diabetologia 2003;46:182–189 [DOI] [PubMed] [Google Scholar]
- 16.World Health Organization Report of a WHO Consultation: Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications. Geneva, World Health Organization, 1999 [Google Scholar]
- 17.Martina IS, van Koningsveld R, Schmitz PI, van der Meché FG, van Doorn PA. Measuring vibration threshold with a graduated tuning fork in normal aging and in patients with polyneuropathy. European Inflammatory Neuropathy Cause and Treatment (INCAT) group. J Neurol Neurosurg Psychiatry 1998;65:743–747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paisley AN, Abbott CA, van Schie CH, Boulton AJ. A comparison of the Neuropen against standard quantitative sensory-threshold measures for assessing peripheral nerve function. Diabet Med 2002;19:400–405 [DOI] [PubMed] [Google Scholar]
- 19.Bongaerts BW, Rathmann W, Kowall B, et al. Postchallenge hyperglycemia is positively associated with diabetic polyneuropathy: the KORA F4 study. Diabetes Care 2012;35:1891–1893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Apelqvist J, Bakker K, van Houtum WH, Schaper NC, International Working Group on the Diabetic Foot (IWGDF) Editorial Board Practical guidelines on the management and prevention of the diabetic foot: based upon the International Consensus on the Diabetic Foot (2007) Prepared by the International Working Group on the Diabetic Foot. Diabetes Metab Res Rev 2008;24(Suppl. 1):S181–S187 [DOI] [PubMed] [Google Scholar]
- 21.Abbott CA, Carrington AL, Ashe H, et al. North-West Diabetes Foot Care Study The North-West Diabetes Foot Care Study: incidence of, and risk factors for, new diabetic foot ulceration in a community-based patient cohort. Diabet Med 2002;19:377–384 [DOI] [PubMed] [Google Scholar]
- 22.Iversen MM, Ostbye T, Clipp E, et al. Regularity of preventive foot care in persons with diabetes: results from the Nord-Trøndelag Health Study. Res Nurs Health 2008;31:226–237 [DOI] [PubMed] [Google Scholar]
- 23.Rasli MH, Zacharin MR. Foot problems and effectiveness of foot care education in children and adolescents with diabetes mellitus. Pediatr Diabetes 2008;9:602–608 [DOI] [PubMed] [Google Scholar]
- 24.Ahluwalia HK, Miller CE, Pickard SP, Mayo MS, Ahluwalia JS, Beckles GL. Prevalence and correlates of preventive care among adults with diabetes in Kansas. Diabetes Care 2000;23:484–489 [DOI] [PubMed] [Google Scholar]
- 25.De Berardis G, Pellegrini F, Franciosi M, et al. QuED Study Group Physician attitudes toward foot care education and foot examination and their correlation with patient practice. Diabetes Care 2004;27:286–287 [DOI] [PubMed] [Google Scholar]


